Recent Advances in Surface-Enhanced Raman Scattering Magnetic Plasmonic Particles for Bioapplications
Abstract
1. Introduction
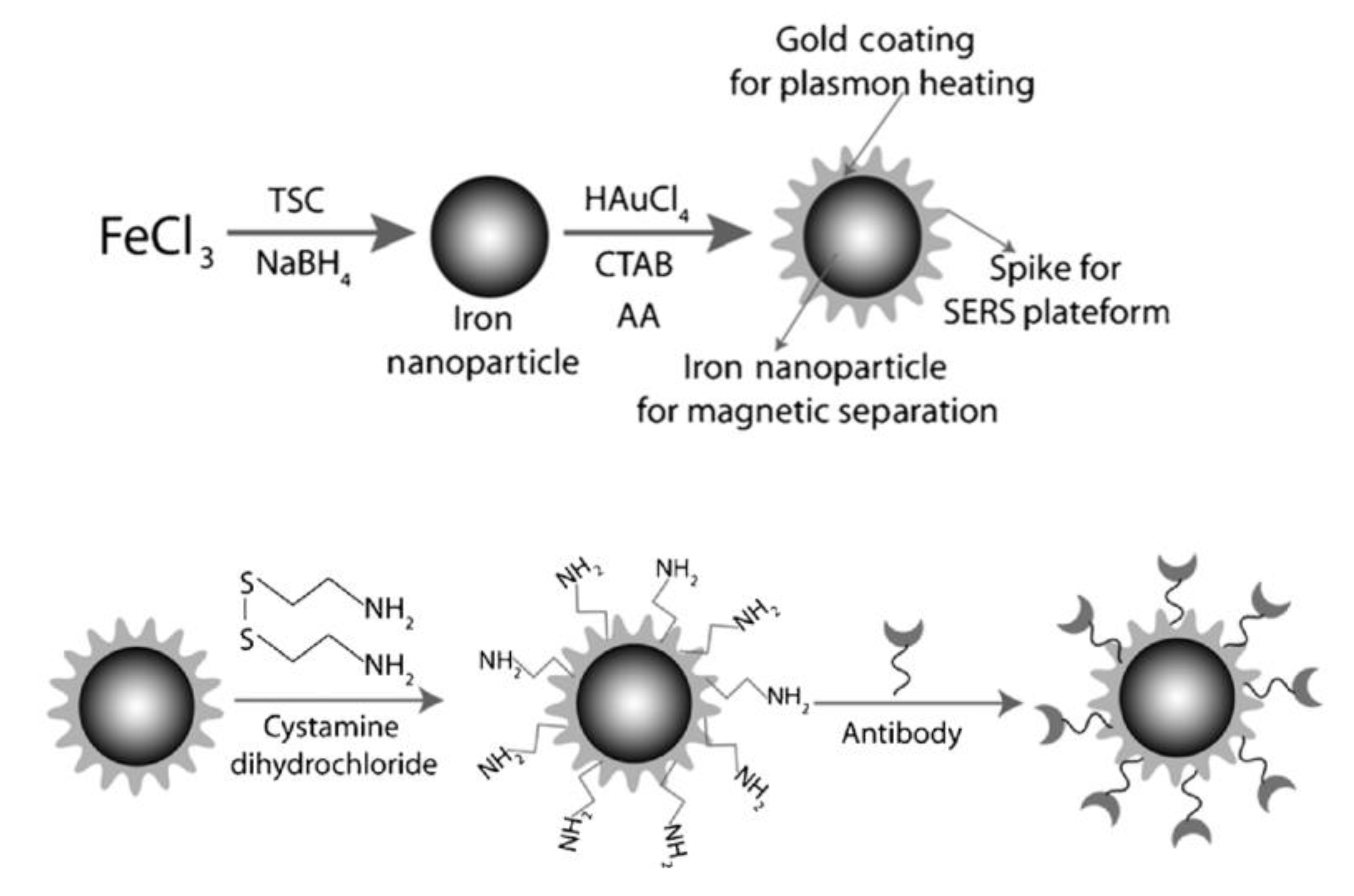
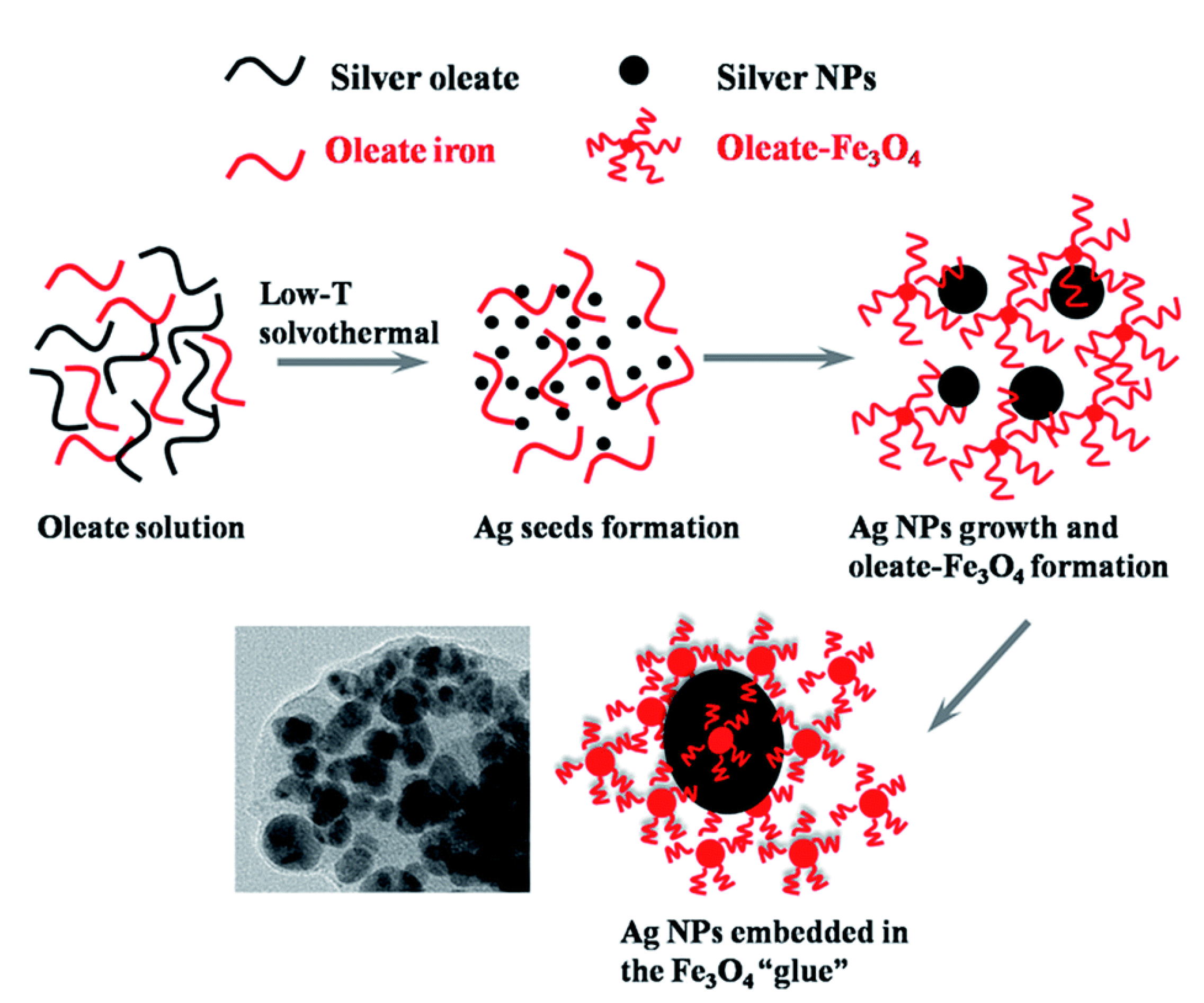
2. Types of MPPs
3. Bioapplication of MPPs
3.1. Detection and Separation
3.1.1. Nucleic Acids
3.1.2. Protein
3.1.3. Small Molecules
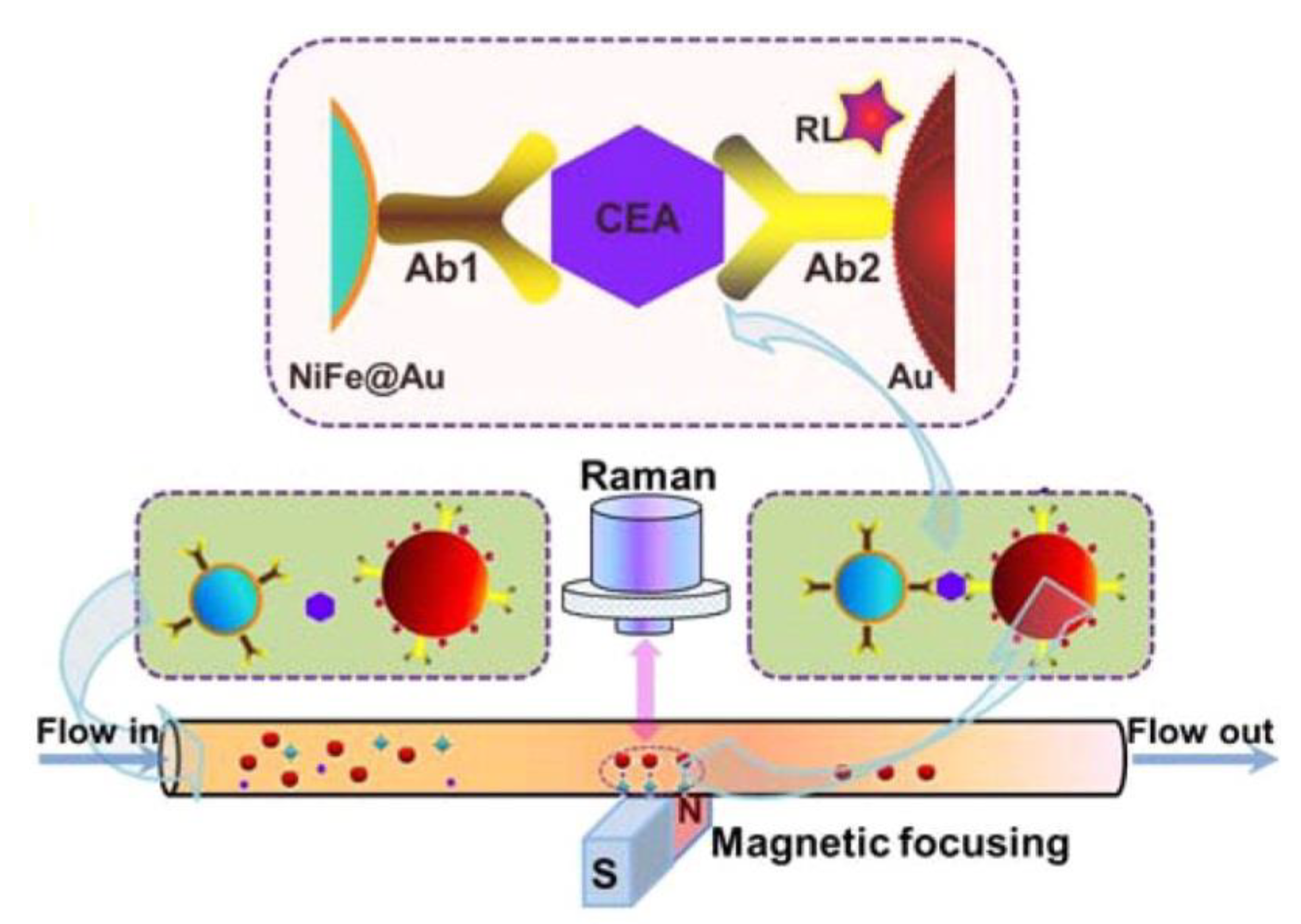
3.1.4. Cancer Diagnostic
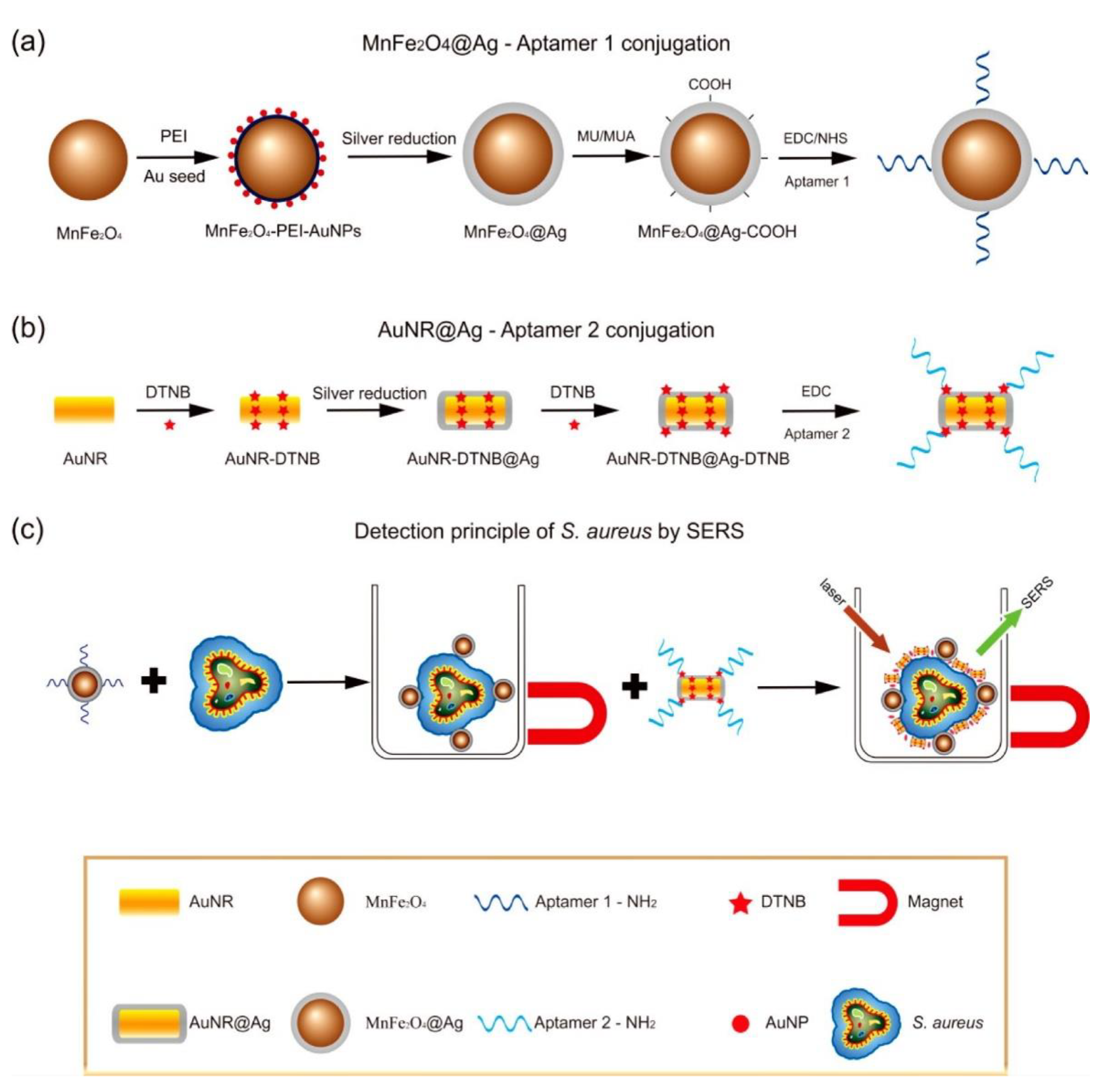
3.1.5. Detection of Pathogens
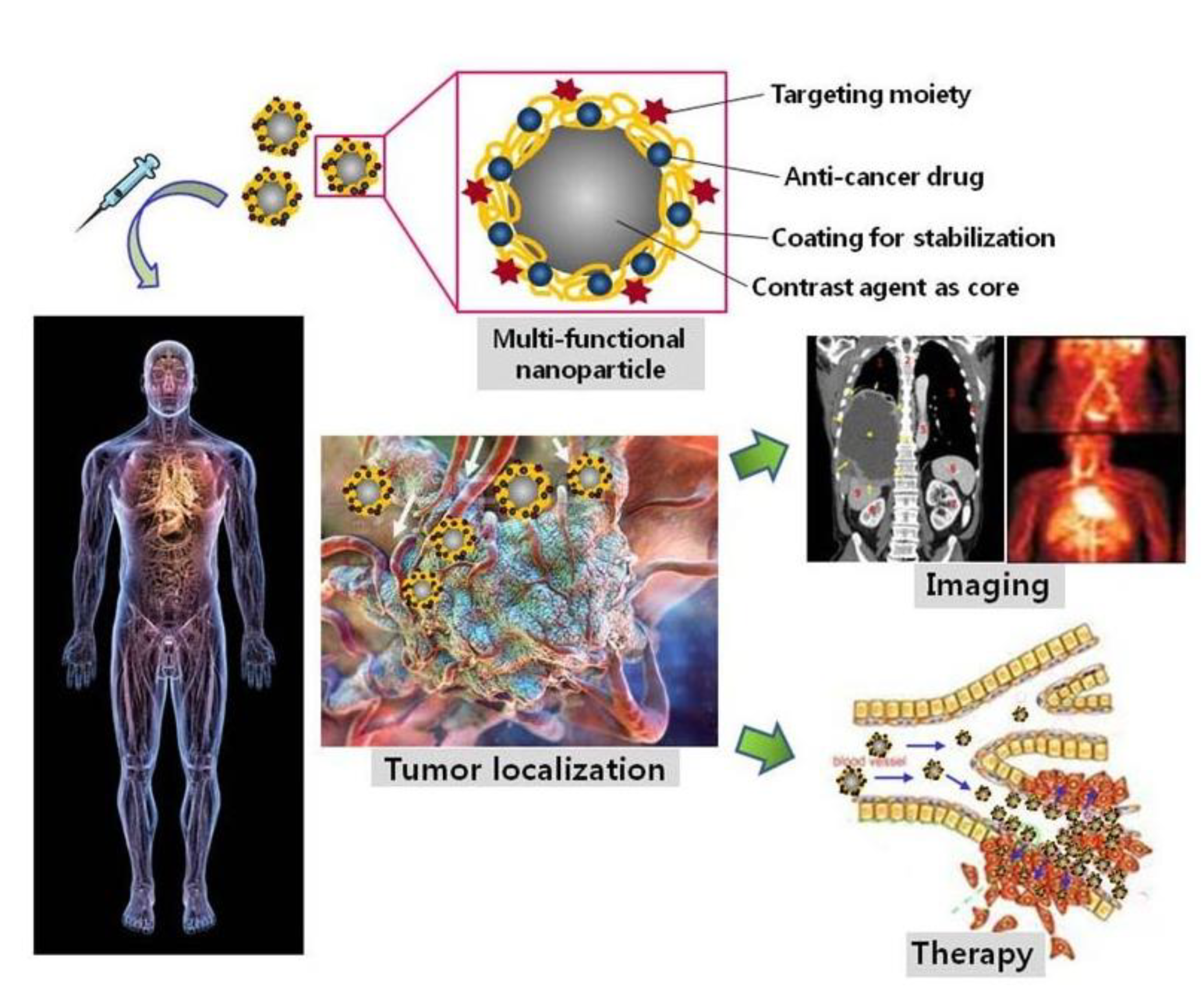
3.2. Drug Delivery and Therapy
3.3. Imaging
4. Conclusion and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A review on surface-enhanced raman scattering. Biosensors 2019, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Schlucker, S. Surface-enhanced raman spectroscopy: Concepts and chemical applications. Angew. Chem. Int. Ed. 2014, 53, 4756–4795. [Google Scholar]
- Etchegoin, P.G.; Le Ru, E.C. A perspective on single molecule sers: Current status and future challenges. Phys. Chem. Chem. Phys. 2008, 10, 6079–6089. [Google Scholar] [CrossRef] [PubMed]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single molecule detection using surface-enhanced raman scattering (sers). Phys. Rev. Lett. 1997, 78, 1667. [Google Scholar] [CrossRef]
- Lee, H.M.; Jin, S.M.; Kim, H.M.; Suh, Y.D. Single-molecule surface-enhanced raman spectroscopy: A perspective on the current status. Phys. Chem. Chem. Phys. 2013, 15, 5276–5287. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, M.; Schraer, S.; Pourrezaei, K.; Tyagi, S. Surface-Enhanced Raman Scattering and Microwave Absorption in Silver Nanoparticle Inks; SPIE BiOS: San Francisco, CA, USA, 2012; Volume 8234. [Google Scholar]
- Figueroa, M.; Pourrezaei, K.; Tyagi, S. Detection of Hyaluronic Acid on a Functionalized Surface Enhanced Raman Scattering Substrate. In Proceedings of the 2011 IEEE 37th Annual Northeast Bioengineering Conference (NEBEC), Troy, NY, USA, 1–3 April 2011; pp. 1–2. [Google Scholar]
- Contarino, M.; Kamat, V.; Keough, E.; Babu, N.; Sergi, M.; Ishino, T.; Papazoglou, E.; Chaiken, I.; Tyagi, S.; Pourrezaei, K. Bio-nano-Optics for Cellular Investigations; SPIE: Philadelphia, PA, USA, 2004; Volume 5588. [Google Scholar]
- Tyagi, S.; Figueroa, M. Flexible Sers Substrates with Filtering Capabilities. United States Patents US9278855B2, 8 March 2016. [Google Scholar]
- Lawandy, N.M. Chemical and Biological Sensing Using Metallic Particles in Amplifying and Absorbing Media. United States Patents US7355704B2, 8 April 2008. [Google Scholar]
- Tyagi, S.; Pourrezaei, K. Method for the Formation of Sers Substrates. United States Patents US8559002B2, 15 October 2013. [Google Scholar]
- Lu, A.H.; Salabas, E.L.; Schuth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar]
- Xie, W.; Schlucker, S. Rationally designed multifunctional plasmonic nanostructures for surface-enhanced raman spectroscopy: A review. Rep. Prog. Phys. 2014, 77, 116502. [Google Scholar] [CrossRef]
- Park, S.-M.; Aalipour, A.; Vermesh, O.; Yu, J.H.; Gambhir, S.S. Towards clinically translatable in vivo nanodiagnostics. Nat. Rev. Mater. 2017, 2, 17014. [Google Scholar] [CrossRef]
- Park, S.-M.; Wong, D.J.; Ooi, C.C.; Kurtz, D.M.; Vermesh, O.; Aalipour, A.; Suh, S.; Pian, K.L.; Chabon, J.J.; Lee, S.H.; et al. Molecular profiling of single circulating tumor cells from lung cancer patients. Proc. Natl. Acad. Sci. USA 2016, 113, E8379. [Google Scholar] [CrossRef]
- Vermesh, O.; Aalipour, A.; Ge, T.J.; Saenz, Y.; Guo, Y.; Alam, I.S.; Park, S.-M.; Adelson, C.N.; Mitsutake, Y.; Vilches-Moure, J.; et al. An intravascular magnetic wire for the high-throughput retrieval of circulating tumour cells in vivo. Nat. Biomed. Eng. 2018, 2, 696–705. [Google Scholar] [CrossRef]
- Lai, H.S.; Xu, F.G.; Wang, L. A review of the preparation and application of magnetic nanoparticles for surface-enhanced raman scattering. J. Mater. Sci. 2018, 53, 8677–8698. [Google Scholar] [CrossRef]
- Wang, C.G.; Meloni, M.M.; Wu, X.Z.; Zhuo, M.; He, T.G.; Wang, J.F.; Wang, C.W.; Dong, P.T. Magnetic plasmonic particles for sers-based bacteria sensing: A review. Aip. Adv. 2019, 9. [Google Scholar] [CrossRef]
- Rhouati, A.; Catanante, G.; Nunes, G.; Hayat, A.; Marty, J.-L. Label-free aptasensors for the detection of mycotoxins. Sensors 2016, 16, 2178. [Google Scholar] [CrossRef] [PubMed]
- Balzerova, A.; Fargasova, A.; Markova, Z.; Ranc, V.; Zboril, R. Magnetically-assisted surface enhanced raman spectroscopy (ma-sers) for label-free determination of human immunoglobulin g (igg) in blood using fe3o4@ag nanocomposite. Anal. Chem. 2014, 86, 11107–11114. [Google Scholar] [CrossRef]
- Han, Y.; Lei, S.-L.; Lu, J.-H.; He, Y.; Chen, Z.-W.; Ren, L.; Zhou, X. Potential use of sers-assisted theranostic strategy based on fe3o4/au cluster/shell nanocomposites for bio-detection, mri, and magnetic hyperthermia. Mater. Sci. Eng. C 2016, 64, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Wang, C.W.; Zhu, A.N.; Long, F. Single functional magnetic-bead as universal biosensing platform for trace analyte detection using sers-nanobioprobe. Biosens. Bioelectron. 2016, 79, 661–668. [Google Scholar] [CrossRef]
- Agoston, R.; Izake, E.L.; Sivanesan, A.; Lott, W.B.; Sillence, M.; Steel, R. Rapid isolation and detection of erythropoietin in blood plasma by magnetic core gold nanoparticles and portable raman spectroscopy. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 633–641. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Senapati, D.; Khan, S.A.; Singh, A.K.; Hamme, A.; Yust, B.; Sardar, D.; Ray, P.C. Popcorn-shaped magnetic core–plasmonic shell multifunctional nanoparticles for the targeted magnetic separation and enrichment, label-free sers imaging, and photothermal destruction of multidrug-resistant bacteria. Chem. A Eur. J. 2013, 19, 2839–2847. [Google Scholar] [CrossRef]
- Wang, J.; Wu, X.; Wang, C.; Shao, N.; Dong, P.; Xiao, R.; Wang, S. Magnetically assisted surface-enhanced raman spectroscopy for the detection of staphylococcus aureus based on aptamer recognition. ACS Appl. Mater. Interfaces 2015, 7, 20919–20929. [Google Scholar] [CrossRef]
- Yang, K.; Hu, Y.; Dong, N.; Zhu, G.; Zhu, T.; Jiang, N. A novel sers-based magnetic aptasensor for prostate specific antigen assay with high sensitivity. Biosens. Bioelectron. 2017, 94, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, Q.; Mehedi Hassan, M.; Chen, X.; Ouyang, Q.; Guo, Z.; Zhao, J. A magnetite/pmaa nanospheres-targeting sers aptasensor for tetracycline sensing using mercapto molecules embedded core/shell nanoparticles for signal amplification. Biosens. Bioelectron. 2017, 92, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Carrouée, A.; Allard-Vannier, E.; Même, S.; Szeremeta, F.; Beloeil, J.-C.; Chourpa, I. Sensitive trimodal magnetic resonance imaging-surface-enhanced resonance raman scattering-fluorescence detection of cancer cells with stable magneto-plasmonic nanoprobes. Anal. Chem. 2015, 87, 11233–11241. [Google Scholar] [CrossRef]
- Yang, X.; He, Y.; Wang, X.; Yuan, R. A sers biosensor with magnetic substrate cofe2o4@ag for sensitive detection of hg2+. Appl. Surf. Sci. 2017, 416, 581–586. [Google Scholar] [CrossRef]
- He, Y.; Wang, Y.; Yang, X.; Xie, S.; Yuan, R.; Chai, Y. Metal organic frameworks combining cofe2o4 magnetic nanoparticles as highly efficient sers sensing platform for ultrasensitive detection of n-terminal pro-brain natriuretic peptide. ACS Appl. Mater. Interfaces 2016, 8, 7683–7690. [Google Scholar] [PubMed]
- Shen, J.; Zhou, Y.; Huang, J.; Zhu, Y.; Zhu, J.; Yang, X.; Chen, W.; Yao, Y.; Qian, S.; Jiang, H.; et al. In-situ sers monitoring of reaction catalyzed by multifunctional fe3o4@tio2@ag-au microspheres. Appl. Catal. B Environ. 2017, 205, 11–18. [Google Scholar] [CrossRef]
- Ilkhani, H.; Hughes, T.; Li, J.; Zhong, C.J.; Hepel, M. Nanostructured sers-electrochemical biosensors for testing of anticancer drug interactions with DNA. Biosens. Bioelectron. 2016, 80, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Srinoi, P.; Chen, Y.T.; Vittur, V.; Marquez, M.D.; Lee, T.R. Bimetallic nanoparticles: Enhanced magnetic and optical properties for emerging biological applications. Appl. Sci. 2018, 8, 1106. [Google Scholar] [CrossRef]
- Wang, C.; Wang, J.; Li, M.; Qu, X.; Zhang, K.; Rong, Z.; Xiao, R.; Wang, S. A rapid sers method for label-free bacteria detection using polyethylenimine-modified au-coated magnetic microspheres and au@ag nanoparticles. Analyst 2016, 141, 6226–6238. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Xu, W.; Wang, Y.; Kuang, Q.; Shi, Y.; Zhong, L.; Zhang, Q. Fabrication of cluster/shell fe3o4/au nanoparticles and application in protein detection via a sers method. J. Phys. Chem. C 2010, 114, 19607–19613. [Google Scholar] [CrossRef]
- Wang, C.; Xu, J.; Wang, J.; Rong, Z.; Li, P.; Xiao, R.; Wang, S. Polyethylenimine-interlayered silver-shell magnetic-core microspheres as multifunctional sers substrates. J. Mater. Chem. C 2015, 3, 8684–8693. [Google Scholar] [CrossRef]
- Wang, C.; Li, P.; Wang, J.; Rong, Z.; Pang, Y.; Xu, J.; Dong, P.; Xiao, R.; Wang, S. Polyethylenimine-interlayered core–shell–satellite 3d magnetic microspheres as versatile sers substrates. Nanoscale 2015, 7, 18694–18707. [Google Scholar] [PubMed]
- He, Q.; Zhao, A.; Li, L.; Sun, H.; Wang, D.; Guo, H.; Sun, M.; Chen, P. Fabrication of fe3o4@sio2@ag magnetic–plasmonic nanospindles as highly efficient sers active substrates for label-free detection of pesticides. New J. Chem. 2017, 41, 1582–1590. [Google Scholar]
- Sun, Z.; Du, J.; Yan, L.; Chen, S.; Yang, Z.; Jing, C. Multifunctional fe3o4@sio2–au satellite structured sers probe for charge selective detection of food dyes. ACS Appl. Mater. Inter. 2016, 8, 3056–3062. [Google Scholar] [CrossRef] [PubMed]
- Jeong, C.; Kim, H.-M.; Park, S.Y.; Cha, M.G.; Park, S.-J.; Kyeong, S.; Pham, X.-H.; Hahm, E.; Ha, Y.; Jeong, D.H.; et al. Highly sensitive magnetic-sers dual-function silica nanoprobes for effective on-site organic chemical detection. Nanomaterials 2017, 7, 146. [Google Scholar] [CrossRef] [PubMed]
- Hou, T.; Liu, Y.; Xu, L.; Wu, Y.; Ying, Y.; Wen, Y.; Guo, X.; Yang, H. Au dotted magnetic graphene sheets for sensitive detection of thiocyanate. Sens. Actuators B Chem. 2017, 241, 376–382. [Google Scholar] [CrossRef]
- Ding, G.; Xie, S.; Zhu, Y.; Liu, Y.; Wang, L.; Xu, F. Graphene oxide wrapped fe3o4@au nanohybrid as sers substrate for aromatic dye detection. Sens. Actuators B Chem. 2015, 221, 1084–1093. [Google Scholar]
- Liu, Z.; Wang, Y.; Deng, R.; Yang, L.; Yu, S.; Xu, S.; Xu, W. Fe3o4@graphene oxide@ag particles for surface magnet solid-phase extraction surface-enhanced raman scattering (smspe-sers): From sample pretreatment to detection all-in-one. ACS Appl. Mater. Inter. 2016, 8, 14160–14168. [Google Scholar]
- Contreras-Cáceres, R.; Abalde-Cela, S.; Guardia-Girós, P.; Fernández-Barbero, A.; Pérez-Juste, J.; Alvarez-Puebla, R.A.; Liz-Marzán, L.M. Multifunctional microgel magnetic/optical traps for sers ultradetection. Langmuir 2011, 27, 4520–4525. [Google Scholar]
- Zhao, M.; Guo, H.; Liu, W.; Tang, J.; Wang, L.; Zhang, B.; Xue, C.; Liu, J.; Zhang, W. Silica cladding of ag nanoparticles for high stability and surface-enhanced raman spectroscopy performance. Nanoscale Res. Lett. 2016, 11, 403. [Google Scholar] [CrossRef]
- Son, H.Y.; Kim, K.R.; Lee, J.B.; Le Kim, T.H.; Jang, J.; Kim, S.J.; Yoon, M.S.; Kim, J.W.; Nam, Y.S. Bioinspired synthesis of mesoporous gold-silica hybrid microspheres as recyclable colloidal sers substrates. Sci. Rep. 2017, 7, 14728. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Chen, H.; Li, Y.; Yuan, X.; Zhao, X.; Chen, W.; Cao, F.; Cai, N.; Huang, X.; Yang, F.; et al. Synthesis of mesoporous-silica coated multi-branched gold nanoparticles for surface enhanced raman scattering evaluation of 4-bromomethcathinone. J. Saudi Chem. Soc. 2019, 23, 378–383. [Google Scholar] [CrossRef]
- Li, M.; Qiu, Y.; Fan, C.; Cui, K.; Zhang, Y.; Xiao, Z. Design of sers nanoprobes for raman imaging: Materials, critical factors and architectures. Acta Pharm. Sin. B 2018, 8, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Huang, Y.; Huang, K.; Lin, J.; Huang, P. Functional magnetic graphene composites for biosensing. Int. J. Mol. Sci. 2020, 21, 390. [Google Scholar] [CrossRef] [PubMed]
- Shan, Y.; Yang, Y.; Cao, Y.; Huang, Z. Facile solvothermal synthesis of ag/fe3o4 nanocomposites and their sers applications in on-line monitoring of pesticide contaminated water. RSC Adv. 2015, 5, 102610–102618. [Google Scholar] [CrossRef]
- Tomitaka, A.; Arami, H.; Ahmadivand, A.; Pala, N.; McGoron, A.J.; Takemura, Y.; Febo, M.; Nair, M. Magneto-plasmonic nanostars for image-guided and nir-triggered drug delivery. Sci. Rep. 2020, 10, 10115. [Google Scholar] [CrossRef] [PubMed]
- McBain, S.C.; Yiu, H.H.P.; Dobson, J. Magnetic nanoparticles for gene and drug delivery. Int. J. Nanomed. 2008, 3, 169–180. [Google Scholar]
- Mou, X.; Ali, Z.; Li, S.; He, N. Applications of magnetic nanoparticles in targeted drug delivery system. J. Nanosci. Nanotechnol. 2015, 15, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, D.; Kiselev, M.A.; Caccamo, M.T. Smart nanoparticles for drug delivery application: Development of versatile nanocarrier platforms in biotechnology and nanomedicine. J. Nanomater. 2019, 2019, 3702518. [Google Scholar] [CrossRef]
- Ulbrich, K.; Holá, K.; Šubr, V.; Bakandritsos, A.; Tuček, J.; Zbořil, R. Targeted drug delivery with polymers and magnetic nanoparticles: Covalent and noncovalent approaches, release control, and clinical studies. Chem. Rev. 2016, 116, 5338–5431. [Google Scholar] [CrossRef] [PubMed]
- Bae, K.H.; Chung, H.J.; Park, T.G. Nanomaterials for cancer therapy and imaging. Mol. Cells 2011, 31, 295–302. [Google Scholar] [CrossRef]
- Jun, B.H.; Noh, M.S.; Kim, J.; Kim, G.; Kang, H.; Kim, M.S.; Seo, Y.T.; Baek, J.; Kim, J.H.; Park, J.; et al. Multifunctional silver-embedded magnetic nanoparticles as sers nanoprobes and their applications. Small 2010, 6, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Doering, W.E.; Piotti, M.E.; Natan, M.J.; Freeman, R.G. Sers as a foundation for nanoscale, optically detected biological labels. Adv. Mater. 2007, 19, 3100–3108. [Google Scholar] [CrossRef]
- Jun, B.H.; Noh, M.S.; Kim, G.; Kang, H.; Kim, J.H.; Chung, W.J.; Kim, M.S.; Kim, Y.K.; Cho, M.H.; Jeong, D.H.; et al. Protein separation and identification using magnetic beads encoded with surface-enhanced raman spectroscopy. Anal. Biochem. 2009, 391, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Padilla-Cruz, A.L.; Garza-Cervantes, J.A.; Vasto-Anzaldo, X.G.; García-Rivas, G.; León-Buitimea, A.; Morones-Ramírez, J.R. Synthesis and design of ag–fe bimetallic nanoparticles as antimicrobial synergistic combination therapies against clinically relevant pathogens. Sci. Rep. 2021, 11, 5351. [Google Scholar] [CrossRef]
- Al-Asfar, A.; Zaheer, Z.; Aazam, E.S. Eco-friendly green synthesis of ag@fe bimetallic nanoparticles: Antioxidant, antimicrobial and photocatalytic degradation of bromothymol blue. J. Photochem. Photobiol. B Biol. 2018, 185, 143–152. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Li, G.; Hu, Y. Aptamer recognition induced target-bridged strategy for proteins detection based on magnetic chitosan and silver/chitosan nanoparticles using surface-enhanced raman spectroscopy. Anal. Chem. 2015, 87, 11039–11047. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Pang, S.; He, L.; Nugen, S.R. Highly sensitive and selective detection of nitrite ions using fe3o4@sio2/au magnetic nanoparticles by surface-enhanced raman spectroscopy. Biosens. Bioelectron. 2016, 85, 726–733. [Google Scholar] [CrossRef]
- Pour Yazdankhah, S.; Hellemann, A.-L.; Rønningen, K.; Olsen, E. Rapid and sensitive detection of staphylococcus species in milk by elisa based on monodisperse magnetic particles. Vet. Microbiol. 1998, 62, 17–26. [Google Scholar] [CrossRef]
- Alarcón, B.; Vicedo, B.; Aznar, R. Pcr-based procedures for detection and quantification of staphylococcus aureus and their application in food. J. Appl. Microbiol. 2006, 100, 352–364. [Google Scholar] [CrossRef]
- Zelada-Guillén, G.A.; Sebastián-Avila, J.L.; Blondeau, P.; Riu, J.; Rius, F.X. Label-free detection of staphylococcus aureus in skin using real-time potentiometric biosensors based on carbon nanotubes and aptamers. Biosens. Bioelectron. 2012, 31, 226–232. [Google Scholar] [CrossRef]
- Wang, X.-Y.; Yang, J.-Y.; Wang, Y.-T.; Zhang, H.-C.; Chen, M.-L.; Yang, T.; Wang, J.-H. M13 phage-based nanoprobe for sers detection and inactivation of staphylococcus aureus. Talanta 2021, 221, 121668. [Google Scholar] [CrossRef]
- Zhu, A.; Ali, S.; Xu, Y.; Ouyang, Q.; Chen, Q. A sers aptasensor based on aunps functionalized pdms film for selective and sensitive detection of staphylococcus aureus. Biosens. Bioelectron. 2021, 172, 112806. [Google Scholar]
- Strickland, A.D.; Batt, C.A. Detection of carbendazim by surface-enhanced raman scattering using cyclodextrin inclusion complexes on gold nanorods. Anal. Chem. 2009, 81, 2895–2903. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, L.; Garcia-Ramos, J.V.; Domingo, C.; Sanchez-Cortes, S. Nanosensors based on viologen functionalized silver nanoparticles: Few molecules surface-enhanced raman spectroscopy detection of polycyclic aromatic hydrocarbons in interparticle hot spots. Anal. Chem. 2009, 81, 1418–1425. [Google Scholar]
- Guerrini, L.; Garcia-Ramos, J.V.; Domingo, C.; Sanchez-Cortes, S. Sensing polycyclic aromatic hydrocarbons with dithiocarbamate-functionalized ag nanoparticles by surface-enhanced raman scattering. Anal. Chem. 2009, 81, 953–960. [Google Scholar] [PubMed]
- Zhang, H.; Li, F.; Chen, H.; Ma, Y.; Qi, S.; Chen, X.; Zhou, L. Aunps colorimetric sensor for detecting platelet-derived growth factor-bb based on isothermal target-triggering strand displacement amplification. Sens. Actuators B Chem. 2015, 207, 748–755. [Google Scholar] [CrossRef]
- Zhu, D.; Zhou, X.; Xing, D. A new kind of aptamer-based immunomagnetic electrochemiluminescence assay for quantitative detection of protein. Biosens. Bioelectron. 2010, 26, 285–288. [Google Scholar]
- Jin, X.; Zhao, J.; Zhang, L.; Huang, Y.; Zhao, S. An enhanced fluorescence polarization strategy based on multiple protein–DNA–protein structures for sensitive detection of pdgf-bb. RSC Adv. 2014, 4, 6850–6853. [Google Scholar] [CrossRef]
- Wang, C.-W.; Chang, H.-T. Sensitive detection of platelet-derived growth factor through surface-enhanced raman scattering. Anal. Chem. 2014, 86, 7606–7611. [Google Scholar] [CrossRef]
- Niu, C.; Zou, B.; Wang, Y.; Cheng, L.; Zheng, H.; Zhou, S. Highly sensitive and reproducible sers performance from uniform film assembled by magnetic noble metal composite microspheres. Langmuir 2016, 32, 858–863. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Fan, C.; Wang, J.; He, J.; Liang, E.; Chao, M. Realization of high sensitive sers substrates with one-pot fabrication of ag–fe3o4 nanocomposites. J. Colloid Interf Sci. 2015, 438, 116–121. [Google Scholar] [CrossRef]
- Toma, S.H.; Santos, J.J.; Araki, K.; Toma, H.E. Pushing the surface-enhanced raman scattering analyses sensitivity by magnetic concentration: A simple non core–shell approach. Anal. Chim. Acta 2015, 855, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Zhu, Y.; Yang, X.; Zong, J.; Li, C. Multifunctional fe3o4@ag/sio2/au core–shell microspheres as a novel sers-activity label via long-range plasmon coupling. Langmuir 2013, 29, 690–695. [Google Scholar] [CrossRef]
- Wang, Y.; Ravindranath, S.; Irudayaraj, J. Separation and detection of multiple pathogens in a food matrix by magnetic sers nanoprobes. Anal. Bioanal. Chem. 2011, 399, 1271–1278. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.; Wang, C.; Wang, J.; Sun, Z.; Xiao, R.; Wang, S. Fe3o4@ag magnetic nanoparticles for microrna capture and duplex-specific nuclease signal amplification based sers detection in cancer cells. Biosens. Bioelectron. 2016, 79, 574–580. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Joshi, P.; Zhou, Y.; Ding, R.; Zhang, P. Quantitative sers-based DNA detection assisted by magnetic microspheres. Chem. Commun. 2015, 51, 15284–15286. [Google Scholar] [CrossRef]
- Wu, L.; Xiao, X.; Chen, K.; Yin, W.; Li, Q.; Wang, P.; Lu, Z.; Ma, J.; Han, H. Ultrasensitive sers detection of bacillus thuringiensis special gene based on au@ag nrs and magnetic beads. Biosens. Bioelectron. 2017, 92, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Alula, M.T.; Yang, J. Photochemical decoration of gold nanoparticles on polymer stabilized magnetic microspheres for determination of adenine by surface-enhanced raman spectroscopy. Microchim. Acta 2015, 182, 1017–1024. [Google Scholar] [CrossRef]
- Ngo, H.T.; Gandra, N.; Fales, A.M.; Taylor, S.M.; Vo-Dinh, T. Sensitive DNA detection and snp discrimination using ultrabright sers nanorattles and magnetic beads for malaria diagnostics. Biosens. Bioelectron. 2016, 81, 8–14. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Hannon, G.J. Micrornas: Small rnas with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Novina, C.D.; Sharp, P.A. The rnai revolution. Nature 2004, 430, 161–164. [Google Scholar] [CrossRef]
- Farazi, T.A.; Spitzer, J.I.; Morozov, P.; Tuschl, T. Mirnas in human cancer. J. Pathol. 2011, 223, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Kang, W.; Tong, J.H.M.; Chan, A.W.H.; Lung, R.W.M.; Chau, S.L.; Wong, Q.W.L.; Wong, N.; Yu, J.; Cheng, A.S.L.; To, K.F. Stathmin1 plays oncogenic role and is a target of microrna-223 in gastric cancer. PLoS ONE 2012, 7, e33919. [Google Scholar] [CrossRef] [PubMed]
- Yanaihara, N.; Caplen, N.; Bowman, E.; Seike, M.; Kumamoto, K.; Yi, M.; Stephens, R.M.; Okamoto, A.; Yokota, J.; Tanaka, T. Unique microrna molecular profiles in lung cancer diagnosis and prognosis. Cancer Cell 2006, 9, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Kilic, T.; Topkaya, S.N.; Ariksoysal, D.O.; Ozsoz, M.; Ballar, P.; Erac, Y.; Gozen, O. Electrochemical based detection of microrna, mir21 in breast cancer cells. Biosens. Bioelectron. 2012, 38, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.P.; Liu, H.; Zhen, S.J.; Li, C.M.; Huang, C.Z. Nanosilver-based surface-enhanced raman spectroscopic determination of DNA methyltransferase activity through real-time hybridization chain reaction. Biosens. Bioelectron. 2015, 73, 228–233. [Google Scholar] [CrossRef]
- Gao, R.; Cheng, Z.; deMello, A.J.; Choo, J. Wash-free magnetic immunoassay of the psa cancer marker using sers and droplet microfluidics. Lab. A Chip. 2016, 16, 1022–1029. [Google Scholar] [CrossRef] [PubMed]
- Selbes, Y.S.; Caglayan, M.G.; Eryilmaz, M.; Boyaci, I.H.; Saglam, N.; Basaran, A.A.; Tamer, U. Surface-enhanced raman probe for rapid nanoextraction and detection of erythropoietin in urine. Anal. Bioanal. Chem. 2016, 408, 8447–8456. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.H.; Shin, Y.; Sim, S.J. Development of sers substrate using phage-based magnetic template for triplex assay in sepsis diagnosis. Biosens. Bioelectron. 2016, 85, 522–528. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Xu, G.; Wei, F.; Zhang, A.; Yang, J.; Hu, Q. Detection of cea in human serum using surface-enhanced raman spectroscopy coupled with antibody-modified au and γ-fe2o3@au nanoparticles. J. Pharm. Biomed. Anal. 2016, 121, 135–140. [Google Scholar] [CrossRef]
- Usta, D.D.; Salimi, K.; Pinar, A.; Coban, İ.; Tekinay, T.; Tuncel, A. A boronate affinity-assisted sers tag equipped with a sandwich system for detection of glycated hemoglobin in the hemolysate of human erythrocytes. ACS Appl. Mater. Inter. 2016, 8, 11934–11944. [Google Scholar] [CrossRef] [PubMed]
- Hassanain, W.A.; Izake, E.L.; Schmidt, M.S.; Ayoko, G.A. Gold nanomaterials for the selective capturing and sers diagnosis of toxins in aqueous and biological fluids. Biosens. Bioelectron. 2017, 91, 664–672. [Google Scholar] [CrossRef]
- Gao, R.; Ko, J.; Cha, K.; Ho Jeon, J.; Rhie, G.-E.; Choi, J.; deMello, A.J.; Choo, J. Fast and sensitive detection of an anthrax biomarker using sers-based solenoid microfluidic sensor. Biosens. Bioelectron. 2015, 72, 230–236. [Google Scholar] [CrossRef]
- Yang, T.; Guo, X.; Wang, H.; Fu, S.; Wen, Y.; Yang, H. Magnetically optimized sers assay for rapid detection of trace drug-related biomarkers in saliva and fingerprints. Biosens. Bioelectron. 2015, 68, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Skeete, Z.; Shan, S.; Yan, S.; Kurzatkowska, K.; Zhao, W.; Ngo, Q.M.; Holubovska, P.; Luo, J.; Hepel, M.; et al. Surface enhanced raman scattering detection of cancer biomarkers with bifunctional nanocomposite probes. Anal. Chem. 2015, 87, 10698–10702. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Zhang, R.; Gao, M.; Zhang, X. A rapid and simple method for efficient capture and accurate discrimination of circulating tumor cells using aptamer conjugated magnetic beads and surface-enhanced raman scattering imaging. Anal. Bioanal Chem. 2015, 407, 8883–8892. [Google Scholar] [CrossRef] [PubMed]
- Noh, M.S.; Jun, B.-H.; Kim, S.; Kang, H.; Woo, M.-A.; Minai-Tehrani, A.; Kim, J.-E.; Kim, J.; Park, J.; Lim, H.-T.; et al. Magnetic surface-enhanced raman spectroscopic (m-sers) dots for the identification of bronchioalveolar stem cells in normal and lung cancer mice. Biomaterials 2009, 30, 3915–3925. [Google Scholar] [CrossRef]
- Jang, H.; Hwang, E.Y.; Kim, Y.; Choo, J.; Jeong, J.; Lim, D.W. Surface-enhanced raman scattering and fluorescence-based dual nanoprobes for multiplexed detection of bacterial pathogens. J. Biomed. Nanotechnol. 2016, 12, 1938–1951. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, X.; Luo, C.; Liu, Z.; Chen, Y.; Dong, S.; Jiang, C.; Yang, S.; Wang, F.; Xiao, X. Volume-enhanced raman scattering detection of viruses. Small 2019, 15, 1805516. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xu, J.; Mi, L.; Gong, H.; Jiang, S.; Yu, Q. Multifunctional magnetic–plasmonic nanoparticles for fast concentration and sensitive detection of bacteria using sers. Biosens. Bioelectron. 2012, 31, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wu, X.; Wang, C.; Rong, Z.; Ding, H.; Li, H.; Li, S.; Shao, N.; Dong, P.; Xiao, R.; et al. Facile synthesis of au-coated magnetic nanoparticles and their application in bacteria detection via a sers method. ACS Appl. Mater. Inter. 2016, 8, 19958–19967. [Google Scholar] [CrossRef] [PubMed]
- Ondera, T.J.; Hamme Ii, A.T. Magnetic-optical nanohybrids for targeted detection, separation, and photothermal ablation of drug-resistant pathogens. Analyst 2015, 140, 7902–7911. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Xu, L.; Zhang, F.; Song, Z.; Hu, Y.; Ji, Y.; Shen, J.; Li, B.; Lu, H.; Yang, H. A promising magnetic sers immunosensor for sensitive detection of avian influenza virus. Biosens. Bioelectron. 2017, 89, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Gong, J.-L.; Huang, Y.; Zheng, Y.; Jiang, J.-H.; Shen, G.-L.; Yu, R.-Q. Biocompatible core-shell nanoparticle-based surface-enhanced raman scattering probes for detection of DNA related to hiv gene using silica-coated magnetic nanoparticles as separation tools. Talanta 2007, 72, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Huynh, K.-H.; Pham, X.-H.; Kim, J.; Lee, S.H.; Chang, H.; Rho, W.-Y.; Jun, B.-H. Synthesis, properties, and biological applications of metallic alloy nanoparticles. Int. J. Mol. Sci. 2020, 21, 5174. [Google Scholar] [CrossRef]
- Maier-Hauff, K.; Rothe, R.; Scholz, R.; Gneveckow, U.; Wust, P.; Thiesen, B.; Feussner, A.; von Deimling, A.; Waldoefner, N.; Felix, R.; et al. Intracranial thermotherapy using magnetic nanoparticles combined with external beam radiotherapy: Results of a feasibility study on patients with glioblastoma multiforme. J. Neurooncol. 2007, 81, 53–60. [Google Scholar] [CrossRef] [PubMed]
- DeNardo, S.J.; DeNardo, G.L.; Miers, L.A.; Natarajan, A.; Foreman, A.R.; Gruettner, C.; Adamson, G.N.; Ivkov, R. Development of tumor targeting bioprobes ((111)in-chimeric l6 monoclonal antibody nanoparticles) for alternating magnetic field cancer therapy. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2005, 11, 7087s–7092s. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, Y.; Yan, C.; Song, L.; Wen, S.; Zang, F.; Chen, G.; Ding, Q.; Yan, C.; Gu, N. High-performance pegylated mn-zn ferrite nanocrystals as a passive-targeted agent for magnetically induced cancer theranostics. Biomaterials 2014, 35, 9126–9136. [Google Scholar] [CrossRef]
- Xie, J.; Yan, C.; Yan, Y.; Chen, L.; Song, L.; Zang, F.; An, Y.; Teng, G.; Gu, N.; Zhang, Y. Multi-modal mn–zn ferrite nanocrystals for magnetically-induced cancer targeted hyperthermia: A comparison of passive and active targeting effects. Nanoscale 2016, 8, 16902–16915. [Google Scholar] [CrossRef] [PubMed]
- Botha, T.L.; Elemike, E.E.; Horn, S.; Onwudiwe, D.C.; Giesy, J.P.; Wepener, V. Cytotoxicity of ag, au and ag-au bimetallic nanoparticles prepared using golden rod (solidago canadensis) plant extract. Sci. Rep. 2019, 9, 4169. [Google Scholar] [CrossRef] [PubMed]
- Chugh, H.; Sood, D.; Chandra, I.; Tomar, V.; Dhawan, G.; Chandra, R. Role of gold and silver nanoparticles in cancer nano-medicine. Artif. CellsNanomed. Biotechnol. 2018, 46, 1210–1220. [Google Scholar]
- Yamada, M.; Foote, M.; Prow, T.W. Therapeutic gold, silver, and platinum nanoparticles. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2015, 7, 428–445. [Google Scholar] [PubMed]
- Conde, J.; Doria, G.; Baptista, P. Noble metal nanoparticles applications in cancer. J. Drug Deliv. 2012, 2012, 751075. [Google Scholar] [CrossRef] [PubMed]
- Shanmugasundaram, T.; Radhakrishnan, M.; Gopikrishnan, V.; Kadirvelu, K.; Balagurunathan, R. Biocompatible silver, gold and silver/gold alloy nanoparticles for enhanced cancer therapy: In vitro and in vivo perspectives. Nanoscale 2017, 9, 16773–16790. [Google Scholar] [CrossRef]
- Katifelis, H.; Lyberopoulou, A.; Mukha, I.; Vityuk, N.; Grodzyuk, G.; Theodoropoulos, G.E.; Efstathopoulos, E.P.; Gazouli, M. Ag/au bimetallic nanoparticles induce apoptosis in human cancer cell lines via p53, caspase-3 and bax/bcl-2 pathways. Artif Cells Nanomed. Biotechnol. 2018, 46, S389–s398. [Google Scholar] [PubMed]
- Shmarakov, I.; Mukha, I.; Vityuk, N.; Borschovetska, V.; Zhyshchynska, N.; Grodzyuk, G.; Eremenko, A. Antitumor activity of alloy and core-shell-type bimetallic agau nanoparticles. Nanoscale Res. Lett. 2017, 12, 333. [Google Scholar] [CrossRef] [PubMed]
- Hao, M.; Kong, C.; Jiang, C.; Hou, R.; Zhao, X.; Li, J.; Wang, Y.; Gao, Y.; Zhang, H.; Yang, B.; et al. Polydopamine-coated au-ag nanoparticle-guided photothermal colorectal cancer therapy through multiple cell death pathways. Acta Biomater. 2019, 83, 414–424. [Google Scholar]
- Gnanasekar, S.; Balakrishnan, D.; Seetharaman, P.; Arivalagan, P.; Chandrasekaran, R.; Sivaperumal, S. Chrysin-anchored silver and gold nanoparticle-reduced graphene oxide composites for breast cancer therapy. ACS Appl. Nano Mater. 2020, 3, 4574–4585. [Google Scholar] [CrossRef]
- Abdel-Fattah, W.; Ali, G. On the anti-cancer activities of silver nanoparticles. J. Appl. Biotechnol. Bioeng. 2018, 5, 43–46. [Google Scholar] [CrossRef]
- Gomathi, A.C.; Xavier Rajarathinam, S.R.; Mohammed Sadiq, A.; Rajeshkumar, S. Anticancer activity of silver nanoparticles synthesized using aqueous fruit shell extract of tamarindus indica on mcf-7 human breast cancer cell line. J. Drug Deliv. Sci. Technol. 2020, 55, 101376. [Google Scholar]
- Xu, L.; Wang, Y.-Y.; Huang, J.; Chen, C.-Y.; Wang, Z.-X.; Xie, H. Silver nanoparticles: Synthesis, medical applications and biosafety. Theranostics 2020, 10, 8996–9031. [Google Scholar] [CrossRef] [PubMed]
- Noh, M.S.; Lee, S.; Kang, H.; Yang, J.-K.; Lee, H.; Hwang, D.; Lee, J.W.; Jeong, S.; Jang, Y.; Jun, B.-H. Target-specific near-ir induced drug release and photothermal therapy with accumulated au/ag hollow nanoshells on pulmonary cancer cell membranes. Biomaterials 2015, 45, 81–92. [Google Scholar] [PubMed]
- Meyers, J.D.; Cheng, Y.; Broome, A.-M.; Agnes, R.S.; Schluchter, M.D.; Margevicius, S.; Wang, X.; Kenney, M.E.; Burda, C.; Basilion, J.P. Peptide-targeted gold nanoparticles for photodynamic therapy of brain cancer. Part. Part. Syst. Charact. 2015, 32, 448–457. [Google Scholar] [PubMed]
- Guo, Z.; Chen, Y.; Wang, Y.; Jiang, H.; Wang, X. Advances and challenges in metallic nanomaterial synthesis and antibacterial applications. J. Mater. Chem. B 2020, 8, 4764–4777. [Google Scholar] [CrossRef] [PubMed]
- Cheeseman, S.; Christofferson, A.J.; Kariuki, R.; Cozzolino, D.; Daeneke, T.; Crawford, R.J.; Truong, V.K.; Chapman, J.; Elbourne, A. Antimicrobial metal nanomaterials: From passive to stimuli-activated applications. Adv. Sci. 2020, 7, 1902913. [Google Scholar] [CrossRef] [PubMed]
- Vimbela, G.V.; Ngo, S.M.; Fraze, C.; Yang, L.; Stout, D.A. Antibacterial properties and toxicity from metallic nanomaterials. Int. J. Nanomed. 2017, 12, 3941–3965. [Google Scholar] [CrossRef]
- Valodkar, M.; Modi, S.; Pal, A.; Thakore, S. Synthesis and anti-bacterial activity of cu, ag and cu–ag alloy nanoparticles: A green approach. Mater. Res. Bull. 2011, 46, 384–389. [Google Scholar] [CrossRef]
- Hu, X.; Zhao, Y.; Hu, Z.; Saran, A.; Hou, S.; Wen, T.; Liu, W.; Ji, Y.; Jiang, X.; Wu, X. Gold nanorods core/agpt alloy nanodots shell: A novel potent antibacterial nanostructure. Nano Res. 2013, 6, 822–835. [Google Scholar] [CrossRef]
- Jiang, F.; Zhu, W.; Zhao, C.; Li, Y.; Wei, P.; Wan, T.; Ye, H.; Pan, S.; Ren, F. A strong, wear- and corrosion-resistant, and antibacterial co–30 at.% cr–5 at.% ag ternary alloy for medical implants. Mater. Des. 2019, 184, 108190. [Google Scholar]
- Mo, A.H.; Landon, P.B.; Gomez, K.S.; Kang, H.; Lee, J.; Zhang, C.; Janetanakit, W.; Sant, V.; Lu, T.; Colburn, D.A.; et al. Magnetically-responsive silica–gold nanobowls for targeted delivery and sers-based sensing. Nanoscale 2016, 8, 11840–11850. [Google Scholar] [CrossRef]
- Li, Y.-Q.; Xu, M.; Dhawan, U.; Liu, W.-C.; Wu, K.-T.; Liu, X.-R.; Lin, C.; Zhao, G.; Wu, Y.-C.; Chung, R.-J. Iron-gold alloy nanoparticles serve as a cornerstone in hyperthermia-mediated controlled drug release for cancer therapy. Int. J. Nanomed. 2018, 13, 5499–5509. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Lau, K.C.; Hsu, J.C.; Hajfathalian, M.; Mian, S.; Chhour, P.; Uppuluri, L.; McDonald, E.S.; Maidment, A.D.; Cormode, D.P. Gold silver alloy nanoparticles (gsan): An imaging probe for breast cancer screening with dual-energy mammography or computed tomography. Nanoscale 2016, 8, 13740–13754. [Google Scholar] [CrossRef]
- Maenosono, S.; Suzuki, T.; Saita, S. Superparamagnetic fept nanoparticles as excellent mri contrast agents. J. Magn. Magn. Mater. 2008, 320, L79–L83. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, J.; Tian, Q.; Hu, H.; Fang, Y.; Wu, H.; Yang, S. One-pot synthesis of amphiphilic superparamagnetic fept nanoparticles and magnetic resonance imaging in vitro. J. Magn. Magn. Mater. 2010, 322, 973–977. [Google Scholar] [CrossRef]
- Yang, H.; Li, X.; Zhou, H.; Zhuang, Y.; Hu, H.; Wu, H.; Yang, S. Monodisperse water-soluble fe–ni nanoparticles for magnetic resonance imaging. J. Alloy. Compd. 2011, 509, 1217–1221. [Google Scholar] [CrossRef]
- Chou, S.-W.; Liu, C.-L.; Liu, T.-M.; Shen, Y.-F.; Kuo, L.-C.; Wu, C.-H.; Hsieh, T.-Y.; Wu, P.-C.; Tsai, M.-R.; Yang, C.-C.; et al. Infrared-active quadruple contrast fept nanoparticles for multiple scale molecular imaging. Biomaterials 2016, 85, 54–64. [Google Scholar] [CrossRef] [PubMed]
- Nune, S.K.; Gunda, P.; Thallapally, P.K.; Lin, Y.-Y.; Laird Forrest, M.; Berkland, C.J. Nanoparticles for biomedical imaging. Expert Opin. Drug Del. 2009, 6, 1175–1194. [Google Scholar] [CrossRef]
- Choi, H.S.; Frangioni, J.V. Nanoparticles for biomedical imaging: Fundamentals of clinical translation. Mol. Imaging 2010, 9, 7290. [Google Scholar] [CrossRef]
- Lussier, F.; Thibault, V.; Charron, B.; Wallace, G.Q.; Masson, J.-F. Deep learning and artificial intelligence methods for raman and surface-enhanced raman scattering. Trac. Trends Anal. Chem. 2020, 124, 115796. [Google Scholar] [CrossRef]
- Hu, W.; Ye, S.; Zhang, Y.; Li, T.; Zhang, G.; Luo, Y.; Mukamel, S.; Jiang, J. Machine learning protocol for surface-enhanced raman spectroscopy. J. Phys. Chem. Lett. 2019, 10, 6026–6031. [Google Scholar] [CrossRef] [PubMed]
- Mandrell, C.T.; Holland, T.E.; Wheeler, J.F.; Esmaeili, S.M.A.; Amar, K.; Chowdhury, F.; Sivakumar, P. Machine learning approach to raman spectrum analysis of mia paca-2 pancreatic cancer tumor repopulating cells for classification and feature analysis. Life 2020, 10, 181. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.H.; Hong, B.; Rubin, S.; Fainman, Y. Machine learning for composition analysis of ssdna using chemical enhancement in sers. Biomed. Opt. Express 2020, 11, 5092–5121. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huynh, K.-H.; Hahm, E.; Noh, M.S.; Lee, J.-H.; Pham, X.-H.; Lee, S.H.; Kim, J.; Rho, W.-Y.; Chang, H.; Kim, D.M.; et al. Recent Advances in Surface-Enhanced Raman Scattering Magnetic Plasmonic Particles for Bioapplications. Nanomaterials 2021, 11, 1215. https://doi.org/10.3390/nano11051215
Huynh K-H, Hahm E, Noh MS, Lee J-H, Pham X-H, Lee SH, Kim J, Rho W-Y, Chang H, Kim DM, et al. Recent Advances in Surface-Enhanced Raman Scattering Magnetic Plasmonic Particles for Bioapplications. Nanomaterials. 2021; 11(5):1215. https://doi.org/10.3390/nano11051215
Chicago/Turabian StyleHuynh, Kim-Hung, Eunil Hahm, Mi Suk Noh, Jong-Hwan Lee, Xuan-Hung Pham, Sang Hun Lee, Jaehi Kim, Won-Yeop Rho, Hyejin Chang, Dong Min Kim, and et al. 2021. "Recent Advances in Surface-Enhanced Raman Scattering Magnetic Plasmonic Particles for Bioapplications" Nanomaterials 11, no. 5: 1215. https://doi.org/10.3390/nano11051215
APA StyleHuynh, K.-H., Hahm, E., Noh, M. S., Lee, J.-H., Pham, X.-H., Lee, S. H., Kim, J., Rho, W.-Y., Chang, H., Kim, D. M., Baek, A., Kim, D.-E., Jeong, D. H., Park, S.-m., & Jun, B.-H. (2021). Recent Advances in Surface-Enhanced Raman Scattering Magnetic Plasmonic Particles for Bioapplications. Nanomaterials, 11(5), 1215. https://doi.org/10.3390/nano11051215






