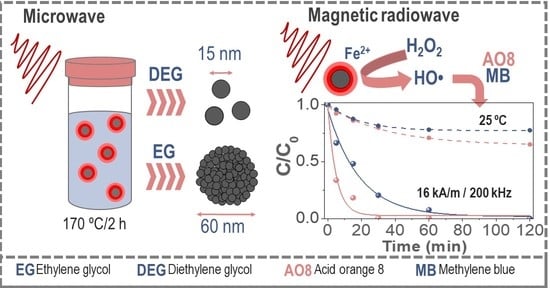
Engineering Iron Oxide Nanocatalysts by a Microwave-Assisted Polyol Method for the Magnetically Induced Degradation of Organic Pollutants
Abstract
1. Introduction
2. Materials and Methods
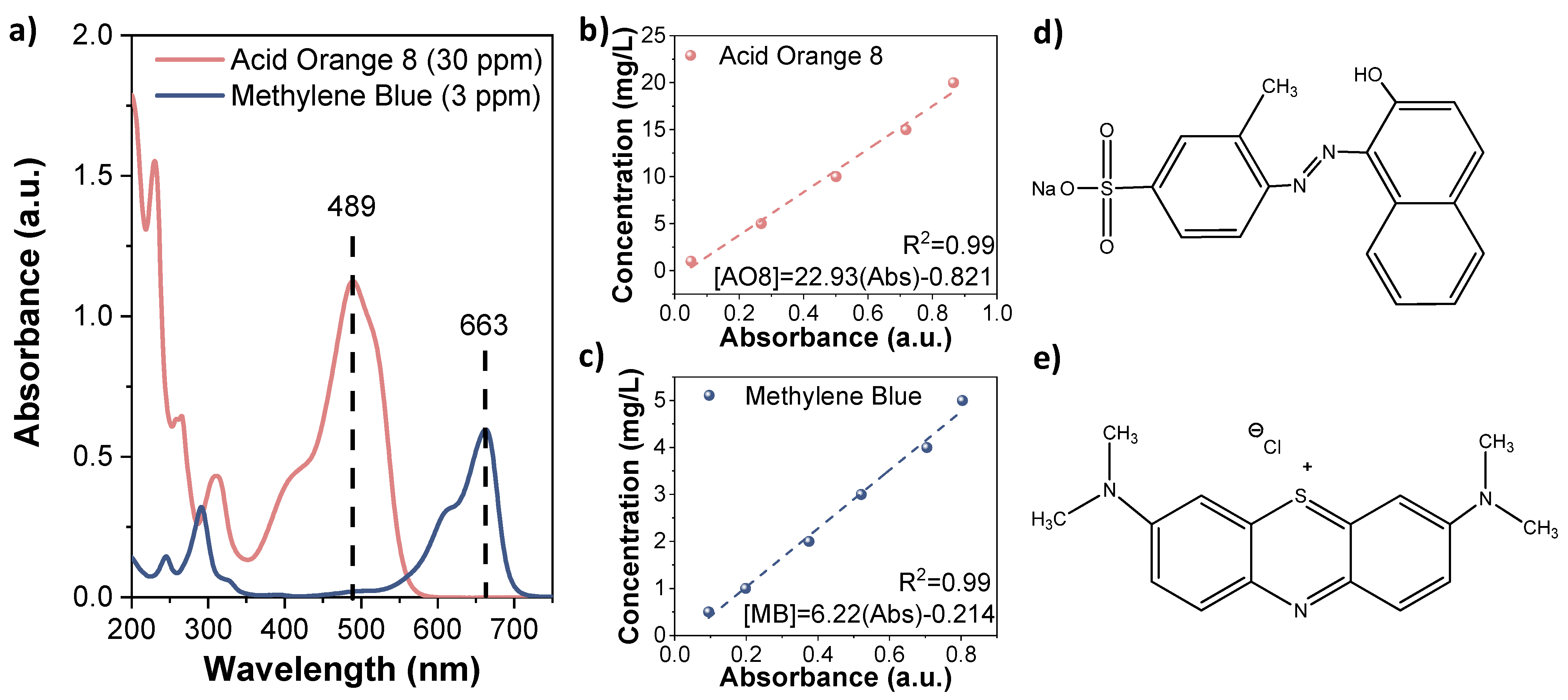
2.1. Chemical Reagents and Analysis
2.2. Synthesis of Iron Oxide Nanoparticles
2.3. Characterization of Iron Oxide Nanoparticles
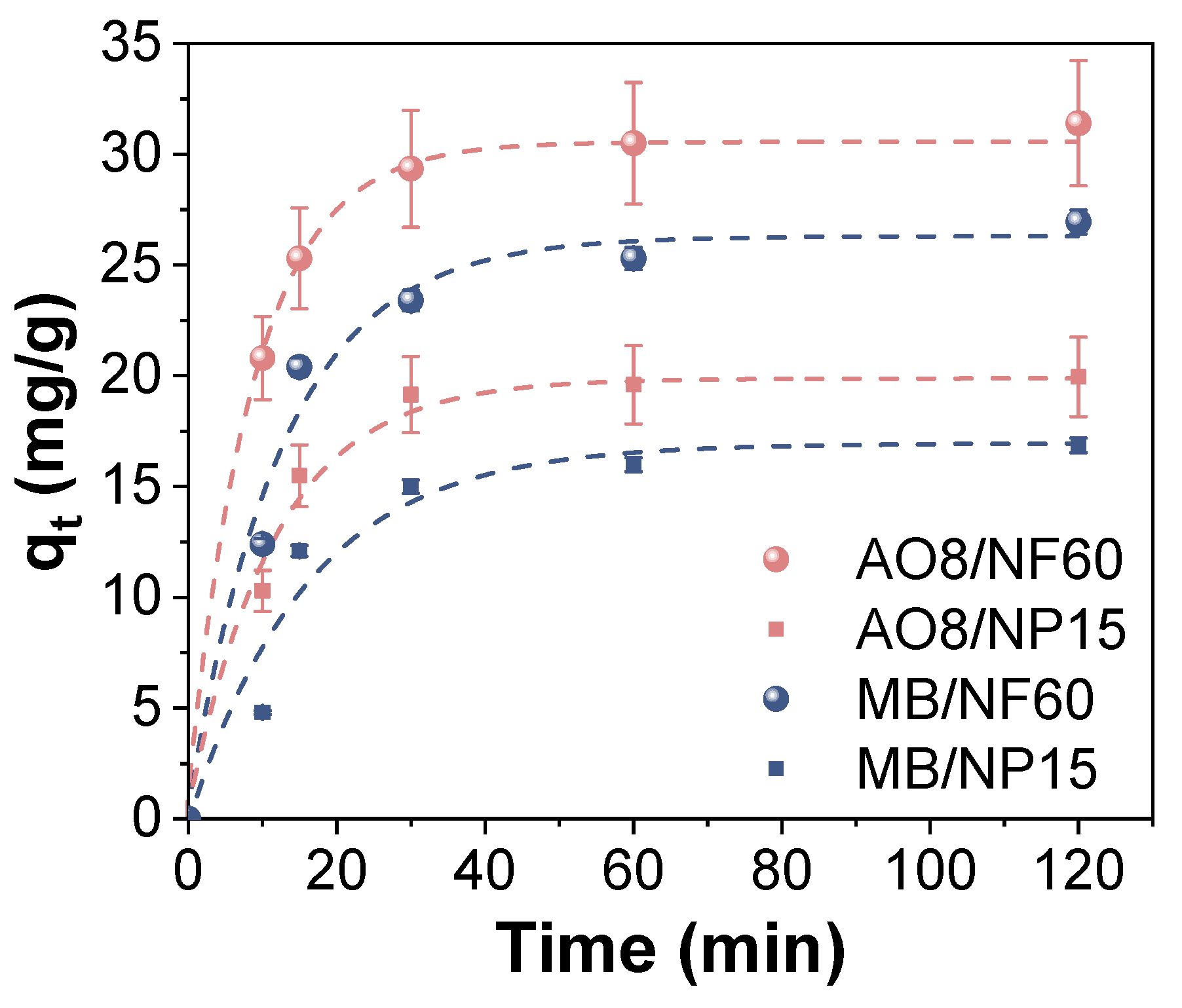
2.4. Adsorption Experimentation
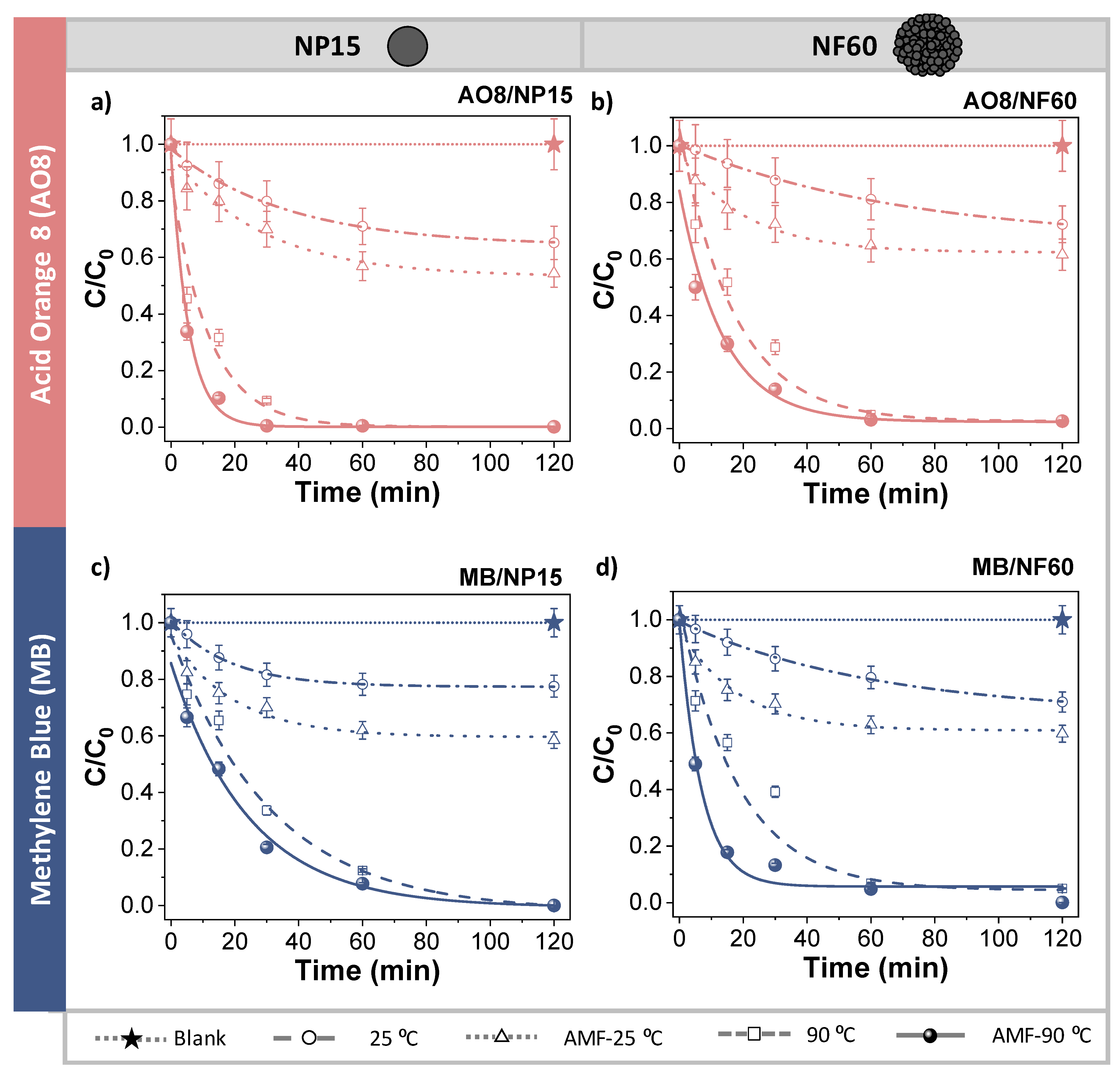
2.5. Thermal and Magnetic Induction Heating-Assisted Degradation
3. Results
3.1. Microwave-Assisted Synthesis
3.2. Iron Oxide Nanoparticles Characterization
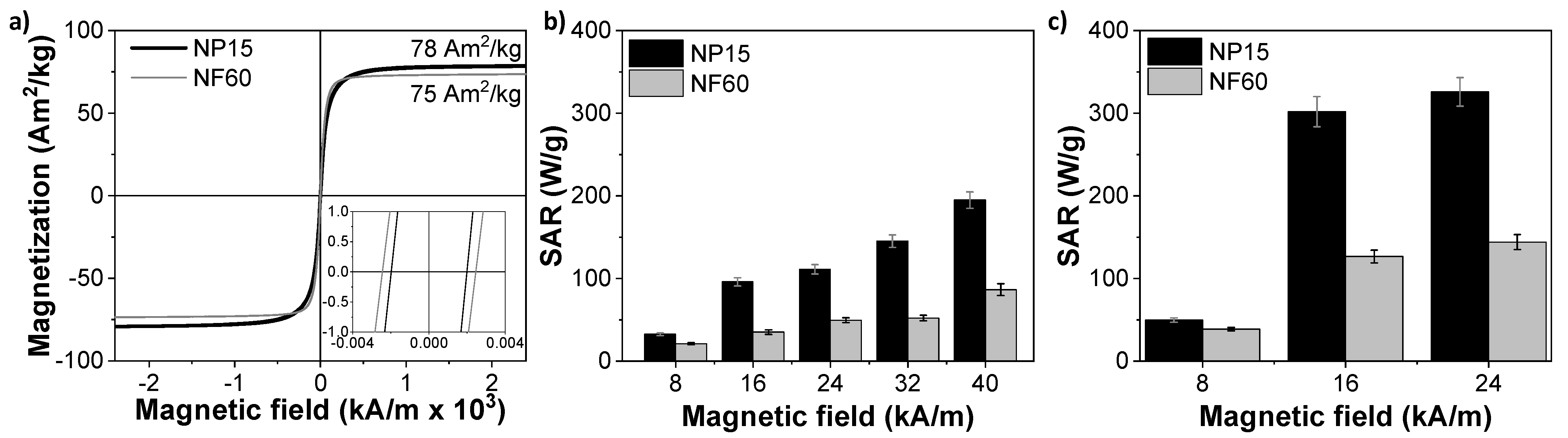
3.3. Magnetic Properties
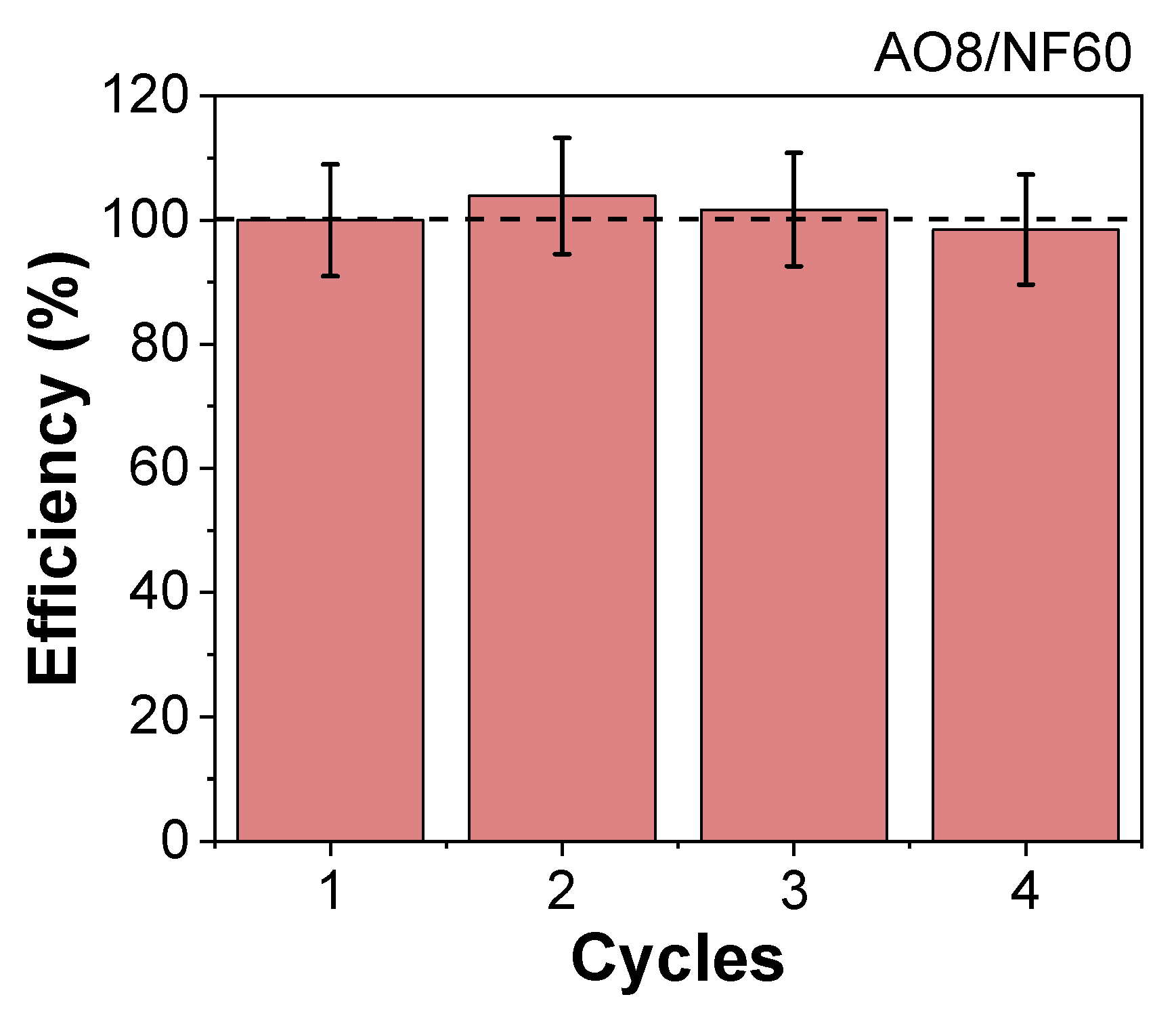
3.4. Thermal Degradation and Magnetic Induction Heating-Assisted Degradation
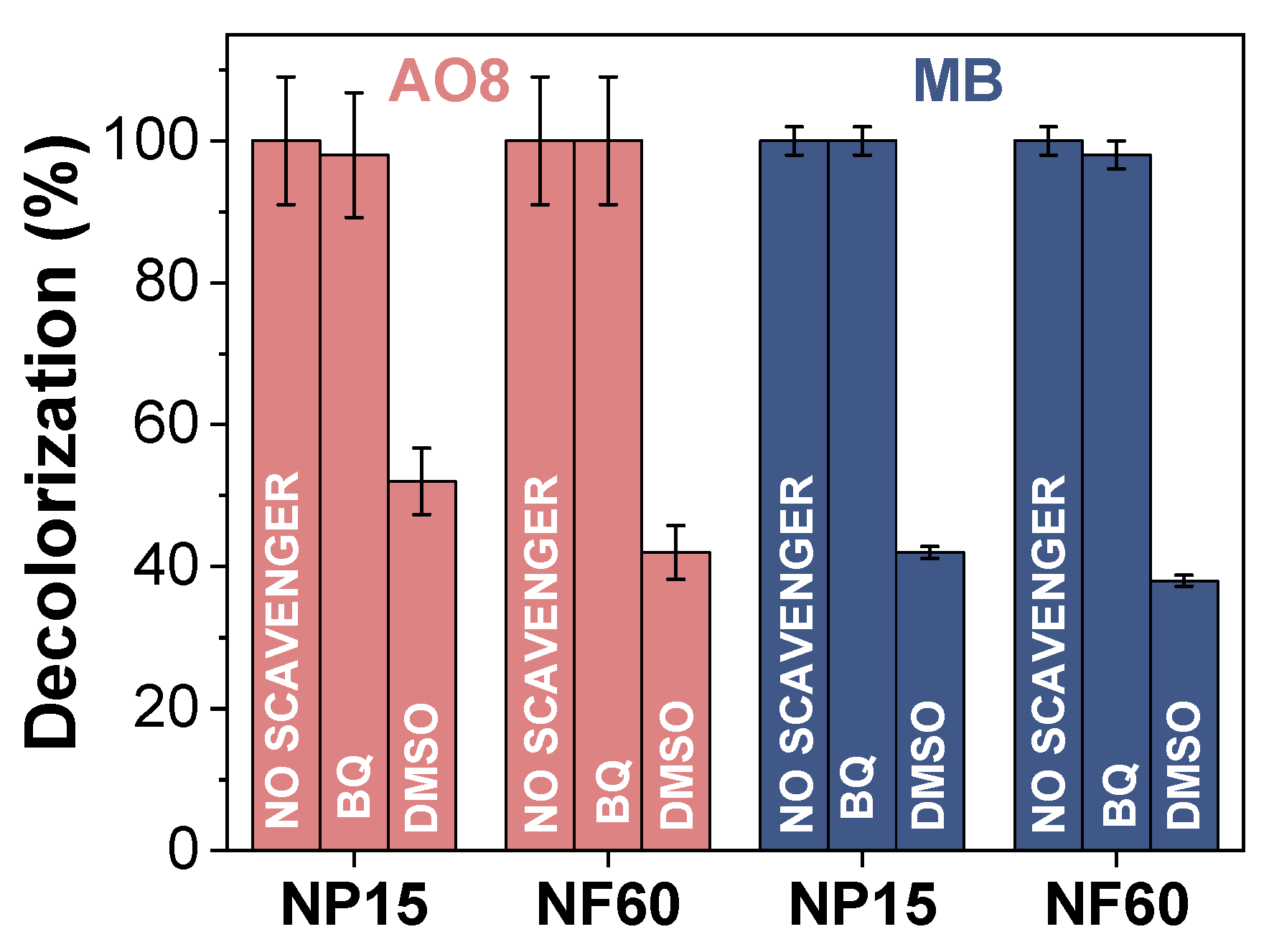
3.5. Proposed Reaction Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kolahalam, L.A.; Kasi Viswanath, I.V.; Diwakar, B.S.; Govindh, B.; Reddy, V.; Murthy, Y.L.N. Review on nanomaterials: Synthesis and applications. Mater. Today Proc. 2019, 18, 2182–2190. [Google Scholar] [CrossRef]
- Yusefi, M.; Shameli, K.; Jumaat, A.F. Preparation and Properties of Magnetic Iron Oxide Nanoparticles for Biomedical Applications: A Brief Review. J. Adv. Res. Mater. Sci. 2020, 75, 10–18. [Google Scholar] [CrossRef]
- Gallo-Cordova, A.; Almeida-Streitwieser, D.; Morales, M.P.; Ovejero, J.G. Magnetic Iron Oxide Colloids for Environmental Applications. In Colloids—Types, Preparation and Applications; Intech Open: London, UK, 2021; pp. 1–25. [Google Scholar]
- Roca, A.G.; Gutiérrez, L.; Gavilán, H.; Fortes Brollo, M.E.; Veintemillas-Verdaguer, S.; Morales, M.P. Design strategies for shape-controlled magnetic iron oxide nanoparticles. Adv. Drug Deliv. Rev. 2019, 138, 68–104. [Google Scholar] [CrossRef] [PubMed]
- Leonel, A.G.; Mansur, A.A.P.; Mansur, H.S. Advanced Functional Nanostructures based on Magnetic Iron Oxide Nanomaterials for Water Remediation: A Review. Water Res. 2021, 190, 116693. [Google Scholar] [CrossRef] [PubMed]
- Tara, N.; Siddiqui, S.I.; Rathi, G.; Chaudhry, S.A.; Inamuddin; Asiri, A.M. Nano-engineered Adsorbent for the Removal of Dyes from Water: A Review. Curr. Anal. Chem. 2019, 16, 14–40. [Google Scholar] [CrossRef]
- Tkaczyk, A.; Mitrowska, K.; Posyniak, A. Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems: A review. Sci. Total Environ. 2020, 717, 137222. [Google Scholar] [CrossRef]
- Chung, K.T. Azo dyes and human health: A review. J. Environ. Sci. Health Part C Environ. Carcinog. Ecotoxicol. Rev. 2016, 34, 233–261. [Google Scholar] [CrossRef]
- Schirmer, R.H.; Adler, H.; Pickhardt, M.; Mandelkow, E. Lest we forget you—Methylene blue…. Neurobiol. Aging 2011, 32, 2325.e7–2325.e16. [Google Scholar] [CrossRef]
- Singh, P.K.; Singh, R.L. Bio-removal of Azo Dyes: A Review. Int. J. Appl. Sci. Biotechnol. 2017, 5, 108–126. [Google Scholar] [CrossRef]
- Yalcin-enis, I.; Kucukali-ozturk, M.; Sezgin, H. Nanoscience and Biotechnology for Environmental Applications; Springer: Berlin, Germany, 2019; Volume 22, ISBN 978-3-319-97921-2. [Google Scholar]
- Kaykhaii, M.; Sasani, M.; Marghzari, S. Removal of Dyes from the Environment by Adsorption Process. Chem. Mater. Eng. 2018, 6, 31–35. [Google Scholar] [CrossRef]
- Rekhate, C.V.; Srivastava, J.K. Recent advances in ozone-based advanced oxidation processes for treatment of wastewater—A review. Chem. Eng. J. Adv. 2020, 3, 100031. [Google Scholar] [CrossRef]
- Brillas, E. A review on the photoelectro-Fenton process as efficient electrochemical advanced oxidation for wastewater remediation. Treatment with UV light, sunlight, and coupling with conventional and other photo-assisted advanced technologies. Chemosphere 2020, 250, 126198. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Lu, J.; Zhou, Y.; Liu, Y. Recent advances for dyes removal using novel adsorbents: A review. Environ. Pollut. 2019, 252, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Tuci, G.; Duong-Viet, C.; Liu, Y.; Rossin, A.; Luconi, L.; Nhut, J.M.; Nguyen-Dinh, L.; Pham-Huu, C.; Giambastiani, G. Induction Heating: An Enabling Technology for the Heat Management in Catalytic Processes. ACS Catal. 2019, 9, 7921–7935. [Google Scholar] [CrossRef]
- Gallo-Cordova, A.; Lemus, J.; Palomares, F.J.; Morales, M.P.; Mazarío, E. Superparamagnetic nanosorbent for water purification: Assessment of the adsorptive removal of lead and methyl orange from aqueous solutions. Sci. Total Environ. 2020, 711, 134644. [Google Scholar] [CrossRef]
- Gallo-Cordova, A.; Morales, M.P.; Mazarío, E. Effect of the surface charge on the adsorption capacity of chromium(VI) of iron oxide magnetic nanoparticles prepared by microwave-assisted synthesis. Water 2019, 11, 2372. [Google Scholar] [CrossRef]
- Lingamdinne, L.P.; Choi, J.S.; Choi, Y.L.; Chang, Y.Y.; Yang, J.K.; Karri, R.R.; Koduru, J.R. Process modeling and optimization of an iron oxide immobilized graphene oxide gadolinium nanocomposite for arsenic adsorption. J. Mol. Liq. 2020, 299, 112261. [Google Scholar] [CrossRef]
- Rivera, F.L.; Recio, F.J.; Palomares, F.J.; Sánchez-Marcos, J.; Menéndez, N.; Mazarío, E.; Herrasti, P. Fenton-like degradation enhancement of methylene blue dye with magnetic heating induction. J. Electroanal. Chem. 2020, 114773. [Google Scholar] [CrossRef]
- Munoz, M.; Nieto-Sandoval, J.; Serrano, E.; De Pedro, Z.M.; Casas, J.A. CWPO intensification by induction heating using magnetite as catalyst. J. Environ. Chem. Eng. 2020, 8, 104085. [Google Scholar] [CrossRef]
- Mustieles Marin, I.; De Masi, D.; Lacroix, L.-M.; Fazzini, P.-F.; van Leeuwen, P.W.N.M.; Asensio, J.M.; Chaudret, B. Hydrodeoxygenation and hydrogenolysis of biomass-based materials using FeNi catalysts and magnetic induction. Green Chem. 2021, 2025–2036. [Google Scholar] [CrossRef]
- Gatard, V.; De Masi, D.; Chattot, R.; Marin, I.M.; Revert, J.M.A.; Fazzini, P.F.; Encinas, T.; Martin, V.; Faure, S.; Deseure, J.; et al. FeNi3 and Ni-Based Nanoparticles as Electrocatalysts for Magnetically Enhanced Alkaline Water Electrolysis. Electrocatalysis 2021, 11, 567–577. [Google Scholar] [CrossRef]
- Rivas-Murias, B.; Asensio, J.M.; Mille, N.; Rodríguez-Gonzµlez, B.; Fazzini, P.; Carrey, J.; Chaudret, B.; Salgueiriço, V. Magnetically Induced CO2 Methanation Using Exchange-Coupled Spinel Ferrites in Cuboctahedron-Shaped Nanocrystals. Angew. Chem. 2020, 59, 15537–15542. [Google Scholar] [CrossRef]
- Fievet, F.; Lagier, J.P.; Figlarz, M. Preparing Monodisperse Metal Powders in Micrometer and Submicrometer Sizes by the Polyol Process. MRS Bull. 1989, 14, 29–34. [Google Scholar] [CrossRef]
- Fievet, F.; Ammar-Merah, S.; Brayner, R.; Chau, F.; Giraud, M.; Mammeri, F.; Peron, J.; Piquemal, J.Y.; Sicard, L.; Viau, G. The polyol process: A unique method for easy access to metal nanoparticles with tailored sizes, shapes and compositions. Chem. Soc. Rev. 2018, 47, 5187–5233. [Google Scholar] [CrossRef] [PubMed]
- Varanda, L.C.; Souza, C.G.S.; Moraes, D.A.; Neves, H.R.; Junior, J.B.S.; Silva, M.F.; Bini, R.A.; Albers, R.F.; Silva, T.L.; Beck, W. Size and shape-controlled nanomaterials based on modified polyol and thermal decomposition approaches. A brief review. An. Acad. Bras. Cienc. 2019, 91, 1–32. [Google Scholar] [CrossRef]
- Brollo, M.E.F.; Veintemillas-Verdaguer, S.; Salván, C.M.; Morales, M.P. Key parameters on the microwave assisted synthesis of magnetic nanoparticles for MRI contrast agents. Contrast Media Mol. Imaging 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Gallo-Cordova, A.; Espinosa, A.; Serrano, A.; Gutiérrez, L.; Menéndez, N.; Morales, M.P.; Mazario, E. New insights into the structural analysis of maghemite and (MFe2O4, M = Co, Zn) ferrite nanoparticles synthesized by a microwave-assisted polyol process. Mater. Chem. Front. 2020, 4, 3063–3073. [Google Scholar] [CrossRef]
- de Greñu, B.D.; Torres, J.; García-González, J.; Muñoz-Pina, S.; de los Reyes, R.; Costero, A.M.; Amorós, P.; Ros-Lis, J.V. Microwave-Assisted Synthesis of Covalent Organic Frameworks: A Review. ChemSusChem 2021, 14, 208–233. [Google Scholar] [CrossRef] [PubMed]
- Bandala, E.R.; Peláez, M.A.; García-López, A.J.; de Salgado, M.J.; Moeller, G. Photocatalytic decolourisation of synthetic and real textile wastewater containing benzidine-based azo dyes. Chem. Eng. Process. Process Intensif. 2008, 47, 169–176. [Google Scholar] [CrossRef]
- Koizumi, N. Dielectric properties of polyethylene glycols at microwave frequencies. J. Chem. Phys. 1957, 27, 625–630. [Google Scholar] [CrossRef]
- Gavilán, H.; Sánchez, E.H.; Brollo, M.E.F.; Asín, L.; Moerner, K.K.; Frandsen, C.; Lázaro, F.J.; Serna, C.J.; Veintemillas-Verdaguer, S.; Morales, M.P.; et al. Formation Mechanism of Maghemite Nanoflowers Synthesized by a Polyol-Mediated Process. ACS Omega 2017, 2, 7172–7184. [Google Scholar] [CrossRef] [PubMed]
- Mirabello, G.; Ianiro, A.; Bomans, P.H.H.; Yoda, T.; Arakaki, A.; Friedrich, H.; de With, G.; Sommerdijk, N.A.J.M. Crystallization by particle attachment is a colloidal assembly process. Nat. Mater. 2020, 19, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Chen, Y.C.; Feldmann, C. Polyol synthesis of nanoparticles: Status and options regarding metals, oxides, chalcogenides, and non-metal elements. Green Chem. 2015, 17, 4107–4132. [Google Scholar] [CrossRef]
- Soltani, N.; Saion, E.; Hussein, M.; Bahrami, A.; Naghavi, K.; Yunus, R. Microwave irradiation effects on hydrothermal and polyol synthesis of ZnS nanoparticles. Chalcogenide Lett. 2012, 9, 265–274. [Google Scholar]
- Hadjiivanov, K.I.; Panayotov, D.A.; Mihaylov, M.Y.; Ivanova, E.Z.; Chakarova, K.K.; Andonova, S.M.; Drenchev, N.L. Power of Infrared and Raman Spectroscopies to Characterize Metal-Organic Frameworks and Investigate Their Interaction with Guest Molecules. Chem. Rev. 2021, 121, 1286–1424. [Google Scholar] [CrossRef] [PubMed]
- Kemp, S.J.; Ferguson, R.M.; Khandhar, A.P.; Krishnan, K.M. Monodisperse magnetite nanoparticles with nearly ideal saturation magnetization. RSC Adv. 2016, 6, 77452–77464. [Google Scholar] [CrossRef]
- Roca, A.G.; Veintemillas-Verdaguer, S.; Port, M.; Robic, C.; Serna, C.J.; Morales, M.P. Agents Based on High Quality Magnetite Nanoparticles. J. Phys. Chem. B 2009, 113, 7033–7039. [Google Scholar] [CrossRef]
- Li, Q.; Kartikowati, C.W.; Horie, S.; Ogi, T.; Iwaki, T.; Okuyama, K. Correlation between particle size/domain structure and magnetic properties of highly crystalline Fe3O4 nanoparticles. Sci. Rep. 2017, 7, 9894. [Google Scholar] [CrossRef]
- Salas, G.; Casado, C.; Teran, F.J.; Miranda, R.; Serna, C.J.; Morales, M.P. Controlled synthesis of uniform magnetite nanocrystals with high-quality properties for biomedical applications. J. Mater. Chem. 2012, 22, 21065–21075. [Google Scholar] [CrossRef]
- Ovejero, J.G.; Cabrera, D.; Carrey, J.; Valdivielso, T.; Salas, G.; Teran, F.J. Effects of inter- and intra-aggregate magnetic dipolar interactions on the magnetic heating efficiency of iron oxide nanoparticles. Phys. Chem. Chem. Phys. 2016, 18, 10954–10963. [Google Scholar] [CrossRef]
- Lartigue, L.; Hugounenq, P.; Alloyeau, D.; Clarke, S.P.; Lévy, M.; Bacri, J.C.; Bazzi, R.; Brougham, D.F.; Wilhelm, C.; Gazeau, F. Cooperative organization in iron oxide multi-core nanoparticles potentiates their efficiency as heating mediators and MRI contrast agents. ACS Nano 2012, 6, 10935–10949. [Google Scholar] [CrossRef] [PubMed]
- Salas, G.; Camarero, J.; Cabrera, D.; Takacs, H.; Varela, M.; Ludwig, R.; Dähring, H.; Hilger, I.; Miranda, R.; Morales, M.P.; et al. Modulation of magnetic heating via dipolar magnetic interactions in monodisperse and crystalline iron oxide nanoparticles. J. Phys. Chem. C 2014, 118, 19985–19994. [Google Scholar] [CrossRef]
- Bokare, A.D.; Choi, W. Zero-valent aluminum for oxidative degradation of aqueous organic pollutants. Environ. Sci. Technol. 2009, 43, 7130–7135. [Google Scholar] [CrossRef] [PubMed]
- Riedinger, A.; Guardia, P.; Curcio, A.; Garcia, M.A.; Cingolani, R.; Manna, L.; Pellegrino, T. Subnanometer local temperature probing and remotely controlled drug release based on Azo-functionalized iron oxide nanoparticles. Nano Lett. 2013, 13, 2399–2406. [Google Scholar] [CrossRef] [PubMed]
- Giwa, A.R.A.; Bello, I.A.; Olabintan, A.B.; Bello, O.S.; Saleh, T.A. Kinetic and thermodynamic studies of fenton oxidative decolorization of methylene blue. Heliyon 2020, 6, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Fu, X.; Yu, J.; Xu, Y.; Huang, J.; Li, Q.; Sun, D. Green synthesized iron nanoparticles as highly efficient fenton-like catalyst for degradation of dyes. Chemosphere 2020, 261, 127618. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Chen, S.; Xiao, H.; Zhang, J.; Lan, J.; Yan, B.; Yan, B.; Zeng, H. Ultra-efficient and stable heterogeneous iron-based Fenton nanocatalysts for degrading organic dyes at neutral pH: Via a chelating effect under nanoconfinement. Chem. Commun. 2020, 56, 6571–6574. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Lee, J.; Zheng, Y.; Duan, Q. A non-traditional energy transfer process in CWPO heterogeneous reaction for wastewater treatment. Chem. Eng. Res. Des. 2016, 114, 142–147. [Google Scholar] [CrossRef]
- Leiw, M.Y.; Guai, G.H.; Wang, X.; Tse, M.S.; Ng, C.M.; Tan, O.K. Dark ambient degradation of Bisphenol A and Acid Orange 8 as organic pollutants by perovskite SrFeO3-δ metal oxide. J. Hazard. Mater. 2013, 260, 1–8. [Google Scholar] [CrossRef]
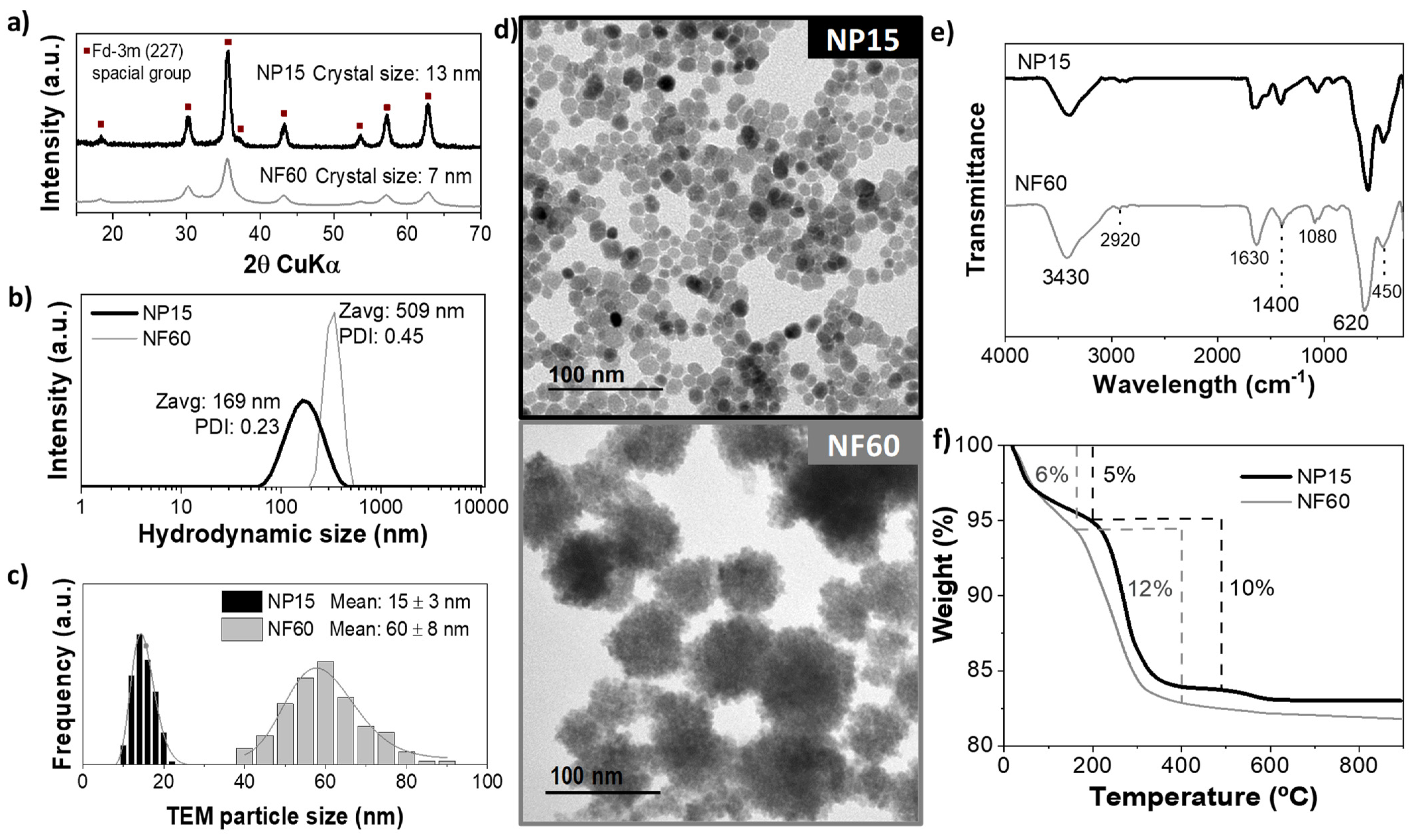
| Sample | Solvent | Water | Heating Rate | Temp. | Structure | TEM Size |
|---|---|---|---|---|---|---|
| (%) | (°C/min) | (°C) | (nm) | |||
| Ramp1 | DEG | 0 | 3.75 | 220 | Single core | 5.8 ± 1.0 |
| Ramp2 | DEG | 0 | 7.30 | 220 | Single core | 6.4 ± 1.0 |
| Ramp3 | DEG | 0 | 14.6 | 220 | Single core | 7 ± 1 |
| NP15 | DEG | 3.7 | 3.75 | 170 | Single core | 15 ± 3 |
| Solv1 | TREG | 3.7 | 3.75 | 170 | Single core | <5 |
| Solv2 | TEG | 3.7 | 3.75 | 170 | Single core | <5 |
| NF60 | EG | 3.7 | 3.75 | 170 | Multicore | 60 ± 8 |
| Parameter | NP15 | NF60 |
|---|---|---|
| BET area (m2/g) | 60.1 ± 0.2 | 118.3 ± 2.1 |
| Pore volume (cm3/g) | 0.14 | 0.19 |
| MIONPs | Dye | Ti Ϯ | Tf ϮϮ | AMF * | DY | kapp1 ** | R2 ** | t1/2 ** | |
|---|---|---|---|---|---|---|---|---|---|
| g/L | °C | °C | % | (10−3) min−1 | min | ||||
| NP15 | 1 | AO8 | 25 | 25 | OFF | 31 | 7.10 | 0.9532 | 98 |
| NP15 | 1 | AO8 | 25 | 50 | ON | 46 | N/C | N/C | N/C |
| NP15 | 1 | AO8 | 90 | 90 | OFF | 100 | 69.9 | 0.8097 | 9.9 |
| NP15 | 2 | AO8 | 90 | 90 | ON | 100 | 106 | 0.8804 | 6.5 |
| NF60 | 1 | AO8 | 25 | 25 | OFF | 33 | 4.40 | 0.9999 | 158 |
| NF60 | 1 | AO8 | 25 | 35 | ON | 40 | N/C | N/C | N/C |
| NF60 | 1 | AO8 | 90 | 90 | OFF | 100 | 23.1 | 0.9143 | 30 |
| NF60 | 3 | AO8 | 90 | 90 | ON | 100 | 61.6 | 0.9295 | 11.2 |
| NP15 | 1 | MB | 25 | 25 | OFF | 22 | 6.80 | 0.9999 | 102 |
| NP15 | 1 | MB | 25 | 50 | ON | 41 | N/C | N/C | N/C |
| NP15 | 1 | MB | 90 | 90 | OFF | 100 | 34.5 | 0.9999 | 20 |
| NP15 | 2 | MB | 90 | 90 | ON | 100 | 46.1 | 0.9341 | 15 |
| NF60 | 1 | MB | 25 | 25 | OFF | 29 | 3.70 | 0.9659 | 157 |
| NF60 | 1 | MB | 25 | 35 | ON | 40 | N/C | N/C | N/C |
| NF60 | 1 | MB | 90 | 90 | OFF | 100 | 35.8 | 0.9134 | 19 |
| NF60 | 3 | MB | 90 | 90 | ON | 100 | 65.4 | 0.8626 | 10.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gallo-Cordova, A.; Veintemillas-Verdaguer, S.; Tartaj, P.; Mazarío, E.; Morales, M.d.P.; Ovejero, J.G. Engineering Iron Oxide Nanocatalysts by a Microwave-Assisted Polyol Method for the Magnetically Induced Degradation of Organic Pollutants. Nanomaterials 2021, 11, 1052. https://doi.org/10.3390/nano11041052
Gallo-Cordova A, Veintemillas-Verdaguer S, Tartaj P, Mazarío E, Morales MdP, Ovejero JG. Engineering Iron Oxide Nanocatalysts by a Microwave-Assisted Polyol Method for the Magnetically Induced Degradation of Organic Pollutants. Nanomaterials. 2021; 11(4):1052. https://doi.org/10.3390/nano11041052
Chicago/Turabian StyleGallo-Cordova, Alvaro, Sabino Veintemillas-Verdaguer, Pedro Tartaj, Eva Mazarío, María del Puerto Morales, and Jesús G. Ovejero. 2021. "Engineering Iron Oxide Nanocatalysts by a Microwave-Assisted Polyol Method for the Magnetically Induced Degradation of Organic Pollutants" Nanomaterials 11, no. 4: 1052. https://doi.org/10.3390/nano11041052
APA StyleGallo-Cordova, A., Veintemillas-Verdaguer, S., Tartaj, P., Mazarío, E., Morales, M. d. P., & Ovejero, J. G. (2021). Engineering Iron Oxide Nanocatalysts by a Microwave-Assisted Polyol Method for the Magnetically Induced Degradation of Organic Pollutants. Nanomaterials, 11(4), 1052. https://doi.org/10.3390/nano11041052