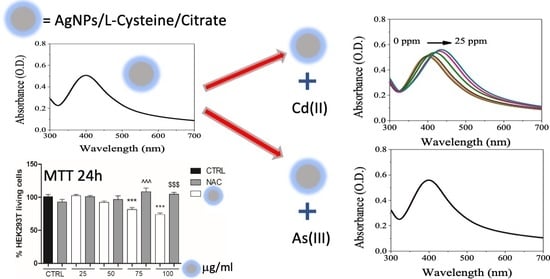
Biocompatible Silver Nanoparticles: Study of the Chemical and Molecular Structure, and the Ability to Interact with Cadmium and Arsenic in Water and Biological Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. AgNPs/L-cys/citr Synthesis and Preliminary Characterization
2.2. UV-Visible Sensing Tests
2.3. Advanced Characterization Methods
2.4. Biological Studies
2.4.1. Materials for Biological Characterization
2.4.2. Cell Cultures
2.4.3. MTT Assay
Gene Expression Analysis
Statistical Analysis
3. Results and Discussion
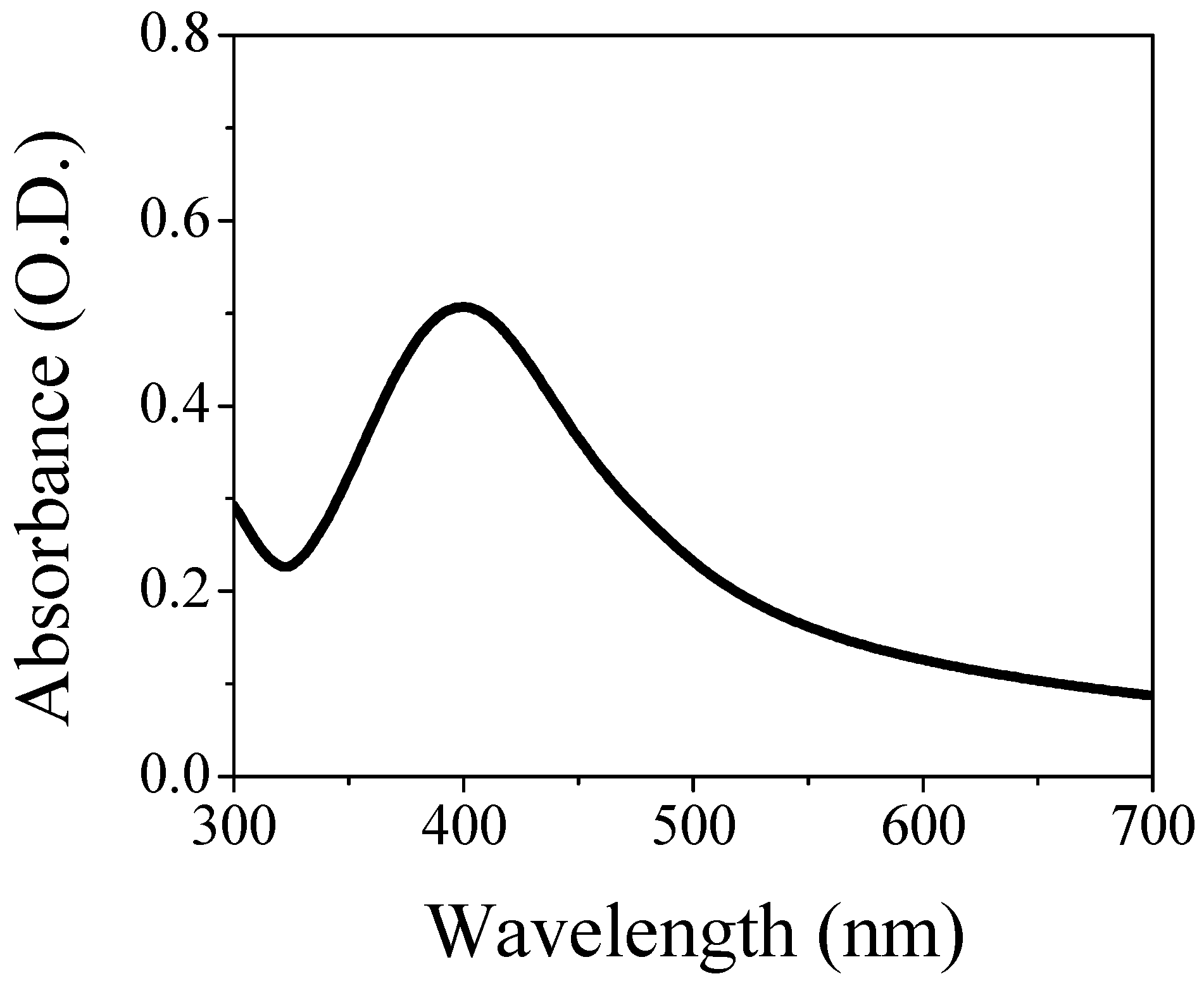
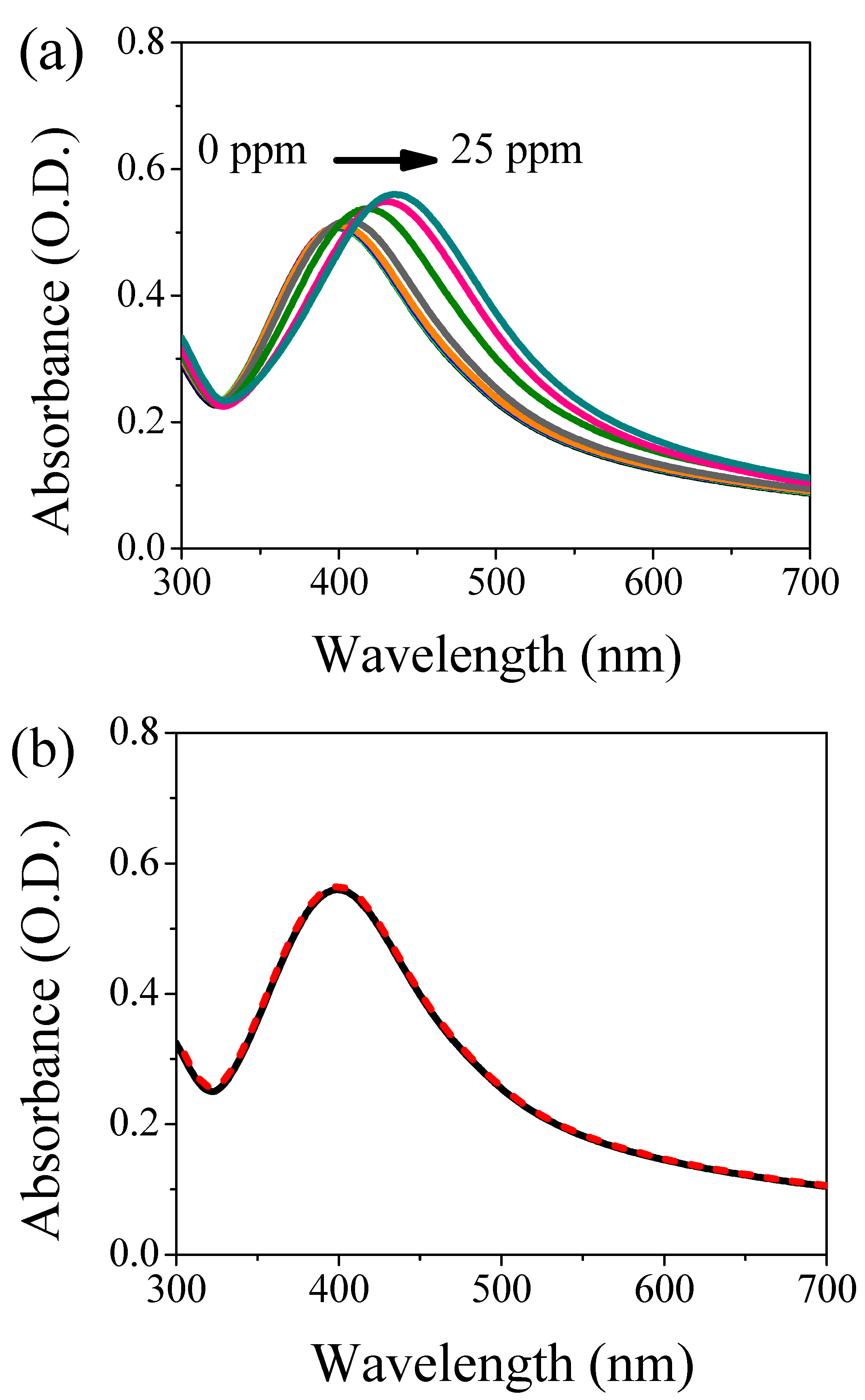
3.1. UV-Visible Assessment of AgNPs/L-cys/citr Responsivity and Selectivity for Heavy Metal Ions
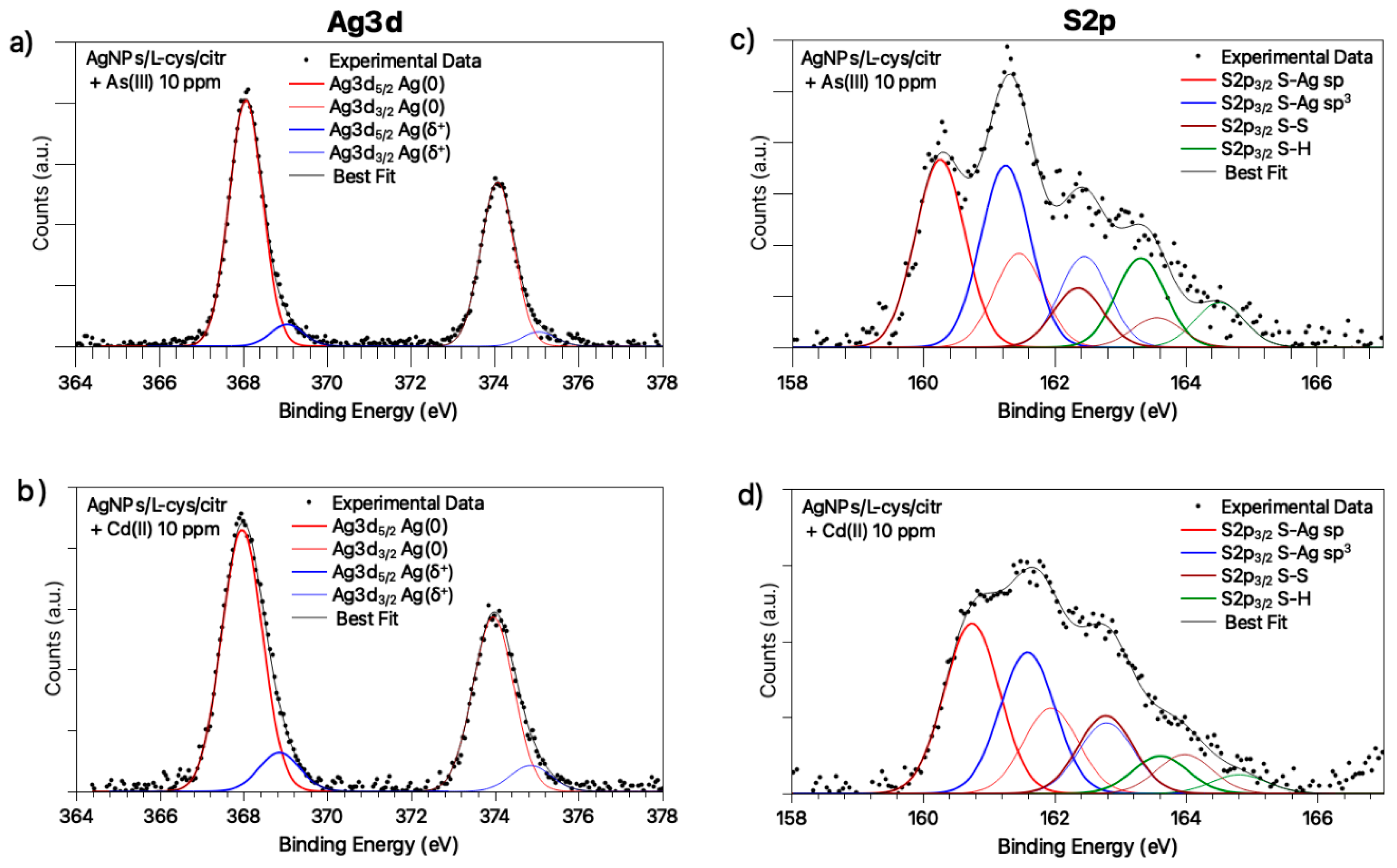
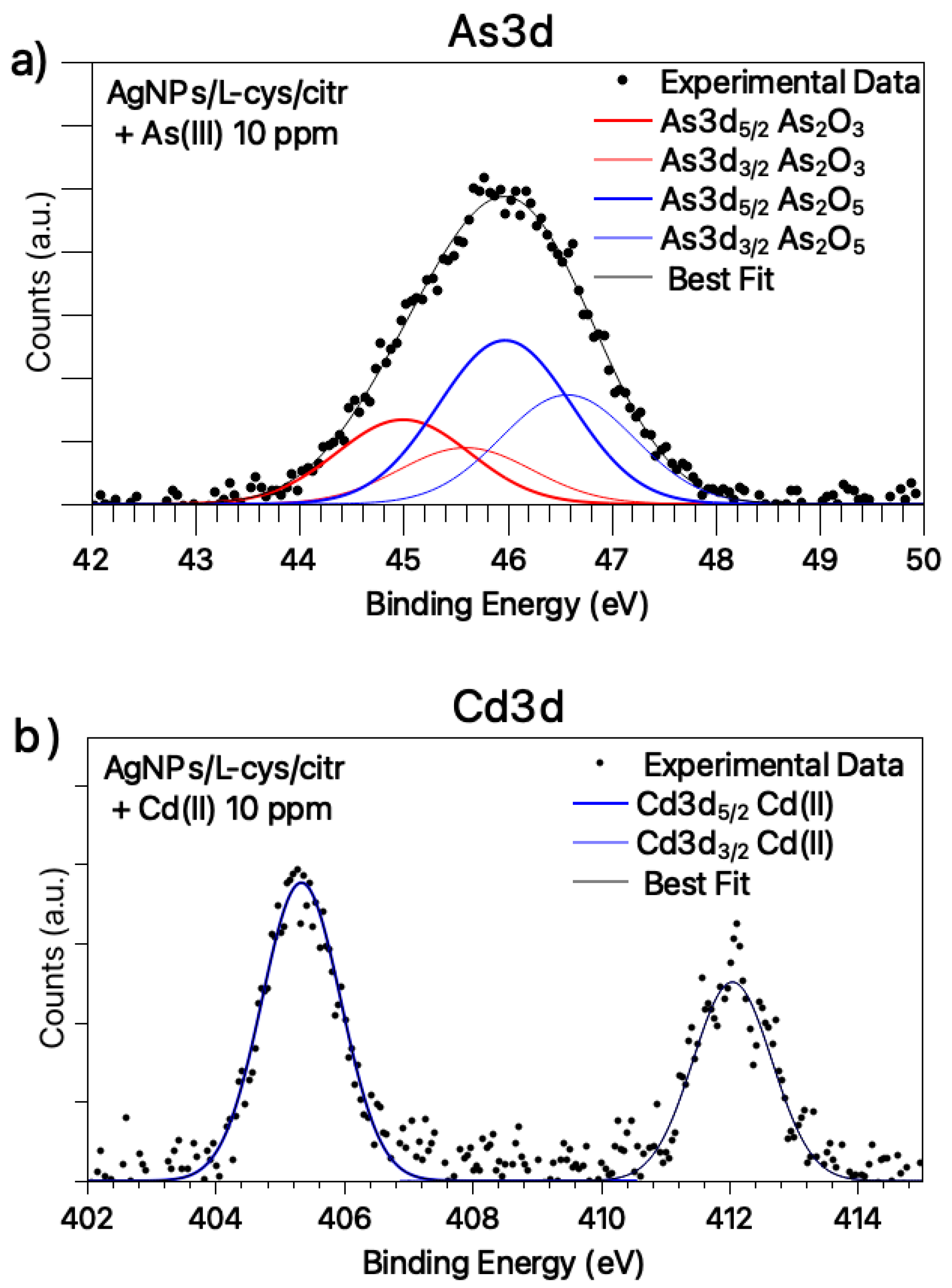
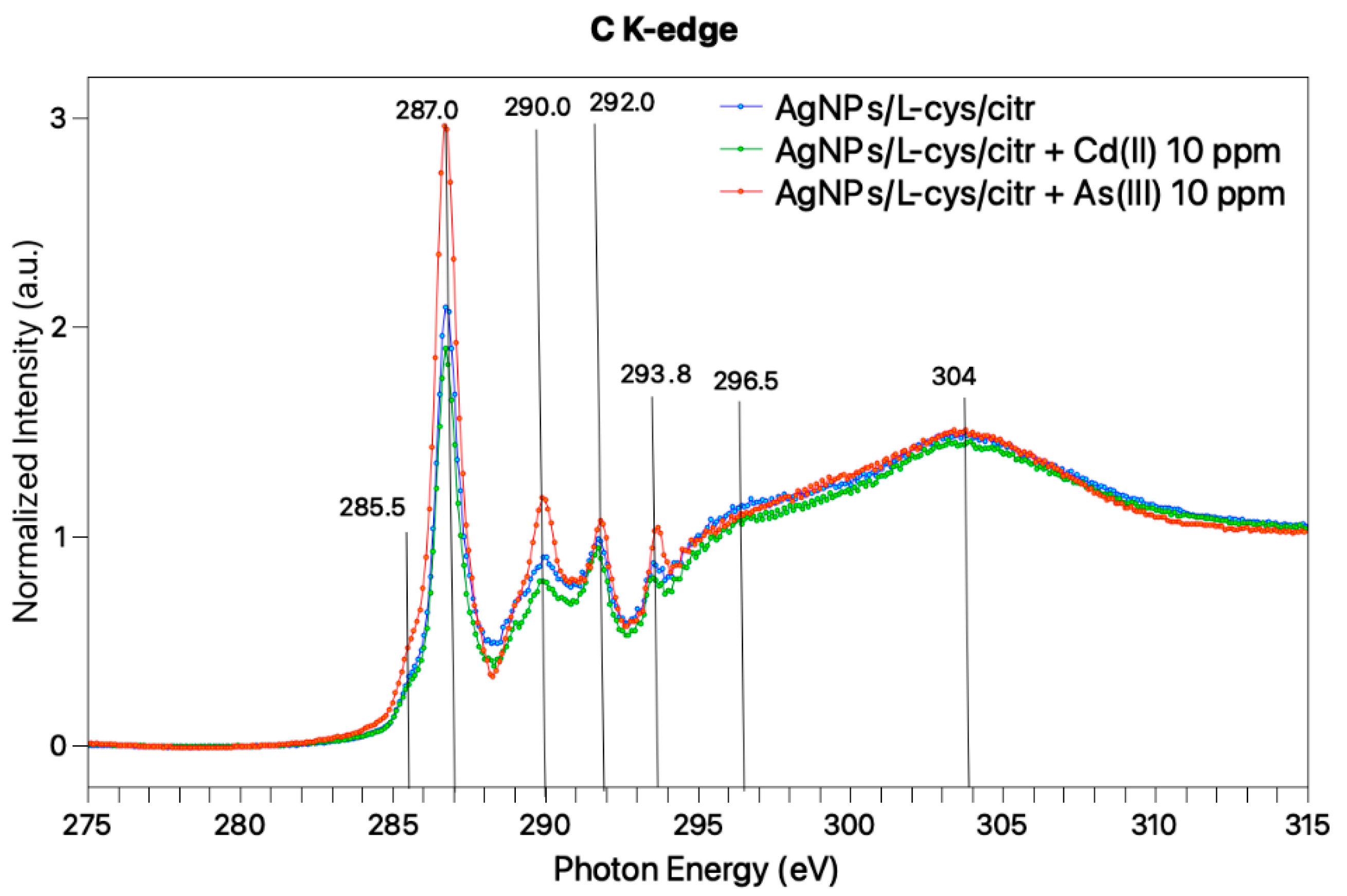
3.2. Molecular and Electronic Structure: SR-XPS, IRRAS and NEXAFS Studies on AgNPs/L-cys/citr-As(III) and Cd(II)
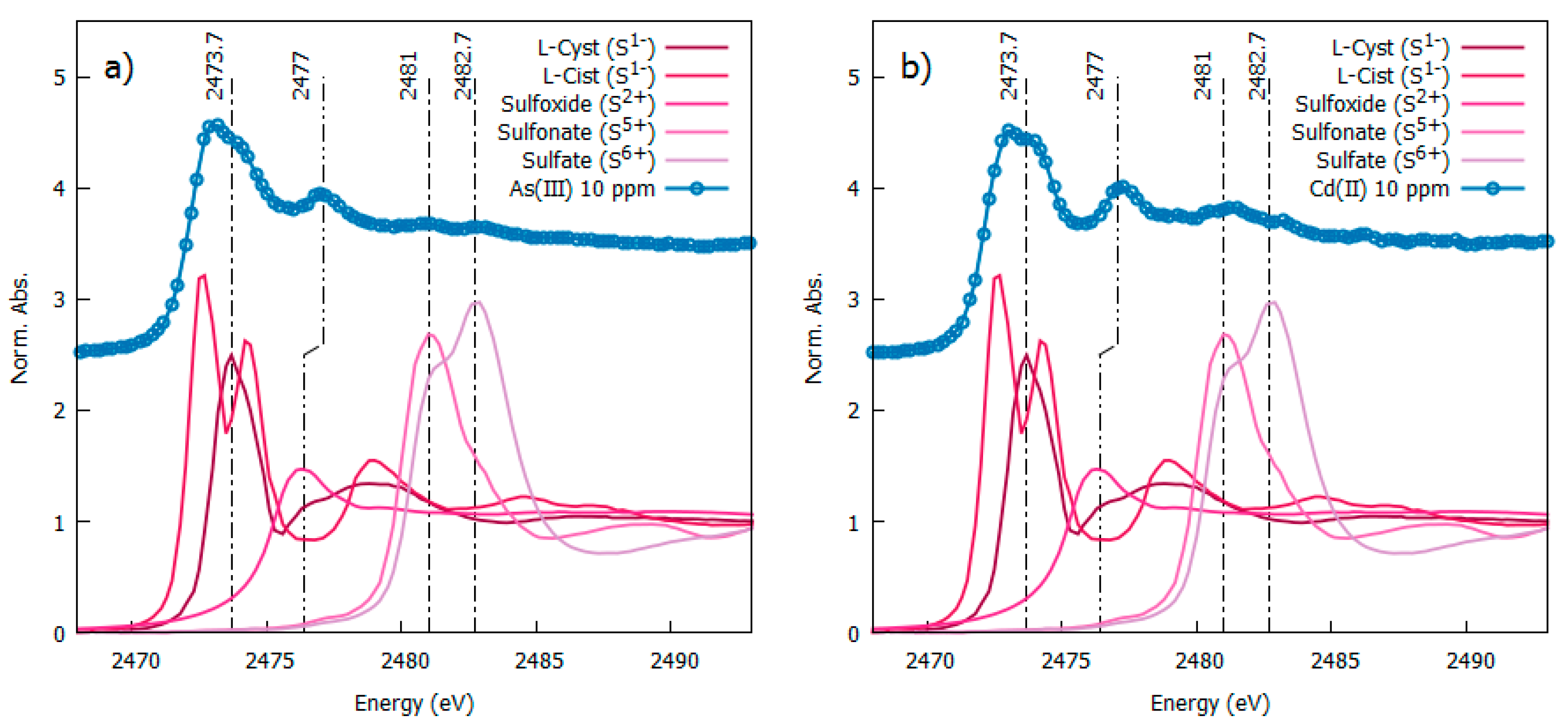
3.3. Local Structure around Silver Nanoparticles: XAS Measurements at the S-K Edge
3.4. Silver Nanoparticles Morphology Assessment: TEM
3.5. Biological Characterization
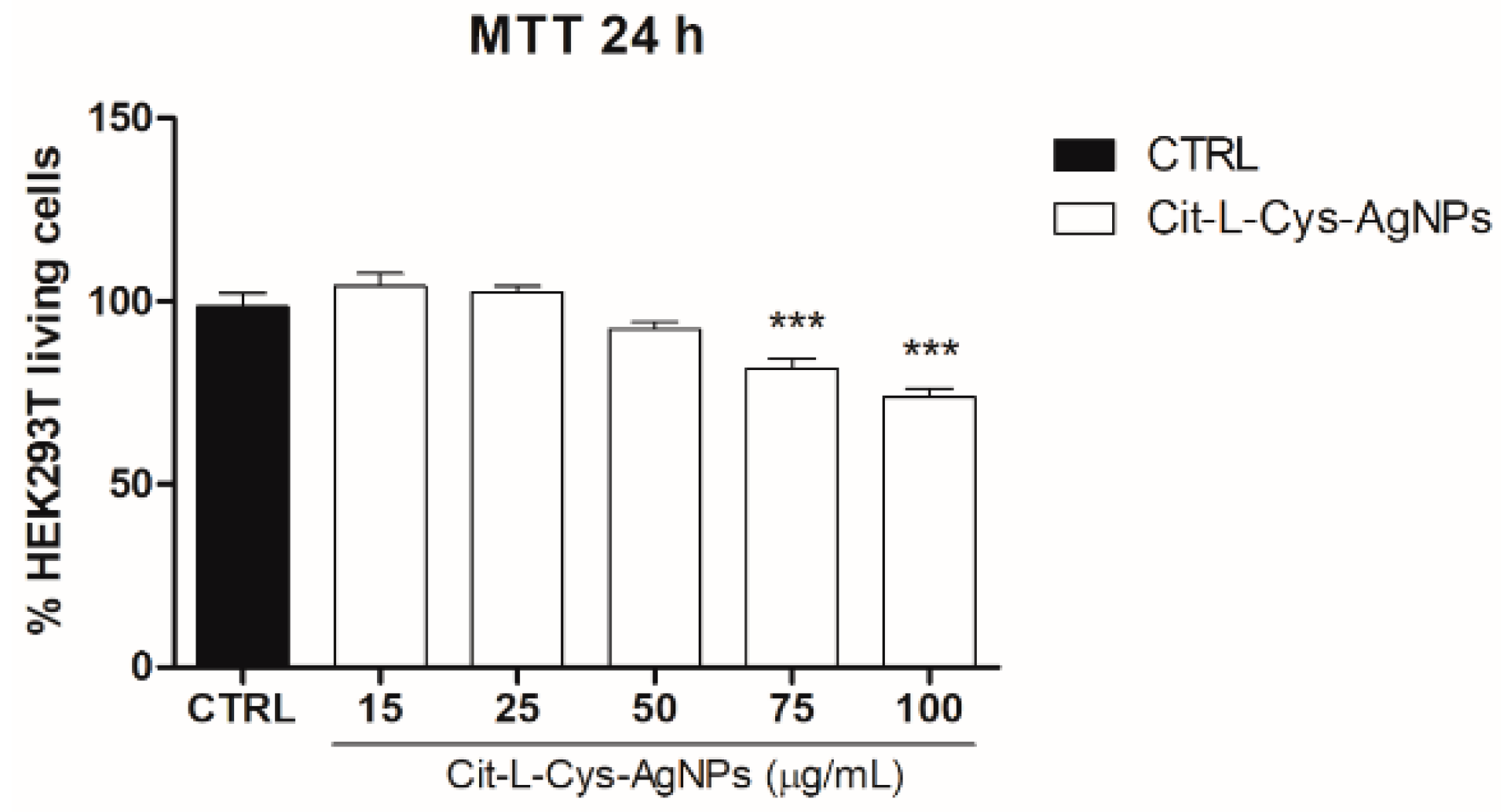
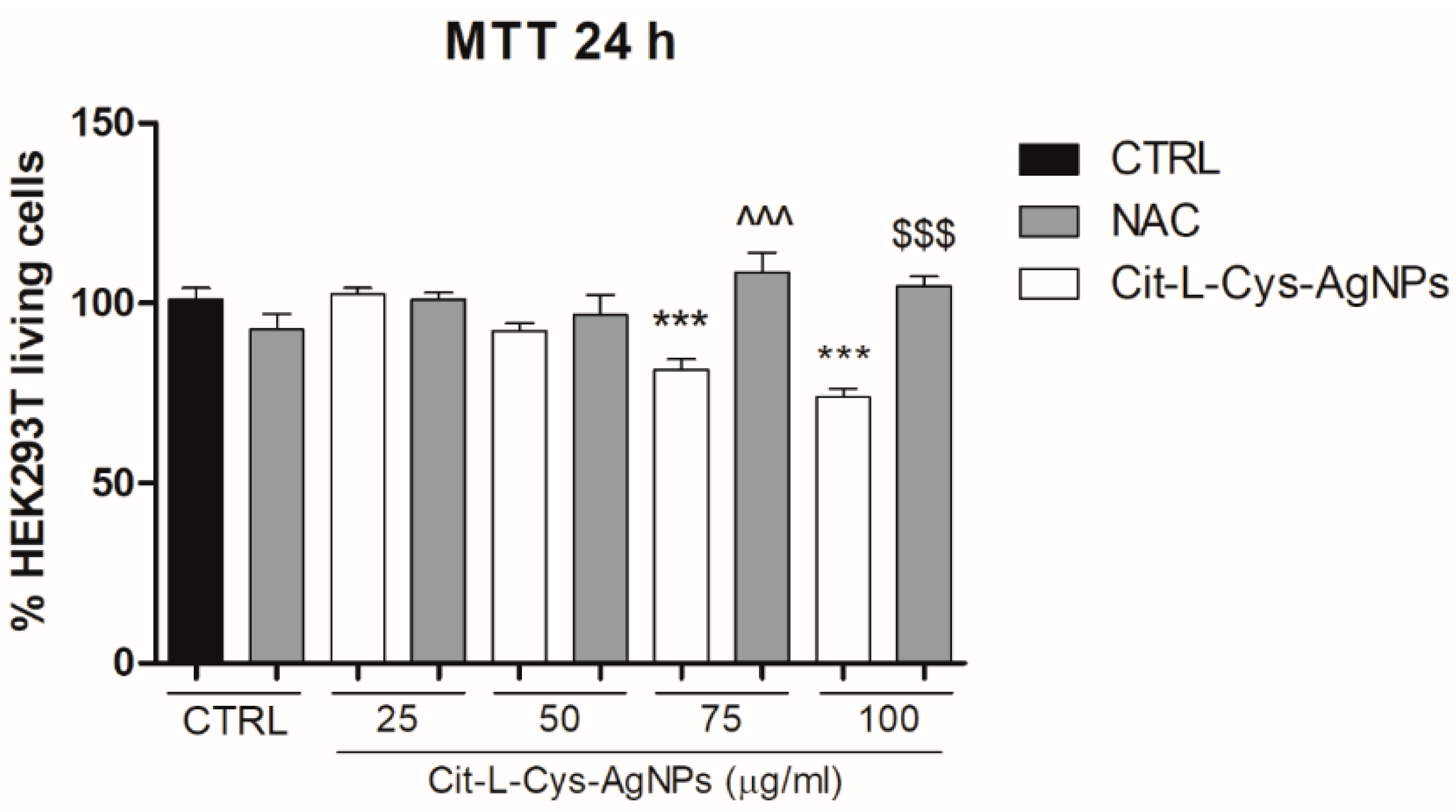
3.5.1. Effect of AgNPs/L-cys/Citron Cell Viability
3.5.2. Effect of the Antioxidant N-Acetyl-Cysteine on AgNPs/L-cys/citr-Induced Cytotoxicity
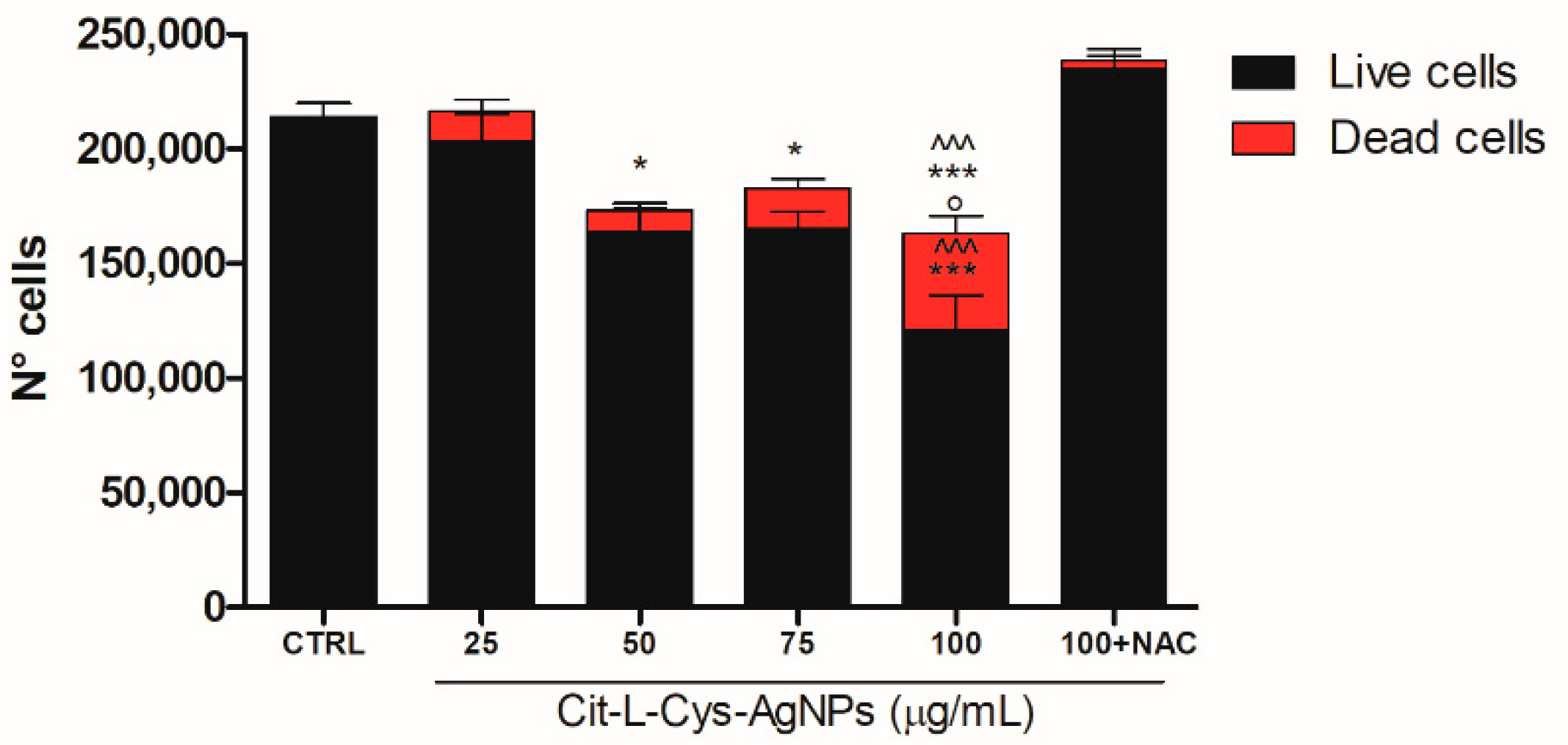
3.5.3. Quantification of Dead and Living Cells after AgNPs/L-cys/citr Treatment
3.5.4. Effect of AgNPs/L-cys/citr on the Cellular Antioxidant Response
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kirk, K.A.; Andreescu, S. Easy-to-Use Sensors for Field Monitoring of Copper Contamination in Water and Pesticide-Sprayed Plants. Anal. Chem. 2019, 91, 13892−13899. [Google Scholar] [CrossRef]
- Islam, M.S.; Ahmed, M.K.; Habibullah-Al-Mamun, M. Heavy Metals in Cereals and Pulses: Health Implications in Bangladesh. J. Agric. Food Chem. 2014, 62, 10828–10835. [Google Scholar] [CrossRef]
- Fu, J.; Zhang, A.; Wang, T.; Qu, G.; Shao, J.; Yuan, B.; Wang, Y.; Jiang, G. Influence of E-Waste Dismantling and Its Regulations: Temporal Trend, Spatial Distribution of Heavy Metals in Rice Grains, and Its Potential Health Risk. Environ. Sci. Technol. 2013, 47, 7437–7445. [Google Scholar] [CrossRef]
- Rahman, M.T.; Kabir, M.F.; Gurung, A.; Reza, K.M.; Pathak, R.; Ghimire, N.; Baride, A.; Wang, Z.; Kumar, M.; Qiao, Q. Graphene Oxide–Silver Nanowire Nanocomposites for Enhanced Sensing of Hg2+. ACS Appl. Nano Mater. 2019, 2, 4842–4851. [Google Scholar] [CrossRef]
- Fiorati, A.; Bellingeri, A.; Punta, C.; Corsi, I.; Venditti, I. Silver Nanoparticles for Water Pollution Monitoring and Treatments: Ecosafety Challenge and Cellulose-Based Hybrids Solution. Polymers 2020, 12, 1635. [Google Scholar] [CrossRef]
- Waheed, A.; Mansha, M.; Ullah, N. Nanomaterials-based electrochemical detection of heavy metals in water: Current status, challenges and future direction. Trends Anal. Chem. 2018, 105, 37–51. [Google Scholar] [CrossRef]
- Yang, Z.; Liu, H.; Li, J.; Yang, K.; Zhang, Z.; Chen, F.; Wang, B. High-Throughput Metal Trap: Sulfhydryl-Functionalized Wood Membrane Stacks for Rapid and Highly Efficient Heavy Metal Ion Removal. ACS Appl. Mater. Interfaces 2020, 12, 15002–15011. [Google Scholar] [CrossRef]
- Ciotta, E.; Paoloni, S.; Richetta, M.; Prosposito, P.; Tagliatesta, P.; Lorecchio, C.; Venditti, I.; Fratoddi, I.; Ciardi, S.; Pizzoferrato, R. Sensitivity to heavy-metal ions of open-cage fullerene quantum dots. Sensors 2017, 17, 2614. [Google Scholar] [CrossRef] [Green Version]
- Galashev, A.Y. Modeling of Forced Desorption Processes in a Regenerable Graphene Sorbent for Elemental Mercury Capture. J. Phys. Chem. C 2016, 120, 13263–13274. [Google Scholar] [CrossRef]
- Boruah, B.S.; Daimari, N.K.; Biswas, R. Functionalized silver nanoparticles as an effective medium towards trace, determination of arsenic (III) in aqueous solution. Results Phys. 2019, 12, 2061–2065. [Google Scholar] [CrossRef]
- Paul, D.; Dutta, S.; Saha, D.; Biswas, R. LSPR based Ultra-sensitive low cost U-bent optical fiber for volatile liquid sensing. Sens. Actuat. B Chem. 2017, 250, 198–207. [Google Scholar] [CrossRef]
- Ferdous, Z.; Nemmar, A. Review Health Impact of Silver Nanoparticles: A Review of the Biodistribution and Toxicity Following Various Routes of Exposure. Int. J. Mol. Sci. 2020, 21, 2375. [Google Scholar] [CrossRef] [Green Version]
- Venditti, I.; Testa, G.; Sciubba, F.; Carlini, L.; Secchi, V.; Krause, S.; Meneghini, C.; Mobilio, S.; Battocchio, C.; Fratoddi, I. Hydrophilic Metal Nanoparticles Functionalized by 2-Diethylaminoethanethiol: A Close Look at the Metal–Ligand Interaction and Interface Chemical Structure. J. Phys. Chem. C 2017, 121, 8002–8013. [Google Scholar] [CrossRef]
- Prosposito, P.; Burratti, L.; Bellingeri, A.; Protano, G.; Faleri, C.; Corsi, I.; Battocchio, C.; Iucci, G.; Tortora, L.; Secchi, V.; et al. Bifunctionalized Silver Nanoparticles as Hg2+ Plasmonic Sensor in Water: Synthesis, Characterizations, and Ecosafety. Nanomaterials 2019, 9, 1353. [Google Scholar] [CrossRef] [Green Version]
- Fratoddi, I.; Battocchio, C.; Cartoni, A.; Iucci, G.; Paladini, A.; Catone, D.; O’Keeffe, P.; Nappini, S.; Cerra, S.; Venditti, I. Silver nanoparticles functionalized by Rhodamine B isothiocyanate or Fluorescein isothiocyanate: Synthesis and studies on fluorescent and plasmonic properties. Appl. Sci. 2021, 11, 2472. [Google Scholar] [CrossRef]
- Mochi, F.; Burratti, L.; Fratoddi, I.; Venditti, I.; Battocchio, C.; Carlini, L.; Iucci, G.; Casalboni, M.; De Matteis, F.; Casciardi, S.; et al. Interaction of colloidal silver nanoparticles with Co2+ and Ni2+ in water for sensing application. Nanomaterials 2018, 8, 488. [Google Scholar] [CrossRef] [Green Version]
- Moulder, J.F.; Stickle, W.F.; Sobol, P.E.; Bomben, K.D. Handbook of X-ray Photoelectron Spectroscopy; Physical Electronics Inc.: Eden Prairie, MN, USA, 1996. [Google Scholar]
- Shirley, D.A. High-resolution X-ray photoemission spectrum of the valence bands of gold. Phys. Rev. B 1972, 12, 4709–4714. [Google Scholar] [CrossRef] [Green Version]
- Beamson, G.; Briggs, D. High Resolution XPS of Organic Polymers, The Scienta ESCA 300 Database; John Wiley & Sons: Chichester, UK, 1992. [Google Scholar]
- Di Cicco, A.; Aquilanti, G.; Minicucci, M.; Principi, E.; Novello, N.; Cognigni, A.; Olivi, L. Novel XAFS capabilities at ELETTRA synchrotron light source. J. Phys. Conf. Ser. 2009, 190, 012043. [Google Scholar] [CrossRef]
- Meneghini, C.; Mobilio, S.; Bardelli, F.; Prestipino, C. International Tables for Crystallography, Vol. I X-ray Absorption Spectroscopy and Related Techniques. Chantler, C.T., Boscherini, F., Bunker, B., Eds.; Wiley Online Library. 2020. Available online: https://onlinelibrary.wiley.com/ (accessed on 22 September 2021).
- Benfatto, M.; Meneghini, C. Synchrotron Radiation: Basics, Methods and Applications; Mobilio, S., Boscherini, F., Meneghini, C., Eds.; Springer: Berlin/Heidelberg, Germany, 2015. [Google Scholar]
- Available online: https://www.esrf.fr/home/UsersAndScience/Experiments/XNP/ID21/php/Database-SCompounds.html (accessed on 10 July 2021).
- Strober, W. Trypan Blue Exclusion Test of Cell Viability. Curr. Protoc. Immunol. 2015, 111, A3.B.1–A3.B.31. [Google Scholar] [CrossRef]
- Shaghayegh, J.; Zanjanchi, M.A. Colorimetric Detection of Cadmium Ions Using Modified Silver Nanoparticles. Appl. Phys. A 2019, 125, 1–10. [Google Scholar]
- Du, J.; Hu, X.; Zhang, G.; Wu, X.; Gong, D. Colorimetric Detection of Cadmium in Water Using L-cysteine Functionalized Gold–Silver anoparticles. Anal. Lett. 2018, 51, 2906–2919. [Google Scholar] [CrossRef]
- Aravind, A.; Maria, S.; Beena, M. Green Silver Nanoparticles as a Multifunctional Sensor for Toxic Cd (ii) Ions. New J. Chem. 2018, 42, 15022–15031. [Google Scholar] [CrossRef]
- Schiesaro, I.; Burratti, L.; Meneghini, C.; Fratoddi, I.; Prosposito, P.; Lim, J.; Scheu, C.; Venditti, I.; Iucci, G.; Battocchio, C. Hydrophilic Silver Nanoparticles for Hg(II) Detection in Water: Direct Evidence for Mercury–Silver Interaction. J. Phys. Chem. C 2020, 124, 25975–25983. [Google Scholar] [CrossRef]
- Porcaro, F.; Battocchio, C.; Antoccia, A.; Fratoddi, I.; Venditti, I.; Fracassi, A.; Luisetto, I.; Russo, M.V.; Polzonetti, G. Synthesis of functionalized gold nanoparticles capped with 3-mercapto-1-propansulfonate and 1-thioglucose mixed thiols and “in vitro” bioresponse. Colloids Surf. B Biointerfaces 2016, 142, 408–416. [Google Scholar] [CrossRef]
- Carlini, L.; Fasolato, C.; Postorino, P.; Fratoddi, I.; Venditti, I.; Testa, G.; Battocchio, C. Comparison between Silver and Gold Nanoparticles Stabilized with Negatively Charged Hydrophilic Thiols: SR-XPS and SERS as Probes for Structural Differences and Similarities. Colloids Surf. A Physicochem. Eng. 2017, 532, 0927–7757. [Google Scholar] [CrossRef]
- NIST X-ray Photoelectron Spectroscopy Database, Version 4.1; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2012. Available online: http://srdata.nist.gov/xps/ (accessed on 5 July 2021).
- Yoshida, T.; Yamasaki, K.; Sawada, S. An X-ray Photoelectron Spectroscopic Study of Biuret Metal Complexes. Bull. Chem. Soc. Jpn. 1978, 51, 1561–1562. [Google Scholar] [CrossRef] [Green Version]
- Blackburn, J.R.; Nordberg, C.R.; Stevie, F.; Albridge, R.G.; Jones, M.M. Photoelectron Spectroscopy of Coordination Compounds. Triphenylphosphine and Its Complexes. Inorg. Chem. 1970, 9, 2374–2376. [Google Scholar] [CrossRef]
- Lindau, I.; Pianetta, P.; Garner, C.M.; Chye, P.E.; Gregory, P.E.; Spicer, W.E. Photoemission studies of the electronic structure of III–V semiconductor surfaces. Surf. Sci. 1977, 63, 45–55. [Google Scholar] [CrossRef]
- Flinn, B.J.; McIntyre, N.S. Studies of the UV/Ozone oxidation of GaAs using angle-resolved X-ray photoelectron spectroscopy. Surf. Interface Anal. 1990, 15, 19–26. [Google Scholar] [CrossRef]
- Hall, G.E.M.; Pelchat, J.C.; Gauthier, G. Stability of inorganic arsenic(III) and arsenic(V) in water samples. J. Anal. At. Spectrom. 1999, 14, 205–213. [Google Scholar] [CrossRef]
- NIST Chemistry WebBook, SRD 69; National Institute of Standards and Technology: Gaithersburg, MD, USA, 2018. Available online: https://webbook.nist.gov/chemistry/ (accessed on 12 July 2021).
- Begonja, S.S.; Rodenas, L.A.G.; Borghi, E.B.; Morando, P.J. Adsorption of cysteine on TiO2 at different pH values: Surface complexes characterization by FTIR-ATR and Langmuir isotherms analysis. Colloids Surf. A Physicochem. Eng. Asp. 2012, 403, 114–120. [Google Scholar] [CrossRef]
- Zubavichus, Y.; Shaporenko, A.; Grunze, M.; Zharnikov, M. NEXAFS spectroscopy of biological molecules: From aminoacids to functional proteins. Nucl. Instrum. Methods Phys. Res. A 2009, 603, 111–114. [Google Scholar] [CrossRef]
- Stöhr, J. Nexafs Spectroscopy; Gomer, C., Ed.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Marchioni, M.; Battocchio, C.; Joly, Y.; Gateau, C.; Nappini, S.; Pis, I.; Delangle, P.; Michaud-Soret, I.; Deniaud, A.; Veronesi, G. Thiolate-Capped Silver Nanoparticles: Discerning Direct Grafting from Sulfidation at the Metal-Ligand Interface by Interrogating the Sulfur Atom. J. Phys. Chem. C 2020, 124, 13467–13478. [Google Scholar] [CrossRef]
- Battocchio, C.; Meneghini, C.; Fratoddi, I.; Venditti, I.; Russo, M.V.; Aquilanti, G.; Maurizio, C.; Bondino, F.; Matassa, R.; Rossi, M.; et al. Silver Nanoparticles Stabilized with Thiols: A Close Look at the Local Chemistry and Chemical Structure. J. Phys. Chem. C 2012, 116, 19571–19578. [Google Scholar] [CrossRef]



| Gene | Accession Number | PCR Product Size | Nucleotide Sequence |
|---|---|---|---|
| CAT | NM_001752 | 139 bp | Forward 5′-TCA GGT TTC TTT CTT GTT CAG-3′ Reverse 5′-CTG GTC AGT CTT ATA ATG GAA TT-3′ |
| SOD2 | NM_000636.4 | 199 bp | Forward 5′-AAT GGT GGT GGT CAT ATC A-3′ Reverse 5′-CCC GTT CCT TAT TGA AAC C-3′ |
| System Xc− | NM_014331.4 | 107 bp | Forward 5′-GGT GGT GTG TTT GCT GTC-3′ Reverse 5′-GCT GGT AGA GGA GTG TGC-3′ |
| GCLC | AB262176.1 | 125 bp | Forward 5′-TTG CAA AGG TGG CAA TGC-3′ Reverse 5′-GAA ACA CAC CTT CCT TCC-3′ |
| GAPDH | NM_002046.7 | 110 bp | Forward 5′- TTG TTG CCA TCA ATG ACC C -3′ Reverse 5′- CTT CCC GTT CTC AGC CTT G -3′ |
| Sample | Signal | BE (eV) | FWHM (eV) | Atomic % | Assignment |
|---|---|---|---|---|---|
| AgNPs/L-cys/citr + As (10 ppm) | Ag3d5/2 | 368.09 368.51 | 0.95 0.95 | 91.8% 8.2% | Ag (0) Ag+ |
| S2p3/2 | 160.25 161.25 162.36 163.31 | 0.89 0.89 0.89 0.89 | 36.2% 35.0% 11.5% 17.3% | RS-Ag (sp) RS-Ag (sp3) RS-SR RS-H physisorbed | |
| AgNPs/L-cys/citr + Cd (10 ppm) | Ag3d5/2 Ag3d5/2 | 367.96 368.85 | 1.14 1.14 | 87.0% 13.0% | Ag (0) Ag+ |
| S2p3/2 | 160.74 161.59 162.78 163.61 | 0.98 0.98 0.98 0.98 | 39.9% 33.1% 18.2% 8.8% | RS-Ag (sp) RS-Ag (sp3) RS-SR RS-H physisorbed |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertelà, F.; Marsotto, M.; Meneghini, C.; Burratti, L.; Maraloiu, V.-A.; Iucci, G.; Venditti, I.; Prosposito, P.; D’Ezio, V.; Persichini, T.; et al. Biocompatible Silver Nanoparticles: Study of the Chemical and Molecular Structure, and the Ability to Interact with Cadmium and Arsenic in Water and Biological Properties. Nanomaterials 2021, 11, 2540. https://doi.org/10.3390/nano11102540
Bertelà F, Marsotto M, Meneghini C, Burratti L, Maraloiu V-A, Iucci G, Venditti I, Prosposito P, D’Ezio V, Persichini T, et al. Biocompatible Silver Nanoparticles: Study of the Chemical and Molecular Structure, and the Ability to Interact with Cadmium and Arsenic in Water and Biological Properties. Nanomaterials. 2021; 11(10):2540. https://doi.org/10.3390/nano11102540
Chicago/Turabian StyleBertelà, Federica, Martina Marsotto, Cecilia Meneghini, Luca Burratti, Valentin-Adrian Maraloiu, Giovanna Iucci, Iole Venditti, Paolo Prosposito, Veronica D’Ezio, Tiziana Persichini, and et al. 2021. "Biocompatible Silver Nanoparticles: Study of the Chemical and Molecular Structure, and the Ability to Interact with Cadmium and Arsenic in Water and Biological Properties" Nanomaterials 11, no. 10: 2540. https://doi.org/10.3390/nano11102540
APA StyleBertelà, F., Marsotto, M., Meneghini, C., Burratti, L., Maraloiu, V.-A., Iucci, G., Venditti, I., Prosposito, P., D’Ezio, V., Persichini, T., & Battocchio, C. (2021). Biocompatible Silver Nanoparticles: Study of the Chemical and Molecular Structure, and the Ability to Interact with Cadmium and Arsenic in Water and Biological Properties. Nanomaterials, 11(10), 2540. https://doi.org/10.3390/nano11102540