One-Pot Synthesis of Thiol-Modified Liquid Crystals Conjugated Fluorescent Gold Nanoclusters
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
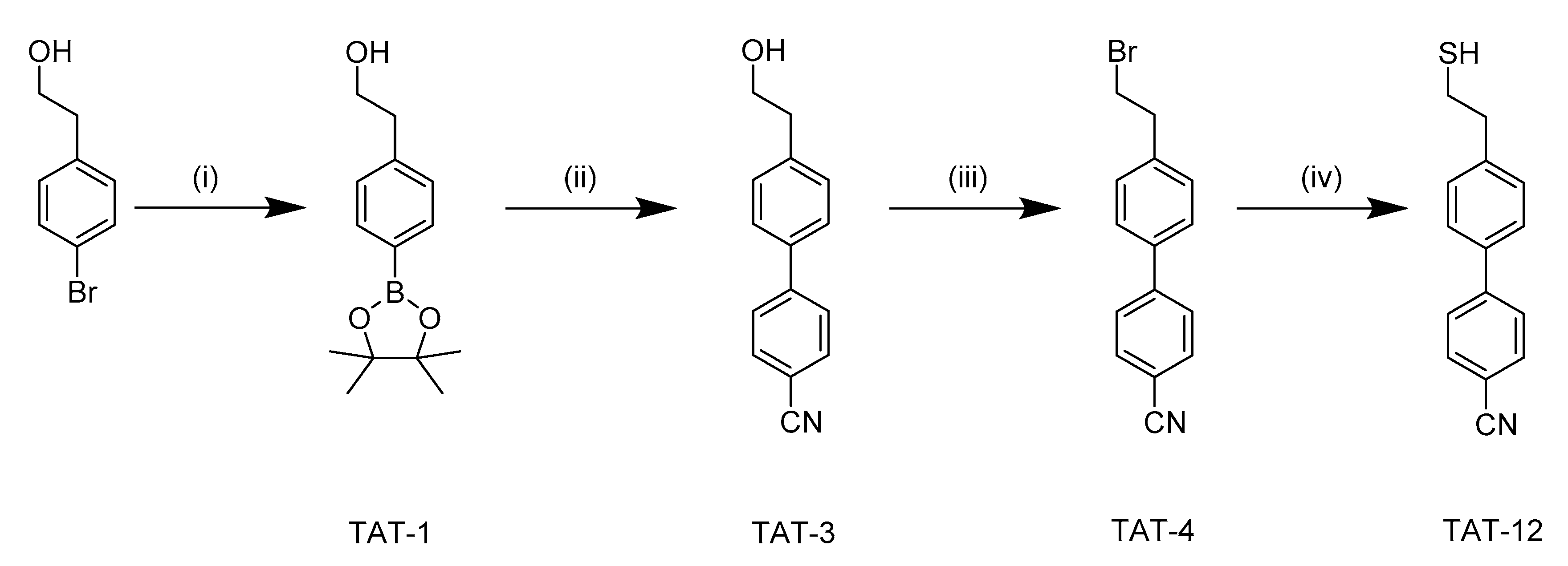
2.2. Synthesis of 4-(4,4,5,5-Tetramethyl-1,3,2-Dioxaborolan-2-yl) Benzeneethanol (TAT-1)
2.3. Synthesis of 4′-(2-Hydroxyethyl)-(1,1′-Biphenyl)-4-Carbonitrile (TAT-3)
2.4. Synthesis of 4′-(2-Bromoethyl)-(1,1′-Biphenyl)-4-Carbonitrile (TAT-4)
2.5. Synthesis of 4′-(2-Mercaptoethyl)-(1,1′-Biphenyl)-4-Carbonitrile (TAT-12)
2.6. Synthesis of Gold Nanoclusters Conjugated with TAT-12
3. Results and Discussion
3.1. Synthetic Pathway of 4′-(2-Mercaptoethyl)-(1,1′-Biphenyl)-4-Carbonitrile (TAT-12)
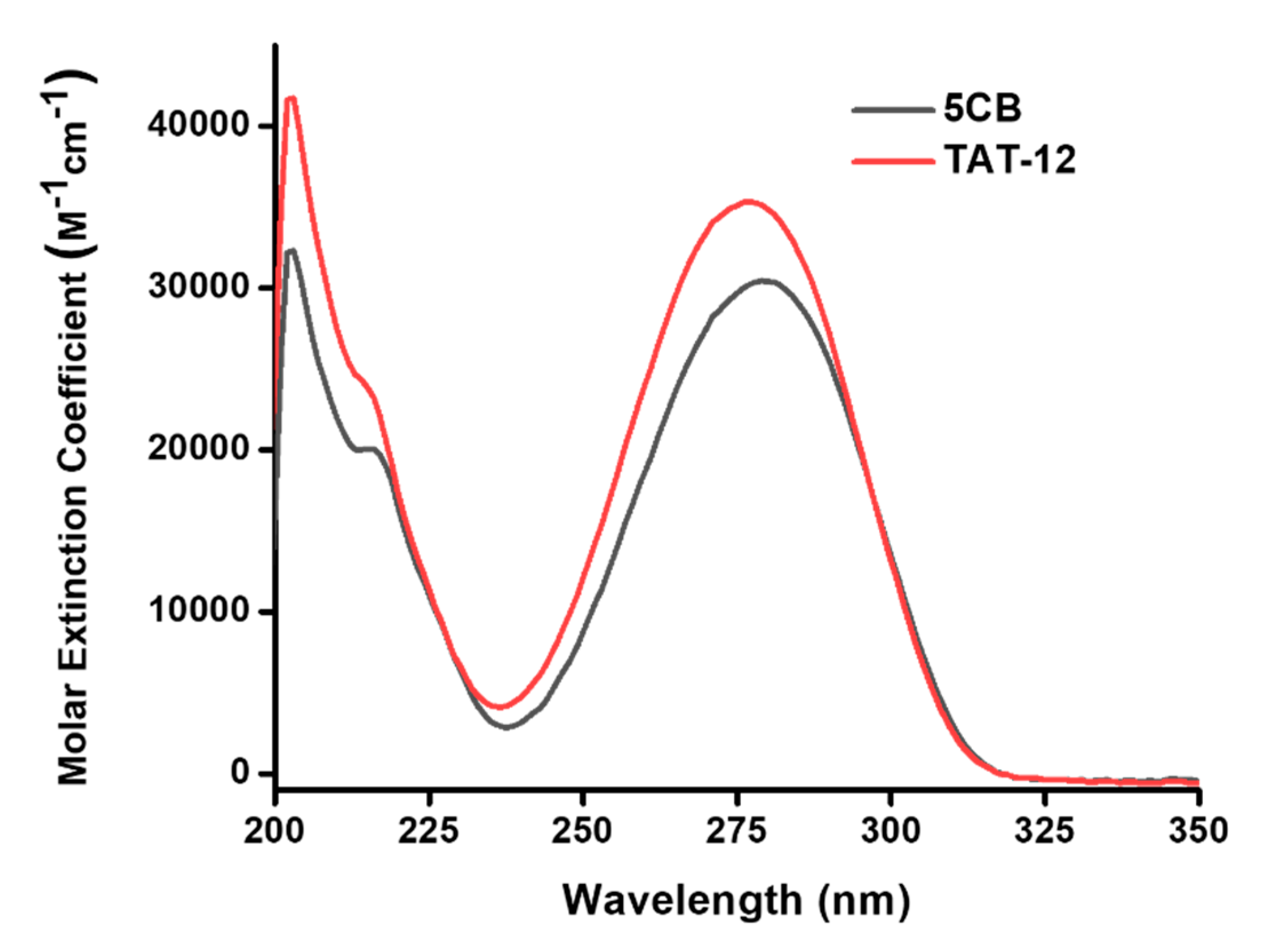
3.2. Characterizations of TAT-12
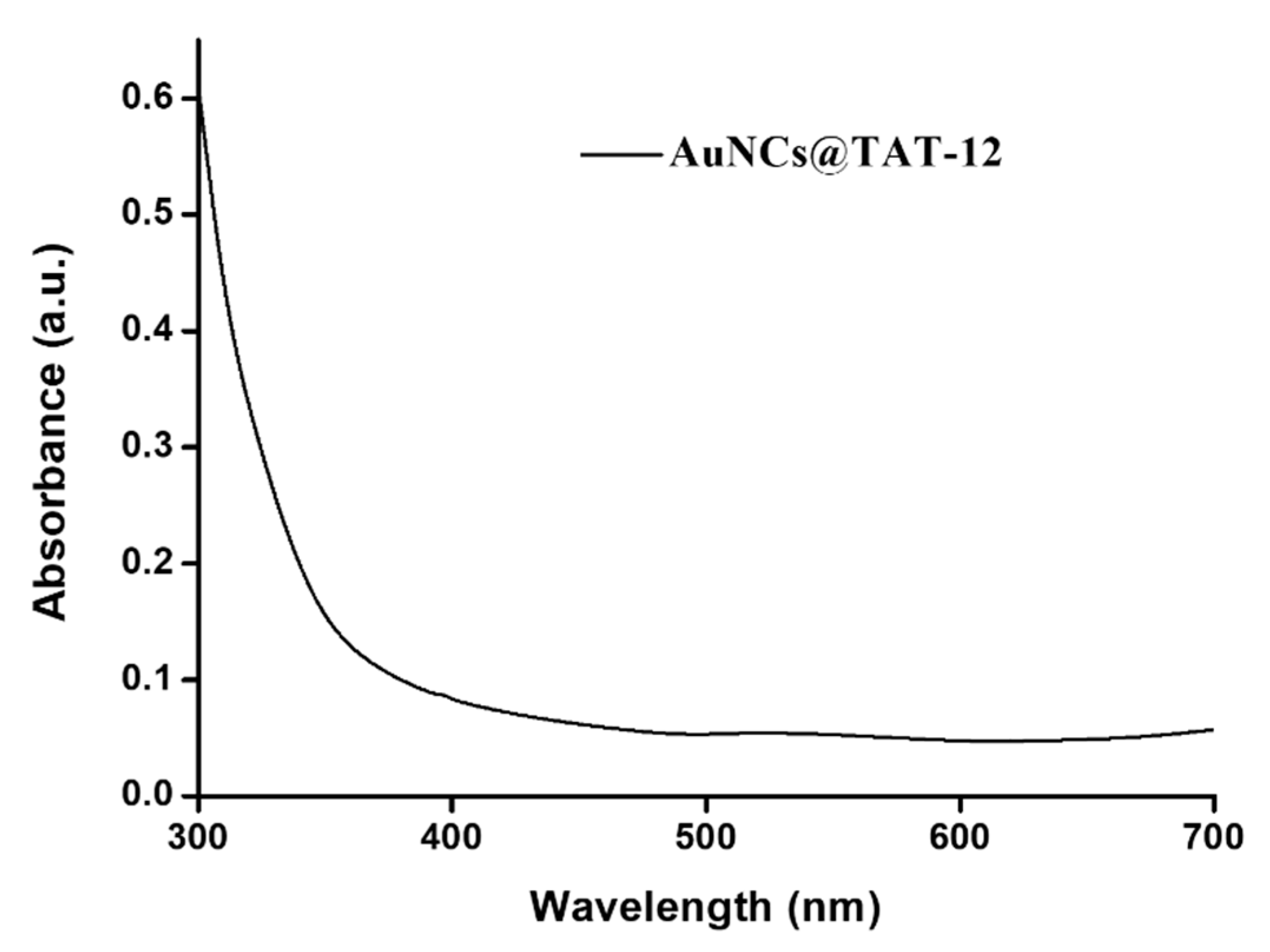
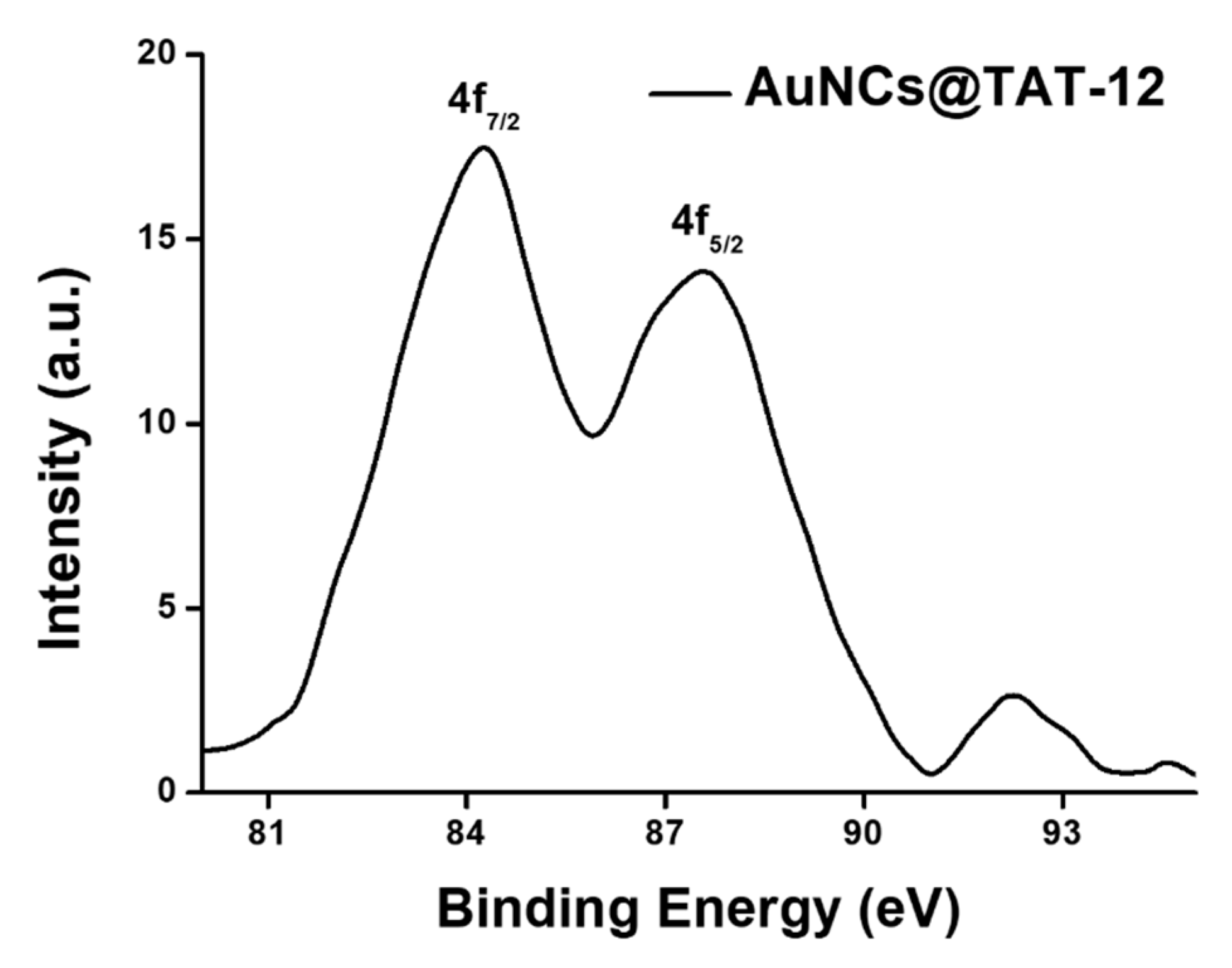
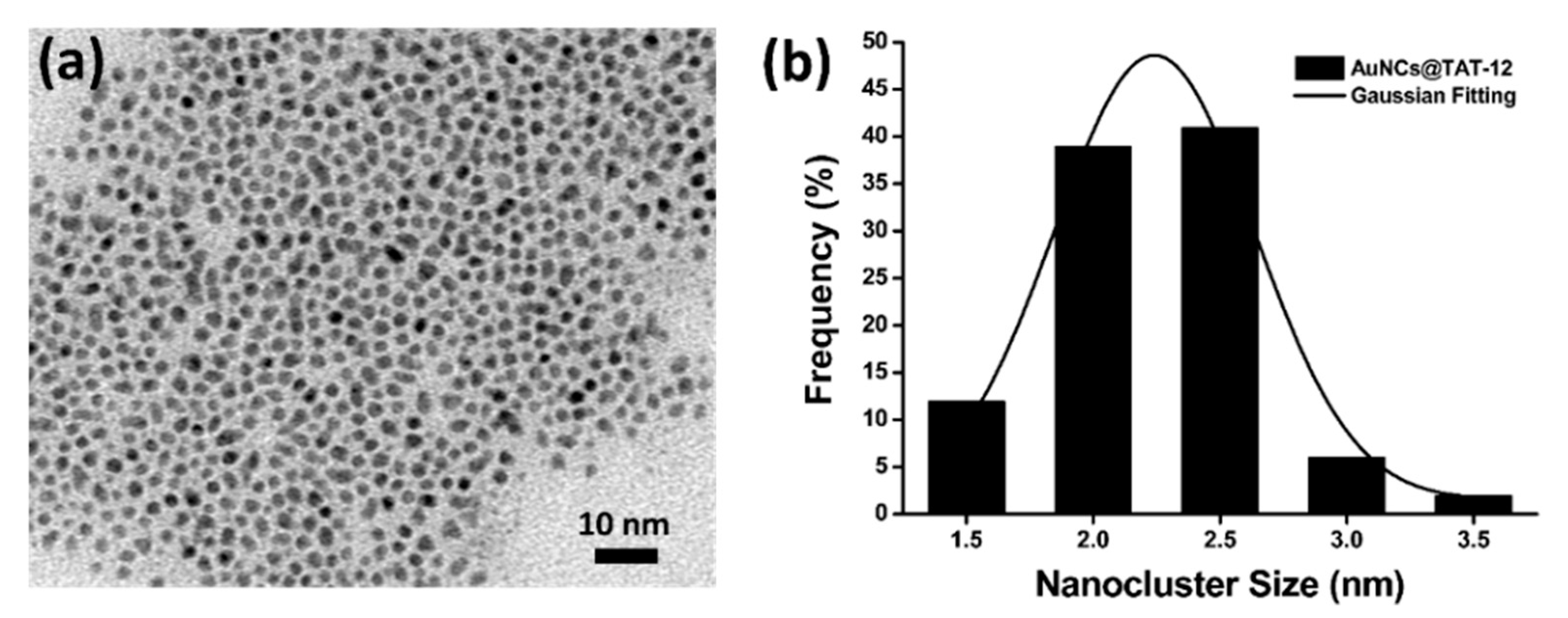
3.3. Optical and Structural Properties of AuNCs@TAT-12
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ray, P.C.; Khan, S.A.; Singh, A.K.; Senapati, D.; Fan, Z. Nanomaterials for targeted detection and photothermal killing of bacteria. Chem. Soc. Rev. 2012, 41, 3193–3209. [Google Scholar] [CrossRef]
- García, I.; Marradi, M.; Penadés, S. Glyconanoparticles: Multifunctional nanomaterials for biomedical applications. Nanomedicine 2010, 5, 777–792. [Google Scholar] [CrossRef]
- Cheng, L.; Wang, C.; Feng, L.; Yang, K.; Liu, Z. Functional nanomaterials for phototherapies of cancer. Chem. Rev. 2014, 114, 10869–10939. [Google Scholar] [CrossRef]
- Yang, X.; Yang, M.; Pang, B.; Vara, M.; Xia, Y. Gold nanomaterials at work in biomedicine. Chem. Rev. 2015, 115, 10410–10488. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Aditya, R.N.; Singh, A.; Kuo, T.-R. Biomedical applications for gold nanoclusters: Recent developments and future perspectives. Nanoscale Res. Lett. 2018, 13, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.-H.; Choi, E.; Ellis, E.; Lee, T.-C. Recent advances in gold nanoparticles for biomedical applications: From hybrid structures to multi-functionality. J. Mater. Chem. B 2019, 7, 3480–3496. [Google Scholar] [CrossRef]
- Zheng, K.; Setyawati, M.I.; Leong, D.T.; Xie, J. Antimicrobial gold nanoclusters. ACS Nano 2017, 11, 6904–6910. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Cao, Y.; Chen, T.; Zang, S.; Xie, J. Ligand-protected atomically precise gold nanoclusters as model catalysts for oxidation reactions. Chem. Commun. 2020, 56, 1163–1174. [Google Scholar] [CrossRef]
- Li, H.; Zhu, W.; Wan, A.; Liu, L. The mechanism and application of the protein-stabilized gold nanocluster sensing system. Analyst 2017, 142, 567–581. [Google Scholar] [CrossRef]
- Shang, L.; Dong, S.; Nienhaus, G.U. Ultra-small fluorescent metal nanoclusters: Synthesis and biological applications. Nano Today 2011, 6, 401–418. [Google Scholar] [CrossRef]
- Zhao, Y.; Tian, Y.; Cui, Y.; Liu, W.; Ma, W.; Jiang, X. Small molecule-capped gold nanoparticles as potent antibacterial agents that target gram-negative bacteria. J. Am. Chem. Soc. 2010, 132, 12349–12356. [Google Scholar] [CrossRef] [PubMed]
- Yougbare, S.; Chang, T.-K.; Tan, S.-H.; Kuo, J.-C.; Hsu, P.-H.; Su, C.-Y.; Kuo, T.-R. Antimicrobial gold nanoclusters: Recent developments and future perspectives. Int. J. Mol. Sci. 2019, 20, 2924. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.M.; Chu, H.L.; Lee, Y.C.; Wang, D.Y.; Chang, C.C.; Chung, K.L.; Yen, H.C.; Hsiao, C.W.; Pan, X.Y.; Kuo, T.R.; et al. Quantitative analysis of glucose metabolic cleavage in glucose transporters overexpressed cancer cells by target-specific fluorescent gold nanoclusters. Anal. Chem. 2018, 90, 3974–3980. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, K.; Higuchi, I.; Akagi, K. Synthesis of liquid crystalline binaphthyl derivatives. Mol. Cryst. Liq. Cryst. 2001, 364, 825–834. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, S.; Wu, X.; Xu, Y.; Li, H.; Liu, Y.; Tong, H.; Wang, L. Triazatruxene-based small molecules with thermally activated delayed fluorescence, aggregation-induced emission and mechanochromic luminescence properties for solution-processable nondoped oleds. J. Mater. Chem. C 2018, 6, 12503–12508. [Google Scholar] [CrossRef]
- Nayek, P.; Li, G. Superior electro-optic response in multiferroic bismuth ferrite nanoparticle doped nematic liquid crystal device. Sci. Rep. 2015, 5, 10845. [Google Scholar] [CrossRef]
- De Sio, L.; Lloyd, P.F.; Tabiryan, N.V.; Placido, T.; Comparelli, R.; Curri, M.L.; Bunning, T.J. Thermoplasmonic activated reverse-mode liquid crystal gratings. ACS Appl. Nano Mater. 2019, 2, 3315–3322. [Google Scholar] [CrossRef]
- Choudhary, A.; Singh, G.; Biradar, A.M. Advances in gold nanoparticle–liquid crystal composites. Nanoscale 2014, 6, 7743–7756. [Google Scholar] [CrossRef]
- Vardanyan, K.K.; Walton, R.D.; Sita, D.M.; Gurfinkiel, I.S.; Saidel, W.M. Study of pentyl-cyanobiphenyl nematic doped with gold nanoparticles. Liq. Cryst. 2012, 39, 595–605. [Google Scholar] [CrossRef]
- Vardanyan, K.K.; Sita, D.M.; Walton, R.D.; Saidel, W.M.; Jones, K.M. Cyanobiphenyl liquid crystal composites with gold nanoparticles. RSC Adv. 2013, 3, 259–273. [Google Scholar] [CrossRef]
- Hadjichristov, G.B.; Marinov, Y.G.; Petrov, A.G.; Bruno, E.; Marino, L.; Scaramuzza, N. Electro-optically responsive composites of gold nanospheres in 5cb liquid crystal under direct current and alternating current joint action. J. Appl. Phys. 2014, 115, 083107. [Google Scholar] [CrossRef]
- Khatua, S.; Manna, P.; Chang, W.-S.; Tcherniak, A.; Friedlander, E.; Zubarev, E.R.; Link, S. Plasmonic nanoparticles− liquid crystal composites. J. Phys. Chem. C 2010, 114, 7251–7257. [Google Scholar] [CrossRef]
- Liao, S.; Qiao, Y.; Han, W.; Xie, Z.; Wu, Z.; Shen, G.; Yu, R. Acetylcholinesterase liquid crystal biosensor based on modulated growth of gold nanoparticles for amplified detection of acetylcholine and inhibitor. Anal. Chem. 2012, 84, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Hartono, D.; Qin, W.J.; Yang, K.-L.; Yung, L.-Y.L. Imaging the disruption of phospholipid monolayer by protein-coated nanoparticles using ordering transitions of liquid crystals. Biomaterials 2009, 30, 843–849. [Google Scholar] [CrossRef]
- Carroll, V.; Michel, B.W.; Blecha, J.; VanBrocklin, H.; Keshari, K.; Wilson, D.; Chang, C.J. A boronate-caged [18f] flt probe for hydrogen peroxide detection using positron emission tomography. J. Am. Chem. Soc. 2014, 136, 14742–14745. [Google Scholar] [CrossRef]
- Muhammed, M.A.H.; Verma, P.K.; Pal, S.K.; Kumar, R.C.A.; Paul, S.; Omkumar, R.V.; Pradeep, T. Bright, nir-emitting au-23 from au-25: Characterization and applications including biolabeling. Chem. Eur. J. 2009, 15, 10110–10120. [Google Scholar] [CrossRef]
- Chang, T.K.; Cheng, T.M.; Chug, H.L.; Tan, S.H.; Kuo, J.C.; Hsu, P.H.; Su, C.Y.; Chen, H.M.; Lee, C.M.; Kuo, T.R. Metabolic mechanism investigation of antibacterial active cysteine-conjugated gold nanoclusters in escherichia coli. ACS Sustain. Chem. Eng. 2019, 7, 15479–15486. [Google Scholar] [CrossRef]
- Ishida, Y.; Akita, I.; Sumi, T.; Matsubara, M.; Yonezawa, T. Thiolate–protected gold nanoparticles via physical approach: Unusual structural and photophysical characteristics. Sci. Rep. 2016, 6, 29928. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hsu, P.-H.; Yougbaré, S.; Kuo, J.-C.; Krisnawati, D.I.; Jazidie, A.; Nuh, M.; Chou, P.-T.; Hsiao, Y.-C.; Kuo, T.-R. One-Pot Synthesis of Thiol-Modified Liquid Crystals Conjugated Fluorescent Gold Nanoclusters. Nanomaterials 2020, 10, 1755. https://doi.org/10.3390/nano10091755
Hsu P-H, Yougbaré S, Kuo J-C, Krisnawati DI, Jazidie A, Nuh M, Chou P-T, Hsiao Y-C, Kuo T-R. One-Pot Synthesis of Thiol-Modified Liquid Crystals Conjugated Fluorescent Gold Nanoclusters. Nanomaterials. 2020; 10(9):1755. https://doi.org/10.3390/nano10091755
Chicago/Turabian StyleHsu, Po-Hsuan, Sibidou Yougbaré, Jui-Chi Kuo, Dyah Ika Krisnawati, Achmad Jazidie, Mohammad Nuh, Po-Ting Chou, Yu-Cheng Hsiao, and Tsung-Rong Kuo. 2020. "One-Pot Synthesis of Thiol-Modified Liquid Crystals Conjugated Fluorescent Gold Nanoclusters" Nanomaterials 10, no. 9: 1755. https://doi.org/10.3390/nano10091755
APA StyleHsu, P.-H., Yougbaré, S., Kuo, J.-C., Krisnawati, D. I., Jazidie, A., Nuh, M., Chou, P.-T., Hsiao, Y.-C., & Kuo, T.-R. (2020). One-Pot Synthesis of Thiol-Modified Liquid Crystals Conjugated Fluorescent Gold Nanoclusters. Nanomaterials, 10(9), 1755. https://doi.org/10.3390/nano10091755





