Argovit™ Silver Nanoparticles Effects on Allium cepa: Plant Growth Promotion without Cyto Genotoxic Damage
Abstract
1. Introduction
| AgNP Source and Physicochemical Characteristics | Shape | Size (nm) | ζ a (mV) | Ag Content b | RP c (nm) | [C] d (µg/mL) | Exposure Time (h) | Cytotoxic and Genotoxic Damage | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Commercial Sigma-Aldrich, | <100 | - | - | 99.5% | - | 25, 50, 75 and 100 µg/mL | 4 h | CA and cell disintegration. | [29] |
| Commercial Sigma-Aldrich | <100 | - | - | 99.5% | - | 5, 10, 20, 40, 80 µg/mL | 2 h of exposure and recovery of 12, 24 and 48 h | 20 and 40 µg/mL. Dose-dependence increase in the frequency of cells with MN and CA ≥10 µg/mL: DNA damage (comet assay) | [37] |
| Synthesized with male inflorescence of screw pine, Pandanus odorifer | - | - | - | - | 5, 10, 20, 40 and 80 µg/mL | 2h of exposure and recovery of 12, 24 and 48 h | Dose-dependence increase in the frequency of cells with CA After 2 h of exposure and 48 h of recovery, no differences in cells with MN between control and lower concentrations (5 and 10 µg/mL). ≥20 µg/mL: DNA damage (comet assay) | [37] | |
| Commercial Sigma-Aldrich | - | TEM 70–130, av. ~125; SEM: 90–180, av. 120 | −4.86 | 99.5% | - | 25, 50 and 75 µg/mL | 24 h | No damage was observed in nuclei isolated from shoots. Nuclei isolated from roots exposed to 25 and 50 µg/mL shown DNA damage determined by comet assay. The major effect was observed with 50 µg/mL. No damage was observed with 75 µg/mL, and the authors suggest agglomeration and precipitation of AgNP. | [42] |
| Synthesis AgNP-citrate AgNP-PVP AgNP-CTAB | Citrate rod-like PVP spherical CTAB spherical | Citrate 61.2 ± 33.9 (TEM) PVP 9.4 ± 1.3 (TEM) CTAB 5.6 ± 2.1 (TEM) | Citrate −39.8 ± 3.4 PVP −4.8 ± 0.6 CTAB 42.5 ± 2.7 | - | - | 25, 50, 75, 100 µM (Quantified by ICP-MS) 10 µM AgNO3, 2.5, 5.0, 7.5, 10 µg/mL | 72 h | No DNA damage was observed with any of the AgNP-citrate concentrations employed. An increase in tail DNA was recorded after exposure to AgNP-PVP at 100 μM concentration. AgNP-CTAB produces DNA damage only with 50 μM concentration. | [14] |
| Commercial Nanotech PVP-AgNPs | - | 20–30 | - | -- | 5, 10, 15 µg/mL | 3, 6, 9 h | The decrease in MI and the increase in CA have a dependence on concentration and exposure time | [43] | |
| AgNPs Synthetized with leaf extract of Swertia chirata Commercial Sigma-Aldrich | - | Synthesis 20 Commercial 20 | - | - | 5, 10, 20 µg/mL | 4 h | The decrease in MI and the increase in CA have a dependence on concentration. Both AgNPs produce cytotoxic and genotoxic damage similar to AgNO3. | [30] | |
| They were synthesized with Cola nitida pod (p), seed (s), and seed shell (ss). | All semi-spherical | p: 12–80 s: 8–50 ss: 5–40 | - | - | p: 431 s: 457 ss: 454 | 0.01, 0.1, 1, 10 and 100 µg/mL | 24, 48 and 72 h | Cytotoxic and genotoxic damage have a dependence on concentration and exposure time. | [31] |
| Synthesized with plant extract | Semi-spherical | 25–40 | - | - | 440 | 1, 5 and 10 µg/mL | 72 h | Produces a reduction in the number and diameter of roots, decreases in MI, and increases the frequency of CA. | [33] |
| Synthesized AgNPs | - | 2–8 | - | - | - | 1.5 and 15 µg/mL With CMC 1.24 and 12.4 µg/mL | 24 h | Cytotoxic and genotoxic effects with concentration-dependence behavior (MI decrease and CA increase). In the presence of CMC, the cytotoxic damage is lower than the observed for AgNPs alone. Genotoxic damage is found only with 12.4 µg/mL. | [34] |
| Synthesized with Althea officinalis leaf extract (E) and dehydrated root infusion (R) | - | E: 157 ± 11 (DLS), 131 ± 5 (NTA) R: 293 ± 12 (DLS) 227 ± 16 (NTA) | E: 20.1 ± 1 R: 26.0 ± 1 | E: 7.2 × 1010 NP/mL (NTA); R: 4.6 × 1010 NP/mL | E 384 R 380 | E: 3 × 1010 NP/mL (3.4 µg/mL) R: 3 × 108 NP/mL | 24 h | An increase in MI and CA observed. AgNPs produce a frequency increase on cells with chromosome damage more than 3-times compared with control, but the extract of Althea officinalis produces a frequency increase of nearly 3-times | [35] |
| Biogenic AgNPs obtained with Fusarium oxysporum. Unwashed (AgNPuw) and washed (AgNPw) with water | - | AgNPuw 40.3 ± 3.5 (TEM) 106.2 ± 13 (DLS) AgNPw 40.3 ± 3.5 (TEM) 145.1 ± 4.5 (DLS) | AgNPuw −37.1 ± 2.6 AgNPw −47.8 ± 1.1 | - | - | 0.5, 1, 5 and 10 µg/mL | 24 h | No difference in the MI compared with control, but 5 and 10 µg/mL of AgNPs increase the frequency of CA. No data of lower concentration was provided. Results of genotoxicity at concentrations 5.0 and 10.0 ug/mL show some response, but at concentrations 0.5 and 1.0 μg/mL, the washed and unwashed silver nanoparticles did not present any effect. | [36] |
| Commercial BioPure Silver Nanospheres–PVP (5, 25, 50, 75 nm) PVP: 40 kDa from nanoComposix® Characterization performed by the authors BE: before exposure AE: After exposure | All nanoparticles are spherical | AgNP5 size: 10.4 ± 4.7 nm (TEM). BE d: 42.6 ± 19.2 nm (DLS); AE d: 161.2 ± 55.5 nm (DLS) AgNP25 size: 20.4 ± 7.2 nm (TEM) BE d: 77.1 ± 26.2 nm (DLS); AE d: 94.5 ± 42.9 nm (DLS) AgNP50 size: 51.3 ± 7.4 nm BE d: 80.5 ± 30.4 nm (DLS); AE d: 103.3 ± 46.5 nm (DLS) AgNP75 size: 73.4 ± 4.7 nm BE d: 124.4 ± 48.1 nm (DLS); AE d: 119.8 ± 42.1 nm (DLS) | AgNP5 BE −15.6 AE −8.35 AgNP25 BE −11.2 AE −6.81 AgNP50 BE −16.3 AE −7.53 AgNP75 BE −13.0 AE −6.42 | AgNP5 Release of Ag+ from PVP-AgNPs in distilled water: 0.75% AgNP25 Release of Ag+ from PVP-AgNPs in distilled water: 0.29% AgNP50 Release of Ag+ from PVP-AgNPs in distilled water: 0.03% AgNP75 Release of Ag+ from PVP-AgNPs in distilled water: < LOQ | - | 100 µg/mL | 48 h | The smaller the AgNPs diameter, the more the MI decrease, the MN frequency increases compared to the control group | [15] |
| Synthesized AgNPs with cocoa pod husk (A = CPHE-AgNPs) and cocoa bean (B = CBE-AgNPs) | A 4–32 (TEM) B 8.9–54.2 (TEM) | - | - | - | A 428 B 438 | 0.01, 0.1, 1, 10 and 100 µg/mL | 24, 48 and 72 h | Cytotoxicity and genotoxicity shown dependence on concentration and time exposure | [32] |
2. Materials and Methods
2.1. Materials
2.2. Experimental Design
2.3. Sample Preparation
2.4. Mitotic Index and Genotoxicity
2.5. Determination of Antioxidant Capacity
2.6. Determination of Reactive Oxygen Species (ROS)
2.7. Determination of Total Phenolic Content (TPC)
2.8. Determination of Lipoperoxidation (LPO)
2.9. Statistical Analysis
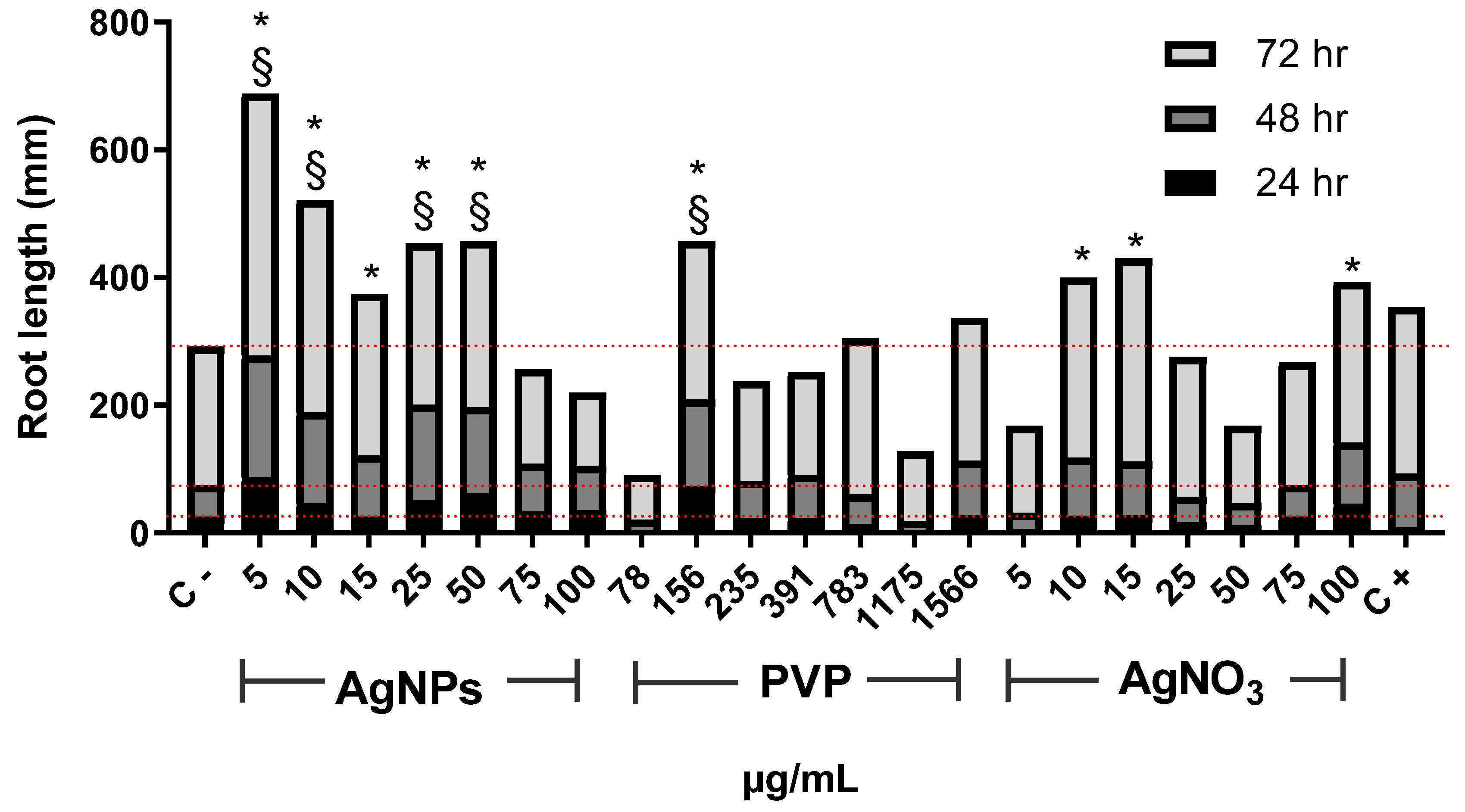
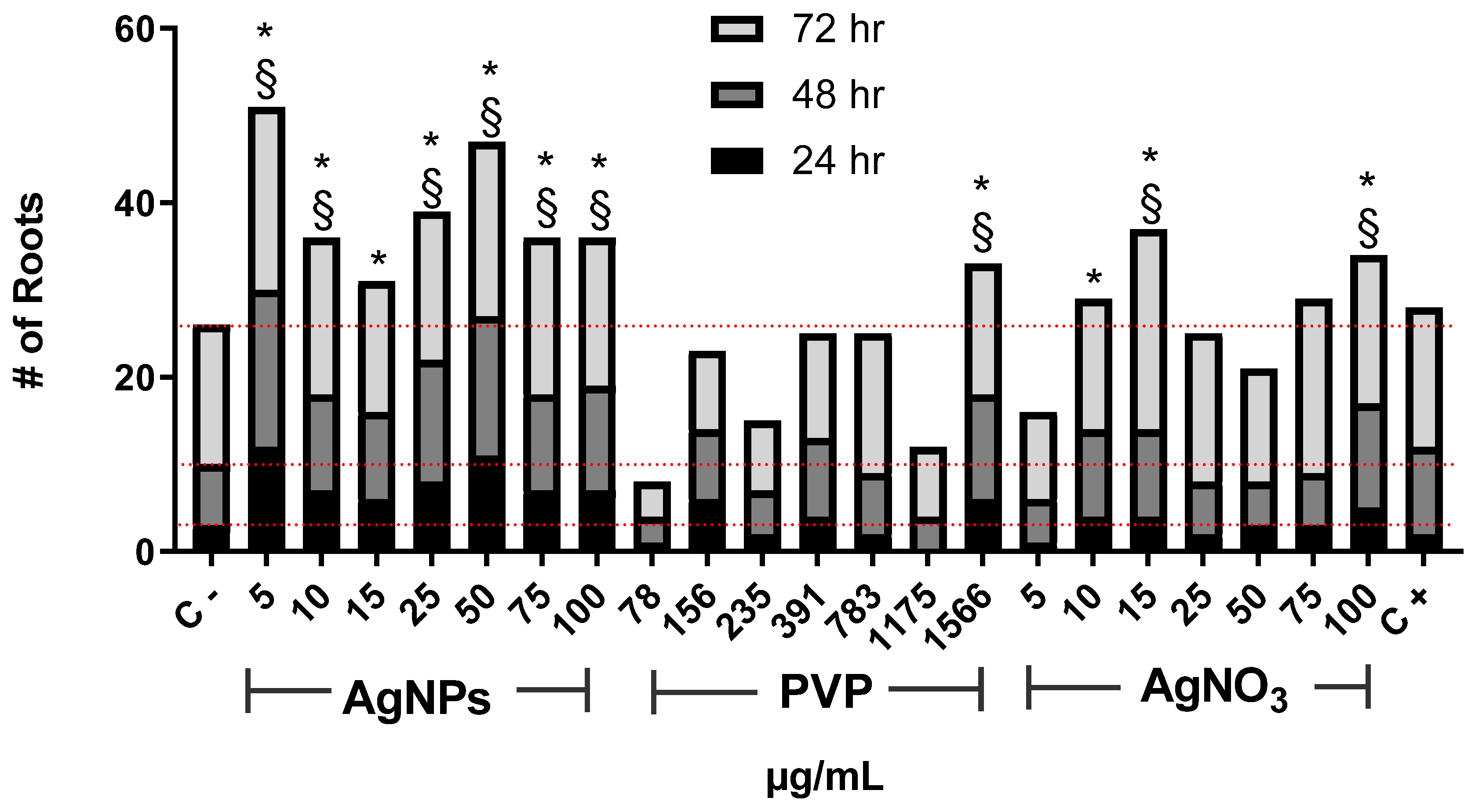
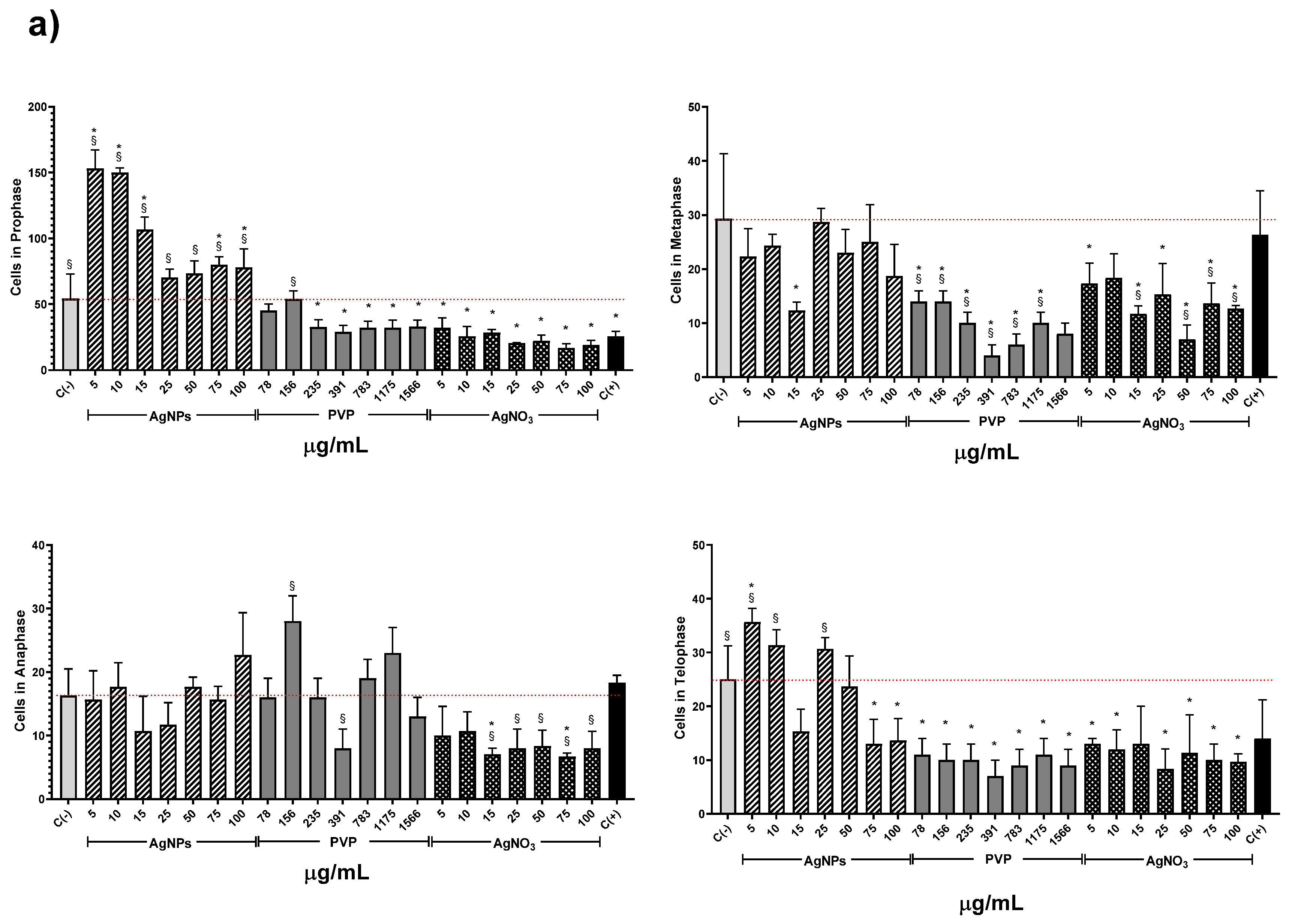
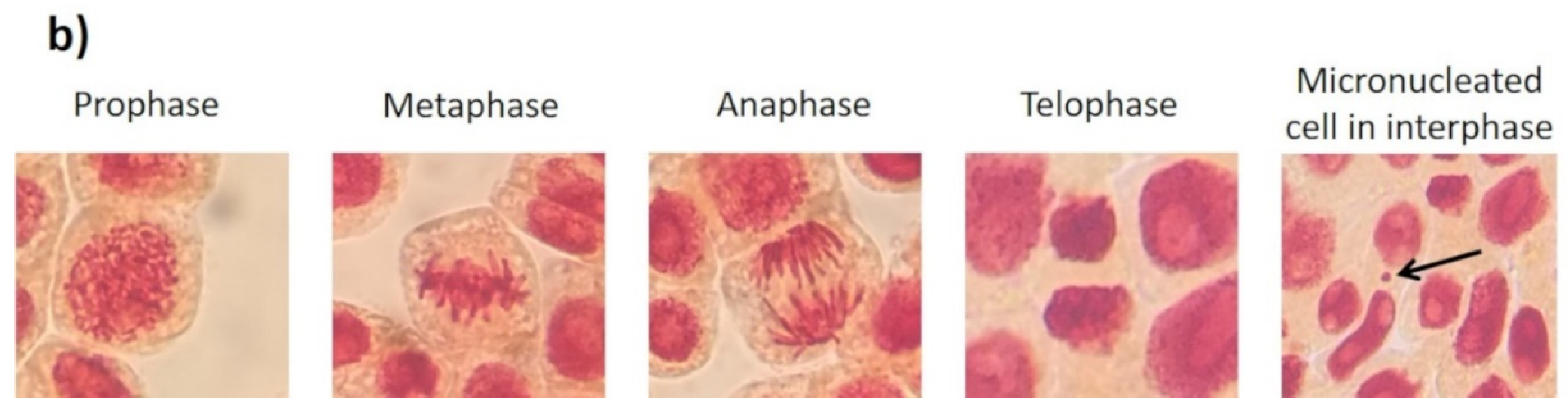
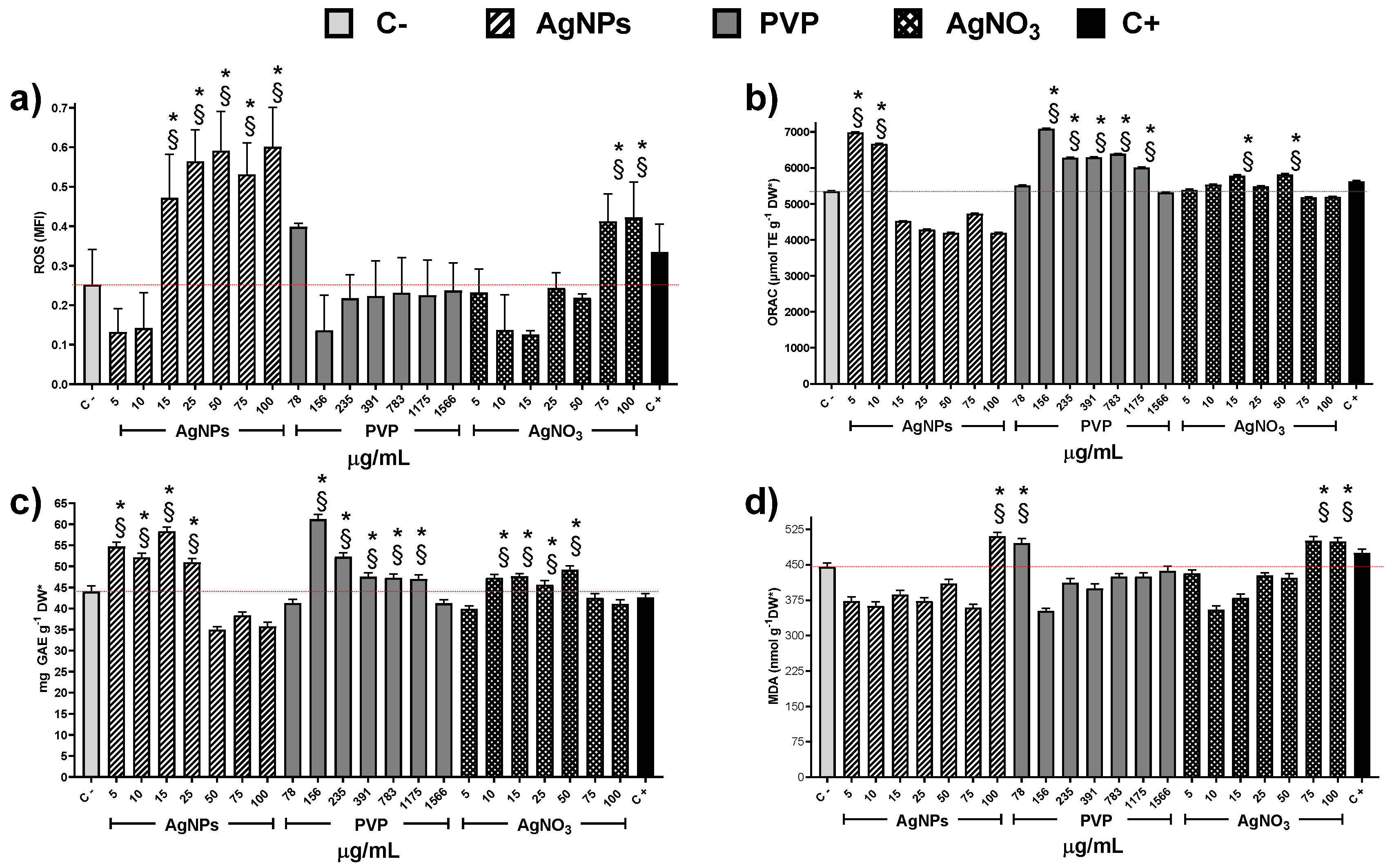
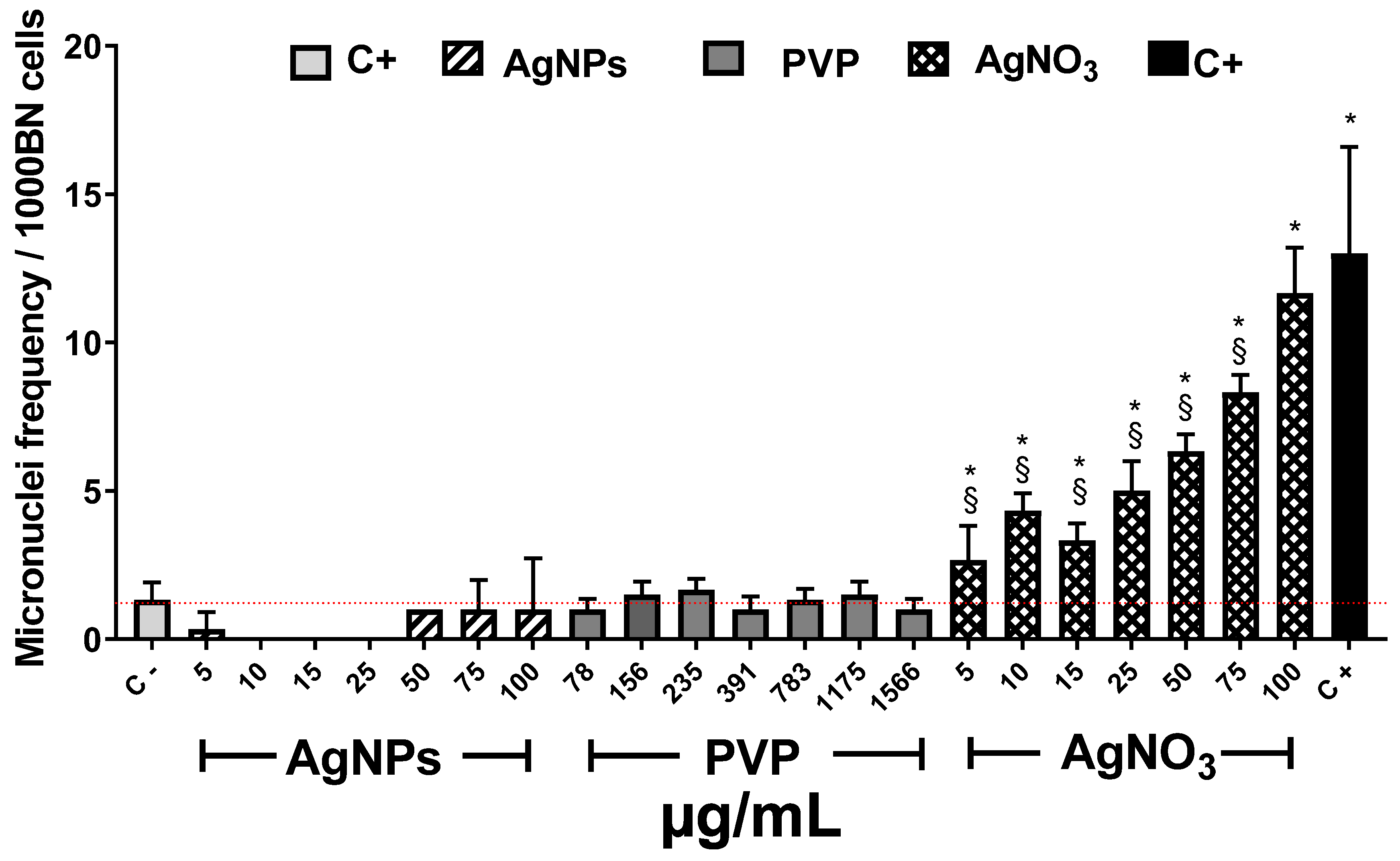
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liao, C.; Li, Y.; Tjong, S.C. Bactericidal and cytotoxic properties of silver nanoparticles. Int. J. Mol. Sci. 2019, 20, 449. [Google Scholar] [CrossRef] [PubMed]
- Yan, A.; Chen, Z. Impacts of silver nanoparticles on plants: A focus on the phytotoxicity and underlying mechanism. Int. J. Mol. Sci. 2019, 20, 1003. [Google Scholar] [CrossRef] [PubMed]
- Burdușel, A.C.; Gherasim, O.; Grumezescu, A.M.; Mogoantă, L.; Ficai, A.; Andronescu, E. Biomedical Applications of Silver Nanoparticles: An Up-to-Date Overview. Nanomaterials 2018, 8, 681. [Google Scholar] [CrossRef] [PubMed]
- Ahlberg, S.; Antonopulos, A.; Diendorf, J.; Dringen, R.; Epple, M.; Flöck, R.; Goedecke, W.; Graf, C.; Haberl, N.; Helmlinger, J.; et al. PVP-coated, negatively charged silver nanoparticles: A multi-center study of their physicochemical characteristics, cell culture and in vivo experiments. Beilstein J. Nanotechnol. 2014, 5, 1944–1965. [Google Scholar] [CrossRef]
- Butler, K.S.; Peeler, D.J.; Casey, B.J.; Dair, B.J.; Elespuru, R.K. Silver nanoparticles: Correlating nanoparticle size and cellular uptake with genotoxicity. Mutagenesis 2015, 30, 577–591. [Google Scholar] [CrossRef] [PubMed]
- Kedziora, A.; Speruda, M.; Krzyzewska, E.; Rybka, J.; Lukoviak, A.; Bugla-Ploskonska, G. Similarities and Differences between Silver Ions and Silver in Nanoforms as Antibacterial Agents. Int. J. Mol. Sci. 2018, 19, 444. [Google Scholar] [CrossRef]
- Rafique, M.; Sadaf, I.; Rafique, M.S.; Tahir, M.B. A review on green synthesis of silver nanoparticles and their applications. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1272–1291. [Google Scholar] [CrossRef]
- Hadrup, N.; Lam, H.R. Oral toxicity of silver ions, silver nanoparticles and colloidal silver—A review. Regul. Toxicol. Pharmacol. 2014, 68, 1–7. [Google Scholar] [CrossRef]
- Akter, M.; Sikder, M.T.; Rahman, M.M.; Ullah, A.K.M.A.; Hossain, K.F.B.; Banik, S.; Hosokawa, T.; Saito, T.; Kurasaki, M. A systematic review on silver nanoparticles-induced cytotoxicity: Physicochemical properties and perspectives. J. Adv. Res. 2018, 9, 1–16. [Google Scholar] [CrossRef]
- Milić, M.; Leitinger, G.; Pavičić, I.; Zebić Avdičević, M.; Dobrović, S.; Goessler, W.; Vinković Vrček, I. Cellular uptake and toxicity effects of silver nanoparticles in mammalian kidney cells. J. Appl. Toxicol. 2015, 35, 581–592. [Google Scholar] [CrossRef]
- Antony, J.J.; Sivalingam, P.; Chen, B. Toxicological effects of silver nanoparticles. Environ. Toxicol. Pharmacol. 2015, 40, 729–732. [Google Scholar] [CrossRef]
- Liu, W.; Wu, Y.; Wang, C.; Li, H.C.; Wang, T.; Liao, C.Y.; Cui, L.; Zhou, Q.F.; Yan, B.; Jiang, G.B. Impact of silver nanoparticles on human cells: Effect of particle size. Nanotoxicology 2010, 4, 319–330. [Google Scholar] [CrossRef] [PubMed]
- Levard, C.; Hotze, E.M.; Lowry, G.V.; Brown, G.E. Environmental transformations of silver nanoparticles: Impact on stability and toxicity. Environ. Sci. Technol. 2012, 46, 6900–6914. [Google Scholar] [CrossRef] [PubMed]
- Cvjetko, P.; Milošić, A.; Domijan, A.M.; Vinković Vrček, I.; Tolić, S.; Peharec Štefanić, P.; Letofsky-Papst, I.; Tkalec, M.; Balen, B. Toxicity of silver ions and differently coated silver nanoparticles in Allium cepa roots. Ecotoxicol. Environ. Saf. 2017, 137, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Scherer, M.D.; Sposito, J.C.V.; Falco, W.F.; Grisolia, A.B.; Andrade, L.H.C.; Lima, S.M.; Machado, G.; Nascimento, V.A.; Gonçalves, D.A.; Wender, H.; et al. Cytotoxic and genotoxic effects of silver nanoparticles on meristematic cells of Allium cepa roots: A close analysis of particle size dependence. Sci. Total Environ. 2019, 660, 459–467. [Google Scholar] [CrossRef]
- Foldbjerg, R.; Jiang, X.; Micləuş, T.; Chen, C.; Autrup, H.; Beer, C. Silver nanoparticles—Wolves in sheep’s clothing? Toxicol. Res. 2015, 4, 563–575. [Google Scholar] [CrossRef]
- Yin, L.; Cheng, Y.; Espinasse, B.; Colman, B.P.; Auffan, M.; Wiesner, M.; Rose, J.; Liu, J.; Bernhardt, E.S. More than the ions: The effects of silver nanoparticles on lolium multiflorum. Environ. Sci. Technol. 2011, 45, 2360–2367. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qin, T.; Ingle, T.; Yan, J.; He, W.; Yin, J.J.; Chen, T. Differential genotoxicity mechanisms of silver nanoparticles and silver ions. Arch. Toxicol. 2017, 91, 509–519. [Google Scholar] [CrossRef]
- Behzadi, S.; Serpooshan, V.; Tao, W.; Hamaly, M.A.; Alkawareek, M.Y.; Dreaden, E.C.; Brown, D.; Alkilany, A.M.; Farokhzad, O.C.; Mahmoudi, M. Cellular uptake of nanoparticles: Journey inside the cell. Chem. Soc. Rev. 2017, 46, 4218–4244. [Google Scholar] [CrossRef]
- Vinković Vrček, I.; Pavičić, I.; Crnković, T.; Jurašin, D.; Babič, M.; Horák, D.; Lovrić, M.; Ferhatović, L.; Ćurlin, M.; Gajović, S. Does surface coating of metallic nanoparticles modulate their interference with in vitro assays? RSC Adv. 2015, 5, 70787–70807. [Google Scholar] [CrossRef]
- Jurašin, D.D.; Ćurlin, M.; Capjak, I.; Crnković, T.; Lovrić, M.; Babič, M.; Horïk, D.; Vrček, I.V.; Gajović, S. Surface coating affects behavior of metallic nanoparticles in a biological environment. Beilstein J. Nanotechnol. 2016, 7, 246–262. [Google Scholar] [CrossRef]
- Nallanthighal, S.; Chan, C.; Bharali, D.J.; Mousa, S.A.; Vásquez, E.; Reliene, R. Particle coatings but not silver ions mediate genotoxicity of ingested silver nanoparticles in a mouse model. NanoImpact 2017, 5, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Pareek, V.; Gupta, R.; Panwar, J. Do physico-chemical properties of silver nanoparticles decide their interaction with biological media and bactericidal action? A review. Mater. Sci. Eng. C 2018, 90, 739–749. [Google Scholar] [CrossRef] [PubMed]
- OCDE. OECD 474 Guideline For The Testing Of Chemicals: Mammalian Erythrocyte Micronucleous Test; OECD Publishing: Paris, France, 2016. [Google Scholar]
- Leme, D.M.; Marin-Morales, M.A. Allium cepa test in environmental monitoring: A review on its application. Mutat. Res. Rev. Mutat. Res. 2009, 682, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Barreto, M.R.; Aleixo, N.A.; Silvestre, R.B.; Fregonezi, F.; Barud, S.; Dias, S.; Ribeiro, C.A.; Resende, R.A. Genotoxicological safety assessment of puree-only edible films from onion bulb (Allium cepa L.) for use in food packaging-related applications. J. Food Sci. 2019. [Google Scholar] [CrossRef]
- De Souza, C.P.; de Guedes, T.A.; Fontanetti, C.S. Evaluation of herbicides action on plant bioindicators by genetic biomarkers: A review. Environ. Monit. Assess. 2016, 188. [Google Scholar] [CrossRef]
- Stapulionytė, A.; Kleizaitė, V.; Šiukšta, R.; Žvingila, D.; Taraškevičius, R.; Čėsnienė, T. Cyto/genotoxicological evaluation of hot spots of soil pollution using Allium bioassays in relation to geochemistry. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2019, 842, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Mukherjee, A.; Chandrasekaran, N. Genotoxicity of silver nanoparticles in Allium cepa. Sci. Total Environ. 2009, 407, 5243–5246. [Google Scholar] [CrossRef]
- Saha, N.; Dutta Gupta, S. Low-dose toxicity of biogenic silver nanoparticles fabricated by Swertia chirata on root tips and flower buds of Allium cepa. J. Hazard. Mater. 2017, 330, 18–28. [Google Scholar] [CrossRef]
- Yekeen, T.A.; Azeez, M.A.; Akinboro, A.; Lateef, A.; Asafa, T.B.; Oladipo, I.C.; Oladokun, S.O.; Ajibola, A.A. Safety evaluation of green synthesized Cola nitida pod, seed and seed shell extract-mediated silver nanoparticles (AgNPs) using an Allium cepa assay. J. Taibah Univ. Sci. 2017, 11, 895–909. [Google Scholar] [CrossRef]
- Yekeen, T.A.; Azeez, M.A.; Lateef, A.; Asafa, T.B.; Oladipo, I.C.; Badmus, J.A.; Adejumo, S.A.; Ajibola, A.A. Cytogenotoxicity potentials of cocoa pod and bean-mediated green synthesized silver nanoparticles on Allium cepa cells. Caryologia 2017, 70, 366–377. [Google Scholar] [CrossRef]
- Debnath, P.; Mondal, A.; Hajra, A.; Das, C.; Mondal, N.K. Cytogenetic effects of silver and gold nanoparticles on Allium cepa roots. J. Genet. Eng. Biotechnol. 2018, 16, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Becaro, A.A.; Siqueira, M.C.; Puti, F.C.; de Moura, M.R.; Correa, D.S.; Marconcini, J.M.; Mattoso, L.H.C.; Ferreira, M.D. Cytotoxic and genotoxic effects of silver nanoparticle/carboxymethyl cellulose on Allium cepa. Environ. Monit. Assess. 2017, 189. [Google Scholar] [CrossRef]
- Rheder, D.T.; Guilger, M.; Bilesky-José, N.; Germano-Costa, T.; Pasquoto-Stigliani, T.; Gallep, T.B.B.; Grillo, R.; dos Carvalho, C.S.; Fraceto, L.F.; Lima, R. Synthesis of biogenic silver nanoparticles using Althaea officinalis as reducing agent: Evaluation of toxicity and ecotoxicity. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Lima, R.; Feitosa, L.O.; Ballottin, D.; Marcato, P.D.; Tasic, L.; Durán, N. Cytotoxicity and genotoxicity of biogenic silver nanoparticles. J. Phys. Conf. Ser. 2013, 429. [Google Scholar] [CrossRef]
- Panda, K.K.; Achary, V.M.M.; Krishnaveni, R.; Padhi, B.K.; Sarangi, S.N.; Sahu, S.N.; Panda, B.B. In vitro biosynthesis and genotoxicity bioassay of silver nanoparticles using plants. Toxicol. Vitr. 2011, 25, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Zhou, T.; Cunningham, C.K.; Chen, T.; Jones, M.Y.; Abbas, M.; Li, Y.; Mei, N.; Guo, X.; Moore, M.M.; et al. Size- and coating-dependent cytotoxicity and genotoxicity of silver nanoparticles evaluated using in vitro standard assays. Nanotoxicology 2016, 10, 1373–1384. [Google Scholar] [CrossRef]
- Andreani, T.; Nogueira, V.; Pinto, V.V.; Ferreira, M.J.; Rasteiro, M.G.; Silva, A.M.; Pereira, R.; Pereira, C.M. Influence of the stabilizers on the toxicity of metallic nanomaterials in aquatic organisms and human cell lines. Sci. Total Environ. 2017, 607, 1264–1277. [Google Scholar] [CrossRef] [PubMed]
- Nymark, P.; Catalán, J.; Suhonen, S.; Järventaus, H.; Birkedal, R.; Clausen, P.A.; Jensen, K.A.; Vippola, M.; Savolainen, K.; Norppa, H. Genotoxicity of polyvinylpyrrolidone-coated silver nanoparticles in BEAS 2B cells. Toxicology 2013, 313, 38–48. [Google Scholar] [CrossRef]
- Panda, K.K.; Achary, V.M.M.; Phaomie, G.; Sahu, H.K.; Parinandi, N.L.; Panda, B.B. Polyvinyl polypyrrolidone attenuates genotoxicity of silver nanoparticles synthesized via green route, tested in Lathyrus sativus L. root bioassay. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2016, 806, 11–23. [Google Scholar] [CrossRef]
- Ghosh, M.; Manivannan, J.; Sinha, S.; Chakraborty, A.; Mallick, S.K.; Bandyopadhyay, M.; Mukherjee, A. In vitro and in vivo genotoxicity of silver nanoparticles. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2012, 749, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Fouad, A.S.; Hafez, R.M. The effects of silver ions and silver nanoparticles on cell division and expression of cdc2 gene in Allium cepa root tips. Biol. Plant. 2018, 62, 166–172. [Google Scholar] [CrossRef]
- Almonaci Hernández, C.A.; Juarez-Moreno, K.; Castañeda Juarez, M.E.; Almanza-Reyes, H.; Pestryakov, A.; Bogdanchikova, N. Silver Nanoparticles for the Rapid Healing of Diabetic Foot Ulcers. Int. J. Med. Nano Res. 2017, 4, 19. [Google Scholar] [CrossRef]
- Valenzuela-Salas, L.M.; Girón-Vázquez, N.G.; García-Ramos, J.C.; Torres-Bugarín, O.; Gómez, C.; Pestryakov, A.; Villarreal-Gómez, L.J.; Toledano-Magaña, Y.; Bogdanchikova, N. Antiproliferative and antitumor effect of non-genotoxic silver nanoparticles on melanoma models. Oxid. Med. Cell. Longev. 2019, 2019, 4528241. [Google Scholar] [CrossRef]
- Ochoa-Meza, A.R.; Álvarez-Sánchez, A.R.; Romo-Quiñonez, C.R.; Barraza, A.; Magallón-Barajas, F.J.; Chávez-Sánchez, A.; García-Ramos, J.C.; Toledano-Magaña, Y.; Bogdanchikova, N.; Pestryakov, A.; et al. Silver nanoparticles enhance survival of white spot syndrome virus infected Penaeus vannamei shrimps by activation of its immunological system. Fish Shellfish Immunol. 2019, 84, 1083–1089. [Google Scholar] [CrossRef]
- Bogdanchikova, N.; Vázquez-Muñoz, R.; Huerta-Saquero, A.; Peña-Jasso, A.; Aguilar-Uzcanga, G.; Picos-Díaz, P.L.; Pestryakov, A.; Burmistrov, V.A.; Martynyuk, O.; Luna-Vázquez-Gómez, R.; et al. Silver nanoparticles composition for treatment of distemper in dogs. Int. J. Nanotechnol. 2016, 13, 227–237. [Google Scholar] [CrossRef]
- Borrego, B.; Lorenzo, G.; Mota-Morales, J.D.; Almanza-Reyes, H.; Mateos, F.; López-Gil, E.; de la Losa, N.; Burmistrov, V.A.; Pestryakov, A.N.; Brun, A.; et al. Potential application of silver nanoparticles to control the infectivity of Rift Valley fever virus in vitro and in vivo. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 1185–1192. [Google Scholar] [CrossRef]
- Pimentel-Acosta, C.A.; Morales-Serna, F.N.; Chávez-Sánchez, M.C.; Lara, H.H.; Pestryakov, A.; Bogdanchikova, N.; Fajer-Ávila, E.J. Efficacy of silver nanoparticles against the adults and eggs of monogenean parasites of fish. Parasitol. Res. 2019, 118, 1741–1749. [Google Scholar] [CrossRef]
- Spinoso-Castillo, J.L.; Chavez-Santoscoy, R.A.; Bogdanchikova, N.; Pérez-Sato, J.A.; Morales-Ramos, V.; Bello-Bello, J.J. Antimicrobial and hormetic effects of silver nanoparticles on in vitro regeneration of vanilla (Vanilla planifolia Jacks. ex Andrews) using a temporary immersion system. Plant Cell. Tissue Organ Cult. 2017, 129, 195–207. [Google Scholar] [CrossRef]
- Bello-Bello, J.; Spinoso-Castillo, J.; Arano-Avalos, S.; Martínez-Estrada, E.; Arellano-García, M.; Pestryakov, A.; Toledano-Magaña, Y.; García-Ramos, J.; Bogdanchikova, N. Cytotoxic, Genotoxic, and Polymorphism Effects on Vanilla planifolia Jacks ex Andrews after Long-Term Exposure to Argovit® Silver Nanoparticles. Nanomaterials 2018, 8, 754. [Google Scholar] [CrossRef]
- Juarez-Moreno, K.; Mejía-Ruiz, C.H.; Díaz, F.; Reyna-Verdugo, H.; Re, A.D.; Vazquez-Felix, E.F.; Sánchez-Castrejón, E.; Mota-Morales, J.D.; Pestryakov, A.; Bogdanchikova, N. Effect of silver nanoparticles on the metabolic rate, hematological response, and survival of juvenile white shrimp Litopenaeus vannamei. Chemosphere 2017, 169, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Fiskesjö, G. Mercury and selenium in a modified Allium test. Hereditas 1979, 91, 169–178. [Google Scholar] [CrossRef]
- Fiskesjö, G. The Allium test as a standard in environmental monitoring. Hereditas 1985, 102, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Yi, H.; Yi, M. Assessment of arsenic toxicity using Allium/Vicia root tip micronucleus assays. J. Hazard. Mater. 2010, 176, 952–956. [Google Scholar] [CrossRef]
- Yi, H.; Wu, L.; Jiang, L. Genotoxicity of arsenic evaluated by Allium-root micronucleus assay. Sci. Total Environ. 2007, 383, 232–236. [Google Scholar] [CrossRef]
- Grant, W.F. Chromosome aberrations in plants as a monitoring system. Environ. Health Perspect. 1978, 27, 37–43. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.A.; Prior, R.L. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 2002, 50, 4437–4444. [Google Scholar] [CrossRef]
- Bartlett, M.S. Properties of sufficiency and statistical tests. Proc. R. Soc. Lond. Ser. A Math. Phys. Sci. 1937, 160, 268–282. [Google Scholar] [CrossRef]
- NIST/SEMATECH e-Handbook of Statistical Methods. Available online: http://www.itl.nist.gov/div898/handbook/ (accessed on 11 May 2020).
- Dizdari, A.M.; Kopliku, D. Cytotoxic and Genotoxic Potency Screening of Two Pesticides on Allium cepa L. Procedia Technol. 2013, 8, 19–26. [Google Scholar] [CrossRef]
- Bello-Bello, J.J.; Chavez-Santoscoy, R.A.; Lecona-Guzmán, C.A.; Bogdanchikova, N.; Salinas-Ruíz, J.; Gómez-Merino, F.C.; Pestryakov, A. Hormetic response by silver nanoparticles on in vitro multiplication of sugarcane (Saccharum spp. Cv. Mex 69-290) using a temporary immersion system. Dose Response 2017, 15, 1–9. [Google Scholar] [CrossRef]
- Castro-González, C.G.; Sánchez-Segura, L.; Gómez-Merino, F.C.; Bello-Bello, J.J. Exposure of stevia (Stevia rebaudiana B.) to silver nanoparticles in vitro: Transport and accumulation. Sci. Rep. 2019, 9, 10372. [Google Scholar] [CrossRef] [PubMed]
- Cox, A.; Venkatachalam, P.; Sahi, S.; Sharma, N. Reprint of: Silver and titanium dioxide nanoparticle toxicity in plants: A review of current research. Plant Physiol. Biochem. 2017, 110, 33–49. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, J.; Mantovani, M.S.; Marin-Morales, M.A. Analysis of the genotoxic potential of low concentrations of Malathion on the Allium cepa cells and rat hepatoma tissue culture. J. Environ. Sci. 2015, 36, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Batista, N.J.C.; de Carvalho Melo Cavalcante, A.A.; de Oliveira, M.G.; Medeiros, E.C.N.; Machado, J.L.; Evangelista, S.R.; Dias, J.F.; dos Santos, C.E.I.; Duarte, A.; da Silva, F.R.; et al. Genotoxic and mutagenic evaluation of water samples from a river under the influence of different anthropogenic activities. Chemosphere 2016, 164, 134–141. [Google Scholar] [CrossRef]
- Leme, D.M.; Marin-Morales, M.A. Chromosome aberration and micronucleus frequencies in Allium cepa cells exposed to petroleum polluted water-A case study. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2008, 650, 80–86. [Google Scholar] [CrossRef]
- Coskun, D.; Britto, D.T.; Jean, Y.K.; Schulze, L.M.; Becker, A.; Kronzucker, H.J. Silver ions disrupt K+ homeostasis and cellular integrity in intact barley (Hordeum vulgare L.) roots. J. Exp. Bot. 2012, 63, 151–162. [Google Scholar] [CrossRef]
- Schaller, G.; Binder, B. Inhibitors of Ethylene Biosynthesis and Signaling. In Methods in Molecular Biology 1573. Ethylene Signaling. Methods and Protocols; Binder, B., Schaller, G., Eds.; Humana Press: New York, NY, USA, 2017; pp. 223–236. [Google Scholar]
- Vecchio, G.; Fenech, M.; Pompa, P.P.; Voelcker, N.H. Lab-on-a-chip-based high-throughput screening of the genotoxicity of engineered nanomaterials. Small 2014, 10, 2721–2734. [Google Scholar] [CrossRef]
- Ghosh, M.; Ghosh, I.; Godderis, L.; Hoet, P.; Mukherjee, A. Genotoxicity of engineered nanoparticles in higher plants. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2019, 842, 132–145. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Casillas-Figueroa, F.; Arellano-García, M.E.; Leyva-Aguilera, C.; Ruíz-Ruíz, B.; Luna Vázquez-Gómez, R.; Radilla-Chávez, P.; Chávez-Santoscoy, R.A.; Pestryakov, A.; Toledano-Magaña, Y.; García-Ramos, J.C.; et al. Argovit™ Silver Nanoparticles Effects on Allium cepa: Plant Growth Promotion without Cyto Genotoxic Damage. Nanomaterials 2020, 10, 1386. https://doi.org/10.3390/nano10071386
Casillas-Figueroa F, Arellano-García ME, Leyva-Aguilera C, Ruíz-Ruíz B, Luna Vázquez-Gómez R, Radilla-Chávez P, Chávez-Santoscoy RA, Pestryakov A, Toledano-Magaña Y, García-Ramos JC, et al. Argovit™ Silver Nanoparticles Effects on Allium cepa: Plant Growth Promotion without Cyto Genotoxic Damage. Nanomaterials. 2020; 10(7):1386. https://doi.org/10.3390/nano10071386
Chicago/Turabian StyleCasillas-Figueroa, Francisco, María Evarista Arellano-García, Claudia Leyva-Aguilera, Balam Ruíz-Ruíz, Roberto Luna Vázquez-Gómez, Patricia Radilla-Chávez, Rocío Alejandra Chávez-Santoscoy, Alexey Pestryakov, Yanis Toledano-Magaña, Juan Carlos García-Ramos, and et al. 2020. "Argovit™ Silver Nanoparticles Effects on Allium cepa: Plant Growth Promotion without Cyto Genotoxic Damage" Nanomaterials 10, no. 7: 1386. https://doi.org/10.3390/nano10071386
APA StyleCasillas-Figueroa, F., Arellano-García, M. E., Leyva-Aguilera, C., Ruíz-Ruíz, B., Luna Vázquez-Gómez, R., Radilla-Chávez, P., Chávez-Santoscoy, R. A., Pestryakov, A., Toledano-Magaña, Y., García-Ramos, J. C., & Bogdanchikova, N. (2020). Argovit™ Silver Nanoparticles Effects on Allium cepa: Plant Growth Promotion without Cyto Genotoxic Damage. Nanomaterials, 10(7), 1386. https://doi.org/10.3390/nano10071386








