Carbonized Lanthanum-Based Metal-Organic Framework with Parallel Arranged Channels for Azo-Dye Adsorption
Abstract
1. Introduction
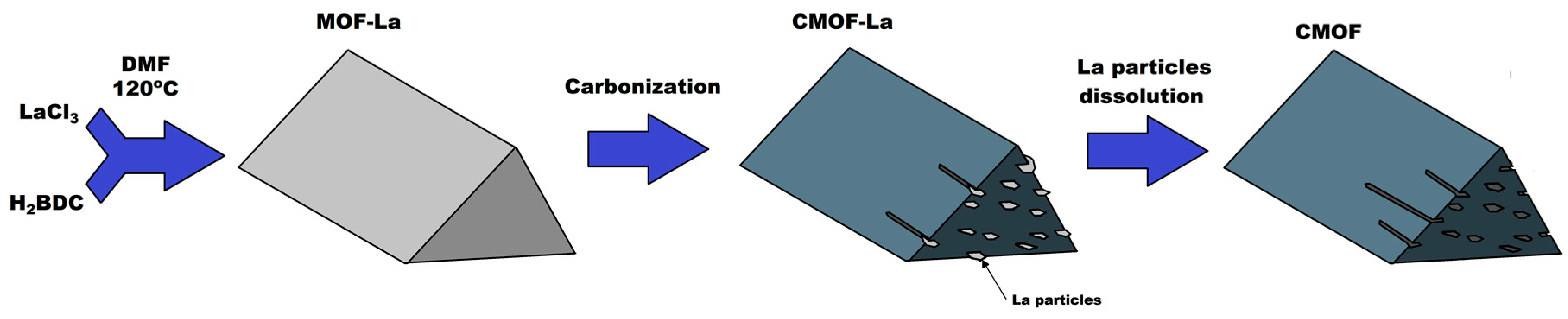
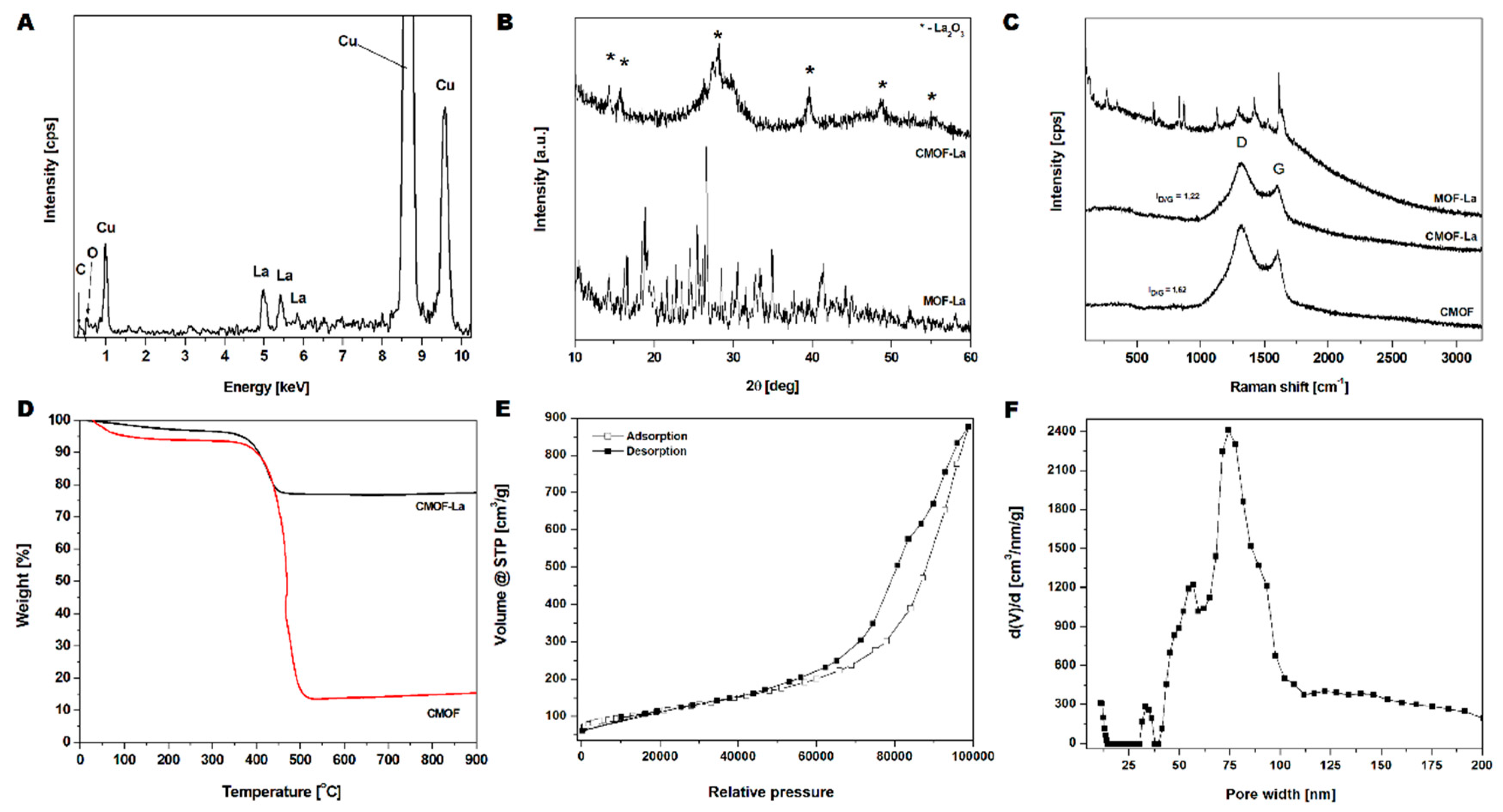
2. Results
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. MOF–La Synthesis
4.3. Carbonization of MOF–La
4.4. Dye Adsorption
4.5. Characterization Techniques
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Srinivasan, A.; Viraraghavan, T. Decolorization of dye wastewaters by biosorbents: A review. J. Environ. Manag. 2010, 91, 1915–1929. [Google Scholar] [CrossRef] [PubMed]
- Wong, Y.C.; Szeto, Y.S.; Cheung, W.H.; McKay, G. Equilibrium studies for acid dye adsorption onto chitosan. Langmuir 2004, 39, 693–702. [Google Scholar] [CrossRef]
- Sarkar, S.; Banerjee, A.; Halder, U.; Biswas, R.; Bandopadhyay, R. Degradation of synthetic azo dyes of textile industry: A sustainable approach using microbial enzymes. Water Conserv. Sci. Eng. 2017, 2, 121–131. [Google Scholar] [CrossRef]
- Wang, S.; Zhai, Y.Y.; Gao, Q.; Luo, W.J.; Xia, H.; Zhou, C.G. Highly efficient removal of Acid Red 18 from aqueous solution by magnetically retrievable chitosan/carbon nanotube: Batch study, isotherms, kinetics, and thermodynamics. J. Chem. Eng. Data 2014, 59, 39–51. [Google Scholar] [CrossRef]
- Janveja, B.; Kant, K.; Sharma, J. A study of activated rice husk charcoal as an adsorbent of congo red dye present in textile industrial waste. J. Punjab Acad. Forensic Med. Toxicol. 2007, 8, 12–15. [Google Scholar]
- Ahmed, M.J.; Theydan, S.K. Physical and chemical characteristics of activated carbon prepared by pyrolysis of chemically treated date stones and its ability to adsorb organics. Powder Technol. 2012, 229, 237–245. [Google Scholar] [CrossRef]
- Yagub, M.T.J.; Sen, T.K.J.; Afroze, S.J.; Ang, H.M.J. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-cost adsorbents for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, B97, 219–243. [Google Scholar] [CrossRef]
- Phan, T.N.T.; Bacquet, M.; Morcellet, M. Synthesis and Characterization of Silica Gels Functionalized with Monochlorotriazinyl β-Cyclodextrin and their Sorption Capacities towards Organic Compounds. J. Incl. Phenom. Macrocycl. Chem. 2000, 38, 345–359. [Google Scholar] [CrossRef]
- Sabarish, R.; Unnikrishnan, G. Polyvinyl alcohol/carboxymethyl cellulose/ZSM-5 zeolite biocomposite membranes for dye adsorption applications. Carbohydr. Polym. 2018, 199, 129–140. [Google Scholar] [CrossRef]
- Nassar, N.N. Kinetics, mechanistic, equilibrium, and thermodynamic studies on the adsorption of acid red dye from wastewater by γ-Fe2O3 nanoadsorbents. Sep. Sci. Technol. 2010, 45, 1092–1103. [Google Scholar] [CrossRef]
- Asgher, M.; Bhatti, H.N. Evaluation of thermodynamics and effect of chemical treatments on sorption potential of (Citrus) waste biomass for removal of anionic dyes from aqueous solutions. Ecol. Eng. 2012, 38, 79–85. [Google Scholar] [CrossRef]
- Molavia, H.; Hakimian, A.; Shojaeia, A.; Raeiszadeh, M. Selective dye adsorption by highly water stable metal-organic framework: Long term stability analysis in aqueous media. Appl. Surf. Sci. 2018, 445, 424–436. [Google Scholar] [CrossRef]
- Song, Y.; Li, X.; Sun, L.; Wang, L. Metal/metal oxide nanostructures derived from metal–organic frameworks. RSC Adv. 2015, 5, 7267–7279. [Google Scholar] [CrossRef]
- Gangu, K.K.; Maddila, S.; Mukkamala, S.B.; Jonnalagadda, S.B. A review on contemporary Metal Organic Framework materials. Inorg. Chim. Acta 2016, 446, 61–74. [Google Scholar] [CrossRef]
- Yang, H.; Chang, Z.; Qiao, Y.; Deng, H.; Mu, X.; He, P.; Zhou, H. Constructing a Super-Saturated Electrolyte Front Surface for Stable Rechargeable Aqueous Zinc Batteries. Angew. Chem. Int. Ed. 2020. [Google Scholar] [CrossRef]
- Qiao, Y.; He, Y.; Wu, S.; Jiang, K.; Li, X.; Guo, S.; He, P.; Zhou, H. MOF-Based Separator in an Li–O2 Battery: An Effective Strategy to Restrain the Shuttling of Dual Redox Mediators. ACS Energy Lett. 2018, 3, 463–468. [Google Scholar] [CrossRef]
- Han, X.; Yang, J.; Liu, Y.Y.; Ma, J.F. Nine coordination polymers assembled with a novel resorcin[4]arene tetracarboxylic acid: Selective luminescent sensing of acetone and Fe3+ ion. Dyes Pigment. 2019, 160, 492–500. [Google Scholar] [CrossRef]
- Shen, J.; Wang, N.; Wang, Y.G.; Yu, D.; Ouyang, X. Effificient Adsorption of Pb(II) from Aqueous Solutions by Metal Organic Framework (Zn-BDC) Coated Magnetic Montmorillonite. Polymers 2018, 10, 1383. [Google Scholar] [CrossRef]
- Juan-Juan, J.; Marco-Lozar, J.P.; Suarez-Garcıa, F.; Cazorla-Amoro´s, D.; Linares-Solano, A. A comparison of hydrogen storage in activated carbons and a metal–organic framework (MOF-5). Carbon 2010, 48, 2906–2909. [Google Scholar] [CrossRef]
- Sarkar, B.; Xi, Y.; Megharaj, M.; Naidu, R. Orange II adsorption on palygorskites modified with alkyl trimethylammonium and dialkyl dimethylammonium bromide—An isothermal and kinetic study. Appl. Clay Sci. 2011, 51, 370–374. [Google Scholar] [CrossRef]
- Zhan, X.Q.; Tsai, F.C.; Xie, L.; Zhang, K.D.; Liu, H.L.; Ma, N.; Shi, D.; Jiang, T. Ligands-Coordinated Zr-Based MOF for Wastewater Treatment. Nanomaterials (Basel) 2018, 8, 655. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, A.; Acharya, H. Facile Synthesis of MgAl-Layered Double Hydroxide Supported Metal Organic Framework Nanocomposite for Adsorptive Removal of Methyl Orange Dye. J. Colloid Interface Sci. Commun. 2018, 24, 35–39. [Google Scholar] [CrossRef]
- Zhan, X.Q.; Yu, X.Y.; Tsai, F.C.; Ma, N.; Liu, H.L.; Han, Y.; Xie, L.; Jiang, T.; Shi, D.; Xiong, Y. Magnetic MOF for AO7 Removal and Targeted Delivery. Crystals 2018, 8, 250. [Google Scholar] [CrossRef]
- Cendrowski, K.; Skumial, P.; Spera, P.; Mijowska, E. Thermally induced formation of zinc oxide nano structures with tailoring morphology during metalorganic framework (MOF-5) carbonization process. Mater. Des. 2016, 110, 740–748. [Google Scholar] [CrossRef]
- García, E.R.; Medina, R.L.; Lozano, M.M.; Pérez, I.H.; Valero, M.J.; Franco, A.M.M. Adsorption of Azo-Dye Orange II from Aqueous Solutions Using a Metal-Organic Framework Material: Iron- Benzenetricarboxylate. Materials 2014, 7, 8037–8057. [Google Scholar] [CrossRef]
- Bünzli, J.C.G. Review: Lanthanide coordination chemistry: From old concepts to coordination polymers. J. Coord. Chem. 2014, 67, 3706–3733. [Google Scholar] [CrossRef]
- Moore, E.G.; Samuel, A.P.S.; Raymond, K.N. From Antenna to Assay: Lessons Learned in Lanthanide Luminescence. Acc. Chem. Res. 2009, 42, 542–552. [Google Scholar] [CrossRef]
- Gao, M.L.; Wang, W.J.; Liu, L.; Han, Z.B.; Wei, N.; Cao, X.M.; Yuan, D.Q. Microporous Hexanuclear Ln(III) Cluster-Based Metal-Organic Frameworks: Color Tunability for Barcode Application and Selective Removal of Methylene Blue. Inorg. Chem. 2017, 56, 511–517. [Google Scholar] [CrossRef]
- Pagis, C.; Ferbinteanu, M.; Rothenberg, G.; Tanase, S. Lanthanide-Based Metal Organic Frameworks: Synthetic Strategies and Catalytic Applications. ACS Catal. 2016, 69, 6063–6072. [Google Scholar] [CrossRef]
- Xue, Y.S.; Chen, Z.L.; Dong, Y.; Cheng, W.W. Two Lanthanide Metal–Organic Frameworks Based on Semi-Rigid T-Shaped Tricarboxylate Ligand: Syntheses, Structures, and Properties. Polymers 2019, 11, 868. [Google Scholar] [CrossRef] [PubMed]
- Prabhu, S.M.; Imamura, S.; Sasaki, K. Mono-, Di-, and Tricarboxylic Acid Facilitated Lanthanum-Based Organic Frameworks: Insights into the Structural Stability and Mechanistic Approach for Superior Adsorption of Arsenate from Water. ACS Sustain. Chem. Eng. 2019, 7, 6917–6928. [Google Scholar] [CrossRef]
- Sheta, M.S.; El-Sheikh, S.M.; Abd-Elzaher, M.M.; Wassel, A.R. A novel nano-size lanthanum metal–organic framework based on 5-amino-isophthalic acid and phenylenediamine: Photoluminescence study and sensing applications. Appl Organometal Chem. 2019, 33, e4777. [Google Scholar] [CrossRef]
- Liu, C.H.; Zhang, L.J.; Bai, F.Y.; Wang, Y.; Hong, Y.Z.; Li, C.R.; Xing, Y.H. Dye Adsorption and Fluorescence Sensing Behaviour About Rare Earth-Indole Carboxylic Acid Complexes. J. Inorg. Organomet. Polym. Mater. 2018, 28, 1839–1849. [Google Scholar] [CrossRef]
- Han, Y.; Li, X.; Li, L.; Ma, C.; Shen, Z.; Song, Y.; You, X. Structures and Properties of Porous Coordination Polymers Based on Lanthanide Carboxylate Building Units. Inorg. Chem. 2010, 49, 10781–10787. [Google Scholar] [CrossRef]
- Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S.; Cancado, L.G.; Jorio, A.; Saito, R. Studying disorder in graphite-based systems by Raman spectroscopy. Phys. Chem. Chem. Phys. 2007, 9, 1276–1291. [Google Scholar] [CrossRef]
- Yan, H.; Wu, H.; Li, K.; Wang, Y.; Tao, X.; Yang, H.; Li, A.; Cheng, R. Influence of the surface structure of graphene oxide on the adsorption of aromatic organic compounds from water. ACS Appl. Mater. Interfaces 2015, 7, 6690–6697. [Google Scholar] [CrossRef]
- Lata, S.; Singh, P.K.; Samadder, S.R. Regeneration of adsorbents and recovery of heavy metals: A review. Int. J. Environ. Sci. Technol. 2015, 12, 1461–1478. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Z.; Wen, H.; Cai, Z.; He, C.; Wang, Z.; Yan, W. Microwave assisted modification of activated carbons by organic acid ammoniums activation for enhanced adsorption of acid red 18. Powder Technol. 2018, 323, 230–237. [Google Scholar] [CrossRef]
- Chen, Y.; Long, W.; Xu, H. Efficient removal of Acid Red 18 from aqueous solution by in-situ polymerization of polypyrrole-chitosan composites. J. Mol. Liq. 2019, 287, 110888. [Google Scholar] [CrossRef]
- Wang, S.; Li, H. Kinetic modelling and mechanism of dye adsorption on unburned carbon. Dyes Pigment. 2007, 72, 308–314. [Google Scholar] [CrossRef]
- Saratale, R.G.; Sivapathan, S.S.; Jung, W.J.; Kim Hee, Y.; Saratale, G.D.; Kim, D.S. Preparation of activated carbons from peach stone by H4P2O7 activation and its application for the removal of Acid Red 18 and dye containing wastewater. J. Environ. Sci. Health Part A 2016, 51, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Cheung, W.H.; Szeto, Y.S.; McKay, G. Enhancing the adsorption capacities of acid dyes by chitosan nano particles. Bioresour. Technol. 2009, 100, 1143–1148. [Google Scholar] [CrossRef] [PubMed]
- Balusamy, B.; Tastan, B.E.; Ergen, S.F.; Uyarbe, T.; Tekinay, T. Toxicity of lanthanum oxide (La2O3) nanoparticles in aquatic environments. Environ. Sci. Process. Impacts 2015, 17, 1265. [Google Scholar] [CrossRef]
- Brabu, B.; Haribabu, S.; Revathy, M.; Anitha, S.; Thangapandiyan, M.; Navaneethakrishnan, K.R.; Gopalakrishnan, C.; Murugana, S.S.; Kumaravel, T.S. Biocompatibility studies on lanthanum oxide nanoparticles. Toxicol. Res. 2015, 4, 1037–1044. [Google Scholar] [CrossRef]
- Lim, C.H. Toxicity of Two Different Sized Lanthanum Oxides in Cultured Cells and Sprague-Dawley Rats. Toxicol. Res. 2015, 31, 181–189. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, F.; Gao, J.; Chen, Y.; Hao, X. Ordered mesoporous TiO2/activated carbon for adsorption and photocatalysis of Acid Red 18 solution. Bioresources 2017, 12, 9086–9102. [Google Scholar]
- Zhao, S.; Zhou, F.; Li, L.; Cao, M.; Zuo, D.; Liu, H. Removal of anionic dyes from aqueous solutions by adsorption of chitosan-based semi-IPN hydrogel composites. Compos. Part B 2012, 43, 1570–1578. [Google Scholar] [CrossRef]
- Vargas, A.M.; Cazetta, A.L.; Martins, A.C.; Moraes, J.C.; Garcia, E.E.; Gauze, G.F.; Costa, W.F.; Almeida, V.C. Kinetic and equilibrium studies: Adsorption of food dyes Acid Yellow 6, Acid Yellow 23, and Acid Red 18 on activated carbon from flamboyant pods. Chem. Eng. J. 2012, 181, 243–250. [Google Scholar] [CrossRef]
- Barylak, M.; Cendrowski, K.; Peruzynska, M.; Augustyniak, A.; Nawrotek, P.; Drozdzik, M.; Mijowska, E. Carbonized metal–organic frameworks with trapped cobalt nanoparticles as biocompatible and efficient azo-dye adsorbent. Environ. Sci. Eur. 2019, 31, 1–15. [Google Scholar]


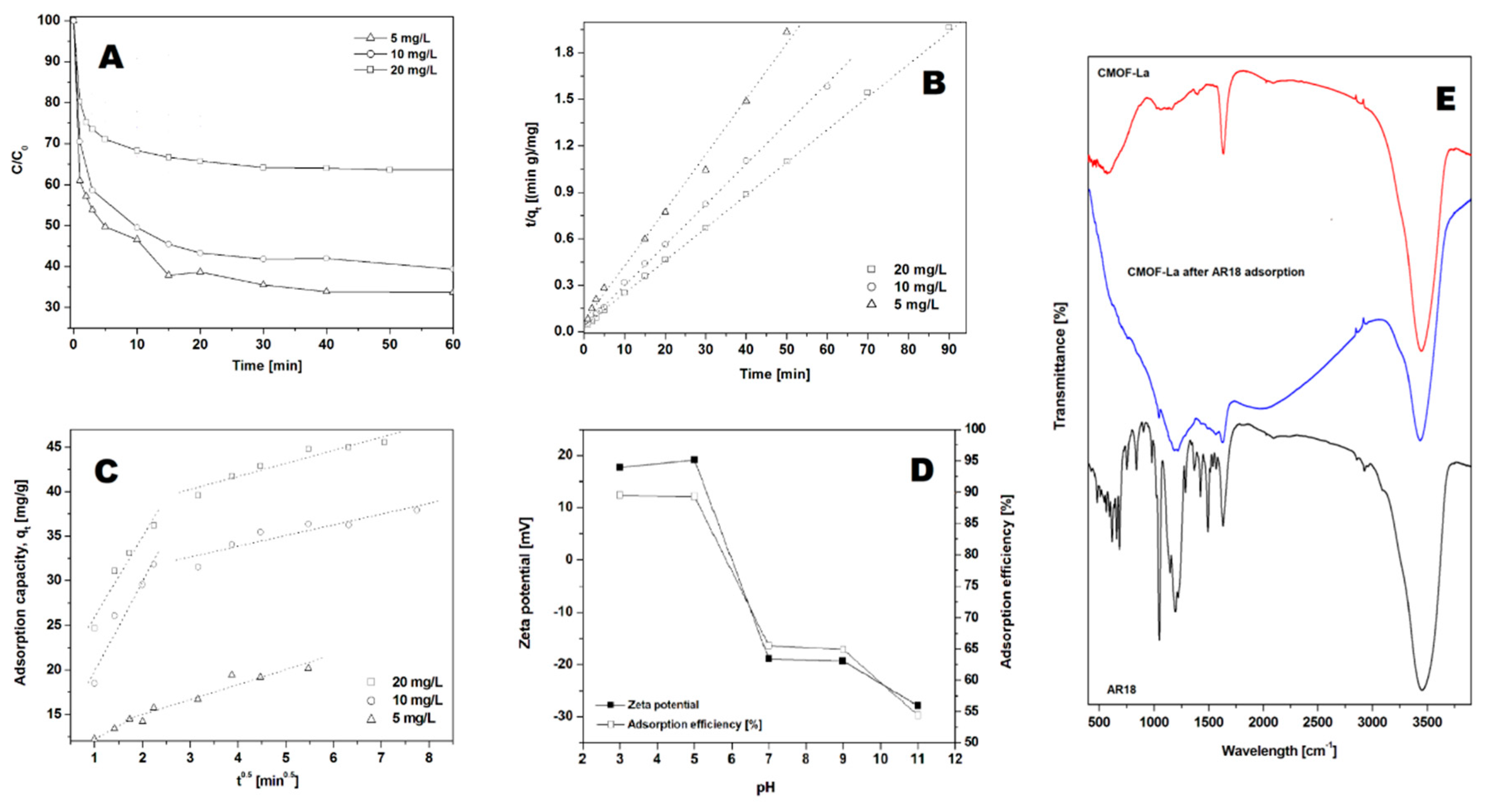
| C0 (mg/L) | Qe, Exp (mg/g) | Pseudo-First-Order | Pseudo-Second-Order | Intraparticle Diffusion Model | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| k1 (min−1) | qe (mg/g) | R2 | k2 (g/mg·min) | qe (mg/g) | R2 | Kp (mg/g min0.5) | C (mg/g) | R2 | ||
| 5 | 20.93 | 0.1365 | 24.34 | 0.9400 | 0.0122 | 27.86 | 0.9994 | 2.99 | 9.21 | 0.996 |
| 10 | 38.28 | 0.3777 | 29.39 | 0.9400 | 0.0183 | 38.27 | 0.9994 | 10.06 | 9.75 | 0.9363 |
| 20 | 47.32 | 0.4609 | 35.02 | 0.9450 | 0.0194 | 47.35 | 0.9944 | 9.01 | 16.88 | 0.9356 |
| Adsorbent Type | Adsorbent [mg] | C0 [mg/L] | Volume [mL] | Temperature [°C] | Time [min] | pH | Qe [mg/g] | Kinetic Model (R2) | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Carbonized Phragmites australis | 400 | 40–140 | 50 | 25 | 450 | 6.6 | 80.94 | Elovich (0.942) | [39] |
| polypyrrole-chitosan composites | 50 | 50 | 100 | 26 | 60 | 7.0 | 100.00 | Second-order (1.0000) | [40] |
| Activated carbon | 1000 | 300 | N/A | 30 | 240 | 7.0 | 29.30 | Second-order (0.998) | [42] |
| Chitosan/Carbon Nanotube | 50 | 300 | 20 | 29 | 120 | 7.0 | 691.0 | Second-order (0.985) | [4] |
| Ordered mesoporous TiO2/activated carbon | 150 | 80 | 250 | 25 | 30 | 7.0 | 138.9 | Second-order (0.996) | [47] |
| Semi-IPN hydrogel composites | 60 | 200 | 100 | 25 | 300 | 2.0 | 345.7 | Second-order (0.9996) | [48] |
| Activated carbon | 25 | 1000 | 25 | 25 | 150 | 7.0 | 599.821 | Second-order (0.987) | [49] |
| Carbonized cobalt based organic framework-CoOF | 10 | 100 | 10 | 22 | 90 | 7.0 | 43.309 | Second-order (1.000) | [50] |
| CMOF | 40 | 10 | 250 | 25 | 30 | 7.0 | 38.27 | Second-order (0.9993) | This work |
| CMOF | 40 | 20 | 250 | 25 | 30 | 7.0 | 47.35 | Second-order (0.9994) | This work |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cendrowski, K.; Opała, K.; Mijowska, E. Carbonized Lanthanum-Based Metal-Organic Framework with Parallel Arranged Channels for Azo-Dye Adsorption. Nanomaterials 2020, 10, 1053. https://doi.org/10.3390/nano10061053
Cendrowski K, Opała K, Mijowska E. Carbonized Lanthanum-Based Metal-Organic Framework with Parallel Arranged Channels for Azo-Dye Adsorption. Nanomaterials. 2020; 10(6):1053. https://doi.org/10.3390/nano10061053
Chicago/Turabian StyleCendrowski, Krzysztof, Karolina Opała, and Ewa Mijowska. 2020. "Carbonized Lanthanum-Based Metal-Organic Framework with Parallel Arranged Channels for Azo-Dye Adsorption" Nanomaterials 10, no. 6: 1053. https://doi.org/10.3390/nano10061053
APA StyleCendrowski, K., Opała, K., & Mijowska, E. (2020). Carbonized Lanthanum-Based Metal-Organic Framework with Parallel Arranged Channels for Azo-Dye Adsorption. Nanomaterials, 10(6), 1053. https://doi.org/10.3390/nano10061053





