3D Porous Ti3C2 MXene/NiCo-MOF Composites for Enhanced Lithium Storage
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Synthesis of Ti3C2 MXene Nanosheets
2.3. Synthesis of Ultrathin NiCo-MOF Nanosheets
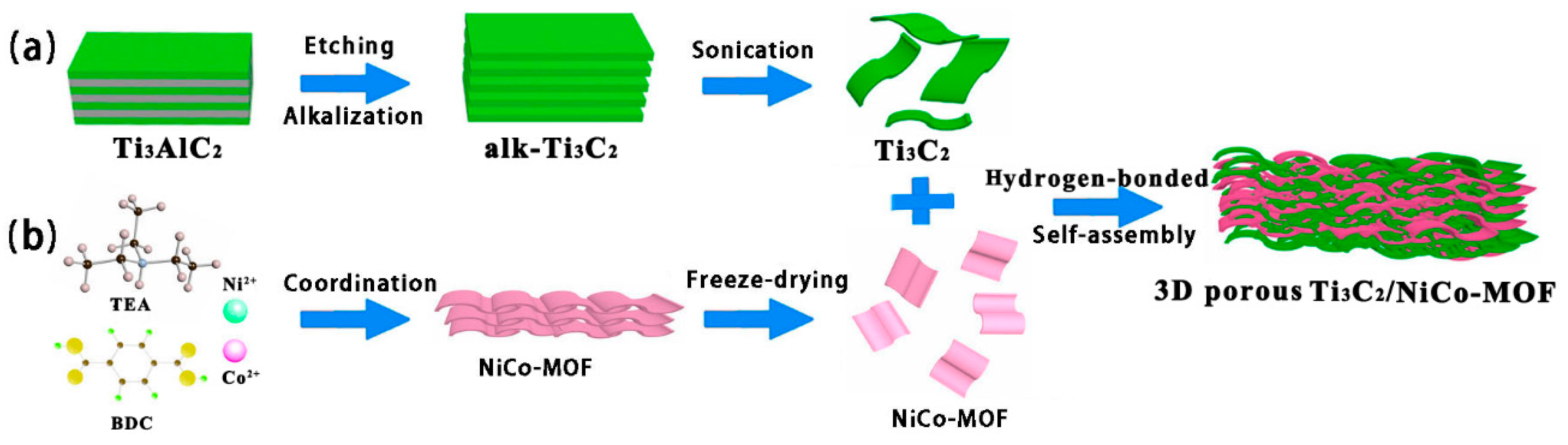
2.4. Fabrication of 3D Porous Ti3C2/NiCo-MOF Composites
2.5. Material Characterization
2.6. Electrochemical Measurements
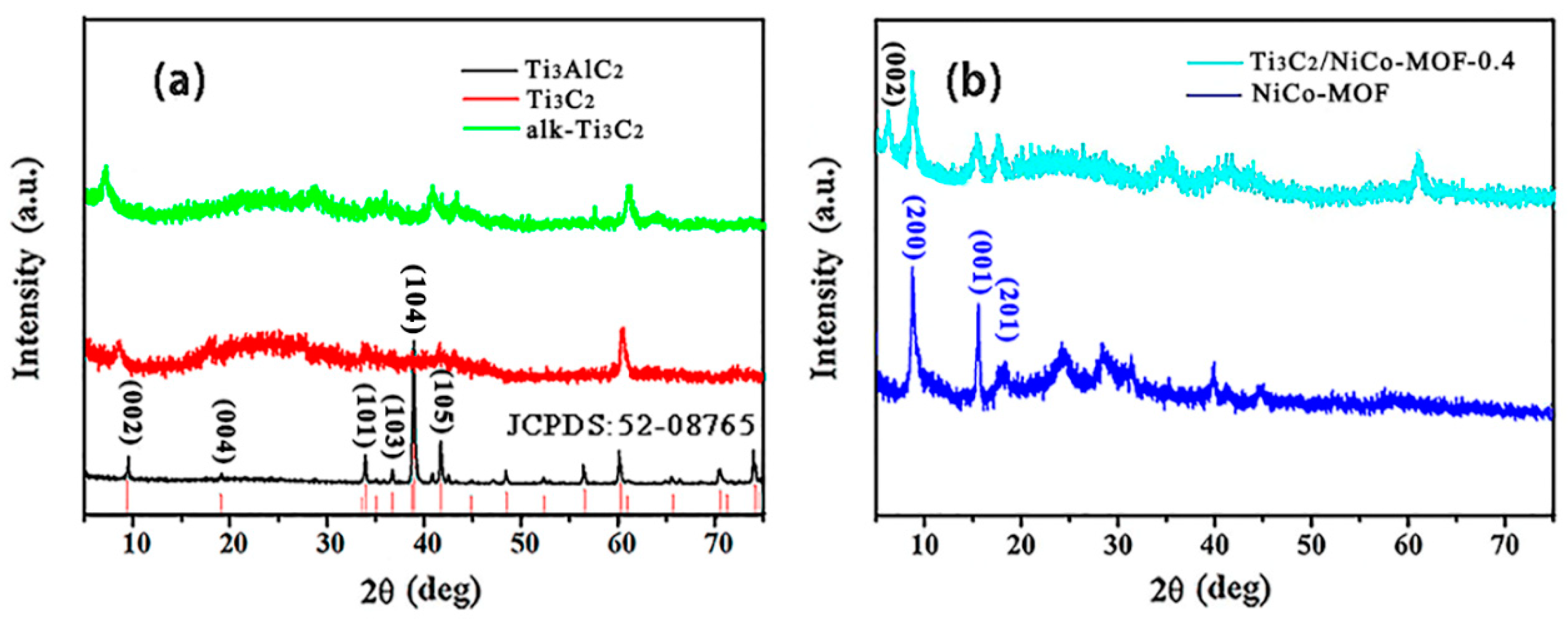
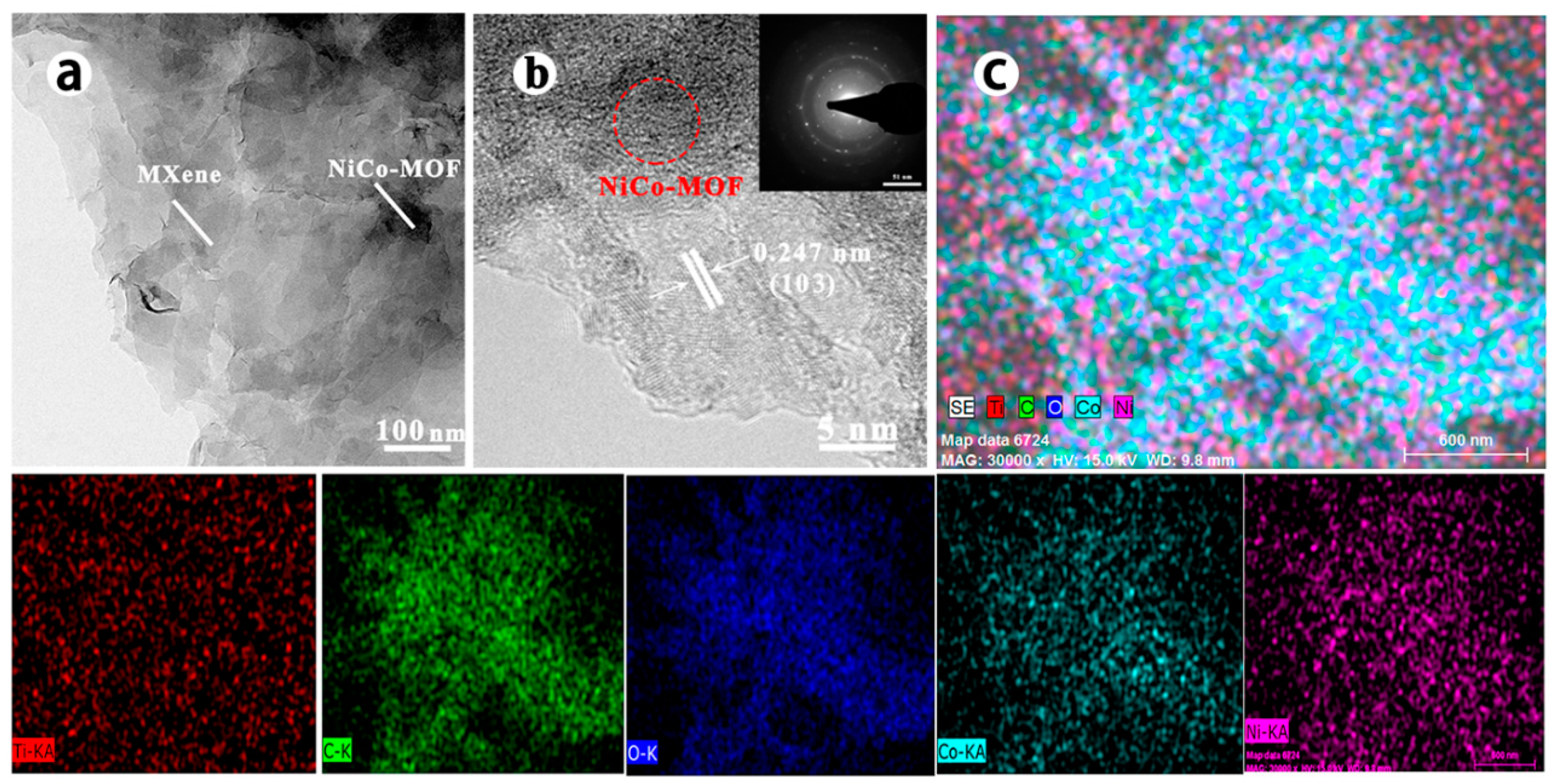
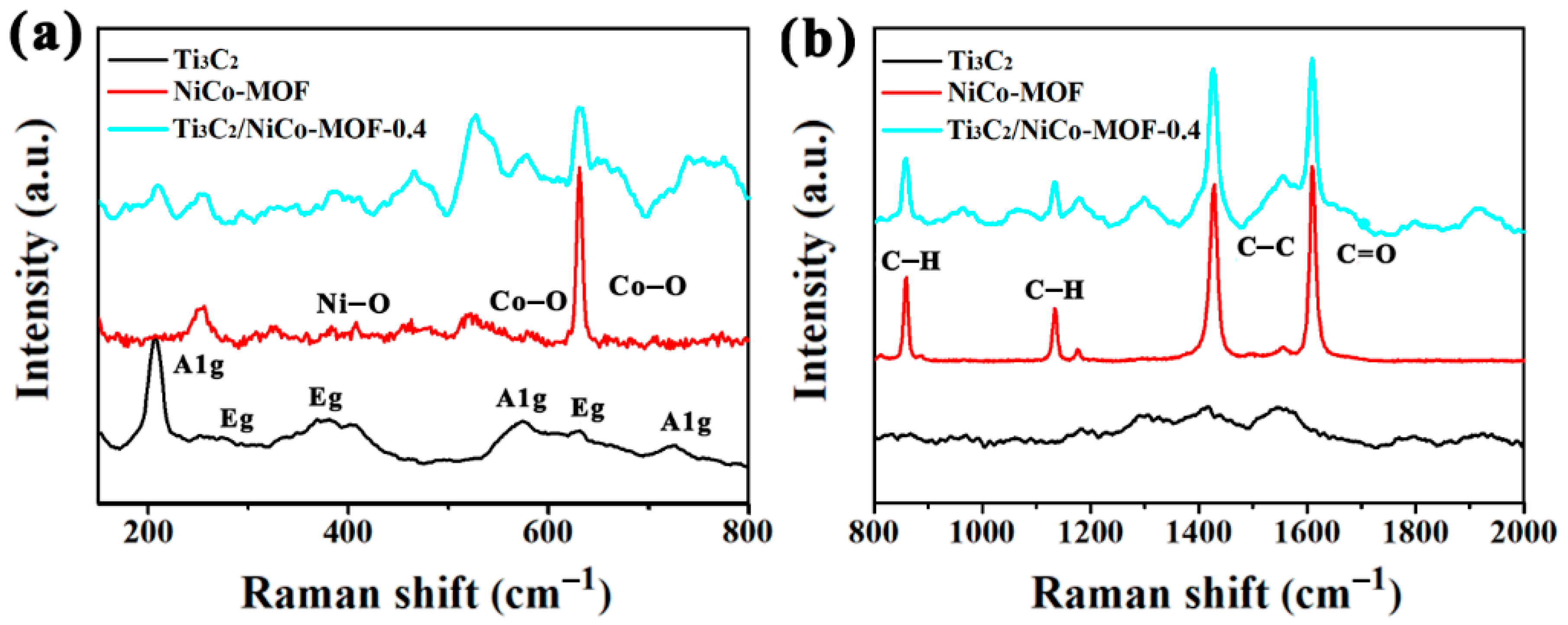
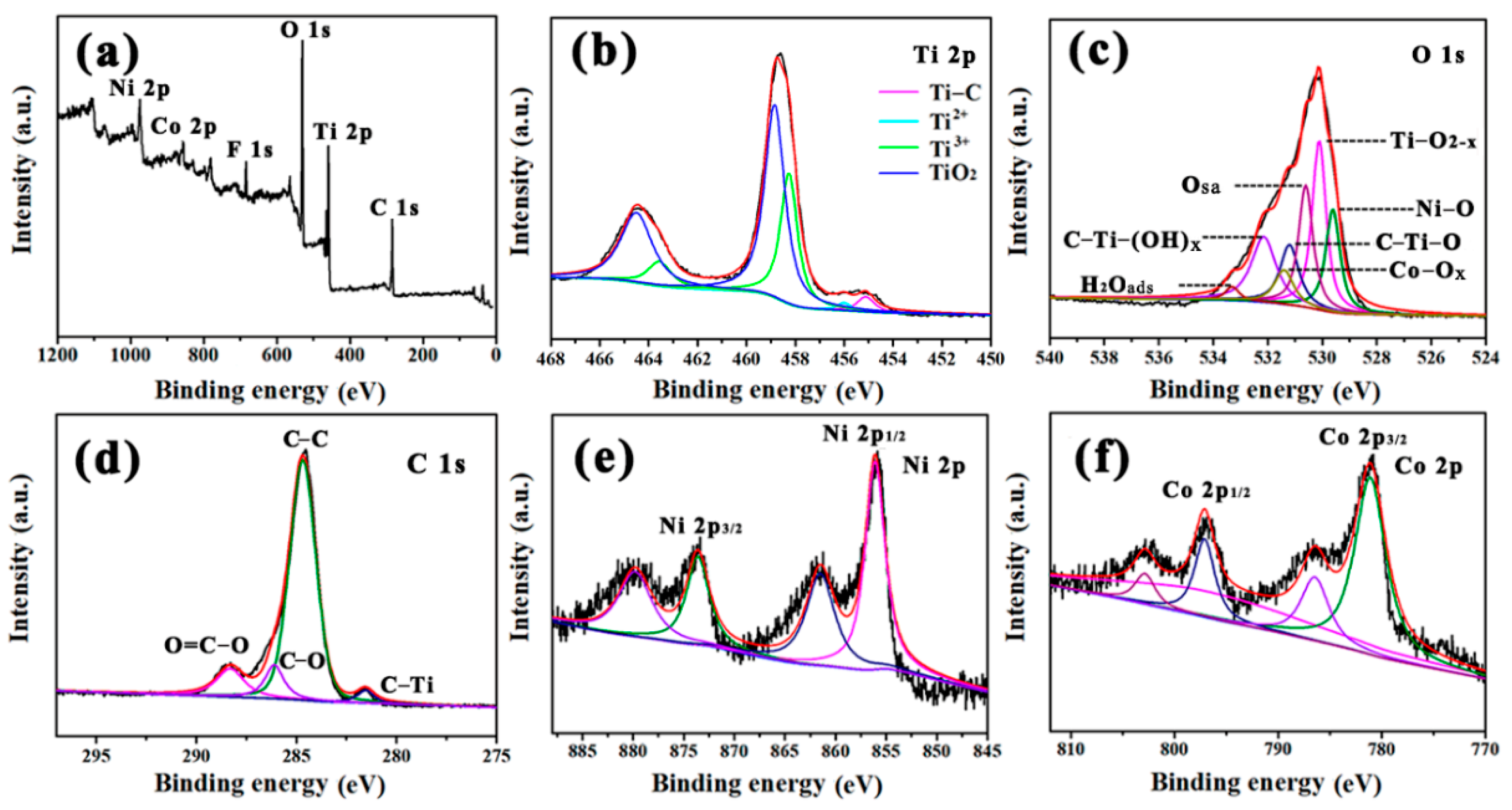
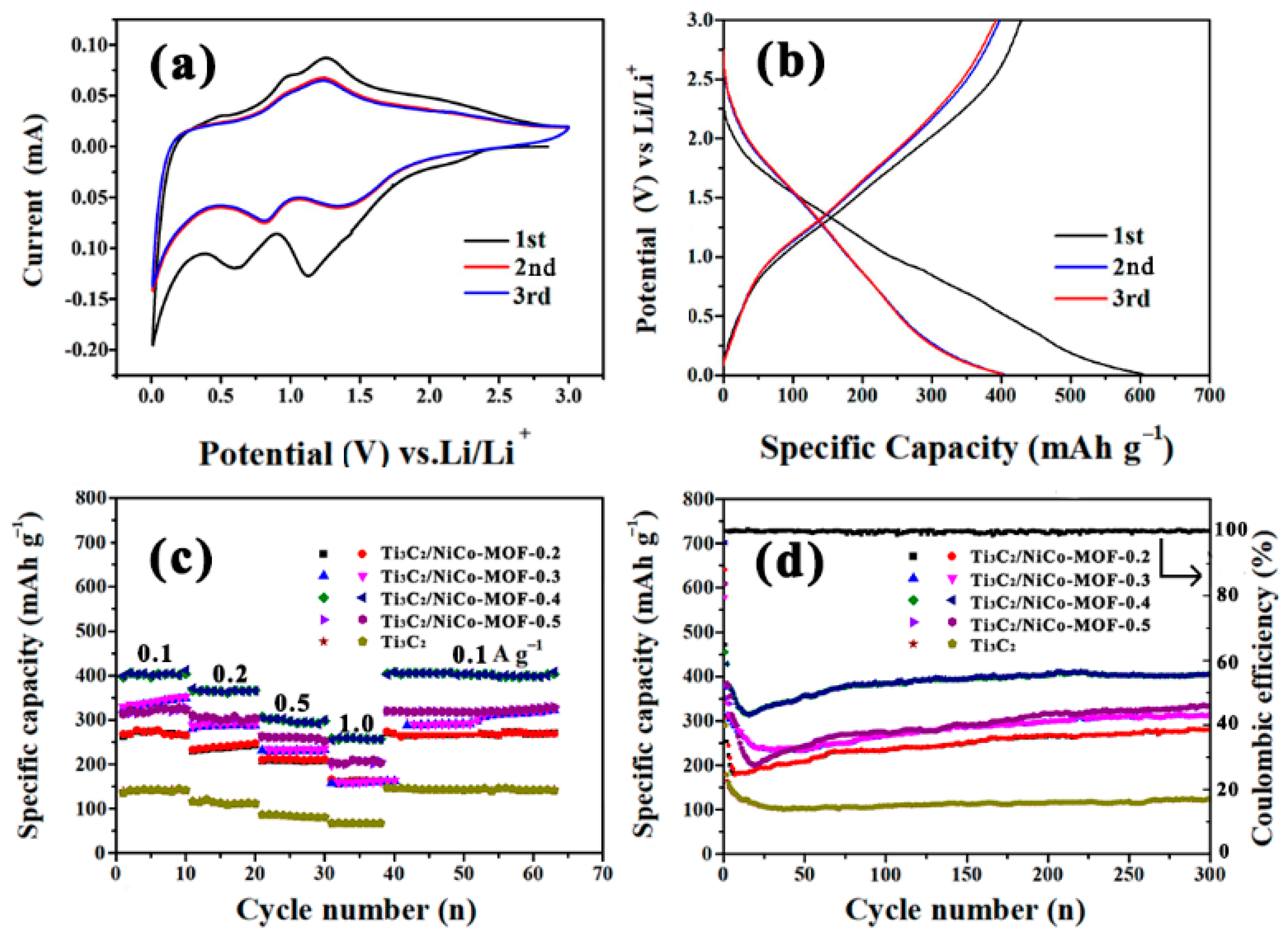
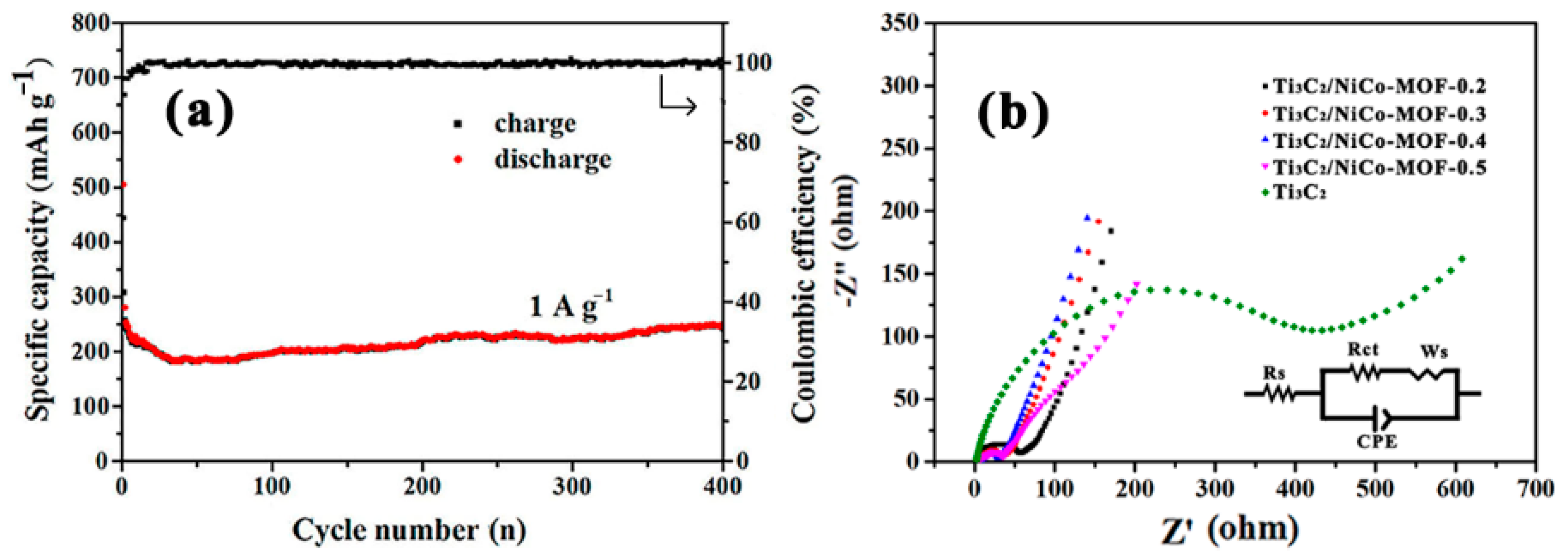
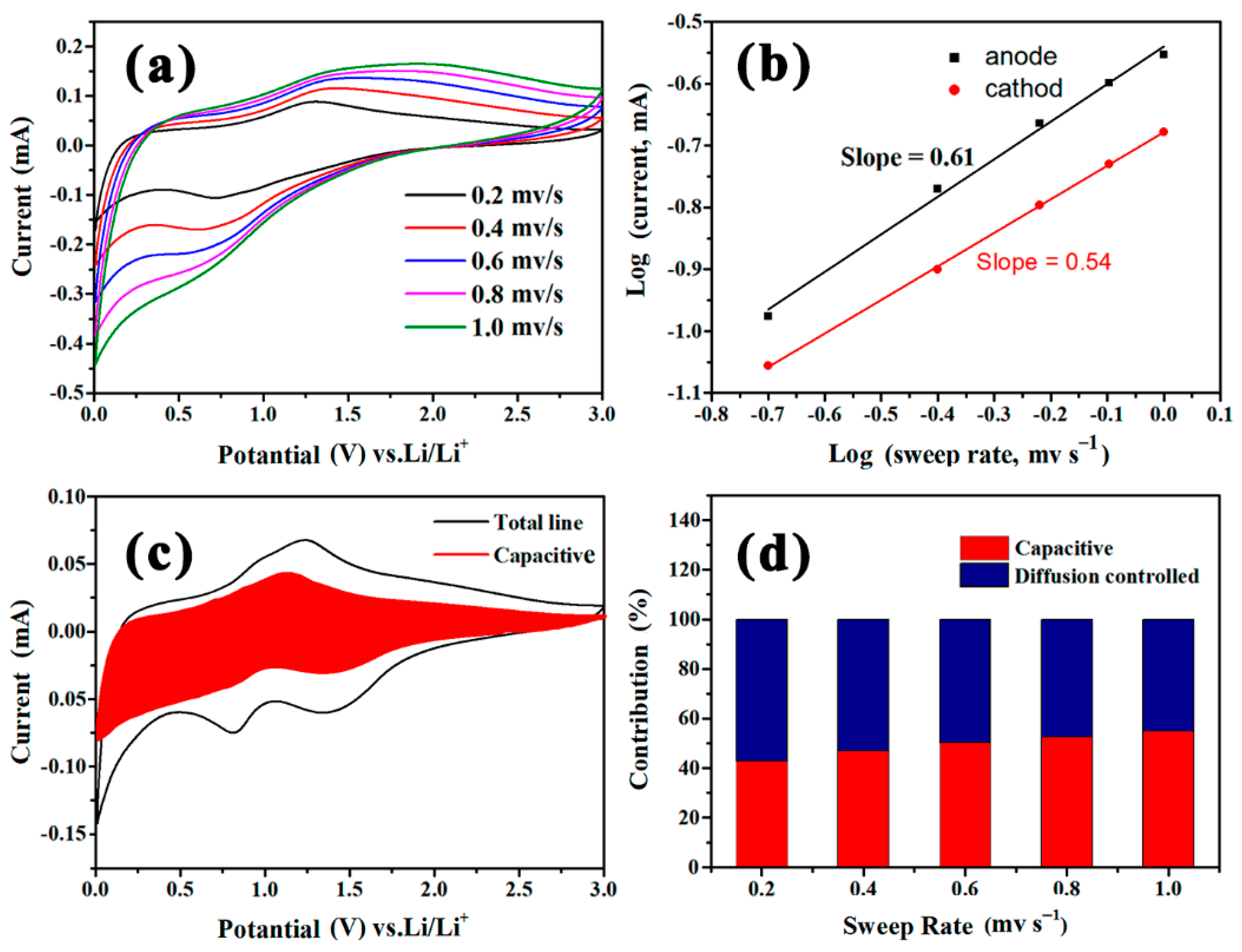
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Simon, P.; Gogotsi, Y.; Dunn, B. Where do batteries end and supercapacitors begain. Science 2014, 343, 1210–1211. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Zhu, C.; Li, H.; Yu, Y.; Li, C.; Tsukimoto, S.; Maier, J.; Ikuhara, Y. Direct observation of lithium staging in partially delithiated LiFePO4 at atomic resolution. J. Am. Chem. Soc. 2011, 133, 4661–4663. [Google Scholar] [CrossRef]
- Dubal, D.P.; Ayyad, O.; Ruiz, V.; Gomez, R.P. Hybrid energy storage: The merging of battery and supercapacitor chemistries. Chem. Soc. Rev. 2015, 44, 1777–1790. [Google Scholar] [CrossRef] [PubMed]
- Pourali, Z.; Sovizi, M.R.; Yaftian, M.R. Two-dimensional Ti3C2TX/CMK-5 nanocomposite as high performance anodes for lithium batteries. J. Alloys Compd. 2018, 738, 130–137. [Google Scholar] [CrossRef]
- Anasori, B.; Lukatskaya, M.R.; Gogotsi, Y. 2D metal carbides and nitrides (MXenes) for energy storage. Nat. Rev. Mater. 2017, 2, 16098. [Google Scholar] [CrossRef]
- Naguib, M.; Kurtoglu, M.; Presser, V.; Lu, J.; Niu, J.; Heon, M.; Hultman, L.; Gogotsi, Y.; Barsoum, M.W. Two-dimensional nanocrystals produced by exfoliation of Ti3AlC2. Adv. Mater. 2011, 23, 4248–4253. [Google Scholar] [CrossRef]
- Liu, L.; Zhao, Q.; Liu, R.; Zhu, L. Hydrogen adsorption-induced catalytic enhancement over Cu nanoparticles immobilized by layered Ti3C2 MXene. Appl. Catal. B Environ. 2019, 252, 198–204. [Google Scholar] [CrossRef]
- Kim, S.J.; Koh, H.J.; Ren, C.E.; Kwon, O.; Maleski, K.; Cho, S.Y.; Anasori, B.; Kim, C.K.; Choi, Y.K.; Kim, J.; et al. Metallic Ti3C2Tx MXene gas sensors with ultrahigh signal-to-noise ratio. ACS Nano 2018, 12, 986–993. [Google Scholar] [CrossRef]
- Wen, Y.; Rufford, T.E.; Chen, X.; Li, N.; Lyu, M.; Dai, L.; Wang, L. Nitrogen-doped Ti3C2Tx MXene electrodes for high-performance supercapacitors. Nano Energy 2017, 38, 368–376. [Google Scholar] [CrossRef]
- Zhu, M.; Huang, Y.; Deng, Q.; Zhou, J.; Pei, Z.; Xue, Q.; Huang, Y.; Wang, Z.; Li, H.; Huang, Q.; et al. Highly flexible, freestanding supercapacitor electrode with enhanced performance obtained by hybridizing polypyrrole chains with MXene. Adv. Energy Mater. 2016, 6, 1600969. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, H.; Zhang, X.; Xu, Y.; Fransaer, J. Cu2O hybridized titanium carbide with open conductive frameworks for lithium-ion batteries. Electrochim. Acta 2016, 202, 24–31. [Google Scholar] [CrossRef]
- Zheng, M.; Guo, R.; Liu, Z.; Wang, B.; Meng, L.; Li, F.; Li, T.; Luo, Y. MoS2 intercalated p-Ti3C2 anode materials with sandwich-like three dimensional conductive networks for lithium-ion batteries. J. Alloys Compd. 2018, 735, 1262–1270. [Google Scholar] [CrossRef]
- Zhan, X.; Si, C.; Zhou, J.; Sun, Z. MXene and MXene-based composites: Synthesis, properties and environment-related applications. Nanoscale Horiz. 2020, 5, 235–258. [Google Scholar] [CrossRef]
- Shen, C.; Wang, L.; Zhou, A.; Wang, B.; Wang, X.; Lian, W.; Hu, Q.; Qin, G.; Liu, X. Synthesis and electrochemical properties of two-dimensional RGO/Ti3C2Tx nanocomposites. Nanomaterials 2018, 8, 80. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mu, Z.; Yang, C.; Xu, Z.; Zhang, S.; Zhang, X.; Li, Y.; Lai, J.; Sun, Z.; Yang, Y.; et al. Rational design of MXene/1T-2H MoS2-C nanohybrids for high-performance lithium-sulfur batteries. Adv. Funct. Mater. 2018, 28, 1707578. [Google Scholar] [CrossRef]
- Huang, J.; Meng, R.; Zu, L.; Wang, Z.; Feng, N.; Yang, Z.; Yu, Y.; Yang, J. Sandwich-like Na0.23TiO2 nanobelt/Ti3C2 MXene composites from a scalable in situ transformation reaction for long-life high-rate lithium/sodium-ion batteries. Nano Energy 2018, 46, 20–28. [Google Scholar] [CrossRef]
- Ma, Y.; Yue, Y.; Zhang, H.; Cheng, F.; Zhao, W.; Rao, J.; Luo, S.; Wang, J.; Jiang, X.; Liu, Z.; et al. 3D synergistical MXene/reduced graphene oxide aerogel for a piezoresistive sensor. ACS Nano 2018, 12, 3209–3216. [Google Scholar] [CrossRef]
- Xie, X.; Zhao, M.Q.; Anasori, B.; Maleski, K.; Ren, C.E.; Li, J.; Byles, B.W.; Pomerantseva, E.; Wang, G.; Gogotsi, Y. Porous heterostructured MXene/carbon nanotube composite paper with high volumetric capacity for sodium-based energy storage devices. Nano Energy 2016, 26, 513–523. [Google Scholar] [CrossRef]
- Ma, Z.Y.; Zhou, X.F.; Deng, W.; Lei, D.; Liu, Z.P. 3D porous MXene (Ti3C2)/reduced graphene oxide hybrid films for advanced lithium storage. ACS Appl. Mater. Interfaces 2018, 10, 3634–3643. [Google Scholar] [CrossRef]
- Fan, Z.; Wang, Y.; Xie, Z.; Xu, X.; Yuan, Y.; Cheng, Z.; Liu, Y. Nanoporous MXene film enabled flexible supercapacitor with high energy storage. Nanoscale 2018, 10, 9642–9652. [Google Scholar] [CrossRef]
- Li, X.; Zhu, J.; Fang, Y.; Lv, W.; Wang, F.; Liu, Y.; Liu, H. Hydrothermal preparation of CoO/Ti3C2 composite material for lithium-ion batteries with enhanced electrochemical performance. J. Electroanal. Chem. 2018, 817, 1–8. [Google Scholar] [CrossRef]
- Zhang, H.B.; Nai, J.W.; Yu, L.; Lou, X.W. Metal-organic-framework-based materials as platforms for renewable energy and environmental applications. Joule 2017, 1, 77–107. [Google Scholar] [CrossRef]
- Wang, H.L.; Zhu, Q.L.; Zou, R.Q.; Xu, Q. Metal-organic frameworks for energy applications. Chem 2017, 1, 52–80. [Google Scholar] [CrossRef]
- Shrivastav, V.; Sundriyal, S.; Goel, P.; Kaur, H.; Tuteja, K.T.; Vikrant, K.; Kim, K.H.; Tiwari, U.K.; Deep, A. Metal-organic frameworks (MOFs) and their composites as electrodes for lithium battery applications: Novel means for alternative energy storage. Coord. Chem. Rev. 2019, 393, 48–78. [Google Scholar] [CrossRef]
- Gou, L.; Hao, L.M.; Shi, Y.X.; Ma, S.L.; Fan, X.Y.; Xu, L.; Li, D.L.; Wang, K. One-pot synthesis of a metal–organic framework as an anode for Li-ion batteries with improved capacity and cycling stability. J. Solid State Chem. 2014, 210, 121–124. [Google Scholar] [CrossRef]
- Zhao, S.L.; Wang, Y.; Dong, J.; He, C.T.; Yin, H.; An, P.; Zhao, K.; Zhang, X.; Gao, C.; Zhang, L.; et al. Ultrathin metal–organic framework nanosheets for electrocatalytic oxygen evolution. Nat. Energy 2016, 1, 16184. [Google Scholar] [CrossRef]
- Simon, P. Two-dimensional MXene with controlled interlayer spacing for electrochemical energy storage. ACS Nano 2017, 11, 2393–2396. [Google Scholar] [CrossRef]
- Luo, J.; Tao, X.; Zhang, J.; Xia, Y.; Huang, H.; Zhang, L.; Gan, Y.; Liang, C.; Zhang, W. Sn4+ ion decorated highly conductive Ti3C2 MXene: Promising lithium-ion anodes with enhanced volumetric capacity and cyclic performance. ACS Nano 2016, 10, 2491–2499. [Google Scholar] [CrossRef]
- Lukatskaya, M.R.; Mashtalir, O.; Ren, C.E.; Dall’Agnese, Y.; Rozier, P.; Taberna, P.L.; Naguib, M.; Simon, P.; Barsoum, M.W.; Gogotsi, Y. Cation intercalation and high volumetric capacitance of two-dimensional titanium carbide. Science 2013, 341, 1502−1505. [Google Scholar] [CrossRef]
- Han, B.; Ou, X.; Deng, Z.; Song, Y.; Tian, C.; Deng, H.; Xu, Y.J.; Lin, Z. Nickel metal-organic framework monolayers for photoreduction of diluted CO2: Metal-node-dependent activity and selectivity. Angew. Chem. Int. Ed. Engl. 2018, 57, 16811–16815. [Google Scholar] [CrossRef]
- Liu, Y.X.; Wang, Y.Z.; Wang, H.Q.; Zhao, P.H.; Hou, H.; Guo, L. Acetylene black enhancing the electrochemical performance of NiCo-MOF nanosheets for supercapacitor electrodes. Appl. Surf. Sci. 2019, 492, 455–463. [Google Scholar] [CrossRef]
- Hu, T.; Wang, J.; Zhang, H.; Li, Z.; Hu, M.; Wang, X. Vibrational properties of Ti3C2 and Ti3C2T2 (T = O, F, OH) monosheets by first-principles calculations: A comparative study. Phys. Chem. Chem. Phys. 2015, 17, 9997–10003. [Google Scholar] [CrossRef]
- Zhao, B.; Song, J.; Liu, P.; Xu, W.; Fang, T.; Jiao, Z.; Zhang, H.; Jiang, Y. Monolayer graphene/NiO nanosheets with two-dimension structure for supercapacitors. J. Mater. Chem. 2011, 21, 18792–18798. [Google Scholar] [CrossRef]
- Victoria, S.G.; Ezhil Raj, A.M.; Ravidhas, C. An insight in the structural, morphological, electrical and optical properties of spray pyrolysed Co3O4 thin films. Mater. Chem. Phys. 2015, 162, 852–859. [Google Scholar] [CrossRef]
- Hu, Y.H.; Zhang, L. Amorphization of metal-organic framework MOF-5 at unusually low applied pressure. Phys. Rev. B 2010, 81, 174103. [Google Scholar] [CrossRef]
- Niu, S.; Wang, Z.; Yu, M.; Yu, M.; Xiu, L.; Wang, S.; Wu, X.; Qiu, J. MXene-based electrode with enhanced pseudocapacitance and volumetric capacity for power-type and ultra-long life lithium storage. ACS Nano 2018, 12, 3928–3937. [Google Scholar] [CrossRef]
- Halim, J.; Cook, K.M.; Naguib, M.; Eklund, P.; Gogotsi, Y.; Rosen, J.; Barsoum, M.W. X-ray photoelectron spectroscopy of select multi-layered transition metal carbides (MXenes). Appl. Surf. Sci. 2016, 362, 406–417. [Google Scholar] [CrossRef]
- Liu, H.; Wang, X.; Xu, H.; Yu, W.; Dong, X.; Yang, Y.; Zhang, H.; Wang, J. Nanostructured CoO/NiO/CoNi anodes with tunable morphology for high performance lithium-ion batteries. Dalton Trans. 2017, 46, 11031–11036. [Google Scholar] [CrossRef]
- Zou, R.; Xu, K.; Wang, T.; He, G.; Liu, Q.; Liu, X.; Zhang, Z.; Hu, J. Chain-like NiCo2O4 nanowires with different exposed reactive planes for high-performance supercapacitors. J. Mater. Chem. A 2013, 1, 8560–8566. [Google Scholar] [CrossRef]
- Jiang, J.; Zhang, A.; Li, L.; Ai, L. Nickel–cobalt layered double hydroxide nanosheets as high-performance electrocatalyst for oxygen evolution reaction. J. Power Sources 2015, 278, 445–451. [Google Scholar] [CrossRef]
- Sun, D.; Wang, M.; Li, Z.; Fan, G.; Fan, L.Z.; Zhou, A. Two-dimensional Ti3C2 as anode material for Li-ion batteries. Electrochem. Commun. 2014, 47, 80–83. [Google Scholar] [CrossRef]
- Sun, H.; Xin, G.; Hu, T.; Yu, M.; Shao, D.; Sun, X.; Lian, J. High-rate lithiation-induced reactivation of mesoporous hollow spheres for long-lived lithium-ion batteries. Nat. Commun. 2014, 5, 4526. [Google Scholar] [CrossRef]
- Zuo, D.C.; Song, S.C.; An, C.S.; Tang, L.B.; He, Z.J.; Zheng, J.C. Synthesis of sandwich-like structured Sn/SnOx@MXene composite through in-situ growth for highly reversible lithium storage. Nano Energy 2019, 62, 401–409. [Google Scholar] [CrossRef]
- Muller, G.A.; Cook, J.B.; Kim, H.S.; Tolbert, S.H.; Dunn, B. High performance pseudocapacitor based on 2D layered metal chalcogenide nanocrystals. Nano Lett. 2015, 15, 1911–1917. [Google Scholar] [CrossRef]
- Augustyn, V.; Come, J.; Lowe, M.A.; Kim, J.W.; Taberna, P.L.; Tolbert, S.H.; Abruna, H.D.; Simon, P.; Dunn, B. High-rate electrochemical energy storage through Li+ intercalation pseudocapacitance. Nat. Mater. 2013, 12, 518–522. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; He, Y.; Vargun, E.; Plachy, T.; Saha, P.; Cheng, Q. 3D Porous Ti3C2 MXene/NiCo-MOF Composites for Enhanced Lithium Storage. Nanomaterials 2020, 10, 695. https://doi.org/10.3390/nano10040695
Liu Y, He Y, Vargun E, Plachy T, Saha P, Cheng Q. 3D Porous Ti3C2 MXene/NiCo-MOF Composites for Enhanced Lithium Storage. Nanomaterials. 2020; 10(4):695. https://doi.org/10.3390/nano10040695
Chicago/Turabian StyleLiu, Yijun, Ying He, Elif Vargun, Tomas Plachy, Petr Saha, and Qilin Cheng. 2020. "3D Porous Ti3C2 MXene/NiCo-MOF Composites for Enhanced Lithium Storage" Nanomaterials 10, no. 4: 695. https://doi.org/10.3390/nano10040695
APA StyleLiu, Y., He, Y., Vargun, E., Plachy, T., Saha, P., & Cheng, Q. (2020). 3D Porous Ti3C2 MXene/NiCo-MOF Composites for Enhanced Lithium Storage. Nanomaterials, 10(4), 695. https://doi.org/10.3390/nano10040695





