Ultrasound-Assisted Hydrazine Reduction Method for the Preparation of Nickel Nanoparticles, Physicochemical Characterization and Catalytic Application in Suzuki-Miyaura Cross-Coupling Reaction
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. The Ways of Ni Nanoparticle Syntheses
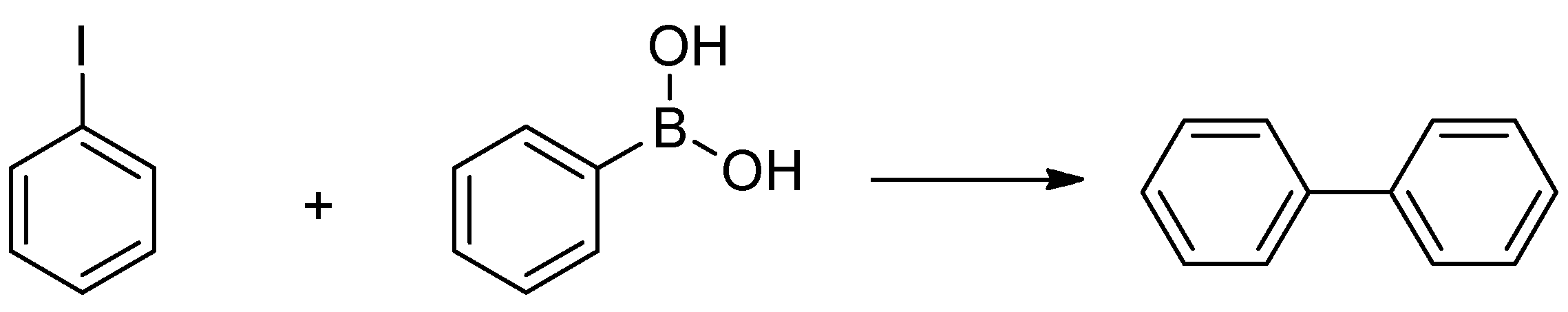
2.3. Procedure of the Suzuki-Miyaura Cross-Coupling Reactions
2.4. Structural Characterization by Instrumental Methods
3. Results and Discussion
3.1. Effects of the Sonication on the Physicochemical Properties of the Nickel Nanoparticles
3.2. Analysis of the Aggregation Tendency of the Nanoparticles
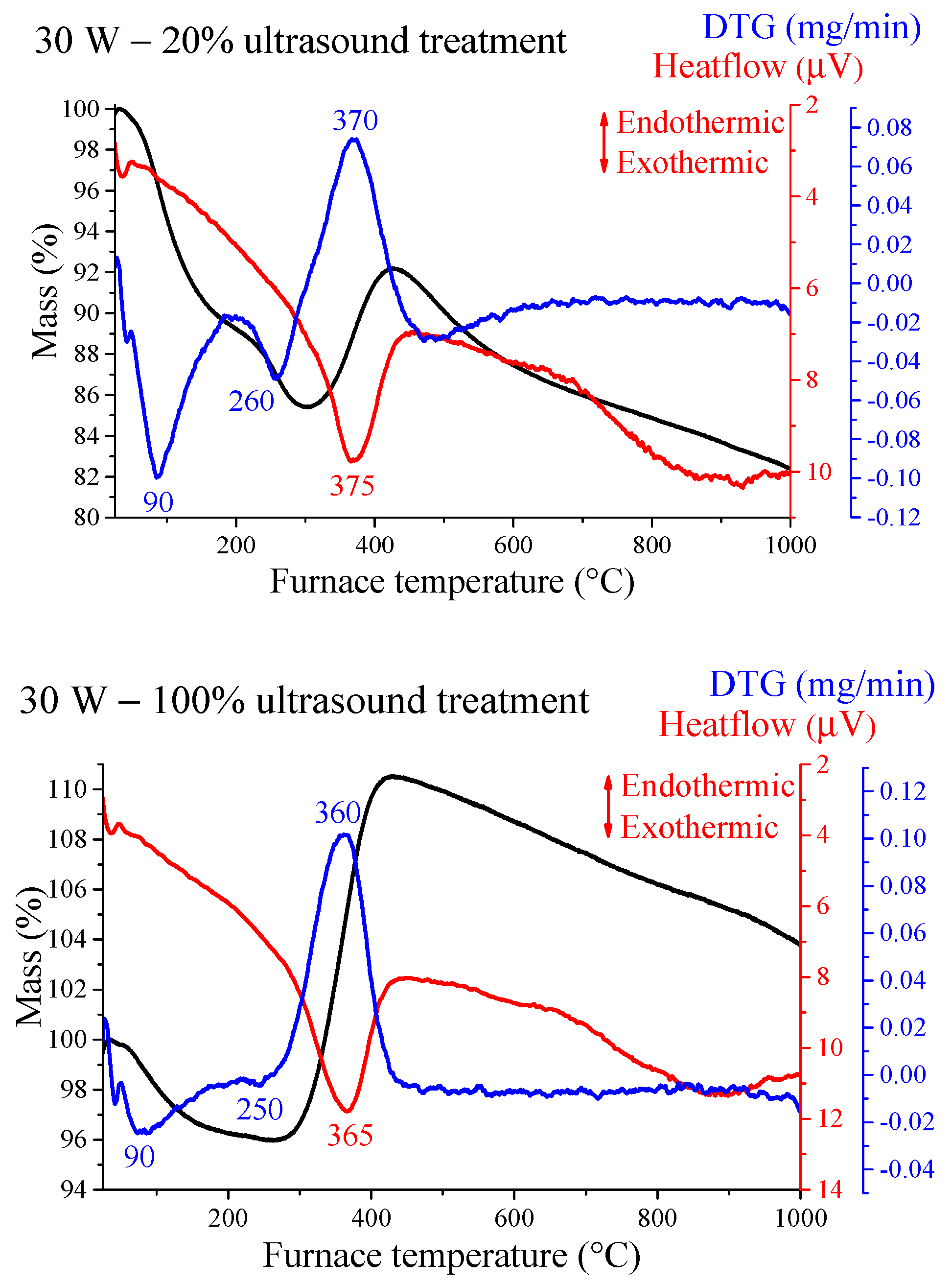
3.3. Thermal Properties of the Nickel Nanoparticles
3.4. Surface and Porous Properties of the Nickel Nanoparticles
3.5. CO2/NH3 Temperate Programmed Desorption (TPD) Studies of the Selected Samples
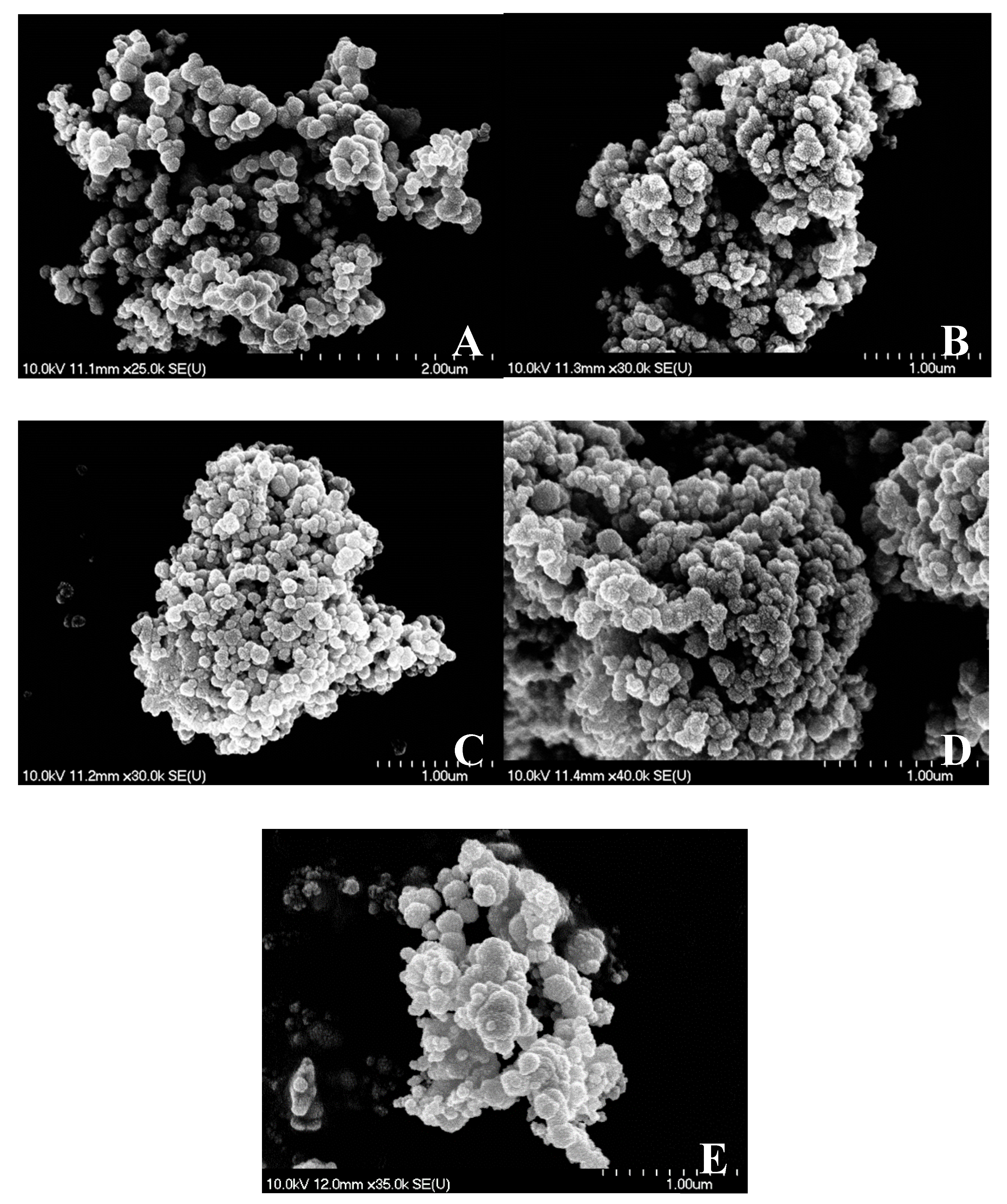
3.6. The selected Materials Studied by Scanning Electron Microscopy
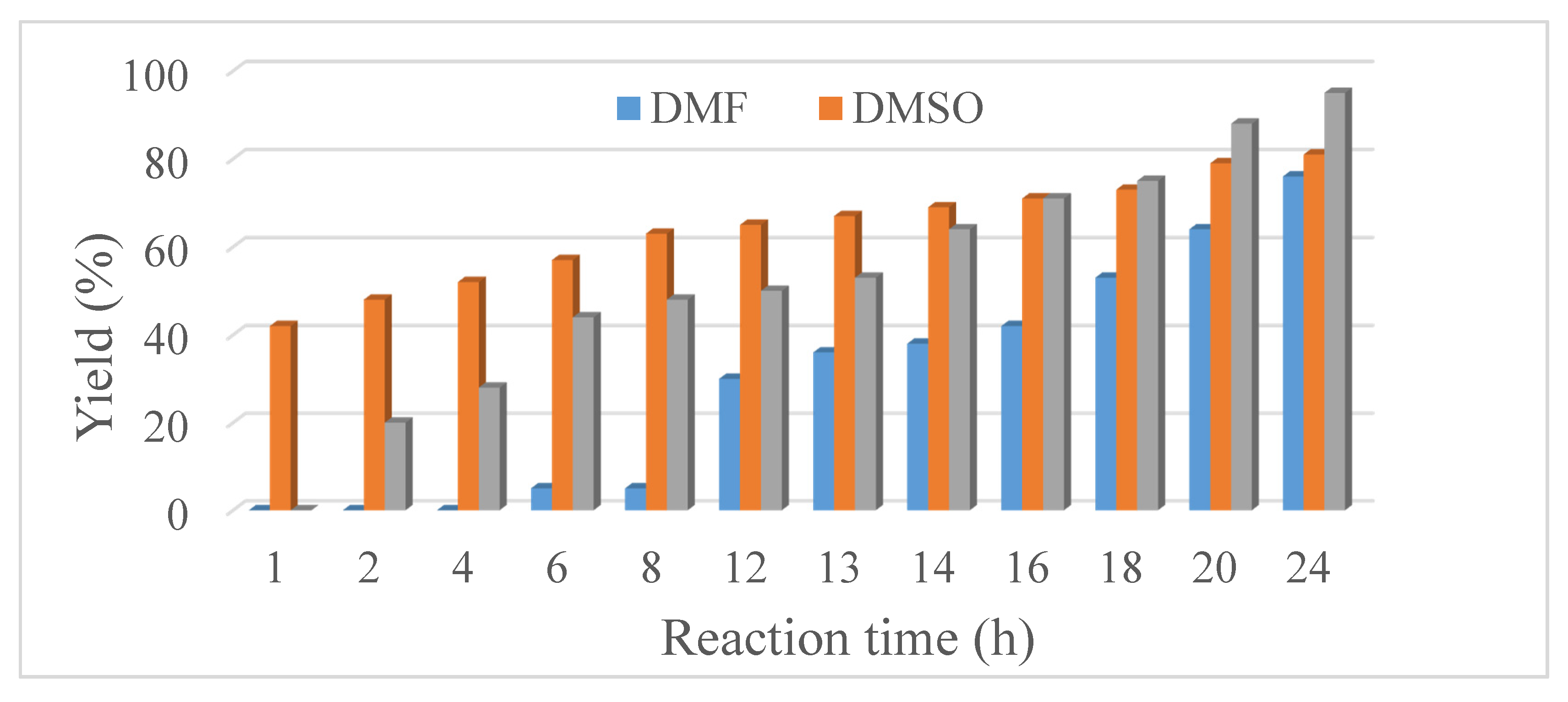
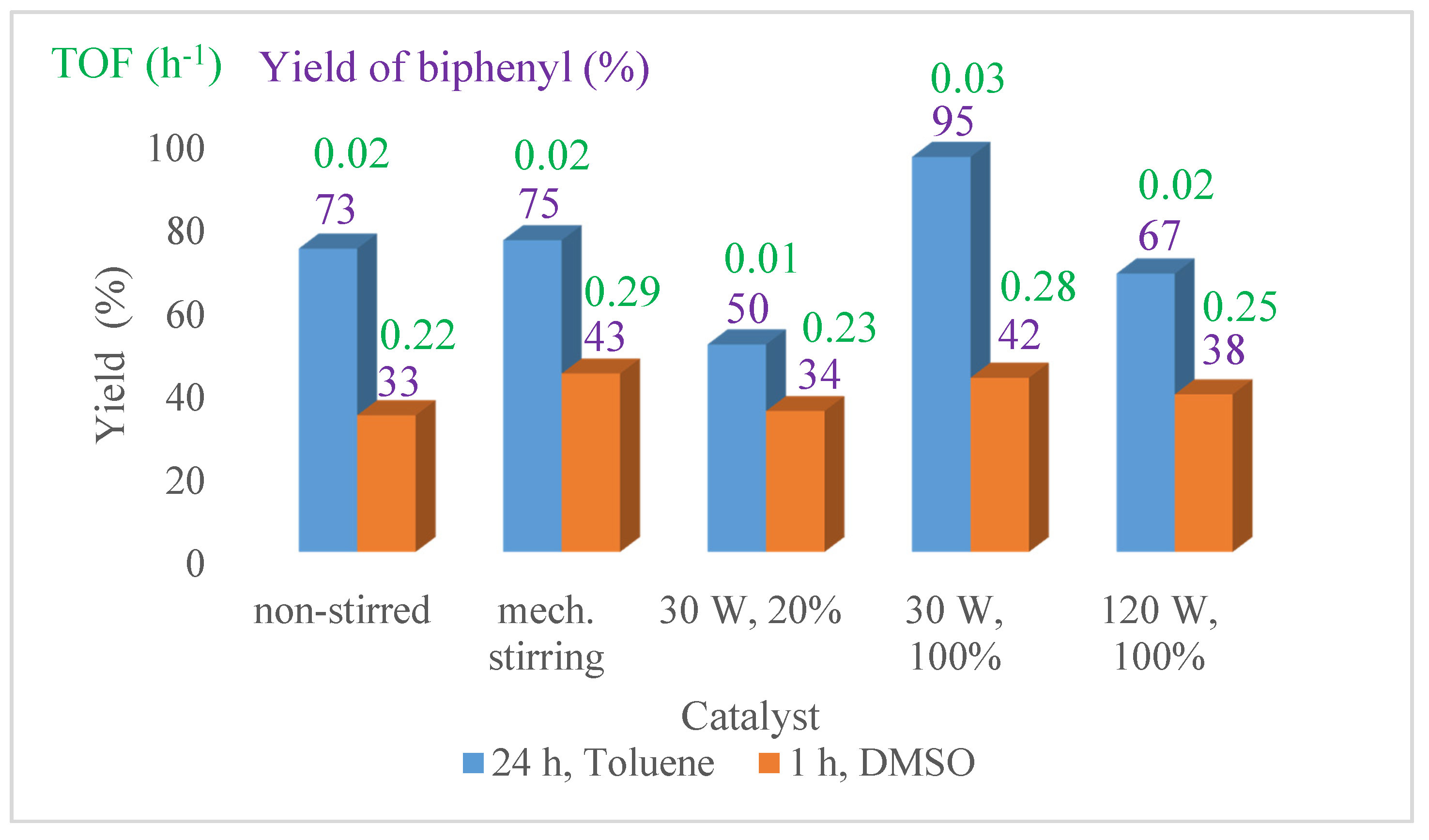
4. Catalytic Application of the Ni NPs in the Suzuki-Miyaura Cross-Coupling Reaction
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Campelo, J.M.; Luna, D.; Luque, R.; Marinas, J.M.; Romero, A.A. Sustainable preparation of supported metal nanoparticles and their applications in catalysis. ChemSusChem 2009, 2, 18–45. [Google Scholar] [CrossRef] [PubMed]
- Schmid, G.; Maihack, V.; Lantermann, F.; Peschel, S. Ligand-stabilized metal clusters and colloids: Properties and applications. J. Chem. Soc. Dalton Trans. 1996, 589–595. [Google Scholar] [CrossRef]
- Huang, H.H.; Yan, F.Q.; Kek, Y.M.; Chew, C.H.; Xu, G.Q.; Ji, W.; Oh, P.S.; Tang, S.H. Synthesis, characterization and nonlinear optical properties of copper nanoparticles. Langmuir 1997, 13, 172–175. [Google Scholar] [CrossRef]
- Fu, H.-B.; Yao, J.-N. Size effect on the optical properties of organic nanoparticles. J. Am. Chem. Soc. 2011, 123, 1434–1439. [Google Scholar] [CrossRef]
- Park, T.-J.; Papaefthymiou, G.C.; Viescas, A.J.; Moodenbaugh, A.R.; Wong, S.S. Size-dependent magnetic properties of single-crystalline multiferroic BiFeO3 nanoparticles. Nano Lett. 2007, 7, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Seo, W.S.; Jo, H.H.; Lee, K.; Kim, B.; Oh, S.J.; Park, J.T. Size-dependent magnetic properties of colloidal Mn3O4 and MnO nanoparticles. Angew. Chem. Int. Ed. 2004, 43, 1115–1117. [Google Scholar] [CrossRef]
- Jagiello, K.; Chomicz, B.; Avramopoulos, A.; Gajawicz, A.; Mikolajczyk, A.; Bonifassi, P.; Papadopoulos, M.P.; Leszczynski, J.; Puzyn, T. Size-dependent electronic properties of nanomaterials: How this novel class on nanodescriptors supposed to be calculated? Struct. Chem. 2017, 28, 635–643. [Google Scholar] [CrossRef]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Application of magnetic nanoparticles in biomedicine. J. Phys. D Appl. Phys. 2003, 36, R167–R181. [Google Scholar] [CrossRef]
- Couvreur, P. Nanoparticles in drug delivery: Past, present and future. Adv. Drug Deliv. Rev. 2013, 65, 21–23. [Google Scholar] [CrossRef]
- Stark, W.J.; Stoessel, P.R.; Wohlleben, W.; Hafner, A. Indutrial applications of nanoparticles. Chem. Soc. Rev. 2015, 44, 5793–5805. [Google Scholar] [CrossRef]
- Musselwhite, N.; Somorjai, G.A. Investigations of structure sensitivity in heterogeneous catalysis: From single crystals to monodisperse nanoparticles. Top. Catal. 2013, 56, 1277–1283. [Google Scholar] [CrossRef]
- Vargas, E.; Romero-Sáez, M.; Denardin, J.C.; Gracia, F. The ultrasound-assisted synthesis of effective monodisperse nickel nanoparticles: Magnetic characterization and its catalytic activity in CO2 methanation. New J. Chem. 2016, 40, 7307–7310. [Google Scholar] [CrossRef]
- Weber, A.P.; Seipenbusch, M.; Kasper, G. Size effect in the catalytic activity of unsupported metallic nanoparticles. J. Nanoparticle Res. 2003, 5, 293–298. [Google Scholar] [CrossRef]
- Shamskar, F.R.; Rezaei, M.; Meshkani, F. The influence of Ni loading on the activity and coke formation on ultrasound-assisted co-precipitated Ni-Al2O3 nanocatalyst in dry reforming of methane. Int. J. Hydrogen Energy 2017, 42, 4155–4164. [Google Scholar] [CrossRef]
- Park, J.; Kang, E.; Son, S.U.; Park, H.M.; Lee, M.K.; Kim, J.; Kim, K.W.; Noh, H.-J.; Park, J.-H.; Bae, C.J.; et al. Monodisperse nanoparticles on Ni and NiO: Synthesis, characterization, self-assebled superlattices and catalytic applications in the Suzuki coupling reaction. Adv. Mater. 2005, 17, 429–434. [Google Scholar] [CrossRef]
- Yurino, T.; Ueda, Y.; Shimizu, Y.; Tanaka, S.; Nishiyama, H.; Tsurugi, H.; Sato, K.; Mashima, K. Salt-free reduction of nonprecious transition-metal compounds: Generation of amorphous Ni nanoparticles for catalytic C-C bond formation. Angew. Chem. Int. Ed. 2015, 54, 14437–14441. [Google Scholar] [CrossRef]
- Hwang, S.J.; Lee, J.-H. Mechanochemical synthesis of Cu-Al2O3 nanocomposites. Mater. Sci. Eng. A 2005, 405, 140–146. [Google Scholar] [CrossRef]
- Balaz, P. From Minerals to Nanoparticles, Mechanochemistry in Nanoscience and Minerals Engineering; Springer: Berlin/Heidelberg, Germany, 2008; pp. 177–256. [Google Scholar]
- Ban, I.; Stergar, J.; Drofenik, M.; Ferk, G.; Makovec, D. Synthesis of copper-nickel nanoparticles prepared by mechanical milling for use in magnetic hyperthermia. J. Magn. Magn. Mater. 2011, 323, 2254–2258. [Google Scholar] [CrossRef]
- Musza, K.; Szabados, M.; Ádám, A.A.; Kónya, Z.; Kukovecz, Á.; Sipos, P.; Pálinkó, I. Ball milling of copper powder under dry and surfactant-assisted conditions-on the way towards Cu/Cu₂O nanocatalyst. J. Nanosci. Nanotechnol. 2019, 19, 389–394. [Google Scholar] [CrossRef]
- Faraday, M. The bakerian lecture: Experimental relations of gold (and other metals) to light. Philos. Trans. R. Soc. 1857, 147, 145–181. [Google Scholar]
- Wu, S.-H.; Chen, D.-W. Synthesis and characterization of nickel nanoparticles by hydrazine reduction in ethylene glycol. J. Colloid Interf. Sci. 2003, 259, 282–286. [Google Scholar] [CrossRef]
- Wu, Z.G.; Munoz, M.; Montero, O. The synthesis of nickel nanoparticles by hydrazine reduction. Adv. Powder Technol. 2010, 21, 165–168. [Google Scholar] [CrossRef]
- Hou, Y.; Kondoh, Y.H.; Ohta, T.; Gao, S. Size-controlled synthesis of nickel nanoparticles. Appl. Surf. Sci. 2005, 241, 218–222. [Google Scholar] [CrossRef]
- Huaman, J.L.C.; Hironaka, N.; Tanaka, S.; Shinoda, K.; Miyamura, H.; Jeyadevan, B. Size-controlled monodispersed nickel nanocrystals using 2-octanol as reducing agent. Cryst. Eng. Comm. 2013, 15, 729–737. [Google Scholar] [CrossRef]
- Chen, D.-H.; He, X.-R. Synthesis of nickel ferrite nanoparticles by sol-gel method. Mater. Res. Bull. 2011, 36, 1369–1377. [Google Scholar] [CrossRef]
- Salavati-Niasari, M.; Davar, F.; Mir, N. Synthesis and characterization on metallic copper nanoparticles via thermal decomposition. Polyhedron 2008, 27, 3514–3518. [Google Scholar] [CrossRef]
- Chandran, S.P.; Chaudhary, M.; Pasricha, R.; Ahmad, A.; Sastry, M. Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract. Biotechnol. Prog. 2006, 22, 577–583. [Google Scholar] [CrossRef]
- Huang, J.; Li, Q.; Sun, D.; Lu, Y.; Su, Y.; Yang, X.; Wang, H.; Wang, Y.; Shao, W.; He, N.; et al. Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphora leaf. Nanotechnology 2007, 18, 105104. [Google Scholar] [CrossRef]
- Narayanan, K.B.; Sakthivel, N. Coriander leaf mediated biosynthesis of gold nanoparticles. Mater. Lett. 2008, 62, 4588–4590. [Google Scholar] [CrossRef]
- Suslick, K.S. Sonochemistry. Science 1990, 247, 1439–1445. [Google Scholar] [CrossRef]
- Boldyrev, V.V. Mechanochemistry and sonochemistry. Ultrason. Sonochem. 1995, 2, S143–S145. [Google Scholar] [CrossRef]
- Cintas, P.; Cravotto, G.; Barge, A.; Martina, K. Interplay between mechanochemistry and sonochemistry. Top. Curr. Chem. 2014, 369, 239–284. [Google Scholar]
- Fujimoto, T.; Terauchi, S.; Umehara, H.; Kojima, I.; Henderson, W. Sonochemical preparation of single-dispersion metal nanoparticles from metal salts. Chem. Mater. 2001, 13, 1057–1060. [Google Scholar] [CrossRef]
- Li, Z.; Tao, X.; Cheng, Y.; Wu, Z.; Zhang, Z.; Dang, H. A simple and rapid method for preparing indium nanoparticles from bulk indium via ultrasound irradiation. Mater. Sci. Eng. A 2005, 407, 7–10. [Google Scholar] [CrossRef]
- Zhu, J.; Liu, S.; Palchik, O.; Koltypin, Y.; Gedanken, A. Shape-controlled synthesis of silver nanoparticles by pulse sonoelectrochemical methods. Langmuir 2000, 16, 6396–6399. [Google Scholar] [CrossRef]
- Okitsu, K.; Ashokkumar, M.; Grieser, F. Sonochemical synthesis of gold nanoparticles: Effects of ultrasound frequency. J. Phys. Chem. B 2005, 109, 20673–20675. [Google Scholar] [CrossRef]
- Mizukoshi, Y.; Okitsu, K.; Maeda, Y.; Yamamoto, T.A.; Oshima, R.; Nagata, Y. Sonochemical preparation of bimetallic nanoparticles of gold/palladium in aqueous solution. J. Phys. Chem. B 1997, 101, 7033–7037. [Google Scholar] [CrossRef]
- Kan, C.; Cai, W.; Li, C.; Zhang, L.; Hofmeister, H. Ultrasonic synthesis and optical properties of Au/Pd bimetallic nanoparticles in ethylene glycol. J. Phys. D Appl. Phys. 2003, 36, 1609–1614. [Google Scholar] [CrossRef]
- Hassanjani-Roshan, A.; Vaezi, M.R.; Shokuhfar, A.; Rajabali, Z. Synthesis of iron oxide nanoparticles via sonochemical method and their characterization. Particuology 2011, 9, 95–99. [Google Scholar] [CrossRef]
- Pol, V.G.; Palchik, O.; Gedanken, A.; Felner, I. Synthesis of europium oxide nanorods by ultrasound irradiation. J. Phys. Chem. 2002, 106, 9737–9743. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, J.-R.; Zhao, X.-N.; Xu, S.; Zhu, J.-J. Preparation of copper monosulfide and nickel monosulfide nanoparticles by sonochemical method. Mater. Lett. 2002, 55, 253–258. [Google Scholar] [CrossRef]
- Daroughegi, R.; Meshkani, F.; Rezaei, M. Enhanced activity of CO2 methanation over mesoporous nanocrystalline Ni-Al2O3 catalysts prepared by ultrasound-assisted co-precipitation method. Int. J. Hydrogen Energy 2017, 42, 15115–15125. [Google Scholar] [CrossRef]
- Rahmani, F.; Haghighi, M.; Vafaeian, Y.; Estifaee, P. Hydrogen production via CO2 reforming of methane over ZrO2-Doped Ni/ZSM-5 nanostructured catalyst prepared by ultrasound assisted sequential impregnation method. J. Power Sources 2014, 272, 816–827. [Google Scholar] [CrossRef]
- Afshari, S.; Montazer, M. In-situ sonosynthesis of hedgehog-like nickel nanoparticles on polyester fabric producing magnetic properties. Ultrason. Sonochem. 2018, 40, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liu, Y.; Gao, Y.; Cheng, Q.; Zhao, L.; Yang, Z. Magnetic properties of aristate spherical Ni nanoparticles synthesized through ultrasound reduction method. Mater. Res. Bull. 2017, 87, 135–139. [Google Scholar] [CrossRef]
- Lorimer, J.P.; Mason, T.J. Sonochemistry. Part 1. The physical aspects. Chem. Soc. Rev. 1987, 16, 239–274. [Google Scholar] [CrossRef]
- Tripathi, B.; Paniwnyk, L.; Cherkasov, N.; Ibhadon, A.O.; Lana-Villarreal, T.; Gómez, R. Ultrasound-assisted selective hydrogenation of C-5 acetylene alcohols with Lindlar catalysts. Ultrason. Sonochem. 2015, 26, 445–451. [Google Scholar] [CrossRef]
- Ádám, A.A.; Szabados, M.; Musza, K.; Bélteky, P.; Kónya, Z.; Kukovecz, Á.; Sipos, P.; Pálinkó, I. Effects of medium and nickel salt source in the synthesis and catalytic performance of nano-sized nickel in the Suzuki-Miyaura cross-coupling reaction. React. Kinet. Mech. Catal. 2019, 126, 841–855. [Google Scholar] [CrossRef]
- Yu, K.; Kim, D.J.; Chung, H.S.; Liang, H. Dispersed rodlike nickel powder synthesized by modified polyol process. Mater. Lett. 2003, 57, 3992–3997. [Google Scholar] [CrossRef]
- Park, J.W.; Chae, E.H.; Kim, S.H.; Lee, J.H.; Kim, J.W.; Yoon, S.M.; Choi, J.-Y. Preparation of fine Ni powders from nickel hydrazine complex. Mater. Chem. Phys. 2006, 97, 371–378. [Google Scholar] [CrossRef]
- Schülein, J.; Minrath, I.; Pommersheim, R.; Löwe, H. Continuous-flow synthesis of Ni(0) nanoparticles using a cone channel nozzle or a micro coaxial-injection mixer. J. Flow Chem. 2014, 4, 44–53. [Google Scholar] [CrossRef]
- Eluri, R.; Paul, B. Synthesis of nickel nanoparticles by hydrazine reduction: Mechanistic study and continuous flow synthesis. J. Nanoparticle Res. 2012, 14, 800. [Google Scholar] [CrossRef]
- Xu, H.; Zeiger, B.W.; Suslick, K.S. Sonochemical synthesis of nanomaterials. Chem. Soc. Rev. 2013, 42, 2555–2567. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, D.; Swindells, R. Hydrazine complexes of nickel(II) chloride. J. Inorg. Nucl. Chem. 1968, 30, 2211–2217. [Google Scholar] [CrossRef]
- İçbudak, H.; Yilmaz, V.T. Synthesis and characterization of new mixed-ligand bis(hydrazine) and bis(ethylenediamine) complexes of cobalt, nickel and copper saccharinates. Synth. React. Inorg. Met. Org. Chem. 1997, 27, 1517–1525. [Google Scholar] [CrossRef]
- Braibanti, A.; Dallavalle, F.; Pellinghelli, M.A.; Leporati, E. The nitrogen-nitrogen stretching band in hydrazine derivatives and complexes. Inorg. Chem. 1968, 7, 1430–1433. [Google Scholar] [CrossRef]
- Ryokichi, T.; Masaaki, Y.; Akira, U.; Eishin, K. Derivatographic studies on transition metal complexes. XIII. thermal decomposition of [Ni(N2H4)6]X2 complexes. Bull. Chem. Soc. Jpn. 1974, 47, 660–664. [Google Scholar]
- Wang, D.; Xu, R.; Wang, X.; Li, Y. NiO nanorings and their unexpected catalytic property for CO oxidation. Nanotechnology 2006, 17, 979–983. [Google Scholar] [CrossRef]
- Parui, J.; Murali, B.; Biradar, B.; Krupanidhi, S.B. Oxygen deficiency induced nickel based oxides for UV & IR sensitive photo-conductive devices. Mater. Res. Bull. 2018, 107, 321–327. [Google Scholar]
- Song, P.; Wen, D.; Guo, Z.X.; Korakianitis, T. Oxidation investigation of nickel nanoparticles. Phys. Chem. Chem. Phys. 2008, 10, 5057–5065. [Google Scholar] [CrossRef]
- Franc, J.; Bastl, Z. Nickel evaporation in high vacuum and formation of nickel oxide nanoparticles on highly oriented pyrolytic graphite. X-ray photoelectron spectroscopy and atomic force microscopy study. Thin Solid Films 2008, 516, 6095–6103. [Google Scholar] [CrossRef]
- Horányi, T.S. The thermal stability of the β-Ni(OH)2-β-NiOOH system. Thermochim. Acta 1989, 137, 247–253. [Google Scholar] [CrossRef]
- Ni, S.; Lv, X.; Li, T.; Yang, X.; Zhang, L. The investigation of Ni(OH)2/Ni as anodes for high performance Li-ion batteries. J. Mater. Chem. A 2013, 1, 1544–1547. [Google Scholar] [CrossRef]
- Sakai, G.; Miyazaki, M.; Kijima, T. Synthesis of high surface area β-Ni(OH)2 and its transformation into nanosheets and nanodisks by hydrothermal treatment. J. Electrochem. Soc. 2010, 157, A480–A486. [Google Scholar] [CrossRef]
- Jung, S.C.; Sim, S.L.; Soon, Y.W.; Lim, C.M.; Hing, P.; Jennings, J.R. Synthesis of nanostructured β-Ni(OH)2 by electrochemical dissolution–precipitation and its application as a water oxidation catalyst. Nanotechnology 2016, 27, 275401. [Google Scholar] [CrossRef]
- Okada, K.; Kaneda, A.; Kameshima, Y.; Yasumori, A. Acidic and basic gas desorption properties in composites of layered double hydroxide/aluminosilicate xerogels. Mater. Res. Bull. 2002, 37, 209–219. [Google Scholar] [CrossRef]
- Stirniman, M.J.; Huang, C.; Smith, R.S.; Joyce, S.A.; Kay, B.D. The adsorption and desorption of water on single crystal MgO(100): The role of surface defects. J. Chem. Phys. 1996, 105, 1295–1298. [Google Scholar] [CrossRef]
- Razali, M.H.; Osman, U.M.; Rozaini, M.Z.H.M.; Yusoff, M. CO2 adsorption study on NiO and Pr2O3-NiO catalyst synthesis using simple sol-gel method. Orient. J. Chem. 2017, 33, 1864–1870. [Google Scholar] [CrossRef][Green Version]
- Chen, Y.; Xiao, M.; Wang, S.; Han, D.; Lu, Y.; Meng, Y. Porous diatomite-immobilized Cu–Ni bimetallic nanocatalysts for direct synthesis of dimethyl carbonate. J. Nanomater. 2012, 2012, 610410. [Google Scholar] [CrossRef]
- Miranda, B.C.; Chimentão, R.J.; Santos, J.B.O.; Gispert-Guirado, F.; Llorca, J.; Medina, F.; López Bonillo, F.; Sueiras, J.E. Conversion of glycerol over 10%Ni/-Al2O3 catalyst. Appl. Catal. B Environ. 2014, 147, 464–480. [Google Scholar] [CrossRef]
- Yao, Y.; Yan, Z.; Chen, L.; Zhou, Z.; Liu, L.; Goodman, D.W. Nickel particle size effects on cyclohexane dehydrogenation: A combined reaction kinetics and surface science study. Catal. Lett. 2016, 6, 134. [Google Scholar] [CrossRef]
- Magano, J.; Dunetz, J.R. Large-scale applications of transition metal-catalyzed couplings for the synthesis of pharmaceuticals. Chem. Rev. 2011, 111, 2177–2250. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Buchwald, S.L. Palladium-catalyzed Suzuki-Miyaura cross-coupling reactions employing dialkylbiaryl phospine ligands. Acc. Chem. Res. 2008, 41, 1461–1473. [Google Scholar] [CrossRef] [PubMed]
- Scheuermann, G.M.; Rumi, L.; Steurer, P.; Bannwarth, W.; Mülhaupt, R. Palladium nanoparticles on graphite oxide and its functionalized graphene derivatives as highly active catalysts for the Suzuki-Miyaura cross-coupling reaction. J. Am. Chem. Soc. 2009, 131, 8262–8270. [Google Scholar] [CrossRef] [PubMed]
- Han, F.-S. Transition-metal-catalyzed Suzuki-Miyaura cross-coupling reactions: A remarkable advance from palladium to nickel catalysts. Chem. Soc. Rev. 2013, 42, 5270–5298. [Google Scholar] [CrossRef] [PubMed]
- Weires, N.A.; Baker, E.L.; Garg, N.K. Nickel-catalysed Suzuki-Miyaura coupling of amides. Nat. Chem. 2016, 8, 75–79. [Google Scholar] [CrossRef]
- Saeki, T.; Son, E.-C.; Tamao, K. Boron trifluoride induced palladium-catalyzed cross-coupling reaction of 1-aryltriazenes with areneboronic acids. Org. Lett. 2004, 6, 617–619. [Google Scholar] [CrossRef]
- Fan, G.; Zhang, H.; Cheng, S.; Ren, Z.; Hu, Z.; Wang, Z. Lewis acid-promoted Suzuki reaction using palladium chloride anchored on a polymer as a catalyst. Aust. J. Chem. 2008, 61, 610–614. [Google Scholar] [CrossRef]
- Sylvester, K.T.; Wu, K.; Doyle, A.G. Mechanistic investigation of the nickel-catalyzed Suzuki reaction of N,O-acetals: Evidence for boronic acid assisted oxidative addition and an iminium activation pathway. J. Am. Chem. Soc. 2012, 134, 16967–16970. [Google Scholar] [CrossRef]
- Jia, X.-G.; Guo, P.; Duan, J.; Shu, X.-Z. Dual nickel and Lewis acid catalysis for cross-electrophile coupling: The allylation of aryl halides with allylic alcohols. Chem. Sci. 2018, 9, 640–645. [Google Scholar] [CrossRef]
- Frohlich, M.T.; Anderson, K.J.; Ugrinov, A.; Boudjouk, P. Breaking carbon–chlorine bonds with the unconventional Lewis acid dodecachlorocyclohexasilane. Inorg. Chem. 2018, 57, 14463–14466. [Google Scholar] [CrossRef] [PubMed]
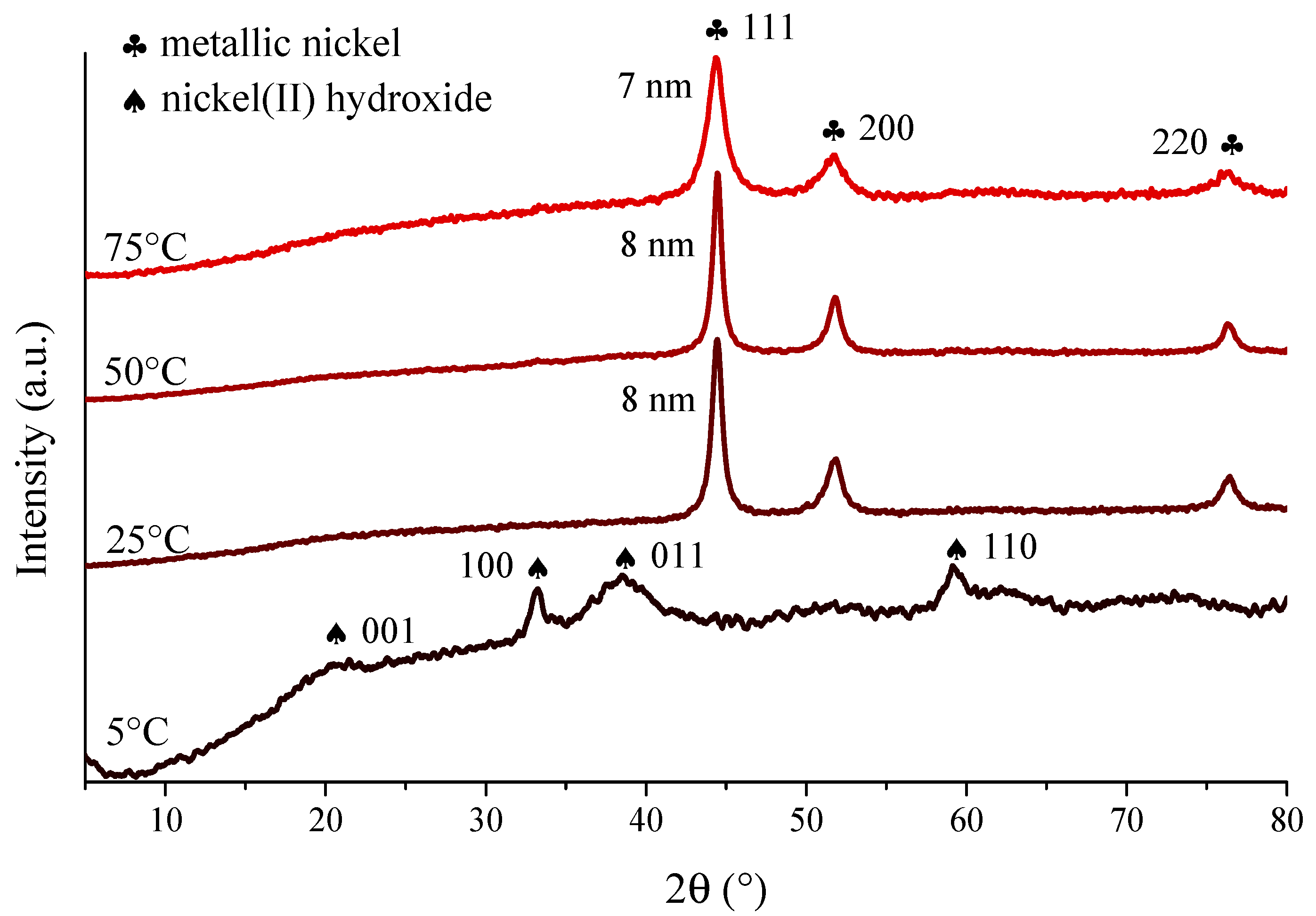
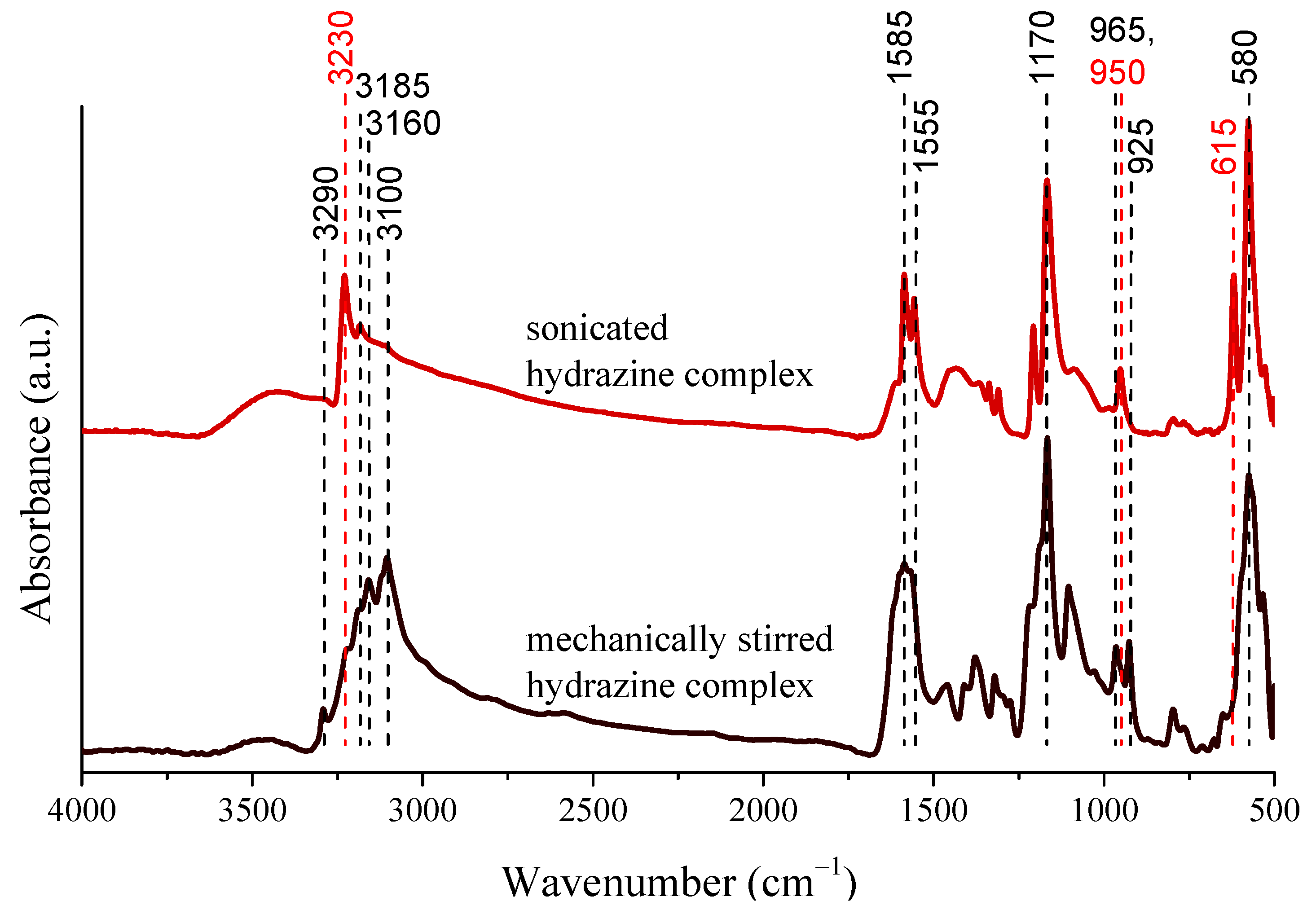
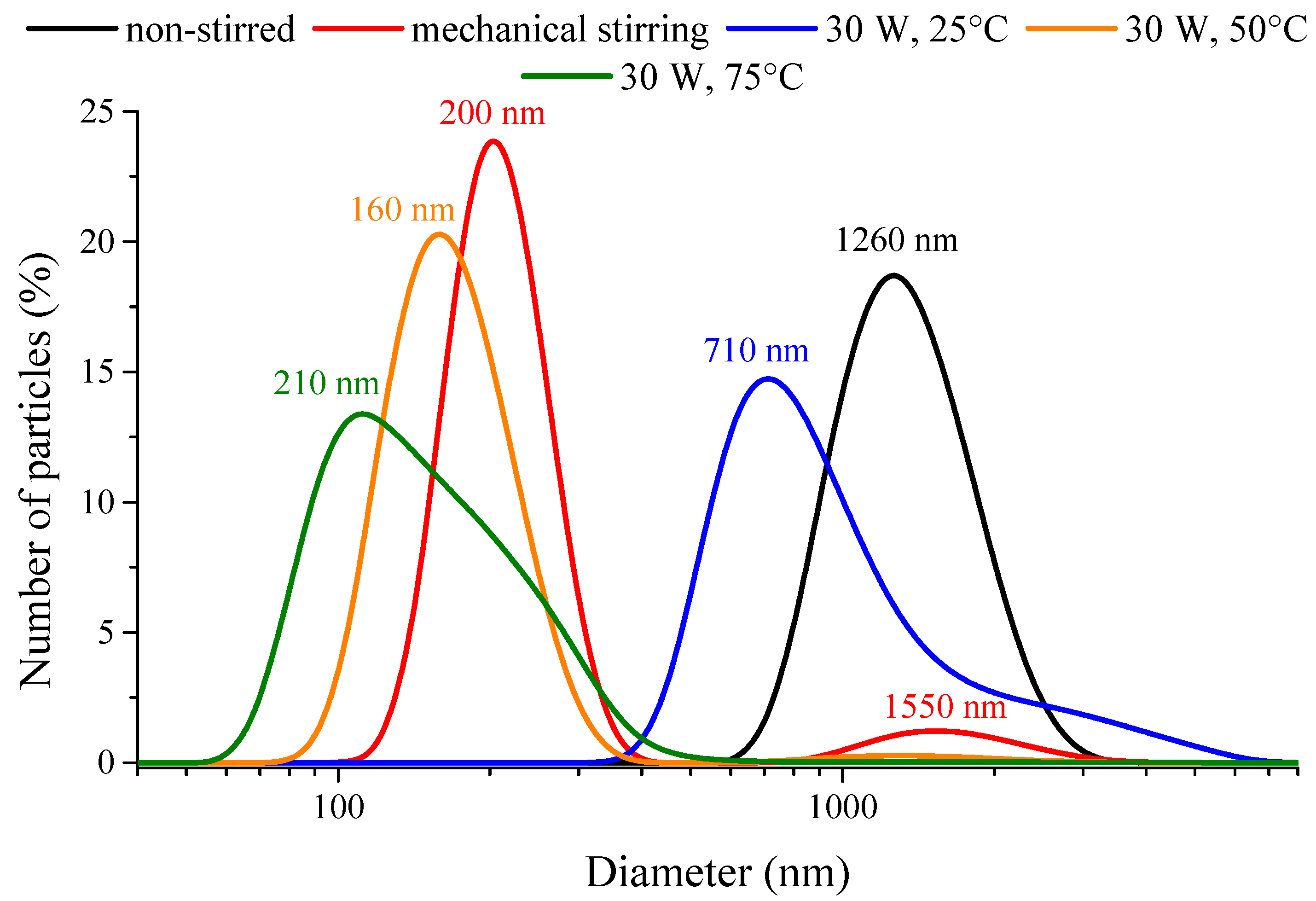
| Ni NPs Samples | Ultrasound Power Density (W/cm3) a | Primary Particle Size (nm) | Predominant Solvodynamic Diameter (nm) | Poly-Dispersity Index | Type of Size Distribution |
|---|---|---|---|---|---|
| non-stirred | − | 12 | 1262 | 0.147 | Unimodal |
| Mechanically stirred | − | 14 | 202 | 0.368 | Bimodal |
| ultrasonic treatment: | |||||
| 30 W – 20% b | 0.007 | 7 | 255 | 0.277 | Unimodal |
| 30 W – 40% | 0.01 | 7 | 396 | 0.199 | Unimodal |
| 30 W – 60% | 0.013 | 7 | 342 | 0.288 | Unimodal |
| 30 W – 80% | 0.017 | 8 | 396 | 0.230 | Unimodal |
| 30 W – 100% | 0.023 | 8 | 712 | 0.170 | Unimodal |
| 60 W – 100% | 0.041 | 8 | 825 | 0.198 | Unimodal |
| 90 W – 100% | 0.057 | 9 | 530 | 0.226 | Unimodal |
| 120 W – 100% | 0.085 | 10 | 190 | 0.404 | Bimodal |
| Ni NP Samples | TG Second Mass Loss (%) | β-Ni(OH)2 Content (m/m%) | Specific Surface Area (m2/g) | Total Pore Volume (cm3/g) | Average Pore Diameter (Å) |
|---|---|---|---|---|---|
| non-stirred | 2.2 | 11.3 | 28.1 | 0.040 | 35.4 |
| mechanical stirring | 0.3 | 1.5 | 21.9 | 0.027 | 35.8 |
| 30 W – 20% a | 3.7 | 19.0 | 39.5 | 0.054 | 38.8 |
| 30 W – 100% | 0.3 | 1.5 | 20.4 | 0.043 | 36.2 |
| 120 W – 100% | 1.0 | 5.1 | 28.6 | 0.036 | 38.8 |
| Ni NP Samples | Total Basicity (mmol CO2/g) | Temperature of Peak Maxima (°C) | Total Acidity (mmol NH3/g) | Temperature of Peak Maxima (°C) |
|---|---|---|---|---|
| non-stirred | 0.051 | 90 and 165 | 0.021 | 185 and – |
| mechanically stirred | 0.046 | 90 and 160 | 0.025 | 150 and 330 |
| 30 W – 20% | 0.054 | 95 and 155 | 0.105 | 175 and – |
| 30 W – 100% | 0.032 | 90 and 175 | 0.257 | 150 and 370 |
| 120 W – 100% | 0.088 | 90 and 165 | 0.054 | 190 and – |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ádám, A.A.; Szabados, M.; Varga, G.; Papp, Á.; Musza, K.; Kónya, Z.; Kukovecz, Á.; Sipos, P.; Pálinkó, I. Ultrasound-Assisted Hydrazine Reduction Method for the Preparation of Nickel Nanoparticles, Physicochemical Characterization and Catalytic Application in Suzuki-Miyaura Cross-Coupling Reaction. Nanomaterials 2020, 10, 632. https://doi.org/10.3390/nano10040632
Ádám AA, Szabados M, Varga G, Papp Á, Musza K, Kónya Z, Kukovecz Á, Sipos P, Pálinkó I. Ultrasound-Assisted Hydrazine Reduction Method for the Preparation of Nickel Nanoparticles, Physicochemical Characterization and Catalytic Application in Suzuki-Miyaura Cross-Coupling Reaction. Nanomaterials. 2020; 10(4):632. https://doi.org/10.3390/nano10040632
Chicago/Turabian StyleÁdám, Adél Anna, Márton Szabados, Gábor Varga, Ádám Papp, Katalin Musza, Zoltán Kónya, Ákos Kukovecz, Pál Sipos, and István Pálinkó. 2020. "Ultrasound-Assisted Hydrazine Reduction Method for the Preparation of Nickel Nanoparticles, Physicochemical Characterization and Catalytic Application in Suzuki-Miyaura Cross-Coupling Reaction" Nanomaterials 10, no. 4: 632. https://doi.org/10.3390/nano10040632
APA StyleÁdám, A. A., Szabados, M., Varga, G., Papp, Á., Musza, K., Kónya, Z., Kukovecz, Á., Sipos, P., & Pálinkó, I. (2020). Ultrasound-Assisted Hydrazine Reduction Method for the Preparation of Nickel Nanoparticles, Physicochemical Characterization and Catalytic Application in Suzuki-Miyaura Cross-Coupling Reaction. Nanomaterials, 10(4), 632. https://doi.org/10.3390/nano10040632