On the Effects of Diluted and Mixed Ionic Liquids as Liquid Substrates for the Sputter Synthesis of Nanoparticles
Abstract
1. Introduction
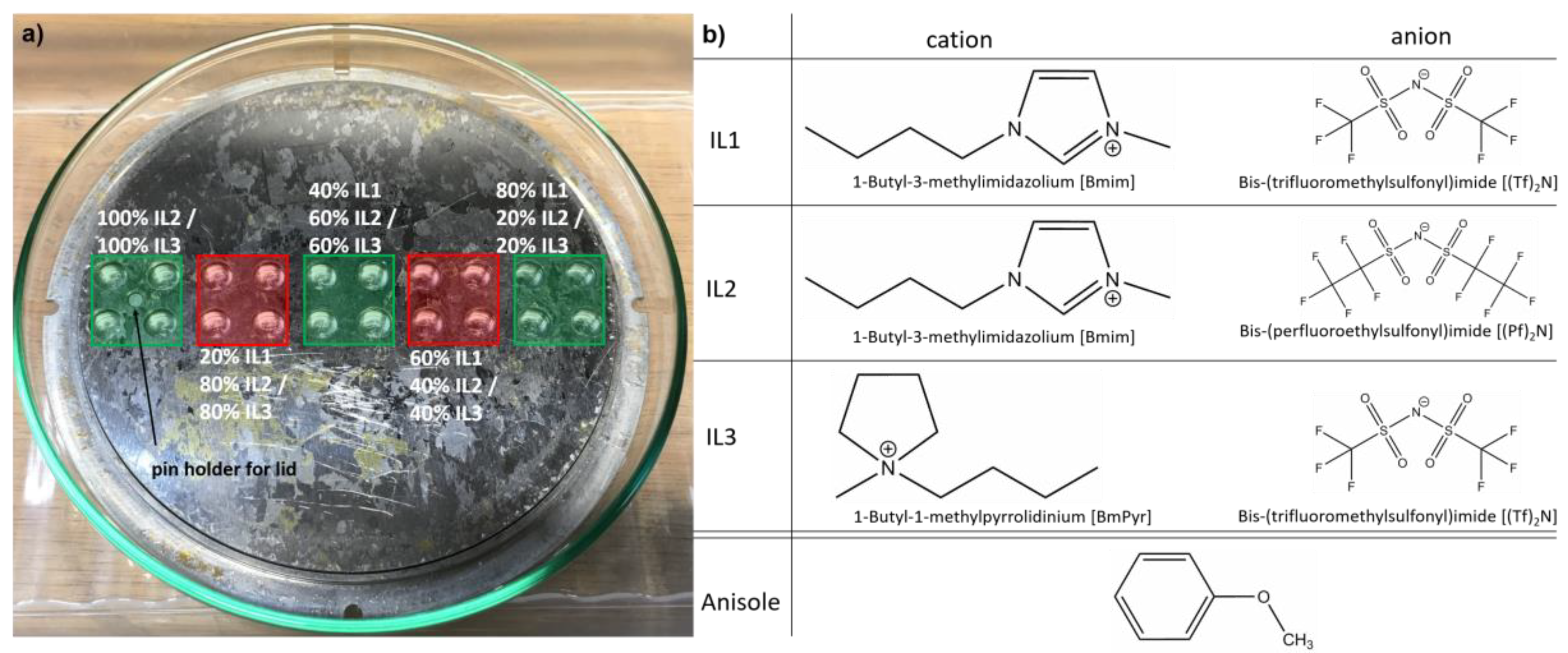
2. Materials and Methods
2.1. Depositions of Ag, Cu, Au into IL and IL Mixtures
2.2. Dilution of Already-Sputtered Ag in IL1 with an Organic Solvent
2.3. TEM Sample Preparation and Analysis
3. Results
3.1. Mixture of Ag/IL1 Suspension with Anisole
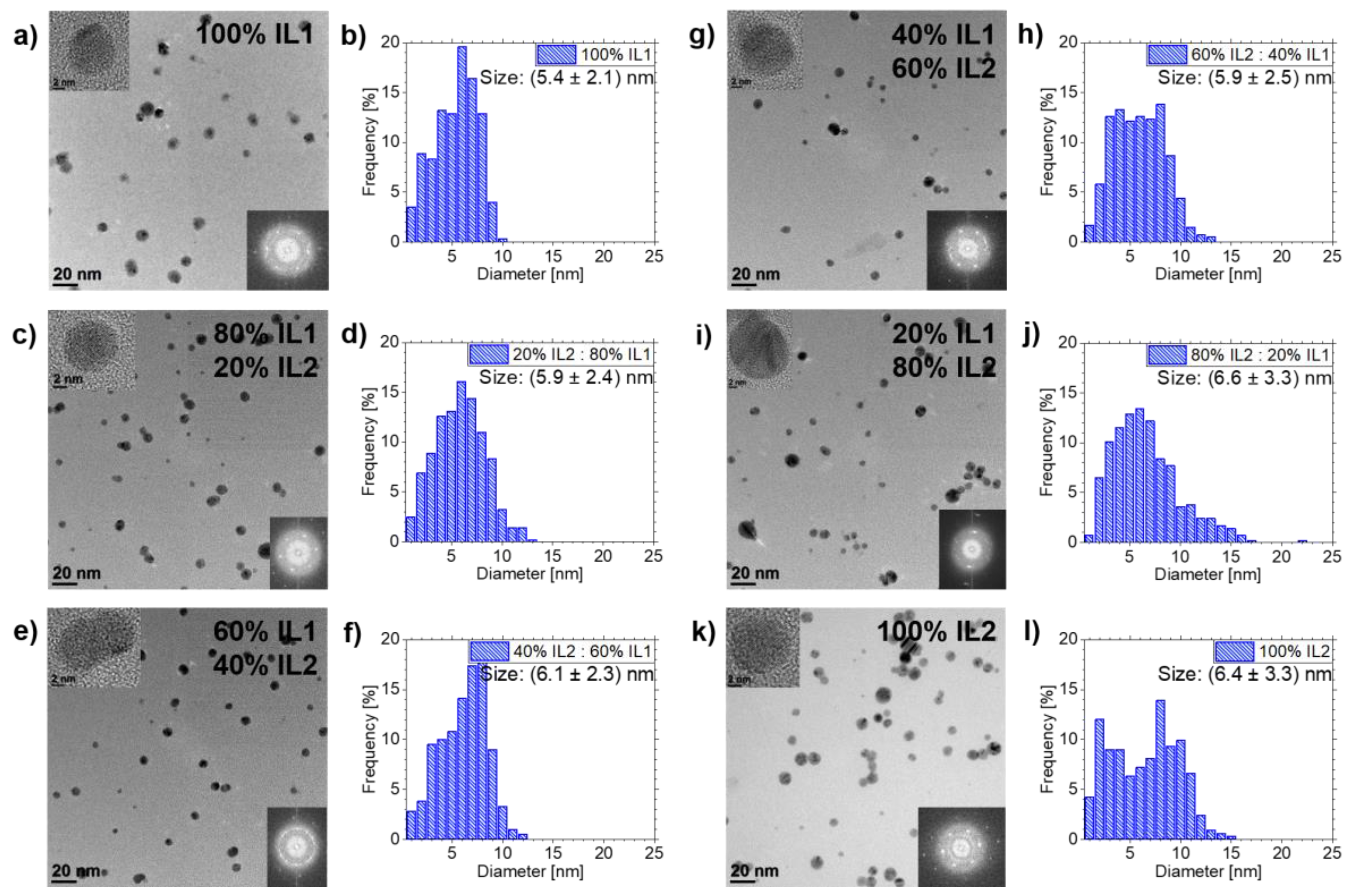
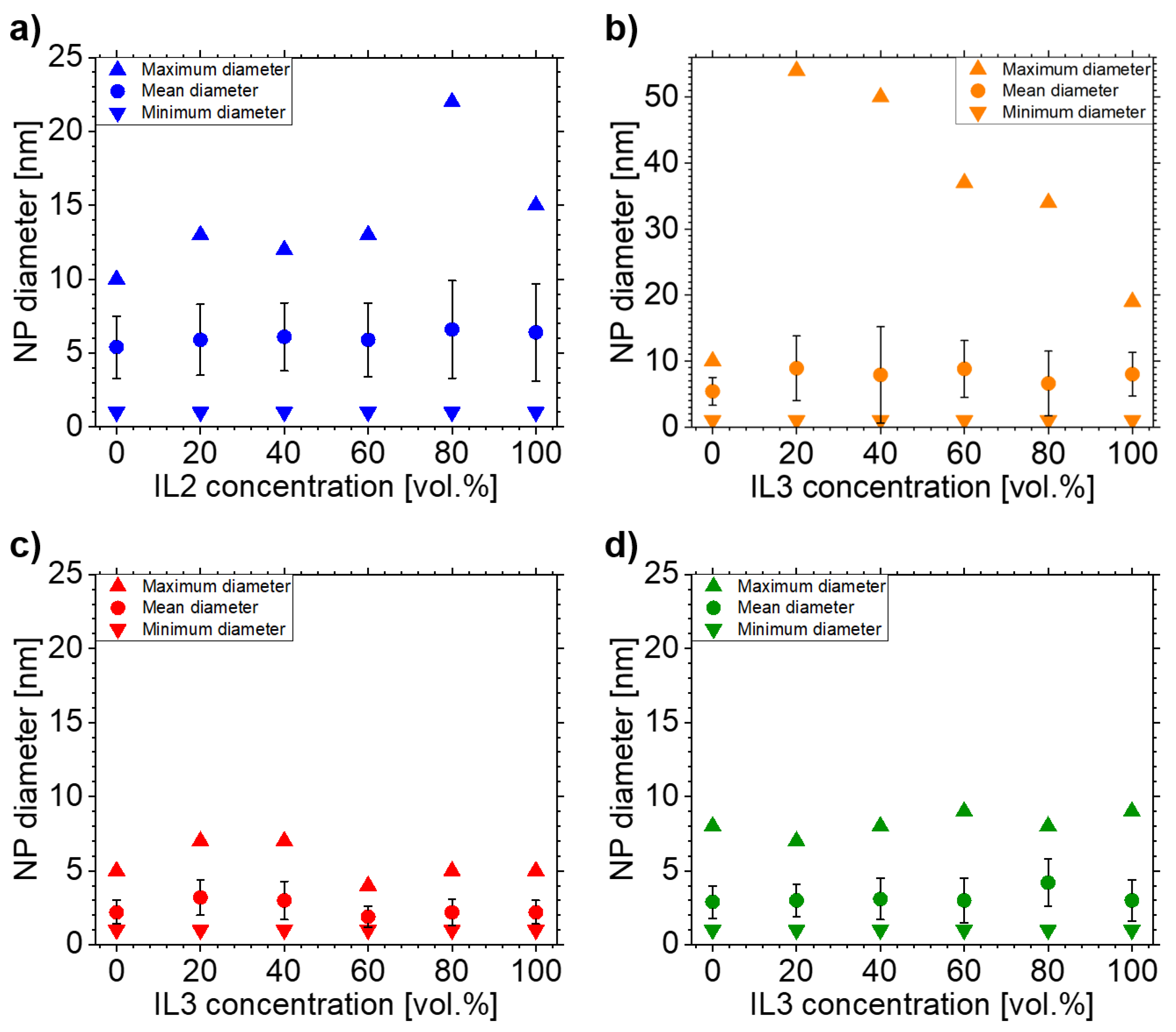
3.2. Ag NPs Sputtered in IL1/IL2-Mixtures
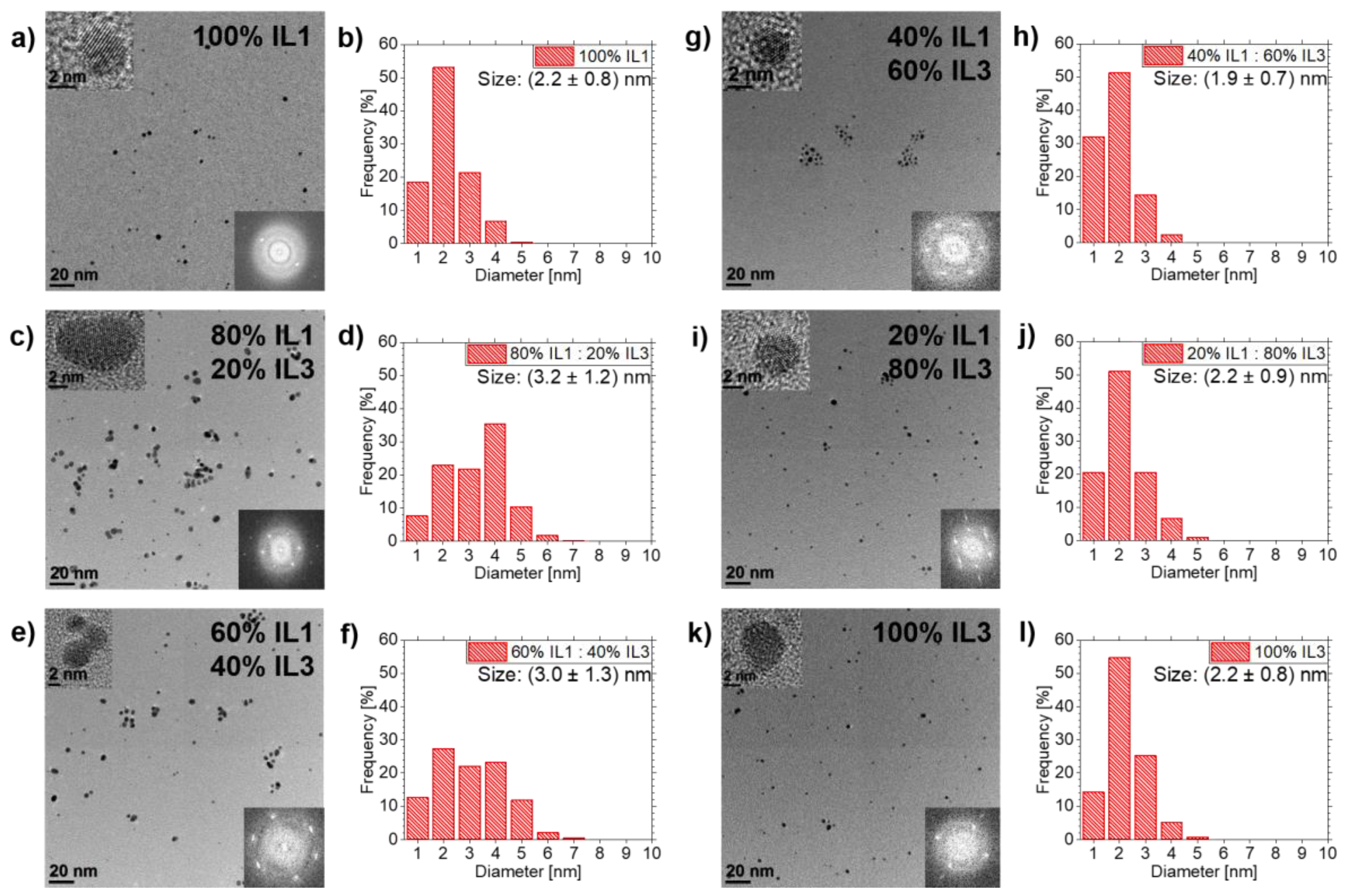
3.3. Ag NPs Sputtered in IL1/IL3-Mixtures
3.4. Au NPs Sputtered in IL1/IL3-Mixtures
3.5. Cu NPs Sputtered in IL1/IL3-Mixtures
4. Discussion
4.1. NP Stabilization in IL Mixed with Organic Solvent
4.2. Effects of IL Mixtures on NPs Sizes
4.3. NP Stabilization in IL1/IL2-Mixtures
4.4. NP Stabilization in IL1/IL3-Mixtures
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Migowski, P.; Dupont, J. Catalytic applications of metal nanoparticles in imidazolium ionic liquids. Chemistry 2007, 13, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Löffler, T.; Meyer, H.; Savan, A.; Wilde, P.; Garzón Manjón, A.; Chen, Y.-T.; Ventosa, E.; Scheu, C.; Ludwig, A.; Schuhmann, W. Discovery of a Multinary Noble Metal-Free Oxygen Reduction Catalyst. Adv. Energy Mater. 2018, 8, 1802269. [Google Scholar] [CrossRef]
- Löffler, T.; Savan, A.; Garzón-Manjón, A.; Meischein, M.; Scheu, C.; Ludwig, A.; Schuhmann, W. Toward a Paradigm Shift in Electrocatalysis Using Complex Solid Solution Nanoparticles. ACS Energy Lett. 2019, 4, 1206–1214. [Google Scholar] [CrossRef]
- Astruc, D. Transition-metal Nanoparticles in Catalysis: From Historical Background to the State-of-the Art. In Nanoparticles and Catalysis; Astruc, D., Ed.; Wiley-VCH: Weinheim, Germany, 2008; pp. 1–48. ISBN 9783527621323. [Google Scholar]
- Conde, J.; Doria, G.; Baptista, P. Noble metal nanoparticles applications in cancer. J. Drug Deliv. 2012, 2012, 751075. [Google Scholar] [CrossRef] [PubMed]
- Pankhurst, Q.A.; Connolly, J.; Jones, S.K.; Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D Appl. Phys. 2003, 36, R167–R181. [Google Scholar] [CrossRef]
- Jain, K.K. Applications of nanobiotechnology in clinical diagnostics. Clin. Chem. 2007, 53, 2002–2009. [Google Scholar] [CrossRef]
- Boroumand Moghaddam, A.; Namvar, F.; Moniri, M.; Md Tahir, P.; Azizi, S.; Mohamad, R. Nanoparticles Biosynthesized by Fungi and Yeast: A Review of Their Preparation, Properties, and Medical Applications. Molecules 2015, 20, 16540–16565. [Google Scholar] [CrossRef]
- Ito, S.; Unemoto, A.; Ogawa, H.; Tomai, T.; Honma, I. Application of quasi-solid-state silica nanoparticles–ionic liquid composite electrolytes to all-solid-state lithium secondary battery. J. Power Sources 2012, 208, 271–275. [Google Scholar] [CrossRef]
- Lu, Y.; Das, S.K.; Moganty, S.S.; Archer, L.A. Ionic liquid-nanoparticle hybrid electrolytes and their application in secondary lithium-metal batteries. Adv. Mater. Weinheim. 2012, 24, 4430–4435. [Google Scholar] [CrossRef]
- Lu, Y.; Korf, K.; Kambe, Y.; Tu, Z.; Archer, L.A. Ionic-liquid-nanoparticle hybrid electrolytes: Applications in lithium metal batteries. Angew. Chem. Int. Ed Engl. 2014, 53, 488–492. [Google Scholar] [CrossRef]
- Garzón-Manjón, A.; Aranda-Ramos, A.; Melara-Benítez, B.; Bensarghin, I.; Ros, J.; Ricart, S.; Nogués, C. Simple Synthesis of Biocompatible Stable CeO2 Nanoparticles as Antioxidant Agents. Bioconjug. Chem. 2018, 29, 2325–2331. [Google Scholar] [CrossRef]
- Garzón-Manjón, A.; Solano, E.; de La Mata, M.; Guzmán, R.; Arbiol, J.; Puig, T.; Obradors, X.; Yáñez, R.; Ricart, S.; Ros, J. Induced shape controllability by tailored precursor design in thermal and microwave-assisted synthesis of Fe3O4 nanoparticles. J. Nanopart. Res. 2015, 17, 329. [Google Scholar] [CrossRef]
- Dorjnamjin, D.; Ariunaa, M.; Shim, Y.K. Synthesis of silver nanoparticles using hydroxyl functionalized ionic liquids and their antimicrobial activity. Int. J. Mol. Sci. 2008, 9, 807–820. [Google Scholar] [CrossRef] [PubMed]
- Schaak, R.E.; Sra, A.K.; Leonard, B.M.; Cable, R.E.; Bauer, J.C.; Han, Y.-F.; Means, J.; Teizer, W.; Vasquez, Y.; Funck, E.S. Metallurgy in a beaker: Nanoparticle toolkit for the rapid low-temperature solution synthesis of functional multimetallic solid-state materials. J. Am. Chem. Soc. 2005, 127, 3506–3515. [Google Scholar] [CrossRef] [PubMed]
- Nikam, A.V.; Prasad, B.L.V.; Kulkarni, A.A. Wet chemical synthesis of metal oxide nanoparticles: A review. CrystEngComm 2018, 20, 5091–5107. [Google Scholar] [CrossRef]
- Lazarus, L.L.; Riche, C.T.; Malmstadt, N.; Brutchey, R.L. Effect of ionic liquid impurities on the synthesis of silver nanoparticles. Langmuir 2012, 28, 15987–15993. [Google Scholar] [CrossRef] [PubMed]
- Torimoto, T.; Okazaki, K.-I.; Kiyama, T.; Hirahara, K.; Tanaka, N.; Kuwabata, S. Sputter deposition onto ionic liquids: Simple and clean synthesis of highly dispersed ultrafine metal nanoparticles. Appl. Phys. Lett. 2006, 89, 243117. [Google Scholar] [CrossRef]
- Hatakeyama, Y.; Okamoto, M.; Torimoto, T.; Kuwabata, S.; Nishikawa, K. Small-Angle X-ray Scattering Study of Au Nanoparticles Dispersed in the Ionic Liquids 1-Alkyl-3-methylimidazolium Tetrafluoroborate. J. Phys. Chem. C 2009, 113, 3917–3922. [Google Scholar] [CrossRef]
- Richter, K.; Birkner, A.; Mudring, A.-V. Stabilizer-free metal nanoparticles and metal-metal oxide nanocomposites with long-term stability prepared by physical vapor deposition into ionic liquids. Angew. Chem. Int. Ed Engl. 2010, 49, 2431–2435. [Google Scholar] [CrossRef]
- Richter, K.; Birkner, A.; Mudring, A.-V. Stability and growth behavior of transition metal nanoparticles in ionic liquids prepared by thermal evaporation: How stable are they really? Phys. Chem. Chem. Phys. PCCP 2011, 13, 7136–7141. [Google Scholar] [CrossRef]
- Helgadottir, I.S.; Arquillière, P.P.; Bréa, P.; Santini, C.C.; Haumesser, P.-H.; Richter, K.; Mudring, A.-V.; Aouine, M. Synthesis of bimetallic nanoparticles in ionic liquids: Chemical routes vs physical vapor deposition. Microelectron. Eng. 2013, 107, 229–232. [Google Scholar] [CrossRef]
- Meyer, H.; Meischein, M.; Ludwig, A. Rapid Assessment of Sputtered Nanoparticle Ionic Liquid Combinations. ACS Comb. Sci. 2018, 20, 243–250. [Google Scholar] [CrossRef]
- König, D.; Richter, K.; Siegel, A.; Mudring, A.-V.; Ludwig, A. High-Throughput Fabrication of Au-Cu Nanoparticle Libraries by Combinatorial Sputtering in Ionic Liquids. Adv. Funct. Mater. 2014, 24, 2049–2056. [Google Scholar] [CrossRef]
- Meischein, M.; Garzón-Manjón, A.; Frohn, T.; Meyer, H.; Salomon, S.; Scheu, C.; Ludwig, A. Combinatorial Synthesis of Binary Nanoparticles in Ionic Liquids by Cosputtering and Mixing of Elemental Nanoparticles. ACS Comb. Sci. 2019. [CrossRef] [PubMed]
- Kokorin, A.I. Ionic Liquids. Theory, Properties, New Approaches; InTech: Rijeka, Croatia, 2011; ISBN 9533073497. [Google Scholar]
- Kadokawa, J.I. (Ed.) Ionic Liquids—New Aspects for the Future; InTech: Rijeka, Croatia, 2014; ISBN 978-953-51-0937-2. [Google Scholar]
- Welton, T. Room-Temperature Ionic Liquids. Solvents for Synthesis and Catalysis. Chem. Rev. 1999, 99, 2071–2084. [Google Scholar] [CrossRef]
- Holbrey, J.D.; Seddon, K.R. Ionic Liquids. Clean Technol. Environ. Policy 1999, 1, 223–236. [Google Scholar] [CrossRef]
- Wasserscheid, P.; Keim, W. Ionic Liquids—New “Solutions” for Transition Metal Catalysis. Angew. Chem. Int. Ed. 2000, 39, 3772–3789. [Google Scholar] [CrossRef]
- Kuwabata, S.; Torimoto, T.; Imanishi, A.; Tsu, T. Use of Ionic Liquid Under Vacuum Conditions. In Ionic Liquids—New Aspects for the Future; Kadokawa, J.-I., Ed.; InTech: Rijeka, Croatia, 2014; ISBN 978-953-51-0937-2. [Google Scholar]
- Vekariya, R.L. A review of ionic liquids: Applications towards catalytic organic transformations. J. Mol. Liq. 2017, 227, 44–60. [Google Scholar] [CrossRef]
- Luska, K.L.; Migowski, P.; Leitner, W. Ionic liquid-stabilized nanoparticles as catalysts for the conversion of biomass. Green Chem. 2015, 17, 3195–3206. [Google Scholar] [CrossRef]
- Plechkova, N.V.; Seddon, K.R. Applications of ionic liquids in the chemical industry. Chem. Soc. Rev. 2008, 37, 123–150. [Google Scholar] [CrossRef]
- Garzón-Manjón, A.; Meyer, H.; Grochla, D.; Löffler, T.; Schuhmann, W.; Ludwig, A.; Scheu, C. Controlling the Amorphous and Crystalline State of Multinary Alloy Nanoparticles in An Ionic Liquid. Nanomaterials 2018, 8, 903. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Suzuki, T.; Tomita, Y.; Hirano, M.; Okazaki, K.-I.; Kuwabata, S.; Torimoto, T. Compositional control of AuPt nanoparticles synthesized in ionic liquids by the sputter deposition technique. CrystEngComm 2012, 14, 4922. [Google Scholar] [CrossRef]
- Hirano, M.; Enokida, K.; Okazaki, K.-I.; Kuwabata, S.; Yoshida, H.; Torimoto, T. Composition-dependent electrocatalytic activity of AuPd alloy nanoparticles prepared via simultaneous sputter deposition into an ionic liquid. Phys. Chem. Chem. Phys. PCCP 2013, 15, 7286–7294. [Google Scholar] [CrossRef] [PubMed]
- Schwartzkopf, M.; Hinz, A.; Polonskyi, O.; Strunskus, T.; Löhrer, F.C.; Körstgens, V.; Müller-Buschbaum, P.; Faupel, F.; Roth, S.V. Role of Sputter Deposition Rate in Tailoring Nanogranular Gold Structures on Polymer Surfaces. ACS Appl. Mater. Interfaces 2017, 9, 5629–5637. [Google Scholar] [CrossRef]
- Schwartzkopf, M.; Santoro, G.; Brett, C.J.; Rothkirch, A.; Polonskyi, O.; Hinz, A.; Metwalli, E.; Yao, Y.; Strunskus, T.; Faupel, F.; et al. Real-Time Monitoring of Morphology and Optical Properties during Sputter Deposition for Tailoring Metal-Polymer Interfaces. ACS Appl. Mater. Interfaces 2015, 7, 13547–13556. [Google Scholar] [CrossRef]
- Scheeren, C.W.; Machado, G.; Teixeira, S.R.; Morais, J.; Domingos, J.B.; Dupont, J. Synthesis and characterization of Pt0 nanoparticles in imidazolium ionic liquids. J. Phys. Chem. B 2006, 110, 13011–13020. [Google Scholar] [CrossRef]
- Ozkar, S.; Finke, R.G. Nanocluster formation and stabilization fundamental studies: Ranking commonly employed anionic stabilizers via the development, then application, of five comparative criteria. J. Am. Chem. Soc. 2002, 124, 5796–5810. [Google Scholar] [CrossRef]
- He, Z.; Alexandridis, P. Nanoparticles in ionic liquids: Interactions and organization. Phys. Chem. Chem. Phys. PCCP 2015, 17, 18238–18261. [Google Scholar] [CrossRef]
- Liang, Y.; Hilal, N.; Langston, P.; Starov, V. Interaction forces between colloidal particles in liquid: Theory and experiment. Adv. Colloid Interface Sci. 2007, 134–135, 151–166. [Google Scholar] [CrossRef]
- Dupont, J. On the solid, liquid and solution structural organization of imidazolium ionic liquids. J. Braz. Chem. Soc. 2004, 15, 341–350. [Google Scholar] [CrossRef]
- Migowski, P.; Machado, G.; Texeira, S.R.; Alves, M.C.M.; Morais, J.; Traverse, A.; Dupont, J. Synthesis and characterization of nickel nanoparticles dispersed in imidazolium ionic liquids. Phys. Chem. Chem. Phys. PCCP 2007, 9, 4814–4821. [Google Scholar] [CrossRef] [PubMed]
- Redel, E.; Thomann, R.; Janiak, C. Use of ionic liquids (ILs) for the IL-anion size-dependent formation of Cr, Mo and W nanoparticles from metal carbonyl M(CO)6 precursors. Chem. Commun. (Camb.) 2008, 1789–1791. [Google Scholar] [CrossRef] [PubMed]
- Redel, E.; Thomann, R.; Janiak, C. First correlation of nanoparticle size-dependent formation with the ionic liquid anion molecular volume. Inorg. Chem. 2008, 47, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, G.S.; Machado, G.; Teixeira, S.R.; Fecher, G.H.; Morais, J.; Alves, M.C.M.; Dupont, J. Synthesis and characterization of catalytic iridium nanoparticles in imidazolium ionic liquids. J. Colloid Interface Sci. 2006, 301, 193–204. [Google Scholar] [CrossRef]
- Wender, H.; de Oliveira, L.F.; Migowski, P.; Feil, A.F.; Lissner, E.; Prechtl, M.H.G.; Teixeira, S.R.; Dupont, J. Ionic Liquid Surface Composition Controls the Size of Gold Nanoparticles Prepared by Sputtering Deposition. J. Phys. Chem. C 2010, 114, 11764–11768. [Google Scholar] [CrossRef]
- Fernandes, A.M.; Rocha, M.A.A.; Freire, M.G.; Marrucho, I.M.; Coutinho, J.A.P.; Santos, L.M.N.B.F. Evaluation of cation-anion interaction strength in ionic liquids. J. Phys. Chem. B 2011, 115, 4033–4041. [Google Scholar] [CrossRef]
- Dupont, J.; Scholten, J.D. On the structural and surface properties of transition-metal nanoparticles in ionic liquids. Chem. Soc. Rev. 2010, 39, 1780–1804. [Google Scholar] [CrossRef]
- Nguyen, M.T.; Yonezawa, T. Sputtering onto a liquid: Interesting physical preparation method for multi-metallic nanoparticles. Sci. Technol. Adv. Mater. 2018, 19, 883–898. [Google Scholar] [CrossRef]
- Mellah, M.; Zeitouny, J.; Gmouh, S.; Vaultier, M.; Jouikov, V. Oxidative self-coupling of aromatic compounds in ionic liquids. Electrochem. Commun. 2005, 7, 869–874. [Google Scholar] [CrossRef]
- Jarosik, A.; Krajewski, S.R.; Lewandowski, A.; Radzimski, P. Conductivity of ionic liquids in mixtures. J. Mol. Liq. 2006, 123, 43–50. [Google Scholar] [CrossRef]
- Baltazar, Q.Q.; Leininger, S.K.; Anderson, J.L. Binary ionic liquid mixtures as gas chromatography stationary phases for improving the separation selectivity of alcohols and aromatic compounds. J. Chromatogr. A 2008, 1182, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Finotello, A.; Bara, J.E.; Narayan, S.; Camper, D.; Noble, R.D. Ideal gas solubilities and solubility selectivities in a binary mixture of room-temperature ionic liquids. J. Phys. Chem. B 2008, 112, 2335–2339. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Rajian, J.R.; Hines, L.G.; Li, S.; Bartsch, R.A.; Quitevis, E.L. Nanostructural organization and anion effects in the optical Kerr effect spectra of binary ionic liquid mixtures. J. Phys. Chem. B 2008, 112, 13316–13325. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Zakeeruddin, S.M.; Humphry-Baker, R.; Grätzel, M. A Binary Ionic Liquid Electrolyte to Achieve ≥7% Power Conversion Efficiencies in Dye-Sensitized Solar Cells. Chem. Mater. 2004, 16, 2694–2696. [Google Scholar] [CrossRef]
- Shi, G.; Wang, Z.; Xia, J.; Bi, S.; Li, Y.; Zhang, F.; Xia, L.; Li, Y.; Xia, Y.; Xia, L. Mixed ionic liquids/graphene-supported platinum nanoparticles as an electrocatalyst for methanol oxidation. Electrochim. Acta 2014, 142, 167–172. [Google Scholar] [CrossRef]
- Cui, Y.; Biondi, I.; Chaubey, M.; Yang, X.; Fei, Z.; Scopelliti, R.; Hartinger, C.G.; Li, Y.; Chiappe, C.; Dyson, P.J. Nitrile-functionalized pyrrolidinium ionic liquids as solvents for cross-coupling reactions involving in situ generated nanoparticle catalyst reservoirs. Phys. Chem. Chem. Phys. PCCP 2010, 12, 1834–1841. [Google Scholar] [CrossRef]
- Zhao, M.; Gao, Y.; Zheng, L. Lyotropic liquid crystalline phases formed in binary mixture of 1-tetradecyl-3-methylimidazolium chloride/ethylammonium nitrate and its application in the dispersion of multi-walled carbon nanotubes. Colloids Surf. A Physicochem. Eng. Asp. 2010, 369, 95–100. [Google Scholar] [CrossRef]
- Morgan, D.; Ferguson, L.; Scovazzo, P. Diffusivities of Gases in Room-Temperature Ionic Liquids: Data and Correlations Obtained Using a Lag-Time Technique. Ind. Eng. Chem. Res. 2005, 44, 4815–4823. [Google Scholar] [CrossRef]
- Krossing, I.; Slattery, J.M.; Daguenet, C.; Dyson, P.J.; Oleinikova, A.; Weingärtner, H. Why are ionic liquids liquid? A simple explanation based on lattice and solvation energies. J. Am. Chem. Soc. 2006, 128, 13427–13434. [Google Scholar] [CrossRef]
- Nazet, A.; Sokolov, S.; Sonnleitner, T.; Makino, T.; Kanakubo, M.; Buchner, R. Densities, Viscosities, and Conductivities of the Imidazolium Ionic Liquids [Emim][Ac], [Emim][FAP], [Bmim][BETI], [Bmim][FSI], [Hmim][TFSI], and [Omim][TFSI]. J. Chem. Eng. Data 2015, 60, 2400–2411. [Google Scholar] [CrossRef]



| Chemical | Purity (%) | Halide Content (ppm) | Water Content (ppm) | Viscosity (mPa s) | Cation Volume (nm3) | Anion Volume (nm3) | Total Molecule Volume (nm3) |
|---|---|---|---|---|---|---|---|
| IL1 [Bmim][(Tf)2N] | >99 | <100 | 51 | 49.00 (25 °C) | 0.196 0.021 [63] | 0.232 0.015 [63] | 0.428 0.036 [63] |
| IL2 [Bmim][(Pf)2N] | >98 | <250 | 60 | 59.50 (40 °C) 115.90 (25 °C) [64] 153.30 (20 °C) [64] | 0.196 0.021 [63] | 0.292 0.021 B | 0.488 A |
| IL3 [BmPyr][(Tf)2N] | >99 | <100 | 80 | 94.00 (20 °C) | 0.221 0.015 [63] | 0.232 0.015 [63] | 0.453 0.030 [63] |
| Anisole | 99 | - | <1000 | 1.52 (15 °C) | - | - | 0.181 C |
| Element in IL Substrate | Start Pressure (Pa) | Ignition Pressure (Pa) | Ignition Power (W) | Pre-Clean Duration (s) | Deposition Pressure (Pa) | Deposition Power (W) | Deposition Duration (min) | Deposition Rate (nm/s) | Film Thickness (nm) |
|---|---|---|---|---|---|---|---|---|---|
| Ag in pure IL1 | 1.33 | 20 | 120 | 0.5 | 30 | 40 | 0.2216 | ||
| Ag in IL1/IL2 mixture | 1.33 | 20 | 120 | 0.5 | 30 | 40 | 0.2216 | ||
| Ag in IL1/IL3 mixture | 1.33 | 20 | 120 | 0.5 | 30 | 40 | 0.2216 | ||
| Au in IL1/IL3 mixture | 1.33 | 20 | 120 | 0.5 | 30 | 30 | 0.2275 | ||
| Cu in IL1/IL3 mixture | 1.33 | 20 | 240 | 0.5 | 30 | 120 | 0.080 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meischein, M.; Fork, M.; Ludwig, A. On the Effects of Diluted and Mixed Ionic Liquids as Liquid Substrates for the Sputter Synthesis of Nanoparticles. Nanomaterials 2020, 10, 525. https://doi.org/10.3390/nano10030525
Meischein M, Fork M, Ludwig A. On the Effects of Diluted and Mixed Ionic Liquids as Liquid Substrates for the Sputter Synthesis of Nanoparticles. Nanomaterials. 2020; 10(3):525. https://doi.org/10.3390/nano10030525
Chicago/Turabian StyleMeischein, Michael, Marvin Fork, and Alfred Ludwig. 2020. "On the Effects of Diluted and Mixed Ionic Liquids as Liquid Substrates for the Sputter Synthesis of Nanoparticles" Nanomaterials 10, no. 3: 525. https://doi.org/10.3390/nano10030525
APA StyleMeischein, M., Fork, M., & Ludwig, A. (2020). On the Effects of Diluted and Mixed Ionic Liquids as Liquid Substrates for the Sputter Synthesis of Nanoparticles. Nanomaterials, 10(3), 525. https://doi.org/10.3390/nano10030525





