Pyrene Coating Transition Metal Disulfides as Protection from Photooxidation and Environmental Aging
Abstract
1. Introduction
2. Materials and Methods
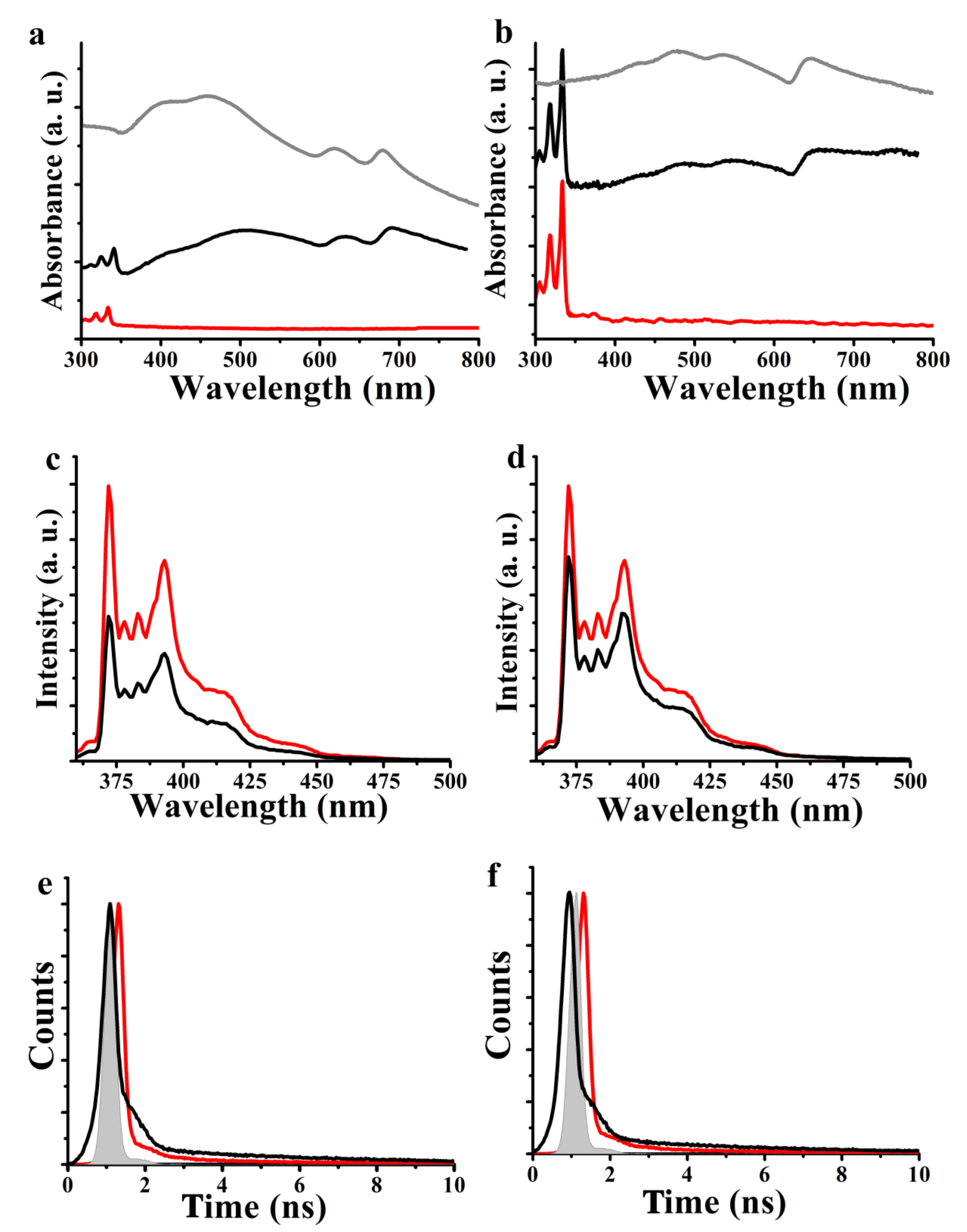
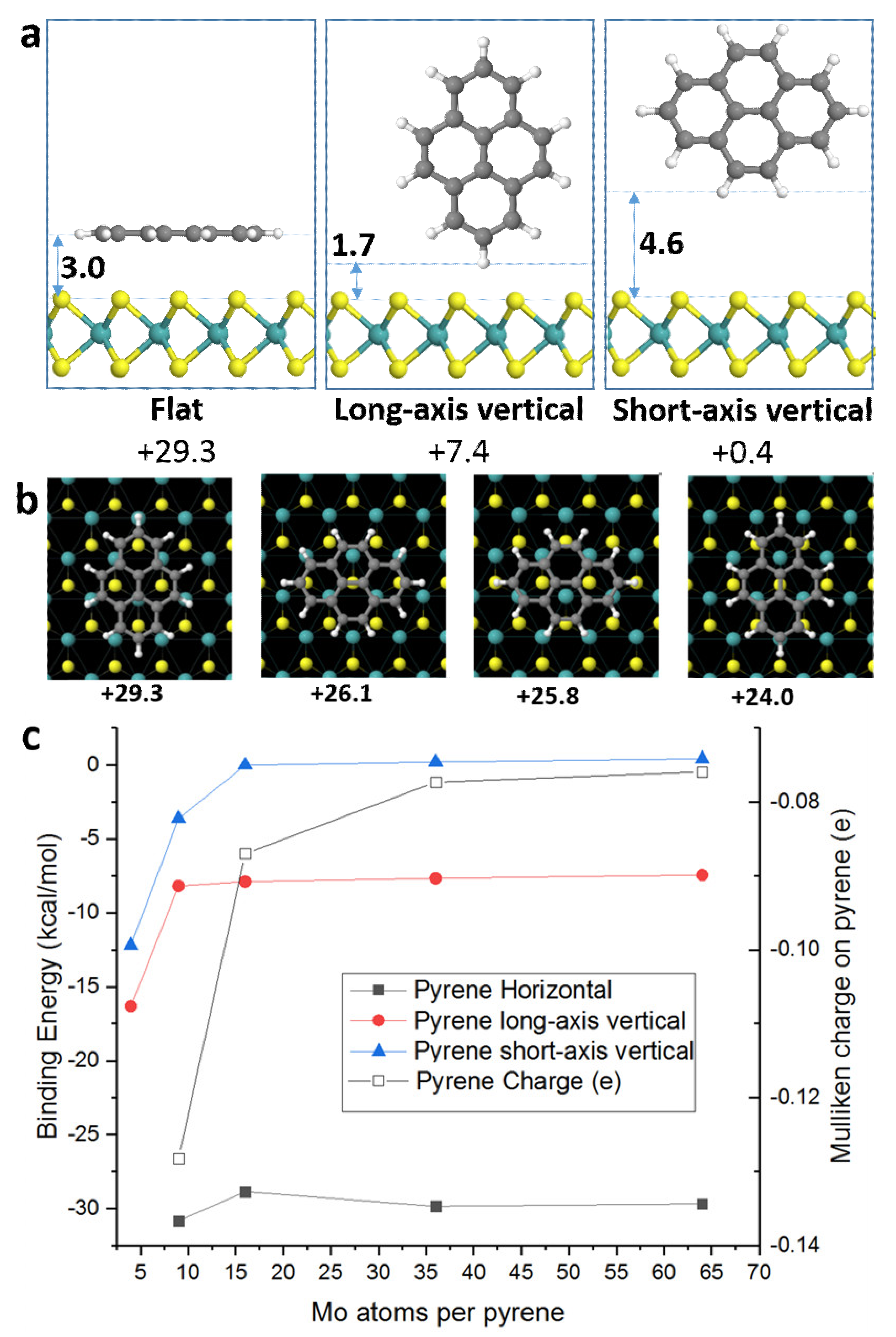
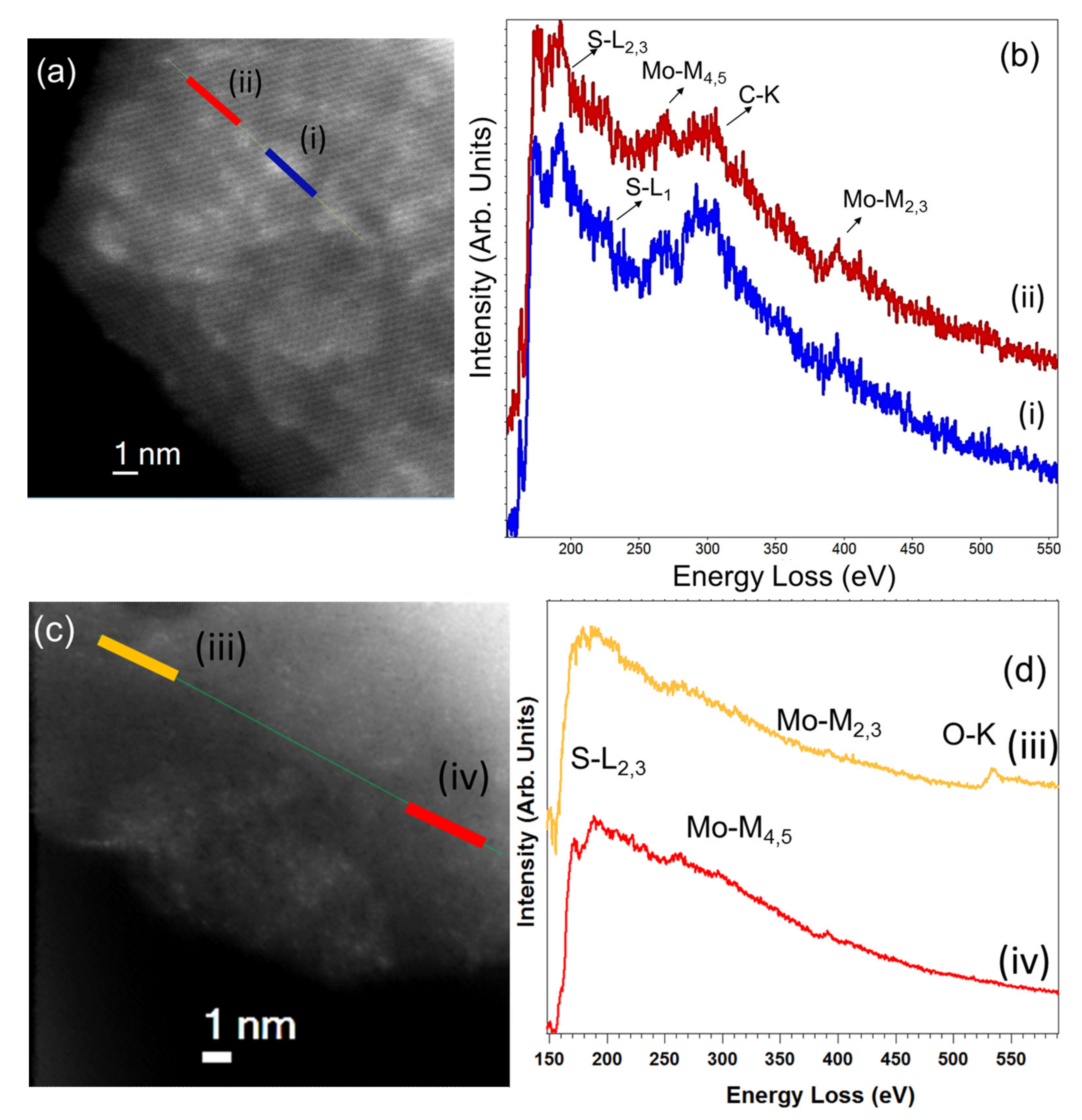
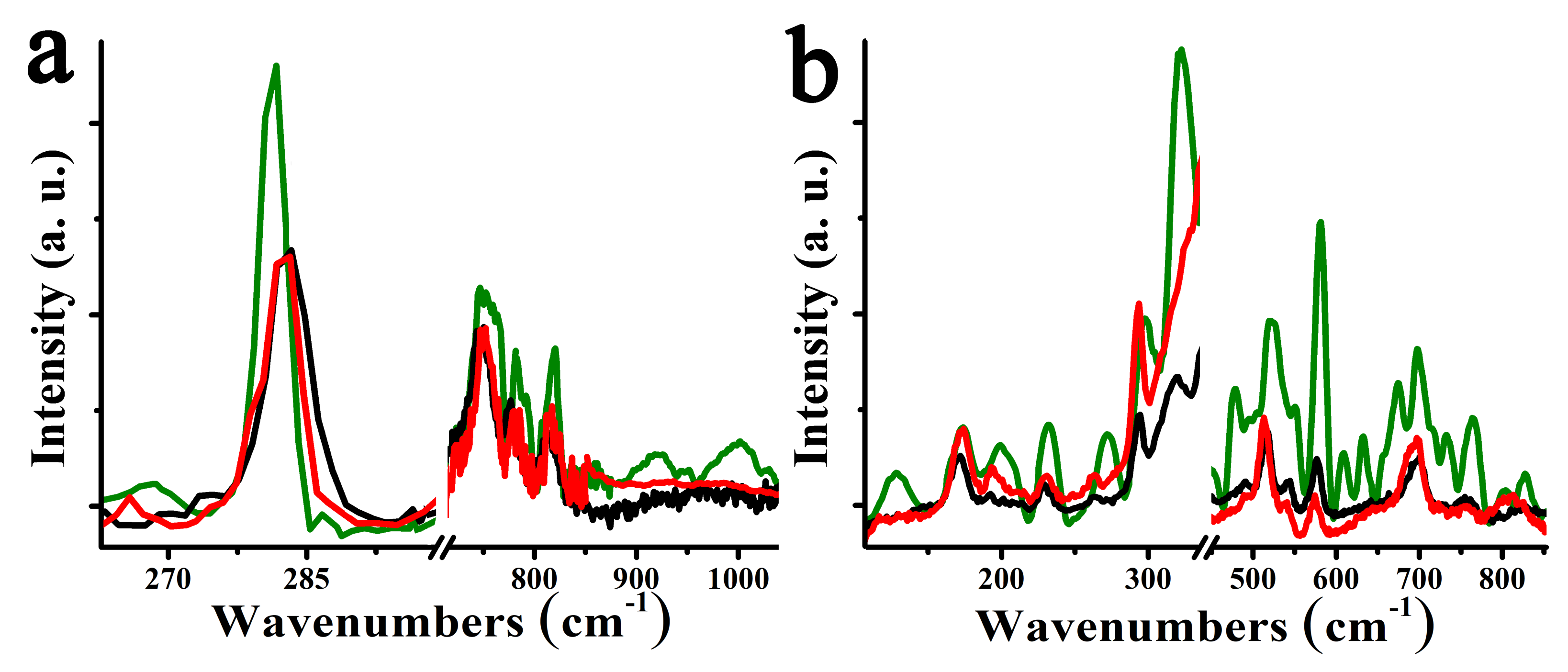
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pagona, G.; Bittencourt, C.; Arenal, R.; Tagmatarchis, N. Exfoliated Semiconducting Pure 2H-MoS2 and 2H-WS2 Assisted by Chlorosulfonic Acid. Chem. Commun. 2015, 51, 12950–12953. [Google Scholar] [CrossRef]
- Fan, X.; Xu, P.; Li, Y.C.; Zhou, D.; Sun, Y.; Nguyen, M.A.T.; Terrones, M.; Mallouk, T.E. Controlled Exfoliation of MoS2 Crystals into Trilayer Nanosheets. J. Am. Chem. Soc. 2016, 138, 5143–5149. [Google Scholar] [CrossRef]
- Niu, L.; Coleman, J.N.; Zhang, H.; Shin, H.; Chhowalla, M.; Zheng, Z. Production of Two-Dimensional Nanomaterials Via Liquid-Based Direct Exfoliation. Small 2016, 12, 272–293. [Google Scholar] [CrossRef]
- Grayfer, E.D.; Kozlova, M.N.; Fedorov, V.E. Colloidal 2D Nanosheets of MoS2 and Other Transition Metal Dichalcogenides Through Liquid-Phase Exfoliation. Adv. Colloid Interface Sci. 2017, 245, 40–61. [Google Scholar] [CrossRef]
- Chow, P.K.; Jacobs-Gedrim, R.B.; Gao, J.; Lu, T.-M.; Yu, B.; Terrones, H.; Koratkar, N. Defect-Induced Photoluminescence in Monolayer Semiconducting Transition Metal Dichalcogenides. ACS Nano 2015, 9, 1520–1527. [Google Scholar] [CrossRef]
- Tongay, S.; Zhou, J.; Ataca, C.; Liu, J.; Kang, J.S.; Matthews, T.S.; You, L.; Li, J.; Grossman, J.C.; Wu, J. Broad-Range Modulation of Light Emission in Two-Dimensional Semiconductors by Molecular Physisorption Gating. Nano Lett. 2013, 13, 2831–2836. [Google Scholar] [CrossRef]
- Pritchard, C.; Midgley, J.W. The Effect of Humidity on the Friction and Life of Unbonded Molybdenum Disulfide Films. Wear 1969, 13, 39–50. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, Y.-J.; Liu, M.; Peterson, A.; Hurt, R.H. Oxidation Suppression During Hydrothermal Phase Reversion Allows Synthesis of Monolayer Semiconducting MoS2 in Stable Aqueous Suspension. Tribol. Lett. 2014, 53, 329–336. [Google Scholar] [CrossRef]
- Tagawa, M.; Muromoto, M.; Hachiue, S.; Yokota, K.; Ohmae, N.; Matsumoto, K.; Suzuki, M. Hyperthermal Atomic Oxygen Interaction with MoS2 Lubricants and Relevance to Space Environmental Effects in Low Earth Orbit-Effects on Friction Coefficient and Wear-Life. Tribol. Lett. 2005, 18, 437–443. [Google Scholar] [CrossRef]
- Liang, T.; Sawyer, W.G.; Perry, S.S.; Sinnott, S.B.; Phillpot, S.R. Energetics of Oxidation in MoS2 Nanoparticles by Density Functional Theory. J. Phys. Chem. C 2011, 115, 10606–10616. [Google Scholar]
- Liang, T.; Sawyer, W.G.; Perry, S.S.; Sinnott, S.B.; Phillpot, S.R. First-Principles Determination of Static Potential Energy Surfaces for Atomic Friction in MoS2 and MoO3. Phys. Rev. B 2008, 77, 104105. [Google Scholar] [CrossRef]
- Gao, J.; Li, B.; Tan, J.; Chow, P.; Lu, T.-M.; Koratkar, N. Aging of Transition Metal Dichalcogenide Monolayers. ACS Nano 2016, 10, 2628–2635. [Google Scholar] [CrossRef] [PubMed]
- Leonhardt, A.; Chiappe, D.; Asselberghs, I.; Radu, I.; de Gendt, S. Improving MOCVD MoS2 Electrical Performance: Impact of Minimized Water and Air Exposure Conditions. IEEE Electron. Dev. Lett. 2017, 38, 1606–1609. [Google Scholar] [CrossRef]
- Lee, G.-H.; Cui, X.; Kim, Y.D.; Arefe, G.; Zhang, X.; Lee, C.-H.; Ye, F.; Watanabe, K.; Taniguchi, T.; Kim, P.; et al. Highly Stable, Dual-Gated MoS2 Transistors Encapsulated by Hexagonal Boron Nitride with Gate-Controllable Contact, Resistance, and Threshold Voltage. ACS Nano 2015, 9, 7019–7026. [Google Scholar] [CrossRef]
- Ahn, S.; Kim, G.; Nayak, P.K.; Yoon, S.I.; Lim, H.; Shin, H.-J.; Shin, H.S. Prevention of Transition Metal Dichalcogenide Photodegradation by Encapsulation with h-BN Layers. ACS Nano 2016, 10, 8973–8979. [Google Scholar] [CrossRef]
- Kim, S.-Y.; Kwak, J.; Kim, J.H.; Lee, J.-U.; Jo, Y.; Kim, S.Y.; Cheong, H.; Lee, Z.; Kwon, S.-Y. Substantial Improvements of Long-Term Stability in Encapsulation-Free WS2 Using Highly Interacting Graphene Substrate. 2D Mater. 2017, 4, 011007. [Google Scholar] [CrossRef]
- Wood, J.D.; Wells, S.A.; Jariwala, D.; Chen, K.; Cho, E.; Sangwan, V.K.; Liu, X.; Lauhon, L.J.; Marks, T.J.; Hersam, M.C. Effective Passivation of Exfoliated Black Phosphorus Transistors Against Ambient Degradation. Nano Lett. 2014, 14, 6964–6970. [Google Scholar] [CrossRef]
- Jeanguillaume, C.; Colliex, C. Spectrum-Image: The Next Step in EELS Digital Acquisition and Processing. Ultramicroscopy 1989, 28, 252–257. [Google Scholar] [CrossRef]
- Arenal, A.; de la Peña, F.; Stephan, O.; Walls, M.; Tence, M.; Loiseau, A.; Colliex, C. Extending the Analysis of EELS Spectrum-Imaging Data, From Elemental to Bond Mapping in Complex Nanostructures. Ultramicroscopy 2008, 109, 32–38. [Google Scholar] [CrossRef]
- Briddon, P.; Jones, R. LDA Calculations Using a Basis of Gaussian Orbitals. Phys. Status Sol. B 2000, 217, 131–171. [Google Scholar] [CrossRef]
- Rayson, M.; Briddon, P. Highly Efficient Method for Kohn-Sham Density Functional Calculations of 500–10,000 Atom Systems. Phys. Rev. B 2009, 80, 205104–205115. [Google Scholar] [CrossRef]
- Briddon, P.R.; Rayson, M.J. Accurate Kohn-Sham DFT with the Speed of Tight Binding: Current Techniques and Future Directions in Materials Modelling. Phys. Status Sol. B 2011, 248, 1309–1318. [Google Scholar] [CrossRef]
- Hartwigsen, C.; Gøedecker, S.; Hutter, J. Relativistic Separable Dual-Space Gaussian Pseudopotentials from H to Rn. Phys. Rev. B 1998, 58, 3641–3662. [Google Scholar] [CrossRef]
- Monkhorst, H.J.; Pack, J.D. Special Points for Brillouin-zone Integrations. Phys. Rev. B 1976, 13, 5188–5192. [Google Scholar] [CrossRef]
- Chou, S.S.; Sai, N.; Lu, P.; Coker, E.N.; Liu, S.; Artyushkova, K.; Luk, T.S.; Kaehr, B.; Brinker, C.J. Understanding Catalysis in a Multiphasic Tw-Dimensional Transition Metal Dichalcogenide. Nat. Commun. 2015, 6, 8311. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, F.; Rotunno, E.; Cinquanta, E.; Campi, D.; Bonnini, E.; Kaplan, D.; Lazzarini, L.; Bernasconi, M.; Ferrari, C.; Longo, M.; et al. Novel Near-Infrared Emission from Crystal Defects in MoS2 Multilayer Flakes. Nat. Commun. 2016, 7, 13044. [Google Scholar] [CrossRef]
- Vallan, L.; Canton-Vitoria, R.; Gobeze, H.B.; Jang, Y.; Arenal, R.; Benito, A.M.; Maser, W.K.; D’Souza, F.; Tagmatarchis, N. Interfacing Transition Metal Dichalcogenides with Carbon Dots for Managing Photoinduced Energy and Charge-Transfer Processes. J. Am. Chem. Soc. 2018, 140, 13488–13496. [Google Scholar] [CrossRef]
- Canton-Vitoria, R.; Sayed-Ahmad-Baraza, Y.; Pelaez-Fernandez, M.; Arenal, R.; Bittencourt, C.; Ewels, C.P.; Tagmatarchis, N. Functionalization of MoS2 with 1,2-Dithiolanes: Toward Donor-Acceptor Nanohybrids for Energy Conversion. npj 2D Mater. Appl. 2017, 1, 13. [Google Scholar] [CrossRef]
- Canton-Vitoria, R.; Gobeze, H.B.; Blas-Ferrando, V.M.; Ortiz, J.; Jang, Y.; Fernandez-Lazaro, F.; Sastre-Santos, A.; Nakanishi, Y.; Shinohara, H.; d’Souza, F.; et al. Excited State Charge Transfer in Covalently Functionalized MoS2 with Zinc Phthalocyanine Donor-Acceptor Hybrid. Angew. Chem. Int. Ed. 2019, 58, 5712–5717. [Google Scholar] [CrossRef]
- Aleksandrov, L.; Komatsu, T.; Iordanova, R.; Dimitriev, Y. Study of Molybdenum Coordination State and Crystallization Behavior in MoO3-La2O3-B2O3 Glasses by Raman Spectroscopy. J. Phys. Chem. Solids 2011, 72, 263–268. [Google Scholar] [CrossRef]
- Windom, B.C.; Sawyer, W.G.; Hahn, D.W. A Raman Spectroscopic Study of MoS2 and MoO3: Applications to Tribological Systems. Tribol. Lett. 2011, 42, 301–310. [Google Scholar] [CrossRef]
- Daniel, M.F.; Desbat, B.; Lassegues, J.C. Infrared and Raman Study of WO3 Tungsten Trioxides and WO3, xH2O Tungsten Trioxide Hydrates. J. Solid State Chem. 1987, 67, 235–247. [Google Scholar] [CrossRef]
- Ayala, P.; Arenal, R.; Rummeli, M.; Rubio, A.; Pichler, T. The Doping of Carbon Nanotubes with Nitrogen and Their Potential Applications. Carbon 2010, 48, 575–586. [Google Scholar] [CrossRef]
- Arenal, R.; Blase, X.; Loiseau, A. Boron-Nitride and Boron-Carbonitride Nanotubes: Synthesis, Characterization and Theory. Adv. Phys. 2010, 59, 101–179. [Google Scholar] [CrossRef]
- Alvarez, L.; Almadori, Y.; Arenal, R.; Babaa, R.; Michel, T.; Leparc, R.; Bantignies, J.-L.; Hermet, P.; Sauvajol, J.-L. Charge Transfer Evidence Between Carbon Nanotubes and Encapsulated Conjugated Oligomers. J. Phys. Chem. C 2011, 115, 11898–11905. [Google Scholar] [CrossRef]
- Arenal, R.; De Matteis, L.; Custardoy, L.; Mayoral, A.; Tence, M.; Grazu, V.; de la Fuente, J.M.; Marquina, C.; Ibarra, M.R. Spatially-Resolved EELS Analysis of Antibody Distribution on Biofunctionalized Magnetic Nanoparticles. ACS Nano 2013, 7, 4006–4013. [Google Scholar] [CrossRef]
- Albiter, M.A.; Huirache-Acuña, R.; Paraguay-Delgado, F.; Rico, J.L.; Alonso-Nuñez, G. Synthesis of MoS2 Nanorods and Their Catalytic Test in the HDS of Dibenzothiophene. Nanotechnology 2006, 17, 3473–3481. [Google Scholar] [CrossRef]
- Santosh, K.C.; Longo, R.C.; Wallace, R.M.; Cho, K. Surface Oxidation Energetics and Kinetics on MoS2 Monolayer. J. Appl. Phys. 2015, 117, 135301–135309. [Google Scholar]
- Addou, R.; McDonnell, S.; Barrera, D.; Guo, Z.; Azcat, A.; Wang, J.; Zhu, H.; Hinkle, C.L.; Quevedo-Lopez, M.; Alshareef, H.N.; et al. Impurities and Electronic Property Variations of Natural MoS2 Crystal Surfaces. ACS Nano 2015, 9, 9124–9133. [Google Scholar] [CrossRef]
- Addou, R.; Colombo, L.; Wallace, R.M. Surface Defects on Natural MoS2. ACS Appl. Mater. Interfaces 2015, 7, 11921–11929. [Google Scholar] [CrossRef]
- Chow, P.; Singh, E.; Viana, B.; Gao, J.; Luo, J.; Li, J.; Lin, Z.; Elías, A.; Shi, Y.; Wang, Z.; et al. Wetting of Mono and Few-Layered WS2 and Mos2 Films Supported on Si/SiO2 Substrates. ACS Nano 2015, 9, 3023–3031. [Google Scholar] [CrossRef] [PubMed]
- Afanasiev, P.; Lorentz, C. Oxidation of Nanodispersed MoS2 in Ambient Air: The Products and the Mechanistic Steps. J. Phys. Chem. C 2019, 123, 7486–7494. [Google Scholar] [CrossRef]
- Kozbial, A.; Gong, X.; Liu, H.; Li, L. Understanding the Intrinsic Water Wettability of Molybdenum Disulfide (MoS2). Langmuir 2015, 31, 8429–8435. [Google Scholar] [CrossRef] [PubMed]
- Barja, S.; Refaely-Abramson, S.; Schuler, B.; Qiu, D.; Pulkin, A.; Wickenburg, S.; Ryu, H.; Ugeda, M.; Kastl, C.; Chen, C. Identifying substitutional oxygen as a prolific point defect in monolayer transition metal dichalcogenides. Nat. Commun. 2019, 10, 3382. [Google Scholar] [CrossRef]
- Hu, Z.; Avila, J.; Wang, X.; Leong, J.; Zhang, Q.; Liu, Y.; Asensio, M.; Lu, J.; Carvalho, A.; Sow, C. The Role of Oxygen Atoms on Excitons at the Edges of Monolayer WS2. Nano Lett. 2019, 19, 4641. [Google Scholar] [CrossRef]
- Canton-Vitoria, R.; Stangel, C.; Tagmatarchis, N. Electrostatic Association of Ammonium-Functionalized Layered-Transition-Metal Dichalcogenides with an Anionic Porphyrin. ACS Appl. Mater. Interfaces 2018, 10, 23476–23480. [Google Scholar] [CrossRef]
- Canton-Vitoria, R.; Vallan, L.; Urriolabeitia, E.; Benito, A.M.; Maser, W.K.; Tagmatarchis, N. Electronic Interactions in Illuminated Carbon Dot/MoS2 Ensembles and Electrocatalytic Activity Towards Hydrogen Evolution. Chem. Eur. J. 2018, 24, 10468–10474. [Google Scholar] [CrossRef]
- Bertolazzi, S.; Gobbi, M.; Zhao, Y.; Backes, C.; Samorì, P. Molecular Chemistry Approaches for Tuning the Properties of Two-Dimensional Transition Metal Dichalcogenides. Chem. Soc. Rev. 2018, 47, 6845–6888. [Google Scholar] [CrossRef]
- Stergiou, A.; Tagmatarchis, N. Molecular Functionalization of 2D MoS2 Nanosheets. Chem. Eur. J. 2018, 24, 18246–18257. [Google Scholar] [CrossRef]
- Canton-Vitoria, R.; Istif, E.; Hernandez-Ferrer, J.; Benito, A.M.; Maser, W.K.; Tagmatarchis, N. Integrating Water-Soluble Polythiophene with Transition Metal Dichalcogenides for Managing Photoinduced Processes. ACS Appl. Mater. Interfaces 2019, 11, 5947–5956. [Google Scholar] [CrossRef]
- Mirabelli, G.; McGeough, C.; Schmidt, M.; McCarthy, E.; Monaghan, S.; Povey, I.; McCarthy, M.; Gity, F.; Nagle, R.; Hughes, G.; et al. Air Sensitivity of MoS2, MoSe2, MoTe2, HfS2, and HfSe2. J. Appl. Phys. 2016, 120, 125102–125111. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canton-Vitoria, R.; Sayed-Ahmad-Baraza, Y.; Humbert, B.; Arenal, R.; Ewels, C.P.; Tagmatarchis, N. Pyrene Coating Transition Metal Disulfides as Protection from Photooxidation and Environmental Aging. Nanomaterials 2020, 10, 363. https://doi.org/10.3390/nano10020363
Canton-Vitoria R, Sayed-Ahmad-Baraza Y, Humbert B, Arenal R, Ewels CP, Tagmatarchis N. Pyrene Coating Transition Metal Disulfides as Protection from Photooxidation and Environmental Aging. Nanomaterials. 2020; 10(2):363. https://doi.org/10.3390/nano10020363
Chicago/Turabian StyleCanton-Vitoria, Ruben, Yuman Sayed-Ahmad-Baraza, Bernard Humbert, Raul Arenal, Christopher P. Ewels, and Nikos Tagmatarchis. 2020. "Pyrene Coating Transition Metal Disulfides as Protection from Photooxidation and Environmental Aging" Nanomaterials 10, no. 2: 363. https://doi.org/10.3390/nano10020363
APA StyleCanton-Vitoria, R., Sayed-Ahmad-Baraza, Y., Humbert, B., Arenal, R., Ewels, C. P., & Tagmatarchis, N. (2020). Pyrene Coating Transition Metal Disulfides as Protection from Photooxidation and Environmental Aging. Nanomaterials, 10(2), 363. https://doi.org/10.3390/nano10020363






