Thiolation of Chitosan Loaded over Super-Magnetic Halloysite Nanotubes for Enhanced Laccase Immobilization
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
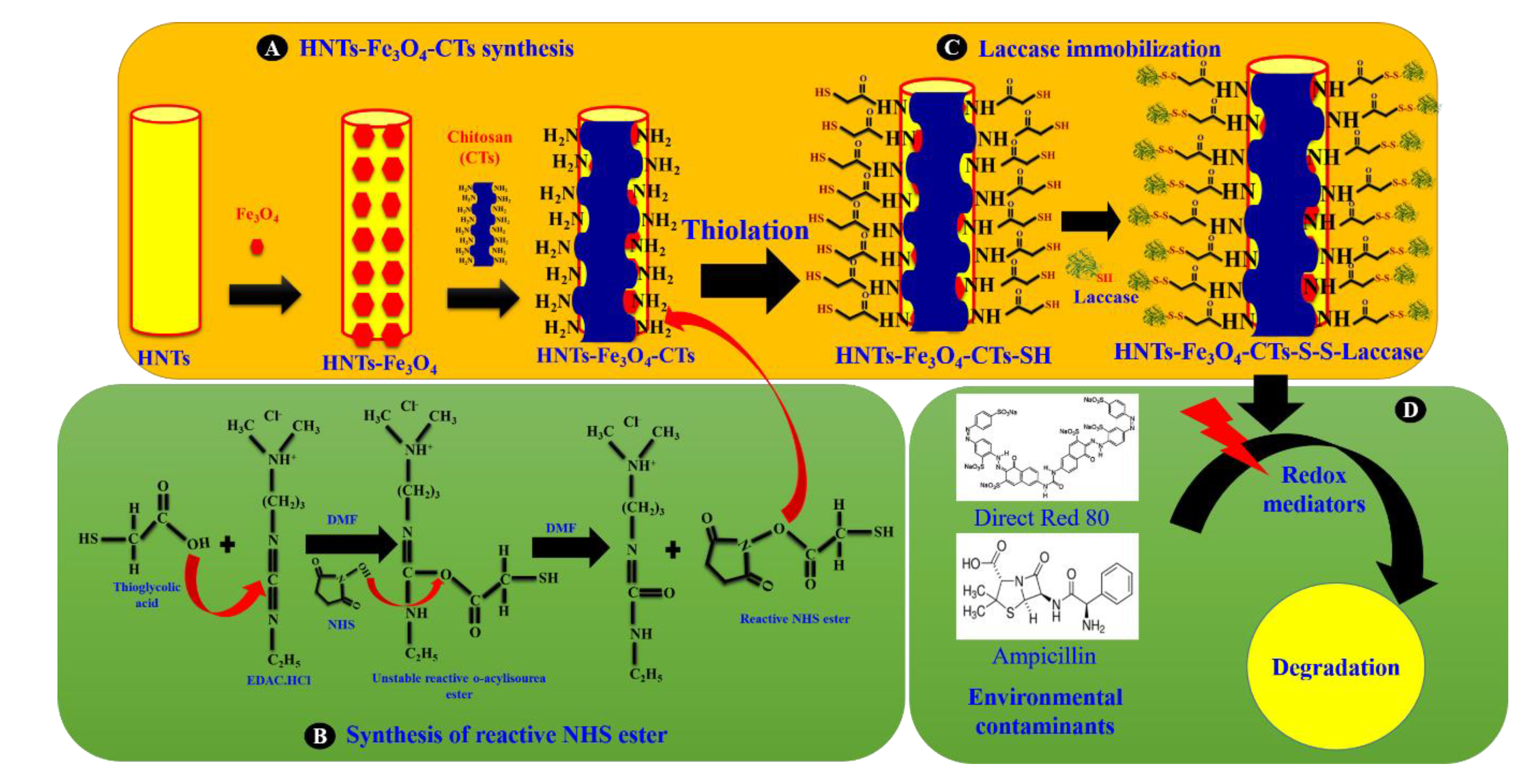
2.2. Synthesis of HNTs-Fe3O4-CTs-SH
2.3. Laccase Immobilization Experiment
2.4. Characterizations
2.5. Biocatalysis of Immobilized Laccase
2.6. Immobilized Laccase Mediated Degradation of Environmental Pollutants
3. Results and Discussion
3.1. Synthesis
3.2. Characterizations
3.2.1. SEM and HR-TEM Analysis
3.2.2. XRD, FT-IR, VSM, and XPS Analysis
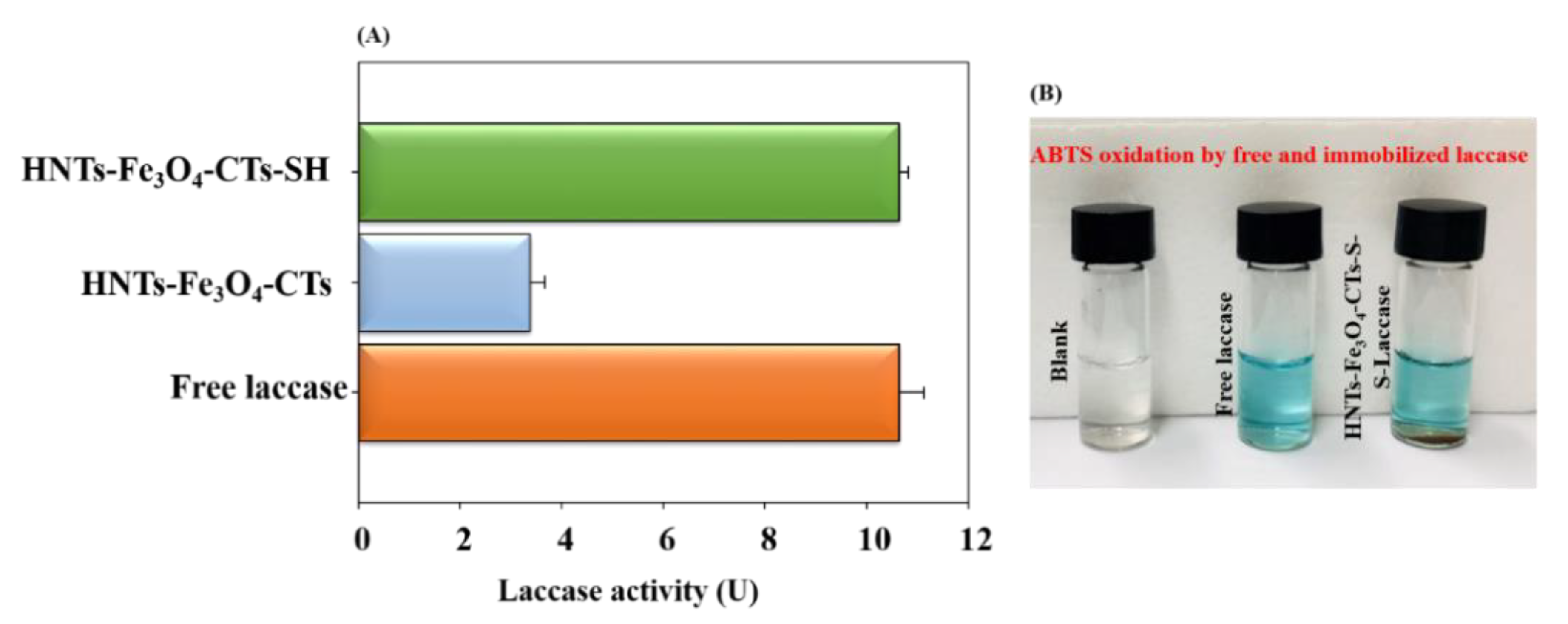
3.3. Laccase Immobilization on HNTs-Fe3O4-CTs-SH
3.4. Biocatalytic Performance of the HNTs-Fe3O4-CTs-S-S-Laccase
3.5. Application of HNTs-Fe3O4-CTs-S-S-Laccase in Environmental Pollutants Removal
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Datta, S.; Veena, R.; Samuel, M.S.; Selvarajan, E. Immobilization of laccases and applications for the detection and remediation of pollutants: A review. Environ. Chem. Lett. 2020, 1–18. [Google Scholar] [CrossRef]
- Datta, S.; Rajnish, K.N.; Samuel, M.S.; Pugazlendhi, A.; Selvarajan, E. Metagenomic applications in microbial diversity, bioremediation, pollution monitoring, enzyme and drug discovery. A review. Environ. Chem. Lett. 2020, 18, 1229–1241. [Google Scholar] [CrossRef]
- Liu, Y.Q.; Maulidiany, N.; Zeng, P.; Heo, S. Decolourization of azo, anthraquinone and triphenylmethane dyes using aerobic granules: Acclimatization and long-term stability. Chemosphere 2020, 263, 128312. [Google Scholar] [CrossRef] [PubMed]
- Taoufik, N.; Boumya, W.; Janani, F.Z.; Elhalil, A.; Mahjoubi, F.Z.; Barka, N. Removal of emerging pharmaceutical pollutants: A systematic mapping study review. J. Environ. Chem. Eng. 2020, 8, 104251. [Google Scholar] [CrossRef]
- Antibiotics, N.; Family, H.; Sanchez, H.M.; Whitener, V.A.; Thulsiraj, V.; Amundson, A.; Collins, C.; Duran-gonzalez, M.; Giragossian, E.; Hornstra, A.; et al. Antibiotic Resistance of Escherichia coli Isolated from Owned Retail Broiler Chicken Meat. Animals 2020, 10, 2217. [Google Scholar]
- Tabla-Hernández, J.; Rodríguez-Espinosa, P.F.; Hernandez-Ramirez, A.G.; Mendoza-Pérez, J.A.; Cano-Aznar, E.R.; Martínez-Tavera, E. Treatment of Eutrophic water and wastewater from Valsequillo Reservoir, Puebla, Mexico by Means of Ozonation: A multiparameter approach. Water 2018, 10, 1790. [Google Scholar] [CrossRef]
- Ulu, A.; Birhanli, E.; Boran, F.; Köytepe, S.; Yesilada, O.; Ateş, B. Laccase-conjugated thiolated chitosan-Fe3O4 hybrid composite for biocatalytic degradation of organic dyes. Int. J. Biol. Macromol. 2020, 150, 871–884. [Google Scholar] [CrossRef]
- Yavaşer, R.; Karagözler, A.A. Laccase immobilized polyacrylamide-alginate cryogel: A candidate for treatment of effluent. Process Biochem. 2010, 101, 137–146. [Google Scholar]
- Ghodake, G.S.; Yang, J.; Shinde, S.S.; Mistry, B.M.; Kim, D.Y.; Sung, J.S.; Kadam, A.A. Paper waste extracted A-cellulose fibers super-magnetized and chitosan-functionalized for covalent laccase immobilization. Bioresour. Technol. 2018, 261, 420–427. [Google Scholar] [CrossRef]
- Ba, S.; Arsenault, A.; Hassani, T.; Jones, J.P.; Cabana, H. Laccase immobilization and insolubilization: From fundamentals to applications for the elimination of emerging contaminants in wastewater treatment. Crit. Rev. Biotechnol. 2013, 33, 404–418. [Google Scholar] [CrossRef]
- Tully, J.; Yendluri, R.; Lvov, Y. Halloysite Clay Nanotubes for Enzyme Immobilization. Biomacromolecules 2016, 17, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Song, W.; Zhao, Q.; Zhang, H. Halloysite nanotubes in polymer science: Purification, characterization, modification and applications. Nanotechnol. Rev. 2020, 9, 323–344. [Google Scholar] [CrossRef]
- Kadam, A.A.; Jang, J.; Jee, S.C.; Sung, J.S.; Lee, D.S. Chitosan-functionalized supermagnetic halloysite nanotubes for covalent laccase immobilization. Carbohydr. Polym. 2018, 194, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Kadam, A.A.; Shinde, S.K.; Ghodake, G.S.; Saratale, G.D.; Saratale, R.G.; Sharma, B.; Hyun, S.; Sung, J.-S. Chitosan-Grafted Halloysite Nanotubes-Fe3O4 Composite for Laccase-Immobilization and Sulfamethoxazole-Degradation. Polymers 2020, 12, 2221. [Google Scholar] [CrossRef] [PubMed]
- Hajizadeh, Z.; Maleki, A.; Rahimi, J.; Eivazzadeh-Keihan, R. Halloysite Nanotubes Modified by Fe3O4 Nanoparticles and Applied as a Natural and Efficient Nanocatalyst for the SymmetricalHantzsch Reaction. Silicon 2020, 12, 1247–1256. [Google Scholar] [CrossRef]
- Kadam, A.A.; Jang, J.; Lee, D.S. Supermagnetically Tuned Halloysite Nanotubes Functionalized with Aminosilane for Covalent Laccase Immobilization. ACS Appl. Mater. Interfaces 2017, 9, 15492–15501. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, Y.; Wu, C.; Xiong, S.; Zhou, C. Chitosan/halloysite nanotubes bionanocomposites: Structure, mechanical properties and biocompatibility. Int. J. Biol. Macromol. 2012, 51, 566–575. [Google Scholar] [CrossRef]
- Kim, M.; Jee, S.C.; Sung, J.-S.; Kadam, A.A. Anti-proliferative applications of laccase immobilized on super-magnetic chitosan-functionalized halloysite nanotubes. Int. J. Biol. Macromol. 2018, 118, 228–237. [Google Scholar] [CrossRef]
- Federer, C.; Kurpiers, M.; Bernkop-Schnürch, A. Thiolated Chitosans: A Multi-talented Class of Polymers for Various Applications. Biomacromolecules 2020. [Google Scholar] [CrossRef]
- Leichner, C.; Jelkmann, M.; Bernkop-Schnürch, A. Thiolated polymers: Bioinspired polymers utilizing one of the most important bridging structures in nature. Adv. Drug Deliv. Rev. 2019, 151–152, 191–221. [Google Scholar] [CrossRef]
- Hanif, M.; Zaman, M.; Qureshi, S. Thiomers: A Blessing to Evaluating Era of Pharmaceuticals. Int. J. Polym. Sci. 2015, 2015, 1–9. [Google Scholar] [CrossRef]
- Zhu, X.; Su, M.; Tang, S.; Wang, L.; Liang, X.; Meng, F.; Hong, Y. Zhu-2012-Synthesis of Thiolat.pdf; 2012; pp. 1973–1982. [Google Scholar]
- Verma, M.L.; Kumar, S.; Das, A.; Randhawa, J.S.; Chamundeeswari, M. Chitin and chitosan-based support materials for enzyme immobilization and biotechnological applications. Environ. Chem. Lett. 2020, 18, 315–323. [Google Scholar] [CrossRef]
- Teymourian, H.; Salimi, A.; Khezrian, S. Biosensors and Bioelectronics Fe3O4 magnetic nanoparticles / reduced graphene oxide nanosheets as a novel electrochemical and bioeletrochemical sensing platform. Biosens. Bioelectron. 2013, 49, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, K.; Singh, P.; Singhal, R.K. New Chitosan-Thiomer: An Efficient Colorimetric Sensor and Effective Sorbent for Mercury at Ultralow Concentration. ACS Appl. Mater. Interfaces 2015, 7, 26069–26078. [Google Scholar] [CrossRef] [PubMed]
- Nandiyanto, A.B.D.; Oktiani, R.; Ragadhita, R. How to read and interpret ftir spectroscope of organic material. Indones. J. Sci. Technol. 2019, 4, 97–118. [Google Scholar] [CrossRef]
- Taylor, P.; Trofimov, B.A.; Sinegovskaya, L.M.; Gusarova, N.K. Vibrations of the S–S bond in elemental sulfur and organic polysulfides: A structural guide. J. Sulfur Chem. 2009, 30, 518–554. [Google Scholar]
- Ji, Y.; Yang, X.; Ji, Z.; Zhu, L.; Ma, N.; Chen, D.; Jia, X.; Tang, J.; Cao, Y. DFT-Calculated IR Spectrum Amide I, II, and III Band Contributions of N-Methylacetamide Fine Components. ACS Omega 2020, 5, 8572–8578. [Google Scholar] [CrossRef]
- Sharma, B.; Kadam, A.A.; Sung, J.S.; Myung, J. ha Surface tuning of halloysite nanotubes with Fe3O4 and 3-D MnO2 nanoflakes for highly selective and sensitive acetone gas sensing. Ceram. Int. 2020, 46, 21292–21303. [Google Scholar] [CrossRef]
- Wan, L.; Yan, D.; Xu, X.; Li, J.; Lu, T.; Gao, Y.; Yao, Y.; Pan, L. Self-assembled 3D flower-like Fe3O4/C architecture with superior lithium ion storage performance. J. Mater. Chem. A 2018, 6, 24940–24948. [Google Scholar] [CrossRef]
- Chandra, S.; Das, R.; Kalappattil, V.; Eggers, T.; Harnagea, C.; Nechache, R.; Phan, M.H.; Rosei, F.; Srikanth, H. Epitaxial magnetite nanorods with enhanced room temperature magnetic anisotropy. Nanoscale 2017, 9, 7858–7867. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, J.; Wang, Y.; Tian, W.; Liu, J.; Zang, J.; Ning, H.; Yang, C.; Wu, M. Novel in-situ redox synthesis of Fe3O4/rGO composites with superior electrochemical performance for lithium-ion batteries. Electrochim. Acta 2018, 262, 233–240. [Google Scholar] [CrossRef]
- Stefanov, I.; Pérez-Rafael, S.; Hoyo, J.; Cailloux, J.; Santana Pérez, O.O.; Hinojosa-Caballero, D.; Tzanov, T. Multifunctional Enzymatically Generated Hydrogels for Chronic Wound Application. Biomacromolecules 2017, 18, 1544–1555. [Google Scholar] [CrossRef] [PubMed]
- Kalkan, N.A.; Aksoy, S.; Aksoy, E.A.; Hasirci, N. Preparation of chitosan-coated magnetite nanoparticles and application for immobilization of laccase. J. Appl. Polym. Sci. 2012, 123, 707–716. [Google Scholar] [CrossRef]
- Zhang, Y.; Piao, M.; He, L.; Yao, L.; Piao, T.; Liu, Z.; Piao, Y. Immobilization of laccase on magnetically separable biochar for highly efficient removal of bisphenol A in water. RSC Adv. 2020, 10, 4795–4804. [Google Scholar] [CrossRef]
- Qiu, X.; Wang, S.; Miao, S.; Suo, H.; Xu, H.; Hu, Y. Co-immobilization of laccase and ABTS onto amino-functionalized ionic liquid-modified magnetic chitosan nanoparticles for pollutants removal. J. Hazard. Mater. 2020, 401, 123353. [Google Scholar] [CrossRef]
- Cao, P.; Liu, H.; Wu, D.; Wang, X. Immobilization of laccase on phase-change microcapsules as self- thermoregulatory enzyme carrier for biocatalytic enhancement. Chem. Eng. J. 2020, 405, 126695. [Google Scholar] [CrossRef]
- Jankowska, K.; Ciesielczyk, F.; Bachosz, K.; Zdarta, J.; Kaczorek, E.; Jesionowski, T. Laccase immobilized onto zirconia-silica hybrid doped with Cu2+ as an effective biocatalytic system for decolorization of dyes. Materials 2019, 12, 1252. [Google Scholar] [CrossRef]
- Zhang, K.; Yang, W.; Liu, Y.; Zhang, K.; Chen, Y.; Yin, X. Laccase immobilized on chitosan-coated Fe3O4 nanoparticles as reusable biocatalyst for degradation of chlorophenol. J. Mol. Struct. 2020, 1220, 128769. [Google Scholar] [CrossRef]
- Qiu, X.; Wang, Y.; Xue, Y.; Li, W.; Hu, Y. Laccase immobilized on magnetic nanoparticles modified by amino-functionalized ionic liquid via dialdehyde starch for phenolic compounds biodegradation. Chem. Eng. J. 2020, 391, 123564. [Google Scholar] [CrossRef]
- Xue, P.; Liu, X.; Gu, Y.; Zhang, W.; Ma, L.; Li, R. Laccase-mediator system assembling co-immobilized onto functionalized calcium alginate beads and its high-efficiency catalytic degradation for acridine. Colloids Surfaces B Biointerfaces 2020, 196, 111348. [Google Scholar] [CrossRef]
- Aricov, L.; Leonties, A.R.; Gîfu, I.C.; Preda, D.; Raducan, A.; Anghel, D.F. Enhancement of laccase immobilization onto wet chitosan microspheres using an iterative protocol and its potential to remove micropollutants. J. Environ. Manage. 2020, 276, 111326. [Google Scholar] [CrossRef] [PubMed]
- Zofair, S.F.F.; Arsalan, A.; Khan, M.A.; Alhumaydhi, F.A.; Younus, H. Immobilization of laccase on Sepharose-linked antibody support for decolourization of phenol red. Int. J. Biol. Macromol. 2020, 161, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Daronch, N.A.; Kelbert, M.; Pereira, C.S.; de Araújo, P.H.H.; de Oliveira, D. Elucidating the choice for a precise matrix for laccase immobilization: A review. Chem. Eng. J. 2020, 397, 125506. [Google Scholar] [CrossRef]
- Shen, L.; Liu, Y.; Xu, H. Lou Treatment of ampicillin-loaded wastewater by combined adsorption and biodegradation. J. Chem. Technol. Biotechnol. 2010, 85, 814–820. [Google Scholar] [CrossRef]
- Elander, R.P. Industrial production of β-lactam antibiotics. Appl. Microbiol. Biotechnol. 2003, 61, 385–392. [Google Scholar] [CrossRef]
- Zhang, C.; You, S.; Zhang, J.; Qi, W.; Su, R.; He, Z. An effective in-situ method for laccase immobilization: Excellent activity, effective antibiotic removal rate and low potential ecological risk for degradation products. Bioresour. Technol. 2020, 308, 123271. [Google Scholar] [CrossRef]
- Kumar, R.; Park, B.J.; Jeong, H.R.; Lee, J.T.; Cho, J.Y. Biodegradation of β-lactam antibiotic “ampicillin” by white rot fungi from aqueous solutions. J. Pure Appl. Microbiol. 2013, 4, 3163. [Google Scholar]
- Pajchel, G.; Pawłowski, K.; Tyski, S. CE versus LC for simultaneous determination of amoxicillin/clavulanic acid and ampicillin/sulbactam in pharmaceutical formulations for injections. J. Pharm. Biomed. Anal. 2002, 29, 75–81. [Google Scholar] [CrossRef]
- Mem, H.; Ma, M. Development and validation of RP-HPLC method for determination of amoxicillin residues and application to NICOMAC coating machine. J. Anal. Pharm. Res. 2018, 7, 586–594. [Google Scholar] [CrossRef][Green Version]
- Yang, J.; Lin, Y.; Yang, X.; Ng, T.B.; Ye, X.; Lin, J. Degradation of tetracycline by immobilized laccase and the proposed transformation pathway. J. Hazard. Mater. 2017, 322, 525–531. [Google Scholar] [CrossRef]









| Used Material | Laccase Loading (mg/g) | Reference |
|---|---|---|
| HNTs-Fe3O4-CTs-SH | 144 | This study |
| Magnetic biochar (L-MBC) | 27 | [35] |
| MACS-NIL-Cu-Laccase | 47 | [36] |
| Polyacrylamide-alginate cryogel | 68 | [8] |
| LA-Au/PDA@SiO2-MEPCM | 50 | [37] |
| ZrO2–SiO2 | 86 | [38] |
| Fe3O4@Chitosan | 32 | [39] |
| ZrO2–SiO2/Cu2+ | 94 | [38] |
| HNTs-M-chitosan (1%) | 100 | [14] |
| Aminosilanized magnetic HNTs | 84 | [16] |
| Fe3O4-NIL-DAS@lac | 60 | [40] |
| Magnetized chitosan modified α-Cellulose | 73 | [9] |
| PD-GMA-Ca@ABTS beads | 8 | [41] |
| Magnetized chitosan modified HNTs | 92 | [13] |
| Chitosan microspheres | 8 | [42] |
| Sepharose-linked antibody | 33 | [43] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kadam, A.A.; Sharma, B.; Shinde, S.K.; Ghodake, G.S.; Saratale, G.D.; Saratale, R.G.; Kim, D.-Y.; Sung, J.-S. Thiolation of Chitosan Loaded over Super-Magnetic Halloysite Nanotubes for Enhanced Laccase Immobilization. Nanomaterials 2020, 10, 2560. https://doi.org/10.3390/nano10122560
Kadam AA, Sharma B, Shinde SK, Ghodake GS, Saratale GD, Saratale RG, Kim D-Y, Sung J-S. Thiolation of Chitosan Loaded over Super-Magnetic Halloysite Nanotubes for Enhanced Laccase Immobilization. Nanomaterials. 2020; 10(12):2560. https://doi.org/10.3390/nano10122560
Chicago/Turabian StyleKadam, Avinash A., Bharat Sharma, Surendra K. Shinde, Gajanan S. Ghodake, Ganesh D. Saratale, Rijuta G. Saratale, Do-Yeong Kim, and Jung-Suk Sung. 2020. "Thiolation of Chitosan Loaded over Super-Magnetic Halloysite Nanotubes for Enhanced Laccase Immobilization" Nanomaterials 10, no. 12: 2560. https://doi.org/10.3390/nano10122560
APA StyleKadam, A. A., Sharma, B., Shinde, S. K., Ghodake, G. S., Saratale, G. D., Saratale, R. G., Kim, D.-Y., & Sung, J.-S. (2020). Thiolation of Chitosan Loaded over Super-Magnetic Halloysite Nanotubes for Enhanced Laccase Immobilization. Nanomaterials, 10(12), 2560. https://doi.org/10.3390/nano10122560






