High Potency of Organic and Inorganic Nanoparticles to Treat Cystic Echinococcosis: An Evidence-Based Review
Abstract
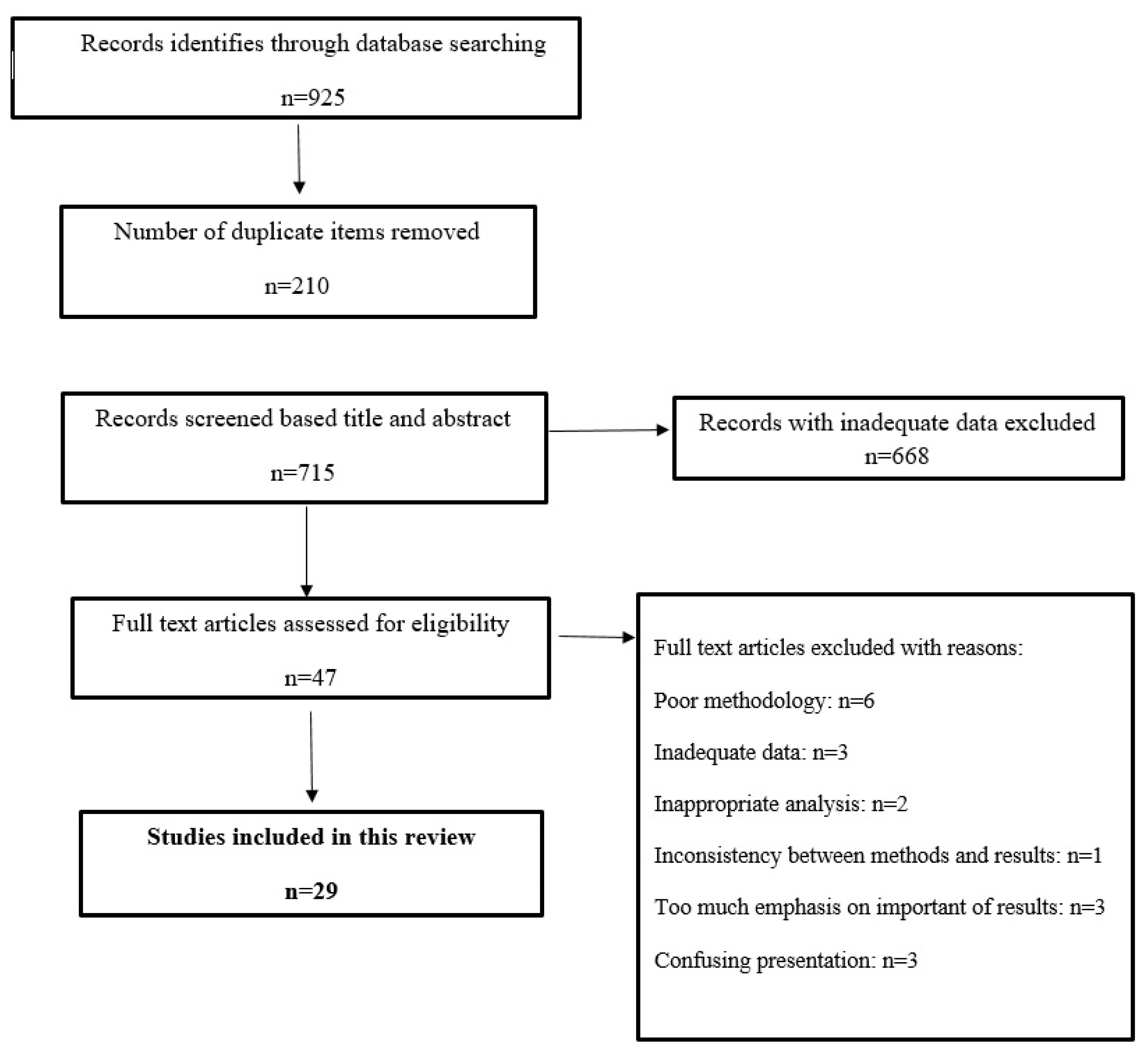
1. Background
2. Materials and Methods
2.1. Search Strategy
2.2. Quality Assessment and Article Selection
2.3. Exclusion Criteria
2.4. Inclusion Criteria
2.5. Data Extraction
3. Results
4. Discussion
4.1. Preparation Methods of Nanoparticles
4.2. Solid Lipid Nanoparticles (SLNs)
4.3. Albendazole (ABZ)-Loaded SLNs
4.4. Albendazole Sulfoxide-Loaded SLNs
4.5. Albendazole- and Praziquantile-Loaded SLNs
4.6. Nanolipid Carriers
Nanolipid Carriers-Loaded Ivermectin
4.7. Lipid Nanocapsules
Albendazole-Lipid Nanocapsules
4.8. Metal Nanoparticles
4.8.1. Selenium (Se) NPs
4.8.2. Silver (Ag) NPs
4.8.3. Gold (Au) NPs
4.9. Non-Metal NPs
ABZ-Loaded Nanoparticles
4.10. Metal Oxide NPs
4.10.1. Zinc Oxide Nanoparticles
4.10.2. Titanium Dioxide (TiO2) Nanoparticles
4.10.3. Cerium Dioxide Nanoparticles
4.10.4. Zirconium Dioxide Nanoparticles
4.11. Nanopolymeric Particles
4.11.1. Chitosan-Curcumin Nanoparticles
4.11.2. Chitosan-Praziquantel and -Albendazole Nanoparticles
4.11.3. Albendazole Sulfoxide-Loaded PLGA-PEG NPs
4.11.4. Flubendazole-Loaded mPEG-PCL NPs
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Availability of Data and Materials
References
- Mcmanus, D.P.; Zhang, W.; Li, J.; Bartley, P.B. Echinococcosis. Lancet 2003, 362, 1295–1304. [Google Scholar] [CrossRef]
- Eckert, J.; Deplazes, P. Biological, Epidemiological, and Clinical Aspects of Echinococcosis, a Zoonosis of Increasing Concern. Clin. Microbiol. Rev. 2004, 17, 107–135. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, E.; Kern, P.; Vuitton, D.A. Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop. 2010, 114, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Junghanss, T.; Da Silva, A.M.; Horton, J.; Chiodini, P.L.; Brunetti, E. Clinical management of cystic echinococcosis: State of the art, problems, and perspectives. Am. J. Trop. Med. Hyg. 2008, 79, 301–311. [Google Scholar] [CrossRef]
- Eckert, J. Guidelines for treatment of cystic and alveolar echinococcosis in humans. Bull. World Health Organ. 1996, 74, 231–242. [Google Scholar]
- Dehkordi, A.B.; Sanei, B.; Yousefi, M.; Sharafi, S.M.; Safarnezhad, F.; Jafari, R.; Darani, H.Y. Albendazole and treatment of hydatid cyst, review of literature. Infect. Disord. Drug Targets 2018, 18, 1. [Google Scholar] [CrossRef]
- Sahin, M.; Eryilmaz, R.; Bulbuloglu, E. The Effect of Scolicidal Agents on Liver and Biliary Tree (Experimental Study). J. Investig. Surg. 2004, 17, 323–326. [Google Scholar] [CrossRef]
- Mishra, S. Nanotechnology in medicine. Indian Heart J. 2016, 68, 437–439. [Google Scholar] [CrossRef]
- Zhu, X.; Radovic-Moreno, A.F.; Wu, J.; Langer, R.; Shi, J. Nanomedicine in the management of microbial infection—Overview and perspectives. Nano Today 2014, 9, 478–498. [Google Scholar] [CrossRef]
- Sengul, A.B.; Asmatulu, E. Toxicity of metal and metal oxide nanoparticles: A review. Environ. Chem. Lett. 2020, 18, 1659–1683. [Google Scholar] [CrossRef]
- Salata, O.V. Applications of nanoparticles in biology and medicine. J. Nanobiotechnol. 2004, 2, 3. [Google Scholar] [CrossRef]
- Rajput, N. Methods of preparation of nanoparticles—A review. Int. J. Adv. Eng. Technol. 2015, 7, 1806. [Google Scholar]
- Shnawa, B.H. Advances in the Use of Nanoparticles as Anti-Cystic Echinococcosis Agents: A Review Article. J. Pharm. Res. Int. 2018, 24, 1–14. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Aminpour, S.; Rafiei, A.; Jelowdar, A.; Kouchak, M. Evaluation of the Protoscolicidal Effects of Albendazole and Albendazole Loaded Solid Lipid Nanoparticles. Iran. J. Parasitol. 2019, 14, 127–135. [Google Scholar] [CrossRef]
- Rafiei, A.; Soltani, S.; Ramezani, Z.; Abbaspour, M.R.; Jelowdar, A.; Kahvaz, M.S. Ultrastructural changes on fertile and infertile hydatid cysts induced by conventional and solid lipid nanoparticles of albendazole and albendazole sulfoxide. Comp. Clin Path. 2019, 28, 1045–1053. [Google Scholar] [CrossRef]
- Soltani, S.; Rafiei, A.; Ramezani, Z.; Abbaspour, M.R.; Jelowdar, A.; Kahvaz, M.S. Evaluation of the hydatid cyst membrane permeability of albendazole and albendazole sulfoxide-loaded solid lipid nanoparticles. Jundishapur J. Nat. Pharm. Prod. 2017, 12, e34723. [Google Scholar]
- Pensel, P.E.; Gamboa, G.V.U.; Fabbri, J.; Ceballos, L.; Bruni, S.S.; Alvarez, L.I.; A Allemandi, D.; Benoît, J.; Palma, S.D.; Elissondo, M.C. Cystic echinococcosis therapy: Albendazole-loaded lipid nanocapsules enhance the oral bioavailability and efficacy in experimentally infected mice. Acta Trop. 2015, 152, 185–194. [Google Scholar] [CrossRef]
- Ahmadpour, E.; Godrati-Azar, Z.; Spotin, A.; Norouzi, R.; Hamishehkar, H.; Nami, S.; Heydarian, P.; Rajabi, S.; Mohammadi, M.; Perez-Cordon, G. Nanostructured lipid carriers of ivermectin as a novel drug delivery system in hydatidosis. Parasites Vectors 2019, 12, 1–9. [Google Scholar] [CrossRef]
- Mahmoudvand, H.; Harandi, M.F.; Shakibaie, M.; Aflatoonian, M.R.; Zia Ali, N.; Makki, M.S.; Jahanbakhsh, S. Scolicidal effects of biogenic selenium nanoparticles against protoscoleces of hydatid cysts. Int. J. Surg. 2014, 12, 399–403. [Google Scholar] [CrossRef]
- Nematollahi, A.; Shahbazi, P.; Rafat, A.; Ghanbarlu, M. Comparative survey on scolicidal effects of selenium and silver nanoparticles on protoscoleces of hydatid cyst. Open Vet. J. 2018, 8, 374. [Google Scholar] [CrossRef] [PubMed]
- Lashkarizadeh, M.R.; Asgaripour, K.; Dezaki, E.S.; Harandi, M.F. Comparison of Scolicidal Effects of Amphotricin B, Silver Nanoparticles, and Foeniculum vulgare Mill on Hydatid Cysts Protoscoleces. Iran. J. Parasitol. 2015, 10, 206–212. [Google Scholar]
- Rahimi, M.T.; Ahmadpour, E.; Esboei, B.R.; Spotin, A.; Koshki, M.H.K.; Alizadeh, A.; Honary, S.; Barabadi, H.; Mohammadi, M.A. Scolicidal activity of biosynthesized silver nanoparticles against Echinococcus granulosus protoscoleces. Int. J. Surg. 2015, 19, 128–133. [Google Scholar] [CrossRef]
- Norouzi, R.; Ataei, A.; Hejazy, M.; Noreddin, A.; Ezzat, M.; Zowalaty, E. Scolicidal Effects of Nanoparticles Against Hydatid Cyst Protoscoleces in vitro. Int. J. Nanomed. 2020, 15, 1095. [Google Scholar] [CrossRef] [PubMed]
- Barabadi, H.; Honary, S.; Ali Mohammadi, M.; Ahmadpour, E.; Rahimi, M.T.; Alizadeh, A.; Naghibi, F.; Saravanan, M. Green chemical synthesis of gold nanoparticles by using Penicillium aculeatum and their scolicidal activity against hydatid cyst protoscoleces of Echinococcus granulosus. Environ. Sci. Pollut. Res. 2017, 24, 5800–5810. [Google Scholar] [CrossRef] [PubMed]
- Napooni, S.; Arbabi, M.; Delavari, M.; Hooshyar, H.; Rasti, S. Lethal effects of gold nanoparticles on protoscoleces of hydatid cyst: In vitro study. Comp. Clin. Path. 2019, 28, 143–150. [Google Scholar] [CrossRef]
- Malekifard, F. Solicidal effect of the gold nanoparticle on protoscoleces of hydratid cyst in vitro. J. URMIA Univ. Med. Sci. 2017, 28, 137. [Google Scholar]
- Norouzi, R.; Hejazy, M.; Ataei, A. Scolicidal effect of zinc oxide nanoparticles against hydatid cyst protoscoleces in vitro. Int. J. Nanomed. 2019, 4, 23–28. [Google Scholar]
- Navvabi, A.; Homaei, A.; Khademvatan, S.; Ansari, M.H.K.; Keshavarz, M. Combination of TiO2 nanoparticles and Echinometra mathaeis gonad extracts: In vitro and in vivo scolicidal activity against hydatid cysts. Biocatal. Agric. Biotechnol. 2019, 22, 101432. [Google Scholar] [CrossRef]
- Aryamand, S.; Khademvatan, S.; Tappeh, K.H.; Heshmatian, B.; Jelodar, A. In Vitro and in Vivo Scolicidal Activities of Holothuria leucospilota Extract and CeO2 Nanoparticles against Hydatid Cyst. Iran. J. Parasitol. 2019, 14, 269–279. [Google Scholar] [CrossRef]
- Ibrahim, A.A.J. Scolicidal Activity of Zirconium Oxide (ZrO2) nanoparticles Against Protoscolices of Hydatid Cysts. Indian J. Forensic Med. Toxicol. 2020, 14, 409. [Google Scholar]
- Napooni, S.; Delavari, M.; Arbabi, M.; Barkheh, H.; Rasti, S.; Hooshyar, H.; Mashkani, S.M.H. Scolicidal Effects of Chitosan–Curcumin Nanoparticles on the Hydatid Cyst Protoscoleces. Acta Parasitol. 2019, 64, 367–375. [Google Scholar] [CrossRef] [PubMed]
- Torabi, N.; Dobakhti, F.; Faghihzadeh, S.; Haniloo, A. In vitro and in vivo effects of chitosan-praziquantel and chitosan-albendazole nanoparticles on Echinococcus granulosus Metacestodes. Parasitol. Res. 2018, 117, 2015–2023. [Google Scholar] [CrossRef]
- Naseri, M.; Akbarzadeh, A.; Spotin, A.; Akbari, N.A.R.; Mahami-Oskouei, M.; Ahmadpour, E. Scolicidal and apoptotic activities of albendazole sulfoxide and albendazole sulfoxide-loaded PLGA-PEG as a novel nanopolymeric particle against Echinococcus granulosus protoscoleces. Parasitol. Res. 2016, 115, 4595–4603. [Google Scholar] [CrossRef] [PubMed]
- Farhadi, M.; Haniloo, A.; Rostamizadeh, K.; Faghihzadeh, S. Efficiency of flubendazole-loaded mPEG-PCL nanoparticles: A promising formulation against the protoscoleces and cysts of Echinococcus granulosus. Acta Trop. 2018, 187, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Darvishi, M.M.; Moazeni, M.; Alizadeh, M.; Abedi, M.; Tamaddon, A.-M. Evaluation of the efficacy of albendazole sulfoxide (ABZ-SO)–loaded chitosan-PLGA nanoparticles in the treatment of cystic echinococcosis in laboratory mice. Parasitol. Res. 2020, 119, 4233–4241. [Google Scholar] [CrossRef] [PubMed]
- Razi, J.M.; Alborzi, A.; Najafzade Varzi, H.; Ghorbanpour, M.; Derakhshan, L. Survey on effects of albendazole, echinacea purpurea, sambucus ebulus and zinc oxide nanoparticles on unilocular hydatid cyst in mice. Sci. Iran. Vet. J. 2015, 11, 68–125. [Google Scholar]
- Nassef, N.E.; Saad, A.-G.E.; Harba, N.M.; Beshay, E.V.N.; Gouda, M.A.; Shendi, S.S.; Mohamed, A.S.E.-D. Evaluation of the therapeutic efficacy of albendazole-loaded silver nanoparticles against Echinococcus granulosus infection in experimental mice. J. Parasit. Dis. 2019, 43, 658–671. [Google Scholar] [CrossRef]
- Gamboa, G.V.U.; Pensel, P.E.; Elissondo, M.C.; Bruni, S.F.S.; Benoît, J.; Palma, S.D.; Allemandi, A. Albendazole-lipid nanocapsules: Optimization, characterization and chemoprophylactic efficacy in mice infected with Echinococcus granulosus. Exp. Parasitol. 2019, 198, 79–86. [Google Scholar] [CrossRef]
- Jelowdar, A.; Rafiei, A.; Abbaspour, M.; Rashidi, I.; Rahdar, M. Efficacy of combined albendazol and praziquntel and their loaded solid lipid nanoparticles components in chemoprophylaxis of experimental hydatidosis. Asian Pac. J. Trop. Biomed. 2017, 7, 549–554. [Google Scholar] [CrossRef]
- Ahmadnia, S.; Moazeni, M.; Mohammadi-Samani, S.; Oryan, A. In vivo evaluation of the efficacy of albendazole sulfoxide and albendazole sulfoxide loaded solid lipid nanoparticles against hydatid cyst. Exp. Parasitol. 2013, 135, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Cong, T.T.; Faivre, V.; Nguyen, T.T.; Heras, H.; Pirot, F.; Walchshofer, N.; Sarciron, M.-E.; Falson, F. Study on the hydatid cyst membrane: Permeation of model molecules and interactions with drug-loaded nanoparticles. Int. J. Pharm. 2008, 353, 223–232. [Google Scholar] [CrossRef]
- Çolak, B.; Aksoy, F.; Yavuz, S.; Demircili, M.E. Investigating the effect of gold nanoparticles on hydatid cyst protoscoleces under low-power green laser irradiation. Turk. J. Surg. 2019, 35, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, K.; Lenggoro, I.W. Preparation of nanoparticles via spray route. Chem. Eng. Sci. 2003, 58, 537–547. [Google Scholar] [CrossRef]
- Jahn, A.; Reiner, J.E.; Vreeland, W.N.; DeVoe, D.L.; Locascio, L.E.; Gaitan, M. Preparation of nanoparticles by continuous-flow microfluidics. J. Nanoparticle. Res. 2008, 10, 925–934. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Li, Y.; Yehui, G.; Hao, L.; Yu, Z.; Jinsong, Y.; Yanyan, C. Enhancement the oral bioavailability of praziquantel by incorporation into solid lipid nanoparticles. Pharmazie 2009, 64, 86–89. [Google Scholar]
- Xie, S.; Pan, B.; Shi, B.; Zhang, Z.; Zhang, X.; Wang, M.; Zhou, W. Solid lipid nanoparticle suspension enhanced the therapeutic efficacy of praziquantel against tapeworm. Int. J. Nanomed. 2011, 6, 2367–2374. [Google Scholar] [CrossRef][Green Version]
- Purohit, D.K.; Nandgude, T.D.; Poddar, S.S. Nano-lipid carriers for topical application: Current scenario. Asian J. Pharm. 2016, 10, S1–S9. [Google Scholar]
- Beloqui, A.; Solinís, M.Á.; Rodríguez-Gascón, A.; Almeida, A.J.; Préat, V. Nanostructured lipid carriers: Promising drug delivery systems for future clinics. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 143–161. [Google Scholar] [CrossRef]
- Cortesi, R.; Valacchi, G.; Muresan, X.M.; Drechsler, M.; Contado, C.; Esposito, E.; Grandini, A.; Guerrini, A.; Forlani, G.; Sacchetti, G. Nanostructured lipid carriers (NLC) for the delivery of natural molecules with antimicrobial activity: Production, characterisation and in vitro studies. J. Microencapsul. 2017, 34, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Huynh, N.; Passirani, C.; Saulnier, P.; Benoit, J.P. Lipid nanocapsules: A new platform for nanomedicine. Int. J. Pharm. 2009, 379, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Conde, J.; Doria, G.; Baptista, P. Noble Metal Nanoparticles Applications in Cancer. J. Drug Deliv. 2012, 2012, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gold, K.; Slay, B.; Knackstedt, M.; Gaharwar, A.K. Antimicrobial Activity of Metal and Metal-Oxide Based Nanoparticles. Adv. Ther. 2018, 1. [Google Scholar] [CrossRef]
- Kumar, G.S.; Kulkarni, A.; Khurana, A.; Kaur, J.; Tikoo, K. Selenium nanoparticles involve HSP-70 and SIRT1 in preventing the progression of type 1 diabetic nephropathy. Chem. Biol. Interact. 2014, 223, 125–133. [Google Scholar] [CrossRef]
- Khurana, A.; Tekula, S.; Saifi, M.A.; Venkatesh, P.; Godugu, C. Therapeutic applications of selenium nanoparticles. Biomed. Pharmacother. 2019, 111, 802–812. [Google Scholar] [CrossRef]
- Wadhwani, S.A.; Shedbalkar, U.U.; Singh, R.; Chopade, B.A. Biogenic selenium nanoparticles: Current status and future prospects. Appl. Microbiol. Biotechnol. 2016, 100, 2555–2566. [Google Scholar] [CrossRef]
- Huang, T.; Holden, J.A.; Heath, D.E.; O’Brien-Simpson, N.M.; O’Connor, A.J. Engineering highly effective antimicrobial selenium nanoparticles through control of particle size. Nanoscale 2019, 11, 14937–14951. [Google Scholar] [CrossRef]
- Wei, L.; Lu, J.; Xu, H.; Patel, A.; Chen, Z.-S.; Chen, G. Silver nanoparticles: Synthesis, properties, and therapeutic applications. Drug Discov. Today 2015, 20, 595–601. [Google Scholar] [CrossRef]
- Gaafar, M.R.; Mady, R.; Diab, R.; Shalaby, T. Chitosan and silver nanoparticles: Promising anti-toxoplasma agents. Exp. Parasitol. 2014, 143, 30–38. [Google Scholar] [CrossRef]
- Allahverdiyev, A.; Abamor, E.Ş.; Bagirova, M.; Ustundag, C.B.; Kaya, C.; Rafailovich, M. Antileishmanial effect of silver nanoparticles and their enhanced antiparasitic activity under ultraviolet light. Int. J. Nanomed. 2011, 6, 2705–2714. [Google Scholar] [CrossRef] [PubMed]
- Galdiero, S.; Falanga, A.; Vitiello, M.; Cantisani, M.; Marra, V.; Galdiero, M. Silver Nanoparticles as Potential Antiviral Agents. Molecules 2011, 16, 8894–8918. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, C.A.; Rai, M.; Ingle, A.P.; Gupta, I.; Galdiero, S.; Galdiero, M.; Gade, A.; Rai, M. Silver Nanoparticles: Therapeutical Uses, Toxicity, and Safety Issues. J. Pharm. Sci. 2014, 103, 1931–1944. [Google Scholar] [CrossRef] [PubMed]
- Thambiraj, S.; Hema, S.; Shankaran, D.R. Functionalized gold nanoparticles for drug delivery applications. Mater. Today Proc. 2018, 5, 16763–16773. [Google Scholar] [CrossRef]
- Benelli, G. Gold nanoparticles—Against parasites and insect vectors. Acta Trop. 2018, 178, 73–80. [Google Scholar] [CrossRef]
- Webster, T.J.T.; Taylor, E. Reducing infections through nanotechnology and nanoparticles. Int. J. Nanomed. 2011, 6, 1463–1473. [Google Scholar] [CrossRef]
- Mirzaei, H.; Darroudi, M. Zinc oxide nanoparticles: Biological synthesis and biomedical applications. Ceram. Int. 2017, 43, 907–914. [Google Scholar] [CrossRef]
- Çeşmeli, S.; Avci, C.B. Application of titanium dioxide (TiO2) nanoparticles in cancer therapies. J. Drug Target. 2019, 27, 762–766. [Google Scholar] [CrossRef]
- Alhadrami, H.A.; Baqasi, A.; Iqbal, J.; Shoudri, R.A.; Ashshi, A.M.; Azhar, E.I.; Al-Hazmi, F.; Al-Ghamdi, A.; Wageh, S. Antibacterial Applications of Anatase TiO2 Nanoparticle. Am. J. Nanomater. 2017, 5, 31–42. [Google Scholar] [CrossRef]
- Peiris, M.M.K.; Guansekera, T.D.C.P.; Jayaweera, P.M.; Fernando, S.S.N. TiO2 nanoparticles from baker’s yeast: A potent antimicrobial. J. Microbiol. Biotechnol. 2018, 28, 1664–1670. [Google Scholar] [CrossRef]
- Dhall, A.; Self, W.T. Cerium Oxide Nanoparticles: A Brief Review of Their Synthesis Methods and Biomedical Applications. Antioxidants 2018, 7, 97. [Google Scholar] [CrossRef] [PubMed]
- Nithya, P.; Sundrarajan, M. Ionic liquid functionalized biogenic synthesis of Ag[sbnd]Au bimetal doped CeO2 nanoparticles from Justicia adhatoda for pharmaceutical applications: Antibacterial and anti-cancer activities. J. Photochem. Photobiol. B Biol. 2020, 202, 111706. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-W.; Moussi, J.; Drury, J.L.; Wataha, J.C. Zirconia in biomedical applications. Expert Rev. Med. Devices 2016, 13, 945–963. [Google Scholar] [CrossRef] [PubMed]
- Larsson, C. Zirconium dioxide based dental restorations. Studies on clinical performance and fracture behaviour. Swed. Dent. J. Suppl. 2011, 213, 9. [Google Scholar]
- Patil, N.A.; Kandasubramanian, B. Biological and mechanical enhancement of zirconium dioxide for medical applications. Ceram. Int. 2020, 46, 4041–4057. [Google Scholar] [CrossRef]
- Fathima, J.B.; Pugazhendhi, A.; Venis, R. Synthesis and characterization of ZrO2 nanoparticles-antimicrobial activity and their prospective role in dental care. Microb. Pathog. 2017, 110, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Gowri, S.; Gandhi, R.R.; Sundrarajan, M. Structural, Optical, Antibacterial and Antifungal Properties of Zirconia Nanoparticles by Biobased Protocol. J. Mater. Sci. Technol. 2014, 30, 782–790. [Google Scholar] [CrossRef]
- Tiyabooncjai, W.; Tiyaboonchai, W. Chitosan nanoparticles: A promising system for drug delivery. Naresuan Univ. J. 2003, 3, 51–66. [Google Scholar]
- Illum, L.; Jabbal-Gill, I.; Hinchcliffe, M.; Fisher, A.; Davis, S. Chitosan as a novel nasal delivery system for vaccines. Adv. Drug Deliv. Rev. 2001, 51, 81–96. [Google Scholar] [CrossRef]
- Divya, K.; Jisha, M. Chitosan nanoparticles preparation and applications. Environ. Chem. Lett. 2018, 16, 101–112. [Google Scholar] [CrossRef]
- Torabi, N.; Dobakhti, F.; Haniloo, A. Albendazole and Praziquantel Chitosan Nanoparticles: Preparation, Characterization, and In Vitro Release Study. Iran. J. Sci. Technol. Trans. A Sci. 2018, 42, 1269–1275. [Google Scholar] [CrossRef]
| Nanoparticles | Drug | Outcome | Ref | ||
|---|---|---|---|---|---|
| Concentration of Drug (µg/mL) | Best Exposure Time | ||||
| Solid lipid nanoparticles (SLNs) | Albendazole Loaded SLN | 250 | Fifth day | [15] | |
| 500 | Fifth day | ||||
| 2 | 72 h | [16] | |||
| Albendazole Sulfoxide Loaded SLN | 2 2.5 | 72 h | [17] | ||
| 2 | 72 h | [16] | |||
| Lipid nanocapsules (LNCs) | Albendazole -LNCs | 0.5 | - | [18] | |
| 1 | - | ||||
| 1.5 | 7 day | ||||
| Nano lipid carriers (NLCs) | NLCs Loaded Ivermectin | 50 | - | [19] | |
| 100 | - | ||||
| 200 | 150 min | ||||
| 400 | 120 min | ||||
| 800 | 60 min | ||||
| Metal NPs | Selenium NPs | 50 | - | [20] | |
| 125 | - | ||||
| 250 | 20 min | ||||
| 500 | 10 min | ||||
| 50 | - | [21] | |||
| 125 | - | ||||
| 250 | - | ||||
| 500 | 60 min | ||||
| Silver NPs | 500 | - | [22] | ||
| 1000 | - | ||||
| 2000 | - | ||||
| 4000 | 60 min | ||||
| 25 | - | [23] | |||
| 50 | - | ||||
| 100 | - | ||||
| 150 | 120 min | ||||
| 50 | - | [21] | |||
| 125 | - | ||||
| 250 | 60 min | ||||
| 500 | - | ||||
| 250 | - | [24] | |||
| 500 | - | ||||
| 1000 | 60 min | ||||
| Gold NPs | 50 | - | [25] | ||
| 100 | - | ||||
| 200 | - | ||||
| 300 | 120 min | ||||
| 250 | - | [26] | |||
| 500 | - | ||||
| 1000 | - | ||||
| 2000 | - | ||||
| 4000 | 60 min | ||||
| 250 | - | [27] | |||
| 500 | - | ||||
| 1000 | 60 min | ||||
| Zinc NPs | 250 | 10 min | [24] | ||
| 500 | - | ||||
| 1000 | - | ||||
| Copper NPs | 250 | - | [24] | ||
| 500 | 60 min | ||||
| 1000 | - | ||||
| Iron NPs | 250 | - | [24] | ||
| 500 | 10 min | ||||
| 1000 | - | ||||
| Metal oxide NPs | Zinc Oxide NPs | 50,000 | 10 min | [28] | |
| 10,000 | - | ||||
| Sea Urchin Gonad Extraction Combined with Tio2 NPs | 1 | - | [29] | ||
| 5 | - | ||||
| 15 | 60 min | ||||
| CeO2 NPs | 1000 | - | [30] | ||
| 5000 | - | ||||
| 10,000 | - | ||||
| 15,000 | - | ||||
| 20,000 | 60 min | ||||
| ZrO2 | 250 | - | [31] | ||
| 500 | - | ||||
| 1000 | 60 min | ||||
| 2000 | 60 min | ||||
| 4000 | 60 min | ||||
| Nonmetals oxide NPs | SiO2 NPs | 250 | - | [24] | |
| 500 | - | ||||
| 1000 | 60 min | ||||
| Nanopolymeric particles | Chitosan NPs | Curcumin | 500 | - | [32] |
| 1000 | - | ||||
| 2000 | - | ||||
| 4000 | 60 min | ||||
| Praziquantel | 1 | - | [33] | ||
| 5 | Tenth days | ||||
| 10 | Tenth days | ||||
| Albendazole | 1 | - | [33] | ||
| 5 | Tenth days | ||||
| 10 | Tenth days | ||||
| Albendazole Sulfoxide Loaded PLGA-PEG NPs | 50 | - | [34] | ||
| 100 | - | ||||
| 150 | All The time | ||||
| 200 | All The time | ||||
| Flubendazole-Loaded mPEG-PCL NPs | 1 | - | [35] | ||
| 5 | - | ||||
| 10 | Fifteenth days | ||||
| Group | Drug | Animal | Outcome | Ref. | |||
|---|---|---|---|---|---|---|---|
| Drug Dose | Duration of Medication | Investigation Timeframe | |||||
| Nanopolymeric particles | Flubendazole-loaded mPEG-PCL NPs | BALB/c mice | 5 mg/kg | 1 month | 8 months | [35] | |
| Chitosan NPs | Praziquantel | Male DBA/2 mice | 25mg/kg | 21 days | 8 months | [33] | |
| Albendazole | Male DBA/2 Mice | 25 mg/kg | 21 days | 8 months | [33] | ||
| Albendazole sulfoxide (ABZ-SO)–loaded chitosan-PLGA nanoparticles | BALB/c mice | 10 mg/kg | 45 days | 10 months | [36] | ||
| Metal oxide NPs | CeO2 NPs | Male BALB/c mice | 50 mg/kg | 1 month | 2 months | [30] | |
| Sea Urchin Gonad Extraction Combined with Tio2 NPs | BALB/c mice | - | 3 month | 3 months | [29] | ||
| Zinc Oxide NPs | [37] | ||||||
| Metal NPs | Albendazole-Loaded Silver NPs | Female albino mice | 100 mg/kg | 2 month | 2 months | [38] | |
| Silver NPs | Female albino mice | 25 mg/kg | 2 month | 2 months | [38] | ||
| Lipid Nanocapsules (LNCs) | Albendazole -LNCs | Female CF-1 mice | 5 mg/kg | 1 month | 6 months | [39] | |
| Female CF-1 mice | 5 mg/kg | 1 month | 8 months | [18] | |||
| Solid lipid nanoparticles (SLNs) | Praziquantel Loaded SLN | Female BALB/c mice | - | 3 month | 5 months | [40] | |
| Albendazole Loaded SLN | Female BALB/c mice | - | 3 month | 5 months | [40] | ||
| Albendazole Sulfoxide Loaded SLN | Male BALB/c mice | 0.5 mg/kg 2 mg/kg | 15 day | 8 months | [41] | ||
| Albendazole Loaded SLN | BALB/c mice | - | - | 3 months | [15] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albalawi, A.E.; Alanazi, A.D.; Baharvand, P.; Sepahvand, M.; Mahmoudvand, H. High Potency of Organic and Inorganic Nanoparticles to Treat Cystic Echinococcosis: An Evidence-Based Review. Nanomaterials 2020, 10, 2538. https://doi.org/10.3390/nano10122538
Albalawi AE, Alanazi AD, Baharvand P, Sepahvand M, Mahmoudvand H. High Potency of Organic and Inorganic Nanoparticles to Treat Cystic Echinococcosis: An Evidence-Based Review. Nanomaterials. 2020; 10(12):2538. https://doi.org/10.3390/nano10122538
Chicago/Turabian StyleAlbalawi, Aishah E., Abdullah D. Alanazi, Parastoo Baharvand, Maryam Sepahvand, and Hossein Mahmoudvand. 2020. "High Potency of Organic and Inorganic Nanoparticles to Treat Cystic Echinococcosis: An Evidence-Based Review" Nanomaterials 10, no. 12: 2538. https://doi.org/10.3390/nano10122538
APA StyleAlbalawi, A. E., Alanazi, A. D., Baharvand, P., Sepahvand, M., & Mahmoudvand, H. (2020). High Potency of Organic and Inorganic Nanoparticles to Treat Cystic Echinococcosis: An Evidence-Based Review. Nanomaterials, 10(12), 2538. https://doi.org/10.3390/nano10122538







