1. Introduction
Nanocomposites (NCs) with silver nanoparticles (AgNPs) as fulfilments and polymers as a matrix are intensively studied due to their useful properties resulting from the unique properties of nanosilver and easy processability of polymers. Among a wide range of applications of such materials, the utilization as antibacterial agents is the one most intensively investigated [
1]. Although the biocidal activity of silver has been known for centuries, the current rapid development of nanotechnology gives new opportunities in designing biomedical materials containing this element in the form of AgNPs with well-defined composition and structure. Noteworthily, AgNPs exhibit not only a broad antibacterial activity but also antifungal, antiviral, anti-inflammatory, anti-angiogenic, and anti-cancer activities [
2]. Whereas, many reports revealed low/relatively low cytotoxicity of AgNPs towards human cells. However, it must be emphasized that the cytotoxicity of nanoparticles depends on many factors. The most important are size, shape, size distribution, surface charge, and kind of the ligands attached on their surface [
3,
4,
5]. Thus, the designing of repeated procedures allowing us to obtain AgNPs with precisely defined morphology and structure is crucial for their biomedical applications. Furthermore, the dispersion of AgNPs in an adequately chosen polymer matrix ensures their long-time stability, enables easy processability of such materials, and allows controllable releasing of the nanoparticles from NCs into the environments [
6].
There are three main types of nanoparticle-polymer composites (AgNCs) differing in the structure of the nanoparticles used for their preparation and the manner of their combination with the polymer matrix: (1) AgNPs with non-polymer coating dispersed in the polymer matrix; (2) AgNPs attached to the polymer net, and (3) AgNPs with the polymer-modified surface (core-shell nanostructures) dispersed in the polymer matrix [
6,
7]. Only in the last case, homogenous composites with a uniform distribution of nanoparticles in a polymer matrix may be achieved [
8]. In the current scientific literature, a layer built of polymer chains covalently grafted onto the nanoparticles’ surface is named a polymer brush. Three parameters are crucial for the controlling of AgNPs dispersion in a polymer matrix: (1) grafting density of polymer brush, (2) degree of polymerization of the grafted macromolecules, and (3) degree of polymerization of macromolecules that building the polymer matrix [
8]. Additionally, when the structure of matrix polymers and grafted polymers is different, this factor should also be taken into consideration.
In the case of biomedical applications of NCs with AgNPs, the uniform dispersion of fulfilment is crucial because it enables gradual and steady releasing of the antibacterial agent in the form of nanoparticles or silver ions. Noteworthily, the releasing also depends on the polymer matrix affinity to the medium where the NCs are located [
9,
10,
11,
12,
13].
Great attention is targeted at the AgNCs fabricated from engineering polymers that ensuring their easy processability by typical polymer processing techniques such as injection molding, extrusion, or thermoforming [
1].
Polystyrene (PS) is one of the most often commercially used engineering polymers. It is a thermoplastic polymer used in the production of packages, insulation boards, and numerous consumer goods [
14,
15] as well as medical products such as surgical instruments, dental tools, dressing materials, prosthesis, and many others [
16]. For all these applications, uses of PS composites that exhibit biocidal activities, for instance, thanks to the dispersed AgNPs, are highly desirable. However, due to the hydrophobic character of this polymer, the preparation of stable and homogeneous composites requires a proper modification of the nanoparticles’ surface. There are a few reports on the modification of AgNPs with hydrophobic molecules such as oleylamine [
17] and polystyrene [
18,
19] via their physical adsorption onto the nanoparticles’ surface. Although hydrophobic modification of AgNPs’ surface undoubtedly facilitates their dispersion in the hydrophobic matrix, this loosely adsorbed layer cannot efficiently protect the nanoparticles against aggregation during the thermal processing of such materials.
Grafting of polymers onto the surface of nanoparticles means anchoring via covalent bonds (also called polymer brush formation), and it enables the preparation of nanohybrids that are miscible in the polymer matrix and also resistant against aggregation [
8].
Recently, our group proposed a novel approach for the preparation of perfectly homogeneous Ag/PS nanocomposites using high-density polystyrene grafted silver nanoparticles. The developed procedure is based on utilizing nitroxide-mediated polymerization (NMRP) and relies on the coupling of growing polystyrene chains with nitroxide-coated silver nanoparticles. These fabricated nanohybrids are readily dispersible in the polystyrene matrix. The prepared composites Ag/PS are thermoforming and exhibit significant antibacterial activity against model Gram(−) (
Pseudomonas aeruginosa) and model Gram(+) (
Staphylococcus aureus) pathogenic bacteria. Nevertheless, the antibacterial effect is strong when silver content is relatively high ≥ 2% (
w/
w) [
20]. Thus, it would be worth to find a means of enabling of decreasing silver content and maintain the same bioactivity. We hypothesized that the introduction into the polystyrene matrix of a hydrophilic polymer admixture would allow for enabling easier silver ion release thanks to more effective water migration inside of the material. Therefore, an excellent antibacterial activity could be achieved for lower silver content. For this purpose, we decided to use poly(vinyl alcohol)—PVA as a hydrophilic admixture for the PS matrix. The PVA is widely used for the fabrication of biomaterials as biocompatible, biodegradable, water-soluble, and at the same time, non-expensive polymers [
21,
22]. Therefore, the bioactive nanocomposites with PVA as a component can find plenty of interesting applications, among others, in medicine, cosmetics, pharmaceuticals, and the packaging industry.
Very recently, Abdallah and co-workers proved that the nanocomposites with AgNPs as fulfilment and PVA as a matrix exhibit high antibacterial activity against a series of Multi-Drug Resistance bacteria strains [
23].
In this work, we present research that reveals the influence of hydrophilic admixture in nanocomposites’ matrix on their antibacterial activity. We developed the procedure allowing us to obtain perfectly homogeneous PS/PVA/Ag nanocomposites containing silver nanoparticles grafted with polystyrene.
Notably, the developed method allows for the preparation of the nanocomposites on a scale of tens of grammes. The developed modification of AgNPs gives them perfect miscibility with the polymer matrix and at the same time high thermal stability. Due to the connection of polystyrene chains via the nitroxide linker with the silver surface, the polymer shell is covalently attached to the nanoparticle. Whereas, the hydrophilic admixture (PVA) allows for gaining efficient antibacterial activity at low silver content.
The prepared nanocomposites with silver content 2%, 1%, and 0.5% were subjected to the comprehensive antibacterial assays towards four strains of Gram(−) type and three strains of Gram(+) using two types of antibacterial assays: the Kirby-Bauer disc susceptibility test and the Dynamic Shake Flask method. The composites fabricated, according to the reported procedure, exhibit high antibacterial activity at silver content as low as 0.5%.
The detailed physicochemical characterizations of the fabricated nanomaterials were carried out using UV-vis spectrophotometry, Size-exclusion chromatography (SEC), Transmission Electron Microscopy (TEM), and Thermogravimetric analysis (TGA).
3. Results and Discussion
The nanocomposites of polystyrene grafted silver nanoparticles (Ag@PS) with a mixture of polystyrene (PS) and polyvinyl alcohol (PVA) were fabricated. The developed procedure allows for preparing perfectly homogeneous nanomaterials exhibiting antibacterial properties at a very low content of nanosilver (0.5% w/w).
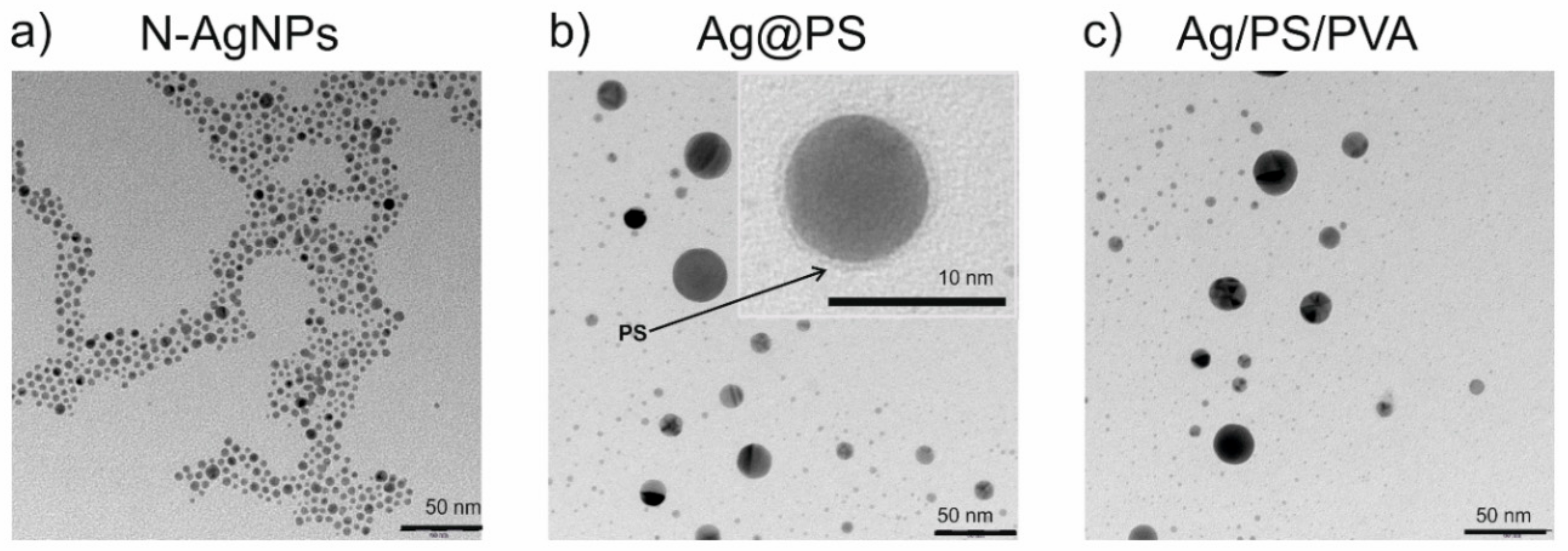
Figure 1 displays TEM images of nitroxide-coated silver nanoparticles (
N-AgNPs), the nanohybrids Ag@PS prepared from them, and the fabricated nanocomposites (Ag/PS/PVA).
As can be seen in
Figure 1a, the nitroxide-coated silver nanoparticles (
N-AgNPs) are narrowly dispersed in terms of their size. Further, they have the ability to self-assemble, which is most probably a consequence of interactions between nitroxide radicals and silver. Such interactions cause accumulation of the nanoparticles in partially ordered structures but importantly do not lead to the aggregation. Recently our group reported the existence of the interactions of nitroxide radicals with silver and gold nanoparticles/surfaces [
24,
26,
27,
28,
29].
The presence of nitroxide radicals covalently connected with the silver surface in
N-AgNPs allows us to connect polystyrene-growing macroradicals during the polymerization and obtain in this way polystyrene grafted silver nanoparticles (Ag@PS). During the polymerization, a fraction of nanoparticles increases in size, as can be seen in
Figure 1b.
The inset in
Figure 1b displays the TEM image of the single silver nanoparticle (magnification with the scale bar 10 nm) where the surrounding polymer shell onto the nanoparticle is visible.
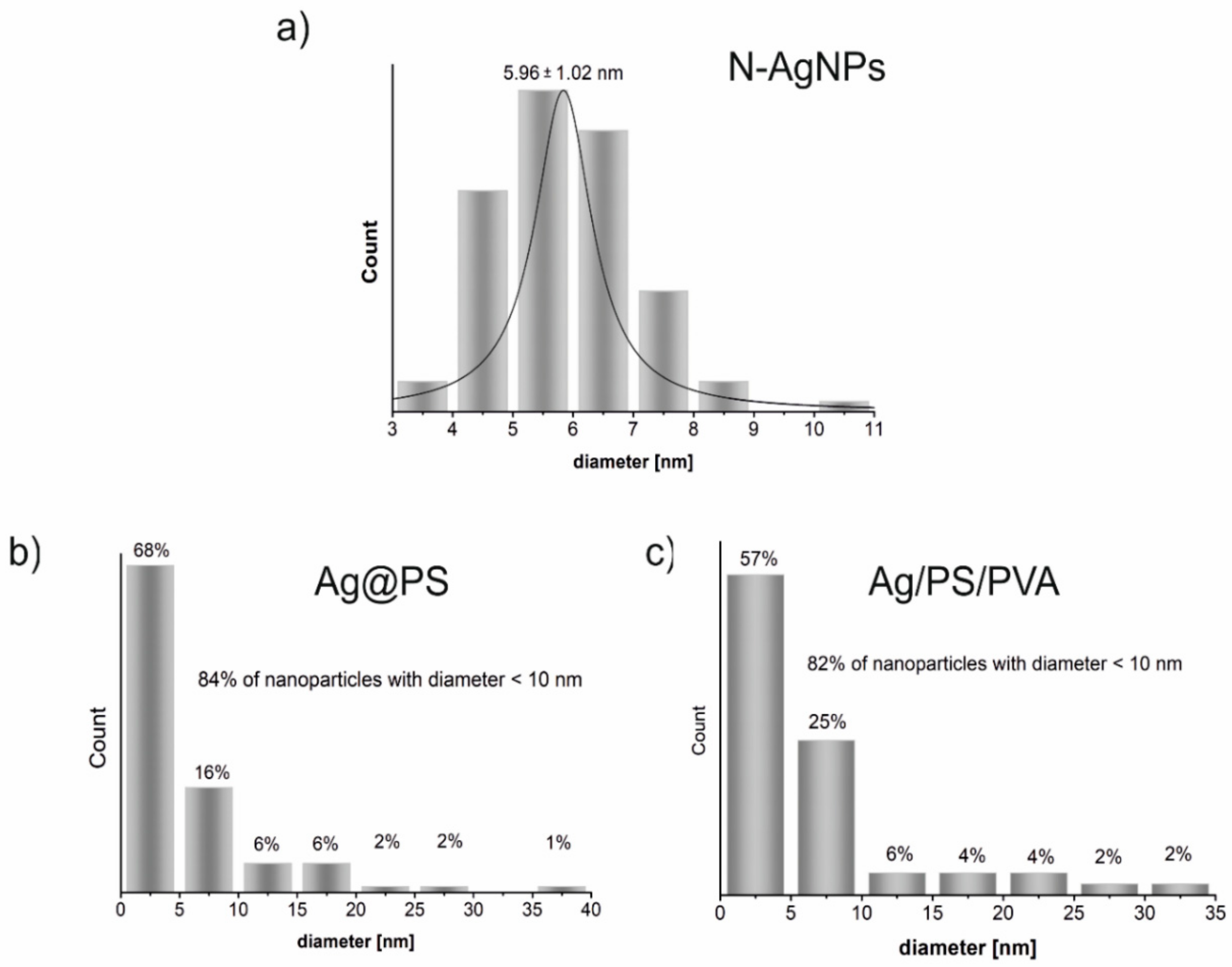
Figure 2 displays the histograms drawn on the base of TEM images shown in
Figure 1. As can be seen in
Figure 2 size distribution of
N-AgNPs is very narrow, and during the polymerization of styrene with
N-AgNPs part of nanoparticles increases their size even to above 35 nm, but 84% of the synthesized nanohybrids (Ag@PS) have a diameter below 10 nm. The size distribution is very similar in the nanocomposites fabricated using Ag@PS as fulfilment and the mixture of PS and PVA as a matrix.
TEM studies revealed that the nanocomposites with homogeneously dispersed, non-aggregated spherical silver nanoparticles with the diameter mostly below 10 nm (see
Figure 2) were prepared.
Due to the strong localized surface plasmon resonance (LSPR) of silver nanoparticles, the UV-vis spectrophotometry is a very convenient tool to analyze their dispersity in a polymer matrix. The position of LSPR band/bands in the absorption spectrum and its full width at half maximum (FWHM) depend not only on the shape, size, and dispersity of AgNPs, but also on the refractive index of the environment (solvent or solid matrix) [
17,
30,
31].
Figure 3 shows the absorption spectra of the prepared silver nanoparticles; polystyrene-grafted nanohybrids and nanocomposites in DMF solutions. The LSPR band in
N-AgNPs spectrum with a maximum at 416 nm is narrow and symmetric and confirms the narrow dispersity of the nanoparticles. The position of the maximum absorption correlates well with the average size of metal cores ca. 6 nm [
28]. For Ag@PS, the LSPR band is red-shifted to 427 nm compared with the band in
N-AgNPs spectrum. This effect can be explained by changing the refractive index of the protecting layer attached to the surface of the nanoparticles. The maximum of the absorption band for the nanocomposite Ag/PS/PVA is significantly more red-shifted to 442 nm, and also the width of LSPR has increased. It indicates that during their thermal processing, widening of the size dispersity of silver nanoparticles occurs. It is consistent with the results from TEM analyses (see
Figure 1 and
Figure 2). Notably, the changes in size dispersity are not significant (ca. 80% of nanoparticles retain the same size). In the spectrum of Ag/PS/PVA, two bands characteristic for polystyrene in the UV range at 220 and 260 nm are visible. The absorption bands of PVA in UV-vis spectra are not visible, due to its low concentration in the nanocomposites (5%
w/
w). We recorded the spectrum of PVA in the same solvent at the same weight concentration, and any bands were not visible.
The developed procedure for the preparation of Ag@PS is based on the employment of the nitroxide mediated polymerization (NMRP) [
32], allowing us to prepare narrowly dispersed polymers with a well-defined molecular weight. Recently, our group developed the method for the grafting of polymer chains on the surface of nanoparticles using NMRP [
20,
28,
29,
33]. In this method, nitroxide-coated nanoparticles are injected to the polymerization system controlled by a nitroxide radical. During the polymerization, the growing macroradicals are captured by the radicals attached on the nanoparticles’ surface. In this way, nanohybrid structures with covalently attached polymer chains are obtained (in the case of silver nanoparticles denoted as Ag@PS). Besides the nanohybrids, in the polymerization system, free ones not attached to the nanoparticles polymer chains are prepared with well-defined molecular weights.
Now we use Ag@PS nanohybrids for the fabrication of the Ag/PS/PVA nanocomposites. The polystyrene applied for this purpose (PSF) was separated from the same polymerization system as Ag@PS. The size exclusion chromatography (SEC) was used to determine average molecular weights and dispersity of PSF.
Table 1 displays the results of the SEC analysis of PSF. The elugram and molecular weights distribution are presented in the
Supplementary Information (Figures S2 and S3, respectively).
Due to the application of the nitroxide radical (TEMPOL) as a mediator, polystyrene formed in the polymerization system (PSF) is narrowly dispersed (polydispersity index PDI = 1.13). In our earlier paper, we proved that the molecular weight of polymers connected with nanoparticles and free in the system is similar. This is so because, during the polymerization, an exchange between attached and free polymer chains is possible [
20]. As a consequence, Ag@PS is perfectly miscible in the polymer matrix (PSF) used for the nanocomposites’ (Ag/PS/PVA) preparation. PVA with an averaged molecular weight M
w = 27 kDa was used for the nanocomposites’ preparation. Notably, the composites with the concentration of PVA above 5% (
w/
w) were becoming non-homogeneous during their thermoforming. Therefore, for all studies, we decided to use only the nanocomposites with 5% PVA.
Thermogravimetric analyses (TGA) were conducted to evaluate the stability of the fabricated nanomaterials.
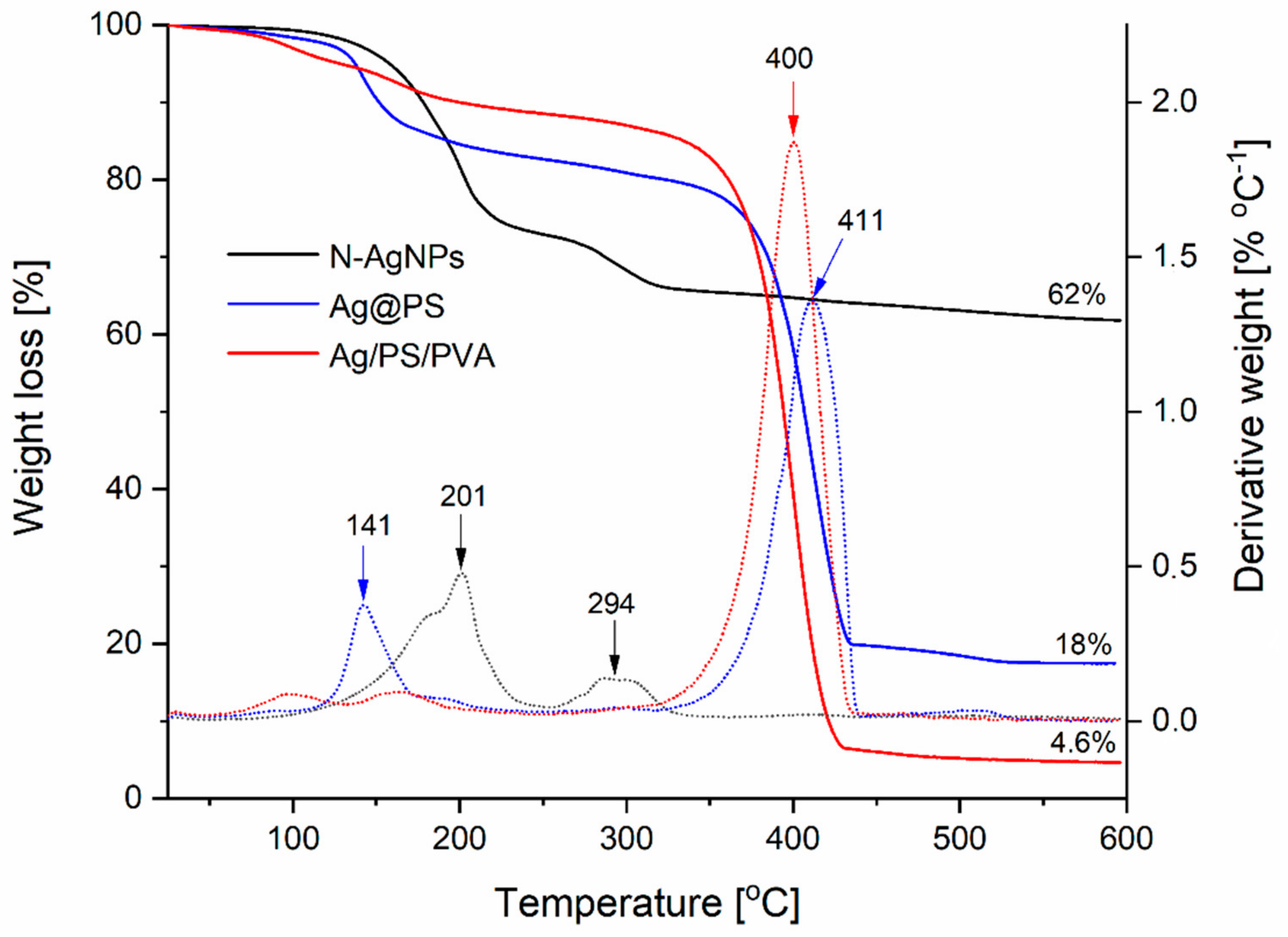
Figure 4 shows TGA curves in the range 20–600 °C and the corresponding first derivatives (DTA).
As can be seen, the decomposition of nitroxide-coated nanoparticles, under the conditions of analysis, is completed below 300 °C and occurs in two steps with the maximum weight loss at 201 °C and 294 °C (see the first derivative curve of corresponding TGA curve). When the polystyrene chains are attached to the surface of the nanoparticles via nitroxide radicals, a significant increase of thermal stability is observed (see TGA curve for Ag@PS), and the decomposition is completed above 450 °C. However, the first step of the decomposition runs at a lower temperature and maximum is located at 141 °C; this step corresponds to only 15% of weight loss. Most likely, part of the nitroxide radicals covering the nanoparticles’ surface remained not connected with polystyrene chains, and their decomposition runs at a lower temperature.
In the case of nanocomposites of Ag@PS with a mixture of PS and PVA (95%: 5%, w/w), the main decomposition step occurs above 300 °C and corresponds to 80% of total weight loss. These results confirm that the fabricated nanocomposites can be processed by thermoforming at the temperatures below 200 °C without decomposition.
The final percentage of the start mass obtained from TGA corresponds to a content of silver in the fabricated materials, and it is 62% for nitroxide-coated nanoparticles and 18% in the nanohybrids (Ag@PS) prepared from them.
As was written earlier, the developed procedure allows for obtaining the nanocomposites with different silver contents depending on the ratio Ag@PS and polymer matrix (PS/PVA). In
Figure 4, the exemplified TGA curve is present for nanocomposite Ag/PS/PVA with silver content 4.6%.
Antibacterial activity of the fabricated nanocomposites was evaluated using two assays, which are commonly used for solid materials, namely the Kirby-Bauer (K-B) disc susceptibility test and the Dynamic Shake Flask (DSF) method. Because of the potential applications of the prepared materials as packaging materials, we decided to exam series of pathogenic bacteria that may be present in food and water. There are Salmonella typhimurium, Escherichia coli, Yersinia enterocolitica, Campylobacter jejuni, Bacillus cereus, and Listeria monocytogenes. Furthermore, antibacterial activity against usually antibiotic-resistant bacterial strains such as Pseudomonas aeruginosa and Staphylococcus aureus was tested.
As it turned out, all fabricated nanocomposites exhibit high antibacterial activity against all studied pathogenic strains, both Gram(−), and Gram(+).
Figure 5 shows photographic images of the results of K-B assays for selected bacterial strains. As can be seen the inhibition zones (IZs), it means the area without bacterial growth is distinctly visible for all materials, also for those with the lowest studied silver content, namely 0.5%.
The IZs determined from the performed K-B assays for all studied nanocomposites against all studied bacterial strains are presented in
Table 2. As can be seen, IZs are in the range 7–34 mm, it means that the antibacterial activity of the fabricated nanocomposites is good or very good. Our results proved that the introducing of PVA as a hydrophilic component significantly increased antibacterial activity of AgNPs dispersed in a hydrophobic polymer matrix. IZ determined against
Pseudomonas aeruginosa, and
Staphylococcus aureus determined for the composite PS/Ag@PS without PVA is 6 mm and 4 mm at 1% of nanosilver. Whereas in the case of reported here Ag/PS/PVA, these IZs are equal to 11 mm, so are almost two times higher. The nanocomposites not containing PVA, when nanosilver concentration is below 1%, do not exhibit antibacterial activity. Thus, the introduction of a small amount of a hydrophilic polymer to the hydrophobic matrix dramatically increased the bioactivity of the composite. Most likely, it facilitates water migration inside of the polymer matrix, and this leads to the easier releasing of silver nanoparticles and silver ions.
The determined IZ values for Gram(−) bacterial strains are higher for all the studied nanocomposites in comparison with these determined for Gram(+). It can be explained by differences in the structure of their cell membranes that cause higher susceptibility of Gram(−) strains [
34].
The highest susceptibility, among those studied, exhibits
Campylobacter jejuni, the IZ value for the nanocomposite with silver content 0.5% is as high as 17 mm. It is worth mentioning that
Campylobacter is a foodborne pathogen which is responsible for many bacterial diarrheal diseases. At the same time, antibiotic resistance of these strains is observed recently more and more frequently [
35]. Thus, all the more, the fabricated nanocomposites have a strong application potential, among others, as food packaging materials.
The Dynamic Shake Flask (DSF) method was also used to evaluate the antibacterial activity of the prepared nanomaterials. This method is commonly used in the case of polymer composites and textiles. We chose three bacterial strains:
Escherichia coli as Gram(−) representative,
Staphylococcus aureus as Gram(+) representative, and
Yersinia enterocolitica—a pathogen often present in food products and water causing acute infections and disease [
36]. It is noteworthy that this bacterial strain can withstand low temperatures, and its growth is observed even at around 4 °C [
37]. The presence of this bacteria strain may be responsible for severe problems with food storage.
The DSF assays were performed under no-growth conditions: without nutrients, only in an aqueous solution containing phosphate buffer (0.3 M, pH = 7.3). The samples were performed in the form of pastilles made of composite (Ag/PS/PVA) with the mass 76 ± 8 mg and such content of Ag@PS to achieve 1% of silver. The buffer solution was inoculated with 10
5 CFU ml
−1. The pastilles were placed in the solution, and afterwards, the mixture was shaken at 200 rpm for 24 h at 37 °C. As a control sample, the pastill made of PS and PVA with a mass ratio 95:5
w/
w was used. The bacterial concentration in the microbial suspension was determined by measuring optical density (O.D.) at wavelength 600 nm. The percentage survival of bacterial cells in the system was calculated according to the following equation:
where
Cs—surviving cell concentration in the mixture with the composite sample after an appropriate time of incubation at 37 °C,
Cc—surviving cell concentration in the mixture with the control sample after the same period of incubation time as
Cs.
The measurements of bacterial concentration were performed every hour for 6 h, and the next measurement was performed after 24 h.
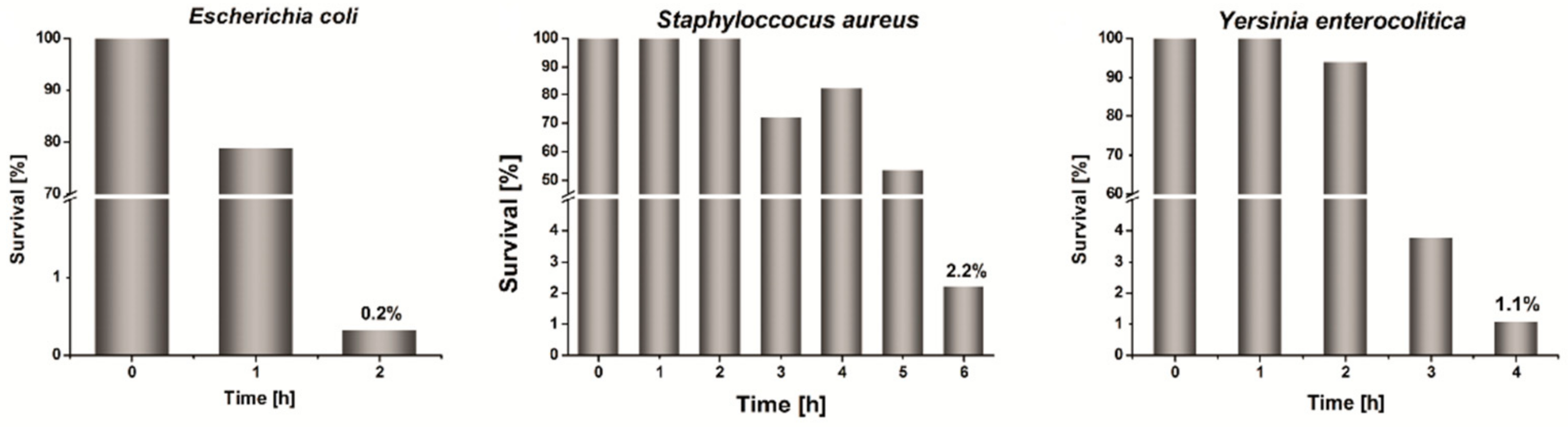
Figure 6 displays the determined percentage survival
S% of the studied bacterial strains vs. time of incubation in the presence of the pastilles made of Ag/PS/PVA composite containing 1% of nanosilver and 5% of PVA. In the case of all studied systems after 24 h of incubation, there were not any survived cells. In a period of only 6 h number of bacterial cells decrease rapidly under the influence of the presence of the pastilles made of the nanocomposite. The survival of
Escherichia coli after only 2 h of incubation with Ag/PS/PVA is 0.2%. After a longer time of incubation, we did not observe any survived cells.
The survival of Staphylococcus aureus in the presence of the studied nanocomposite is higher and after 6 h is 2.2%, but after 24 h of incubation, we did not observe survived cells. The observed differences in susceptibility between Escherichia coli and Staphylococcus aureus are a consequence of the different structure and the composition of their cell membranes. Staphylococcus aureus is a representative of Gram(+) bacteria; therefore, its cell membrane is significantly thicker, and permeation through the membrane is more complicated than in the case of Gram(−) bacteria.
The survival of Yersinia enterocolitica is higher than Escherichia coli and lower than Staphylococcus aureus. After 4 h of incubation, in the presence of Ag/PS/PVA, the survival is 1.1%, whereas after 5 h the surviving cells were not observed.
Notably, the biocidal activity of the developed nanocomposites is significantly higher than for the analogical composites without the addition of PVA. The survival of
Staphylococcus aureus after 24 h of incubation in the presence of pastille, made of Ag@PS and dispersed in PS with 4% silver, is almost 90% [
20].
Our results revealed that the doping of polystyrene nanocomposites with a hydrophilic polymer increases their bioactivity.
The performed antibacterial assays proved that the fabricated nanocomposites could be successfully used as biocidal materials against pathogenic bacteria, both Gram(−) and Gram(+).
4. Conclusions
In summary, we propose a new approach for the preparation of highly bioactive and thermoprocessable nanocomposites. In the prepared materials, polystyrene-grafted silver nanoparticles (Ag@PS) play the role of bioactive fulfilment, and a mixture of polystyrene (PS) and poly(vinyl alcohol) PVA is the polymer matrix.
We also report the method allowing us to fabricate narrowly sized dispersive (5.96 ± 1.02 nm) silver nanoparticles (N-AgNPs) on a scale of hundreds of milligrams/several grams. Nitroxide radicals stabilize the synthesized nanoparticles, and thanks to this, they can be readily grafted with polymer chains via radical coupling during radical polymerization. Polystyrene-grafted silver nanoparticles (Ag@PS), prepared in this way, are perfectly dispersible in a mixture of PS with PVA (95:5 w/w). Such nanocomposites are thermally stable and, thus, can be thermally processed into pellets, pastilles, foils, plates, and other elements.
Kirby-Bauer (K-B) and Dynamic Shake Flask (DSF) assays showed high antibacterial activity against a series of Gram(−) and Gram(+) bacteria strains of the fabricated nanocomposites. Regarding the silver content, 2% of the biocidal activity of the nanocomposites is very high, and when the content is only 0.5%, the activity can be evaluated as being effective.
The doping of Ag/PS composites with PVA gives antibacterial activity at a low concentration of nanosilver (0.5%). Most likely, the presence of hydrophilic admixture in the nanocomposites enables water migration inside the material, which makes the releasing of silver nanoparticles and ions from the polymer matrix easier.
Due to the thermal processability and antibacterial activity at low silver content, the fabricated nanocomposites are highly promising materials for the fabrication of medical equipment such as, among others, surgical instruments, prosthesis, coatings, and dental tools.