Performant Composite Materials Based on Oxide Semiconductors and Metallic Nanoparticles Generated from Cloves and Mandarin Peel Extracts
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples Preparation
- (i)
- Preparation of extracts
- (a)
- the cloves extractA quantity of 50 g of the cloves powder was introduced into 250 mL of hot distilled water. The suspension was boiled for 5 min, then cooled to room temperature and filtered using Whatman filter paper no.1. The aqueous extract was used in subsequent experiments to obtain AgNPs and ZnO-based structures.
- (b)
- mandarin peels extractA quantity of 50 g of mandarin peels (previously washed with distilled water) was introduced into 250 mL of hot distilled water. The suspension was boiled for 45 min, then cooled to room temperature and filtered using filter paper (Whatman filter paper no.1). The aqueous mandarin extract was used in subsequent experiments to obtain AgNPs and ZnO-based structures.
- (ii)
- Preparation of Ag nanoparticles by bioreduction of Ag+ in the presence of cloves or mandarin extractsIn a volume of cloves extract, an equal volume of 1 mM aqueous solution of AgNO3 was added under continuous stirring. Over time, the color of the extract changed from pink to reddish brown, indicating the formation of AgNPs.In an adequate volume of mandarin extract, AgNO3 crystals were added up to the final concentration of 4 mM under continuous stirring. The change of color from yellow–orange to green–gray was observed, indicating the formation of AgNPs.The obtaining of AgNPs was monitored by recording the |Ultraviolet-Visible (UV-Vis) absorption spectra, which shows the appearance of an absorption band in the wavelength region of 400–450 nm, characteristic for the SPR (surface plasmon resonance) band of the AgNPs with the quasi-spherical shape [28].
- (iii)
- Preparation of ZnO particles by the chemical reaction between Zn (NO3)2 and NaOH, in the presence of Ag nanoparticles in cloves, respectively mandarins extractThe suspension of AgNPs in the cloves or mandarins extract, represents solution A. The precursors: Zn (NO3)2·6H2O and NaOH were dissolved in distilled water (under continuous stirring), obtaining solutions B and, respectively, C. Subsequently solutions B and C were added dropwise into solution A (stirring continued for 30 min). The precipitate was centrifuged and washed several times until a neutral pH was obtained and finally dried in vacuum at 100 °C for 2 h. Additionally, maintaining the same reaction conditions, the ZnO nanoparticles were also obtained in the absence of Ag nanoparticles, in the two extracts: cloves and mandarins. The abbreviations: CUI and MAND correspond to cloves and mandarin derived samples, respectively. The investigated samples are noted as follows: AgZnO-CUI; AgZnO-MAND, ZnO-CUI, ZnO-MAND, Ag-CUI and Ag-MAND.
- (i)
- AgNO3 + phyto-molecules (proteins, sugars and phenolic compounds)→Ag0
- (ii)
- Ag0→AgNPs through the process of capturing Ag0 by the extract biomolecules
- (iii)
- Zn(NO3)2 + 2NaOH→Zn(OH)2↓ + 2NaNO3
- (iv)
- phyto-AgNPs +
- (i)
- Zn(NO3)2 + 2NaOH + plant extract →Zn(OH)2↓ + 2NaNO3 + plant extract
- (ii)
- plant extract +
2.2. Physico-Chemical Characterization of Phyto-Developed Materials
2.3. Biological Investigation Methods
- i.
- Cell Viability
- ii.
- Antibacterial Assay
- iii.
- Hemolytic Assay
3. Results and Discussions
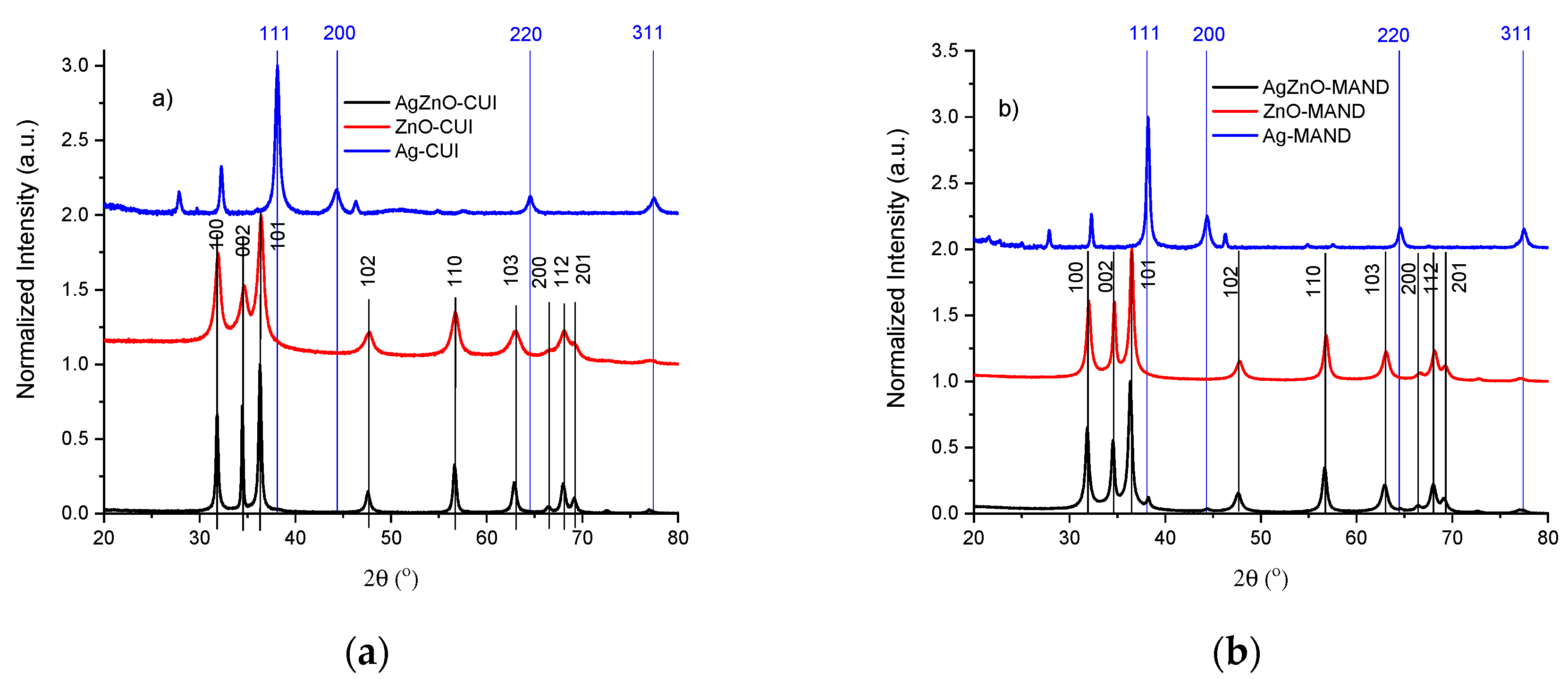
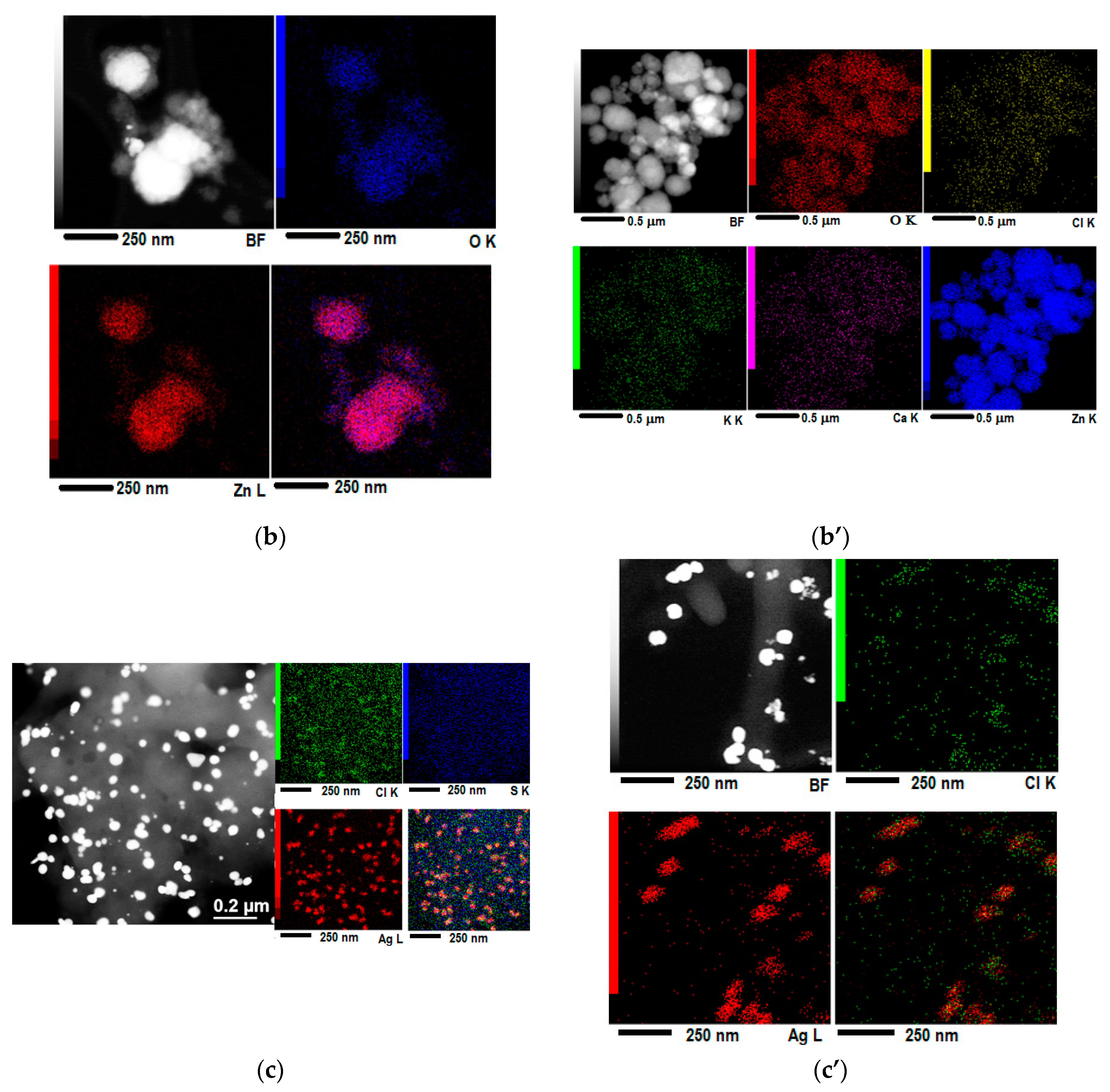
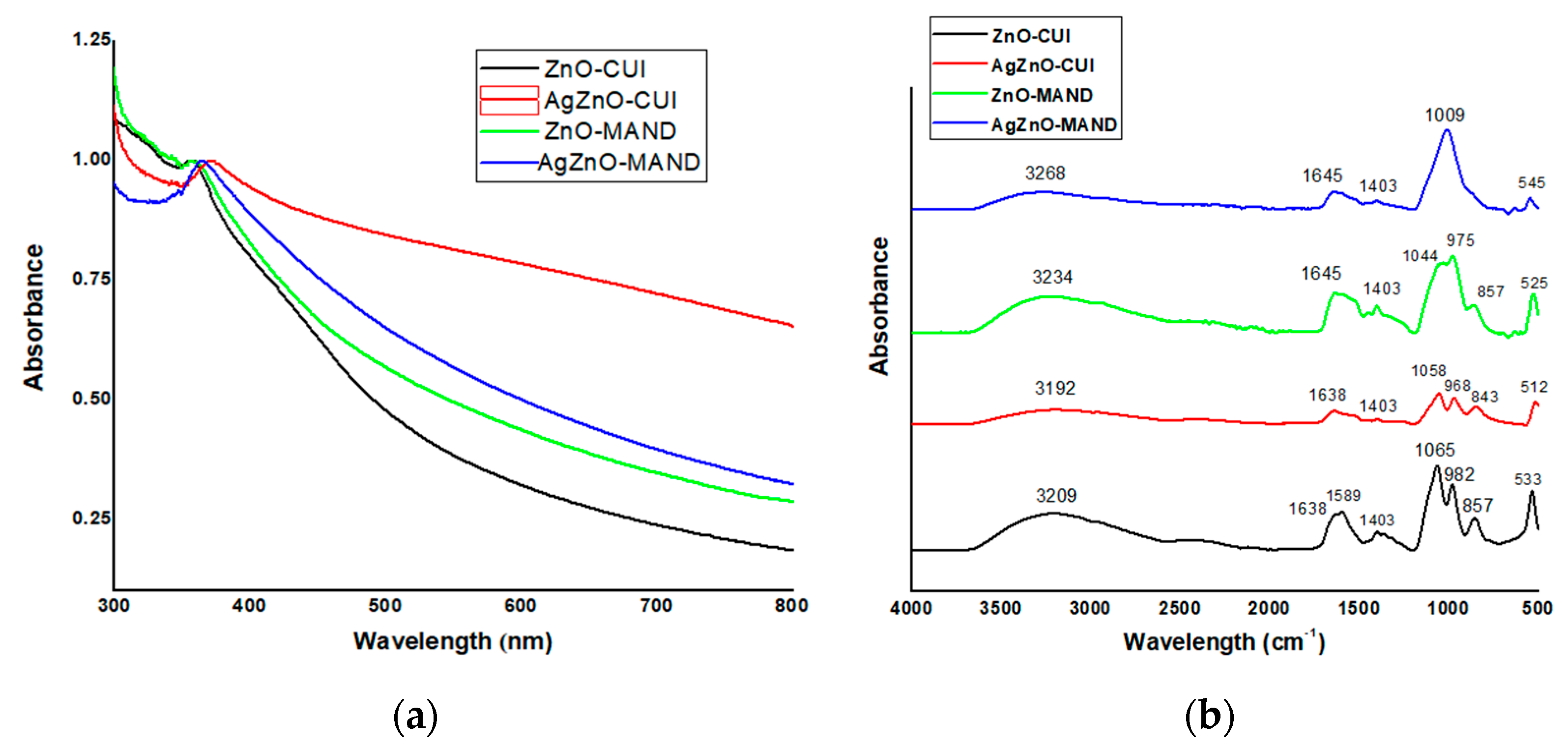
3.1. Structural, Morphological and Spectral Characterization of “Green” Synthesized Materials
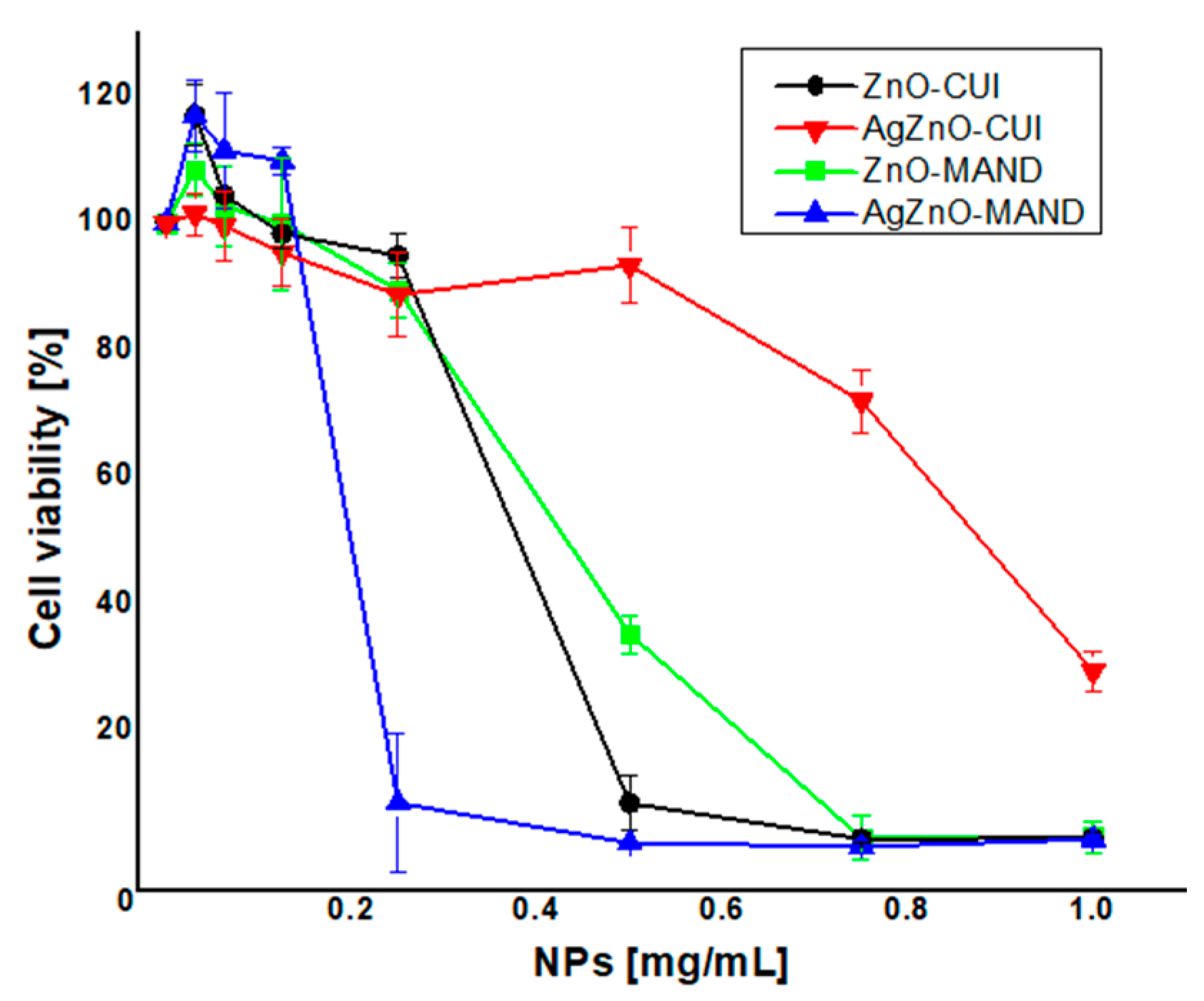
3.2. Biological Characterization of Phytosynthesized Composites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kumar, H.; Bhardwaj, K.; Kuča, K.; Kalia, A.; Nepovimova, E.; Verma, R.; Kumar, D. Flower-Based Green Synthesis of Metallic Nanoparticles: Applications beyond Fragrance. Nanomaterials 2020, 10, 766. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, K.S.; ur Rahman, A.; Tajuddin; Husen, A. Properties of Zinc Oxide Nanoparticles and Their Activity Against Microbes. Nanoscale Res. Lett. 2018, 13, 141. [Google Scholar] [CrossRef] [PubMed]
- Some, S.; Kumar Sen, I.; Mandal, A.; Aslan, T.; Ustun, Y.; Yilmaz, E.Ş.; Kati, A.; Demirbas, A.; Mandal, A.K.; Ocsoy, I. Biosynthesis of silver nanoparticles and their versatile antimicrobial properties. Mater. Res. Express 2018, 6, 012001. [Google Scholar] [CrossRef]
- Gajbhiye, S.; Sakharwade, S. Silver nanoparticles in cosmetics. J. Cosmet. Dermatol. Sci. Appl. 2016, 6, 48–53. [Google Scholar] [CrossRef]
- Frederickson, C.J.; Koh, J.Y.; Bush, A.I. The neurobiology of zinc in health and disease. Nat. Rev. Neurosci. 2005, 6, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, H.; Venkat Kumar, S.; Rajeshkumar, S. A review on green synthesis of zinc oxide nanoparticles—An eco-friendly approach. Resour. Eff. Technol. 2017, 3, 406–413. [Google Scholar] [CrossRef]
- Elshama, S.; Abdallah, M.; Abdel-Karim, R. Zinc Oxide Nanoparticles: Therapeutic Benefits and Toxicological Hazards. Open Nanomed. J. 2018, 5, 16–22. [Google Scholar] [CrossRef]
- Jiang, J.; Pi, J.; Cai, J. The advancing of zinc oxide nanoparticles for biomedical applications. Bioinorg. Chem. Appl. 2018, 18, 1062562. [Google Scholar] [CrossRef]
- Martínez-Carmona, M.; Gun’ko, Y.; Vallet-Regí, M. ZnO Nanostructures for Drug Delivery and Theranostic Applications. Nanomaterials 2018, 8, 268. [Google Scholar] [CrossRef]
- Arshad, R.; Sohail, M.F.; Sarwar, H.S.; Saeed, H.; Ali, I.; Akhtar, S.; Hussain, S.Z.; Afzal, I.; Jahan, S.; Anees ur, R.; et al. ZnO-NPs embedded biodegradable thiolated bandage for postoperative surgical site infection: In vitro and in vivo evaluation. PLoS ONE 2019, 14, e0217079. [Google Scholar] [CrossRef]
- Rajendran, S. Advanced Textiles for Wound Care, 2nd ed.; The Textile Institute Book Series; Woodhead Publishing: Cambridge, UK, 2018; p. 26. [Google Scholar]
- Naseri, N.; Algan, C.; Jacobs, V.; John, M.; Oksman, K.; Mathew, A.P. Electrospun chitosan-based nanocomposite mats reinforced with chitin nanocrystals for wound dressing. Carbohydr. Polym. 2014, 109, 7–15. [Google Scholar] [CrossRef]
- Ahmad, K.S.; Jaffri, S.B. Phytosynthetic Ag doped ZnO nanoparticles: Semiconducting green remediators. Open Chem. 2018, 16, 556–570. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Badea, N.; Bacalum, M.; Ungureanu, C.; Suica-Bunghez, I.R.; Iordache, S.M.; Pirvu, C.; Zgura, I.; Maraloiu, V.A. 3D hybrid structures based on biomimetic membranes and Caryophyllus aromaticus-“Green” synthesized nano-silver with improved bioperformances. Mater. Sci. Eng. C 2019, 101, 120–137. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Ungureanu, C.; Badea, N.; Bacalum, M.; Lazea-Stoyanova, A.; Zgura, I.; Negrila, C.; Enculescu, M.; Burnei, C. Novel Ecogenic Plasmonic Biohybrids as Multifunctional Bioactive Coatings. Coatings 2020, 10, 659. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Ungureanu, C.; Badea, N.; Constantin, M.; Purcar, V.; Ispas, A. Bioperformances of honey-phytonanosilver in silica materials. J. Optoelectron. Adv. Mater. 2020, 22, 310–315. [Google Scholar]
- Barbinta-Patrascu, M.E.; Badea, N.; Constantin, M.; Ungureanu, C.; Nichita, C.; Iordache, S.M.; Vlad, A.; Antohe, S. Bio-activity of organic/inorganic phyto-generated composites in bio-inspired systems. Rom. J. Phys. 2018, 63, 702. [Google Scholar]
- Patra, S.; Mukherjee, S.; Barui, A.K.; Ganguly, A.; Sreedhar, B.; Patra, C.R. Green synthesis, characterization of gold and silver nanoparticles and their potential application for cancer therapeutics. Mater. Sci. Eng. C 2015, 53, 298–309. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Ungureanu, C.; Iordache, S.M.; Bunghez, I.R.; Badea, N.; Rau, I. Green silver nanobioarchitectures with amplified antioxidant and antimicrobial properties. J. Mater. Chem. B 2014, 2, 3221–3231. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Badea, N.; Ungureanu, C.; Constantin, M.; Pirvu, C.; Rau, I. Silver-based biohybrids “green” synthesized from Chelidonium majus L. Opt. Mater. 2016, 56, 94–99. [Google Scholar] [CrossRef]
- Abbasi, B.H.; Shah, M.; Hashmi, S.S.; Nazir, M.; Naz, S.; Ahmad, W.; Khan, I.U.; Hano, C. Green Bio-Assisted Synthesis, Characterization and Biological Evaluation of Biocompatible ZnO NPs Synthesized from Different Tissues of Milk Thistle (Silybum marianum). Nanomaterials 2019, 9, 1171. [Google Scholar] [CrossRef]
- Cortés-Rojas, D.F.; Fernandes de Souza, C.R.; Oliveira, W.P. Clove (Syzygium aromaticum): A precious spice. Asian Pac. J. Trop. Biomed. 2014, 4, 90–96. [Google Scholar] [CrossRef]
- Sharma, K.; Mahato, N.; Cho, M.H.; Lee, Y.R. Converting citrus wastes into value-added products: Economic and environmently friendly approaches. Nutrition 2017, 34, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Heying, E.; Tanumihardjo, S.A. History, global distribution, and nutritional importance of citrus fruits. Compr. Rev. Food Sci. Food Saf. 2012, 11, 530–545. [Google Scholar] [CrossRef]
- Prezotti, F.G.; Boni, F.I.; Ferreira, N.N.; de Souza e Silva, D.; Campana-Filho, S.P.; Almeida, A.; Vasconcelos, T.; Daflon Gremião, M.P.; Stringhetti Ferreira Cury, B.; Sarmento, B. Gellan Gum/Pectin Beads Are Safe and Efficient for the Targeted Colonic Delivery of Resveratrol. Polymers 2018, 10, 50. [Google Scholar] [CrossRef]
- Wong, T.W.; Colombo, G.; Sonvico, F. Pectin Matrix as Oral Drug Delivery Vehicle for Colon Cancer Treatment. AAPS Pharm. Sci. Tech. 2011, 12, 201–214. [Google Scholar] [CrossRef] [PubMed]
- De Fátima Sato, M.; Rigoni, D.C.; Giovanetti Canteri, M.H.; de Oliveira Petkowicz, C.L.; Nogueira, A.; Wosiacki, G. Chemical and instrumental characterization of pectin from dried pomace of eleven apple cultivars. Acta Sci. Agron. 2011, 33, 383–389. [Google Scholar]
- Raut, R.W.; Lakkakula, J.R.; Kolekar, N.S.; Mendhulkar, V.D.; Kashid, S.B. Phytosynthesis of silver nanoparticle using Gliricidia Sepium. Curr. Nanosci. 2009, 5, 117–122. [Google Scholar]
- CLSI. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 11th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 1990. [Google Scholar]
- Wiegand, I.; Hilpert, K.; Hancock, R.E. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat. Protoc. 2008, 3, 163–175. [Google Scholar] [CrossRef]
- ASTM. ASTM F 756-00—Standard Practice for Assessment of Hemolytic Properties of Materials; ASTM International: West Conshohocken, PA, USA, 2000. [Google Scholar]
- Roopan, S.M.; Rohit Madhumitha, G.; Rahuman, A.A.; Kamaraj, C.; Bharathi, A.; Surendra, T.V. Low-cost and ecofriendly phyto-synthesis of silver nanoparticles using Cocos nucifera coir extract and its larvicidal activity. Ind. Crop. Prod. 2013, 43, 631–635. [Google Scholar] [CrossRef]
- Ibrahim, H.M.M. Green synthesis and characterization of silver nanoparticles using banana peel extract and their antimicrobial activity against representative microorganisms. J. Radiat. Res. Appl. Sci. 2015, 8, 265–275. [Google Scholar] [CrossRef]
- Za, A.K.; Abrishami, M.E.; Majid, W.H.A.; Yousefi, R.; Hosseini, S.M. Effects of annealing temperature on some structural and optical properties of ZnO nanoparticles prepared by a modified sol–gel combustion method. Ceram. Int. 2011, 37, 393–398. [Google Scholar]
- Nguyen, V.T.; Vu, V.T.; Nguyen, T.H.; Nguyen, T.A.; Tran, V.K.; Nguyen-Tri, P. Antibacterial Activity of TiO2- and ZnO-Decorated with Silver Nanoparticles. J. Compos. Sci. 2019, 3, 61. [Google Scholar] [CrossRef]
- Lu, W.; Liu, G.; Gao, S.; Xing, S.; Wang, J. Tyrosine-assisted preparation of Ag/ZnO nanocomposites with enhanced photocatalytic performance and synergistic antibacterial activities. Nanotechnology 2008, 19, 445711. [Google Scholar] [CrossRef] [PubMed]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on zinc oxide nanoparticles: Antibacterial activity and toxicity mechanism. Nano Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef]
- Talebian, N.; Amininezhad, S.M.; Doudi, M. Controllable synthesis of ZnO nanoparticles and their morphology-dependent antibacterial and optical properties. J. Photochem. Photobiol. 2013, 120, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Mant, C.T.; Farmer, S.W.; Hancock, R.E.; Vasil, M.L.; Hodges, R.S. Rational design of alpha-helical antimicrobial peptides with enhanced activities and specificity/therapeutic index. J. Biol. Chem. 2005, 280, 12316–12329. [Google Scholar] [CrossRef]
- Muller, P.; Milton, M. The determination and interpretation of the therapeutic index in drug development. Nat. Rev. Drug Discov. 2012, 11, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Kondejewski, L.H.; Jelokhani-Niaraki, M.; Farmer, S.W.; Lix, B.; Kay, C.M.; Sykes, B.D.; Hancock, R.E.W.; Hodges, R.S. Dissociation of Antimicrobial and Hemolytic Activities in Cyclic Peptide Diastereomers by Systematic Alterations in Amphipathicity. J. Biol. Chem. 1999, 274, 13181–13192. [Google Scholar] [CrossRef] [PubMed]




| Sample | MIC (mg/mL) | MBC (mg/mL) | MHC (mg/mL) | IC50 (mg/mL) | TI |
|---|---|---|---|---|---|
| ZnO-CUI | 0.37 | 3.4 | >1 | 0.35 | 0.95 |
| AgZnO-CUI | 0.11 | 2.68 | >1 | 0.90 | 8.18 |
| ZnO-MAND | 0.25 | 4 | >1 | 0.43 | 1.72 |
| AgZnO-MAND | 0.34 | 2.93 | >1 | 0.22 | 0.65 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zgura, I.; Enculescu, M.; Istrate, C.; Negrea, R.; Bacalum, M.; Nedelcu, L.; Barbinta-Patrascu, M.E. Performant Composite Materials Based on Oxide Semiconductors and Metallic Nanoparticles Generated from Cloves and Mandarin Peel Extracts. Nanomaterials 2020, 10, 2146. https://doi.org/10.3390/nano10112146
Zgura I, Enculescu M, Istrate C, Negrea R, Bacalum M, Nedelcu L, Barbinta-Patrascu ME. Performant Composite Materials Based on Oxide Semiconductors and Metallic Nanoparticles Generated from Cloves and Mandarin Peel Extracts. Nanomaterials. 2020; 10(11):2146. https://doi.org/10.3390/nano10112146
Chicago/Turabian StyleZgura, Irina, Monica Enculescu, Cosmin Istrate, Raluca Negrea, Mihaela Bacalum, Liviu Nedelcu, and Marcela Elisabeta Barbinta-Patrascu. 2020. "Performant Composite Materials Based on Oxide Semiconductors and Metallic Nanoparticles Generated from Cloves and Mandarin Peel Extracts" Nanomaterials 10, no. 11: 2146. https://doi.org/10.3390/nano10112146
APA StyleZgura, I., Enculescu, M., Istrate, C., Negrea, R., Bacalum, M., Nedelcu, L., & Barbinta-Patrascu, M. E. (2020). Performant Composite Materials Based on Oxide Semiconductors and Metallic Nanoparticles Generated from Cloves and Mandarin Peel Extracts. Nanomaterials, 10(11), 2146. https://doi.org/10.3390/nano10112146





