Improving the Tribological and Anticorrosion Performance of Waterborne Polyurethane Coating by the Synergistic Effect between Modified Graphene Oxide and Polytetrafluoroethylene
Abstract
1. Introduction
2. Experimental Details
2.1. Reagents and Materials
2.2. Preparation of Modified Graphene Oxide
2.3. Characterization
2.4. Preparation of MGO/WPU Composite Coating and MGO-PTFE/WPU Composite Coating
2.5. Tribological Measurements
2.6. Electrochemical Measurements
3. Results and Discussion
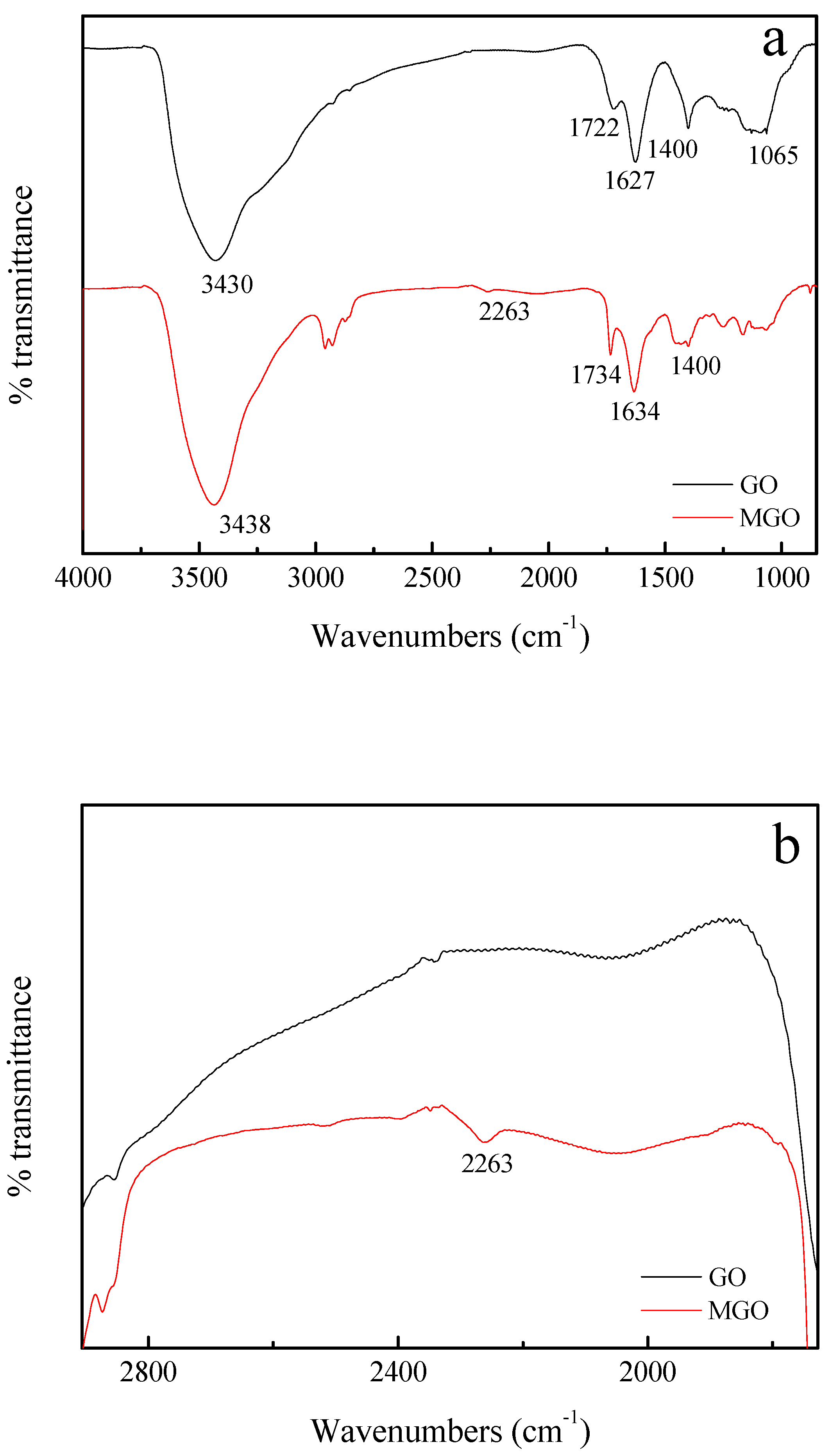
3.1. FTIR Characterization
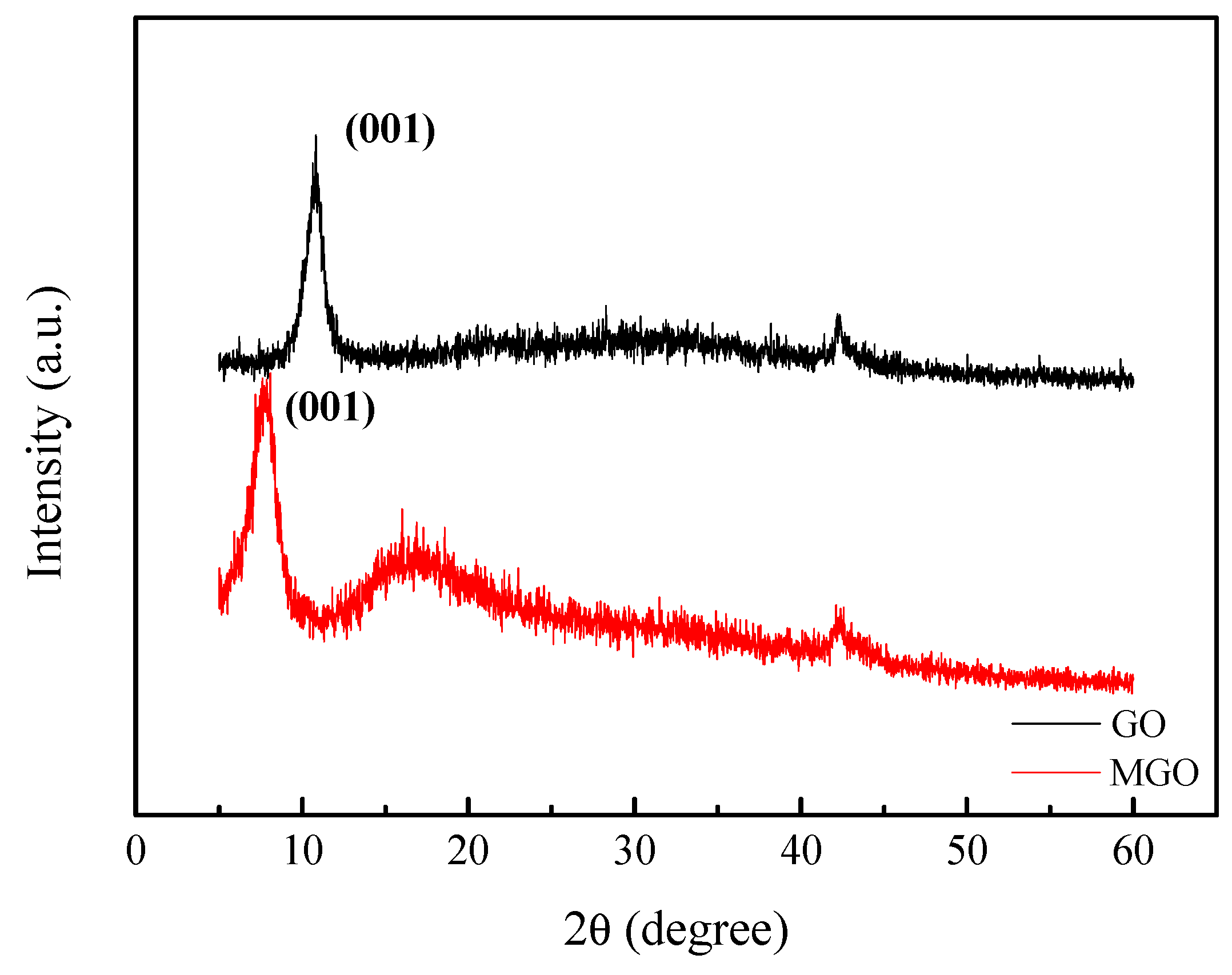
3.2. XRD Characterization
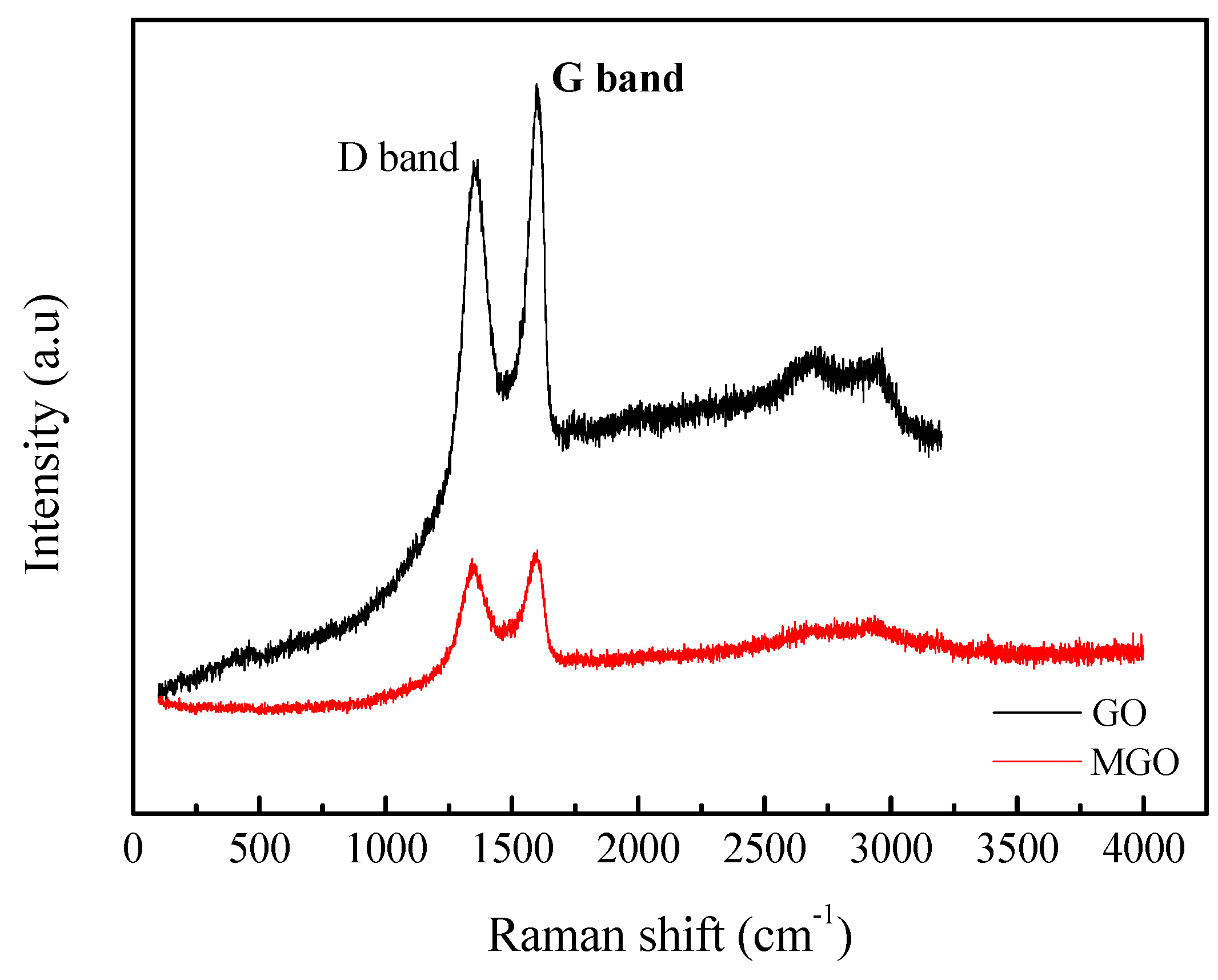
3.3. Raman Characterization
3.4. Elemental Analysis
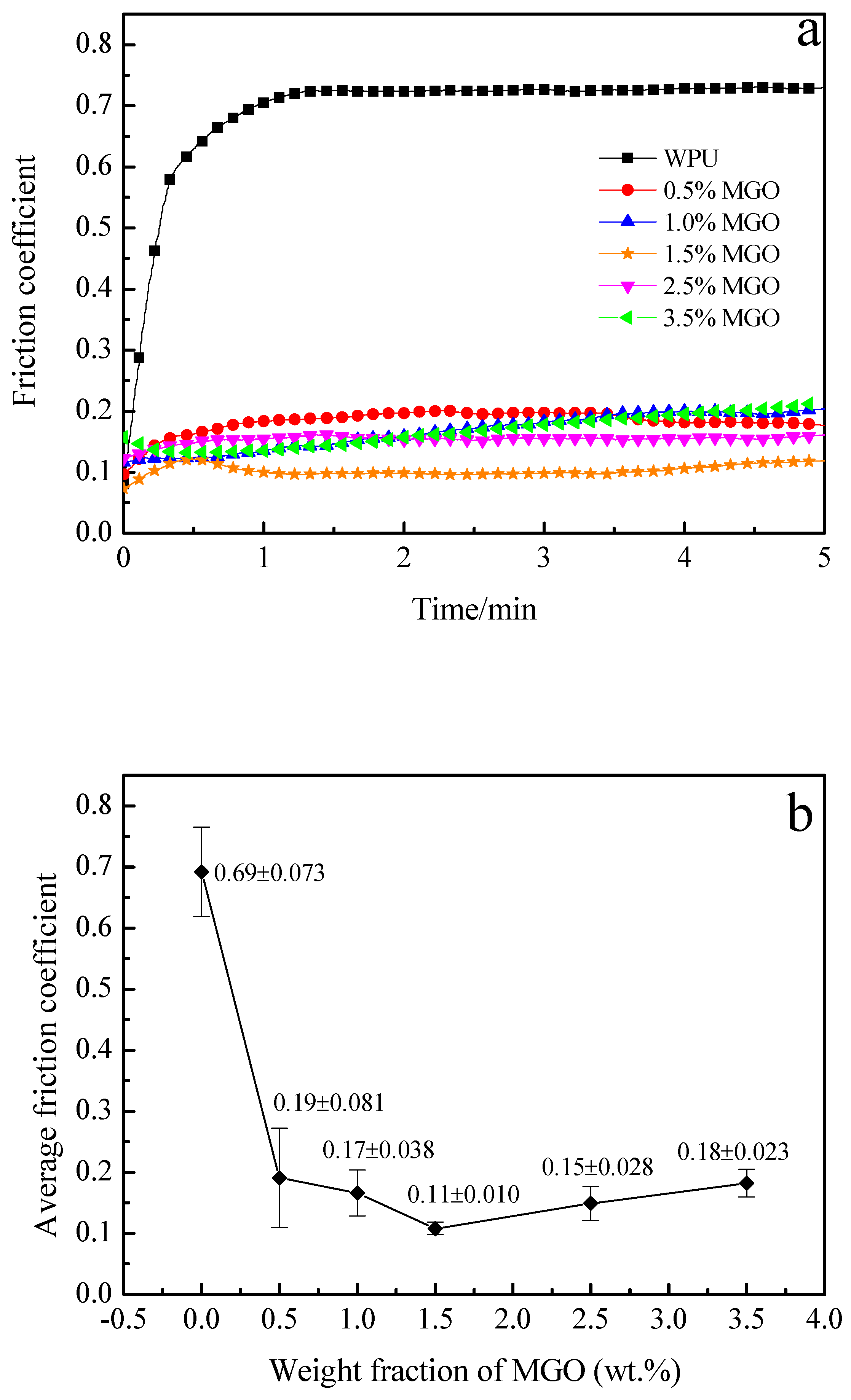
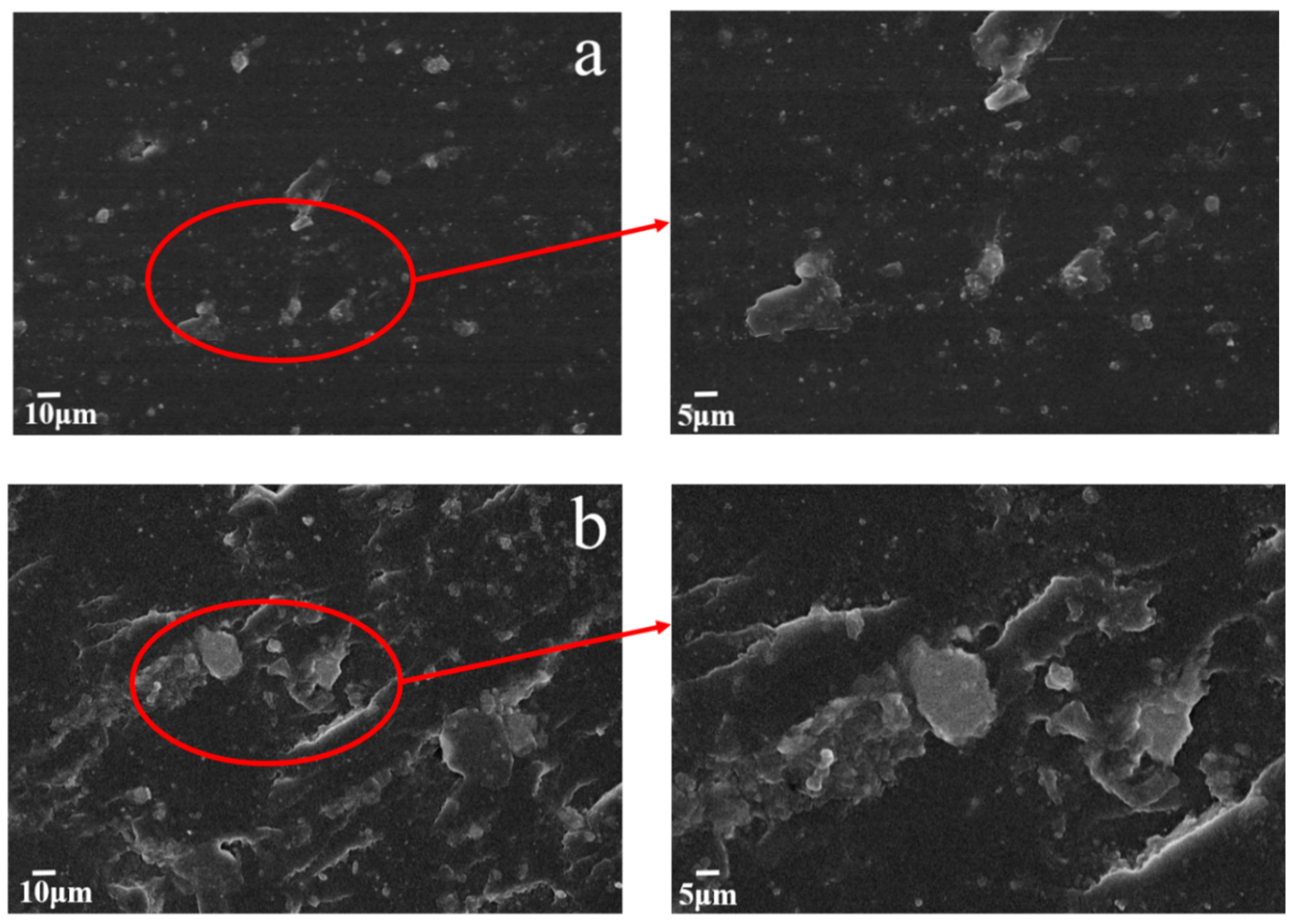
3.5. Tribological Performance of the MGO/WPU Composite Coating
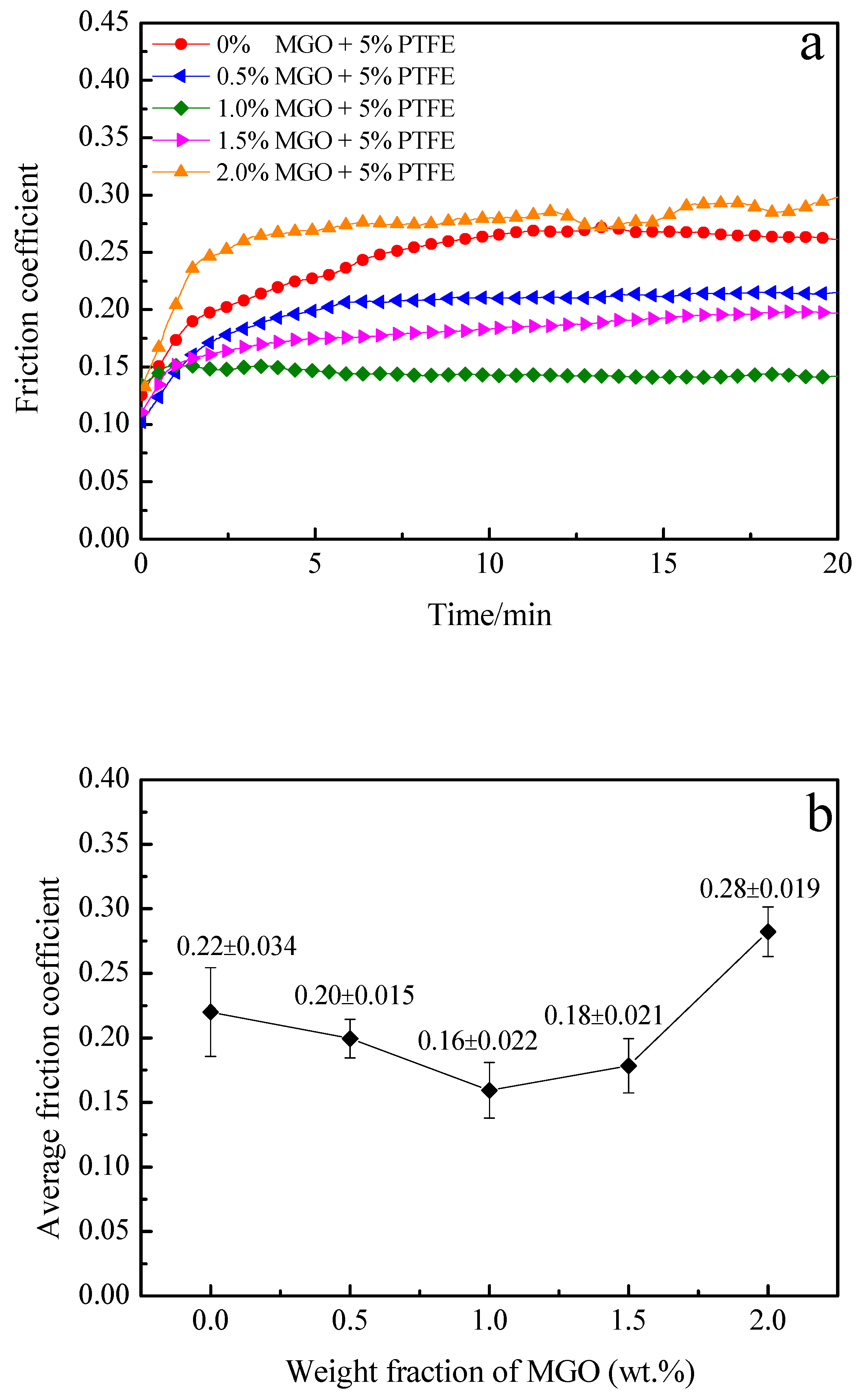
3.6. Tribological Performance of the MGO-PTFE/WPU Composite Coating
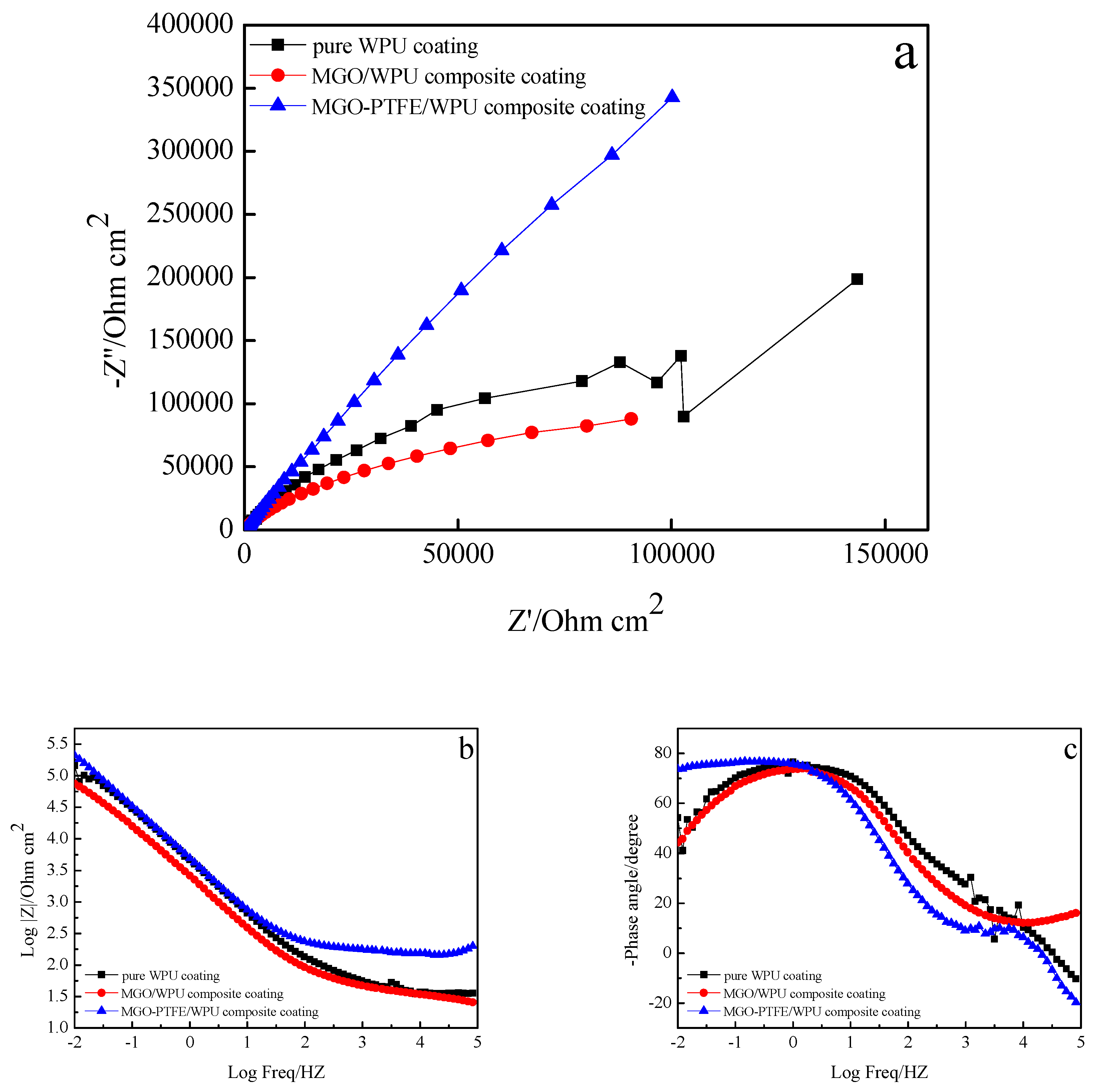
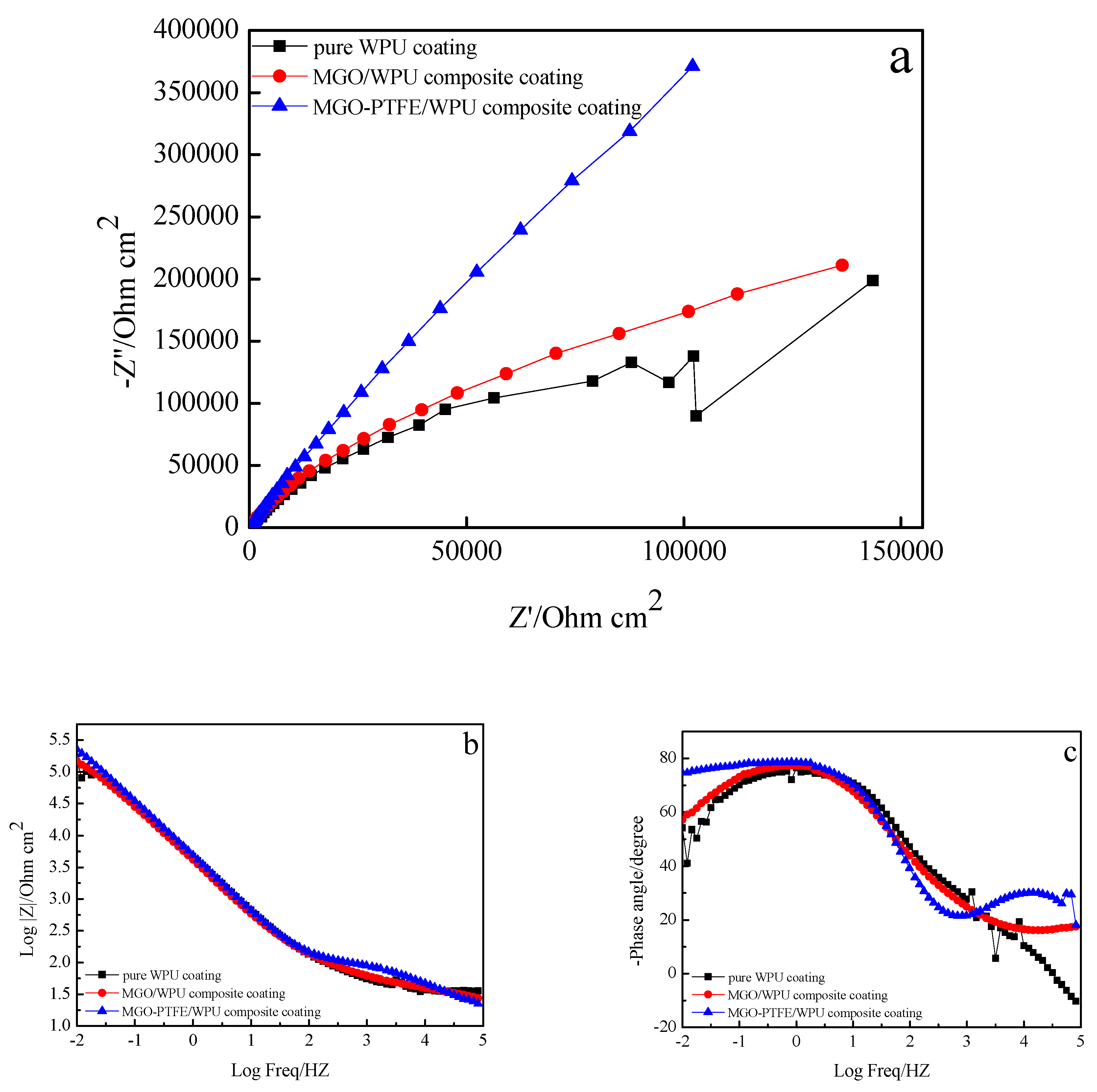
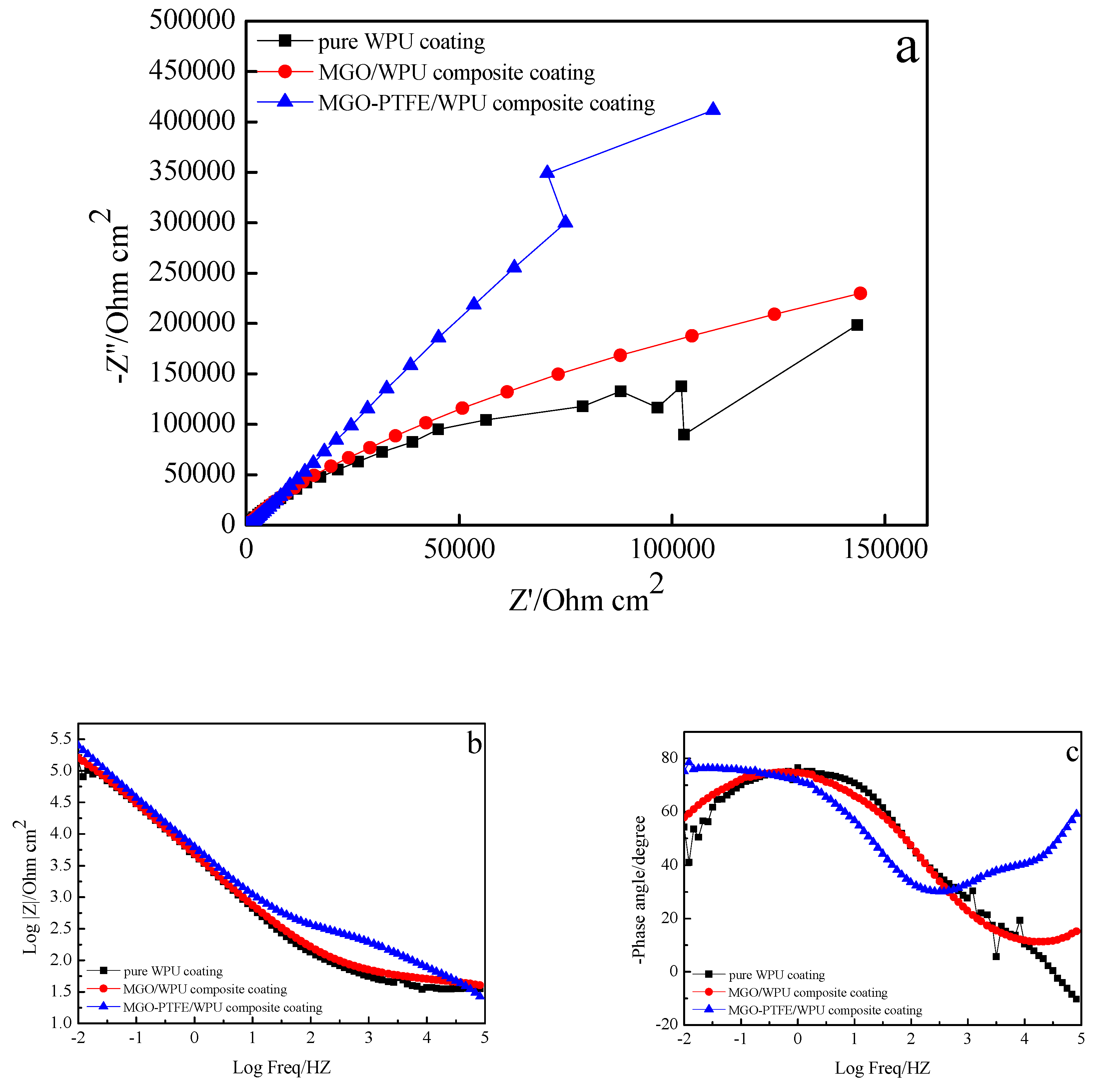
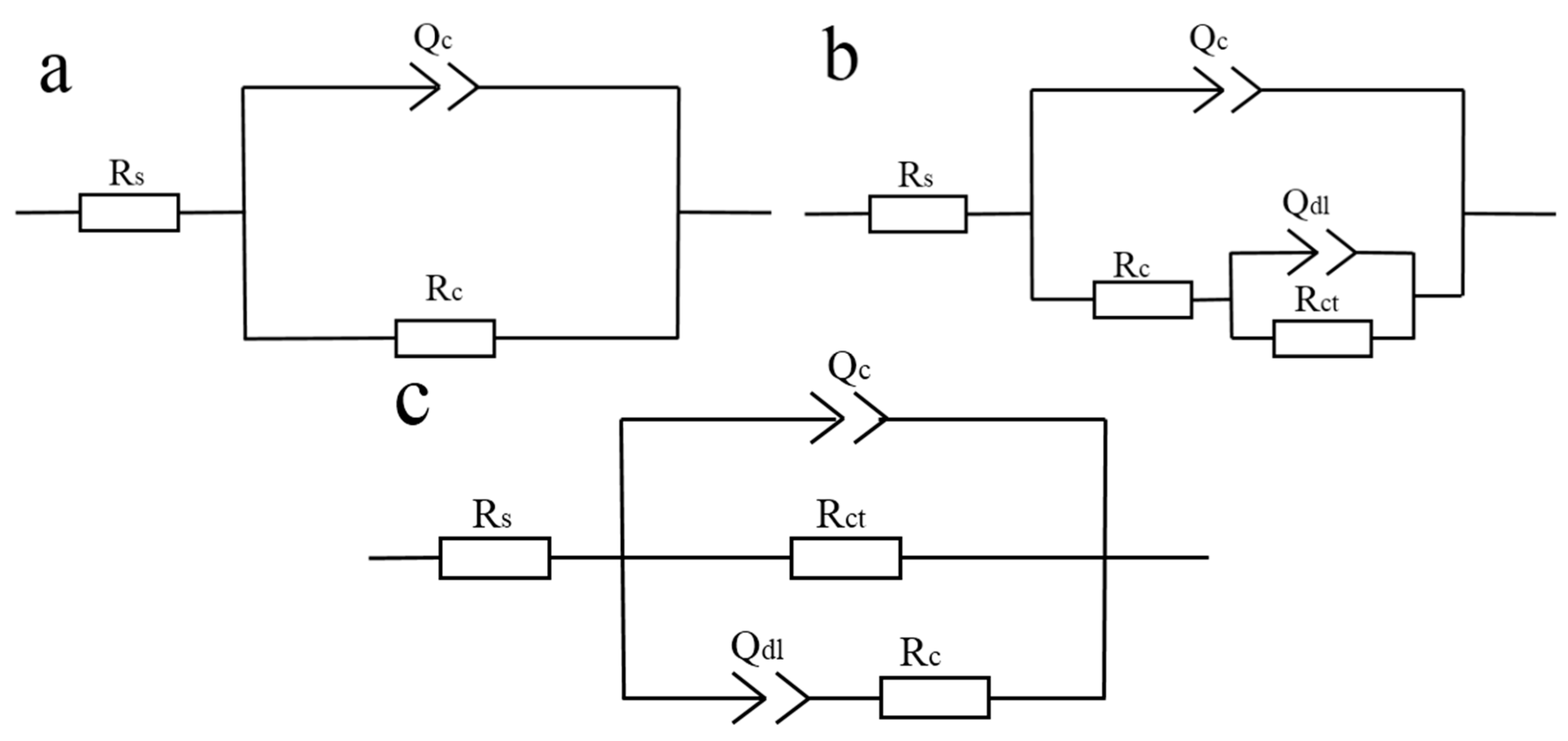
3.7. Anticorrosion Performance of MGO/WPU Composite Coating and MGO-PTFE/WPU Composite Coating
4. Conclusions
- (1)
- After the surface modification of GO by IPDI, the -NCO functional group is successfully grafted on the surface of MGO.
- (2)
- The tribological performance of MGO-PTFE/WPU composite coating are significantly improved after the addition of MGO and PTFE. This is because both MGO and PTFE have excellent self-lubricating properties, and PTFE can improve the ability of the MGO-PTFE/WPU composite coating to withstand the applied load. Therefore, under the synergistic effect of MGO and PTFE, the MGO-PTFE/WPU composite coating shows the excellent tribological performance.
- (3)
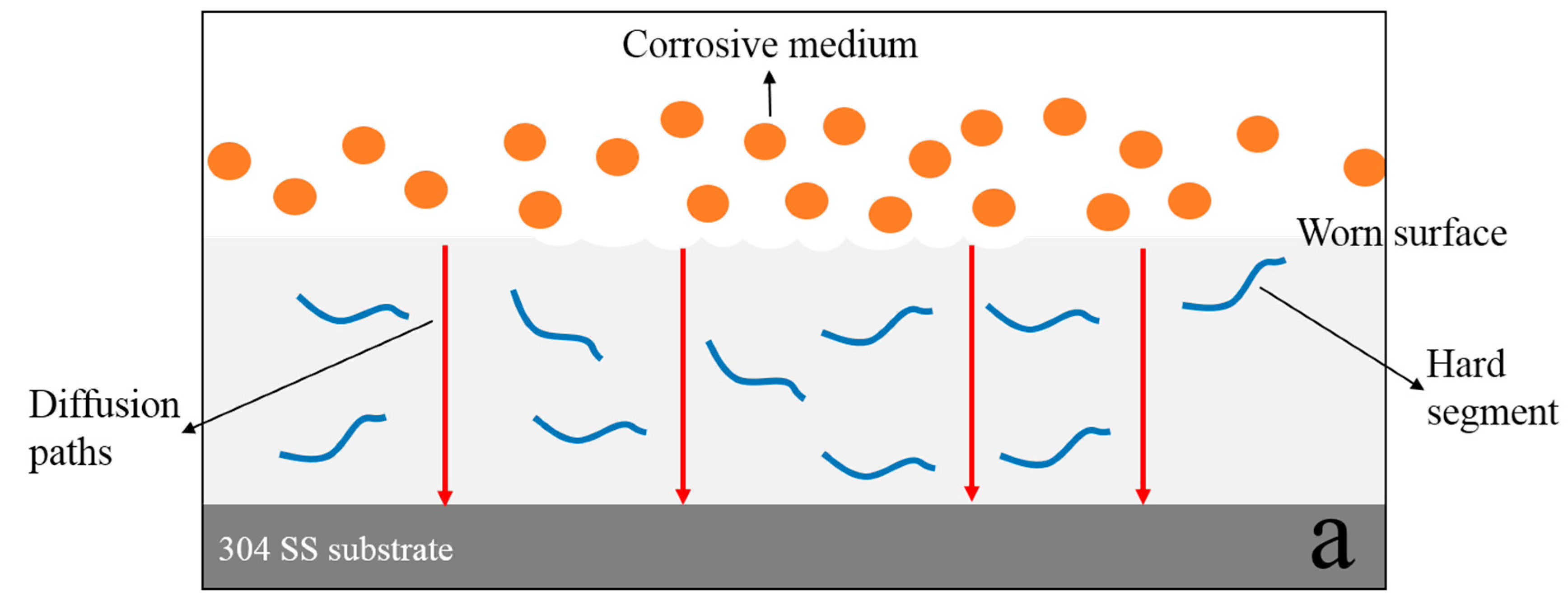
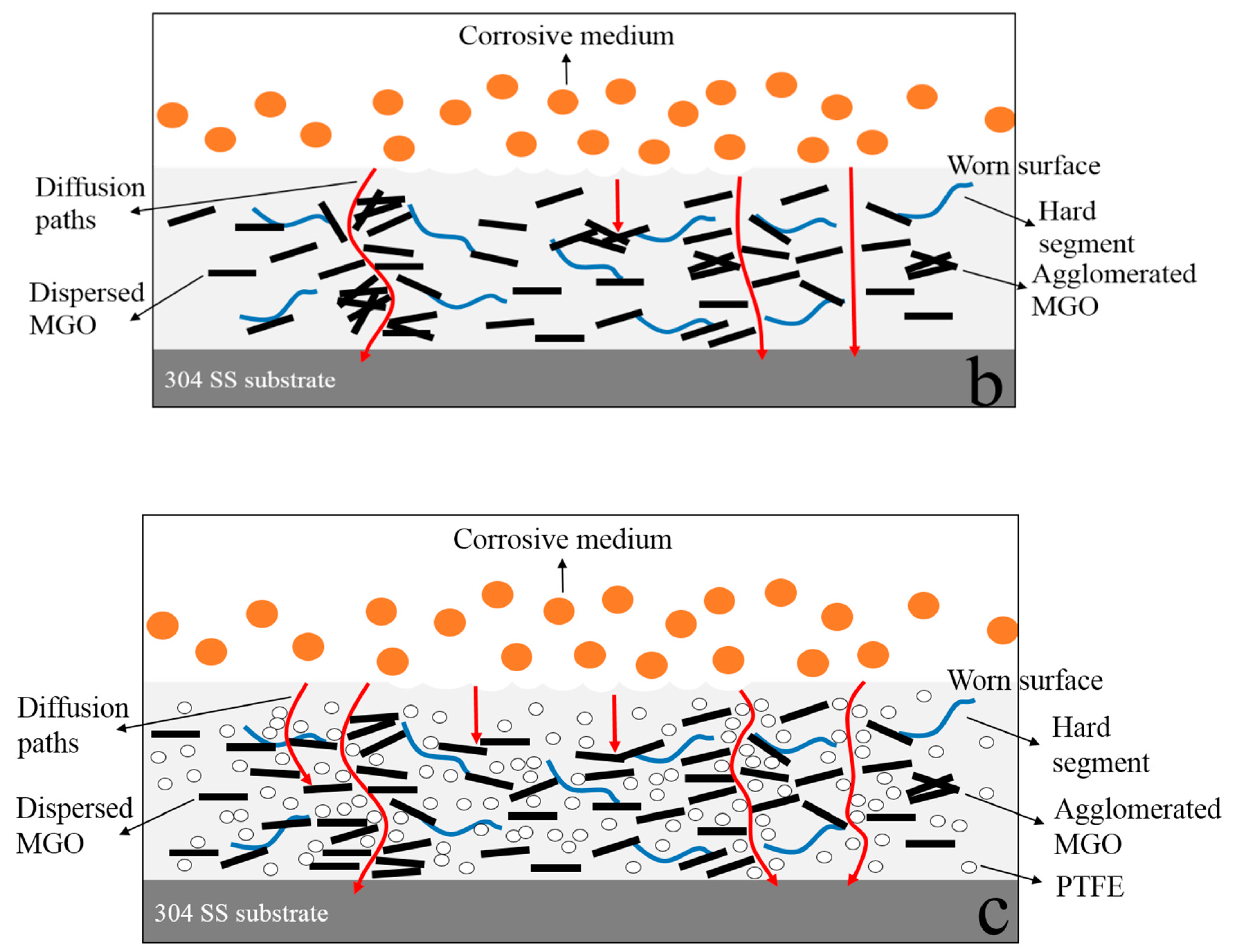
- MGO left in the worn composite coating can act as an excellent barrier to the corrosive medium, and they can zigzag and block the diffusion path of corrosive medium through coating to the metal/coating interface. After continuing to add PTFE, PTFE can promote the dispersion of MGO, and it can fill the gaps between MGO, which further increases the tortuosity of the diffusion paths of corrosive medium. Therefore, the worn MGO-PTFE/WPU composite coating shows the best corrosion resistance.
Author Contributions
Funding
Conflicts of Interest
References
- Lei, L.; Xia, Z.; Zhang, L.; Zhang, Y.; Zhang, L. Preparation and properties of amino-functional reduced graphene oxide/waterborne polyurethane hybrid emulsions. Prog. Org. Coat. 2016, 97, 19–27. [Google Scholar] [CrossRef]
- Zhang, P.; Xu, P.; Fan, H.; Sun, Z.; Wen, J. Covalently functionalized graphene towards molecular-level dispersed waterborne polyurethane nanocomposite with balanced comprehensive performance. Appl. Surf. Sci. 2019, 471, 595–606. [Google Scholar] [CrossRef]
- Guo, Y.-H.; Guo, J.-J.; Miao, H.; Teng, L.-J.; Huang, Z. Properties and paper sizing application of waterborne polyurethane emulsions synthesized with isophorone diisocyanate. Prog. Org. Coat. 2014, 77, 988–996. [Google Scholar] [CrossRef]
- Cheng, D.; Wen, Y.; An, X.; Zhu, X.; Ni, Y. TEMPO-oxidized cellulose nanofibers (TOCNs) as a green reinforcement for waterborne polyurethane coating (WPU) on wood. Carbohydr. Polym. 2016, 151, 326–334. [Google Scholar] [CrossRef]
- Qi, K.; Sun, Y.; Duan, H.; Guo, X. A corrosion-protective coating based on a solution-processable polymer-grafted graphene oxide nanocomposite. Corros. Sci. 2015, 98, 500–506. [Google Scholar] [CrossRef]
- Kousalya, A.S.; Kumar, A.; Paul, R.; Zemlyanov, D.; Fisher, T.S. Graphene: An effective oxidation barrier coating for liquid and two-phase cooling systems. Corros. Sci. 2013, 69, 5–10. [Google Scholar] [CrossRef]
- Kirkland, N.T.; Schiller, T.; Medhekar, N.; Birbilis, N. Exploring graphene as a corrosion protection barrier. Corros. Sci. 2012, 56, 1–4. [Google Scholar] [CrossRef]
- Wang, X.; Xing, W.; Song, L.; Yang, H.; Hu, Y.; Yeoh, G.-H. Fabrication and characterization of graphene-reinforced waterborne polyurethane nanocomposite coatings by the sol–gel method. Surf. Coat. Technol. 2012, 206, 4778–4784. [Google Scholar] [CrossRef]
- Wan, T.; Chen, D. Mechanical enhancement of self-healing waterborne polyurethane by graphene oxide. Prog. Org. Coat. 2018, 121, 73–79. [Google Scholar] [CrossRef]
- Zhang, F.; Liu, W.; Wang, S.; Jiang, C.; Xie, Y.; Yang, M.; Shi, H. A novel and feasible approach for polymer amine modified graphene oxide to improve water resistance, thermal, and mechanical ability of waterborne polyurethane. Appl. Surf. Sci. 2019, 491, 301–312. [Google Scholar] [CrossRef]
- Zhang, D.; Ho, J.K.L.; Dong, G.; Zhang, H.; Hua, M. Tribological properties of Tin-based B abbitt bearing alloy with Polyurethane coating under dry and starved lubrication conditions. Tribol. Int. 2015, 90, 22–31. [Google Scholar] [CrossRef]
- Masood, M.T.; Papadopoulou, E.L.; Heredia-Guerrero, J.A.; Bayer, I.S.; Athanassiou, A.; Ceseracciu, L. Graphene and polytetrafluoroethylene synergistically improve the tribological properties and adhesion of nylon 66 coatings. Carbon 2017, 123, 26–33. [Google Scholar] [CrossRef]
- Zhao, B.; Bai, T. Improving the tribological performance of epoxy coatings by the synergistic effect between dehydrated ethylenediamine modified graphene and polytetrafluoroethylene. Carbon 2019, 144, 481–491. [Google Scholar] [CrossRef]
- Stankovich, S.; Piner, R.D.; Nguyen, S.T.; Ruoff, R.S. Synthesis and exfoliation of isocyanate-treated graphene oxide nanoplatelets. Carbon 2006, 44, 3342–3347. [Google Scholar] [CrossRef]
- Xu, C.; Wu, X.; Zhu, J.; Wang, X. Synthesis of amphiphilic graphite oxide. Carbon 2008, 46, 365–389. [Google Scholar] [CrossRef]
- Parhizkar, N.; Ramezanzadeh, B.; Shahrabi, T. Corrosion protection and adhesion properties of the epoxy coating applied on the steel substrate pre-treated by a sol-gel based silane coating filled with amino and isocyanate silane functionalized graphene oxide nanosheets. Appl. Surf. Sci. 2018, 439, 45–59. [Google Scholar] [CrossRef]
- Parhizkar, N.; Shahrabi, T.; Ramezanzadeh, B. Synthesis and characterization of a unique isocyanate silane reduced graphene oxide nanosheets; Screening the role of multifunctional nanosheets on the adhesion and corrosion protection performance of an amido-amine cured epoxy composite. J. Taiwan Inst. Chem. Eng. 2018, 82, 281–299. [Google Scholar] [CrossRef]
- Lin, P.; Meng, L.; Huang, Y.; Liu, L.; Fan, D. Simultaneously functionalization and reduction of graphene oxide containing isocyanate groups. Appl. Surf. Sci. 2015, 324, 784–790. [Google Scholar] [CrossRef]
- Nimita Jebaranjitham, J.; Mageshwari, C.; Saravanan, R.; Naushad, M. Fabrication of amine functionalized graphene oxide—AgNPs nanocomposite with improved dispersibility for reduction of 4-nitrophenol. Compos. Part B Eng. 2019, 171, 302–309. [Google Scholar] [CrossRef]
- Qin, C.; Liu, D.; Jin, K.; Fang, L.; Xie, G.; Robertson, J. Electrochemical functionalization of 316 stainless steel with polyaniline-graphene oxide: Corrosion resistance study. Mater. Chem. Phys. 2017, 198, 90–98. [Google Scholar] [CrossRef]
- Ding, J.-H.; Zhao, H.-R.; Zheng, Y.; Zhao, X.; Yu, H.-B. A long-term anticorrsive coating through graphene passivation. Carbon 2018, 138, 197–206. [Google Scholar] [CrossRef]
- Dong, Y.; Zhou, Q.; Meng, X.; Cong, C.; Su, X. Anti-H2S corrosion property of bipolar epoxy-resin coatings. Prog. Org. Coat. 2019, 130, 66–74. [Google Scholar] [CrossRef]
- Yan, M.C.; Xu, J.; Yu, L.B.; Wu, T.Q.; Sun, C.; Ke, W. EIS analysis on stress corrosion initiation of pipeline steel under disbanded coating innear-neutral pH simulated soil electrolyte. Corros. Sci. 2016, 110, 23–34. [Google Scholar] [CrossRef]
- Chen, L.; Bai, S.-L.; Ge, Y.-Y.; Wang, Q.-Y. Erosion-corrosion behavior and electrochemical performance of Hastelloy C22 coatings under impingement. Appl. Surf. Sci. 2018, 456, 985–998. [Google Scholar] [CrossRef]
- Pourhashem, S.; Rashidi, A.; Vaezi, M.R.; Bagherzadeh, M.R. Excellent corrosion protection performance of epoxy composite coatings filled with amino-silane functionalized graphene oxide. Surf. Coat. Technol. 2017, 317, 1–9. [Google Scholar] [CrossRef]
- Liu, D.; Zhao, W.J.; Liu, S.; Cen, Q.H.; Xue, Q.J. Comparative tribological and corrosion resistance properties of epoxy composite coatings reinforced with functionalized fullerene C60 and grapheme. Surf. Coat. Technol. 2016, 286, 354–364. [Google Scholar] [CrossRef]


| Sample | C/% | H/% | N/% |
|---|---|---|---|
| GO | 45.82 | 3.07 | ≤0.05 |
| MGO | 58.18 | 7.73 | 9.87 |
| WPU | MGO/WPU | MGO-PTFE/WPU | |
|---|---|---|---|
| Rs/(Ω cm−2) | 6.90 × 10+1 | 4.00 × 10+1 | 2.25 × 10+1 |
| Qc-Yo/(Ω−1 cm−2 s−n) | 2.93 × 10−5 | 4.20 × 10−5 | 6.02 × 10−6 |
| Qc-n | 0.7964 | 0.8239 | 0.6969 |
| Rc/(Ω cm−2) | 7.28 × 10+5 | 2.48 × 10+5 | 3.46 × 10+13 |
| Qdl-Yo/(Ω−1 cm−2 s−n) | / | 1.02 × 10−5 | 2.65 × 10−5 |
| Qdl-n | / | 0.7272 | 0.8267 |
| Rct/(Ω cm−2) | / | 5.06 × 10+1 | 6.01 × 10+1 |
| WPU | MGO/WPU | MGO-PTFE/WPU | |
|---|---|---|---|
| Rs/(Ω cm−2) | 6.90 × 10+1 | 4.79 × 10+1 | 2.75 × 10+1 |
| Qc-Yo/(Ω−1 cm−2 s−n) | 2.93 × 10−5 | 2.28 × 10−5 | 5.80 × 10−6 |
| Qc-n | 0.7964 | 0.8492 | 0.7016 |
| Rc/(Ω cm−2) | 7.28 × 10+5 | 8.61 × 10+5 | 7.37 × 10+13 |
| Qdl-Yo/(Ω−1 cm−2 s−n) | / | 8.29 × 10−6 | 1.97 × 10−5 |
| Qdl-n | / | 0.7583 | 0.8904 |
| Rct/(Ω cm−2) | / | 1.00 × 10+2 | 1.64 × 10+2 |
| WPU | MGO/WPU | MGO-PTFE/WPU | |
|---|---|---|---|
| Rs/(Ω cm−2) | 6.90 × 10+1 | 6.75 × 10+1 | 1.11 × 10+1 |
| Qc-Yo/(Ω−1 cm−2 s−n) | 2.93 × 10−5 | 2.20 × 10−5 | 4.81 × 10−6 |
| Qc-n | 0.7964 | 0.8051 | 0.6552 |
| Rc/(Ω cm−2) | 7.28 × 10+5 | 1.20 × 10+6 | 2.62 × 10+15 |
| Qdl-Yo/(Ω−1 cm−2 s−n) | / | 5.90 × 10−6 | 1.71 × 10−5 |
| Qdl-n | / | 0.7771 | 0.8238 |
| Rct/(Ω cm−2) | / | 1.21 × 10+2 | 5.67 × 10+2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bai, T.; Lv, L.; Du, W.; Fang, W.; Wang, Y. Improving the Tribological and Anticorrosion Performance of Waterborne Polyurethane Coating by the Synergistic Effect between Modified Graphene Oxide and Polytetrafluoroethylene. Nanomaterials 2020, 10, 137. https://doi.org/10.3390/nano10010137
Bai T, Lv L, Du W, Fang W, Wang Y. Improving the Tribological and Anticorrosion Performance of Waterborne Polyurethane Coating by the Synergistic Effect between Modified Graphene Oxide and Polytetrafluoroethylene. Nanomaterials. 2020; 10(1):137. https://doi.org/10.3390/nano10010137
Chicago/Turabian StyleBai, Tao, Lei Lv, Weiping Du, Wenqi Fang, and Yansong Wang. 2020. "Improving the Tribological and Anticorrosion Performance of Waterborne Polyurethane Coating by the Synergistic Effect between Modified Graphene Oxide and Polytetrafluoroethylene" Nanomaterials 10, no. 1: 137. https://doi.org/10.3390/nano10010137
APA StyleBai, T., Lv, L., Du, W., Fang, W., & Wang, Y. (2020). Improving the Tribological and Anticorrosion Performance of Waterborne Polyurethane Coating by the Synergistic Effect between Modified Graphene Oxide and Polytetrafluoroethylene. Nanomaterials, 10(1), 137. https://doi.org/10.3390/nano10010137




