Bioactive Glasses: Where Are We and Where Are We Going?
Abstract
1. Introduction—The Invention of Bioactive Glass
2. Current Clinical Applications of BGs—Where Are We?
3. Grand Challenges for the Future—Where Are We Going?
3.1. Challenge No. 1: Reliable BG Coatings
3.2. Challenge No. 2: Mechanical Properties—BG-Based Strong Scaffolds and Self-Healing Implants
3.3. Challenge No. 3: Beyond Bone Repair—BGs in Contact with Soft Tissues and Interfacial Tissue Engineering
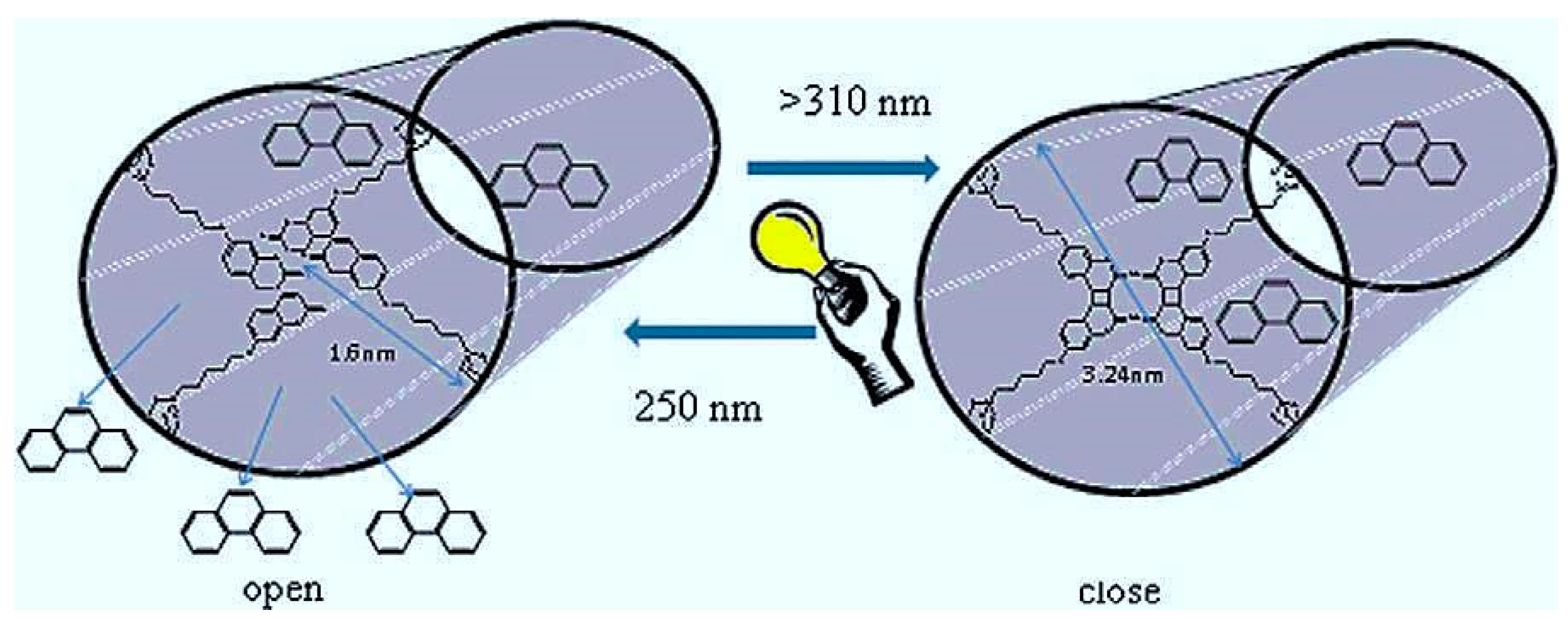
3.4. Challenge No. 4: Bioactive Glasses as Vehicles for the Controlled Release of Biomolecules
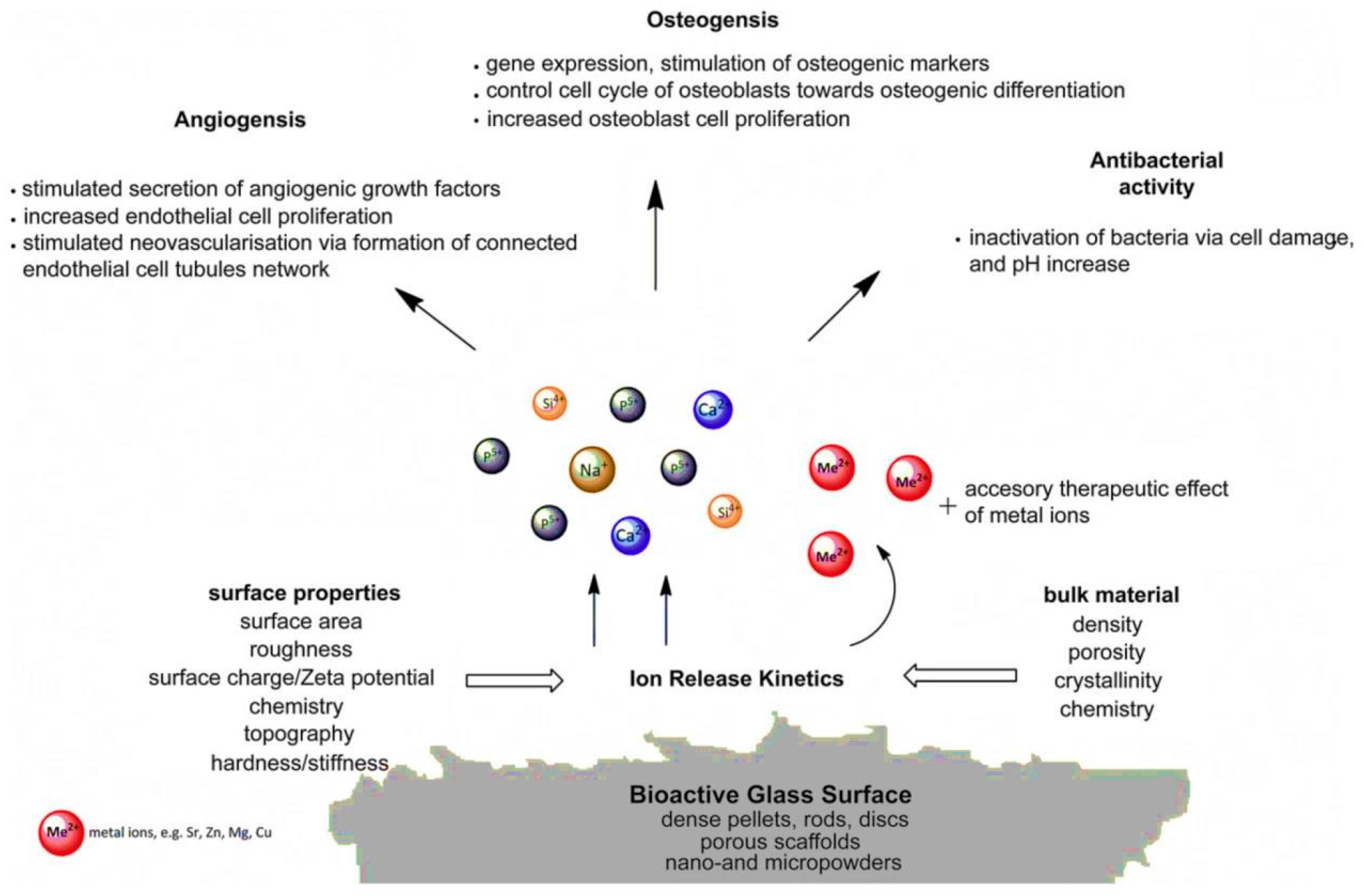
3.5. Challenge No. 5: BGs as Vehicles for the Controlled Release of Therapeutic Ions—Beyond the Pharmaceutical Approach
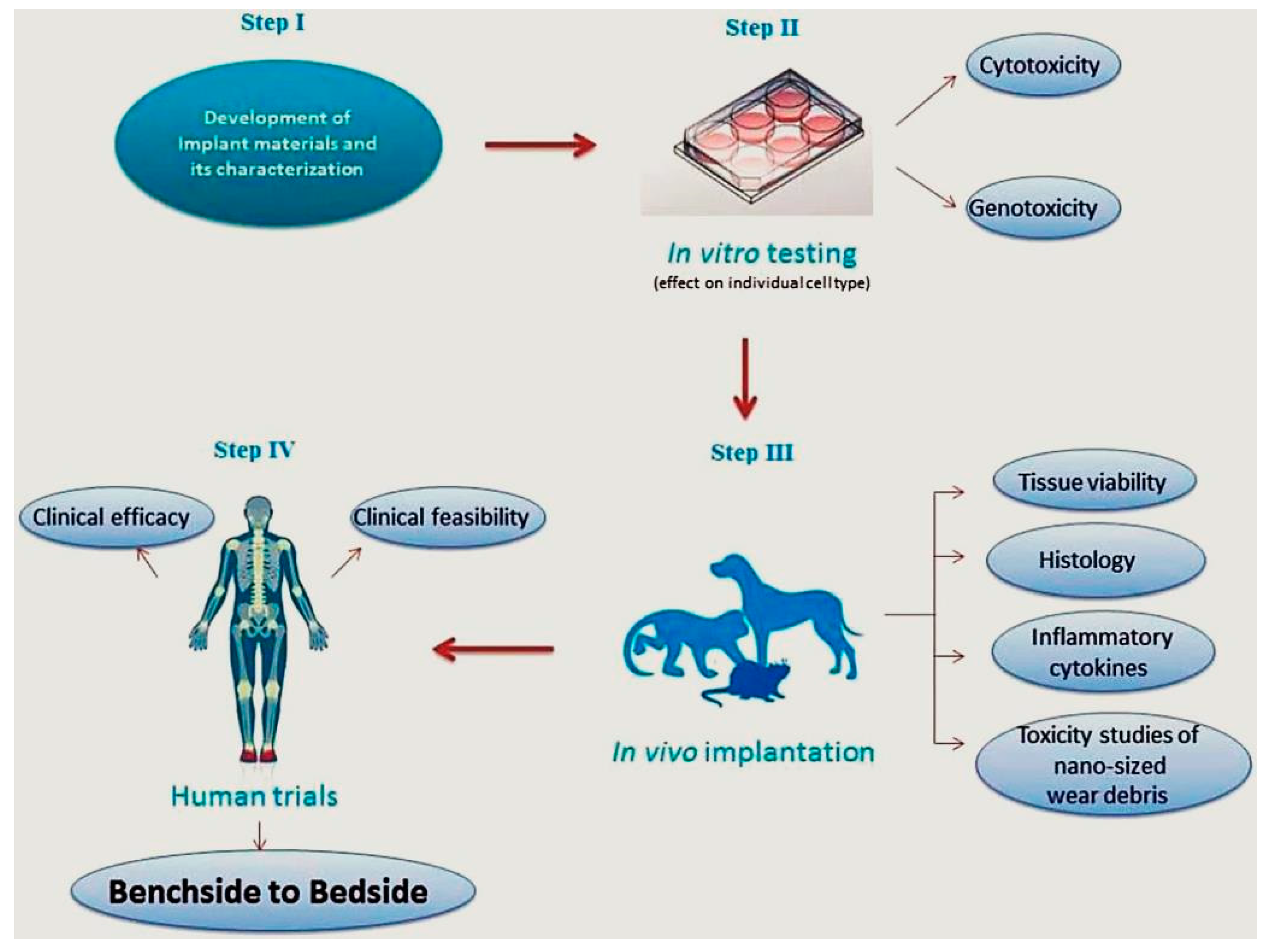
3.6. Challenge No. 6: BGs In Vitro and In Vivo—The Challenge of Having Reliable Testing Methodologies
4. Conclusions
Conflicts of Interest
References
- Williams, D.F. On the mechanisms of biocompatibility. Biomaterials 2008, 29, 2941–2953. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Splinter, R.J.; Allen, W.; Greenlee, T. Bonding mechanisms at the interface of ceramic prosthetic materials. J. Biomed. Mater. Res. Part A 1971, 5, 117–141. [Google Scholar] [CrossRef]
- Hench, L.L.; Polak, J.M. Third-generation biomedical materials. Science 2002, 295, 1014–1017. [Google Scholar] [CrossRef] [PubMed]
- Xynos, I.; Hukkanen, M.; Batten, J.; Buttery, L.; Hench, L.; Polak, J. Bioglass® 45S5 stimulates osteoblast turnover and enhances bone formation in vitro: Implications and applications for bone tissue engineering. Calcif. Tissue Int. 2000, 67, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. The story of bioglass®. J. Mater. Sci. Mater. Med. 2006, 17, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.; Pigott, G.; Schoen, F.; Hench, L. Toxicology and biocompatibility of bioglasses. J. Biomed. Mater. Res. Part A 1981, 15, 805–817. [Google Scholar] [CrossRef] [PubMed]
- Montazerian, M.; Zanotto, E.D. A guided walk through larry hench’s monumental discoveries. J. Mater. Sci. 2017, 52, 8695–8732. [Google Scholar] [CrossRef]
- Rahaman, M.N.; Day, D.E.; Bal, B.S.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive glass in tissue engineering. Acta Biomater. 2011, 7, 2355–2373. [Google Scholar] [CrossRef] [PubMed]
- Rabiee, S.M.; Nazparvar, N.; Azizian, M.; Vashaee, D.; Tayebi, L. Effect of ion substitution on properties of bioactive glasses: A review. Ceram. Int. 2015, 41, 7241–7251. [Google Scholar] [CrossRef]
- Bellantone, M.; Coleman, N.J.; Hench, L.L. Bacteriostatic action of a novel four-component bioactive glass. J. Biomed. Mater. Res. Part A 2000, 51, 484–490. [Google Scholar] [CrossRef]
- Miola, M.; Verné, E.; Vitale-Brovarone, C.; Baino, F. Antibacterial bioglass-derived scaffolds: Innovative synthesis approach and characterization. Int. J. Appl. Glass Sci. 2016, 7, 238–247. [Google Scholar] [CrossRef]
- Gentleman, E.; Fredholm, Y.C.; Jell, G.; Lotfibakhshaiesh, N.; O’Donnell, M.D.; Hill, R.G.; Stevens, M.M. The effects of strontium-substituted bioactive glasses on osteoblasts and osteoclasts in vitro. Biomaterials 2010, 31, 3949–3956. [Google Scholar] [CrossRef] [PubMed]
- Molino, G.; Bari, A.; Baino, F.; Fiorilli, S.; Vitale-Brovarone, C. Electrophoretic deposition of spray-dried Sr-containing mesoporous bioactive glass spheres on glass–ceramic scaffolds for bone tissue regeneration. J. Mater. Sci. 2017, 52, 9103–9114. [Google Scholar] [CrossRef]
- Jones, J.R. Reprint of: Review of bioactive glass: From hench to hybrids. Acta Biomater. 2015, 23, S53–S82. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. Genetic design of bioactive glass. J. Eur. Ceram. Soc. 2009, 29, 1257–1265. [Google Scholar] [CrossRef]
- Jones, J.R.; Brauer, D.S.; Hupa, L.; Greenspan, D.C. Bioglass and bioactive glasses and their impact on healthcare. Int. J. Appl. Glass Sci. 2016, 7, 423–434. [Google Scholar] [CrossRef]
- Merwin, G.E. Bioglass middle ear prosthesis: Preliminary report. Ann. Otol. Rhinol. Laryngol. 1986, 95, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Greenspan, D. Interactions between bioactive glass and collagen: A review and new perspectives. J. Aust. Ceram. Soc. 2013, 49, 1–40. [Google Scholar]
- Wilson, J.; Douek, D.E.; Rust, K. Bioglass middle ear devices: Ten year clinical results. Bioceramics 1995, 8, 239–245. [Google Scholar]
- Bahmad, F., Jr.; Merchant, S.N. Histopathology of ossicular grafts and implants in chronic otitis media. Ann. Otol. Rhinol. Laryngol. 2007, 116, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Reck, R.; Störkel, S.; Meyer, A. Bioactive glass-ceramics in middle ear surgery an 8-year review. Ann. N. Y. Acad. Sci. 1988, 523, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Walliker, J.; Carson, H.; Douek, E.; Fourcin, A.; Rosen, S. An extracochlear auditory prosthesis. In Proceedings of the Cochlear Implant Symposium, Düren, Germany, 7–12 September 1987; Volume 265. [Google Scholar]
- Downing, M.; Johansson, U.; Carlsson, L.; Walliker, J.; Spraggs, P.; Dodson, H.; Hochmair-Desoyer, I.; Albrektsson, T. A bone-anchored percutaneous connector system for neural prosthetic applications. Ear Nose Throat J. 1997, 76, 328–332. [Google Scholar] [PubMed]
- Stanley, H.R.; Hall, M.B.; Clark, A.E.; King, C.J., III; Hench, L.L.; Berte, J.J. Using 45S5 bioglass cones as endosseous ridge maintenance implants to prevent alveolar ridge resorption: A 5-year evaluation. Int. J. Oral Maxillofac. Implants 1997, 12, 95–105. [Google Scholar] [PubMed]
- Thompson, I. Clinical applications of bioactive glasses for maxillo-facial repair. New Mater. Technol. Healthc. 2012, 377, 77–96. [Google Scholar]
- Suominen, E.; Kinnunen, J. Bioactive glass granules and plates in the reconstruction of defects of the facial bones. Scand. J. Plast. Reconstr. Surg. Hand Surg. 1996, 30, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Kinnunen, I.; Aitasalo, K.; Pöllönen, M.; Varpula, M. Reconstruction of orbital floor fractures using bioactive glass. J. Cranio-Maxillofac. Surg. 2000, 28, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Aitasalo, K.; Kinnunen, I.; Palmgren, J.; Varpula, M. Repair of orbital floor fractures with bioactive glass implants. J. Oral Maxillofac. Surg. 2001, 59, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.; Hench, J.W.; Greenspan, D. Bioglass: A short history and bibliography. J. Australas. Ceram. Soc. 2004, 40, 1–42. [Google Scholar]
- Zamet, J.; Darbar, U.; Griffiths, G.; Bulman, J.; Brägger, U.; Bürgin, W.; Newman, H. Particulate Bioglass® as a grafting material in the treatment of periodontal intrabony defects. J. Clin. Periodontol. 1997, 24, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Ilharreborde, B.; Morel, E.; Fitoussi, F.; Presedo, A.; Souchet, P.; Penneçot, G.-F.; Mazda, K. Bioactive glass as a bone substitute for spinal fusion in adolescent idiopathic scoliosis: A comparative study with iliac crest autograft. J. Pediatr. Orthop. 2008, 28, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Tadjoedin, E.S.; De Lange, G.L.; Lyaruu, D.; Kuiper, L.; Burger, E.H. High concentrations of bioactive glass material (Biogran®) vs. Autogenous bone for sinus floor elevation. Clin. Oral Implants Res. 2002, 13, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Fujikura, K.; Karpukhina, N.; Kasuga, T.; Brauer, D.; Hill, R.; Law, R. Influence of strontium substitution on structure and crystallisation of Bioglass® 45S5. J. Mater. Chem. 2012, 22, 7395–7402. [Google Scholar] [CrossRef]
- Hill, R.G.; Stevens, M.M. Bioactive Glass. U.S. Patent US2,009,020,842,8A1, 20 August 2009. [Google Scholar]
- Peltola, M.; Aitasalo, K.; Suonpää, J.; Varpula, M.; Yli-Urpo, A. Bioactive glass S53P4 in frontal sinus obliteration: A long-term clinical experience. Head Neck 2006, 28, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Lindfors, N.C.; Koski, I.; Heikkilä, J.T.; Mattila, K.; Aho, A.J. A prospective randomized 14-year follow-up study of bioactive glass and autogenous bone as bone graft substitutes in benign bone tumors. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 94, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Stoor, P.; Pulkkinen, J.; Grénman, R. Bioactive glass s53p4 in the filling of cavities in the mastoid cell area in surgery for chronic otitis media. Ann. Otol. Rhinol. Laryngol. 2010, 119, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Silvola, J.T. Mastoidectomy cavity obliteration with bioactive glass: A pilot study. Otolaryngol. Head Neck Surg. 2012, 147, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.Z.; Thompson, I.D.; Boccaccini, A.R. 45S5 Bioglass®-derived glass–ceramic scaffolds for bone tissue engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Baino, F.; Spriano, S.; Pugno, N.M.; Vitale-Brovarone, C. Modelling of the strength-porosity relationship in glass-ceramic foam scaffolds for bone repair. J. Eur. Ceram. Soc. 2014, 34, 2663–2673. [Google Scholar] [CrossRef]
- Fu, Q.; Rahaman, M.N.; Bal, B.S.; Brown, R.F.; Day, D.E. Mechanical and in vitro performance of 13–93 bioactive glass scaffolds prepared by a polymer foam replication technique. Acta Biomater. 2008, 4, 1854–1864. [Google Scholar] [CrossRef] [PubMed]
- Gillam, D.; Tang, J.; Mordan, N.; Newman, H. The effects of a novel Bioglass® dentifrice on dentine sensitivity: A scanning electron microscopy investigation. J. Oral Rehabilit. 2002, 29, 305–313. [Google Scholar] [CrossRef]
- Tai, B.J.; Bian, Z.; Jiang, H.; Greenspan, D.C.; Zhong, J.; Clark, A.E.; Du, M.Q. Anti-gingivitis effect of a dentifrice containing bioactive glass (Novamin®) particulate. J. Clin. Periodontol. 2006, 33, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.; Sharma, A. Comparison of clinical efficacy of a dentifrice containing calcium sodium phosphosilicate to a dentifrice containing potassium nitrate and to a placebo on dentinal hypersensitivity: A randomized clinical trial. J. Periodontol. 2010, 81, 1167–1173. [Google Scholar] [CrossRef] [PubMed]
- Golpayegani, M.V.; Sohrabi, A.; Biria, M.; Ansari, G. Remineralization effect of topical Novamin versus sodium fluoride (1.1%) on caries-like lesions in permanent teeth. J. Dent. (Tehran, Iran) 2012, 9, 68–75. [Google Scholar]
- Banerjee, A.; Hajatdoost-Sani, M.; Farrell, S.; Thompson, I. A clinical evaluation and comparison of bioactive glass and sodium bicarbonate air-polishing powders. J. Dent. 2010, 38, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Xie, D.; Feng, D.; Chung, I.-D.; Eberhardt, A.W. A hybrid zinc–calcium–silicate polyalkenoate bone cement. Biomaterials 2003, 24, 2749–2757. [Google Scholar] [CrossRef]
- Boyd, D.; Clarkin, O.; Wren, A.; Towler, M. Zinc-based glass polyalkenoate cements with improved setting times and mechanical properties. Acta Biomater. 2008, 4, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Brauer, D.S.; Gentleman, E.; Farrar, D.F.; Stevens, M.M.; Hill, R.G. Benefits and drawbacks of zinc in glass ionomer bone cements. Biomed. Mater. 2011, 6, 045007. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.; Towler, M.; Law, R.; Hill, R. An investigation into the structure and reactivity of calcium-zinc-silicate ionomer glasses using mas-nmr spectroscopy. J. Mater. Sci. Mater. Med. 2006, 17, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, P.; Strobel, L.A.; Kneser, U.; Boccaccini, A.R. Zinc-containing bioactive glasses for bone regeneration, dental and orthopedic applications. Biomed. Glasses 2015, 1. [Google Scholar] [CrossRef]
- Baino, F.; Novajra, G.; Miguez-Pacheco, V.; Boccaccini, A.R.; Vitale-Brovarone, C. Bioactive glasses: Special applications outside the skeletal system. J. Non-Cryst. Solids 2016, 432, 15–30. [Google Scholar] [CrossRef]
- Wray, P. Cotton candy’ that heals. Am. Ceram. Soc. Bull. 2011, 90, 24–31. [Google Scholar]
- Jung, S.; Day, D.; Day, T.; Stoecker, W.; Taylor, P. Treatment of non-healing diabetic venous stasis ulcers with bioactive glass nanofibers. Wound Repair Regen. 2011, 19, A30. [Google Scholar]
- Lin, Y.; Brown, R.F.; Jung, S.B.; Day, D.E. Angiogenic effects of borate glass microfibers in a rodent model. J. Biomed. Mater. Res. Part A 2014, 102, 4491–4499. [Google Scholar] [CrossRef] [PubMed]
- Baino, F. How can bioactive glasses be useful in ocular surgery? J. Biomed. Mater. Res. Part A 2015, 103, 1259–1275. [Google Scholar] [CrossRef] [PubMed]
- Naik, M.N.; Murthy, R.K.; Honavar, S.G. Comparison of vascularization of medpor and medpor-plus orbital implants: A prospective, randomized study. Ophthalmic Plast. Reconstr. Surg. 2007, 23, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Schou, K.R.; Maloney-Schou, M.; Harwin, F.M.; Ng, J.D. The porous polyethylene/bioglass spherical orbital implant: A retrospective study of 170 cases. Ophthalmic Plast. Reconstr. Surg. 2011, 27, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, T.; Glasby, M.; Healy, D.; Kelly, G.; Lenihan, D.; McDowall, K.; Miller, I.; Myles, L. In vitro nerve repair—In vivo. The reconstruction of peripheral nerves by entubulation with biodegradeable glass tubes—A preliminary report. Br. J. Plast. Surg. 1998, 51, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Ehrhardt, G.J.; Day, D.E. Therapeutic use of 90y microspheres. Int. J. Radiat. Appl. Instrum. Part B Nucl. Med. Biol. 1987, 14, 233–242. [Google Scholar] [CrossRef]
- Hench, L.L.; Wilson, J. An Introduction to Bioceramics; World Scientific: Singapore, 1993; Volume 1. [Google Scholar]
- Day, D.E. Glasses for radiotherapy. In Bio-Glasses; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2012; pp. 203–228. [Google Scholar]
- Baskar, R.; Lee, K.A.; Yeo, R.; Yeoh, K.-W. Cancer and radiation therapy: Current advances and future directions. Int. J. Med. Sci. 2012, 9, 193–199. [Google Scholar] [CrossRef] [PubMed]
- McEntire, B.; Bal, B.; Rahaman, M.; Chevalier, J.; Pezzotti, G. Ceramics and ceramic coatings in orthopaedics. J. Eur. Ceram. Soc. 2015, 35, 4327–4369. [Google Scholar] [CrossRef]
- Sun, L.; Berndt, C.C.; Gross, K.A.; Kucuk, A. Material fundamentals and clinical performance of plasma-sprayed hydroxyapatite coatings: A review. J. Biomed. Mater. Res. Part A 2001, 58, 570–592. [Google Scholar] [CrossRef] [PubMed]
- Sola, A.; Bellucci, D.; Cannillo, V.; Cattini, A. Bioactive glass coatings: A review. Surf. Eng. 2011, 27, 560–572. [Google Scholar] [CrossRef]
- Alonso-Barrio, J.; Sanchez-Herraez, S.; Fernandez-Hernandez, O.; Betegon-Nicolas, J.; Gonzalez-Fernandez, J.; Lopez-Sastre, A. Bioglass-coated femoral stem. Orthop. Proc. 2004, 86, 138. [Google Scholar]
- Baino, F.; Verné, E. Glass-based coatings on biomedical implants: A state-of-the-art review. Biomed. Glasses 2017, 3, 1–17. [Google Scholar] [CrossRef]
- Gomez-Vega, J.M.; Saiz, E.; Tomsia, A.P.; Oku, T.; Suganuma, K.; Marshall, G.W.; Marshall, S.J. Novel bioactive functionally graded coatings on ti6al4v. Adv. Mater. 2000, 12, 894–898. [Google Scholar] [CrossRef]
- Boccaccini, A.; Keim, S.; Ma, R.; Li, Y.; Zhitomirsky, I. Electrophoretic deposition of biomaterials. J. R. Soc. Interface 2010, 7, S581–S613. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Ferraris, S.; Miola, M.; Perero, S.; Verné, E.; Coggiola, A.; Dolcino, D.; Ferraris, M. Novel antibacterial ocular prostheses: Proof of concept and physico-chemical characterization. Mater. Sci. Eng. C 2016, 60, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Bretcanu, O.; Chen, Q.; Misra, S.K.; Boccaccini, A.R.; Roy, I.; Verne, E.; Brovarone, C.V. Biodegradable polymer coated 45s5 bioglassderived glass-ceramic scaffolds for bone tissue engineering. Glass Technol.-Eur. J. Glass Sci. Technol. Part A 2007, 48, 227–234. [Google Scholar]
- Řehořek, L.; Chlup, Z.; Meng, D.; Yunos, D.; Boccaccini, A.; Dlouhý, I. Response of 45s5 bioglass® foams to tensile loading. Ceram. Int. 2013, 39, 8015–8020. [Google Scholar] [CrossRef]
- Gmeiner, R.; Deisinger, U.; Schönherr, J.; Lechner, B.; Detsch, R.; Boccaccini, A.R.; Stampfl, J. Additive manufacturing of bioactive glasses and silicate bioceramics. J. Ceram. Sci. Technol. 2015, 6, 75–86. [Google Scholar]
- Baino, F.; Fiorilli, S.; Vitale-Brovarone, C. Bioactive glass-based materials with hierarchical porosity for medical applications: Review of recent advances. Acta Biomater. 2016, 42, 18–32. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.R.; Ehrenfried, L.M.; Hench, L.L. Optimising bioactive glass scaffolds for bone tissue engineering. Biomaterials 2006, 27, 964–973. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhang, Y.; Zhu, Y.; Friis, T.; Xiao, Y. Structure–property relationships of silk-modified mesoporous bioglass scaffolds. Biomaterials 2010, 31, 3429–3438. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Luo, Y.; Cuniberti, G.; Xiao, Y.; Gelinsky, M. Three-dimensional printing of hierarchical and tough mesoporous bioactive glass scaffolds with a controllable pore architecture, excellent mechanical strength and mineralization ability. Acta Biomater. 2011, 7, 2644–2650. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhao, S.; Zhu, Y.; Huang, Y.; Zhu, M.; Tao, C.; Zhang, C. Three-dimensional printing of strontium-containing mesoporous bioactive glass scaffolds for bone regeneration. Acta Biomater. 2014, 10, 2269–2281. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Fiorilli, S.; Vitale-Brovarone, C. Composite biomaterials based on sol-gel mesoporous silicate glasses: A review. Bioengineering 2017, 4, 15. [Google Scholar] [CrossRef] [PubMed]
- Novak, B.M. Hybrid nanocomposite materials—Between inorganic glasses and organic polymers. Adv. Mater. 1993, 5, 422–433. [Google Scholar] [CrossRef]
- Wen, J.; Wilkes, G.L. Organic/inorganic hybrid network materials by the sol−gel approach. Chem. Mater. 1996, 8, 1667–1681. [Google Scholar] [CrossRef]
- Owens, G.J.; Singh, R.K.; Foroutan, F.; Alqaysi, M.; Han, C.-M.; Mahapatra, C.; Kim, H.-W.; Knowles, J.C. Sol–gel based materials for biomedical applications. Prog. Mater. Sci. 2016, 77, 1–79. [Google Scholar] [CrossRef]
- Martín, A.; Salinas, A.; Vallet-Regí, M. Bioactive and degradable organic–inorganic hybrids. J. Eur. Ceram. Soc. 2005, 25, 3533–3538. [Google Scholar] [CrossRef]
- Kamitakahara, M.; Kawashita, M.; Miyata, N.; Kokubo, T.; Nakamura, T. Bioactivity and mechanical properties of polydimethylsiloxane (PDMS)–CaO–SiO2 hybrids with different calcium contents. J. Mater. Sci. Mater. Med. 2002, 13, 1015–1020. [Google Scholar] [CrossRef] [PubMed]
- Maçon, A.L.; Kasuga, T.; Becer, C.R.; Jones, J.R. Silica/methacrylate class ii hybrid: Telomerisation vs. Raft polymerisation. Polym. Chem. 2017, 8, 3603–3611. [Google Scholar] [CrossRef]
- Miguez-Pacheco, V.; Greenspan, D.; Hench, L.; Boccaccini, A. Bioactive glasses in soft tissue repair. Am. Ceram. Soc. Bull. 2015, 94, 27–31. [Google Scholar]
- Kargozar, S.; Hamzehlou, S.; Baino, F. Potential of bioactive glasses for cardiac and pulmonary tissue engineering. Materials 2017, 10, 1429. [Google Scholar] [CrossRef] [PubMed]
- Bührer, G.; Rottensteiner, U.; Hoppe, A.; Detsch, R.; Dafinova, D.; Fey, T.; Greil, P.; Weis, C.; Beier, J.P.; Boccacini, A.R. Evaluation of in vivo angiogenetic effects of copper doped bioactive glass scaffolds in the av loop model. Biomed. Glasses 2016, 2. [Google Scholar] [CrossRef]
- Urso, E.; Maffia, M. Behind the link between copper and angiogenesis: Established mechanisms and an overview on the role of vascular copper transport systems. J. Vasc. Res. 2015, 52, 172–196. [Google Scholar] [CrossRef] [PubMed]
- Rigiracciolo, D.C.; Scarpelli, A.; Lappano, R.; Pisano, A.; Santolla, M.F.; De Marco, P.; Cirillo, F.; Cappello, A.R.; Dolce, V.; Belfiore, A. Copper activates HIF-1α/GPER/VEGF signalling in cancer cells. Oncotarget 2015, 6, 34158–34177. [Google Scholar] [CrossRef] [PubMed]
- Mavria, G.; Vercoulen, Y.; Yeo, M.; Paterson, H.; Karasarides, M.; Marais, R.; Bird, D.; Marshall, C.J. Erk-mapk signaling opposes rho-kinase to promote endothelial cell survival and sprouting during angiogenesis. Cancer Cell 2006, 9, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Baino, F.; Hamzehlou, S.; Hill, R.G.; Mozafari, M. Bioactive glasses: Sprouting angiogenesis in tissue engineering. Trends Biotechnol. 2018, 36, 430–444. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Zhou, Y.; Fan, W.; Han, P.; Chang, J.; Yuen, J.; Zhang, M.; Xiao, Y. Hypoxia-mimicking mesoporous bioactive glass scaffolds with controllable cobalt ion release for bone tissue engineering. Biomaterials 2012, 33, 2076–2085. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Lotfibakhshaiesh, N.; Ai, J.; Mozafari, M.; Milan, P.B.; Hamzehlou, S.; Barati, M.; Baino, F.; Hill, R.G.; Joghataei, M.T. Strontium-and cobalt-substituted bioactive glasses seeded with human umbilical cord perivascular cells to promote bone regeneration via enhanced osteogenic and angiogenic activities. Acta Biomater. 2017, 58, 502–514. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Xiang, L.; Ou, B.; Huang, T.; Zhou, H.; Zeng, W.; Liu, L.; Liu, Q.; Zhao, Y.; He, S. Biological assessment in-vivo of gel-ha scaffold materials containing nano-bioactive glass for tissue engineering. J. Macromol. Sci. Part A 2014, 51, 572–576. [Google Scholar] [CrossRef]
- Meseguer-Olmo, L.; Ros-Nicolás, M.; Clavel-Sainz, M.; Vicente-Ortega, V.; Alcaraz-Baños, M.; Lax-Pérez, A.; Arcos, D.; Ragel, C.; Vallet-Regí, M. Biocompatibility and in vivo gentamicin release from bioactive sol–gel glass implants. J. Biomed. Mater. Res. Part A 2002, 61, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Lai, W.; Garino, J.; Flaitz, C.; Ducheyne, P. Excretion of resorption products from bioactive glass implanted in rabbit muscle. J. Biomed. Mater. Res. Part A 2005, 75, 398–407. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.W.; Lee, J.-S.; Cho, D.-W. Computer-aided multiple-head 3D printing system for printing of heterogeneous organ/tissue constructs. Sci. Rep. 2016, 6, 21685. [Google Scholar] [CrossRef] [PubMed]
- Liverani, L.; Roether, J.; Nooeaid, P.; Trombetta, M.; Schubert, D.; Boccaccini, A. Simple fabrication technique for multilayered stratified composite scaffolds suitable for interface tissue engineering. Mater. Sci. Eng. A 2012, 557, 54–58. [Google Scholar] [CrossRef]
- Galarraga-Vinueza, M.; Mesquita-Guimarães, J.; Magini, R.; Souza, J.; Fredel, M.; Boccaccini, A. Anti-biofilm properties of bioactive glasses embedding organic active compounds. J. Biomed. Mater. Res. Part A 2017, 105, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.; Thakur, S.; Gupta, A.; Kaur, G.; Pandey, O.P. Antibacterial and anticancerous drug loading kinetics for (10 − x)CuO–xZnO–20CaO–60SiO2–10P2O5 (2 ≤ x ≤ 8) mesoporous bioactive glasses. J. Mater. Sci. Mater. Med. 2016, 28, 11. [Google Scholar] [CrossRef] [PubMed]
- Domingues, Z.; Cortés, M.; Gomes, T.; Diniz, H.; Freitas, C.; Gomes, J.; Faria, A.; Sinisterra, R. Bioactive glass as a drug delivery system of tetracycline and tetracycline associated with β-cyclodextrin. Biomaterials 2004, 25, 327–333. [Google Scholar] [CrossRef]
- Pace, C.N.; Treviño, S.; Prabhakaran, E.; Scholtz, J.M. Protein structure, stability and solubility in water and other solvents. Philos. Trans. R. Soc. B Biol. Sci. 2004, 359, 1225–1235. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.W.; Lee, E.J.; Jun, I.K.; Kim, H.E.; Knowles, J.C. Degradation and drug release of phosphate glass/polycaprolactone biological composites for hard-tissue regeneration. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 75, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Farag, M.; Abd-Allah, W.; Ibrahim, A. Effect of gamma irradiation on drug releasing from nano-bioactive glass. Drug Deliv. Transl. Res. 2015, 5, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J. Mesoporous bioactive glasses: Structure characteristics, drug/growth factor delivery and bone regeneration application. Interface Focus 2012, 2, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Xia, W.; Chang, J. Well-ordered mesoporous bioactive glasses (mbg): A promising bioactive drug delivery system. J. Control. Release 2006, 110, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, X.; Zhang, L.; Chen, H.; Shi, J. MBG/PLGA composite microspheres with prolonged drug release. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 89, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-M.; Wang, W.-K.; Hsiung, P.-A.; Shyu, S.-G. Light-sensitive intelligent drug delivery systems of coumarin-modified mesoporous bioactive glass. Acta Biomater. 2010, 6, 3256–3263. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Lotfibakhshaiesh, N.; Ai, J.; Samadikuchaksaraie, A.; Hill, R.G.; Shah, P.A.; Milan, P.B.; Mozafari, M.; Fathi, M.; Joghataei, M.T. Synthesis, physico-chemical and biological characterization of strontium and cobalt substituted bioactive glasses for bone tissue engineering. J. Non-Cryst. Solids 2016, 449, 133–140. [Google Scholar] [CrossRef]
- Miguez-Pacheco, V.; de Ligny, D.; Schmidt, J.; Detsch, R.; Boccaccini, A. Development and characterization of niobium-releasing silicate bioactive glasses for tissue engineering applications. J. Eur. Ceram. Soc. 2018, 38, 871–876. [Google Scholar] [CrossRef]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef] [PubMed]
- Brückner, R.; Tylkowski, M.; Hupa, L.; Brauer, D.S. Controlling the ion release from mixed alkali bioactive glasses by varying modifier ionic radii and molar volume. J. Mater. Chem. B 2016, 4, 3121–3134. [Google Scholar] [CrossRef]
- Fabert, M.; Ojha, N.; Erasmus, E.; Hannula, M.; Hokka, M.; Hyttinen, J.; Rocherullé, J.; Sigalas, I.; Massera, J. Crystallization and sintering of borosilicate bioactive glasses for application in tissue engineering. J. Mater. Chem. B 2017, 5, 4514–4525. [Google Scholar] [CrossRef]
- Jones, J.R.; Ehrenfried, L.M.; Saravanapavan, P.; Hench, L.L. Controlling ion release from bioactive glass foam scaffolds with antibacterial properties. J. Mater. Sci. Mater. Med. 2006, 17, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chang, J. Multifunctional mesoporous bioactive glasses for effective delivery of therapeutic ions and drug/growth factors. J. Control. Release 2014, 193, 282–295. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Mouriño, V.; Boccaccini, A.R. Therapeutic inorganic ions in bioactive glasses to enhance bone formation and beyond. Biomater. Sci. 2013, 1, 254–256. [Google Scholar] [CrossRef]
- Mijnendonckx, K.; Leys, N.; Mahillon, J.; Silver, S.; Van Houdt, R. Antimicrobial silver: Uses, toxicity and potential for resistance. Biometals 2013, 26, 609–621. [Google Scholar] [CrossRef] [PubMed]
- Sinha, D.; Wang, Z.; Ruchalski, K.L.; Levine, J.S.; Krishnan, S.; Lieberthal, W.; Schwartz, J.H.; Borkan, S.C. Lithium activates the wnt and phosphatidylinositol 3-kinase AKT signaling pathways to promote cell survival in the absence of soluble survival factors. Am. J. Physiol.-Renal Physiol. 2005, 288, F703–F713. [Google Scholar] [CrossRef] [PubMed]
- Satija, N.K.; Sharma, D.; Afrin, F.; Tripathi, R.P.; Gangenahalli, G. High throughput transcriptome profiling of lithium stimulated human mesenchymal stem cells reveals priming towards osteoblastic lineage. PLoS ONE 2013, 8, e55769. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Shi, X.; Liu, S.; Guo, X.; Zhao, M.; Cai, R.; Sun, G. Fluoride promotes osteoblastic differentiation through canonical wnt/β-catenin signaling pathway. Toxicol. Lett. 2014, 225, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Aquino-Martínez, R.; Artigas, N.; Gámez, B.; Rosa, J.L.; Ventura, F. Extracellular calcium promotes bone formation from bone marrow mesenchymal stem cells by amplifying the effects of BMP-2 on SMAD signalling. PLoS ONE 2017, 12, e0178158. [Google Scholar] [CrossRef] [PubMed]
- Munaron, L. Intracellular calcium, endothelial cells and angiogenesis. Recent Pat. Anti-Cancer Drug Discov. 2006, 1, 105–119. [Google Scholar] [CrossRef]
- Yang, F.; Yang, D.; Tu, J.; Zheng, Q.; Cai, L.; Wang, L. Strontium enhances osteogenic differentiation of mesenchymal stem cells and in vivo bone formation by activating wnt/catenin signaling. Stem Cells 2011, 29, 981–991. [Google Scholar] [CrossRef] [PubMed]
- Nardone, V.; Zonefrati, R.; Mavilia, C.; Romagnoli, C.; Ciuffi, S.; Fabbri, S.; Palmini, G.; Galli, G.; Tanini, A.; Brandi, M.L. In vitro effects of strontium on proliferation and osteoinduction of human preadipocytes. Stem Cells Int. 2015, 2015, 871863. [Google Scholar] [CrossRef] [PubMed]
- Hurtel-Lemaire, A.S.; Mentaverri, R.; Caudrillier, A.; Cournarie, F.; Wattel, A.; Kamel, S.; Terwilliger, E.F.; Brown, E.M.; Brazier, M. The calcium-sensing receptor is involved in strontium ranelate-induced osteoclast apoptosis new insights into the associated signaling pathways. J. Biol. Chem. 2009, 284, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Sasaki, T.; Tsouderos, Y.; Suda, T. S 12911-2 inhibits osteoclastic bone resorption in vitro. J. Bone Min. Res. 2003, 18, 1082–1087. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Tian, Y.; Qiao, Y.; Liu, X. Mn-containing titanium surface with favorable osteogenic and antimicrobial functions synthesized by PIII&D. Colloids Surf. B Biointerfaces 2017, 152, 376–384. [Google Scholar] [PubMed]
- Sharma, N.; Jandaik, S.; Kumar, S.; Chitkara, M.; Sandhu, I.S. Synthesis, characterisation and antimicrobial activity of manganese- and iron-doped zinc oxide nanoparticles. J. Exp. Nanosci. 2016, 11, 54–71. [Google Scholar] [CrossRef]
- Díaz-Tocados, J.M.; Herencia, C.; Martínez-Moreno, J.M.; De Oca, A.M.; Rodríguez-Ortiz, M.E.; Vergara, N.; Blanco, A.; Steppan, S.; Almadén, Y.; Rodríguez, M. Magnesium chloride promotes osteogenesis through notch signaling activation and expansion of mesenchymal stem cells. Sci. Rep. 2017, 7, 7839. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, S.; Brown, A.; Barchowsky, A.; Sfeir, C. Magnesium ion stimulation of bone marrow stromal cells enhances osteogenic activity, simulating the effect of magnesium alloy degradation. Acta Biomater. 2014, 10, 2834–2842. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M. Role of nutritional zinc in the prevention of osteoporosis. Mol. Cell. Biochem. 2010, 338, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Pasquet, J.; Chevalier, Y.; Couval, E.; Bouvier, D.; Noizet, G.; Morlière, C.; Bolzinger, M.-A. Antimicrobial activity of zinc oxide particles on five micro-organisms of the challenge tests related to their physicochemical properties. Int. J. Pharm. 2014, 460, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Zinc is an antioxidant and anti-inflammatory agent: Its role in human health. Front. Nutr. 2014, 1, 14. [Google Scholar] [CrossRef] [PubMed]
- Lowe, N.M.; Fraser, W.D.; Jackson, M.J. Is there a potential therapeutic value of copper and zinc for osteoporosis? Proc. Nutr. Soc. 2002, 61, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Borkow, G.; Gabbay, J. Copper as a biocidal tool. Curr. Med. Chem. 2005, 12, 2163–2175. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Kojima, I.; Ohse, T.; Ingelfinger, J.R.; Adler, S.; Fujita, T.; Nangaku, M. Cobalt promotes angiogenesis via hypoxia-inducible factor and protects tubulointerstitium in the remnant kidney model. Lab. Investig. 2005, 85, 1292–1307. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Du, Y.; Jiang, H.; Jiang, G.-S. Cerium promotes bone marrow stromal cells migration and osteogenic differentiation via SMAD1/5/8 signaling pathway. Int. J. Clin. Exp. Pathol. 2014, 7, 5369–5378. [Google Scholar] [PubMed]
- Liu, D.D.; Zhang, J.C.; Zhang, Q.; Wang, S.X.; Yang, M.S. TGF-β/bmp signaling pathway is involved in cerium-promoted osteogenic differentiation of mesenchymal stem cells. J. Cell. Biochem. 2013, 114, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Alpaslan, E.; Geilich, B.M.; Yazici, H.; Webster, T.J. pH-controlled cerium oxide nanoparticle inhibition of both gram-positive and gram-negative bacteria growth. Sci. Rep. 2017, 7, 45859. [Google Scholar] [CrossRef] [PubMed]
- Verron, E.; Bouler, J.; Scimeca, J. Gallium as a potential candidate for treatment of osteoporosis. Drug Discov. Today 2012, 17, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Bonchi, C.; Imperi, F.; Minandri, F.; Visca, P.; Frangipani, E. Repurposing of gallium-based drugs for antibacterial therapy. Biofactors 2014, 40, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Capati, M.L.F.; Nakazono, A.; Igawa, K.; Ookubo, K.; Yamamoto, Y.; Yanagiguchi, K.; Kubo, S.; Yamada, S.; Hayashi, Y. Boron accelerates cultured osteoblastic cell activity through calcium flux. Biol. Trace Element Res. 2016, 174, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Dzondo-Gadet, M.; Mayap-Nzietchueng, R.; Hess, K.; Nabet, P.; Belleville, F.; Dousset, B. Action of boron at the molecular level. Biol. Trace Element Res. 2002, 85, 23–33. [Google Scholar] [CrossRef]
- Wang, Q.; Chen, B.; Cao, M.; Sun, J.; Wu, H.; Zhao, P.; Xing, J.; Yang, Y.; Zhang, X.; Ji, M.; et al. Response of mapk pathway to iron oxide nanoparticles in vitro treatment promotes osteogenic differentiation of hbmscs. Biomaterials 2016, 86, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Patra, C.R.; Bhattacharya, R.; Patra, S.; Vlahakis, N.E.; Gabashvili, A.; Koltypin, Y.; Gedanken, A.; Mukherjee, P.; Mukhopadhyay, D. Pro-angiogenic properties of europium (III) hydroxide nanorods. Adv. Mater. 2008, 20, 753–756. [Google Scholar] [CrossRef]
- Zhai, D.; Xu, M.; Liu, L.; Chang, J.; Wu, C. Silicate-based bioceramics regulating osteoblast differentiation through a BMP2 signalling pathway. J. Mater. Chem. B 2017, 5, 7297–7306. [Google Scholar] [CrossRef]
- Li, H.; Chang, J. Bioactive silicate materials stimulate angiogenesis in fibroblast and endothelial cell co-culture system through paracrine effect. Acta Biomater. 2013, 9, 6981–6991. [Google Scholar] [CrossRef] [PubMed]
- Julien, M.; Khoshniat, S.; Lacreusette, A.; Gatius, M.; Bozec, A.; Wagner, E.F.; Wittrant, Y.; Masson, M.; Weiss, P.; Beck, L. Phosphate-dependent regulation of mgp in osteoblasts: Role of erk1/2 and fra-1. J. Bone Miner. Res. 2009, 24, 1856–1868. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; McKinnon, K.E.; Ha, S.W.; Beck, G.R. Inorganic phosphate induces cancer cell mediated angiogenesis dependent on forkhead box protein c2 (foxc2) regulated osteopontin expression. Mol. Carcinog. 2015, 54, 926–934. [Google Scholar] [CrossRef] [PubMed]
- Coleman, K.; Dai, X.; Deng, X.; Lakehal, F.; Tang, X. Medical device biocompatibility evaluation: An industry perspective a2—Boutrand, jean-pierre. Biocompat. Perform. Med. Devices 2012, 1, 201e–227e. [Google Scholar]
- Thrivikraman, G.; Madras, G.; Basu, B. In vitro/in vivo assessment and mechanisms of toxicity of bioceramic materials and its wear particulates. RSC Adv. 2014, 4, 12763–12781. [Google Scholar] [CrossRef]
- Anderson, J.M. Future challenges in the in vitro and in vivo evaluation of biomaterial biocompatibility. Regen. Biomater. 2016, 3, 73–77. [Google Scholar] [CrossRef] [PubMed]
- El-Rashidy, A.A.; Roether, J.A.; Harhaus, L.; Kneser, U.; Boccaccini, A.R. Regenerating bone with bioactive glass scaffolds: A review of in vivo studies in bone defect models. Acta Biomater. 2017, 62, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Mozafari, M.; Hashemian, S.J.; Brouki Milan, P.; Hamzehlou, S.; Soleimani, M.; Joghataei, M.T.; Gholipourmalekabadi, M.; Korourian, A.; Mousavizadeh, K. Osteogenic potential of stem cells-seeded bioactive nanocomposite scaffolds: A comparative study between human mesenchymal stem cells derived from bone, umbilical cord wharton’s jelly, and adipose tissue. J. Biomed. Mater. Res. Part B Appl. Biomater. 2018, 106, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Kargozar, S.; Hashemian, S.J.; Soleimani, M.; Milan, P.B.; Askari, M.; Khalaj, V.; Samadikuchaksaraie, A.; Hamzehlou, S.; Katebi, A.R.; Latifi, N.; et al. Acceleration of bone regeneration in bioactive glass/gelatin composite scaffolds seeded with bone marrow-derived mesenchymal stem cells over-expressing bone morphogenetic protein-7. Mater. Sci. Eng. C 2017, 75, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Johari, B.; Kadivar, M.; Lak, S.; Gholipourmalekabadi, M.; Urbanska, A.M.; Mozafari, M.; Ahmadzadehzarajabad, M.; Azarnezhad, A.; Afshari, S.; Zargan, J. Osteoblast-seeded bioglass/gelatin nanocomposite: A promising bone substitute in critical-size calvarial defect repair in rat. Int. J. Artif. Organs 2016, 39, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Li, Y.; Zuo, Y.; Li, J.; Ma, S.; Cheng, L. Biocompatibility and osteogenesis of biomimetic nano-hydroxyapatite/polyamide composite scaffolds for bone tissue engineering. Biomaterials 2007, 28, 3338–3348. [Google Scholar] [CrossRef] [PubMed]
- Bessa, P.C.; Casal, M.; Reis, R. Bone morphogenetic proteins in tissue engineering: The road from laboratory to clinic, part II (BMP delivery). J. Tissue Eng. Regen. Med. 2008, 2, 81–96. [Google Scholar] [CrossRef] [PubMed]
- Festing, S.; Wilkinson, R. The ethics of animal research. Talking point on the use of animals in scientific research. EMBO Rep. 2007, 8, 526–530. [Google Scholar] [CrossRef] [PubMed]
- Arinzeh, T.L.; Peter, S.J.; Archambault, M.P.; Van Den Bos, C.; Gordon, S.; Kraus, K.; Smith, A.; Kadiyala, S. Allogeneic mesenchymal stem cells regenerate bone in a critical-sized canine segmental defect. JBJS 2003, 85, 1927–1935. [Google Scholar] [CrossRef]
- Cortez, P.P.; Brito, A.F.; Kapoor, S.; Correia, A.F.; Atayde, L.M.; Dias-Pereira, P.; Maurício, A.C.; Afonso, A.; Goel, A.; Ferreira, J.M. The in vivo performance of an alkali-free bioactive glass for bone grafting, fastos® bg, assessed with an ovine model. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017, 105, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Niemeyer, P.; Fechner, K.; Milz, S.; Richter, W.; Suedkamp, N.P.; Mehlhorn, A.T.; Pearce, S.; Kasten, P. Comparison of mesenchymal stem cells from bone marrow and adipose tissue for bone regeneration in a critical size defect of the sheep tibia and the influence of platelet-rich plasma. Biomaterials 2010, 31, 3572–3579. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L.; Jones, J.R. Bioactive glasses: Frontiers and challenges. Front. Bioeng. Biotechnol. 2015, 3, 194. [Google Scholar] [CrossRef] [PubMed]
- Egermann, M.; Goldhahn, J.; Schneider, E. Animal models for fracture treatment in osteoporosis. Osteoporos. Int. 2005, 16, S129–S138. [Google Scholar] [CrossRef] [PubMed]
- Levy, N. The use of animal as models: Ethical considerations. Int. J. Stroke 2012, 7, 440–442. [Google Scholar] [CrossRef] [PubMed]



| Year (First Experimental Use) | Achievement/Application |
|---|---|
| 1969 | Invention of the 45S5 glass composition (45S5 Bioglass®) |
| 1977 | Treatment of ear diseases by using Ceravital® glass-ceramics (replacement of middle ear small bones) |
| 1978 | Ocular implant (biocompatibility with corneal tissue) |
| 1985 | Approval by Food and Drug Administration (FDA) of the first 45S5 Bioglass® implant (MEP® implant for middle ear ossicular repair) |
| 1987 | Treatment of liver cancer (radioactive glasses) |
| 1988 | Clinical use of the 45S5 Bioglass®-based Endosseous Ridge Maintenance Implant (ERMI) in human patients |
| 1993 | FDA approval of PerioGlas (45S5 Bioglass® particulate used for bone and dental repair) |
| 1998 | Peripheral nerve repair |
| 1999 | FDA approval of radioactive glasses (TheraSphere®) for cancer treatment |
| 2000 | Wound healing |
| 2002 | FDA approval of Medpor®-PlusTM (polyethylene/45S5 Bioglass® composite porous orbital implants). |
| 2003 | Antibacterial (Zn-containing) bone/dental cements |
| 2004 | Lung tissue engineering |
| 2004 | Use of mesoporous bioactive glass (MBG) as a drug delivery system |
| 2005 | Skeletal muscle and ligament repair |
| 2005 | Treatment of gastrointestinal ulcers |
| 2010 | Cardiac tissue engineering |
| 2011 | Commercialization of a cotton-candy borate bioactive glass for wound healing in veterinarian medicine. FDA approval is pending. |
| 2012 | Embolization of uterine fibroids |
| 2012 | Spinal cord repair |
| 2018 | Use of radioactive glasses (TheraSphere®) in patients with metastatic colorectal carcinoma of the liver |
| Therapeutic ions | Biological Effects | Mechanism of Action | References | |
|---|---|---|---|---|
| Monovalent | Silver (Ag) | Antibacterial activity |
| [119] |
| Lithium (Li) | Osteogenesis |
| [120,121] | |
| Fluoride (F) | Osteogenesis |
| [122] | |
| Divalent | Calcium (Ca) | Osteogenesis |
| [123] |
| Angiogenesis activity |
| [124] | ||
| Strontium (Sr) | Osteogenesis |
| [125,126,127,128] | |
| Manganese (Mn) | Osteogenesis |
| [129] | |
| Antibacterial activity |
| [130] | ||
| Magnesium (Mg) | Osteogenesis |
| [131] | |
| Angiogenesis activity |
| [132] | ||
| Zinc (Zn) | Osteogenesis |
| [133] | |
| Antibacterial activity |
| [134] | ||
| Anti-inflammation activity |
| [135] | ||
| Copper (Cu) | Osteogenesis |
| [136] | |
| Angiogenesis |
| [91] | ||
| Antibacterial activity |
| [137] | ||
| Cobalt (Co) | Angiogenesis |
| [95,138] | |
| Trivalent | Cerium (Ce) | Osteogenesis |
| [139,140] |
| Antibacterial activity |
| [141] | ||
| Gallium (Ga) | Osteogenesis |
| [142] | |
| Antibacterial activity |
| [143] | ||
| Boron (B) | Osteogenesis |
| [144] | |
| Angiogenesis |
| [145] | ||
| Iron (Fe) | Osteogenesis |
| [146] | |
| Europium (Eu) | Angiogenesis |
| [147] | |
| Tetravalent | Silicon (Si) | Osteogenesis |
| [148] |
| Angiogenesis activity |
| [149] | ||
| Pentavalent | Phosphate (P) | Osteogenesis |
| [150] |
| Angiogenesis activity |
| [151] | ||
| Niobium (Nb) | Angiogenesis |
| [112] | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baino, F.; Hamzehlou, S.; Kargozar, S. Bioactive Glasses: Where Are We and Where Are We Going? J. Funct. Biomater. 2018, 9, 25. https://doi.org/10.3390/jfb9010025
Baino F, Hamzehlou S, Kargozar S. Bioactive Glasses: Where Are We and Where Are We Going? Journal of Functional Biomaterials. 2018; 9(1):25. https://doi.org/10.3390/jfb9010025
Chicago/Turabian StyleBaino, Francesco, Sepideh Hamzehlou, and Saeid Kargozar. 2018. "Bioactive Glasses: Where Are We and Where Are We Going?" Journal of Functional Biomaterials 9, no. 1: 25. https://doi.org/10.3390/jfb9010025
APA StyleBaino, F., Hamzehlou, S., & Kargozar, S. (2018). Bioactive Glasses: Where Are We and Where Are We Going? Journal of Functional Biomaterials, 9(1), 25. https://doi.org/10.3390/jfb9010025






