Treatment of Silk Fibroin with Poly(ethylene glycol) for the Enhancement of Corneal Epithelial Cell Growth
Abstract
:1. Introduction
2. Results and Discussion
2.1. Background
| Aspect | Reference [56] | This report |
|---|---|---|
| Cells | Primary rabbit CLECs | Primary human CLECs; SV40-immortalized HCECs |
| Feeder cells | Always present in cultures | Growth of primary cultures compared in the presence and absence of feeder cells |
| Maximum duration of cultures | 7 days | 12 days |
| Substrata | BMSF films coated onto porous cell culture membranes | Freestanding BMSF membranes |
| Control substrata | Non-treated BMSF film; AM | Non-treated BMSF membrane; TCP |
| Ratio PEG/BMSF (by wt.) | 0 to 38 (assessed); 2 (recommended) | 2 |
| Mol. wt. of molecules assessed for permeability | 0.376 to 15 kDa | 26–28 kDa |
| Modification of membranes | No | Yes (by chemical crosslinking) |
| In vivo evaluation | Yes (animals) | No |
2.2. Characterization of Silk Fibroin Membranes

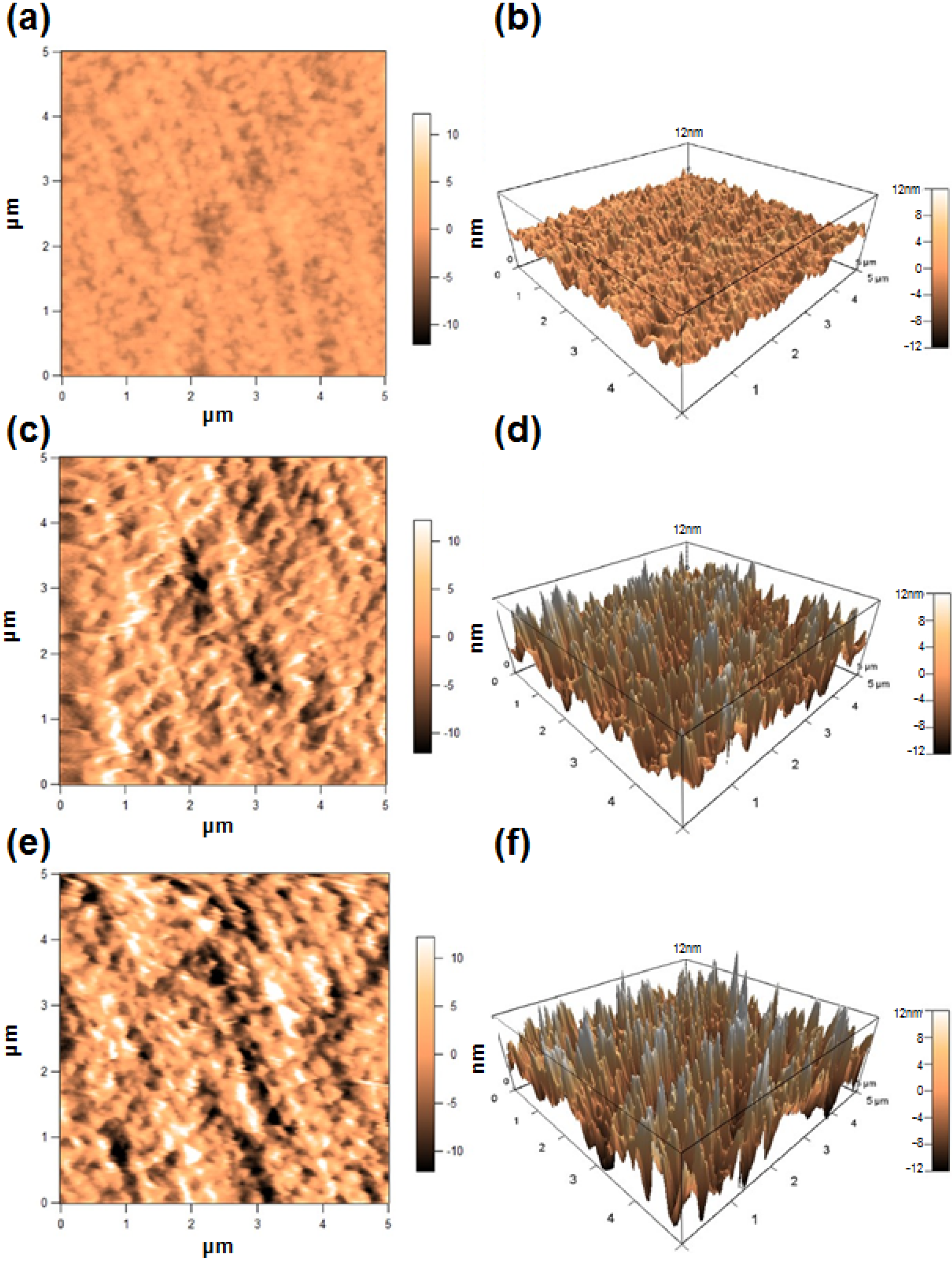
| Fibroin membrane | Ra (nm) |
|---|---|
| Non-treated | 1.3 |
| PEG-treated | 4.4 |
| Genipin-crosslinked PEG-treated | 5.9 |
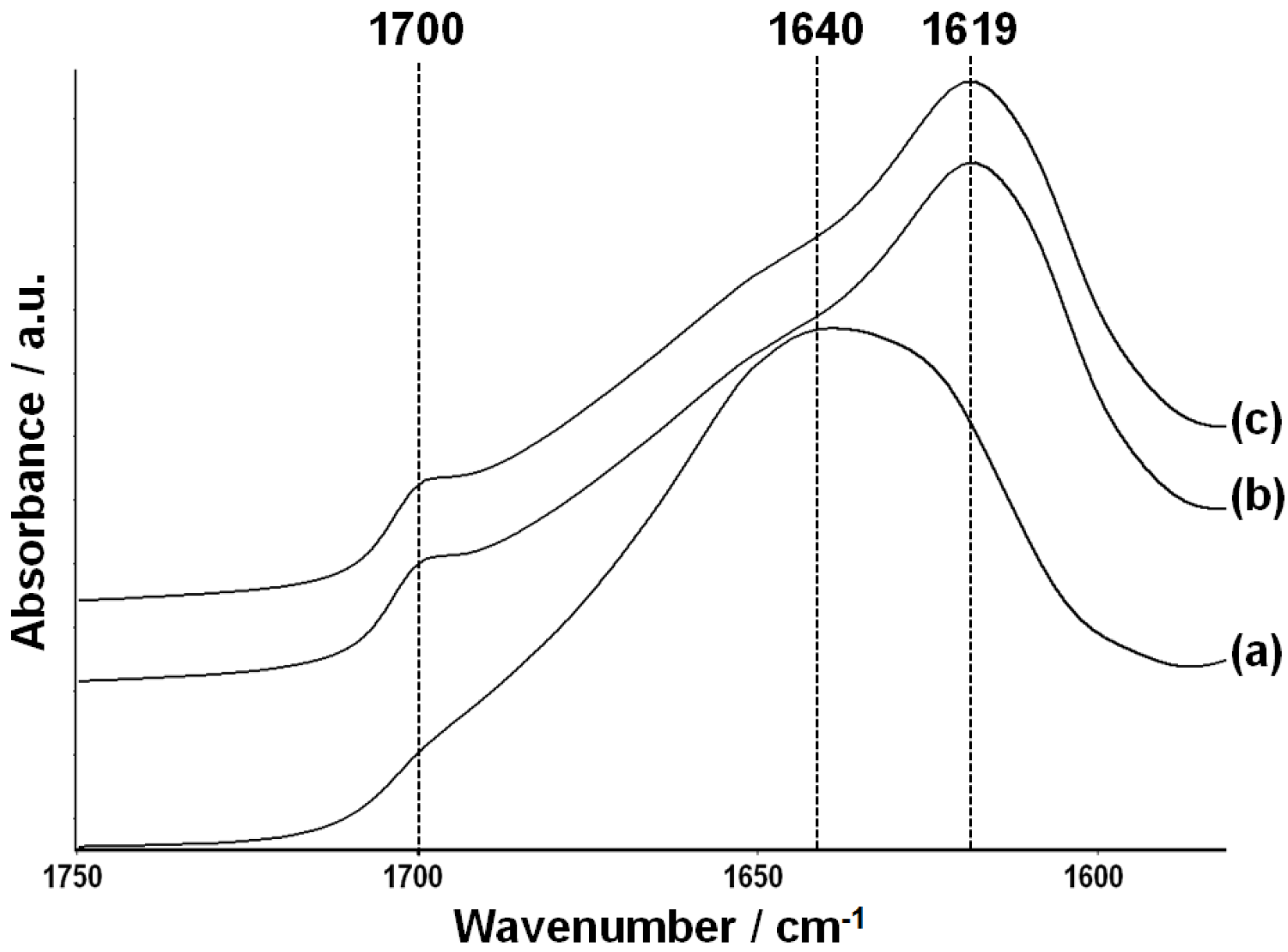
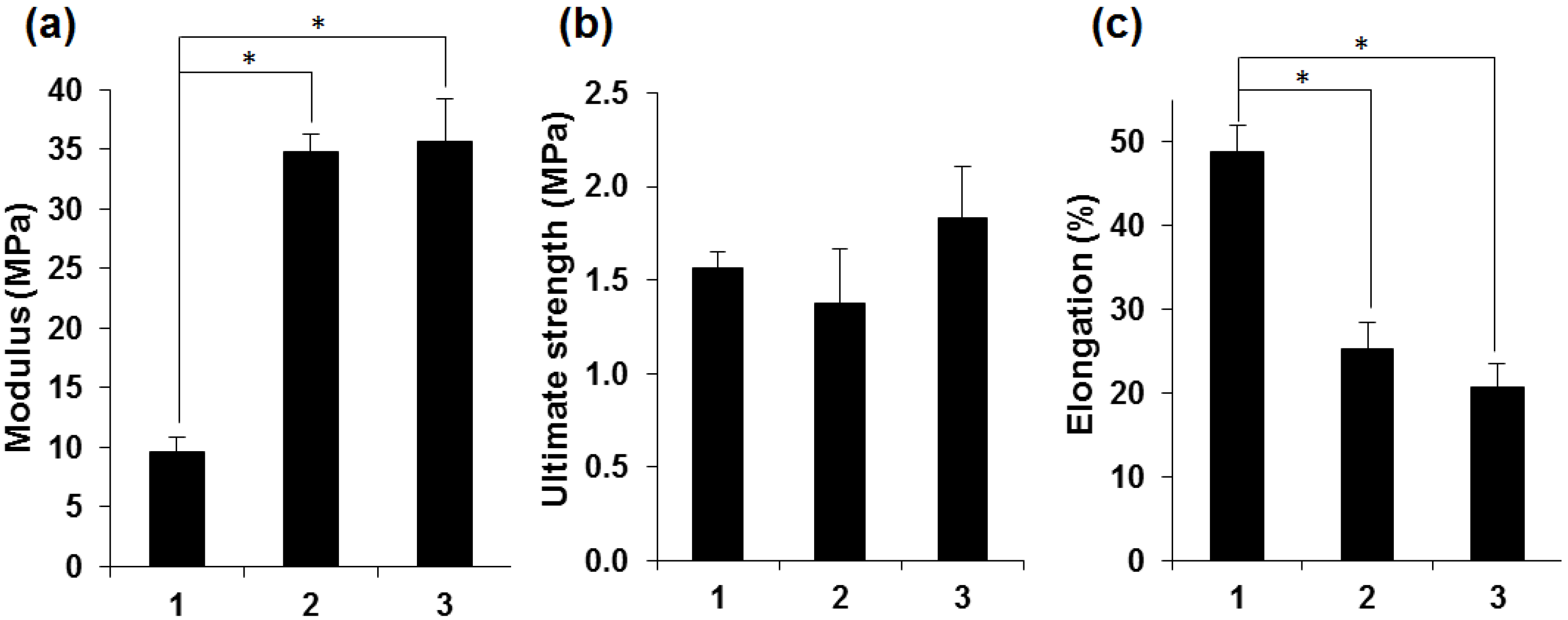
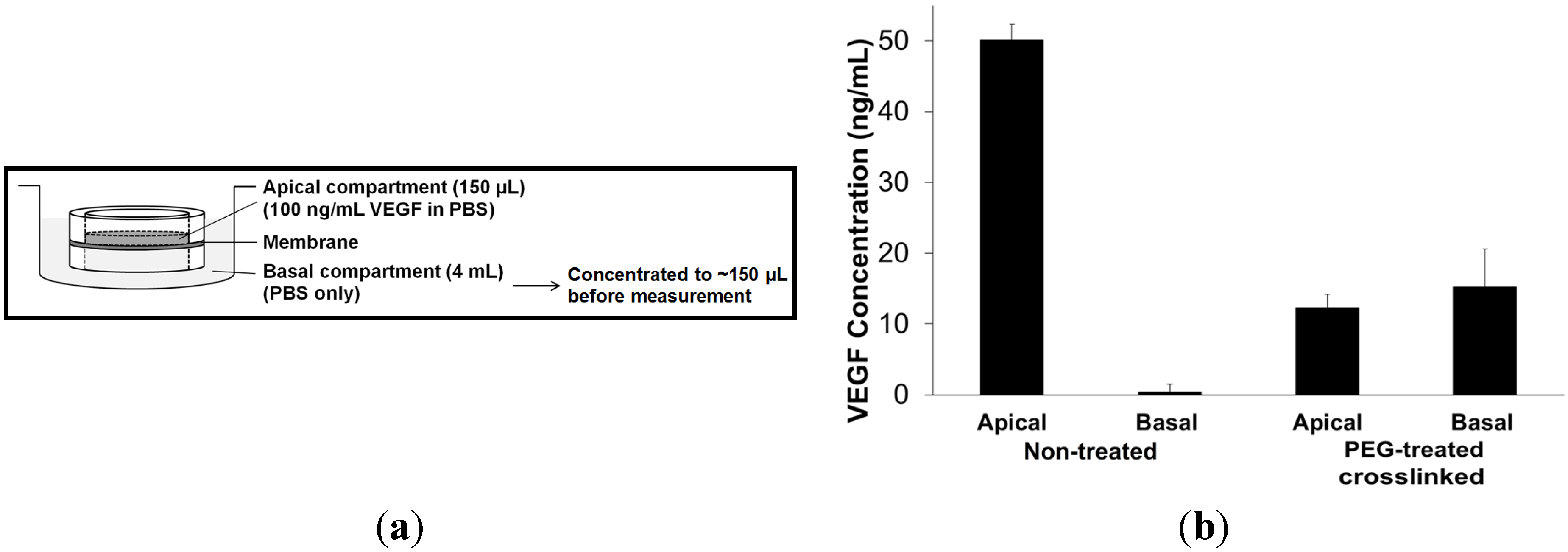
2.3. Attachment and Proliferation of HCE-T Cell Line
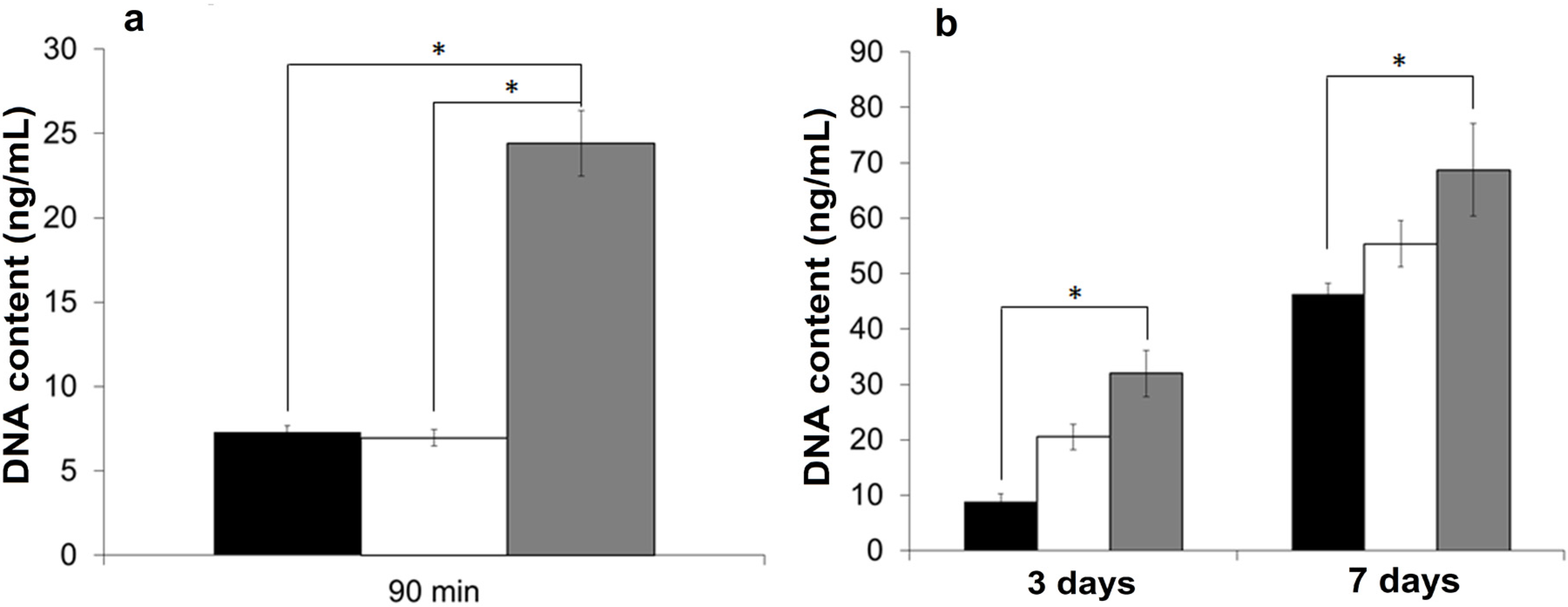
2.4. Growth of Primary Human Corneal Limbal Epithelial Cells (CLECs)

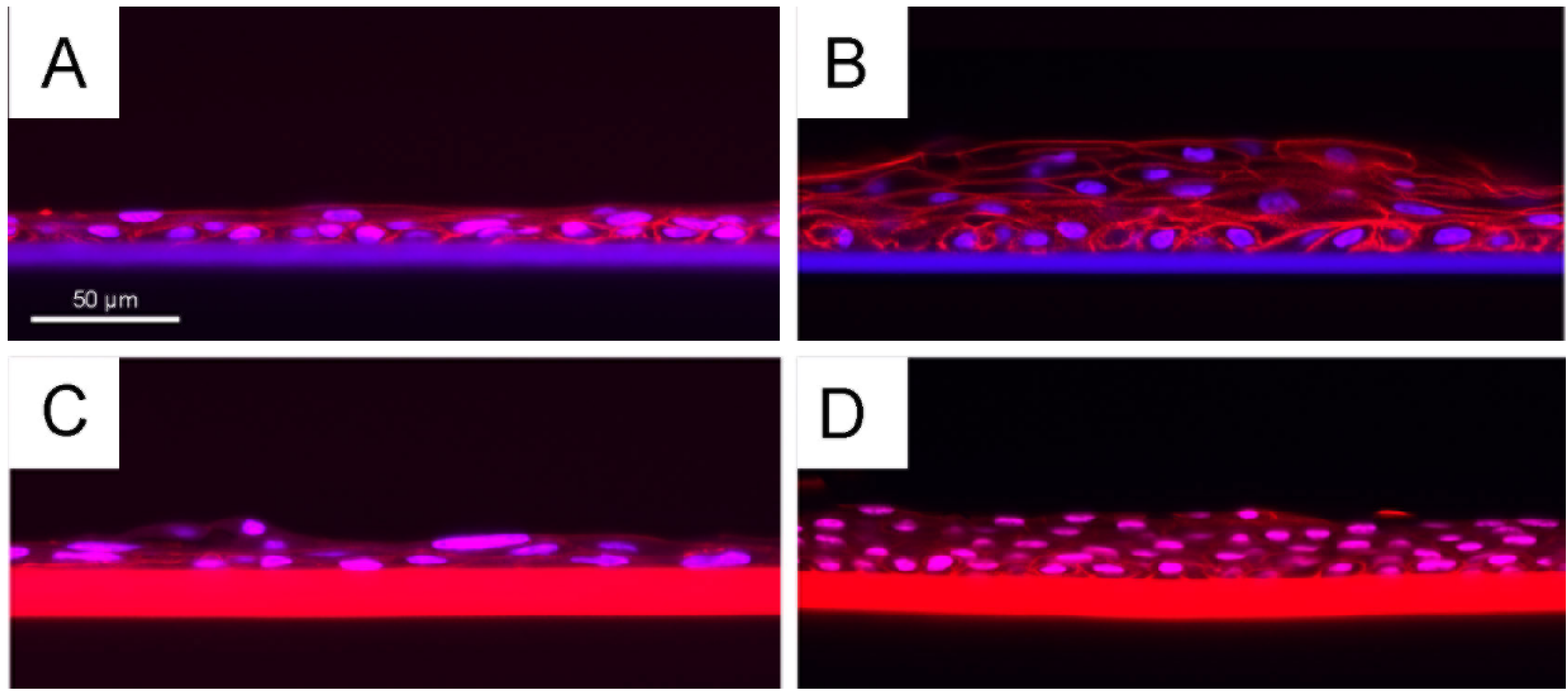
2.5. Summary
3. Experimental Section
3.1. Materials
3.2. Preparation of Fibroin Membranes
3.3. Scanning Electron Microscopy (SEM)
3.4. Atomic Force Microscopy (AFM)
3.5. Fourier-Transform Infrared Spectroscopy (FTIR)
3.6. Tensile Testing
3.7. Permeability of the Membranes
3.8. Culture and Growth of Transformed Human CECs on BMSF Substrata
3.9. Culture and Growth of Primary Human CLECs on BMSF Substrata
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gervers, V. Studies in Textile History; Royal Ontario Museum: Toronto, Canada, 1977. [Google Scholar]
- Barber, E.J.W. Prehistoric Textiles; Princeton University Press: Princeton, NJ, USA, 1991. [Google Scholar]
- Good, I.L.; Kenoyer, J.M.; Meadow, R.H. New evidence for early silk in the Indus Civilization. Archaeometry 2009, 51, 457–466. [Google Scholar] [CrossRef]
- Hansen, V. The Silk Road: A New History; Oxford University Press: New York, NY, USA, 2012; pp. 2–23. [Google Scholar]
- Mackenzie, D. The history of sutures. Med. Hist. 1973, 17, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Muffly, T.M.; Tizzano, A.P.; Walters, M.D. The history and evolution of sutures in pelvic surgery. J. R. Soc. Med. 2011, 104, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Edgar, I.I. Modern surgery and Lord Lister. J. Hist. Med. Allied Sci. 1961, 16, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Kiliani, O.G.T. On traumatic keloid of the median nerve, with observations upon the absorption of silk sutures. Ann. Surg. 1901, 33, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Halsted, W.S. Ligature and suture material. The employment of fine silk in preference to catgut and the advantages of transfixion of tissues and vessels in control of hemorrhage. J. Am. Med. Assoc. 1913, 60, 1119–1126. [Google Scholar] [CrossRef]
- Smit, I.B.; Witte, E.; Brand, R.; Trimbos, J.B. Tissue reaction to suture materials revisited: Is there argument to change our views? Eur. Surg. Res. 1991, 23, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Deveikis, J.P.; Manz, H.J.; Luessenhop, A.J.; Caputy, A.J.; Kobrine, A.I.; Schellinger, D.; Patronas, N. A clinical and neuropathologic study of silk suture as an embolic agent for brain arteriovenous malformations. Am. J. Neuroradiol. 1994, 15, 263–271. [Google Scholar] [PubMed]
- Williams, H.W. Recent Advances in Ophthalmic Science; Ticknor & Fields: Boston, MA, USA, 1866; pp. 90–92. [Google Scholar]
- Kuhnt, H. Beiträge zur operativen Augenheilkunde; Verlag Gustav Fischer: Jena, Germany, 1883; pp. 69–97. [Google Scholar]
- McLean, J.M. A new corneoscleral suture. Arch. Ophthalmol. 1940, 23, 554–559. [Google Scholar] [CrossRef]
- Hughes, W.L.; Guy, L.P.; Romaine, H.H. Use of absorbable sutures in cataract surgery. Arch. Ophthalmol. 1944, 32, 362–367. [Google Scholar] [CrossRef]
- Larmi, T. Sutures in eye surgery. Acta Ophthalmol. 1961, 39(S63), 15–19. [Google Scholar]
- Salthouse, T.N.; Matlaga, B.F.; Wykoff, M.H. Comparative tissue response to six suture materials in rabbit cornea, sclera, and ocular muscle. Am. J. Ophthalmol. 1977, 84, 224–233. [Google Scholar] [CrossRef]
- Minoura, N.; Tsukada, M.; Nagura, M. Physico-chemical properties of silk fibroin membrane as a biomaterial. Biomaterials 1990, 11, 430–434. [Google Scholar] [CrossRef]
- Minoura, N.; Aiba, S.; Higuchi, M.; Gotoh, Y.; Tsukada, M.; Imai, Y. Attachment and growth of fibroblast cells on silk fibroin. Biochem. Biophys. Res. Commun. 1995, 208, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Minoura, N.; Aiba, S.; Gotoh, Y.; Tsukada, M.; Imai, Y. Attachment and growth of cultured fibroblast cells on silk protein matrices. J. Biomed. Mater. Res. 1995, 29, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Altman, G.H.; Diaz, F.; Jakuba, C.; Calabro, T.; Horan, R.L.; Chen, J.; Lu, H.; Richmond, J.; Kaplan, D.L. Silk-based biomaterials. Biomaterials 2003, 24, 401–416. [Google Scholar] [CrossRef]
- Wang, Y.; Kim, H.-J.; Vunjak-Novakovic, G.; Kaplan, D.L. Stem cell-based tissue engineering with silk biomaterials. Biomaterials 2006, 27, 6064–6082. [Google Scholar] [CrossRef] [PubMed]
- Vepari, C.; Kaplan, D.L. Silk as a biomaterial. Prog. Polym. Sci. 2007, 32, 991–1007. [Google Scholar] [CrossRef] [PubMed]
- Hakimi, O.; Knight, D.P.; Vollrath, F.; Vadgama, P. Spider and mulberry silkworm silks as compatible biomaterials. Composites B 2007, 38, 324–337. [Google Scholar] [CrossRef]
- Kundu, S.C.; Dash, B.C.; Dash, R.; Kaplan, D.L. Natural protective glue protein, sericin bioengineered by silkworms: potential for biomedical and biotechnological applications. Prog. Polym. Sci. 2008, 33, 998–1012. [Google Scholar] [CrossRef]
- Wang, X.; Cebe, P.; Kaplan, D.L. Silk proteins—Biomaterials and bioengineering. In Protein Engineering Handbook; Lutz, S., Bornscheuer, U.T., Eds.; Wiley-VCH Verlag: Weinheim, Germany, 2009; pp. 939–959. [Google Scholar]
- Murphy, A.R.; Kaplan, D.L. Biomedical applications of chemically-modified silk fibroin. J. Mater. Chem. 2009, 19, 6443–6450. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.G.; Scheibel, T.R. Composite materials based on silk proteins. Prog. Polym. Sci. 2010, 35, 1093–1115. [Google Scholar] [CrossRef]
- Wenk, E.; Merkle, H.P.; Meinel, L. Silk fibroin as a vehicle for drug delivery applications. J. Control. Rel. 2011, 150, 128–141. [Google Scholar] [CrossRef] [PubMed]
- Kasoju, N.; Bora, U. Silk fibroin in tissue engineering. Adv. Healthc. Mater. 2012, 1, 393–412. [Google Scholar] [CrossRef] [PubMed]
- Gil, E.S.; Panilaitis, B.; Bellas, E.; Kaplan, D.L. Functionalized silk biomaterials for wound healing. Adv. Healthc. Mater. 2013, 2, 206–217. [Google Scholar] [CrossRef] [PubMed]
- Kundu, B.; Rajkhowa, R.; Kundu, S.C.; Wang, X. Silk fibroin biomaterials for tissue regenerations. Adv. Drug Deliv. Rev. 2013, 65, 457–470. [Google Scholar] [CrossRef] [PubMed]
- Pereira, R.F.P.; Silva, M.M.; de Zea Bermudez, V. Bombyx mori silk fibers: An outstanding family of materials. Macromol. Mater. Eng. 2014. [Google Scholar] [CrossRef]
- Chirila, T.; Barnard, Z.; Zainuddin; Harkin, D. Silk as substratum for cell attachment and proliferation. Mater. Sci. Forum 2007, 561–565, 1549–1552. [Google Scholar] [CrossRef]
- Chirila, T.V.; Barnard, Z.; Zainuddin; Harkin, D.G.; Schwab, I.R.; Hirst, L.W. Bombyx mori silk fibroin membranes as potential substrata for epithelial constructs used in the management of ocular surface disorders. Tissue Eng. Part A 2008, 14, 1203–1211. [Google Scholar] [PubMed]
- Chirila, T.V.; Hirst, L.W.; Barnard, Z.; Harkin, D.G.; Schwab, I.R. Reconstruction of the ocular surface using biomaterials. In Biomaterials and Regenerative Medicine in Ophthalmology; Chirila, T., Ed.; Woodhead Publishing Ltd.: Cambridge, UK, 2010; pp. 213–242. [Google Scholar]
- Kwan, A.S.L.; Chirila, T.V.; Cheng, S. Development of tissue-engineered membranes for the culture and transplantation of retinal pigment epithelial cells. In Biomaterials and Regenerative Medicine in Ophthalmology; Chirila, T., Ed.; Woodhead Publishing Ltd.: Cambridge, UK, 2010; pp. 390–408. [Google Scholar]
- Harkin, D.G.; George, K.A.; Madden, P.W.; Schwab, I.R.; Hutmacher, D.W.; Chirila, T.V. Silk fibroin in ocular tissue reconstruction. Biomaterials 2011, 32, 2445–2458. [Google Scholar] [CrossRef] [PubMed]
- Harkin, D.G.; Chirila, T.V. Silk fibroin in ocular surface reconstruction: What is its potential as a biomaterial in ophthalmics? Future Med. Chem. 2012, 4, 2145–2147. [Google Scholar] [CrossRef] [PubMed]
- Chirila, T.V.; Suzuki, S.; Bray, L.J.; Barnett, N.L.; Harkin, D.G. Evaluation of silk sericin as a biomaterial: in vitro growth of human corneal limbal epithelial cells on Bombyx mori sericin membranes. Prog. Biomater. 2013, 2. [Google Scholar] [CrossRef]
- Bray, L.J.; Suzuki, S.; Harkin, D.G.; Chirila, T.V. Incorporation of exogenous RGD peptide and inter-species blending as strategies for enhancing human corneal limbal epithelial cell growth on bombyx mori silk fibroin membrane. J. Funct. Biomater. 2013, 4, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Hogerheyde, T.A.; Suzuki, S.; Stephenson, S.A.; Richardson, N.A.; Chirila, T.V.; Harkin, D.G.; Bray, L.J. Assessment of freestanding membranes prepared from Antheraea pernyi silk fibroin as a potential vehicle for corneal epithelial cell transplantation. Biomed. Mater. 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Bray, L.J.; George, G.A.; Ainscough, S.L.; Hutmacher, D.W.; Chirila, T.V.; Harkin, D.G. Human corneal epithelial equivalents constructed on Bombyx mori silk fibroin membranes. Biomaterials 2011, 32, 5086–5091. [Google Scholar] [CrossRef] [PubMed]
- Bray, L.J.; George, K.A.; Hutmacher, D.W.; Chirila, T.V.; Harkin, D.G. A dual-layer silk fibroin scaffold for reconstructing the human corneal limbus. Biomaterials 2012, 33, 3529–3538. [Google Scholar] [CrossRef] [PubMed]
- Bray, L.J.; George, K.A.; Suzuki, S.; Chirila, T.V.; Harkin, D.G. Fabrication of a corneal-limbal tissue substitute using silk fibroin. In Corneal Regenerative Medicine. Methods and Protocols; Wright, B., Connon, C.J., Eds.; Humana Press: New York, NY, USA, 2013; pp. 165–178. [Google Scholar]
- Madden, P.W.; Lai, J.N.X.; George, K.A.; Giovenco, T.; Harkin, D.G.; Chirila, T.V. Human corneal endothelial cell growth on a silk fibroin membrane. Biomaterials 2011, 32, 4076–4084. [Google Scholar] [CrossRef] [PubMed]
- Shadforth, A.M.; George, K.A.; Kwan, A.S.; Chirila, T.V.; Harkin, D.G. The cultivation of human retinal pigment epithelial cells on Bombyx mori silk fibroin. Biomaterials 2012, 33, 4110–4117. [Google Scholar] [CrossRef] [PubMed]
- Asakura, T.; Demura, M.; Tsutsumi, M. 23Na and 27Al NMR studies of the interaction between Bombyx mori silk fibroin and metal ions trapped in the porous silk fibroin membrane. Makromol. Chem. Rapid Commun. 1988, 9, 835–839. [Google Scholar] [CrossRef]
- Demura, M.; Asakura, T. Porous membrane of Bombyx mori silk fibroin: structure characterization, physical properties and application to glucose oxidase immobilization. J. Membr. Sci. 1991, 59, 39–52. [Google Scholar] [CrossRef]
- Kuga, S. Pore size distribution analysis of gel substances by size exclusion chromatography. J. Chromatog. 1981, 206, 449–461. [Google Scholar] [CrossRef]
- Jin, H.-J.; Fridrich, S.V.; Rutledge, G.C.; Kaplan, D.L. Electrospinning Bombyx mori silk with poly(ethylene oxide). Biomacromolecules 2002, 3, 1233–1239. [Google Scholar]
- Jin, H.-J.; Park, J.; Valluzzi, R.; Cebe, P.; Kaplan, D.L. Biomaterial films of Bombyx mori silk fibroin with poly(ethylene oxide). Biomacromolecules 2004, 5, 711–717. [Google Scholar]
- Lawrence, B.D.; Omenetto, F.; Chui, K.; Kaplan, D.L. Processing methods to control silk fibroin film biomaterial features. J. Mater. Sci. 2008, 6967–6985. [Google Scholar] [CrossRef]
- Jin, H.-J.; Kaplan, D.L. Mechanism of silk processing in insect and spiders. Nature 2003, 1057–1061. [Google Scholar]
- Lawrence, B.D.; Marchant, J.K.; Pindrus, M.A.; Omenetto, F.G.; Kaplan, D.L. Silk film biomaterials for cornea tissue engineering. Biomaterials 2009, 30, 1299–1308. [Google Scholar] [CrossRef] [PubMed]
- Higa, K.; Takeshima, N.; Moro, F.; Kawakita, T.; Kawashima, M.; Demura, M.; Shimazaki, J.; Asakura, T.; Tsubota, K.; Shimmura, S. Porous silk fibroin film as a transparent carrier for cultivated corneal epithelial sheets. J. Biomater. Sci. Polym. Edn. 2010, 22, 2261–2276. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, B.D.; Pan, Z.; Liu, A.; Kaplan, D.L.; Rosenblatt, M.I. Human corneal limbal epithelial cell response to varying silk film geometric topography in vitro. Acta Biomater. 2012, 8, 3732–3743. [Google Scholar] [CrossRef] [PubMed]
- Harley, B.A.; Yannas, I.V. In vivo synthesis of tissues and organs. In Principles of Tissue Engineering, 3rd ed.; Lanza, R., Langer, R., Vacanti, J., Eds.; Elsevier Academic Press: Burlington, MA, USA, 2007; pp. 219–238. [Google Scholar]
- Williams, D. Essential Biomaterials Science; Cambridge University Press: Cambridge, UK, 2014; pp. 340–355. [Google Scholar]
- Fitton, J.H.; Dalton, B.A.; Beumer, G.; Johnson, G.; Griesser, H.J.; Steele, J.G. Surface topography can interfere with epithelial tissue migration. J. Biomed. Mater. Res. 1998, 42, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Evans, M.D.; Dalton, B.A.; Steele, J.G. Persistent adhesion of epithelial tissue is sensitive to polymer topography. J. Biomed. Mater. Res. 1999, 46, 485–493. [Google Scholar] [CrossRef]
- Steele, J.G.; Johnson, G.; McLean, K.M.; Beumer, G.; Griesser, H.J. Effect of porosity and surface hydrophilicity on migration of epithelial tissue over synthetic polymer. J. Biomed. Mater. Res. 2000, 50, 475–482. [Google Scholar] [CrossRef]
- Teixeira, A.I.; Abrams, G.A.; Bertics, P.J.; Murphy, C.J.; Nealey, P.F. Epithelial contact guidances on well-defined micro- and nanostructured substrates. J. Cell Sci. 2003, 116, 1881–1892. [Google Scholar] [CrossRef] [PubMed]
- Karuri, N.W.; Liliensiek, S.; Teixeira, A.I.; Abrams, G.; Campbell, S.; Nealey, P.F.; Murphy, C.J. Biological length scale topography enhances cell-substratum adhesion of human corneal epithelial cells. J. Cell Sci. 2004, 117, 3153–3164. [Google Scholar] [CrossRef] [PubMed]
- Diehl, K.A.; Foley, J.D.; Nealey, P.F.; Murphy, C.J. Nanoscale topography modulates corneal epithelial cell migration. J. Biomed. Mater. Res. A 2005, 75, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Karuri, N.W.; Porri, T.J.; Albrecht, R.M.; Murphy, C.J.; Nealey, P.F. Nano- and microscale holes modulate cell-substrate adhesion, cytoskeletal organization, and -β1 integrin localization in Sv40 human corneal epithelial cells. IEEE Trans. Nanobiosci. 2006, 5, 273–280. [Google Scholar] [CrossRef]
- Liliensiek, S.J.; Campbell, S.; Nealey, P.F.; Murphy, C.J. The scale of substratum topographic features modulates proliferation of corneal epithelial cells and corneal fibroblasts. J. Biomed. Mater. Res. A 2006, 79, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Mo, X.; Huang, C.; He, C.; Wang, H. Electrospun scaffolds from silk fibroin and their cellular compatibility. J. Biomed. Mater. Res. A 2010, 93, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Kaplan, D.; Cebe, P. Determining beta-sheet crystallinity in fibrous proteins by thermal analysis and infrared spectroscopy. Macromolecules 2006, 39, 6161–6170. [Google Scholar] [CrossRef]
- Hu, X.; Shmelev, K.; Sun, L.; Gil, E.-S.; Park, S.-H.; Cebe, P.; Kaplan, D.L. Regulation of silk material structure by temperature-controlled water vapour annealing. Biomacromolecules 2011, 12, 1686–1696. [Google Scholar] [CrossRef] [PubMed]
- Leal-Egaña, A.; Scheibel, T. Interactions of cells with silk surfaces. J. Mater. Chem. 2012, 22, 14330–14336. [Google Scholar] [CrossRef]
- Djerassi, C.; Gray, J.D.; Kincl, F.A. Naturally occurring oxygen heterocycles. IX. Isolation and characterization of genipin. J. Org. Chem. 1960, 25, 2174–2177. [Google Scholar] [CrossRef]
- Dawson, R.A.; Upton, Z.; Malda, J.; Harkin, D.G. Preparation of cultured skin for transplantation using insulin-like growth factor I in conjunction with insulin-like growth factor binding protein 5, epidermal growth factor, and vitronectin. Transplantation 2006, 81, 1668–1676. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzuki, S.; Dawson, R.A.; Chirila, T.V.; Shadforth, A.M.A.; Hogerheyde, T.A.; Edwards, G.A.; Harkin, D.G. Treatment of Silk Fibroin with Poly(ethylene glycol) for the Enhancement of Corneal Epithelial Cell Growth. J. Funct. Biomater. 2015, 6, 345-366. https://doi.org/10.3390/jfb6020345
Suzuki S, Dawson RA, Chirila TV, Shadforth AMA, Hogerheyde TA, Edwards GA, Harkin DG. Treatment of Silk Fibroin with Poly(ethylene glycol) for the Enhancement of Corneal Epithelial Cell Growth. Journal of Functional Biomaterials. 2015; 6(2):345-366. https://doi.org/10.3390/jfb6020345
Chicago/Turabian StyleSuzuki, Shuko, Rebecca A. Dawson, Traian V. Chirila, Audra M. A. Shadforth, Thomas A. Hogerheyde, Grant A. Edwards, and Damien G. Harkin. 2015. "Treatment of Silk Fibroin with Poly(ethylene glycol) for the Enhancement of Corneal Epithelial Cell Growth" Journal of Functional Biomaterials 6, no. 2: 345-366. https://doi.org/10.3390/jfb6020345
APA StyleSuzuki, S., Dawson, R. A., Chirila, T. V., Shadforth, A. M. A., Hogerheyde, T. A., Edwards, G. A., & Harkin, D. G. (2015). Treatment of Silk Fibroin with Poly(ethylene glycol) for the Enhancement of Corneal Epithelial Cell Growth. Journal of Functional Biomaterials, 6(2), 345-366. https://doi.org/10.3390/jfb6020345





