Antimicrobial PVA Hydrogels with Tunable Mechanical Properties and Antimicrobial Release Profiles
Abstract
1. Introduction
2. Methods
2.1. Materials
2.2. Synthesis
2.2.1. Chemically Incorporated PA-PVA Films
2.2.2. Physically Incorporated PA-PVA Films
2.3. Characterization
2.3.1. Gel Fraction
2.3.2. Swelling
2.3.3. Physically Incorporated PA Content
2.3.4. Mechanical Properties
2.3.5. Release Rates
2.4. Cell Viability and Growth
2.5. Antimicrobial Properties
2.6. Biofilm Formation
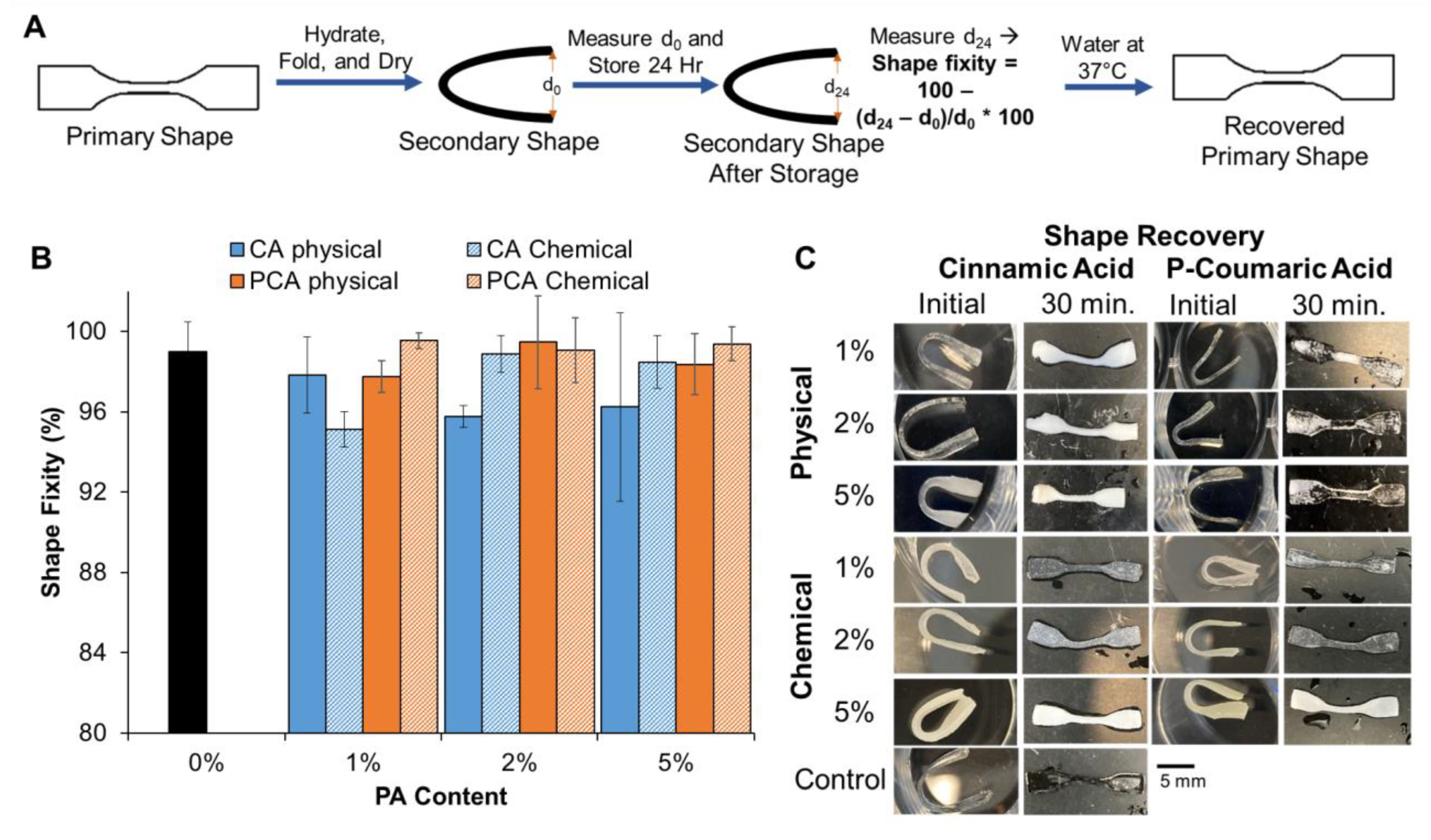
2.7. Shape Memory Properties
2.8. Statistical Analysis
3. Results
3.1. Characterization
3.1.1. Gel Fraction and Swelling Ratio
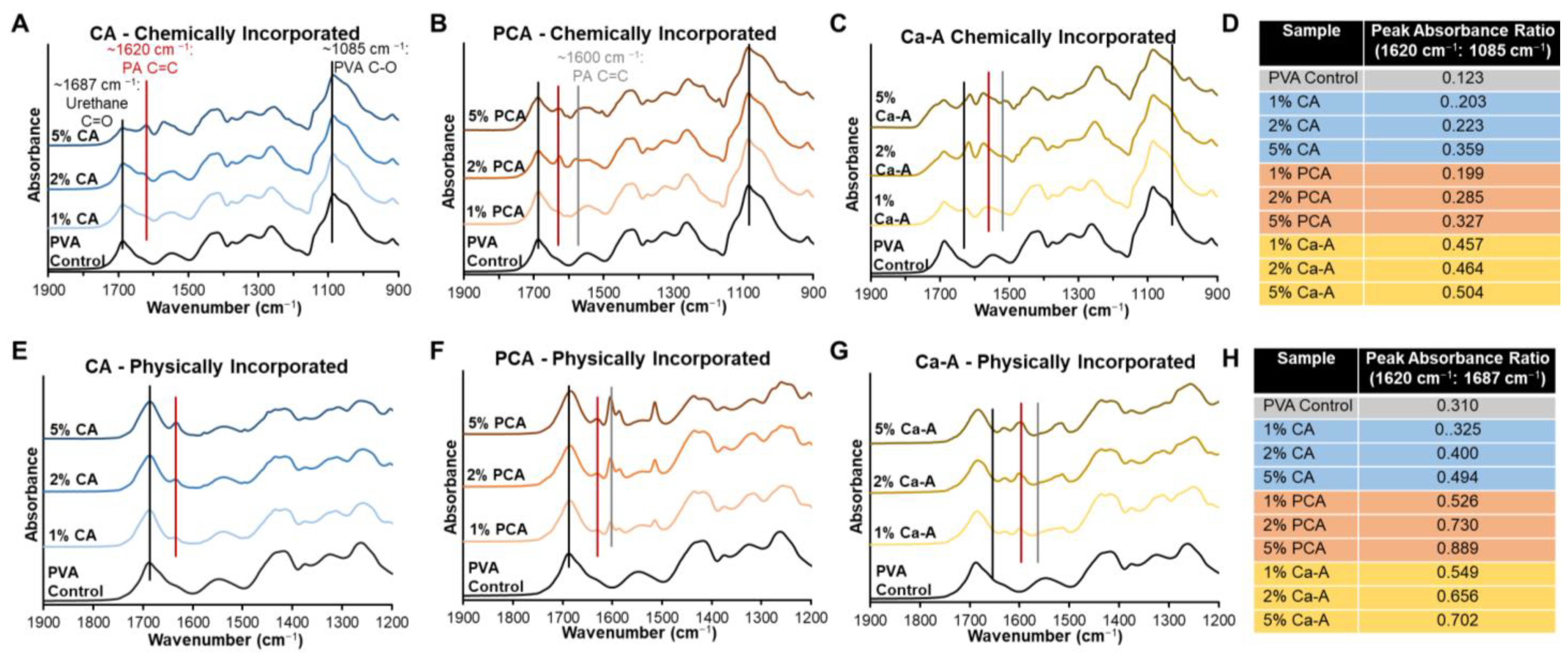
3.1.2. Surface Chemistry
3.1.3. Physically Incorporated PA Content
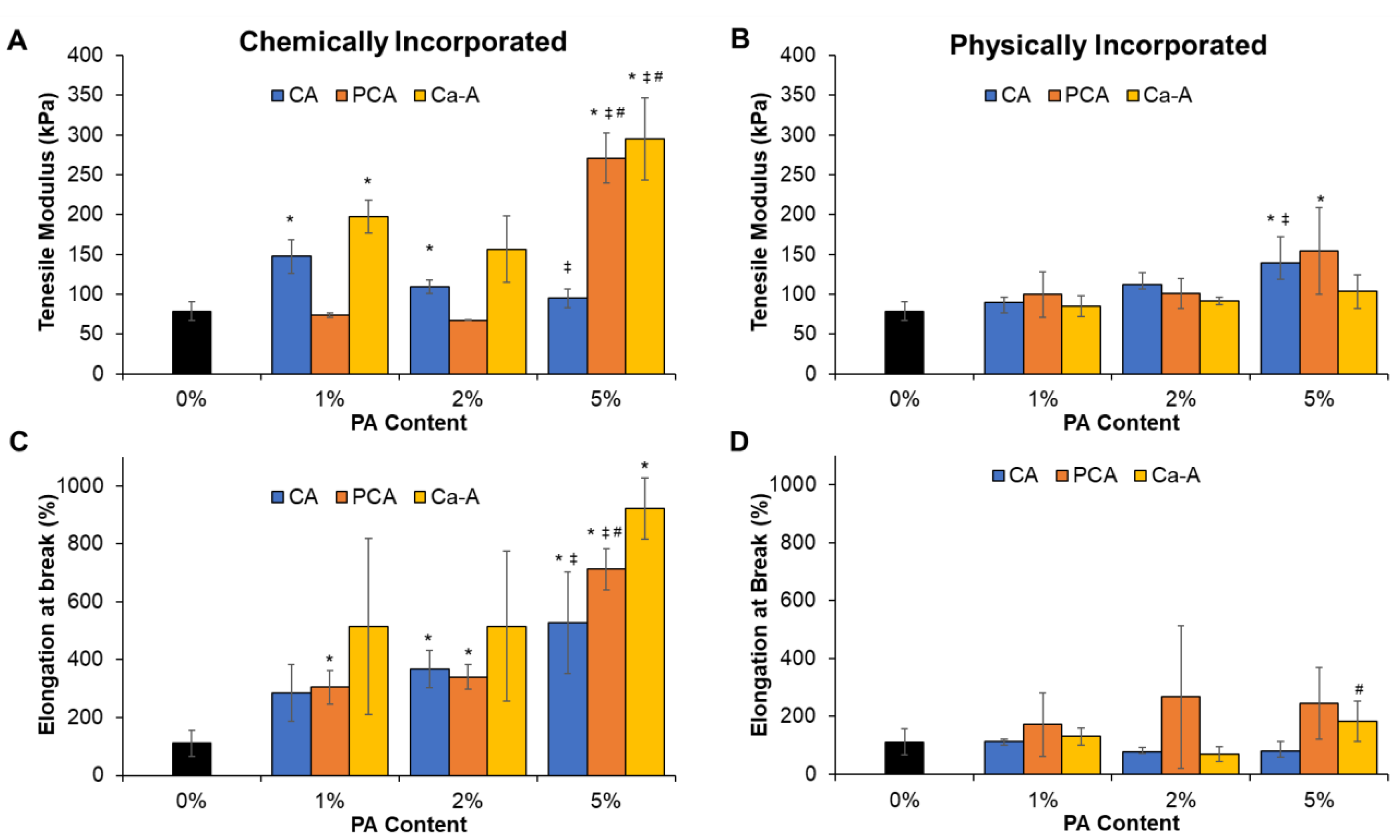
3.1.4. Mechanical Properties
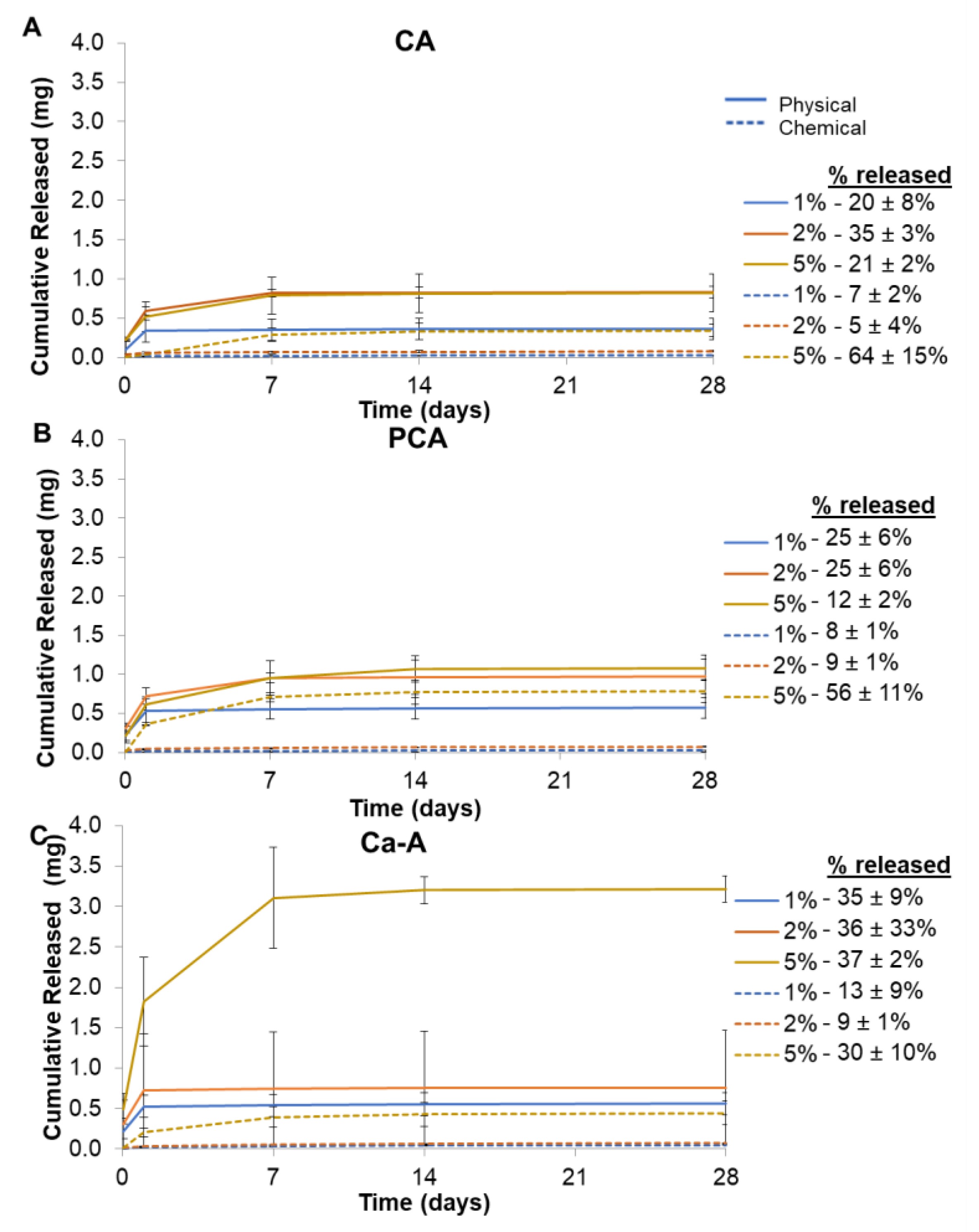
3.1.5. PA Release Rates
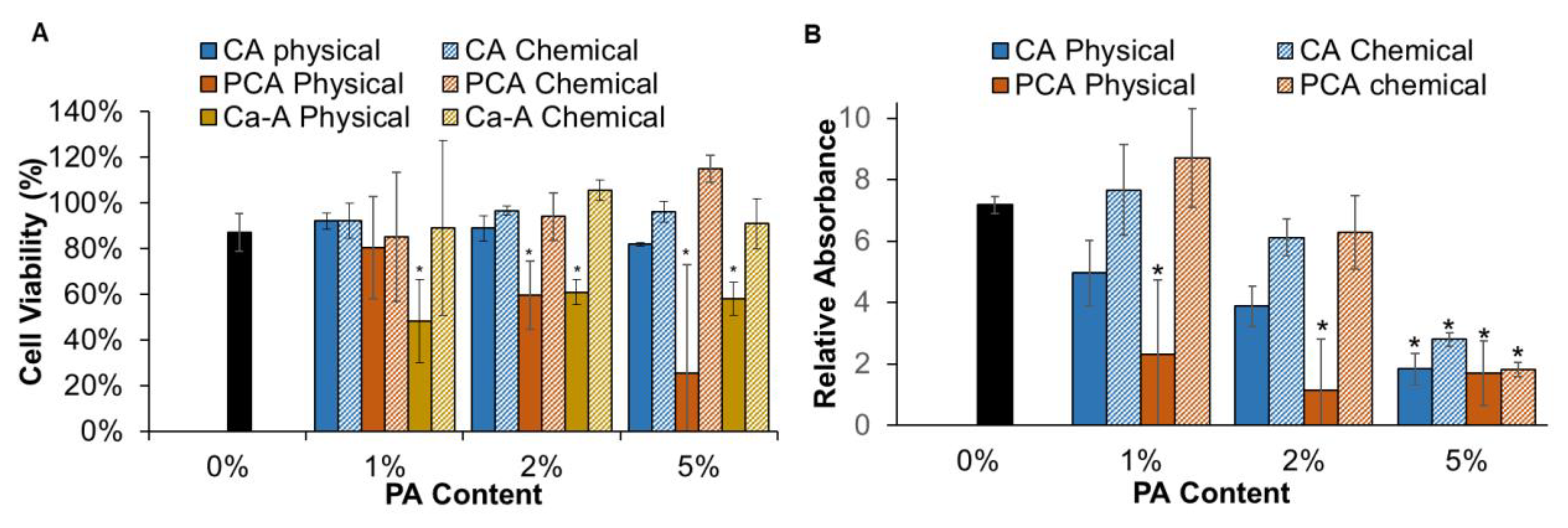
3.2. Cell Viability
3.3. Antimicrobial Properties
3.3.1. Colony Forming Units (CFUs) of E. coli (1 h Incubation)
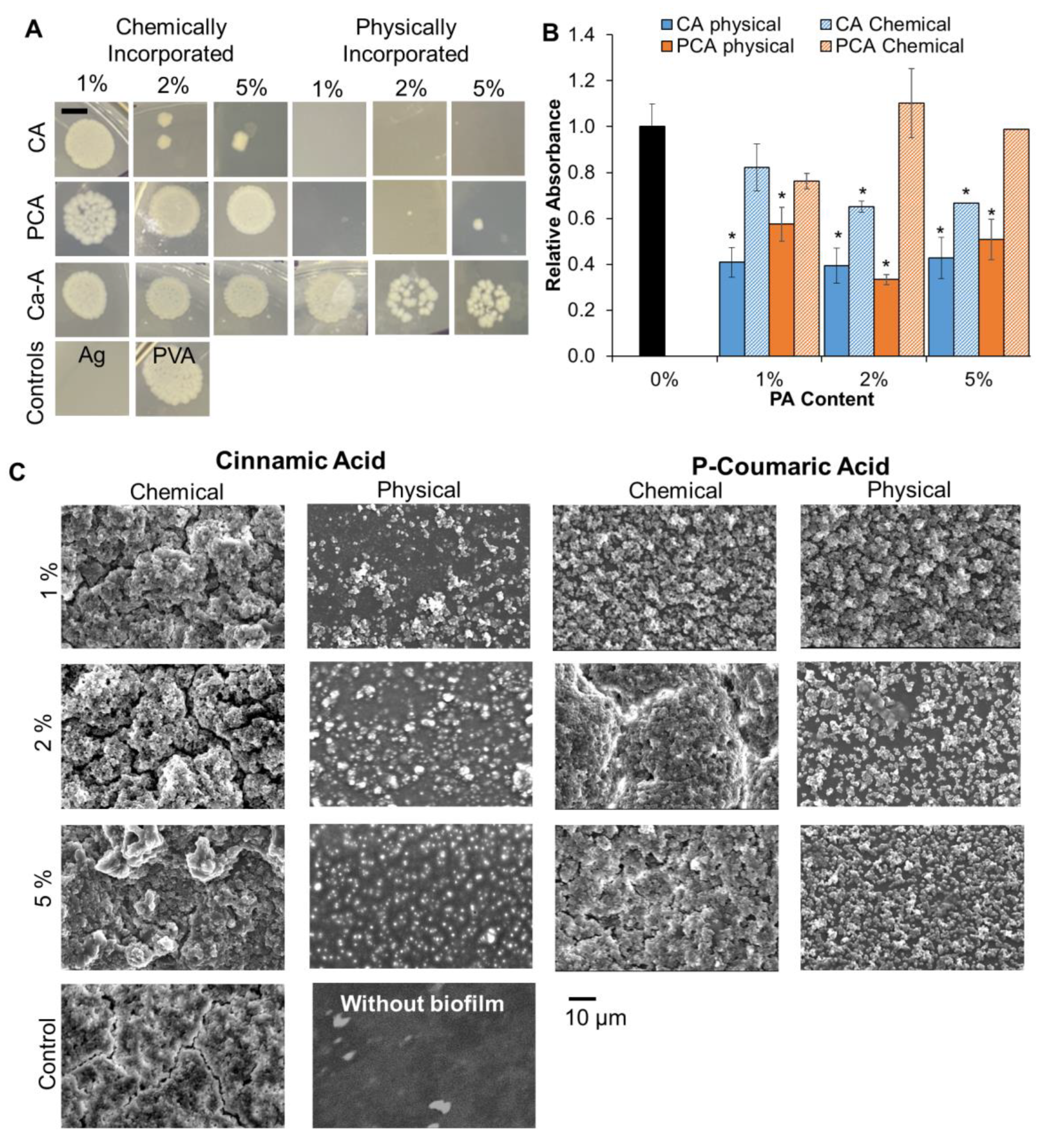
3.3.2. Staph. aureus Biofilm Formation (24-h Incubation)
3.4. Shape Memory Properties
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Loftus, E.V., Jr. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology 2004, 126, 1504–1517. [Google Scholar] [CrossRef] [PubMed]
- Frolkis, A.D.; Dykeman, J.; Negrón, M.E.; Debruyn, J.; Jette, N.; Fiest, K.M.; Frolkis, T.; Barkema, H.; Rioux, K.P.; Panaccione, R.; et al. Risk of Surgery for Inflammatory Bowel Diseases Has Decreased Over Time: A Systematic Review and Meta-analysis of Population-Based Studies. Gastroenterology 2013, 145, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Present, D.H. Crohn’s fistula: Current concepts in management. Gastroenterology 2003, 124, 1629–1635. [Google Scholar] [CrossRef] [PubMed]
- Altomare, N.F.; Greco, V.J.; Tricomi, N.; Arcanà, F.; Mancini, S.; Rinaldi, M.; D’urso, A.P.; La Torre, F. Seton or glue for trans-sphincteric anal fistulae: A prospective randomized crossover clinical trial. Color. Dis. 2011, 13, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Loungnarath, R.; Dietz, D.W.; Mutch, M.G.; Birnbaum, E.H.; Kodner, I.J.; Fleshman, J.W. Fibrin Glue Treatment of Complex Anal Fistulas Has Low Success Rate. Dis. Colon Rectum 2004, 47, 432–436. [Google Scholar] [CrossRef]
- Maralcan, G.; Başkonuş, I.; Gökalp, A.; Borazan, E.; Balk, A. Long-term results in the treatment of fistula-in-ano with fibrin glue: A prospective study. J. Korean Surg. Soc. 2011, 81, 169–175. [Google Scholar] [CrossRef]
- Bernell, O.; Lapidus, A.; Hellers, G. Risk Factors for Surgery and Postoperative Recurrence in Crohn’s Disease. Ann. Surg. 2000, 231, 38–45. [Google Scholar] [CrossRef]
- Mirsepasi-Lauridsen, H.C.; Vallance, B.A.; Krogfelt, K.A.; Petersen, A.M. Escherichia coli Pathobionts Associated with Inflammatory Bowel Disease. Clin. Microbiol. Rev. 2019, 32, e00060-18. [Google Scholar] [CrossRef]
- Sandborn, W.J.; Fazio, V.W.; Feagan, B.G.; Hanauer, S.B. AGA technical review on perianal Crohn’s disease. Gastroenterology 2003, 125, 1508–1530. [Google Scholar] [CrossRef]
- Keshaw, H.; Foong, K.S.; Forbes, A.; Day, R.M. Perianal fistulae in Crohn’s Disease: Current and future approaches to treatment. Inflamm. Bowel Dis. 2010, 16, 870–880. [Google Scholar] [CrossRef]
- Brandt, L.J.; Bernstein, L.H.; Boley, S.J.; Frank, M.S. Metronidazole Therapy for Perineal Crohn’s Disease: A Follow-Up Study. Gastroenterology 1982, 83, 383–387. [Google Scholar] [CrossRef]
- Poggioli, G.; Laureti, S.; Pierangeli, F.; Rizzello, F.; Ugolini, F.; Gionchetti, P.; Campieri, M. Local Injection of Infliximab for the Treatment of Perianal Crohn’s Disease. Dis. Colon Rectum 2005, 48, 768–774. [Google Scholar] [CrossRef]
- Wahdan, H.A.L. Causes of the antimicrobial activity of honey. Infection 1998, 26, 26–31. [Google Scholar] [CrossRef]
- Merkl, R.; Hrádková, I.; Filip, V.; Šmidrkal, J. Antimicrobial and antioxidant properties of phenolic acids alkyl esters. Czech J. Food Sci. 2010, 28, 275–279. [Google Scholar] [CrossRef]
- Nascimento, G.G.F.; Locatelli, J.; Freitas, P.C.; Silva, G.L. Antibacterial activity of plant extracts and phytochemicals on antibiotic-resistant bacteria. Braz. J. Microbiol. 2000, 31, 247–256. [Google Scholar] [CrossRef]
- Liu, J.; Du, C.; Beaman, H.T.; Monroe, M.B.B. Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships. Pharmaceutics 2020, 12, 419. [Google Scholar] [CrossRef]
- Scharl, M.; Rogler, G. Pathophysiology of fistula formation in Crohn’s disease. World J. Gastrointest. Pathophysiol. 2014, 5, 205–212. [Google Scholar] [CrossRef]
- Wei, M.-G.; Sun, W.; He, W.-M.; Ni, L.; Yang, Y.-Y. Ferulic Acid Attenuates TGF-β1-Induced Renal Cellular Fibrosis in NRK-52E Cells by Inhibiting Smad/ILK/Snail Pathway. Evidence-Based Complement. Altern. Med. 2015, 2015, 1–7. [Google Scholar] [CrossRef]
- Gowda, S.S.; Rajasowmiya, S.; Vadivel, V.; Devi, S.B.; Jerald, A.C.; Marimuthu, S.; Devipriya, N. Gallic acid-coated sliver nanoparticle alters the expression of radiation-induced epithelial-mesenchymal transition in non-small lung cancer cells. Toxicol. Vitr. 2018, 52, 170–177. [Google Scholar] [CrossRef]
- Beaman, H.T.; Howes, B.; Ganesh, P.; Monroe, M.B.B. Shape memory polymer hydrogels with cell-responsive degradation mechanisms for Crohn’s fistula closure. J. Biomed. Mater. Res. Part A 2022, 110, 1329–1340. [Google Scholar] [CrossRef]
- Huang, W.M.; Yang, B.; An, L.; Li, C.; Chan, Y.S. Water-driven programmable polyurethane shape memory polymer: Demonstration and mechanism. Appl. Phys. Lett. 2005, 86, 114105. [Google Scholar] [CrossRef]
- Baer, G.M.; SmallIV, W.; Wilson, T.S.; Benett, W.J.; Matthews, D.L.; Hartman, J.; Maitland, D.J. Fabrication and in vitro deployment of a laser-activated shape memory polymer vascular stent. Biomed. Eng. Online 2007, 6, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Gordon, R.F. The Properties and Applications of Shape Memory Polyurethanes. Mater. Technol. 1993, 8, 254–258. [Google Scholar] [CrossRef]
- Hearon, K.; Wierzbicki, M.A.; Nash, L.D.; Landsman, T.L.; Laramy, C.; Lonnecker, A.T.; Gibbons, M.C.; Ur, S.; Cardinal, K.O.; Wilson, T.; et al. A Processable Shape Memory Polymer System for Biomedical Applications. Adv. Health Mater. 2015, 4, 1386–1398. [Google Scholar] [CrossRef]
- Lv, Y.; Chen, X.; Zhang, P.; Zhou, S.; Li, J.; Bai, R.; Wang, L. Hydrophilic shape memory polymer hydrogels with virous pore structures and shape changing performance. Polym. Adv. Technol. 2022, 33, 3532–3539. [Google Scholar] [CrossRef]
- Xiang, T.; Wang, J.; Jia, L.; Wang, P.; Zhou, S. Semicrystalline polymer networks with a swelling-enhanced water-triggered two-way shape-memory effect for programmable deformation and smart actuation. Polym. Chem. 2022, 13, 6614–6624. [Google Scholar] [CrossRef]
- Wu, S.; Shao, Z.; Xie, H.; Xiang, T.; Zhou, S. Salt-mediated triple shape-memory ionic conductive polyampholyte hydrogel for wearable flexible electronics. J. Mater. Chem. A 2021, 9, 1048–1061. [Google Scholar] [CrossRef]
- Du, C.; Liu, J.; Fikhman, D.A.; Dong, K.S.; Monroe, M.B.B. Shape Memory Polymer Foams With Phenolic Acid-Based Antioxidant and Antimicrobial Properties for Traumatic Wound Healing. Front. Bioeng. Biotechnol. 2022, 10, 809361. [Google Scholar] [CrossRef]
- Monroe, M.B.B.; Easley, A.D.; Grant, K.; Fletcher, G.K.; Boyer, C.; Maitland, D.J. Multifunctional Shape-Memory Polymer Foams with Bio-inspired Antimicrobials. ChemPhysChem 2017, 19, 1999–2008. [Google Scholar] [CrossRef]
- Herigstad, B.; Hamilton, M.; Heersink, J. How to optimize the drop plate method for enumerating bacteria. J. Microbiol. Methods 2001, 44, 121–129. [Google Scholar] [CrossRef]
- ISO 10993-5; 2009 Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity. International Organization for Standardization: Geneve, Switzerland, 2009.
- Cane, G.; Ginouvès, A.; Marchetti, S.; Buscà, R.; Pouysségur, J.; Berra, E.; Hofman, P.; Vouret-Craviari, V. HIF-1α mediates the induction of IL-8 and VEGF expression on infection with Afa/Dr diffusely adhering E. coli and promotes EMT-like behaviour. Cell. Microbiol. 2010, 12, 640–653. [Google Scholar] [CrossRef]
- West, R.L.; Van Der Woude, C.J.; Endtz, H.P.; Hansen, B.E.; Ouwedijk, M.; Boelens, H.A.M.; Kusters, J.G.; Kuipers, E.J. Perianal Fistulas in Crohn’s Disease Are Predominantly Colonized by Skin Flora: Implications for Antibiotic Treatment? Dig. Dis. Sci. 2005, 50, 1260–1263. [Google Scholar] [CrossRef]
- Antonino, L.D.; Gouveia, J.R.; Júnior, R.R.d.S.; Garcia, G.E.S.; Gobbo, L.C.; Tavares, L.B.; dos Santos, D.J. Reactivity of Aliphatic and Phenolic Hydroxyl Groups in Kraft Lignin towards 4,4′ MDI. Molecules 2021, 26, 2131. [Google Scholar] [CrossRef]
- Sharmin, E.; Zafar, F. Polyurethane: An Introduction. In Polyurethane, 1st ed.; Zafar, F., Sharmin, E., Eds.; InTechOpen: Rijeka, Croatia, 2012; pp. 3–16. [Google Scholar]
- Sonnenschein, M. Polyurethanes: Science, Technology, Markets, and Trends, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2021. [Google Scholar]
- Guzman, J.D. Natural Cinnamic Acids, Synthetic Derivatives and Hybrids with Antimicrobial Activity. Molecules 2014, 19, 19292–19349. [Google Scholar] [CrossRef]
- Chen, Y.-N.; Peng, L.; Liu, T.; Wang, Y.; Shi, S.; Wang, H. Poly(vinyl alcohol)–Tannic Acid Hydrogels with Excellent Mechanical Properties and Shape Memory Behaviors. ACS Appl. Mater. Interfaces 2016, 8, 27199–27206. [Google Scholar] [CrossRef]
- Sakai, S.; Khanmohammadi, M.; Khoshfetrat, A.B.; Taya, M. Horseradish peroxidase-catalyzed formation of hydrogels from chitosan and poly(vinyl alcohol) derivatives both possessing phenolic hydroxyl groups. Carbohydr. Polym. 2014, 111, 404–409. [Google Scholar] [CrossRef]
- Skelton, S.; Bostwick, M.; O’Connor, K.; Konst, S.; Casey, S.; Lee, B.P. Biomimetic adhesive containing nanocomposite hydrogel with enhanced materials properties. Soft Matter 2013, 9, 3825–3833. [Google Scholar] [CrossRef]
- Le, H.H.; Tran, V.T.; Mredha, T.I.; Na, J.Y.; Seon, J.-K.; Jeon, I. Thin-film hydrogels with superior stiffness, strength, and stretchability. Extrem. Mech. Lett. 2020, 37, 100720. [Google Scholar] [CrossRef]
- Tran, V.T.; Mredha, T.I.; Jeon, I. High-water-content hydrogels exhibiting superior stiffness, strength, and toughness. Extrem. Mech. Lett. 2020, 37, 100691. [Google Scholar] [CrossRef]
- Contardi, M.; Alfaro-Pulido, A.; Picone, P.; Guzman-Puyol, S.; Goldoni, L.; Benítez, J.J.; Heredia, A.; Barthel, M.J.; Ceseracciu, L.; Cusimano, G.; et al. Low molecular weight ε-caprolactone-p-coumaric acid copolymers as potential biomaterials for skin regeneration applications. PLoS ONE 2019, 14, e0214956. [Google Scholar] [CrossRef]
- Kumar, A.; Han, S.S. PVA-based hydrogels for tissue engineering: A review. Int. J. Polym. Mater. Polym. Biomater. 2016, 66, 159–182. [Google Scholar] [CrossRef]
- Davis, N.; Mulvihill, J.; Mulay, S.; Cunnane, E.; Bolton, D.; Walsh, M. Urinary Bladder vs Gastrointestinal Tissue: A Comparative Study of Their Biomechanical Properties for Urinary Tract Reconstruction. Urology 2018, 113, 235–240. [Google Scholar] [CrossRef]
- Pailler-Mattei, C.; Bec, S.; Zahouani, H. In vivo measurements of the elastic mechanical properties of human skin by indentation tests. Med Eng. Phys. 2008, 30, 599–606. [Google Scholar] [CrossRef]
- Watters, A.D.; Smith, A.N.; Eastwood, A.M.; Anderson, K.C.; Elton, A.R.; Mugerwa, J.W. Mechanical properties of the colon: Comparison of the features of the African and European colon in vitro. Gut 1985, 26, 384–392. [Google Scholar] [CrossRef]
- Glavind, E.B.; Forman, A.; Madsen, G.; Svane, D.; Andersson, K.E.; Tottrup, A. Mechanical properties of isolated smooth muscle from human rectum and internal anal sphincter. Am. J. Physiol. Liver Physiol. 1993, 265, G792–G798. [Google Scholar] [CrossRef]
- Manschot, J.; Brakkee, A. The measurement and modelling of the mechanical properties of human skin in vivo—I. Meas. J. Biomech. 1986, 19, 511–515. [Google Scholar] [CrossRef]
- Velada, J.L.; Hollingsbee, D.A.; Menzies, A.R.; Cornwell, R.; Dodd, R.A. Reproducibility of the mechanical properties of Vivostat® system patient-derived fibrin sealant. Biomaterials 2002, 23, 2249–2254. [Google Scholar] [CrossRef]
- Cirocchi, R.; Farinella, E.; La Mura, F.; Cattorini, L.; Rossetti, B.; Milani, D.; Ricci, P.; Covarelli, P.; Coccetta, M.; Noya, G.; et al. Fibrin glue in the treatment of anal fistula: A systematic review. Ann. Surg. Innov. Res. 2009, 3, 12. [Google Scholar] [CrossRef]
- CDRH. Use of International Standard ISO 10993-1, “Biological Evaluation of Medical Devices—Part 1: Evaluation and Testing within a Risk Management Process”. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/use-international-standard-iso-10993-1-biological-evaluation-medical-devices-part-1-evaluation-and (accessed on 3 February 2023).
- Manna, C.; Napoli, D.; Cacciapuoti, G.; Porcelli, M.; Zappia, V. Olive Oil Phenolic Compounds Inhibit Homocysteine-Induced Endothelial Cell Adhesion Regardless of Their Different Antioxidant Activity. J. Agric. Food Chem. 2009, 57, 3478–3482. [Google Scholar] [CrossRef]
- Rezaei-Seresht, H.; Cheshomi, H.; Falanji, F.; Movahedi-Motlagh, F.; Hashemian, M.; Mireskandari, E. Cytotoxic activity of caffeic acid and gallic acid against MCF-7 human breast cancer cells: An in silico and in vitro study. Avicenna J. Phytomedicine 2019, 9, 574–586. [Google Scholar] [CrossRef]
- Cos, P.; Rajan, P.; Vedernikova, I.; Calomme, M.; Pieters, L.; Vlietinck, A.J.; Augustyns, K.; Haemers, A.; Berghe, D.V. In Vitro Antioxidant Profile of Phenolic Acid Derivatives. Free. Radic. Res. 2009, 36, 711–716. [Google Scholar] [CrossRef]
- Wypij, M.; Świecimska, M.; Czarnecka, J.; Dahm, H.; Rai, M.; Golinska, P. Antimicrobial and cytotoxic activity of silver nanoparticles synthesized from two haloalkaliphilic actinobacterial strains alone and in combination with antibiotics. J. Appl. Microbiol. 2018, 124, 1411–1424. [Google Scholar] [CrossRef]
- Suflet, D.M.; Popescu, I.; Pelin, I.M.; Ichim, D.L.; Daraba, O.M.; Constantin, M.; Fundueanu, G. Dual Cross-Linked Chitosan/PVA Hydrogels Containing Silver Nanoparticles with Antimicrobial Properties. Pharmaceutics 2021, 13, 1461. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Rao, S.; Sun, J.; Ma, C.; Li, J. p-Coumaric acid kills bacteria through dual damage mechanisms. Food Control. 2012, 25, 550–554. [Google Scholar] [CrossRef]
- Ojha, D.; Patil, K.N. p-Coumaric acid inhibits the Listeria monocytogenes RecA protein functions and SOS response: An antimicrobial target. Biochem. Biophys. Res. Commun. 2019, 517, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Tamer, T.M.; Hassan, M.A.; Omer, A.M.; Baset, W.M.; El-Shafeey, M.E.; Eldin, M.S.M. Synthesis, characterization and antimicrobial evaluation of two aromatic chitosan Schiff base derivatives. Process. Biochem. 2016, 51, 1721–1730. [Google Scholar] [CrossRef]
- Guaresti, O.; García–Astrain, C.; Palomares, T.; Alonso–Varona, A.; Eceiza, A.; Gabilondo, N. Synthesis and characterization of a biocompatible chitosan–based hydrogel cross–linked via ‘click’ chemistry for controlled drug release. Int. J. Biol. Macromol. 2017, 102, 1–9. [Google Scholar] [CrossRef]
- Norbury, W.; Herndon, D.N.; Tanksley, J.; Jeschke, M.G.; Finnerty, C.C. Infection in Burns. Surg. Infect. 2016, 17, 250–255. [Google Scholar] [CrossRef]
- Smith, D.J.; Thomson, P.D.; Garner, W.L.; Rodriguez, J.L. Burn wounds: Infection and healing. Am. J. Surg. 1994, 167, S46–S48. [Google Scholar] [CrossRef]
- Leaper, D.; Assadian, O.; Edmiston, C. Approach to chronic wound infections. Br. J. Dermatol. 2015, 173, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.N.; Carville, K.; Newall, N.; Kapp, S.; Lewin, G.; Karimi, L.; Santamaria, N. Assessing bacterial burden in wounds: Comparing clinical observation and wound swabs. Int. Wound J. 2011, 8, 45–55. [Google Scholar] [CrossRef]
- Khansa, I.; Schoenbrunner, A.R.; Kraft, C.T.; Janis, J.E. Silver in Wound Care—Friend or Foe?: A Comprehensive Review. Plast. Reconstr. Surg. Glob. Open 2019, 7, e2390. [Google Scholar] [CrossRef]
- Kramer, S.A. Effect of povidone-iodine on wound healing: A review. J. Vasc. Nurs. 1999, 17, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Goldenheim, P.D. An appraisal of povidone-iodine and wound healing. Postgrad. Med. J. 1993, 69, S97–S105. [Google Scholar]
- Allsworth, J.E.; Peipert, J.F. Prevalence of bacterial vaginosis: 2001–2004 National Health and Nutrition Examination Survey data. Obstet. Gynecol. 2007, 109, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Sewankambo, N.; Gray, R.H.; Wawer, M.J.; Paxton, L.; McNairn, D.; Wabwire-Mangen, F.; Serwadda, D.; Li, C.; Kiwanuka, N.; Hillier, S.L.; et al. HIV-1 infection associated with abnormal vaginal flora morphology and bacterial vaginosis. Lancet 1997, 350, 546–550. [Google Scholar] [CrossRef]
- Kurki, T.; Sivonen, A.; Renkonen, O.V.; Savia, E.; Ylikorkala, O. Bacterial vaginosis in early pregnancy and pregnancy outcome. Obstet. Gynecol. 1992, 80, 173–177. [Google Scholar] [CrossRef]
- Watts, D.H.; Krohn, M.A.; Hillier, S.L.; Eschenbach, D.A. Bacterial vaginosis as a risk factor for post-cesarean endometritis. Obstet. Gynecol. 1990, 75, 52–58. [Google Scholar] [PubMed]

| CA | PCA | Ca-A | Control | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Amount of PA | 1% | 2% | 5% | 1% | 2% | 5% | 1% | 2% | 5% | 0% |
| Gel Fraction (%) (Chemically Incorporated) | 98 ± 2 | 94 ± 9 | 100 ± 3 | 92 ± 18 | 94 ± 2 | 83 ± 1 | 85 ± 2 | 89 ± 10 | 77 ± 4 | 99 ± 4 |
| Swelling Ratio (Chemically Incorporated) | 1.46 ± 0.02 | 2.42 ± 0.67 | 1.85 ± 0.01 | 2.14 ± 0.11 | 2.05 ± 0.01 | 1.40 ± 0.11 | 1.85 ± 0.02 | 1.86 ± 0.68 | 2.31 ± 0.01 | 2.87 ± 0.03 |
| PA Absorption (w/w %) (Physically Incorporated) | 25 ± 14 | 32 ± 1 | 47 ± 14 | 37 ± 16 | 70 ± 17 | 144 ± 26 | 35 ± 16 | 52 ± 27 | 125 ± 3 | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greene, C.; Beaman, H.T.; Stinfort, D.; Ramezani, M.; Monroe, M.B.B. Antimicrobial PVA Hydrogels with Tunable Mechanical Properties and Antimicrobial Release Profiles. J. Funct. Biomater. 2023, 14, 234. https://doi.org/10.3390/jfb14040234
Greene C, Beaman HT, Stinfort D, Ramezani M, Monroe MBB. Antimicrobial PVA Hydrogels with Tunable Mechanical Properties and Antimicrobial Release Profiles. Journal of Functional Biomaterials. 2023; 14(4):234. https://doi.org/10.3390/jfb14040234
Chicago/Turabian StyleGreene, Caitlyn, Henry T. Beaman, Darnelle Stinfort, Maryam Ramezani, and Mary Beth B. Monroe. 2023. "Antimicrobial PVA Hydrogels with Tunable Mechanical Properties and Antimicrobial Release Profiles" Journal of Functional Biomaterials 14, no. 4: 234. https://doi.org/10.3390/jfb14040234
APA StyleGreene, C., Beaman, H. T., Stinfort, D., Ramezani, M., & Monroe, M. B. B. (2023). Antimicrobial PVA Hydrogels with Tunable Mechanical Properties and Antimicrobial Release Profiles. Journal of Functional Biomaterials, 14(4), 234. https://doi.org/10.3390/jfb14040234






