Abstract
Biomass is one of the most widespread and accessible energy source and steam gasification is one of the most important processes to convert biomass into combustible gases. However, to date the difference of results between the main models used to predict steam gasification producer gas composition have been not analyzed in details. Indeed, gasification, involving heterogeneous reactions, does not reach thermodynamic equilibrium and so thermodynamic models with experimental corrections and kinetic models are mainly applied. Thus, this paper compares a 1-D kinetic model developed in MATLAB, combining hydrodynamics and reaction kinetics, and a 0-D thermodynamic model developed in Aspen Plus, based on Gibbs free energy minimization applying the quasi-equilibrium approach, calibrated by experimental data. After a comparison of the results of the models against experimental data at two S/B ratios, a sensitivity analysis for a wide range of S/B ratios has been performed. The experimental comparison and sensitivity analysis shows that the two models provide sufficiently similar data in terms of the main components of the syngas although the thermodynamic model shows, with increasing S/B, a greater increase of H2 and CO2 and lower decrease of CH4 and CO respect to the kinetic one and the experimental data. Thus, the thermodynamic model, despite being calibrated by experimental data, can be used mainly to analyze global plant performance due to the reduced importance of the discrepancy from a global energy and plant perspective. Meanwhile, the more complex kinetic model should be used when a more precise gas composition is needed and, of course, for reactor design.
1. Introduction
In the last decades, global warming, climate change issues, national energy security and energy dependency issues have led to the need for an alternative to fossil fuels. Biomass, the fourth largest source of energy in the world after oil, coal and natural gas, seems one of the most favorable renewable energy source to replace fossil fuels [1,2,3]. Biomass can be converted in various forms of energy by various processes, according to its characteristics. Biomass use is a carbon-free process since the producing CO2 was previously captured by the plants. At present, biomass use in modern big plants (e.g., Integrated Gasification Combined Cycle, IGCC, for power or Biomass To Liquid, BTL, plants) is the most cost-effective biomass use for power (efficiencies up to 45%) or biofuel (efficiency up to 80%) generation [4,5,6,7,8,9]. Analysis shows that every additional 1% in energy savings leads to a reduction of about 2.6% in gas imports [10]. The Renewable Energy Directive, 2009/28/EC, has driven a rapid deployment of renewable energy. In 2012, energy from renewable sources was estimated to have contributed 14.1% of EU final energy consumption meanwhile the EU target to 2040 is 50% of EU primary energy [11,12,13].
Gasification is considered a very efficient technology for the thermo-chemical conversion of biomass, becoming one of the preferable pathways for the reuse of solid waste [2,14]. Gasification is performed by using a gasifying agent (air, oxygen and/or steam) in order to convert biomass into a combustible gas mixture by the partial oxidation at high temperature (in the range of 800–1000 °C) [2,15]. The produced gas is called syngas and its composition depends on several parameters, such as feedstock composition, gasifying medium, operating temperature and pressure, gasifier design, etc. For this reason it is very difficult to predict the exact composition of the syngas from gasifier [2,16,17]. Syngas is mainly composed by hydrogen, methane, carbon monoxide, carbon dioxide and steam along with several undesired by-products, which concentrations depend on oxidant (i.e., gasifying medium), process conditions (e.g., temperature and pressure), addition of catalysts and/or sorbents (e.g., a CO2 sorbent, as CaO, can shift the thermodynamic equilibrium leading to a H2 content up to 90%), gasifier design (e.g., residence time, etc.), feedstock composition [2,18,19,20]. In any case, high quality syngas is characterized by low level of N2 and CO2, high level of H2 and CO and low level of contaminants and high Low Heating Value (LHV, that is defined by literature as the amount of heat released by combusting an amount of fuel from 25 °C and returning the temperature of combustion products to 150 °C, not recovering the latent heat of vaporization of water in the reaction products). The investigation of syngas composition, varying the operative parameters, is necessary for the optimization of the design and operation of biomass gasification. In addition, conducting experiments on a wide range of operating conditions at large scale could be problematic for safety and cost reasons [21,22,23]. For this reason, mathematical simulation models have acquired great interest in the prediction of process performance, providing a faithful representation of both chemical and physical phenomena occurring into the gasifier and allowing to evaluate the syngas composition with the aim of optimize the gasifier/plant design and its operation [24]. Gasification, involving heterogeneous reactions, does not reach thermodynamic equilibrium (gasification reaction rates are not fast enough and residence times are not long enough for the equilibrium state to be reached) and so thermodynamic models with experimental corrections and kinetic models are mainly applied. Thermodynamic models, depending only on thermodynamic properties, i.e., temperature and pressure, are independent from reactor/particle typologies. On the other hand, kinetic models can be more realistic but are more complex, requiring the implementation of reaction kinetics, hydrodynamic equations. Aspen Plus and MATLAB represent two of the most used simulation tools for biomass gasification [25,26,27,28,29].
Aspen Plus, a chemical engineering process optimization software developed by Massachusetts Institute of Technology (MIT), uses unit operation blocks, such as reactors, columns, pumps, heat exchangers, etc. The unit blocks are connected by material and energy streams in a flow sheet workspace, utilizing sub-sequential modular approach and in-built physical property databases [30]. Although Aspen Plus presents thermodynamic (e.g., GIBBS, i.e., reactor based on the minimization of the free Gibbs energy) and kinetic (e.g., RYIELD, i.e., a simple kinetic reactor based on the reaction yield) reactors, it is typically used for thermodynamic simulation. In order to obtain results closer to experimental values also in thermodynamic equilibrium models, many authors adopt the quasi-equilibrium approach. This approach was introduced by Gumz [31] and it is based on the use of QET (Quasi-Equilibrium Temperature) at which the specific chemical reaction is assumed to reach equilibrium [14], instead of the actual operating temperature of the reactor. This approach does not require specific information regarding the dimensions, capacity and structure of the gasifier but only a set of experimental data. Doherty et al. [32] used the quasi-equilibrium approach based on Gibbs free energy minimization and the restricted equilibrium method to calibrate it against experimental data by the specification of a temperature approach for the gasification reactions. In this way the model was able to evaluate all the main gasification parameters (syngas composition, conversion efficiencies and heating values) and the effect of several variables (like gasification temperature and equivalence ratio (ER, defined as the ratio of the actual fuel/air ratio to the stoichiometric fuel/air ratio)) on such parameters. Arteaga-Pèrez et al. [33] implemented a quasi-equilibrium biomass gasification system and, by changing the gasifier temperature and the air factor, identified that the maximum yield of syngas is achieved at 850 °C and at air ER equal to 0.3. Mirmoshtaghi et al. [34] built a model for biomass gasification in a fluidized-bed gasifier with air oxidant with QET, predicting the volume fraction of the major components (hydrogen, carbon monoxide, carbon dioxide and methane) in product gas. The temperature range of the gasification was set to 730–815 °C, with an ER between 0.22 and 0.53. Giuliano et al. [35] describes the biorefinery process by Aspen Plus, using corn stover as feedstock. Due to the lack of flexible and fast but also accurate models of biomass gasification usable with all the combinations of oxidizing agents, the authors, in Marcantonio et al. [36], have developed a biomass gasification model that is based on the Gibbs free energy minimization. The approach followed included the restricted quasi-equilibrium approach via data-fit regression from experimental data. The simulation results achieved, taking into account several mixes of gasifying agents, were compared and validated against experimental data reported in literature. The values obtained by the developed simulation are in good agreement with literature data. Thus, Aspen Plus is widely applied to the biomass valorization processes but, up to now, no specific comparison has been done between the gas composition resulting from a Aspen plus thermodynamic (with experimental calibration) model and a kinetic model (normally done via MATLAB).
MATLAB is a customizable programming environment for numerical calculation and statistical analysis, created by MathWorks, which can be tailored for system analysis and simulation. Regarding biomass thermo-chemical conversion, many efforts have been made in order to obtain a modelling tool which predicts the effects of different operating conditions such as steam to biomass ratio (S/B) and reactor temperature. Inayat et al. [26] developed a simulation model in MATLAB in order to evaluate the influence of temperature and S/B ratio on biomass gasification for hydrogen production, based on kinetic models and preliminary results. Giuliano et al. [37] implemented a mathematical program consists in discrete optimization problems concerning a multiproduct lignocellulosic biorefinery using MATLAB. Hosseini et al. [38] developed a MATLAB model for air and steam biomass gasification. Lu et al. [39] also developed air-steam gasification in fluidized bed which accounts for both hydrodynamics and chemical kinetics. The customizable nature of MATLAB modelling is advantageous for simulating unconventional concepts and components, which lack of specific property libraries implemented in commercial software. In this sense, Di Carlo et al. [5] developed a 1-D semi-empirical model of a fluidized bed steam-steam/oxygen gasifier by combining governing hydrodynamic equations and kinetic reaction rates. The 1-D modelling approach allows to implement a resolution of the mass and energy balance along the axis of the reactor (considering uniform conditions in the cross section) obtaining the trend of syngas composition and characteristics along the axis of the reactor.
Thus, taking the set of values obtained at the exit section of the kinetic models as outlet composition, is possible to compare with thermodynamic.
Thus, even though kinetic models are the only ones that can be used to design the reactor (encompassing time, dimensions, etc.), both models can be used to evaluate the steam gasification producer gas composition (i.e., gas flow and yield, gas composition, gasification efficiency, carbon and water conversion, etc.). In literature no specific comparison of the two models has been presented in order to understand the differences and so applicability of the model regarding the prediction of the steam gasification producer gas composition. And, considering that this is necessary to understand the specific differences and the way of usability, the scope of this work is to investigate the difference in syngas composition of the two models assessing their suitability (e.g., thermodynamic seems more suitable than kinetic ones due to their enhanced simplicity and generalized approach but they still can assess, also if they are improved with overall mass and energy balance of the plant and the composition of the main gases).
In order to do that, this paper shows a comparison of the gas composition from the two 0-D and 1-D models developed by the authors: the one described in Marcantonio et al. [36] and the other described in Di Carlo et al. [5], considering the same biomass and operating conditions (and no catalysts and/or sorbents addition except for the bed material in the kinetic model). The aim of the work is to evaluate model’s discrepancy from real values within different conditions in order to highlight possible improvements and in which case one model is more suitable than the other.
2. Materials and Methods
2.1. Biomass Choice and Characteristics
The biomass chosen is hazelnut shells because, being a waste biomass, it has a low price (30–80 €/t) and represent a significant agro-industrial by-product in regions of moderate climate [23,24]. The hazelnut shell chemical composition can represent the large lignocellulosic biomass sector (typically presenting a dry and ash free basis composition in the order of 40–50% of C, 35–45% of O2, 5–6% of H2, less than 1% of N2, Cl and S and less than 10% ashes [2]. The % is referred to mol basis). Furthermore, shells present compatible dimensions with fluidized bed (i.e., small size) gasification reactors and moisture content lower than 10%. The proximate and ultimate analysis of hazelnut shells are:
- Proximate analysis (%wt, dry basis): 1.16 of Ash, 72.45 of Volatile Matter and 26.39 of Fixed Carbon;
- Ultimate analysis (%wt, dry basis): 50.38 of C, 6.03 of H, 0.22 of N, 42.32 of O, 0.38 of Cl and 0.67 of S.
More characteristics of hazelnut shells are reported in detail in [36].
2.2. Aspen Plus Modelling
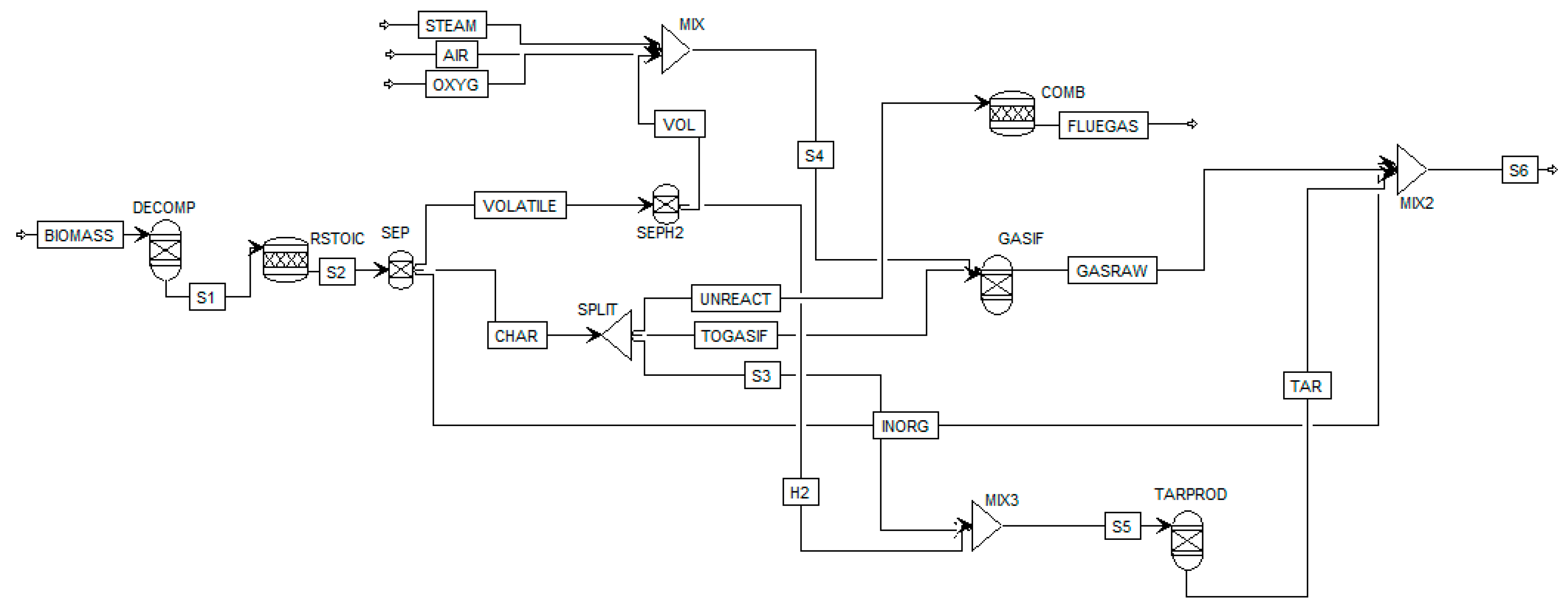
Figure 1 shows the analyzed Aspen Plus biomass gasification model flow sheet. After defining the non-conventional component biomass based on the proximate (i.e., LHV) and ultimate (i.e., composition) analysis, a RYIELD reactor DECOMP is used to convert biomass in conventional components (e.g., C, O2, H2, N2, Cl, S, according to the ultimate analysis). Since the repartition of the products (gas, char unreacted, tar and contaminants) is unknown a DECOMP is considered more suitable than a RYIELD fixing the products based on specific experimental conditions. Products exiting the DECOMP block are moved to the RSTOIC block to simulate the production of H2S, HCl and NH3 (N2, Cl and S as elemental components are known to produce mainly H2S, HCl and NH3, and a fractional conversion of 1 is quite in line with experimental data which represents the worst case of maximum contaminants [36]). The products are moved to a SEP block to separate volatile, char and inorganic (H2S, HCl and NH3) fractions in order to separate (with other SEP) char (i.e., C) and H2 to form tar in the RYIELD block TARPROD where tar is considered to be formed, using experimental data of 18 g/Nm3, repartitioned into 60% benzene, that does not condense, so it is not a “real” tar but it is the most present hydrocarbon in biomass gasification after methane, 20% toluene and 20% naphthalene [36]) and unreacted C to be sent to the combustor. The gasifier, considered as an indirectly heated fluidized-bed reactor, is modeled by a RGibbs reactor (GASIF in Figure 1) and the bed material is sand. Within the reactor, the restricted chemical equilibrium of the specified reactions is simulated in order to set the product gas composition by specifying a temperature approach for each individual reaction. The reactions considered in the simulation are reported in Table 1. The gasifying agent considered in this paper is steam, however in Figure 1 also the streams of oxygen OXYG and air AIR are reported since the model is able to work with all the combination of oxidizing agents, their mass flow has been set to zero. The stream called UNREACT represents the unreacted char, set as 11% of biomass inlet (dry) according to [40] and feeds the combustor COMB. More details of the model can be found in the work of Marcantonio et al. [36].
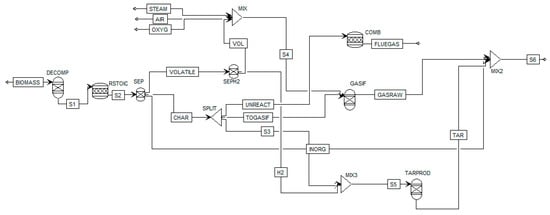
Figure 1.
Flowsheet of the Aspen Plus simulation.

Table 1.
Gasification reactions [36].
2.3. MATLAB Modelling
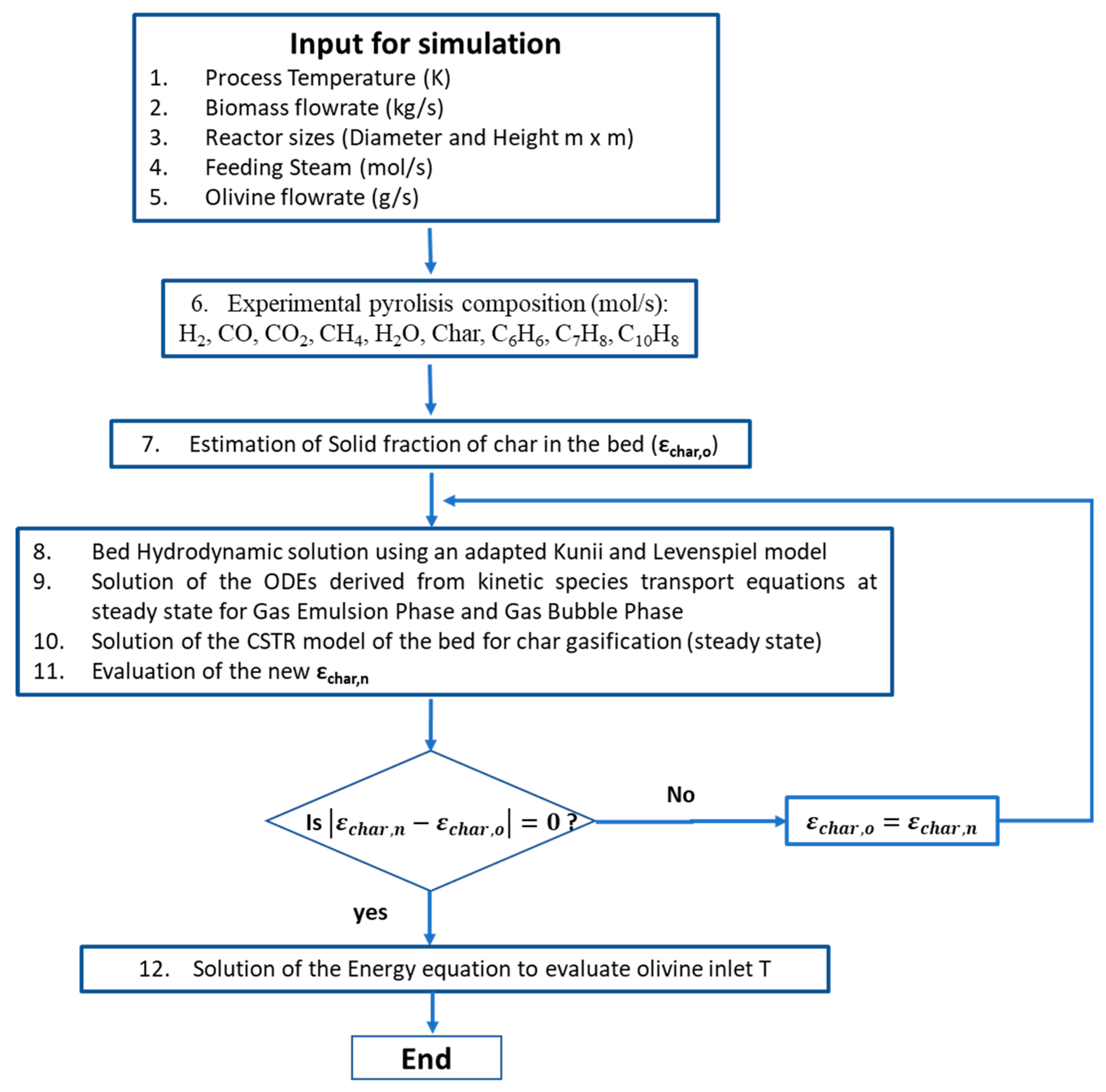
Steam gasification is normally done via fluidized bed gasifier that can allow the recirculation of materials in order to provide the necessary heat for the gasification reactions. Thus, the reactor design is a simple cylinder. Details of the model can be found in Di Carlo et al. [5]. It is a semi-empirical model for steam gasification which implements a 1-D resolution of the mass and energy balance along the axis of the cylinder in order to calculate the syngas composition. In this model, the pyrolysis reactions were considered instantaneous, because the operating temperature is quite high in the gasifier where biomass is fed. For this reason, the de-volatilization time was considered negligible, assuming that biomass is instantaneously converted in its de-volatilization products. In order to obtain realistic values for the pyrolysis products and to validate the biomass steam gasification model, experimental tests on a bench scale fluidized bed reactor were used [41]. A hydrodynamic model according to Kunii and Levenspiel [42] (Geldart group B powder) was used to describe the bubble and emulsion phases and their interaction in the fluidized bed. The rates of the gasification reactions (heterogeneous and homogeneous reactions, including one-ring tar reforming) have been implemented using literature kinetic correlations [9]. In Figure 2 the MATLAB algorithm flow chart is illustrated, more details of the model can be found in the works of Di Carlo et al. [5,43].
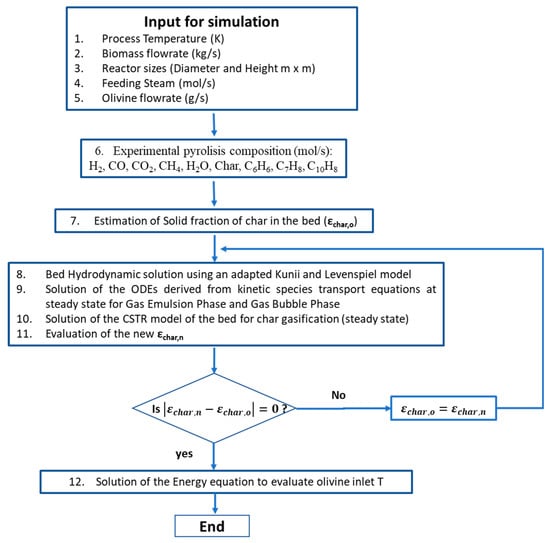
Figure 2.
MATLAB model algorithm flow chart.
The LHV of the produced syngas can be calculated from the standard low calorific values (MJ/kg) of the combustible components of the syngas (120 MJ/kg, 10 MJ/kg and 50 MJ/kg for H2, CO and CH4 respectively) considering their mass-based concentrations in the total gas mixture. The thermal balance of the gasifier determines the temperature drop of the circulating bed material (olivine), considering a fixed ratio of 50 kgolivine/kgbiomass as heat transfer media between the gasifier and the combustor. The specific heat of the olivine is fixed at 750 kJ/(kg·K). Using olivine as bed material also provides a moderate catalytic effect for the heterogeneous reactions in the MATLAB model [5], modifying the syngas composition. The model has been tuned and validated using the experimental results obtained in a bench scale fluidized bed gasifier [43]. The reactions considered in the model are the same of the Aspen Plus (which are reported in Table 1) plus the Boudouard reaction (R6) and the tar reforming (R7), shown below, which were not considered in the Aspen Plus model due to the fact that they are very far from thermodynamic equilibrium. Adding such reactions would lead to inaccurate results respect to the experimental ones (i.e., more CO and H2 and no toluene).
| CO2 + C → 2 CO | (+172 MJ/kmol) | (R6) |
| C7H8 + 7 H2O → 7 CO + 11 H2 | (+881.74 MJ/kmol) | (R7) |
3. Results and Discussion
3.1. Models Comparison
The operation conditions, at which the two models under investigation are simulated, are reported in Table 2.

Table 2.
Common operating conditions of Aspen Plus and MATLAB models.
The size of 350 kWth (kilowatt-thermal) has been chosen as a trade-off between the applicability of fluidized bed technology and biomass availability (e.g., hazelnut shells in a hazelnut production plant, i.e., about 500 t/year). Furthermore, a similar scale gasifier is suitable to integrate with small-to-medium scale CHP end users (as the hazelnut production plants, e.g., few tens of kWe (kilowatt-electric)). The temperature of 850 °C is used since, as reported in [5,36], it represents a trade-off for syngas composition and characteristics, leading to a reasonable value for both gas yield and LHV (consequently optimizing the cold gas efficiency , following the Formula (1)).
where m is the mass in kg.
Atmospheric pressure is typical for small-to-medium scale plants which do not present criticalities in footprint and occupancy of the balance of plant. Table 3 and Table 4 compare syngas composition, LHV and yield obtained by the two models respect to the literature data, Fercher et al. and Hofbauer et al. [44,45,46], for S/B equal to 0.25 and 0.5 respectively; Table 5 and Table 6 show the absolute and relative error committed by each of the two models respect to the reference literature data. The experimental data are always for steam gasification temperature at 850 °C but, owing to the fact that no experimental data are available in literature for hazelnut shells (at 850 °C and S/B of 0.25 and 0.5), the validation has been done considering wood chips that, as already described, have very similar characteristics within the category of lignocellulosic biomass.

Table 3.
Comparison of syngas composition and characteristics (S/B = 0.25).

Table 4.
Comparison of syngas composition and characteristics (S/B = 0.5).

Table 5.
Validation of syngas composition and characteristics (S/B = 0.25).

Table 6.
Validation of syngas composition and characteristics (S/B = 0.5).
For S/B = 0.25 (kgsteam/kgbiomass,dry) the Aspen Plus and MATLAB values of H2 and CH4 are similar and are lower and higher than the reference value, respectively. The AE is within −8 and −3 for H2 and +3 and +5 CH4 with a RE within −20% and −8% for H2 and within +33% and +61% for CH4 due to its lower values in the syngas. This is due to the presence of olivine used as natural catalytic bed material in the experiments of Hofbauer et al. [45] and Fercher et al. [44] that emphasizes the water-gas shift (WGS) reaction (R4) and steam-methane reforming (SMR) reaction (R5), determining in this way the reduction of CO that, vice versa, is higher in the two models (AE +17 and +13 which leads to a RE up to +100%). The MATLAB model, that considers the olivine effect, has fewer differences although the olivine contribution is still lower than what seen in the literature reference. The overestimation of CO consequently shifts the equilibrium of (R4) towards the reagents reducing the CO2 prediction (AE −10 and −14, RE in the range of −34% and −45%). Despite the differences in gas composition, regarding LHV, yield and cold gas efficiency, the models shows good agreement with the experimental values (Aspen Plus maximum AE 1.7 and RE 12%, MATLAB maximum AE 0.7 and RE 5%), confirming that, for plant analysis also the thermodynamic models can be used meanwhile for specific gas prediction the kinetic models are more suitable.
For S/B = 0.5 (kgsteam/kgbiomass,dry) from the comparison of the experimental values obtained by Hofbauer [45] using 0% of nickel catalyst and quartz as bed material it is possible to observe the good correspondence of the developed models for CH4, CO and CO2 (Aspen Plus maximum AE 4 and RE 31%, MATLAB maximum AE 3 and RE 31%). Instead the H2 is overestimated (Aspen Plus maximum AE 8 and RE 23%, MATLAB maximum AE 7 and RE 21%) mainly because the Aspen Plus model, under equal uncatalyzed conditions, considers equilibrium conditions meanwhile MATLAB overestimates the steam reforming of tars, both factors that increase the H2 content of the syngas at higher S/B. At S/B 0.5 the LHV, yield and cold gas efficiency are in good agreement (maximum AE −3 and RE +32%), the LHV of the MATLAB model, in this case, is in a better agreement (AE −0.45 and RE −3%) respect to Aspen Plus (AE −2.95 and RE −20.13%) since it considers the reforming of tars.
In order to get the values from Aspen Plus closer to the ones from literature, and reduce the error, it is possible to improve the data-fit of experimental data used for the QET and make a deeper differentiation among the values come from the using of catalyst inside the reactor. Instead, about MATLAB model, in order to reduce the error between simulation values and experimental ones, the key is to improve the data on kinetic constant and residential time, as well as a differentiation among the values come from the using of catalyst inside the reactor.
3.2. Sensitivity Analysis
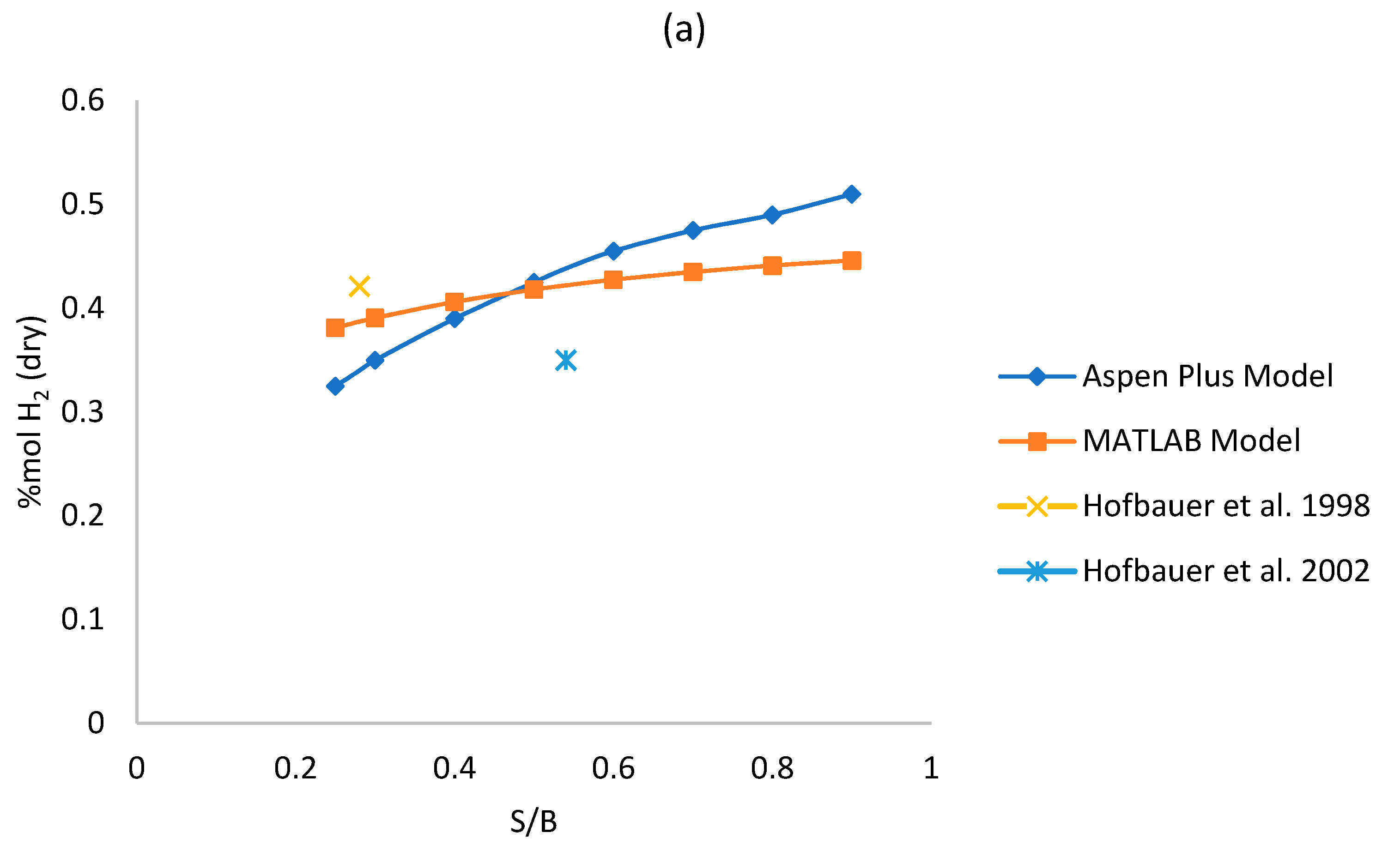
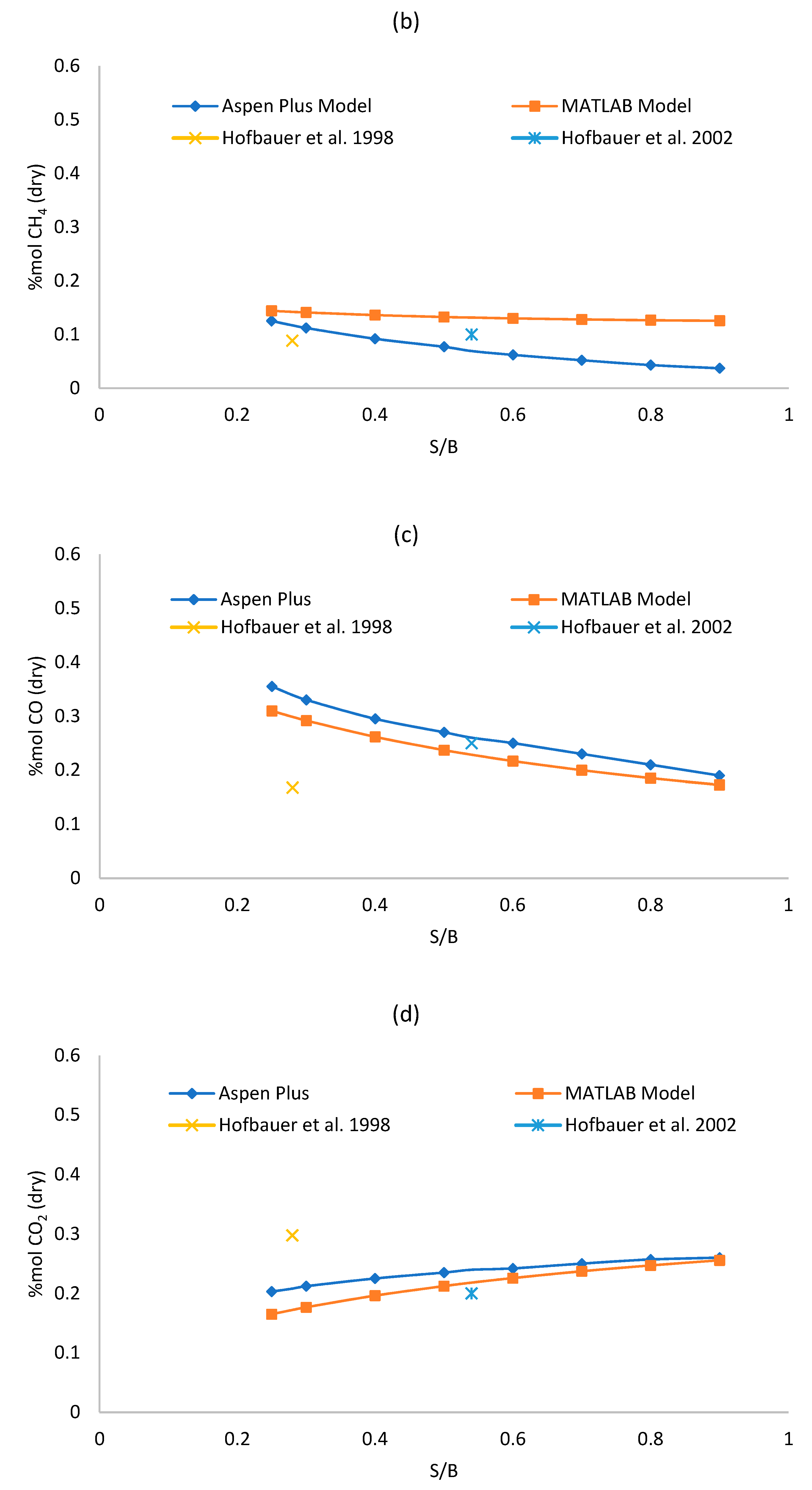
Figure 3a–d shows the sensitivity analysis results for each model, varying the S/B ratio from 0.25 to 0.9 and keeping the gasifier temperature fixed at 850 °C. The effect over syngas composition is observed. The absolute and relative error of each of the two models respect to the reference literature data of Table 5 and Table 6 in S/B equal to 0.25 and 0.5 have been reported in the graphs. A complete analysis of the difference between the Aspen Plus and MATLAB outputs is reported in Table 7.
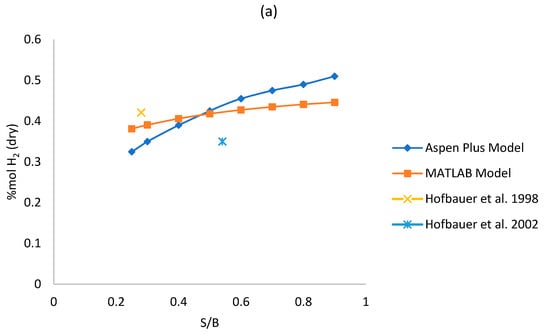
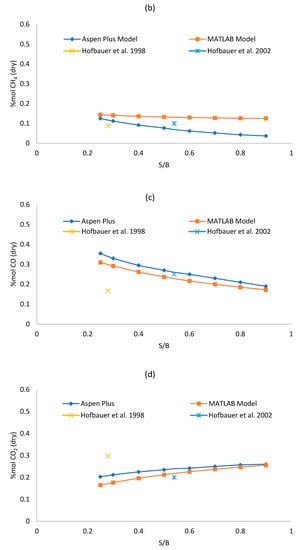
Figure 3.
(a–d) Effect of S/B ratio on syngas (a) hydrogen, (b) methane, (c) carbon monoxide; (d) carbon dioxide dry mole fraction.

Table 7.
Absolute error (AE) and Relative Error (RE) between the Aspen Plus and MATLAB model for each S/B operating point. Maximum values (red), minimum values (green). Mean Absolute Error (MAE) and Mean Relative Error (MRE) over the whole range of S/B.
errors are with sign respect to the median of the reference values.
In order to compare the results of the two models a simple error analysis is taken out evaluating the differences of the estimated syngas composition in relation to the S/B simulated point (Table 7). By comparing only the results of the two models—without a third full reference dataset (which is instead taken out for specific S/B points in Table 5 and Table 6)—the error analysis does not claim to validate the two models for all S/B points, whereas to understand the degree of similarity of the results for the two approaches and, consequently, understand the limits in their applicability in relation to the modelling purpose. The Absolute Error (AE) and Relative Error (RE) between the outputs of the two models in all simulated points are reported in Table 7, together with the mean values of the AE and RE, in order to assess the overall similarity of the datasets along the whole range of the S/B variation.
As shown in Figure 3a, the concentration of H2 increases while steam increases for both models, this is due to the WGS reaction that is favored by the increase of steam. For S/B ratio equal to 0.25, the concentration of H2 given by the Aspen Plus model is lower than the one given by the MATLAB model, but the yield achieved by Aspen Plus is higher. H2 grows faster in the Aspen Plus model since it is a thermodynamic model. As shown in Figure 3c,d, an increase of the S/B ratio results in an increase of CO2 and in a decrease of CO, this can be explained by the influence of the WGS reaction that consumes CO and produces CO2. In fact, even though the WGS is disadvantaged by high temperature respect to the steam-methane reforming (SMR) reaction which is instead favored at high temperature, we should be considered that the quantity of reagents in the WGS reaction are significantly higher compared to the ones of the SMR reaction. So, it is true that the constant of equilibrium of the WGS reaction is lower than the one of the SMR reaction at 850 °C but, since the quantity of reagents of the WGS reaction are higher, the WGS is the reaction that dominates the trends of CO and CO2. Figure 3b reports a clear reducing trend of CH4, as the steam increases, following the SMR reaction. The reduction of the methane in the Aspen Plus model is greater than the one in the MATLAB model with increasing S/B because, being the Aspen Plus a thermodynamic model, it neglects the residence time (more CH4 is converted). Similar trends were reported in literature references [26,47]. High values of RE for CH4 can be justified due to its low molar fraction. The maximum Relative Error can reach 108.92% although in that operating point (S/B = 0.9) the CH4 molar fraction is quite low (0.04 for Aspen Plus and 0.12 for MATLAB) with an AE equal to 0.08, which is acceptable. The overall error analysis shows that the MAE is between 2.11–5.80 for all syngas components. For H2, CO and CO2—which present higher molar fractions (>0.15 mol/molsyngas)—the MRE is contained between 8.95% and 12.64%. The CH4 prediction instead is affected by a larger MRE of 61.45% which is however acceptable, for plant analysis purpose, since the absolute molar fraction is low (0.03–0.15 molCH4/molsyngas).
4. Conclusions
In this work the results of the main used models to predict steam gasification producer gas composition have been compared. One is a thermodynamic quasi-equilibrium model and it is realized by means of Aspen Plus, the second is a 1-D kinetic model developed by means of MATLAB. The composition of the product gas given by the models (at the gasification standard temperature of 850 °C and standard atmospheric pressure) has been first compared against experimental data at S/B ratio equal to 0.25 and 0.5 and after, among the models, over a full range of S/B and. As a general conclusion it can be said that the two models provide sufficiently similar data in terms of the main components of the syngas composition at the outlet of the gasifier. Mean Absolute Error is within 2.11 and 5.80 for all syngas component gases. The Mean Relative Error is acceptable between 8.95% and 12.64% for H2, CO and CO2 gases while for CH4 it is equal to 61.45%. Such high value can be justified by the fact that the contribution of CH4 in the syngas composition is low (<0.15) which affects the overall syngas composition prediction less significantly with an AE of 0.08. The influence of the steam to biomass ratio on the syngas composition of both models was investigated. The concentration of H2 increases while steam increases for both models, due to the WGS reaction, and it grows faster in the Aspen Plus model since it is a thermodynamic model. The values obtained from the simulation by Aspen Plus have a higher error, compared to the literature values, than those of MATLAB. However, this error is acceptable for what regards system simulation (LHV, yield, cold gas efficiency and main gas component) because it is within an error range of 10–20%. For this reason, if the objective of the process modelling is to investigate system coupling and/or integration, thermodynamic models seem to be more suitable than kinetic ones, due to their enhanced simplicity and general applicability; while, at the same time assessing with sufficient accuracy the overall mass and energy balance. On the other hand, if the objective is to predict specific gas composition and design and/or optimize an actual gasifier system, a kinetic model is needed, providing a better accuracy in the syngas composition and, of course, the trends and distributions of the analyzed quantities along the axis of reactor and data regarding the hydrodynamics of the system. For future works, the aim will be to reduce the error between the values obtained from the model and the ones came from experimental data. This could be done, first of all, make a deeper differentiation among the values come from the using of catalyst inside the reactor, and then improving the data-fit of experimental data used for the QET, for Aspen Plus model, and improving the data on kinetic constant and residence time, for MATLAB model.
Author Contributions
Conceptualization, E.B. and A.D.C.; methodology, V.M., E.B., A.M.F.; software, V.M., A.M.F. and A.D.C.; validation, V.M., A.M.F. and E.B.; resources, E.B. and A.D.C. and L.D.Z..; data curation, V.M., E.B. and A.M.F.; writing—original draft preparation, V.M.; writing—review and editing, V.M., A.M.F., A.D.C., L.D.Z.; supervision, E.B. and D.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No 815284 BLAZE project.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pala, L.P.R.; Wang, Q.; Kolb, G.; Hessel, V. Steam gasification of biomass with subsequent syngas adjustment using shift reaction for syngas production: An Aspen Plus model. Renew. Energy 2017, 101, 484–492. [Google Scholar] [CrossRef]
- Bocci, E.; Sisinni, M.; Moneti, M.; Vecchione, L.; Di Carlo, A.; Villarini, M. State of Art of Small Scale Biomass Gasification Power Systems: A Review of the Different Typologies. Energy Proced. 2014, 45, 247–256. [Google Scholar] [CrossRef]
- Thapa, S.; Bhoi, P.; Kumar, A.; Huhnke, R.L. Effects of Syngas Cooling and Biomass Filter Medium on Tar Removal. Energies 2017, 10, 349. [Google Scholar] [CrossRef]
- IEA-Analysis. Biomass for Power Generation and CHP; essentials3; International Energy Agency: Paris, France, 2007; Available online: https://www.iea.org/reports/biomass-for-power-generation-and-chp (accessed on 20 September 2020).
- Di Carlo, A.; Borello, D.; Bocci, E. Process simulation of a hybrid SOFC/mGT and enriched air/steam fluidized bed gasifier power plant. Int. J. Hydrogen Energy 2013, 38, 5857–5874. [Google Scholar] [CrossRef]
- Thakkar, M.; Mohanty, P.; Shah, M.; Singh, V. An Overview of Biomass Gasification. In Recent Advancements in Biofuels and Bioenergy Utilization; Springer: Singapore, 2018; pp. 147–176. [Google Scholar] [CrossRef]
- Sikarwar, V.S.; Zhao, M.; Clough, P.T.; Yao, J.; Zhong, X.; Memon, M.Z.; Shah, N.; Anthony, E.; Fennell, P. An overview of advances in biomass gasification. Energy Environ. Sci. 2016, 9, 2939–2977. [Google Scholar] [CrossRef]
- Sarangi, P.; Nanda, S.; Mohanty, P. Recent Advancements in Biofuels and Bioenergy Utilization; Springer: Berlin, Germany, 2018. [Google Scholar]
- Di Carlo, A.; Bocci, E.; Naso, V. Process simulation of a SOFC and double bubbling fluidized bed gasifier power plant. Int. J. Hydrogen Energy 2013, 38, 532–542. [Google Scholar] [CrossRef]
- “Impact Assessment on the Energy Efficiency Directive Review, European Commission, 2014.” Energy Efficiency and Its Contribution to Energy Security and the 2030 Framework for Climate and Energy Policy. Available online: https://ec.europa.eu/energy/sites/ener/files/documents/2014_eec_ia_adopted_part1_0.pdf (accessed on 30 September 2020).
- Communication from the Commission to the European Parliament and the Council. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52014DC0330&from=EN (accessed on 5 October 2020).
- Bocci, E.L.; Zotto, D.; Monforti, A.F.; Marcantonio, V.; Di Alessandro, C.A.; Giuliano, D.E.; Barisano, S.; Stephen, D.; Massimiliano, P.M.; Pietra, D.; et al. First Results of the H2020-Lc-Sc3-Res.-11 Blaze Project: Biomass Low Cost Advanced Zero Emission Small-To-Medium Scale Integrated Gasifier Fuel Cell Combined Heat and Power Plan. In Proceedings of the 8th European Fuel Cell Technology & Applications Piero Lunghi Conference—EFC19, Napoli, Italy, 9–11 December 2019. [Google Scholar]
- SET Plan Delivering Results: The Implementation Plans, SET Plan 2018 ed.; European Commission: Brussels, Belgium, 2019.
- Arnavat, M.P.; Bruno, J.C.; Coronas, A. Review and analysis of biomass gasification models. Renew. Sustain. Energy Rev. 2010, 14, 2841–2851. [Google Scholar] [CrossRef]
- Ramzan, N.; Ashraf, A.; Naveed, S.; Malik, A. Simulation of hybrid biomass gasification using Aspen plus: A comparative performance analysis for food, municipal solid and poultry waste. Biomass Bioenergy 2011, 35, 3962–3969. [Google Scholar] [CrossRef]
- Villarini, M.; Marcantonio, V.; Colantoni, A.; Bocci, E.; Villarini, M.; Marcantonio, V.; Colantoni, A.; Bocci, E. Sensitivity Analysis of Different Parameters on the Performance of a CHP Internal Combustion Engine System Fed by a Biomass Waste Gasifier. Energies 2019, 12, 688. [Google Scholar] [CrossRef]
- Bocci, E.; Di Carlo, A.; McPhail, S.J.; Gallucci, K.; Foscolo, P.U.; Moneti, M.; Villarini, M.; Carlini, M. Biomass to fuel cells state of the art: A review of the most innovative technology solutions. Int. J. Hydrogen Energy 2014, 39, 21876–21895. [Google Scholar] [CrossRef]
- Dascomb, J.; Krothapalli, A.; Fakhrai, R. Thermal conversion efficiency of producing hydrogen enriched syngas from biomass steam gasification. Int. J. Hydrogen Energy 2013, 38, 11790–11798. [Google Scholar] [CrossRef]
- Kumar, A.; Jones, D.D.; Hanna, M.A. Thermochemical Biomass Gasification: A Review of the Current Status of the Technology. Energies 2009, 2, 556–581. [Google Scholar] [CrossRef]
- Marcantonio, V.; Bocci, E.; Ouweltjes, J.P.; Del Zotto, L.; Monarca, D. Evaluation of sorbents for high temperature removal of tars, hydrogen sulphide, hydrogen chloride and ammonia from biomass-derived syngas by using Aspen Plus. Int. J. Hydrogen Energy 2020, 45, 6651–6662. [Google Scholar] [CrossRef]
- Hamel, S.; Krumm, W. Mathematical modelling and simulation of bubbling fluidised bed gasifiers. Powder Technol. 2001, 120, 105–112. [Google Scholar] [CrossRef]
- Moneti, M.; Di Carlo, A.; Bocci, E.; Foscolo, P.; Villarini, M.; Carlini, M. Influence of the main gasifier parameters on a real system for hydrogen production from biomass. Int. J. Hydrogen Energy 2016, 41, 11965–11973. [Google Scholar] [CrossRef]
- Pallozzi, V.; Di Carlo, A.; Bocci, E.; Villarini, M.; Foscolo, P.; Carlini, M. Performance evaluation at different process parameters of an innovative prototype of biomass gasification system aimed to hydrogen production. Energy Convers. Manag. 2016, 130, 34–43. [Google Scholar] [CrossRef]
- Ahmed, T.Y.; Ahmad, M.M.; Yusup, S.; Inayat, A.; Khan, Z. Mathematical and computational approaches for design of biomass gasification for hydrogen production: A review. Renew. Sustain. Energy Rev. 2012, 16, 2304–2315. [Google Scholar] [CrossRef]
- Marcantonio, V.; De Falco, M.; Capocelli, M.; Bocci, E.; Colantoni, A.; Villarini, M. Process analysis of hydrogen production from biomass gasification in fluidized bed reactor with different separation systems. Int. J. Hydrogen Energy 2019, 44, 10350–10360. [Google Scholar] [CrossRef]
- Inayat, A.; Ahmad, M.M.; Yusup, S.; Mutalib, M.I.A. 2010, Biomass Steam Gasification with In-Situ CO2 Capture for Enriched Hydrogen Gas Production: A Reaction Kinetics Modelling Approach. Energies 2010, 3, 1472–1484. [Google Scholar] [CrossRef]
- Liao, C.-H.; Summers, M.; Seiser, R.; Cattolica, R.; Herz, R. Simulation of a pilot-scale dual-fluidized-bed gasifier for biomass. Environ. Prog. Sustain. Energy 2014, 33, 732–736. [Google Scholar] [CrossRef]
- Silva, V.; Rouboa, A.I. Using a two-stage equilibrium model to simulate oxygen air enriched gasification of pine biomass residues. Fuel Process. Technol. 2013, 109, 111–117. [Google Scholar] [CrossRef]
- Moradi, R.; Marcantonio, V.; Cioccolanti, L.; Bocci, E. Integrating biomass gasification with a steam-injected micro gas turbine and an Organic Rankine Cycle unit for combined heat and power production. Energy Convers. Manag. 2020, 205, 112464. [Google Scholar] [CrossRef]
- Patra, T.K.; Sheth, P.N. Biomass gasification models for downdraft gasifier: A state-of-the-art review. Renew. Sustain. Energy Rev. 2015, 50, 583–593. [Google Scholar] [CrossRef]
- Gumz, W. Gas. Producers and Blast Furnaces; Wiley: Hoboken, NJ, USA, 1950. [Google Scholar]
- Doherty, W.; Reynolds, A.; Kennedy, D. The effect of air preheating in a biomass CFB gasifier using ASPEN Plus simulation. Biomass Bioenergy 2009, 33, 1158–1167. [Google Scholar] [CrossRef]
- Arteaga-Perez, L.E.; Casas-Ledón, Y.; Prins, W.; Radovic, L. Thermodynamic predictions of performance of a bagasse integrated gasification combined cycle under quasi-equilibrium conditions. Chem. Eng. J. 2014, 258, 402–411. [Google Scholar] [CrossRef]
- Mirmoshtaghi, G.; Li, H.; Thorin, E.; Dahlquist, E. Evaluation of different biomass gasification modeling approaches for fluidized bed gasifiers. Biomass BioEnergy 2016, 91, 69–82. [Google Scholar] [CrossRef]
- Giuliano, A.; Barletta, D.; De Bari, I.; Poletto, M. Techno-economic assessment of a lignocellulosic biorefinery co-producing ethanol and xylitol or furfural. In Computer Aided Molecular Design: Theory and Practice; Elsevier: Amsterdam, The Netherlands, 2018; Volume 43, pp. 585–590. [Google Scholar] [CrossRef]
- Marcantonio, V.; Bocci, E.; Monarca, D. Development of a Chemical Quasi-Equilibrium Model of Biomass Waste Gasification in a Fluidized-Bed Reactor by Using Aspen Plus. Energies 2019, 13, 53. [Google Scholar] [CrossRef]
- Giuliano, A.; Cerulli, R.; Poletto, M.; Raiconi, G.; Barletta, D. Optimization of a Multiproduct Lignocellulosic Biorefinery using a MILP Approximation. In Computer Aided Molecular Design: Theory and Practice; Elsevier: Amsterdam, The Netherlands, 2014; Volume 33, pp. 1423–1428. [Google Scholar] [CrossRef]
- Hosseini, M.; Dincer, I.; Rosen, M.A. Steam and air fed biomass gasification: Comparisons based on energy and exergy. Int. J. Hydrogen Energy 2012, 37, 16446–16452. [Google Scholar] [CrossRef]
- Lu, P.; Kong, X.; Wu, C.; Yuan, Z.; Ma, L.; Chang, J. Modeling and simulation of biomass air-steam gasification in a fluidized bed. Front. Chem. Eng. China 2008, 2, 209–213. [Google Scholar] [CrossRef]
- Kaushal, P.; Pröll, T.; Hofbauer, H. Model development and validation: Co-combustion of residual char, gases and volatile fuels in the fast fluidized combustion chamber of a dual fluidized bed biomass gasifier. Fuel 2007, 86, 2687–2695. [Google Scholar] [CrossRef]
- Jand, N.; Foscolo, P.U. Decomposition of Wood Particles in Fluidized Beds. Ind. Eng. Chem. Res. 2005, 44, 5079–5089. [Google Scholar] [CrossRef]
- Kunii, D.; Levenspiel, O. Fluidized reactor models. 1. For bubbling beds of fine, intermediate, and large particles. 2. For the lean phase: Freeboard and fast fluidization. Ind. Eng. Chem. Res. 1990, 29, 1226–1234. [Google Scholar] [CrossRef]
- Hermann, H.; Reinhard, R.; Klaus, B.; Reinhard, K. Biomass CHP Plant Güssing—A Success Story. In Proceedings of the Expert Meeting on Pyrolysis and Gasification of Biomass and Waste, Strasbourg, France, 30 September–1 October 2002. [Google Scholar]
- Hofbauer, H.; Fercher, E.; Fleck, T.; Rauch, R.; Veronik, G. Two Years Experience with the FICFB-Gasification Process. In Proceedings of the 10th European Conference and Technology Exhibition, Wurzburg, Germany, 8–11 June 1998; pp. 3–6. [Google Scholar]
- Hofbauer, H.; Rauch, R.; Loeffler, G.; Kaiser, S.; Fercher, E.; Tremmel, H. Six Years Experience with the FICFB-Gasification Process. In Proceedings of the 12th European Conference and Technology Exhibition Biomass Energy, Amsterdam, The Netherlands, 17–21 June 2002; pp. 982–985. [Google Scholar]
- Pfeifer, C.; Rauch, A.R.; Hofbauer, H. In-Bed Catalytic Tar Reduction in a Dual Fluidized Bed Biomass Steam Gasifier. Ind. Eng. Chem. Res. 2004, 43, 1634–1640. [Google Scholar] [CrossRef]
- Florin, N.H.; Harris, A.T. Hydrogen production from biomass coupled with carbon dioxide capture: The implications of thermodynamic equilibrium. Int. J. Hydrogen Energy 2007, 32, 4119–4134. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).