Towards a Multiscale Model of Acute HIV Infection
Abstract
:1. Introduction
2. Methods
2.1. Multiscale Framework
2.1.1. Cell Displacement
2.1.2. Cell Division and Differentiation
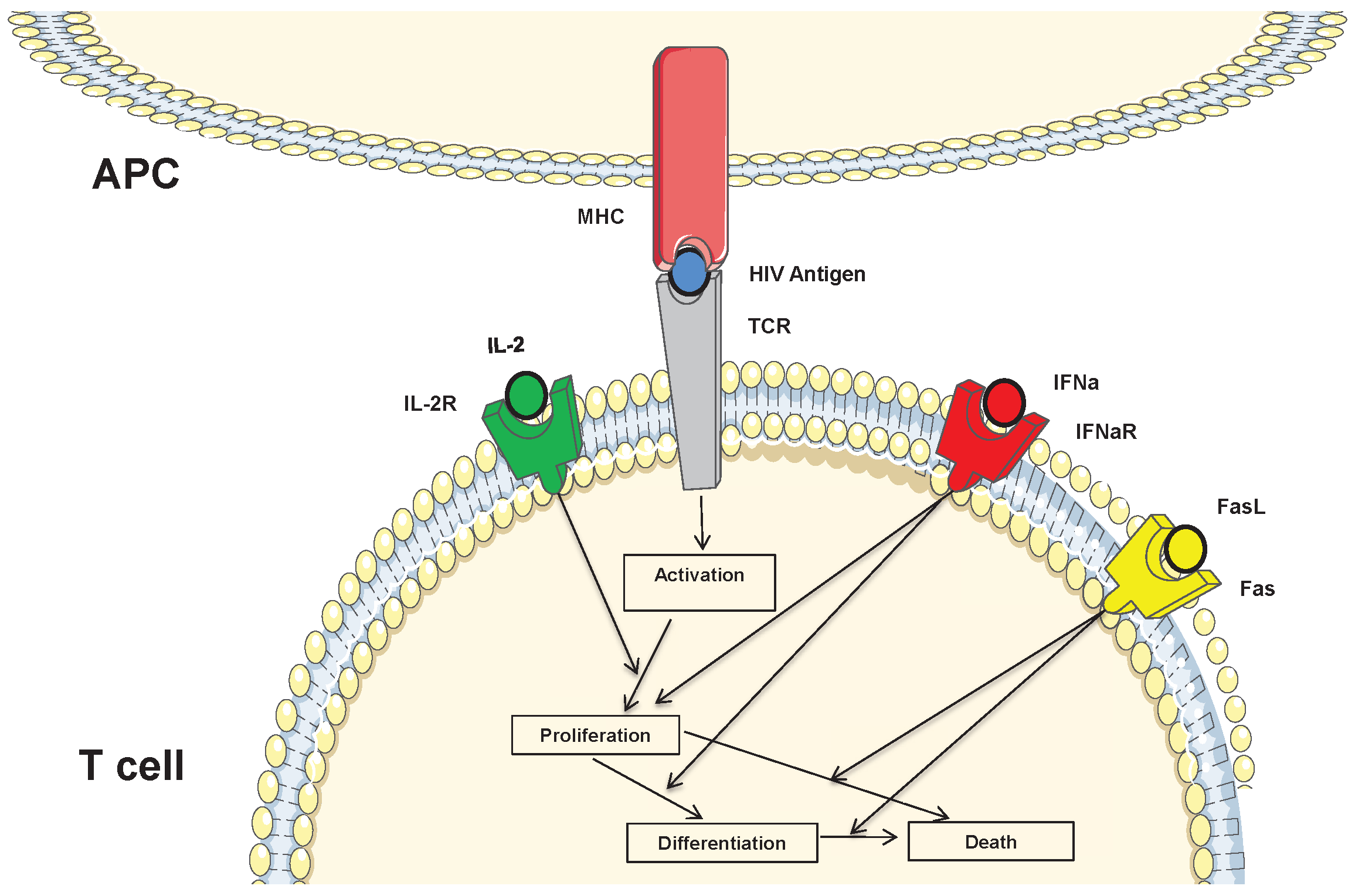
2.2. Biological Assumptions
- HIV enters the lymph node with infected antigen-presenting cells. These cells secrete HIV and transmit it to uninfected (both HIV-specific and non-specific) CD4+ T cells by cell-cell interaction;
- Each APC or CD4+ T cell is determined by an intracellular viral RNA concentration (). The cell is considered to be infected if reaches some threshold level . HIV upregulates the concentration of caspase, which can result in cell apoptosis;
- The CD8+ T cells produce FasL, as well as other apoptosis-inducing factors. They also activate the caspase cascade through direct cell-cell contact with infected cells resulting in target cell apoptosis. These two mechanisms only affect infected CD4+ T cells and not uninfected cells;
- HIV impairs the immune response by reducing the number of CD4+ T cells in the lymph node. These cells are killed by cytotoxic CD8+ T cells or by the virus itself. As a result, fewer mature helper CD4+ T cells are left, and less IL-2 and type I IFN are produced. This leads to a decrease in the survival and differentiation of CD8+ T cells, which ultimately results in the relapse of the infection.
- 1.
- , the viral load in blood;
- 2.
- , the abundance of infected CD4+ T cells in blood;
- 3.
- , the abundance of uninfected CD4+ T cells in blood;
- 4.
- , the abundance of HIV-specific effector CD8+ T cells in blood.
- 5.
- , the density of APCs in the T cell zone of LN, uninfected or infected;
- 6.
- , the density of uninfected CD4+ T cells in the T cell zone of LN; and , the density of infected cells;
- 7.
- , the density of CD8+ T cells in the T cell zone of LN;
- 8.
- , the concentration of free HIV in the T cell zone;
- 9.
- , the concentration of IL-2 in the T cell zone;
- 10.
- , the concentration of type I IFN in the T cell zone;
- 11.
- , the concentration FasL in the T cell zone.
- 12.
- , the intracellular concentration of viral genomes in the i-th cell;
- 13.
- , the intracellular concentration of IL-2-induced signaling molecules in the i-th cell;
- 14.
- , the intracellular concentration of type I IFN-induced signaling molecules in the i-th cell;
- 15.
- , the intracellular concentration of FasL-induced signaling molecules in the i-th cell.
3. The Model of Acute HIV Infection
3.1. Blood Compartment
- 1.
- , rate of T cell production and release into the body: ;
- 2.
- , death rate of T cells in the body: ;
- 3.
- , elimination rate of the infected cells by T cells ;
- 4.
- a, a growth rate parameter of infected cells: ;
- 5.
- h, a parameter in the growth function of infected cells: .
- 6.
- , migration rate constant of CD8+ T cells from LN to blood. The fraction of HIV-specific CTLs in acute HIV infection can reach 10% of the total number of CD8+ T cells, i.e., about 100 cell/L [11]. Therefore, the value of should be within the range 0.004–0.04 ;
- 7.
- , the disappearance rate of HIV-specific effector CD8+ T cells from blood [11]: = 0.0013 .
- 8.
- a, the accumulation rate constant of HIV-infected CD4+ T cells in blood due to the migration of cells from LNs and other peripheral tissues: the reference value 0.1 suggested in [1] was tuned to 0.35 ;
- 9.
- h, the inverse of the threshold of infected T cells density in LN that leads to 50% reduction of the maximal accumulation rate of the infected cells in blood: 1/100 ·L;
- 10.
- , the elimination rate of HIV-infected CD4+ T cells by CTL-mediated killing. We used the value estimated in [9] 0.0004 L/(cell·h). For a strong T cell response, this value reaching 10% of the total CTL population in blood, this value will ensure the elimination rate of the infected cells from blood of about 1 per day;
- 11.
- 12.
- λ, the influx rate of CD4+ from the lymph nodes: 0.45 (L·h)−1;
- 13.
- , the inverse of the threshold of uninfected T cells density in LN that leads to 50% reduction of the maximal accumulation rate of the uninfected cells in blood: 1/200 ·L;
- 14.
- , the infection rate of the CD4+ T cells in blood: 0.00042 (L·h)−1;
- 15.
- , the death rate of uninfected CD4+ T cells in blood: .
- 16.
- k the influx of HIV from LNs to blood was estimated in [40]. Taking into account the volume differences of our computational domain (0.001 L) and the unit of blood volume L, we used the following value 100 virion/(L·h);
- 17.
3.2. Population Dynamics of Infection in LN
3.2.1. Free HIV
3.2.2. HIV Infection in Target Cells
- , the infection rate constant for susceptible cells by free virus. It can be evaluated using the estimates in [44]: L/(virion·h);
- , specifies the maximal production rate of HIV DNA in the activated cell. We used the following estimate virion/(cell·h) [11];
- , specifies the protective effect of type I interferon on HIV DNA growth in the activated cell. The following estimate [45] is used to reproduce a 50% reduction in the viral DNA synthesis rate due to the availability of type I IFN: = 467 L/pg;
- , the probability that a susceptible cell is infected when it contacts infected cells was estimated in [44] to be .
3.3. Cytokine Fields in LN
3.3.1. IL-2
- , the secretion rate of IL-2 by a single CD4+ T cell: molec/h;
- , the degradation rate of extracellular IL-2: 0.5 .
3.3.2. Type I IFN
- , the secretion rate of type I IFN by single activated APC (plasmacytoid dendritic cell): molec/h;
- , the degradation rate of extracellular type I IFN: 0.012 .
3.3.3. FasL
3.4. Intracellular Regulation of Cell Fate
3.4.1. IL-2 Signaling
- , the number of IL-2 molecules internalized by T cells via IL-2 receptors: 2000–5000 per T cell, with used in simulations;
- , the saturation concentration of IL-2 for T cell division in vitro: molec/mL for cells/mL.
3.4.2. Type 1 IFN Signaling
- C1
- If the concentration of activation signals induced by type I IFN, , is greater than some critical level at the beginning of the cell cycle and that of is smaller than the critical level at the end of the cell cycle, then the cell will differentiate, resulting in a mature cell.
- C2
- If the concentration of activation signals induced by IL-2, , is greater than some critical level at the end of the cell cycle, then the cell will divide, producing two more mature cells.
- C3
- If at the beginning of cell cycle and at the end of cell cycle, then the cell will die by apoptosis and will be removed from the computational domain.
3.4.3. FasL Signaling
- , the rate of pro-apoptotic signals accumulation because of the viral replication. As the death rate of the infected CD4 T cells is , then we used it to quantify the cytopathic effect of the intracellular HIV DNA on the cell. It can be further scaled depending on the choice of the threshold . The latter was estimated in [12], and we set it M;
- , specifies the FasL-Fas-induced caspase accumulation rate. It was estimated to be in [12];
- , the killing rate of infected cells by effector CTL. It has been indicated in [46] that once in contact with a target cells, the CTL can program them to undergo apoptosis within 5 min. This value results in the following estimate for the impact of CTL on death likelihood ;
- , the caspase degradation rate is taken from [18] .
3.5. Population Dynamics of Immune Response in LN
4. Numerical Simulation Results
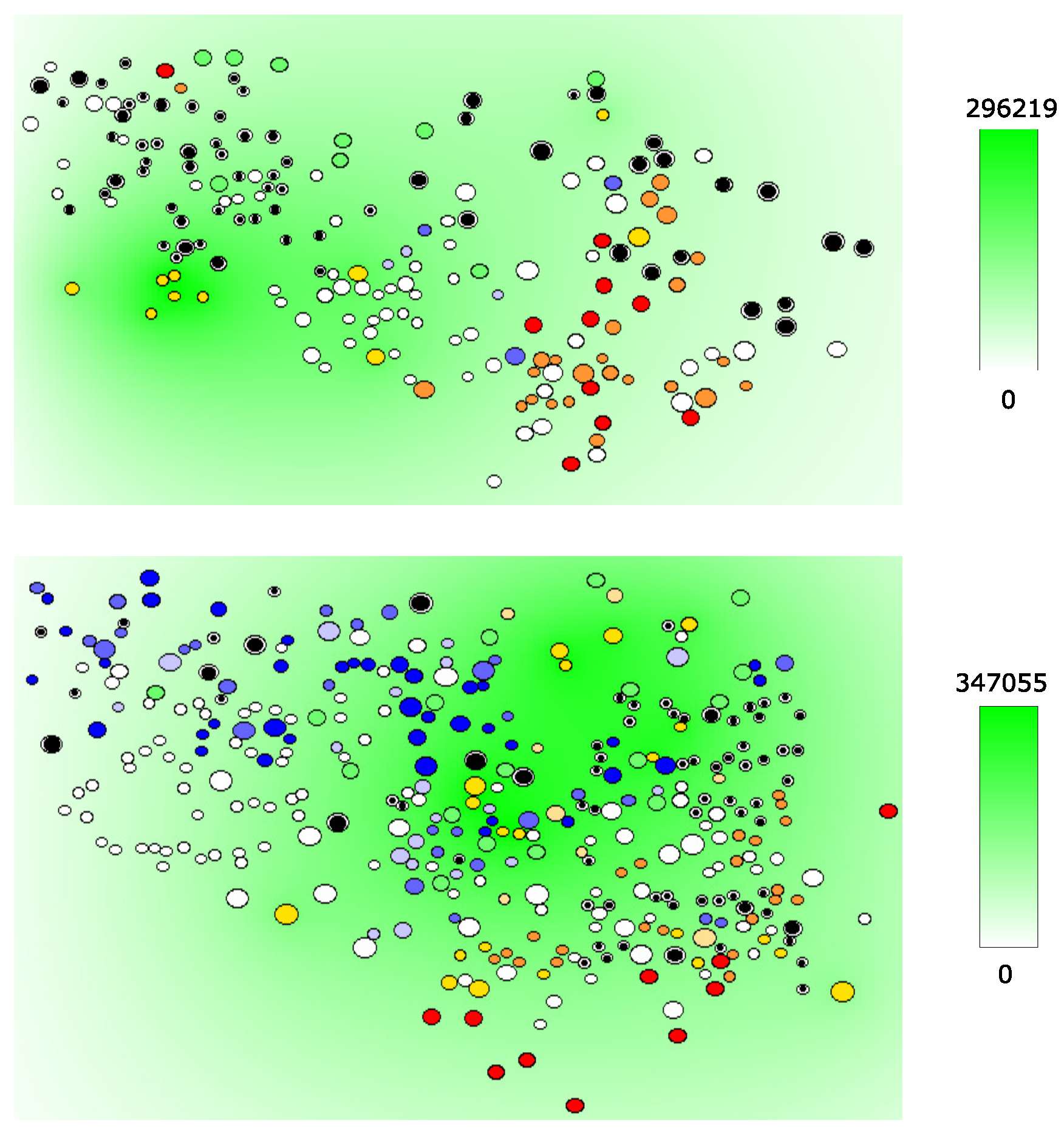
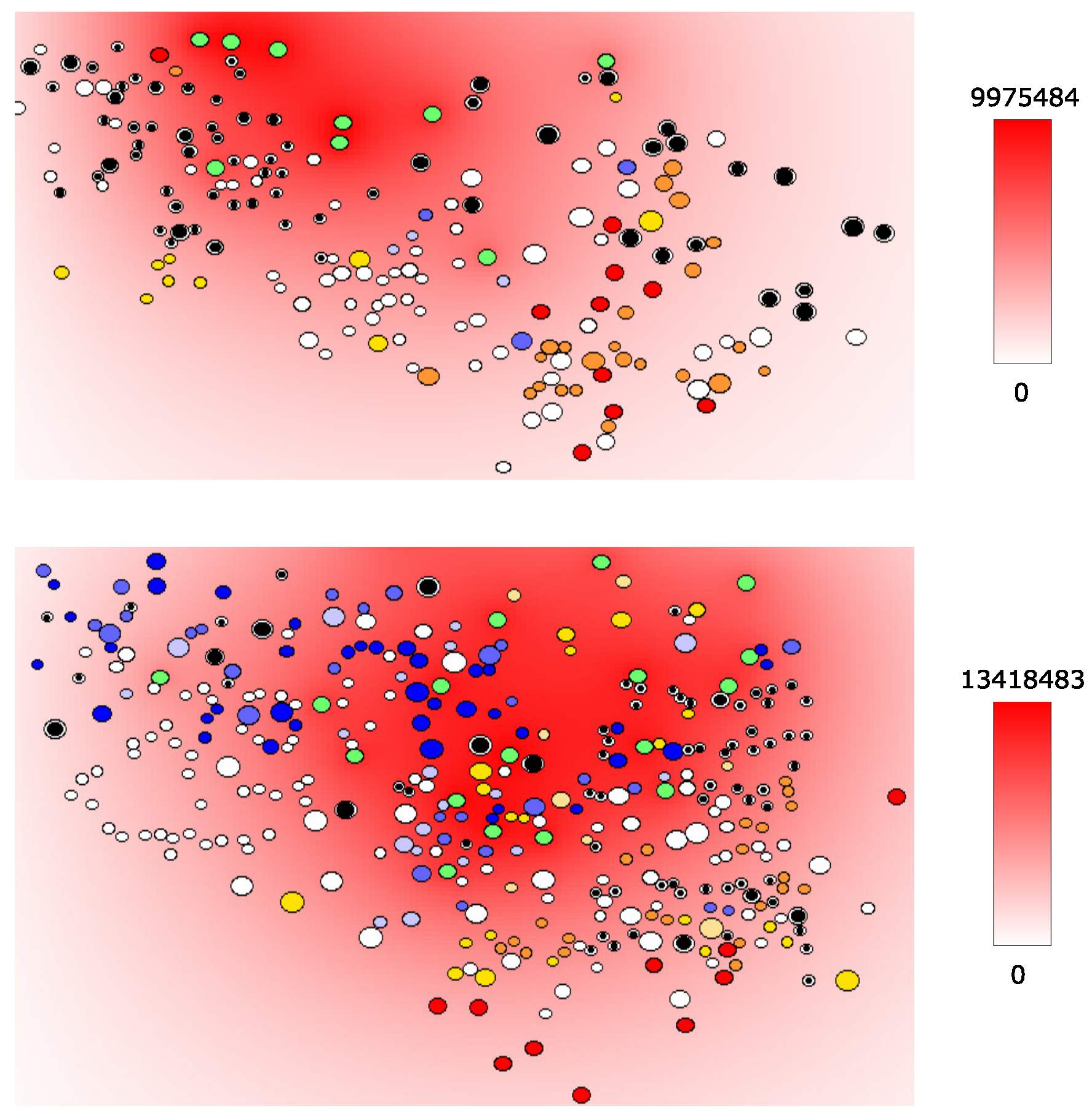
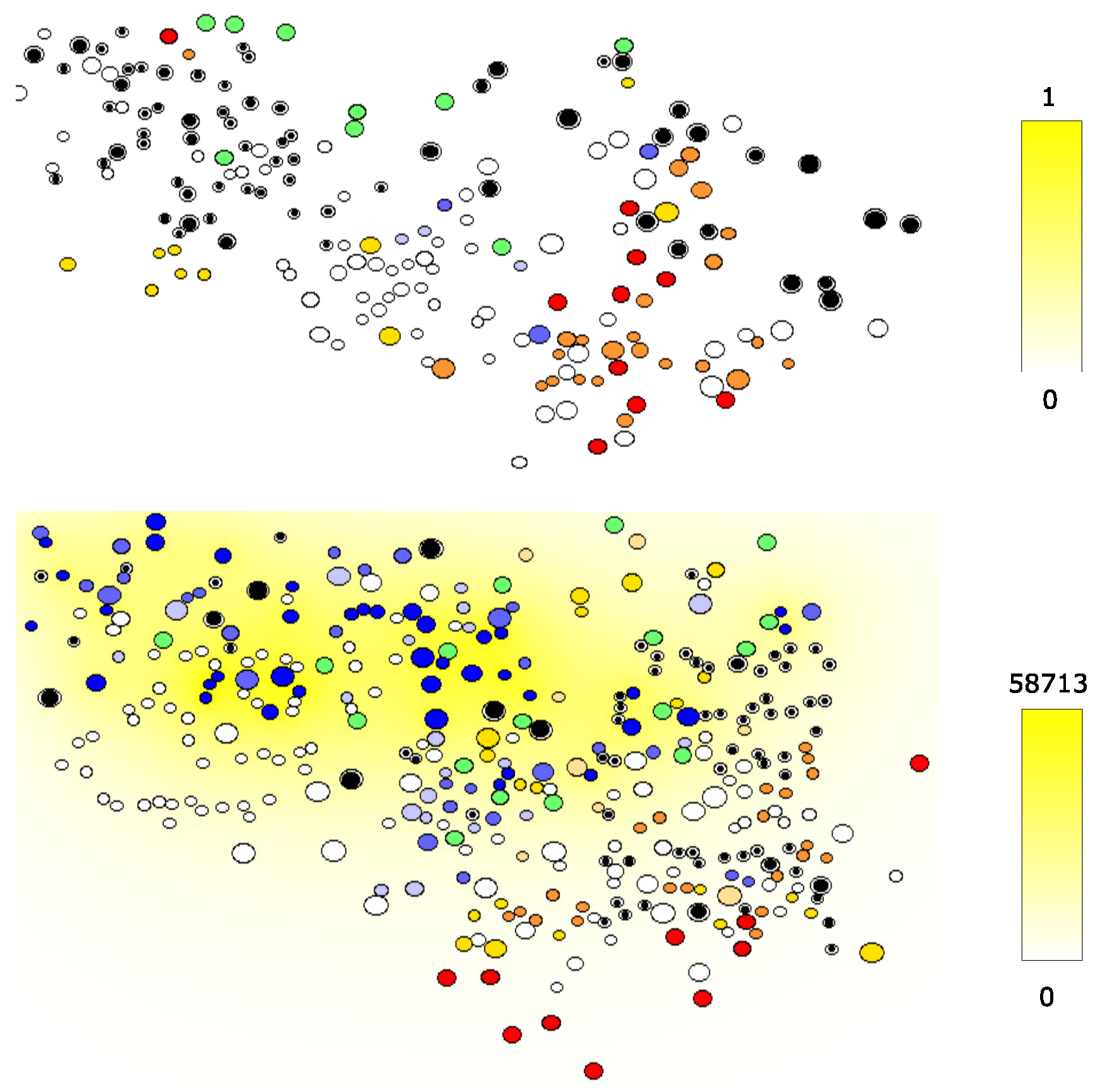
4.1. Dynamics of APCs, CD4+ and CD8+ T Cells in LN
4.2. HIV and Cytokine Fields in LN
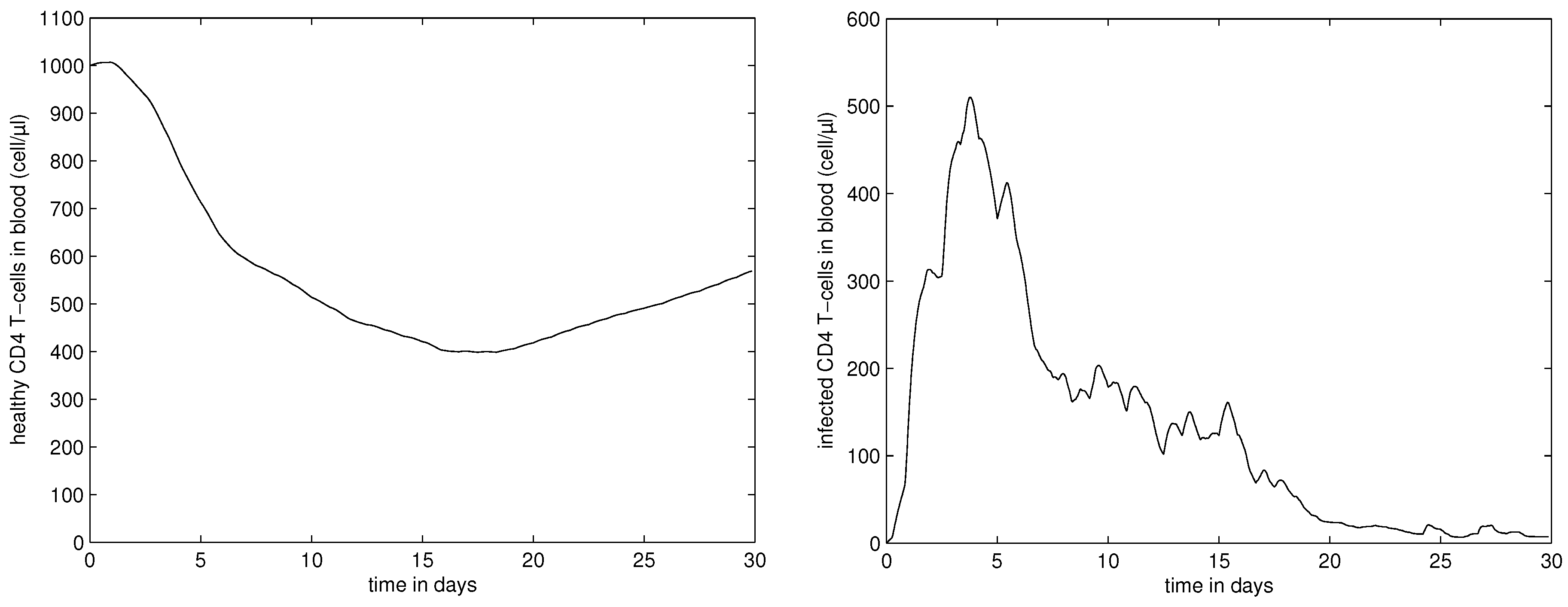
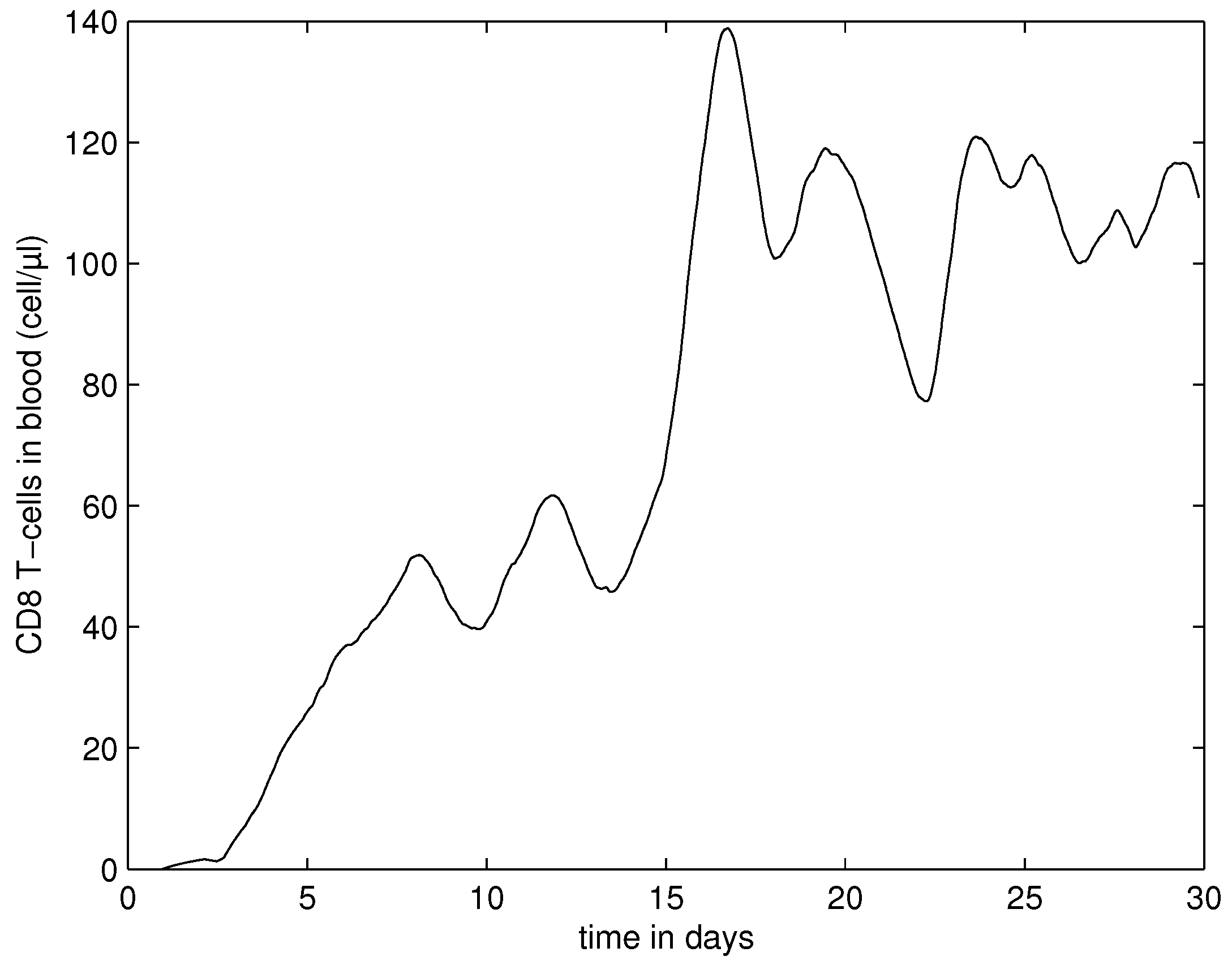
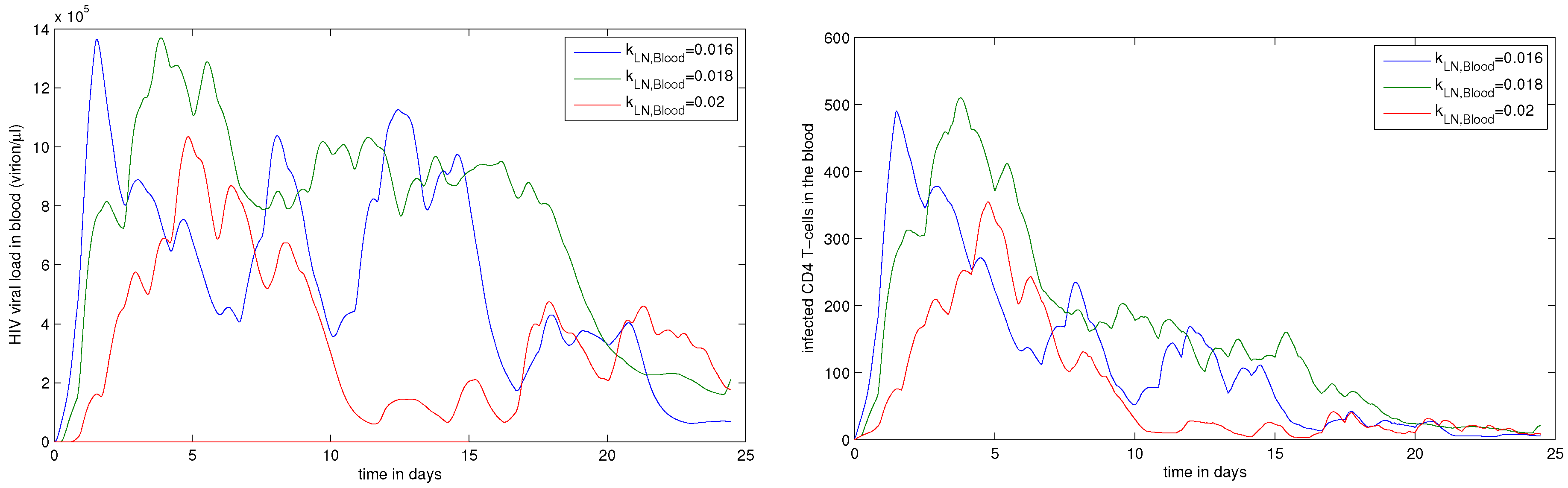
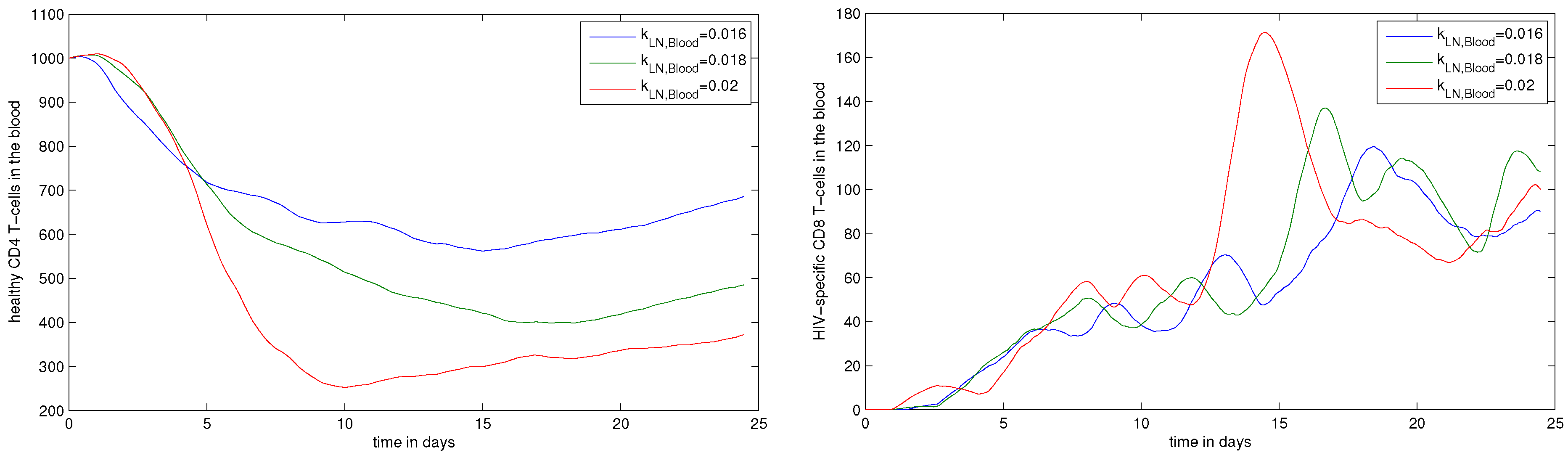
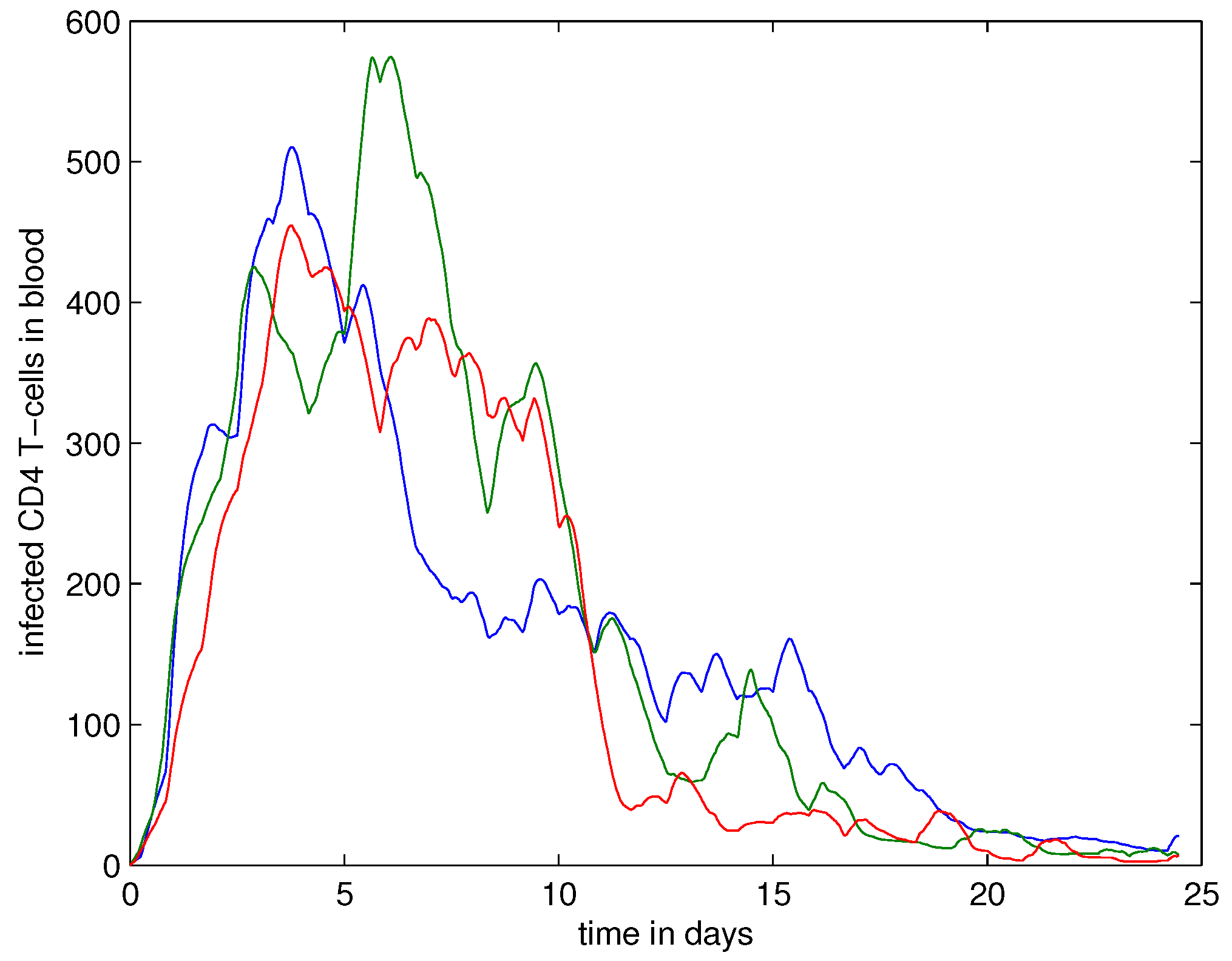
4.3. Systemic Dynamics of HIV Infection in Blood
5. Discussion
- the spatial dynamics of cells and cytokines in LNs is considered in a 2D regular domain;
- the model is restricted to primary acute HIV infection and concomitant cytotoxic T cell responses;
- intracellular regulation of cell fate by multiple cytokine signaling is described via a hierarchy of activation thresholds;
- HIV infection is considered in LN and blood compartments.
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| HIV | Human Immunodeficiency Virus |
| LN | Lymph Mode |
| FRC | Fibroblastic Reticular Cell |
| APC | Antigen-Presenting Cell |
| IL-2 | Interleukin 2 |
| IFN | Type I Interferon |
| FasL | Fas Ligand |
References
- Levy, J.A. HIV and the Pathogenesis of AIDS, 3rd ed.; ASN Press: Washington, DC, USA, 2007. [Google Scholar]
- Grossman, Z.; Meier-Schellersheim, M.; Paul, W.E.; Picker, L.J. Pathogenesis of HIV infection: What the virus spares is as important as what it destroys. Nat. Med. 2006, 12, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Grossman, Z.; Meier-Schellersheim, M.; Sousa, A.E.; Victorino, R.M.; Paul, W.E. CD4+ T cell depletion in HIV infection: Are we closer to understanding the cause? Nat. Med. 2002, 8, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Chereshnev, V.A.; Bocharov, G.; Bazhan, S.; Bachmetyev, B.; Gainova, I.; Likhoshvai, V.; Argilaguet, J.M.; Martinez, J.P.; Rump, J.A.; Mothe, B.; et al. Pathogenesis and treatment of HIV infection: The cellular, the immune system and the neuroendocrine systems perspective. Int. Rev. Immunol. 2013, 32, 282–306. [Google Scholar] [CrossRef] [PubMed]
- Kent, S.J.; Reece, J.C.; Petravic, J.; Martyushev, A.; Kramski, M.; De Rose, R.; Cooper, D.A.; Kelleher, A.D.; Emery, S.; Cameron, P.U.; et al. The search for an HIV cure: Tackling latent infection. Lancet Infect. Dis. 2013, 13, 614–621. [Google Scholar] [CrossRef]
- Melkova, Z.; Shankaran, P.; Madlenakova, M.; Bodor, J. Current views on HIV-1 latency, persistence, and cure. Folia Microbiol. 2016, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kidd, B.A.; Peters, L.A.; Schadt, E.E.; Dudley, J.T. Unifying immunology with informatics and multiscale biology. Nat. Immunol. 2014, 15, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Carlson, J.M.; Le, A.Q.; Shahid, A.; Brumme, Z.L. HIV-1 adaptation to HLA: A window into virus-host immune interactions. Trends Microbiol. 2015, 23, 212–224. [Google Scholar] [CrossRef] [PubMed]
- Banks, H.T.; Davidian, M.; Hu, S.; Kepler, G.M.; Rosenberg, E.S. Modelling HIV immune response and validation with clinical data. J. Biol. Dyn. 2008, 2, 357–385. [Google Scholar] [CrossRef] [PubMed]
- Ludewig, B.; Stein, J.V.; Sharpe, J.; Cervantes-Barragan, L.; Thiel, V.; Bocharov, G. A global “imaging” view on systems approaches in immunology. Eur. J. Immunol. 2012, 42, 3116–3125. [Google Scholar] [CrossRef] [PubMed]
- Bocharov, G.; Chereshnev, V.; Gainova, I.; Bazhan, S.; Bachmetyev, B.; Argilaguet, J.; Martinez, J.; Meyerhans, A. Human Immunodeficiency Virus Infection: From Biological Observations to Mechanistic Mathematical Modelling. Math. Model. Nat. Phenom. 2012, 7, 78–104. [Google Scholar] [CrossRef]
- Alizon, S.; Magnus, C. Modelling the course of an HIV infection: Insights from ecology and evolution. Viruses 2012, 4, 1984–2013. [Google Scholar] [CrossRef] [PubMed]
- Canini, L.; Perelson, A.S. Viral kinetic modeling: State of the art. J. Pharmacokinet. Pharmacodyn. 2014, 41, 431–443. [Google Scholar] [CrossRef] [PubMed]
- Weinan, E. Principles of Multiscale Modelling; Cambridge University Press: Cambridge, UK, 2011. [Google Scholar]
- Fallahi-Sichani, M.; El-Kebir, M.; Marino, S.; Kirschner, D.E.; Linderman, J.J. Multi-scale computational modeling reveals a critical role for TNF receptor 1 dynamics in tuberculosis granuloma formation. J. Immunol. 2011, 186, 3472–3483. [Google Scholar] [CrossRef] [PubMed]
- Cilfone, N.A.; Kirschner, D.E.; Linderman, J.J. Strategies for efficient numerical implementation of hybrid multi-scale agent-based models to describe biological systems. Cell. Mol. Bioeng. 2015, 8, 119–136. [Google Scholar] [CrossRef] [PubMed]
- Simeone Marino, S.; Kirschner, D.E. A multi-compartment hybrid computational model predicts key roles for dendritic cells in tuberculosis infection. Computation 2016, 4, 39. [Google Scholar] [CrossRef]
- Prokopiou, S.A.; Barbarroux, L.; Bernard, S.; Mafille, J.; Leverrier, Y.; Arpin, C.; Marvel, J.; Gandrillon, O.; Crauste, F. Multiscale Modeling of the Early CD8 T-Cell Immune Response in Lymph Nodes: An Integrative Study. Computation 2014, 2, 159–181. [Google Scholar] [CrossRef]
- Gao, X.; Arpin, C.; Marvel, J.; Prokopiou, S.A.; Gandrillon, O.; Crauste, F. IL-2 sensitivity and exogenous IL-2 concentration gradient tune the productive contact duration of CD8(+) T cell-APC: A multiscale modeling study. BMC Syst. Biol. 2016, 10. [Google Scholar] [CrossRef] [PubMed]
- Williams, R.A.; Jon Timmis, J.; Qwarnstrom, E.E. Computational Models of the NF-κB Signaling Pathway. Computation 2014, 2, 131–158. [Google Scholar] [CrossRef]
- Baldazzi, V.; Paci, P.; Bernaschi, M.; Castiglione, F. Modeling lymphocyte homing and encounters in lymph nodes. BMC Bioinform. 2009, 10. [Google Scholar] [CrossRef] [PubMed]
- Gong, C.; Mattila, J.T.; Miller, M.; Flynn, J.L.; Linderman, J.J.; Kirschner, D. Predicting lymph node output efficiency using systems biology. J. Theor. Biol. 2013, 335, 169–184. [Google Scholar] [CrossRef] [PubMed]
- Palsson, S.; Hickling, T.P.; Bradshaw-Pierce, E.L.; Zager, M.; Jooss, K.; O’Brien, P.J.; Spilker, M.E.; Palsson, B.O.; Vicini, P. The development of a fully-integrated immune response model (FIRM) simulator of the immune response through integration of multiple subset models. BMC Syst. Biol. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Germain, R.; Meier-Schellersheim, M.; Nita-Lazar, A.; Fraser, I. Systems biology in immunology—A computational modeling perspective. Annu. Rev. Immunol. 2011, 29, 527–585. [Google Scholar] [CrossRef] [PubMed]
- Donovan, G.M.; Lythe, G. T cell and reticular network co-dependence in HIV infection. J. Theor. Biol. 2016, 395, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Lackner, A.A.; Lederman, M.M.; Rodriguez, B. HIV pathogenesis: The host. Cold Spring Harb. Perspect. Med. 2012, 2, a007005. [Google Scholar] [CrossRef] [PubMed]
- McMichael, A.; Dorrell, L. The immune response to HIV. Medicine 2009, 37, 321–325. [Google Scholar] [CrossRef]
- Walker, B.; McMichael, A. The T cell response to HIV. Cold Spring Harb. Perspect. Med. 2012, 2, a007054. [Google Scholar] [CrossRef] [PubMed]
- Ndhlovu, Z.M.; Kamya, P.; Mewalal, N.; Kløverpris, H.N.; Nkosi, T.; Pretorius, K.; Laher, F.; Ogunshola, F.; Chopera, D.; Shekhar, K.; et al. Magnitude and Kinetics of CD8+ T Cell Activation during Hyperacute HIV Infection Impact Viral Set Point. Immunity 2015, 43, 591–604. [Google Scholar] [CrossRef] [PubMed]
- Petrovas, C.; Mueller, Y.M.; Katsikis, P.D. Apoptosis of HIV-specific CD8+ T cells: An HIV evasion strategy. Cell Death Differ. 2005, 12, 859–870. [Google Scholar] [CrossRef] [PubMed]
- Bessonov, N.; Eymard, N.; Kurbatova, P.; Volpert, V. Mathematical modeling of erythropoiesis in vivo with multiple erythroblastic islands. Appl. Math. Lett. 2012, 25, 1217–1221. [Google Scholar] [CrossRef]
- Fischer, S.; Kurbatova, P.; Bessonov, N.; Gandrillon, O.; Volpert, V.; Crauste, F. Modelling erythroblastic islands: Using a hybrid model to assess the function of central macrophage. J. Theor. Biol. 2012, 298, 92–106. [Google Scholar] [CrossRef] [PubMed]
- Kurbatova, P.; Bernard, S.; Bessonov, N.; Crauste, F.; Demin, I.; Dumontet, C.; Fischer, S.; Volpert, V. Hybrid model of erythropoiesis and leukemia treatment with cytosine arabinoside. SIAM J. Appl. Math. 2011, 71, 2246–2268. [Google Scholar] [CrossRef]
- Volpert, V.; Bessonov, N.; Eymard, N.; Tosenberger, A. Modèle multi-échelle de la dynamique cellulaire. In Le Vivant Discret et Continu; Glade, N., Stephanou, A., Eds.; Editions Materiologiques: Paris, France, 2013. [Google Scholar]
- Kurbatova, P.; Eymard, N.; Volpert, V. Hybrid Model of Erythropoiesis. Acta Biotheor. 2013, 61, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Eymard, N.; Bessonov, N.; Gandrillon, O.; Koury, M.J.; Volpert, V. The role of spatial organization of cells in erythropoiesis. J. Math. Biol. 2015, 70, 71–97. [Google Scholar] [CrossRef] [PubMed]
- Yeghiazarian, L.; Cumberland, W.G.; Yang, O.O. A stochastic multi-scale model of HIV-1 transmission for decision-making: Application to a MSM population. PLoS ONE 2013, 8, e70578. [Google Scholar] [CrossRef] [PubMed]
- Stéphanou, A.; Volpert, V. Hybrid modeling in biology: A classification review. Math. Model. Nat. Phenom. 2016, 11, 37–48. [Google Scholar] [CrossRef]
- Gottschalk, R.A.; Martins, A.J.; Angermann, B.R.; Dutta, B.; Ng, C.E.; Uderhardt, S.; Tsang, J.S.; Fraser, I.D.; Meier-Schellersheim, M.; Germain, R.N. Distinct NFkB and MAPK Activation Thresholds Uncouple Steady-State Microbe Sensing from Anti-pathogen Inflammatory Responses. Cell Syst. 2016, 2, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Nakaoka, S.; Iwami, S.; Sato, K. Dynamics of HIV infection in lymphoid tissue network. J. Math. Biol. 2016, 72, 909–938. [Google Scholar] [CrossRef] [PubMed]
- Stancevic, O.; Angstmann, C.N.; Murray, J.M.; Henry, B.I. Turing patterns from dynamics of early HIV infection. Bull. Math. Biol. 2013, 7, 774–795. [Google Scholar] [CrossRef] [PubMed]
- Dunia, R.; Bonnecaze, R. Mathematical modeling of viral infection dynamics in spherical organs. J. Math. Biol. 2013, 67, 1425–1455. [Google Scholar] [CrossRef] [PubMed]
- Strain, M.C.; Richman, D.D.; Wong, J.K.; Levine, H. Spatiotemporal dynamics of HIV propagation. J. Theor. Biol. 2002, 218, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhou, S.; Groppelli, E.; Pellegrino, P.; Williams, I.; Borrow, P.; Chain, B.M.; Jolly, C. Hybrid spreading mechanisms and T cell activation shape the dynamics of HIV-1 infection. PLoS Comput. Biol. 2015, 11, e1004179. [Google Scholar] [CrossRef] [PubMed]
- Bocharov, G.; Züst, R.; Cervantes-Barragan, L.; Luzyanina, T.; Chiglintsev, E.; Chereshnev, V.A.; Thiel, V.; Ludewig, B. A Systems Immunology Approach to Plasmacytoid Dendritic Cell Function in Cytopathic Virus Infections. PLoS Pathog. 2010, 6, e1001017. [Google Scholar] [CrossRef] [PubMed]
- Webb, S.D.; Sherratt, J.A.; Fish, R.G. Cells behaving badly: A theoretical model for the Fas/FasL system in tumour immunology. Math. Biosci. 2002, 179, 113–129. [Google Scholar] [CrossRef]
- Baker, C.T.H.; Bocharov, G.A.; Paul, C.A.H. Mathematical Modelling of the Interleukin-2 T-Cell System: A Comparative Study of Approaches Based on Ordinary and Delay Differential Equation. J. Theor. Med. 1997, 1, 117–128. [Google Scholar] [CrossRef]
- Grimm, V.; Berger, U.; Bastiansen, F.; Eliassen, S.; Ginot, V.; Giske, J.; Goss-Custard, J.; Grand, T.; Heinz, S.K.; Huse, G.; et al. A standard protocol for describing individual-based and agent-based models. Ecol. Model. 2006, 198, 115–126. [Google Scholar] [CrossRef]
- Grimm, V.; Berger, U.; DeAngelis, D.L.; Polhill, J.G.; Giske, J.; Railsback, S.F. The ODD protocol: A review and first update. Ecol. Model. 2010, 221, 2760–2768. [Google Scholar] [CrossRef]
- Bouchnita, A.; Bocharov, G.; Meyerhans, A.; Volpert, V. Hybrid approach to model the spatial regulation of T cell responses. BMC Immunol. 2016. accepted. [Google Scholar]
- Ananworanich, J.; Chomont, N.; Eller, L.A.; Kroon, E.; Tovanabutra, S.; Bose, M.; Nau, M.; Fletcher, J.L.; Tipsuk, S.; Vandergeeten, C.; et al. HIV DNA Set Point is Rapidly Established in Acute HIV Infection and Dramatically Reduced by Early ART. EBioMedicine 2016, 11, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Ananworanich, J.; Sacdalan, C.P.; Pinyakorn, S.; Chomont, N.; de Souza, M.; Luekasemsuk, T.; Schuetz, A.; Krebs, S.J.; Dewar, R.; Jagodzinski, L.; et al. Virological and immunological characteristics of HIV-infected individuals at the earliest stage of infection. J. Virus Erad. 2016, 2, 43–48. [Google Scholar] [PubMed]
- Grossman, Z.; Polis, M.; Feinberg, M.B.; Grossman, Z.; Levi, I.; Jankelevich, S.; Yarchoan, R.; Boon, J.; de Wolf, F.; Lange, J.M.; et al. Ongoing HIV dissemination during HAART. Nat. Med. 1999, 5, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- Likhoshvai, V.A.; Khlebodarova, T.M.; Bazhan, S.I.; Gainova, I.A.; Chereshnev, V.A.; Bocharov, G.A. Mathematical model of the Tat-Rev regulation of HIV-1 replication in an activated cell predicts the existence of oscillatory dynamics in the synthesis of viral components. BMC Genom. 2014, 15. [Google Scholar] [CrossRef] [PubMed]
- Petrovas, C.; Yamamoto, T.; Price, D.A.; Rao, S.S.; Klatt, N.R.; Brenchley, J.M.; Douek, D.C.; Gostick, E.; Angermann, B.R.; Grossman, Z.; et al. High production rates sustain in vivo levels of PD-1high simian immunodeficiency virus-specific CD8 T cells in the face of rapid clearance. J. Virol. 2013, 87, 9836–9844. [Google Scholar] [CrossRef] [PubMed]
- Peligero, C.; Argilaguet, J.; Güerri-Fernandez, R.; Torres, B.; Ligero, C.; Colomer, P.; Plana, M.; Knobel, H.; García, F.; Meyerhans, A. PD-L1 Blockade Differentially Impacts Regulatory T Cells from HIV-Infected Individuals Depending on Plasma Viremia. PLoS Pathog. 2015, 11, e1005270. [Google Scholar] [CrossRef] [PubMed]
- Grossman, Z.; Paul, W.E. Dynamic tuning of lymphocytes: Physiological basis, mechanisms, and function. Annu. Rev. Immunol. 2015, 33, 677–713. [Google Scholar] [CrossRef] [PubMed]
- Paul, W.E.; Grossman, Z. Pathogen-sensing and regulatory T cells: Integrated regulators of immune responses. Cancer Immunol. Res. 2014, 2, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Bocharov, G.A.; Telatnikov, I.S.; Chereshnev, V.A.; Martinez, J.; Meyerhans, A. Mathematical modeling of the within-host HIV quasispecies dynamics in response to antiviral treatment. Russ. J. Numer. Anal. Math. Model. 2015, 30, 157–170. [Google Scholar] [CrossRef]
- Okoye, A.; Meier-Schellersheim, M.; Brenchley, J.M.; Hagen, S.I.; Walker, J.M.; Rohankhedkar, M.; Lum, R.; Edgar, J.B.; Planer, S.L.; Legasse, A.; et al. Progressive CD4+ central memory T cell decline results in CD4+ effector memory insufficiency and overt disease in chronic SIV infection. J. Exp. Med. 2007, 204, 2171–2185. [Google Scholar] [CrossRef] [PubMed]
- Paul, W.E.; Milner, J.D.; Grossman, Z. Pathogen-sensing, regulatory T cells, and responsiveness-tuning collectively regulate foreign- and self-antigen mediated T cell responses. Cold Spring Harb. Symp. Quant. Biol. 2013, 78, 265–276. [Google Scholar] [CrossRef] [PubMed]







© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouchnita, A.; Bocharov, G.; Meyerhans, A.; Volpert, V. Towards a Multiscale Model of Acute HIV Infection. Computation 2017, 5, 6. https://doi.org/10.3390/computation5010006
Bouchnita A, Bocharov G, Meyerhans A, Volpert V. Towards a Multiscale Model of Acute HIV Infection. Computation. 2017; 5(1):6. https://doi.org/10.3390/computation5010006
Chicago/Turabian StyleBouchnita, Anass, Gennady Bocharov, Andreas Meyerhans, and Vitaly Volpert. 2017. "Towards a Multiscale Model of Acute HIV Infection" Computation 5, no. 1: 6. https://doi.org/10.3390/computation5010006
APA StyleBouchnita, A., Bocharov, G., Meyerhans, A., & Volpert, V. (2017). Towards a Multiscale Model of Acute HIV Infection. Computation, 5(1), 6. https://doi.org/10.3390/computation5010006






