Deep Learning Models for Colorectal Polyps
Abstract
1. Introduction
2. Background
3. Materials and Methods
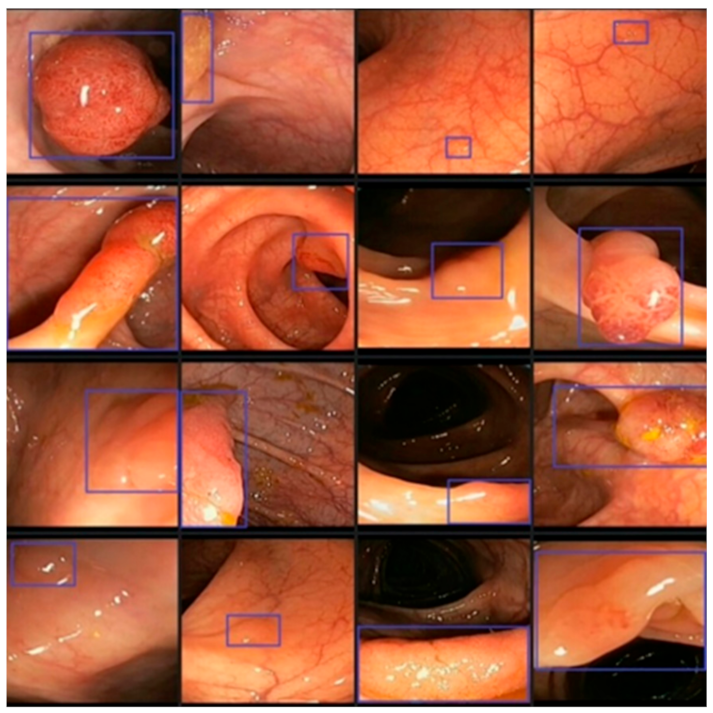
3.1. Databases
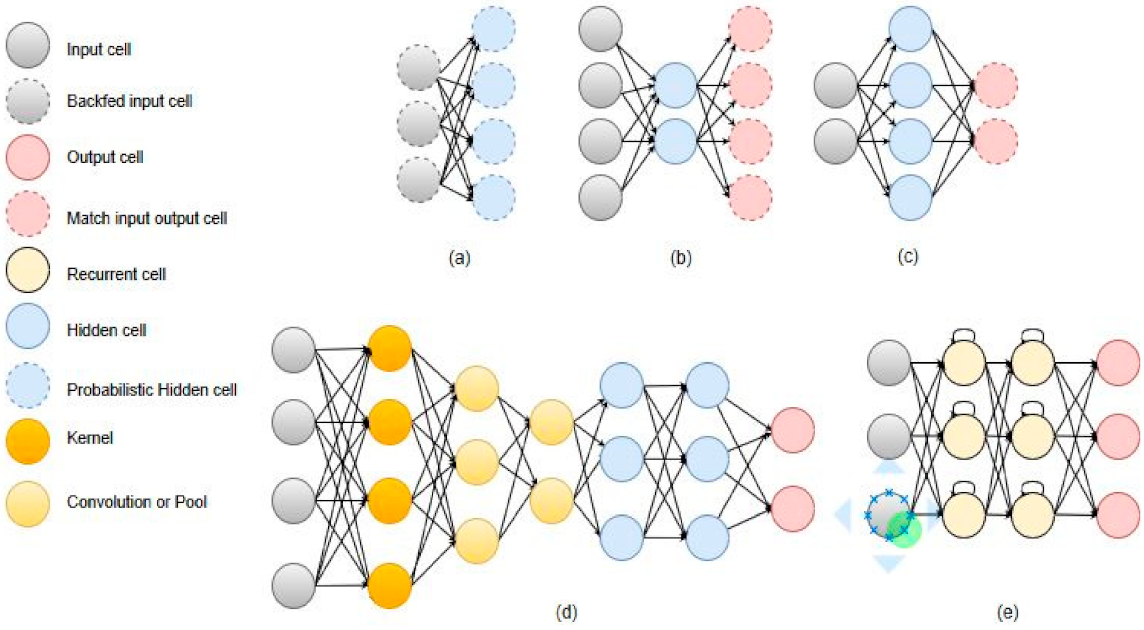
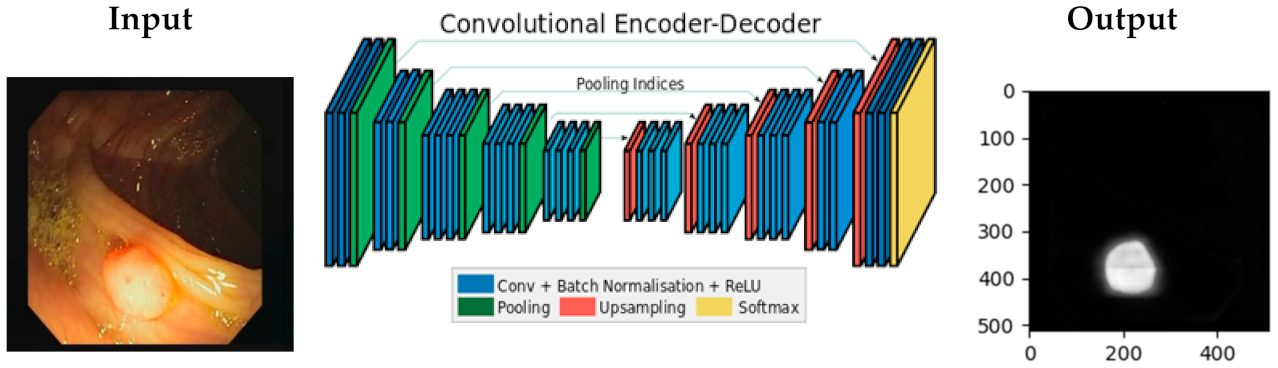
3.2. The Proposed Model
- use the same layer for the non-shrinking convolution layer.
- use transposed deconvolution for the shrinking convolution layer adjusted with the same parameters.
- use the nearest neighbor upsampling for the max_pooling layer.
- Crop—parameter: px = (0, 16) which crops images from each side by 0 to 16 pixels chosen randomly.
- Fliplr—parameter: 0.5 which flips horizontally 50% of all images.
- Flipud—parameter: 0.5 which flips vertically 50% of all images.
- GaussianBlur—parameter: (0, 3.0), blurs each image with varying strength using gaussian blur (sigma between 0 and 3.0).
- Dropout—parameter: (0.02, 0.1), drop randomly 2 to 10% of all pixels (i.e., set them to black).
- AdditiveGaussianNoise—parameter: scale = 0.01*255, adds white noise pixel by pixel to images.
- Affine—parameter: translate_px = {“x”: (-network.IMAGE_HEIGHT // 3, network.IMAGE_WIDTH // 3)}, applies translate/move of images (affine transformation).
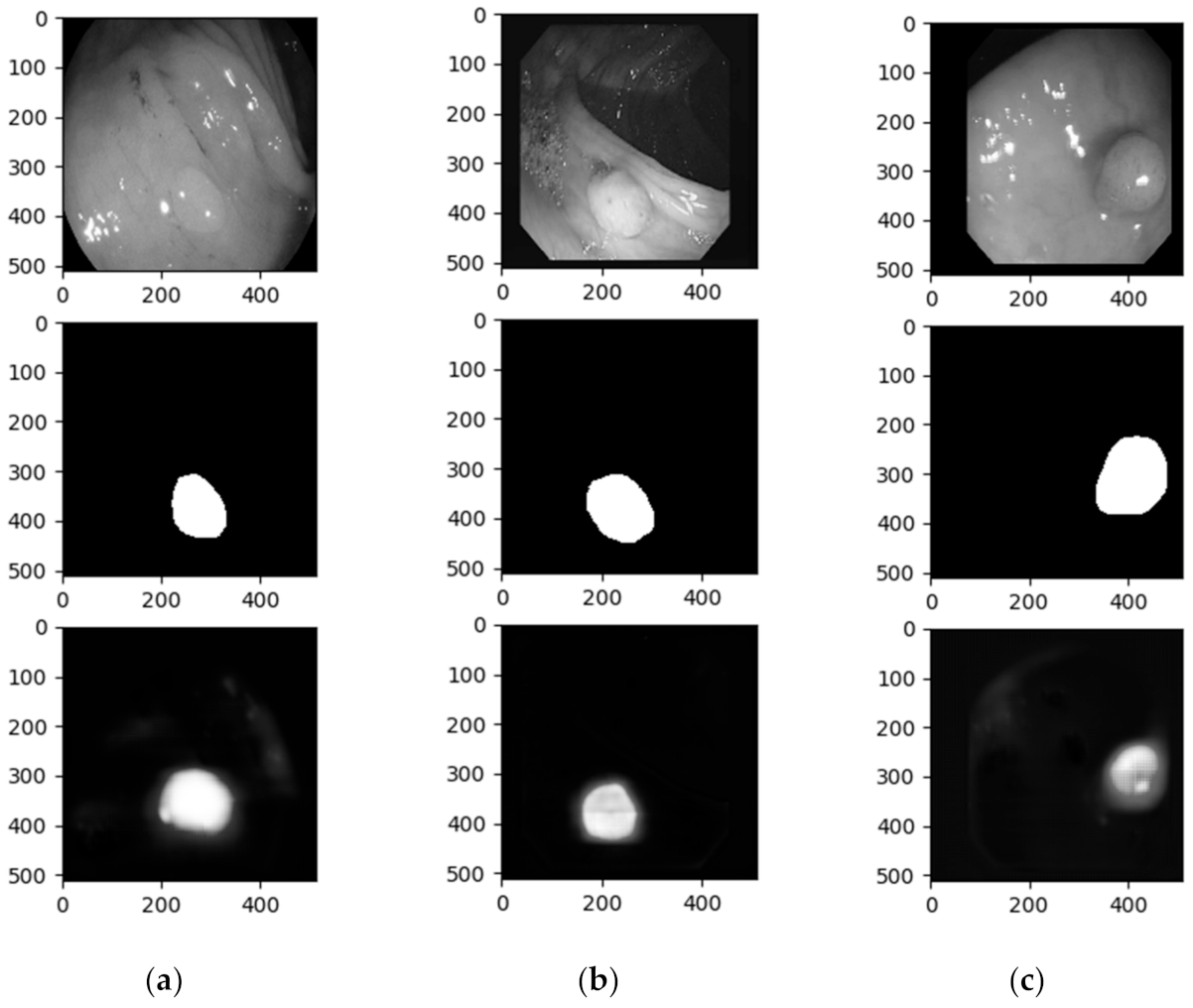
4. Results
5. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Esteva, A.; Robicquet, A.; Ramsundar, B.; Kuleshov, V.; DePristo, M.; Chou, K.; Cui, C. A guide to deep learning in healthcare. Nat. Med. 2019, 25, 24–29. [Google Scholar] [CrossRef]
- Litjens, G.; Kooi, T.; Bejnordi, B.E.; Setio, A.A.A.; Ciompi, F.; Ghafoorian, M.; Van Der Laak, J.A.; Van Ginneken, B.; Sánchez, C.I. A survey on deep learning in medical image analysis. Med. Image Anal. 2017, 42, 60–88. [Google Scholar] [CrossRef]
- World Health Organization and International Agency for Research on Cancer. Cancer Today. 2020. Available online: https://bit.ly/37jXYER (accessed on 17 November 2020).
- Lieberman, D. Quality and colonoscopy: A new imperative. Gastrointest. Endosc. 2005, 61, 392–394. [Google Scholar] [CrossRef]
- Carioli, G.; Bertuccio, P.; Boffetta, P.; Levi, F.; La Vecchia, C.; Negri, E.; Malvezzi, M. European cancer mortality predictions for the year 2020 with a focus on prostate cancer. Ann. Oncol. 2020, 31, 650–658. [Google Scholar] [CrossRef]
- Zauber, A.G. The Impact of Screening on Colorectal Cancer Mortality and Incidence: Has It Really Made a Difference? Dig. Dis. Sci. 2015, 60, 681–691. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Lee, J.; Park, S.W.; Kim, Y.S.; Lee, K.J.; Sung, H.; Song, P.H.; Yoon, W.J.; Moon, J.S. Risk factors of missed colorectal lesions after colonoscopy. Medicine 2017, 96, e7468. [Google Scholar] [CrossRef]
- Bonnington, S.N.; Rutter, M.D. Surveillance of colonic polyps: Are we getting it right? World J. Gastroenterol. 2016, 22, 1925–1934. [Google Scholar] [CrossRef]
- Tajbakhsh, N.; Shin, J.Y.; Gurudu, S.R.; Hurst, R.T.; Kendall, C.B.; Gotway, M.B.; Liang, J. Convolutional Neural Networks for Medical Image Analysis: Full Training or Fine Tuning? IEEE Trans. Med. Imaging 2016, 35, 1299–1312. [Google Scholar] [CrossRef] [PubMed]
- Poon, C.C.; Jiang, Y.; Zhang, R.; Lo, W.W.; Cheung, M.S.; Yu, R.; Zheng, Y.; Wong, J.C.; Liu, Q.; Wong, S.H.; et al. AI-doscopist: A real-time deep-learning-based algorithm for localising polyps in colonoscopy videos with edge computing devices. NPJ Digit. Med. 2020, 3, 73. [Google Scholar]
- Ribeiro, E.; Uhl, A.; Häfner, M. Colonic polyp classification with convolutional neural networks. In Proceedings of the IEEE 29th International Symposium on Computer-Based Medical Systems (CBMS), Dublin, Ireland, 20–24 June 2016. [Google Scholar]
- Ribeiro, E.; Häfner, M.; Wimmer, G.; Tamaki, T.; Tischendorf, J.J.; Yoshida, S.; Tanaka, S.; Uhl, A. Exploring texture transfer learning for colonic polyp classification via convolutional neural networks. In Proceedings of the 2017 IEEE 14th International Symposium on Biomedical Imaging (ISBI 2017), Melbourne, Australia, 18–21 April 2017. [Google Scholar]
- Ribeiro, E.; Uhl, A.; Wimmer, G.; Häfner, M. Exploring Deep Learning and Transfer Learning for Colonic Polyp Classification. Comput. Math. Methods Med. 2016, 2016, 6584725. [Google Scholar] [CrossRef]
- Zhang, R.; Zheng, Y.; Mak, T.W.C.; Yu, R.; Wong, S.H.; Lau, J.Y.; Poon, C.C. Automatic Detection and Classification of Colorectal Polyps by Transferring Low-Level CNN Features from Nonmedical Domain. IEEE J. Biomed. Health Inform. 2017, 21, 41–47. [Google Scholar] [CrossRef]
- Shin, Y.; Qadir, H.A.; Aabakken, L.; Bergsland, J.; Balasingham, I. Automatic Colon Polyp Detection Using Region Based Deep CNN and Post Learning Approaches. IEEE Access 2018, 6, 40950–40962. [Google Scholar] [CrossRef]
- Shin, Y.; Balasingham, I. Comparison of hand-craft feature based SVM and CNN based deep learning framework for automatic polyp classification. In Proceedings of the 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Jeju Island, Korea, 11–15 July 2017. [Google Scholar]
- Korbar, B.; Olofson, A.M.; Miraflor, A.P.; Nicka, C.M.; Suriawinata, M.A.; Torresani, L.; Suriawinata, A.A.; Hassanpour, S. Deep Learning for Classification of Colorectal Polyps on Whole-slide Images. J. Pathol. Inform. 2017, 8, 30. [Google Scholar] [PubMed]
- Mahmood, F.; Durr, N.J. Deep learning and conditional random fields-based depth estimation and topographical reconstruction from conventional endoscopy. Med. Image Anal. 2018, 48, 230–243. [Google Scholar] [CrossRef]
- Carneiro, G.; Pu, L.Z.C.T.; Singh, R.; Burt, A. Deep learning uncertainty and confidence calibration for the five-class polyp classification from colonoscopy. Med. Image Anal. 2020, 62, 101653. [Google Scholar] [CrossRef]
- Gao, J.; Guo, Y.; Sun, Y.; Qu, G. Application of Deep Learning for Early Screening of Colorectal Precancerous Lesions under White Light Endoscopy. Comput. Math. Methods Med. 2020, 2020, 8374317. [Google Scholar] [CrossRef]
- Song, E.M.; Park, B.; Ha, C.A.; Hwang, S.W.; Park, S.H.; Yang, D.H.; Ye, B.D.; Myung, S.J.; Yang, S.K.; Kim, N.; et al. Endoscopic diagnosis and treatment planning for colorectal polyps using a deep-learning model. Sci. Rep. 2020, 10, 30. [Google Scholar] [CrossRef] [PubMed]
- Urban, G.; Tripathi, P.; Alkayali, T.; Mittal, M.; Jalali, F.; Karnes, W.; Baldi, P. Deep Learning Localizes and Identifies Polyps in Real Time With 96% Accuracy in Screening Colonoscopy. Gastroenterology 2018, 155, 1069–1078.e8. [Google Scholar] [CrossRef] [PubMed]
- Misawa, M.; Kudo, S.E.; Mori, Y.; Cho, T.; Kataoka, S.; Yamauchi, A.; Ogawa, Y.; Maeda, Y.; Takeda, K.; Ichimasa, K.; et al. Artificial Intelligence-Assisted Polyp Detection for Colonoscopy: Initial Experience. Gastroenterology 2018, 154, 2027–2029.e3. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.K.; Yildirim, S.; Farup, I.; Pederse, M.; Hovde, O. Y-Net: A deep Convolutional Neural Network for Polyp Detection. 2018. Available online: http://arxiv.org/abs/1806.01907 (accessed on 10 June 2020).
- Yu, L.; Chen, H.; Dou, Q.; Qin, J.; Heng, P.A. Integrating Online and Offline Three-Dimensional Deep Learning for Automated Polyp Detection in Colonoscopy Videos. IEEE J. Biomed. Health Inform. 2017, 21, 65–75. [Google Scholar] [CrossRef]
- Byrne, M.F.; Chapados, N.; Soudan, F.; Oertel, C.; Pérez, M.L.; Kelly, R.; Iqbal, N.; Chandelier, F.; Rex, D.K. Real-time differentiation of adenomatous and hyperplastic diminutive colorectal polyps during analysis of unaltered videos of standard colonoscopy using a deep learning model. Gut 2019, 68, 94. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Liu, X.; Berzin, T.M.; Brown, J.R.G.; Liu, P.; Zhou, C.; Lei, L.; Li, L.; Guo, Z.; Lei, S.; et al. Effect of a deep-learning computer-aided detection system on adenoma detection during colonoscopy (CADe-DB trial): A double-blind randomised study. Lancet Gastroenterol. Hepatol. 2020, 5, 343–351. [Google Scholar] [CrossRef]
- Yamada, M.; Saito, Y.; Imaoka, H.; Saiko, M.; Yamada, S.; Kondo, H.; Takamaru, H.; Sakamoto, T.; Sese, J.; Kuchiba, A.; et al. Development of a real-time endoscopic image diagnosis support system using deep learning technology in colonoscopy. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bernal, J.; Sánchez, J.; Vilariño, F. Towards automatic polyp detection with a polyp appearance model. Pattern Recognit. 2012, 45, 3166–3182. [Google Scholar] [CrossRef]
- Bernal, J.; Sánchez, F.J.; Fernández-Esparrach, G.; Gil, D.; Rodríguez, C.; Vilariño, F. WM-DOVA maps for accurate polyp highlighting in colonoscopy: Validation vs. saliency maps from physicians. Comput. Med Imaging Graph. 2015, 43, 99–111. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.; Histace, A.; Romain, O.; Dray, X.; Granado, B. Toward embedded detection of polyps in WCE images for early diagnosis of colorectal cancer. Int. J. Comput. Assist. Radiol. Surg. 2014, 9, 283–293. [Google Scholar] [CrossRef]
- Abadi, M.; Barham, P.; Chen, J.; Chen, Z.; Davis, A.; Dean, J.; Devin, M.; Ghemawat, S.; Irving, G.; Isard, M.; et al. TensorFlow: A system for large-scale machine learning. In Proceedings of the 12th USENIX Conference on Operating Systems Design and Implementation, Savannah, GA, USA, 2–4 November 2016; pp. 265–283. [Google Scholar]
- Badrinarayanan, V.; Kendall, A.; Cipolla, R. SegNet: A Deep Convolutional Encoder-Decoder Architecture for Image Segmentation. IEEE Trans. Pattern Anal. Mach. Intell. 2017, 39, 2481–2495. [Google Scholar] [CrossRef]
- Jung, A. imgaug 0.2.5 [Internet]. 2017. Available online: http://imgaug.readthedocs.io/en/latest (accessed on 10 June 2020).


| Year | Authors | Nr of images | Format | Objective | Network | Metrics | Datasets | Novelties |
|---|---|---|---|---|---|---|---|---|
| 2016 | Yu et al. [26] | Train: 1.1 M non-med Test: 20 | Video | Detection | 3D-FCN | F1 = 78.6%, F2 = 73.9% | Asu-Mayo Clinic Polyp Database | An integrated framework with online and offline 3D representation learning |
| 2017 | Byrne et al. [27] | Train: 223 Test: 125 | Video | Detection | DCNN based on inception network architecture | Accu = 94%, Sens = 98%, Spec = 83%, NPV = 97%, PPV = 90% | Private dataset | AI differentiating diminutive adenomas from hyperplastic polyps on unaltered videos of colon polyps. The model operates in quasi-real-time |
| 2017 | Shin & Balasingham [17] | Train: 1525 Test: 366 | Image | Classification | HOG + SVM, Combined feature + SVM, CNN (gray), CNN(RGB) | Accu = 91.3%, Sens = 90.8%, Spec = 91.8%, Prec = 92.7% | CVC-Clinic, ETIS-Larib, Asu-Mayo | Compare handcraft feature based SVM method and CNN method for polyp image frame classification |
| 2017 | Korbar et al. [18] | Train: 2074 crop images Test: 239 full images | Image | Classification | AlexNet8, VGG19, GoogleNet22, ResNet50, ResNet101, ResNet152, ResNet152 | Accu = 93.0%, Prec = 89.7%, Rec = 88.3%, F1 = 88.8% | Private dataset | Identify polyps and their types on whole-slide images by breaking them into smaller, overlapping patches |
| 2018 | Mahmood & Durr [19] | Synthetic colon: 100,000 Phantom colon: 100,000 Porcine colon: 1460 | Image | Detection | CNN + CRF | RE = 0.242 | synthetic data, real endoscopy images from a porcine colon | Synthetically generated endoscopy images |
| 2018 | Urban et al. [23] | Train: 8641 images Test: 20 videos | Image/Video | Detection | CNN | Accu = 96.4%, AUC ROC = 0.991 | Private dataset | Localization model by optimizing the size and location, optimizing the Dice loss, and a variation of the “you only look once” algorithm (“internal ensemble”) |
| 2019 | Yamada et al. [29] | Train: 4840 images Test: 77 videos | Image/Video | Detection | Faster R-CNN + VGG16 | Sens = 97.3%, Spec = 99.0%, ROC = 0.975 | Private dataset | Included 5000 images of more than 2000 lesions, and 3000 images of more than 500 non-polypoid superficial lesions It is nearly real-time processing |
| 2020 | Carneiro et al. [20] | 940 | Image | Classification | ResNet-101 & DenseNet-121 | Accu = 51%, Avg Prec = 48% (Z = 0.7) | Private dataset (Australian & Japanese) | Deep learning classifier using classification uncertainty and calibrated confidence to reject the classification of test samples |
| 2020 | Gao et al. [21] | 3413 | Image | Detection + Classification | AlexNet, VGG19, ResNet18, GoogLeNet, ResNet50, Mask R-CNN | Accu = 93.0%, Sens = 94.3%, Spec = 90.6% | Private dataset | Detection and classification models based on white light endoscopic images |
| 2020 | Poon et al. [11] | Pre-trained: 1.2 M non-med images Fine-tuned: 291,090 polyp & non-med images Test: 144 videos | Video | Localizing | ResNet50 + YOLOv2 + a temporal tracking algorithm | Sens = 96.9%, Spec = 93.3% | CVC-ColonDB, CVC-ClinicDB, ETIS-LaribDB, AsuMayoDB, CU-ColonDB, ACP-ColonDB, Selected Google Images | Real-time AI algorithm for localizing polyps in colonoscopy videos, using different medical and non-medical datasets for training |
| 2020 | Song et al. [22] | Train: 12,480 image patches of 624 polyps Test: two DBs of 545 polyp images | Image | Classification | CAD based on NBI near-focus images + ResNet-50, DenseNet-201 | Accu = 82.4% | Private dataset | A CAD system for predicting CR polyp histology using near-focus narrow-band imaging (NBI) pictures and deep-learning technology |
| 2020 | Wang et al. [28] | CADe group: 484 patients non-CADe group: 478 patients | Video | Detection | CAD + AI | ADR = 34% | Private dataset | The first double-blind, randomized controlled trial to assess the effectiveness of automatic polyp detection using a CADe system during colonoscopy. |
| Datasets | Nr of Images |
|---|---|
| CVC-ColonDB [30] | 380 |
| CVC-ClinicDB [31] | 612 |
| ETIS-LaribPolypDB [32] | 196 |
| Datasets | Best Accuracy | Batch | Total Time |
|---|---|---|---|
| ETIS-LaribPolypDB | 0.967 | 1300 | 1120.48 |
| CVC-ClinicDB | 0.951 | 2200 | 2186.97 |
| CVC-ColonDB | 0.937 | 2000 | 3659.52 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bardhi, O.; Sierra-Sosa, D.; Garcia-Zapirain, B.; Bujanda, L. Deep Learning Models for Colorectal Polyps. Information 2021, 12, 245. https://doi.org/10.3390/info12060245
Bardhi O, Sierra-Sosa D, Garcia-Zapirain B, Bujanda L. Deep Learning Models for Colorectal Polyps. Information. 2021; 12(6):245. https://doi.org/10.3390/info12060245
Chicago/Turabian StyleBardhi, Ornela, Daniel Sierra-Sosa, Begonya Garcia-Zapirain, and Luis Bujanda. 2021. "Deep Learning Models for Colorectal Polyps" Information 12, no. 6: 245. https://doi.org/10.3390/info12060245
APA StyleBardhi, O., Sierra-Sosa, D., Garcia-Zapirain, B., & Bujanda, L. (2021). Deep Learning Models for Colorectal Polyps. Information, 12(6), 245. https://doi.org/10.3390/info12060245








