Critical In-Water Habitats for Post-Nesting Sea Turtles from the Southern Gulf of Mexico
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Turtle Tracking and Data Processing
2.3. Critical Habitats Delineation
2.4. Physical Features of Critical Habitats
2.5. Movement and Aggregation Patterns
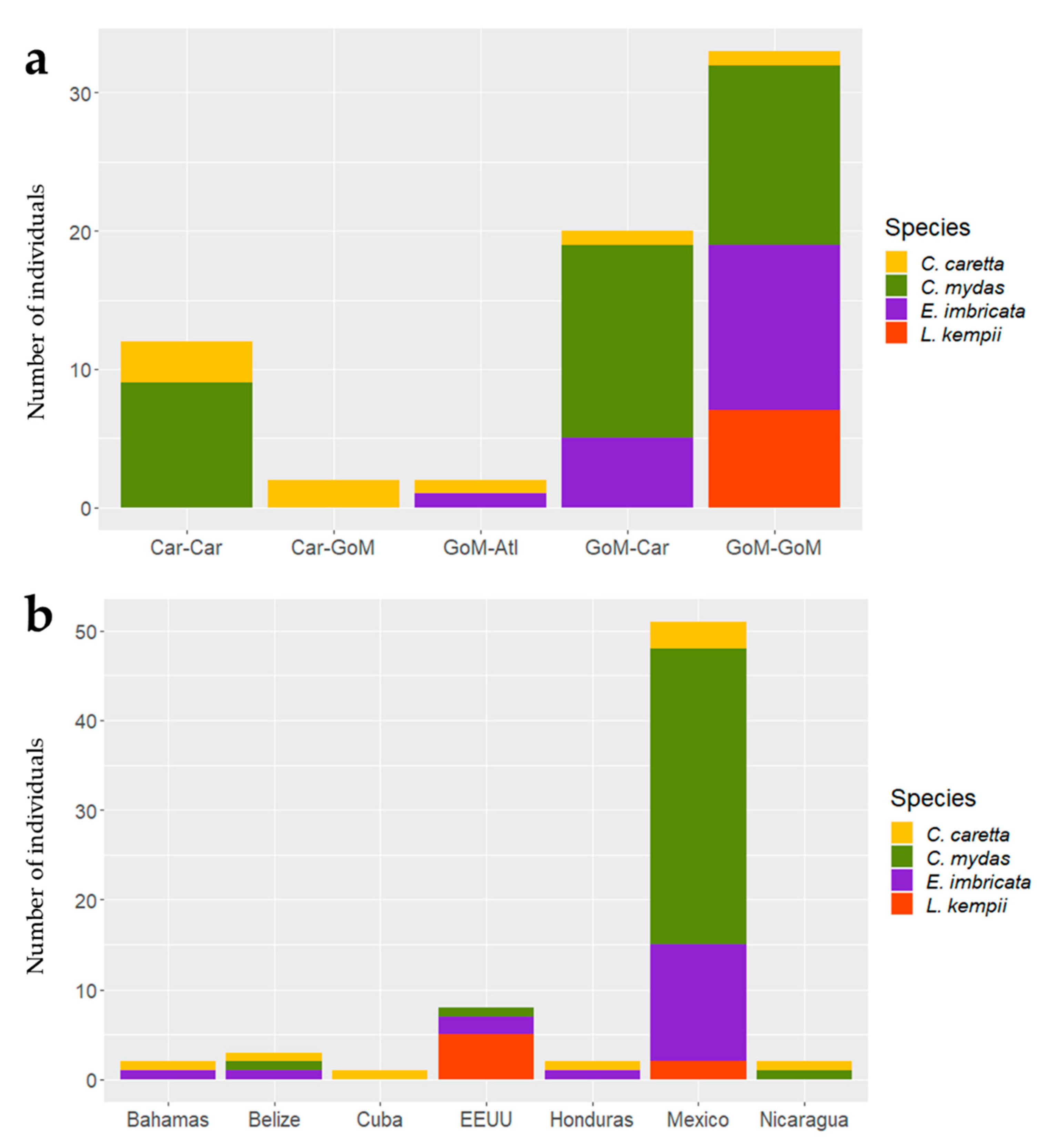
3. Results
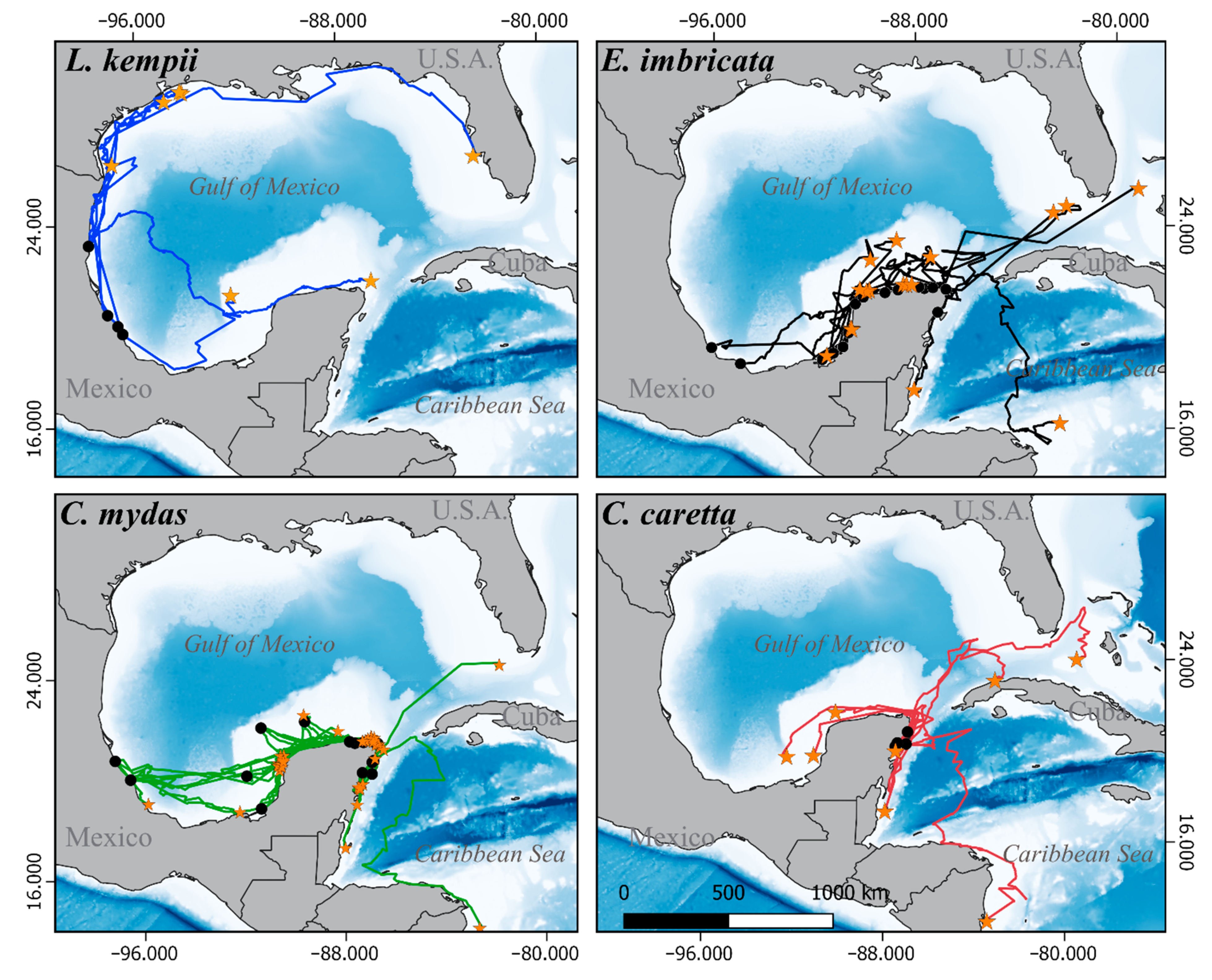
3.1. Satellite Tracking
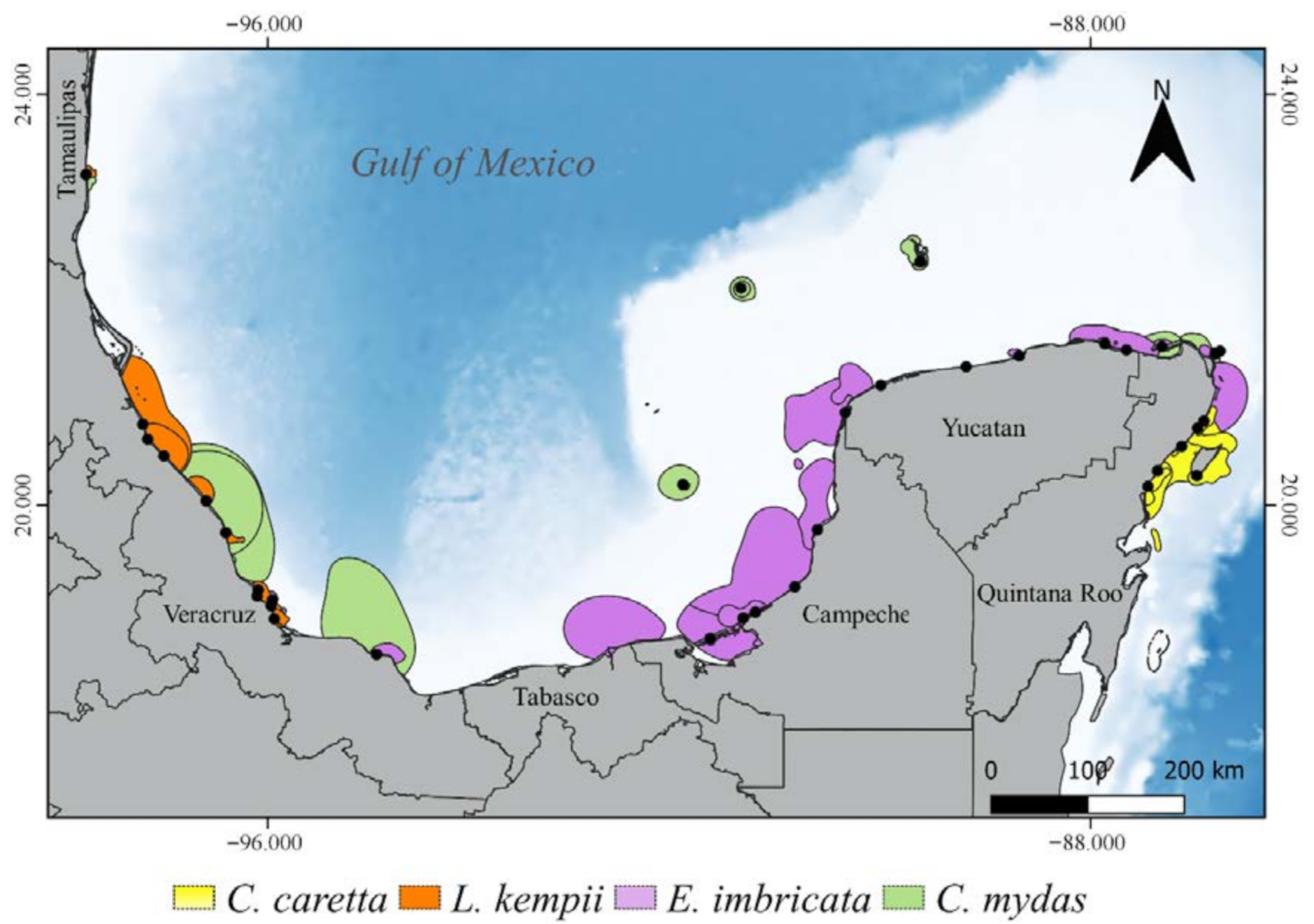
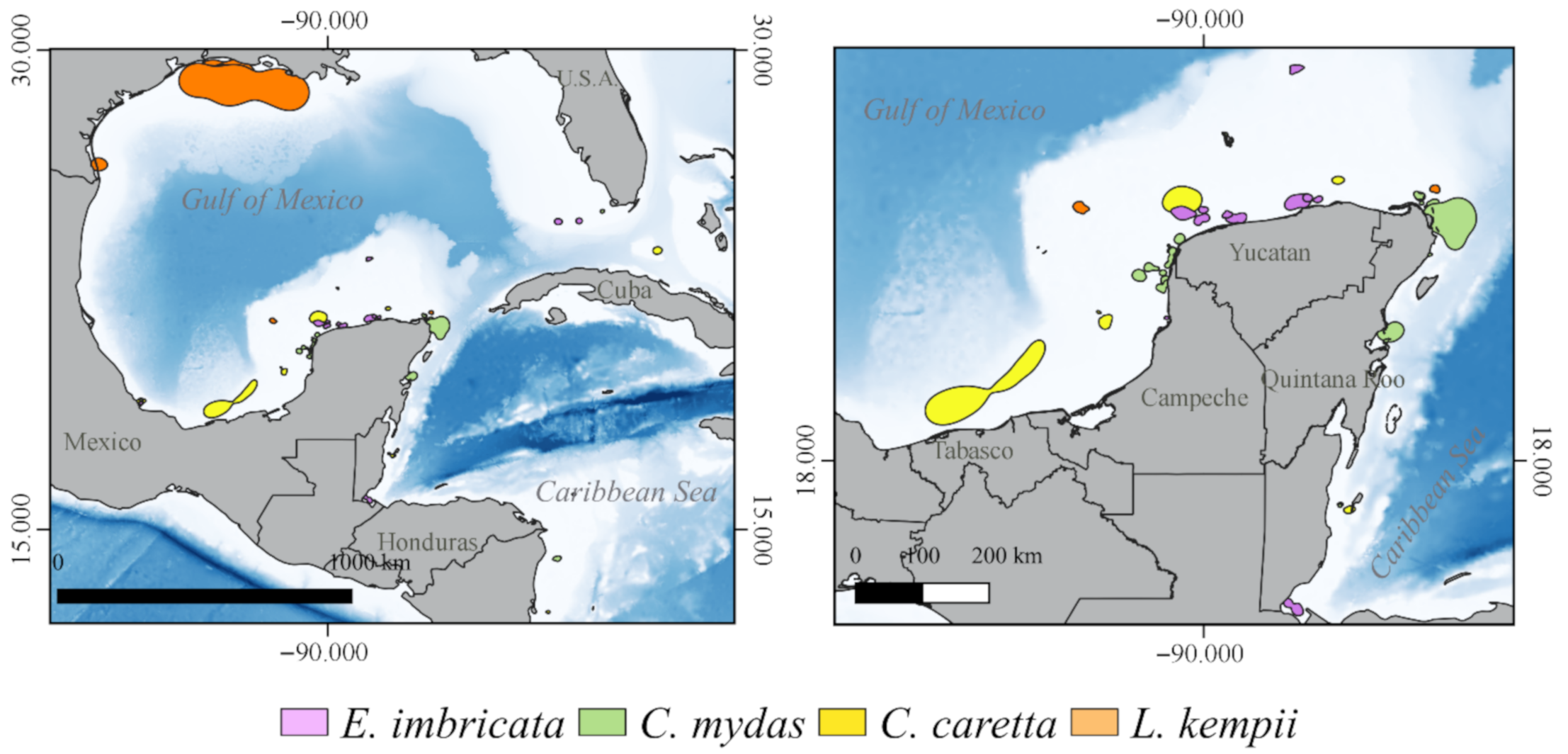
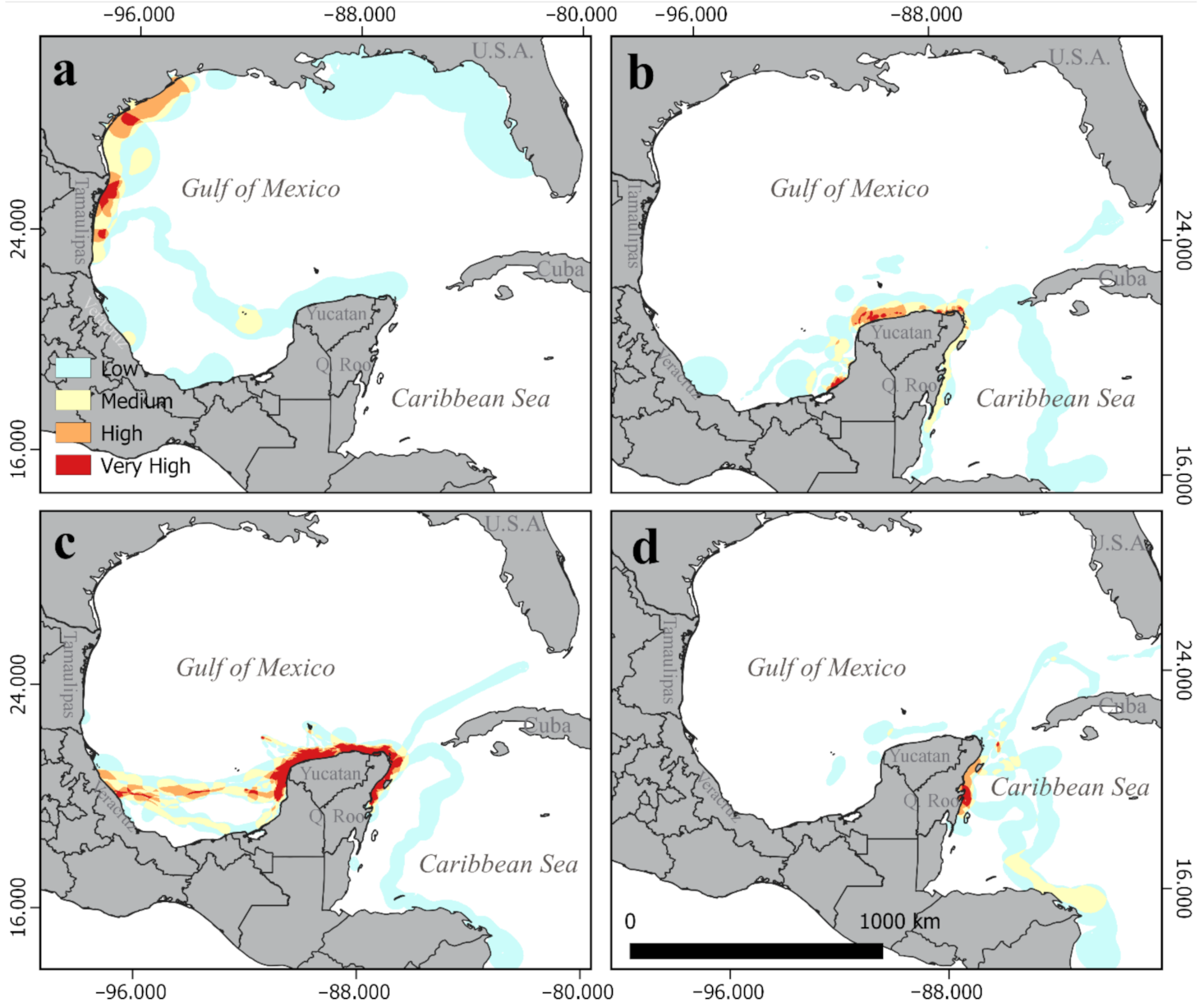
3.2. Critical Habitats Delineation
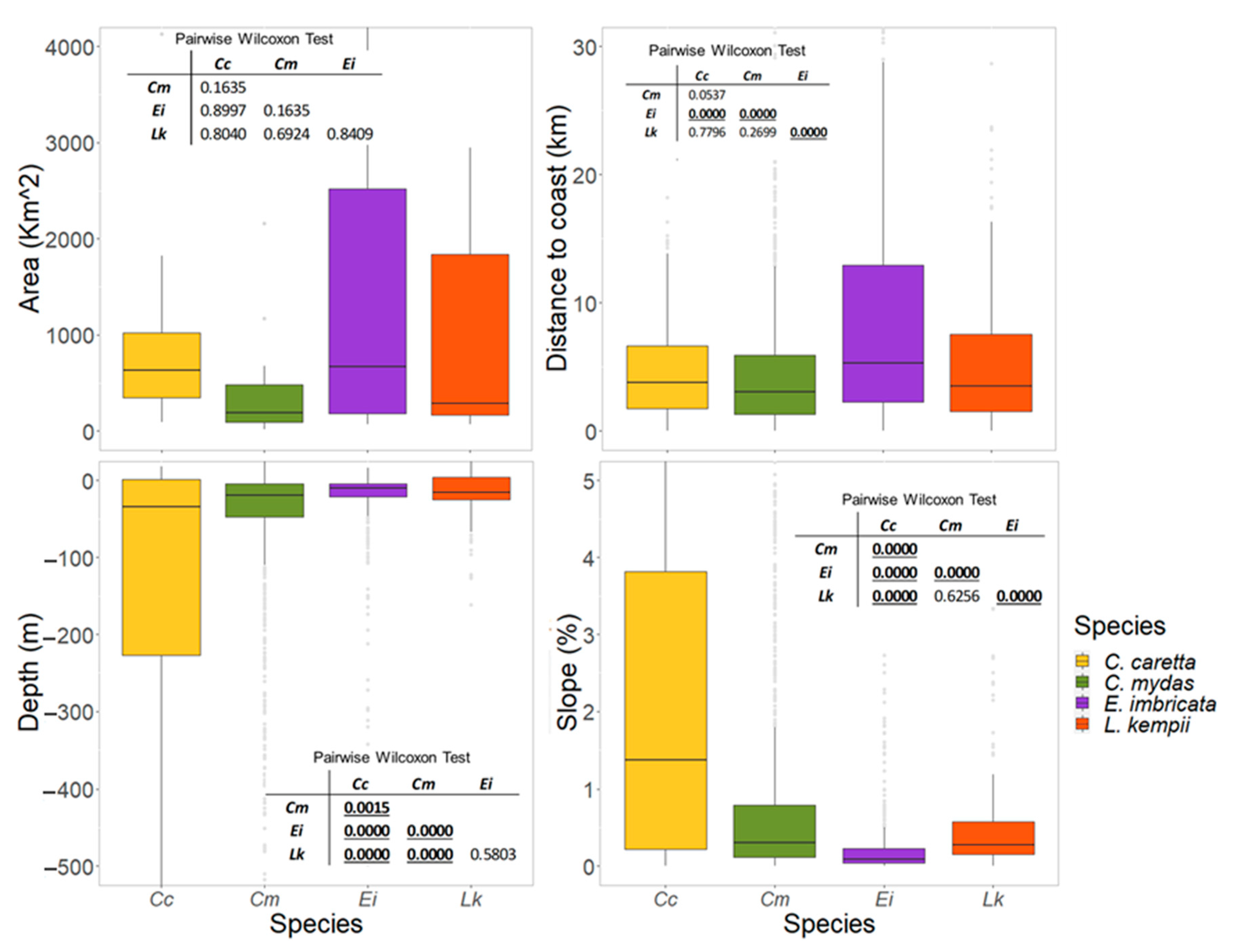
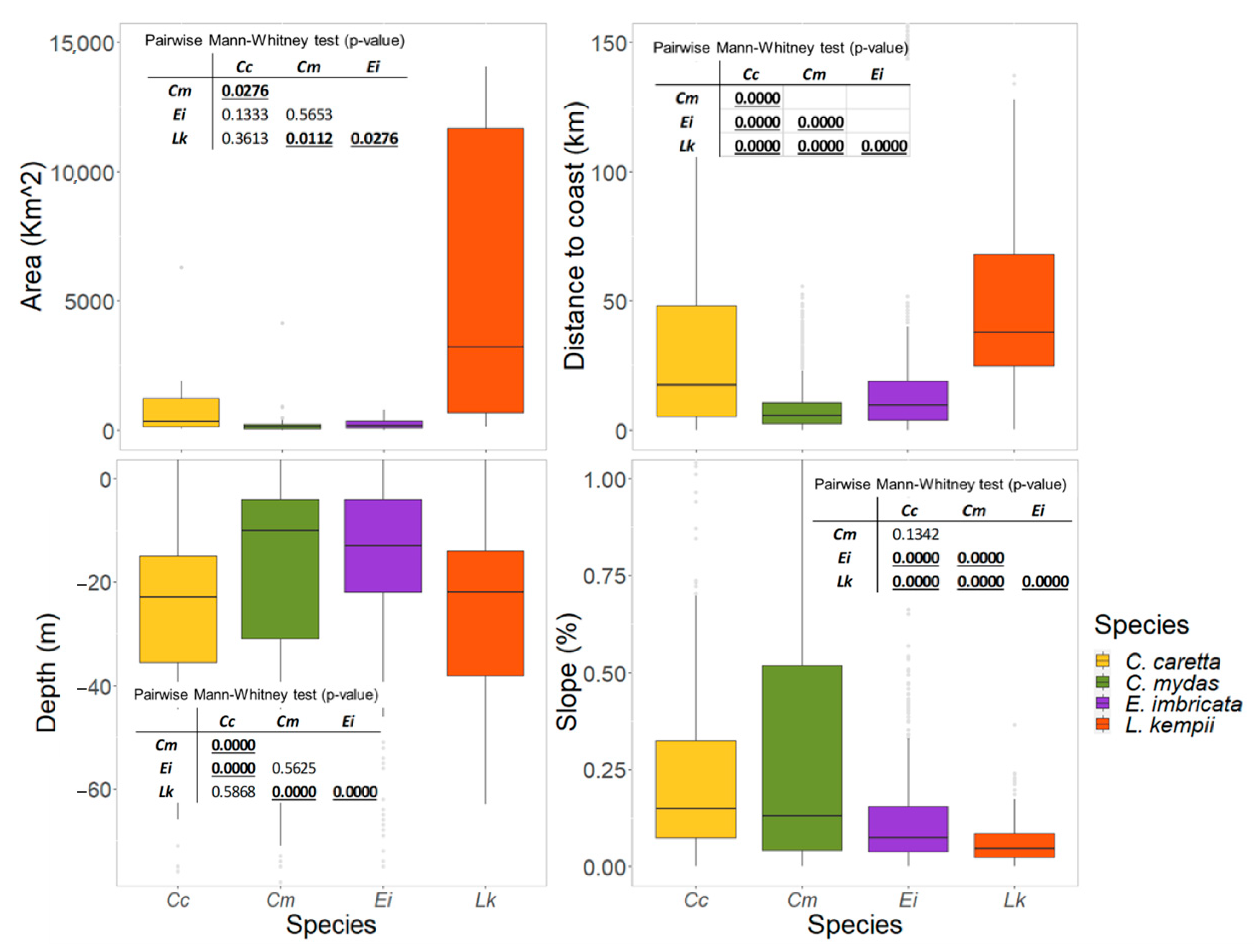
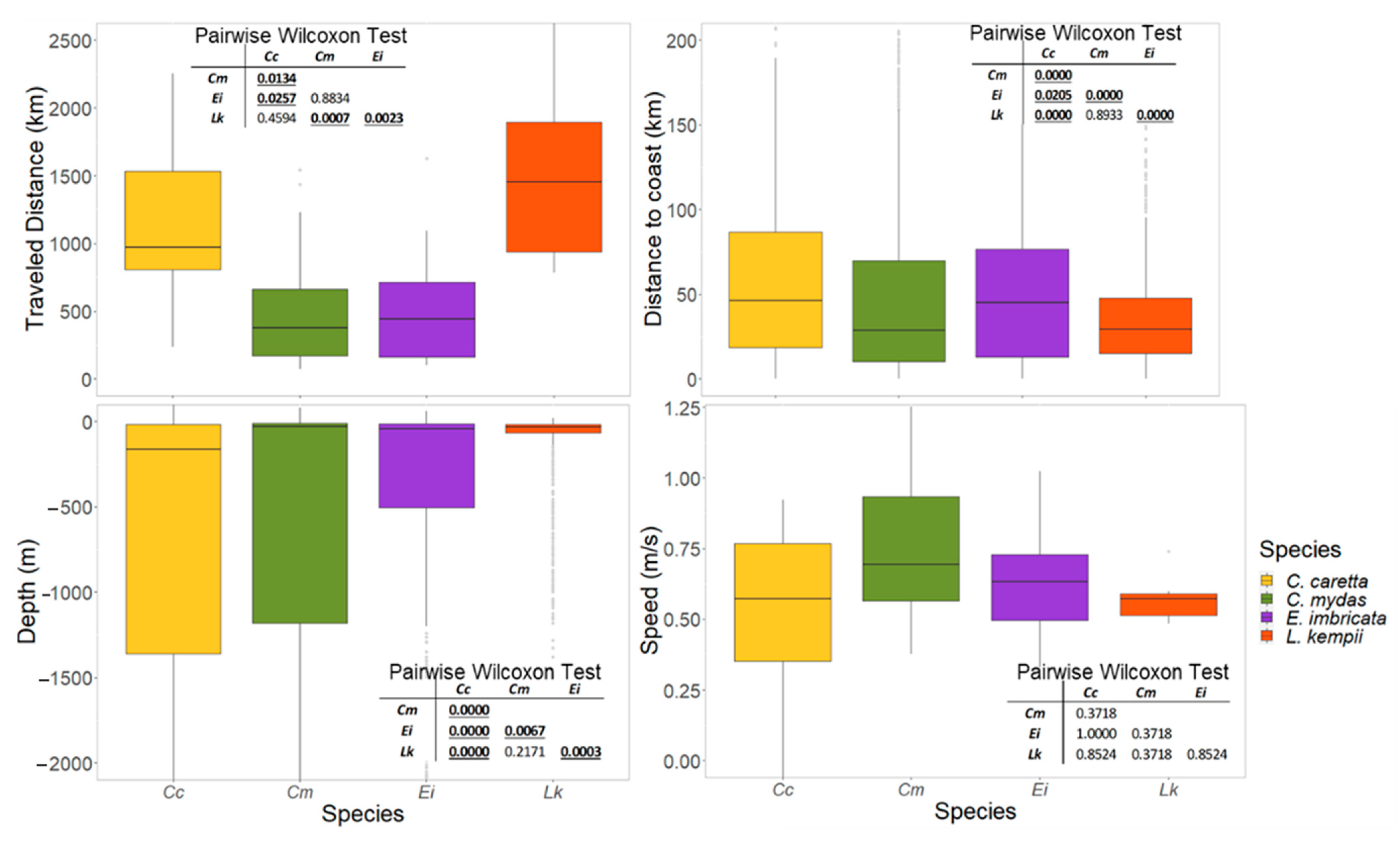
3.3. Physical Features of the Critical Habitats
3.3.1. Internesting Habitats
3.3.2. Feeding and Residency Areas
3.3.3. Movement and Aggregation Patterns
3.3.4. Migratory Routes
4. Discussion
4.1. Critical Habitats Delineation and Physical Features
4.2. Migratory Corridors
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lennox, R.J.; Engler-Palma, C.; Kowarski, K.; Filous, A.; Whitlock, R.; Cooke, S.J.; Auger-Méthé, M. Optimizing marine spatial plans with animal tracking data. Can. J. Fish. Aquat. Sci. 2019, 76, 497–509. [Google Scholar] [CrossRef]
- Augé, A.A.; Dias, M.P.; Lascelles, B.; Baylis, A.M.M.; Black, A.; Boersma, P.D.; Catry, P.; Crofts, S.; Galimberti, F.; Granadeiro, J.P.; et al. Framework for mapping key areas for marine megafauna to inform Marine Spatial Planning: The Falkland Islands case study. Mar. Policy 2018, 92, 61–72. [Google Scholar] [CrossRef]
- Spotila, J.R. Sea Turtles: A Complete Guide to the Biology, Behavior, and Conservation; The Johns Hopkins University Press: Baltimore, MD, USA, 2004; 227p. [Google Scholar]
- Cuevas, E. Spatial and temporal dimensions of the critical habitat selection processes by sea turtles. Rev. Biol. Mar. Oceanogr. 2017, 52, 187–199. [Google Scholar] [CrossRef]
- Meylan, A.B.; Donnelly, M. Status justification for listing the hawksbill turtle (Eretmochelys imbricata) as critically endangered on the 1996 IUCN Red List of Threatened Animals. Chelonian Conserv. Biol. 1999, 3, 200–224. [Google Scholar]
- Dow-Piniak, W.E.; Eckert, K.L. Sea turtle nesting habitat in the Wider Caribbean Region. Endanger. Species Res. 2011, 15, 129–141. [Google Scholar] [CrossRef][Green Version]
- Valverde, R.A.; Holzwart, K.R. Sea turtles of the Gulf of Mexico. In Habitats and Biota of the Gulf of Mexico: Before the Deepwater Horizon Oil Spill; Ward, S.H., Ed.; Springer Nature: New York, NY, USA, 2017; Volume 2, pp. 1189–1352. [Google Scholar]
- Caillouet, C.W., Jr.; Raborn, S.W.; Shaver, D.J.; Putman, N.F.; Gallaway, B.J.; Mansfield, K.L. Did declining carrying capacity for the Kemp’s Ridley sea turtle population within the Gulf of Mexico contribute to the nesting setback in 2010–2017? Chelonian Conserv. Biol. 2018, 17, 123–133. [Google Scholar] [CrossRef]
- Ceriani, S.S.; Casale, P.; Brost, M.; Leone, E.H.; Witherington, B.E. Conservation implications of sea turtle nesting trends: Elusive recovery of a globally important loggerhead population. Ecosphere 2019, 10, e02936. [Google Scholar] [CrossRef]
- Heileman, S.; Rabalais, N. Gulf of Mexico LME. In The UNEP Large Marine Ecosystem Report: A Perspective on Changing Conditions in LME’s of the World Regional Seas; Sherman, K., Hempel, G., Eds.; UNEP Regional Seas Report and Studies No. 182; UNEP: Nairobi, Kenya, 2008; pp. 673–698. [Google Scholar]
- Fanning, L.; Mahon, R.; McConney, P. Focusing on living marine resource governance: The Caribbean large marine ecosystem and adjacent areas project. Coast. Manag. 2009, 37, 219–234. [Google Scholar] [CrossRef]
- Putman, N. Marine migrations. Curr. Biol. 2018, 28, 952–1008. [Google Scholar] [CrossRef] [PubMed]
- Moorcroft, P.R.; Lewis, M.A. Mechanistic Home Range Analysis; Princeton University Press: Princeton, NJ, USA, 2006. [Google Scholar]
- Broderick, A.C.; Coyne, M.S.; Fuller, W.J.; Glen, F.; Godley, B.J. Fidelity and over-wintering of sea turtles. Proc. R. Soc. B 2007, 274, 1533–1539. [Google Scholar] [CrossRef]
- Diario de la Federación. Ley General de Vida Silvestre. Available online: http://www.diputados.gob.mx/LeyesBiblio/pdf/146_190118.pdf (accessed on 1 March 2021).
- Bowen, B.W.; Grant, W.S.; Hillis-Starr, Z.; Shaver, D.J.; Bjorndal, K.A.; Bolten, A.B.; Bass, A.L. Mixed-stock analysis reveals the migrations of juvenile hawksbill turtles (Eretmochelys imbricata) in the Caribbean Sea. Mol. Ecol. 2007, 16, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Abreu-Grobois, F.A. Generalidades de las tortugas marinas. In Las Tortugas Marinas en México: Logros y Perspectivas para su Conservación; Gaona-Pineda, O., Barragán-Rocha, A.R., Eds.; Comisión Nacional de Áreas Naturales Protegidas: Mexico City, Mexico, 2016; pp. 19–34. [Google Scholar]
- Hart, K.M.; Iverson, A.R.; Fujisaki, I.; Lamont, M.M.; Bucklin, D.; Shaver, S.J. Sympatry or syntopy? Investigating drivers of distribution and co-occurrence for two imperiled sea turtles species in the Gulf of Mexico neritic waters. Ecol. Evol. 2018, 8, 12656–12669. [Google Scholar] [CrossRef] [PubMed]
- Godley, B.J.; Blumenthal, J.M.; Broderick, A.C.; Coyne, M.S.; Godfrey, M.H.; Hawkes, L.A.; Witt, M.J. Satellite tracking of sea turtles: Where have we been and where do we go next? Endanger. Species Res. 2008, 4, 3–22. [Google Scholar] [CrossRef]
- Cuevas, E.; Putman, N.F.; Uribe-Martínez, A.; López-Castro, M.C.; Guzmán-Hernández, V.; Gallegos-Fernández, S.A.; Liceaga-Correa, M.A.; Trujillo-Córdova, J.A.; González-Díaz-Mirón, R.J.; Negrete-Phillipe, A.; et al. First spatial distribution analysis of male sea turtles in the Southern Gulf of Mexico. Front. Mar. Sci. 2020, 7, 561846. [Google Scholar] [CrossRef]
- Heppell, S.S. Application on life-history theory and population model analysis to turtle conservation. Copeia 1998, 2, 367–375. [Google Scholar] [CrossRef]
- Hart, K.M.; Hyrenbach, K.D. Satellite telemetry of marine megavertebrates: The coming of age of an experimental science. Endanger. Species Res. 2009, 10, 9–20. [Google Scholar] [CrossRef]
- Hussey, N.E.; Kessel, S.T.; Aarestrup, K.; Cooke, S.J.; Cowley, P.D.; Fisk, A.T.; Harcourt, R.G.; Holland, K.N.; Iverson, S.J.; Kocik, J.F.; et al. Aquatic animal telemetry: A panoramic window into the underwater world. Science 2015, 348, 1–10. [Google Scholar] [CrossRef]
- Hays, G.C.; Ferreira, L.C.; Sequeira, A.M.M.; Meekan, M.G.; Duarte, C.M.; Bailey, H.; Bailleul, F.; Bowen, W.D.; Caley, M.J.; Costa, D.P.; et al. Key questions in marine megafauna movement ecology. Trends Ecol. Evol. 2016, 31, 463–475. [Google Scholar] [CrossRef]
- Jeffers, V.F.; Godley, B.J. Satellite tracking in sea turtles: How do we find our way to the conservation dividends? Biol. Conserv. 2016, 199, 172–184. [Google Scholar] [CrossRef]
- Cuevas, E.; Abreu-Grobois, F.A.; Guzmán-Hernández, V.; Liceaga-Correa, M.A.; van Damo, R.P. Post-nesting migratory movements of hawksbill turtles Eretmochelys imbricata in waters adjacent to the Yucatan Peninsula, Mexico. Endanger. Species Res. 2008, 10, 123–133. [Google Scholar] [CrossRef]
- Shaver, D.J.; Hart, K.M.; Fujisaki, I.; Rubio, C.; Sartain, A.R.; Peña, J.; Burchfield, P.M.; Gómez-Gámez, D.; Ortíz, J. Foraging area fidelity for Kemp’s ridleys in the Gulf of Mexico. Ecol. Evol. 2013, 3, 2002–2012. [Google Scholar] [CrossRef]
- Schofield, G.; Dimadi, A.; Fossette, S.; Katselidis, K.A.; Koutsoubas, D.; Lilley, M.K.S.; Luckman, A.; Pantis, J.D.; Karagouni, A.D.; Hays, G.C. Satellite tracking large numbers of individuals to infer population level dispersal and core areas for the protection of an endangered species. Divers. Distrib. 2013, 19, 834–844. [Google Scholar] [CrossRef]
- Foley, A.M.; Schroeder, B.A.; Hardy, R.; MacPherson, S.L.; Nicholas, M. Long-term behavior at foraging sites of adult female loggerhead sea turtles (Caretta caretta) from three Florida rookeries. Mar. Biol. 2014, 161, 1251–1262. [Google Scholar] [CrossRef] [PubMed]
- Putman, N.F.; Mansfield, K.L. Direct evidence of swimming demonstrates active dispersal in the sea turtle “Lost Years”. Curr. Biol. 2015, 25, 1221–1227. [Google Scholar] [CrossRef]
- Hart, K.M.; Iverson, A.R.; Fujisaki, I.; Lamont, M.M.; Bucklin, D.; Shaver, D.J. Marine threats overlap key foraging habitat for two imperiled sea turtle species in the Gulf of Mexico. Front. Mar. Sci. 2018, 5, 36. [Google Scholar] [CrossRef]
- Luschi, P.; Lutjeharms, J.R.E.; Lambardi, P.; Mencacci, R.; Hughes, G.R.; Hays, G.C. A review of migratory behavior of sea turtles off southeastern Africa. S. Afr. J. Sci. 2006, 102, 51–58. [Google Scholar]
- Cerritelli, G.; Bianco, G.; Santini, G.; Broderick, A.C.; Godley, B.J.; Hays, G.C.; Luschi, P.; Åkesson, S. Assessing reliance on vector navigation in the long-distance oceanic migrations of green sea turtles. Behav. Ecol. 2019, 30, 68–79. [Google Scholar] [CrossRef]
- Huang, H.W. Conservation hotspots for the turtles on the high seas of the Atlantic Ocean. PLoS ONE 2015, 10, e0133614. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, E.; Guzmán-Hernández, V.; Uribe-Martínez, A.; Raymundo-Sánchez, A.; Herrera-Pavón, R. Identification of potential sea turtle bycatch hotspots using a spatially explicit approach in the Yucatan peninsula, Mexico. Chelonian Conserv. Biol. 2018, 17, 78–93. [Google Scholar] [CrossRef]
- Cuevas, E.; Liceaga-Correa, M.A.; Uribe-Martínez, A. Ecological vulnerability of two sea turtle species in the Gulf of Mexico: An integrated spatial approach. Endanger. Species Res. 2019, 40, 337–356. [Google Scholar] [CrossRef]
- Welch, H.; Hazen, E.L.; Briscoe, D.K.; Bograd, S.J.; Jacox, M.G.; Eguchi, T.; Benson, S.R.; Fahy, C.C.; Garfield, T.; Robinson, D.; et al. Environmental indicators to reduce loggerhead turtle bycatch offshore of Southern California. Ecol. Indic. 2019, 98, 657–664. [Google Scholar] [CrossRef]
- Moncada, F.G.; Hawkes, L.A.; Fish, M.R.; Godley, B.J.; Manolis, S.C.; Medina, Y.; Nodarse, G.; Webb, G.J.W. Patterns of dispersal of hawksbill turtles from the Cuban shelf inform scale of conservation and management. Biol. Conserv. 2012, 148, 191–199. [Google Scholar] [CrossRef]
- Dawson, T.M.; Formia, A.; Agamboué, P.D.; Asseko, G.M.; Boussamba, F.; Cardiec, F.; Chartrain, E.; Doherty, P.D.; Fay, J.M.; Godley, B.J.; et al. Designation and mangament for nesting olive ridley sea turtles using satellite tracking. Front. Mar. Sci. 2017, 4, 312. [Google Scholar] [CrossRef]
- Niviere, M.; Chambault, P.; Pérez, T.; Etienne, D.; Bonola, M.; Martin, J.; Barnéiras, C.; Védie, F.; Mailles, J.; Dumont-Dayot, É.; Gresser, J.; et al. Identification of marine key areas across the Caribbean to ensure the conservation of the critically endangered hawksbill turtle. Biol. Conserv. 2018, 233, 170–180. [Google Scholar] [CrossRef]
- Bolten, A.B.; Crowder, L.B.; Dodd, M.G.; MacPherson, S.L.; Musick, J.A.; Schroeder, B.A.; Witherington, B.E.; Long, K.J.; Snover, M.L. Quantifying multiple threats to endangered species: An example from loggerhead sea turtles. Front. Ecol. Environ. 2011, 9, 295–301. [Google Scholar] [CrossRef]
- Iverson, A.R.; Benscoter, A.M.; Fujisaki, I.; Lamont, M.M.; Hart, K.M. Migration corridors and threats in the Gulf of Mexico and Florida Straits for Loggerhead sea turtles. Front. Mar. Sci. 2020, 7, 208. [Google Scholar] [CrossRef]
- Wallace, B.P.; Stacey, B.A.; Cuevas, E.; Holyoake, C.; Lara, P.H.; Marcondes, A.C.J.; Miller, J.D.; Nijkamp, H.; Pilcher, N.J.; Robinson, I.; et al. Oil spills and sea turtles: Documented effects and considerations for response and assessment efforts. Endanger. Species Res. 2020, 41, 17–37. [Google Scholar] [CrossRef]
- Burges, M.G.; McDermott, G.R.; Owashi, B.; Reeves, L.E.P.; Clavelle, T.; Ovando, D.; Wallace, B.P.; Lewison, R.L.; Gaines, S.D.; Costello, C. Protecting marine mammals, turtles, and birds by rebuilding global fisheries. Science 2018, 359, 1255–1258. [Google Scholar] [CrossRef]
- Scales, K.L.; Hazen, E.L.; Jacox, M.G.; Castruccio, F.; Maxwell, S.M.; Lewison, R.L.; Bograd, S.J. Fisheries bycatch risk to marine megafauna is intensified in Lagrangian coherent structures. Proc. Natl. Acad. Sci. USA 2018, 115, 7362–7367. [Google Scholar] [CrossRef]
- Wallace, B.P.; Brosnan, T.; McLamb, D.; Rowles, T.; Ruder, E.; Schroeder, B.; Schwacke, L.; Stacy, B.; Sullivan, L.; Takeshita, R.; et al. Effects of the Deepwater Horizon oil spill on protected species. Endanger. Species Res. 2017, 33, 1–7. [Google Scholar] [CrossRef]
- Hamann, M.; Godfrey, M.H.; Seminoff, J.A.; Arthur, K.; Barata, P.C.R.; Bjorndal, K.A.; Bolten, A.B.; Broderick, A.C.; Campbell, L.M.; Carreras, C.; et al. Global research priorities for sea turtles: Informing management and conservation in the 21st century. Endanger. Species Res. 2010, 11, 245–269. [Google Scholar] [CrossRef]
- Hays, G.C.; Bailey, H.; Bograd, S.J.; Don-Bowes, W.; Campagna, C.; Carmichael, R.H.; Casale, P.; Chiaradia, A.; Costa, D.P.; Cuevas, E.; et al. Translating marine animal tracking data into conservation policy and management. Trends Ecol. Evol. 2019, 34, 459–473. [Google Scholar] [CrossRef]
- Méndez, D.; Cuevas, E.; Navarro, J.; González-Garza, B.I.; Guzmán-Hernández, V. Satellite tracking of Green turtle females Chelonia mydas and the evaluation of their home ranges in the north coast of the Yucatan Peninsula, Mexico. Rev. Biol. Mar. Oceanogr. 2013, 48, 497–509. [Google Scholar] [CrossRef]
- Vázquez, M.; Cuevas, E. Hábitats críticos de tortuga blanca (Chelonia mydas) y carey (Eretmochelys imbricata) en la península de Yucatán y su coincidencia espacial con zonas de pesca artesanal. In El uso del Conocimiento de las Tortugas Marinas Como Herramienta para la Restauración de sus Poblaciones y Hábitats Asociados en México; Cuevas-Flores, E., Guzmán-Hernández, V., Guerra-Santos, J.J., Rivas-Hernández, G.A., Eds.; Universidad Autónoma del Carmen: Ciudad del Carmen, Mexico, 2019; pp. 57–62. [Google Scholar]
- Gallegos-Fernández, S.A.; Cuevas, E.; Liceaga-Correa, M.A. Methodological processes for the placement of satellite transmitters in hard-shelled sea turtles on nesting beaches. Rev Biol. Mar. Oceanogr. 2018, 53, 147–156. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 31 January 2020).
- Collecte Localisation Satellite. Argos User’s Manual 2007–2016. Available online: https://www.argos-system.org/wp-content/uploads/2016/08/r363_9_argos_users_manual-v1.6.6.pdf (accessed on 11 April 2021).
- Freitas, C.; Lydersen, C.; Ims, R.A.; Fedak, M.A.; Kovacs, K.M. A simple new algorithm to filter marine mammal Argos locations. Mar. Mammal Sci. 2012, 24, 315–325. [Google Scholar] [CrossRef]
- Boyd, J.D.; Brightsmith, D.J. Error properties of Argos satellite telemetry locations using least squares and Kalman filtering. PLoS ONE 2013, 8, e63051. [Google Scholar] [CrossRef] [PubMed]
- Lopez, R.; Malardé, J.P. Improving ARGOS Doppler Location Using Kalman Filtering; Collecte Localisation Satellites: Ramonville Saint-Agne, France, 2011. [Google Scholar]
- McClintock, B.T.; London, J.M.; Cameron, M.F.; Boveng, P.L. Modelling animal movement using the Argos satellite telemetry location error ellipse. Methods Ecol. Evol. 2015, 6, 266–277. [Google Scholar] [CrossRef]
- Shaver, D.J.; Hart, K.M.; Fujisaki, I.; Rubio, C.; Sartain-Iverson, A.R.; Peña, J.; Gómez-Gámez, D.; González-Días-Miron, R.J.; Burchfield, P.M.; Martinez, H.J.; et al. Migratory corridors of adult female Kemp’s ridley turtles in the Gulf of Mexico. Biol. Conserv. 2016, 194, 158–167. [Google Scholar] [CrossRef]
- Bentivegna, F.; Valentino, F.; Falco, P.; Zambianchi, E.; Hochscheid, S. The relationship between loggerhead turtle (Caretta caretta) movement patterns and Mediterranean currents. Mar. Biol. 2007, 151, 1605–1614. [Google Scholar] [CrossRef]
- Bailey, H.; Fossette, S.; Bograd, S.J.; Shillinger, G.L.; Swithenbank, A.M.; Georges, J.Y.; Gaspar, P.; Strömber, K.H.P.; Paladino, F.V.; Spotila, J.R.; et al. Movement patterns for a critically endangered species, the Leatherback turtle (Dermochelys coriacea), linked to foraging success and population status. PLoS ONE 2012, 7, e36401. [Google Scholar] [CrossRef]
- McConnell, B.J.; Chambers, C.; Fedak, M.A. Foraging ecology of southern elephant seals in relation to the bathymetry and productivity of the Southern Ocean. Antarct. Sci. 1992, 4, 393–398. [Google Scholar] [CrossRef]
- Luschi, P.; Hays, G.C.; Del Seppia, C.; Marsh, R.; Papi, F. The navigational feats of green sea turtles migrating from Ascencion Island investigate by satellite telemetry. Proc. R. Soc. Lond. B 1998, 265, 2279–2284. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Kapoor, J.; Wieczorek, J.; Garcia, N.; Maunahan, A.; Rala, A.; Mandel, A. Gadm database of Global Administrative Areas: 2016. Available online: https://gadm.org/ (accessed on 7 July 2021).
- Hart, K.M.; Lamont, M.M.; Iverson, A.R.; Smith, B.J. The importance of the northeastern Gulf of Mexico to foraging loggerhead sea turtles. Front. Mar. Sci. 2020, 7, 330. [Google Scholar] [CrossRef]
- Worton, B.J. Kernel methods for estimating the utilization distribution in home-range studies. Ecology 1989, 70, 164–168. [Google Scholar] [CrossRef]
- Calenge, C. The package adehabitat for the R software: A tool for the analysis of space and habitat use by animals. Ecol. Model. 2006, 197, 516–519. [Google Scholar] [CrossRef]
- Bradshaw, C.J.A.; Sims, D.W.; Hays, G.C. Measurement error causes scale-dependent threshold erosion of biological signals in animal movement data. Ecol. App. 2007, 17, 628–638. [Google Scholar] [CrossRef]
- Lowther, A.D.; Lydersen, C.; Fedak, M.A.; Lovell, P.; Kovacs, K.M. The Argos-CLS Kalman filter: Error structures and state-space modelling relative to fastloc GPS data. PLoS ONE 2015, 10, e0124754. [Google Scholar] [CrossRef]
- Horne, J.S.; Garton, E.O.; Krone, S.M.; Lewis, J.S. Analyzing animal movements using Brownian bridges. Ecology 2007, 88, 2354–2363. [Google Scholar] [CrossRef]
- Nielson, R.M.; Sawyer, H.; McDonald, T.L. R Package ‘BBMM’ Brownian Bridge Movement Model. WEST, Inc., 2015. Available online: https://www.west-inc.com (accessed on 7 July 2020).
- North, M.A. A method for implementing a statistically significant number of data classes in the Jenks algorithm. In Proceedings of the Sixth International Conference, Fuzzy Systems and Knowledge Discovery (IEEE), Tianjin, China, 14–16 August 2009; pp. 35–38. [Google Scholar]
- Block, B.A.; Jonsen, I.D.; Jorgensen, S.J.; Winship, A.J.; Shaffer, S.A.; Bograd, S.J.; Hazen, E.L.; Foley, E.L.; Breed, G.A.; Harrison, A.L.; et al. Tracking apex marine predator movements in a dynamic ocean. Nature 2011, 475, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Muller-Karger, F.E.; Walsh, J.J.; Evans, R.H.; Meyers, M.B. On the seasonal phytoplankton concentration and sea surface temperature cycles of the Gulf of Mexico as determined by satellites. J. Geophys. Res. Ocean 1991, 96, 12645–12665. [Google Scholar] [CrossRef]
- Uribe-Martínez, A.; Liceaga-Correa, M.A.; Cuevas, E. Idoneidad ambiental de las tortugas marinas en el Golfo de México: Visión futura en un océano más caliente. In Vulnerabilidad de las Zonas Costeras de Latinoamérica al Cambio Climático; Botello, A., Villanueva, S., Gutiérrez, J., Rojas-Galaviz, L., Eds.; UJAT/UNAM/UAC: Campeche, Mexico, 2017; pp. 379–414. [Google Scholar]
- Uribe-Martínez, A.; Aguirre-Gómez, R.; Zavala-Hidalgo, J.; Ressl, R.; Cuevas, E. Oceanographic units of the Gulf of Mexico and adjacent areas: The monthly integration of surface biophysical features. Geofísica Int. 2019, 58, 295–315. [Google Scholar] [CrossRef]
- Bivand, R.; Rundel, C. R Package: Misc-gDistance: Distance between Geometries. Available online: https://rdrr.io/rforge/rgeos/man/misc-gDistance.html (accessed on 12 May 2021).
- General Bathymetric Chart of the Oceans. Available online: https://www.gebco.net/ (accessed on 12 May 2021).
- Sequeira, A.M.M.; Heupel, M.R.; Lea, M.A.; Eguíluz, V.M.; Duarte, C.M.; Meekan, M.G.; Thums, M.; Calich, H.J.; Carmichael, R.H.; Ferreira, L.C.; et al. The importance of sample size in marine megafauna tagging studies. Ecol. Appl. 2019, 29, e01947. [Google Scholar] [CrossRef] [PubMed]
- Hulin, V.; Guillon, J.M. Female philopatry in a heterogeneous environment: Ordinary conditions leading to extraordinary ESS sex ratios. BMC Evol. Biol. 2007, 7, 13. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.L.M.; Luschi, P.; Hays, G.C. Detecting female precise natal philopatry in green turtles using assignment methods. Mol. Ecol. 2007, 16, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Stiebens, V.A.; Merino, S.E.; Roder, C.; Chain, F.J.J.; Lee, P.L.M.; Eizaguirre, C. Living on the edge: How philopatry maintains adaptive potential. Proc. R. Soc. B 2013, 280, 20130305. [Google Scholar] [CrossRef]
- Stokes, L.L.; Broderick, A.C.; Canbolat, A.F.; Candan, O.; Fuller, W.J.; Glen, F.; Levy, Y.; Rees, A.F.; Rilov, G.; Snape, R.T.; et al. Migratory corridors and foraging hotspots: Critical habitats identified for Mediterranean green turtles. Divers. Distrib. 2015, 21, 665–674. [Google Scholar] [CrossRef]
- Mettler, E.K.; Clyde-Brockway, C.E.; Sinclair, E.M.; Paladino, F.V.; Honarvar, S. Determining critical inter-nesting, migratory, and foraging habitats for the conservation of East Atlantic green turtles (Chelonia mydas). Mar. Biol. 2020, 167, 106. [Google Scholar] [CrossRef]
- Zbinden, J.A.; Aebischer, A.; Margaritoulis, D.; Arlettaz, R. Insights into the management of sea turtle internesting are through satellite telemetry. Biol. Conserv. 2007, 137, 157–162. [Google Scholar] [CrossRef]
- Shillinger, G.L.; Swithenbank, A.M.; Bograd, S.J.; Bailey, H.; Castelton, M.R.; Wallace, B.P.; Spotila, J.R.; Paladino, F.V.; Piedra, R.; Block, B.A. Identification of high-use internesting habitats for eastern Pacific leatherback turtles: Role of the environment and implications for conservation. Endanger. Species Res. 2010, 10, 215–232. [Google Scholar] [CrossRef][Green Version]
- Shaver, D.J.; Hart, K.M.; Fuhisaki, I.; Bucklin, D.; Iverson, A.R.; Rubio, C.; Backof, T.F.; Burchfield, P.M.; González-Díaz-Mirón, R.J.; Dutton, P.H.; et al. Inter-nesting movements and habitat-use of adult female Kemp’s ridley turtles in the Gulf of Mexico. PLoS ONE 2017, 12, e0174248. [Google Scholar] [CrossRef]
- Diario Oficial de la Federacion—Secretaria de Gobernacion. Available online: https://www.dof.gob.mx/nota_detalle.php?codigo=5381585&fecha=11/02/2015 (accessed on 11 April 2021).
- Kakai, T. Assessing the effectiveness of LED lights for the reduction of sea turtle bycatch in an artisanal gillnet fishery—A case study from north of Kenya. WIO J. Mar. Sci. 2019, 18, 37–44. [Google Scholar] [CrossRef]
- Putman, N.F.; Hawkins, J.; Gallaway, B.J. Managing fisheries in a world with more sea turtles. Proc. R. Soc. B 2020, 287, 20200220. [Google Scholar] [CrossRef]
- Bielli, A.; Alfaro-Shigueto, J.; Doherty, P.D.; Godley, B.J.; Ortiz, C.; Pasara, A.; Wang, J.H.; Mangel, J.C. An illuminating idea to reduce bycatch in the Peruvian small-scale gillnet. Biol. Conserv. 2020, 241, 108277. [Google Scholar] [CrossRef]
- Dujon, A.M.; Schofield, G.; Lester, R.E.; Esteban, N.; Hays, G.C. Fastloc-GPS reveals daytime departure and arrival during long-distance migration and the use of different resting strategies. Mar. Biol. 2017, 164, 187. [Google Scholar] [CrossRef]
- Healy, S.D.; Braithwaite, V.A. The role of landmarks in small- and large-scale navigation. In Spatial Cognition, Spatial Perception: Mapping the Self and Space; Dolins, F.L., Mitchell, R.W., Eds.; Cambridge University Press: New York, NY, USA, 2010; pp. 152–179. [Google Scholar]
- Painter, K.J.; Plochocka, A.Z. Efficiency of island homing by sea turtles under multimodal navigating strategies. Ecol. Modell. 2019, 391, 40–52. [Google Scholar] [CrossRef]
- Johnsen, S.; Lohmann, K.J.; Warrant, E.J. Animal navigation: A noisy magnetic sense? J. Exp. Biol. 2020, 223, jeb164921. [Google Scholar] [CrossRef]
- Baudouin, M.; Thoisy, B.; Chambault, P.; Brzins, R.; Entraygues, M.; Kelle, L.; Turny, A.; Le Maho, Y.; Chevallier, D. Identification of key marine areas for conservation based on satellite tracking of post-nesting migrating green turtles (Chelonia mydas). Biol. Conserv. 2015, 184, 36–41. [Google Scholar] [CrossRef]
- Hart, K.M.; Lamont, M.M.; Fujisaki, I.; Tucker, A.D.; Carthy, R.R. Common coastal foraging areas for loggerheads in the Gulf of Mexico: Opportunity for marine conservation. Biol. Conserv. 2012, 145, 185–194. [Google Scholar] [CrossRef]
- van Dam, R.P.; Diez, C.E.; Balazs, G.H.; Colón-Colón, L.A.; McMillan, W.O.; Schroeder, B. Sex-specific migration patterns of hawksbill turtles breeding at Mona Island, Puerto Rico. Endanger. Species Res. 2008, 4, 85–94. [Google Scholar] [CrossRef]
- Hays, G.C.; Metcalfe, J.D.; Walne, A.W. The implications of lung-regulated buoyancy control for dive depth and duration. Ecology 2004, 85, 1137–1145. [Google Scholar] [CrossRef]
- Bresette, M.J.; Witherington, B.E.; Herren, R.M.; Bagley, D.A.; Gorham, J.C.; Traxler, S.L.; Crady, C.K.; Hardy, R. Size-class partitioning and herding in a foraging group of green turtles Chelonia mydas. Endanger. Species Res. 2010, 9, 105–116. [Google Scholar] [CrossRef]
- Herren, R.M.; Bagley, D.A.; Bresette, M.J.; Holloway-Adkins, K.G.; Clark, D.; Witherington, B.E. Sea turtle abundance and demographic measurements in a marine protected area in the Florida Keys, USA. Herpetol. Conserv. Biol. 2018, 13, 224–239. [Google Scholar]
- Tröeng, S.; Evans, D.R.; Harrison, E.; Lagueux, C.J. Migration of green turtle Chelona mydas from Tortuguero, Costa Rica. Mar. Biol. 2005, 148, 435–447. [Google Scholar] [CrossRef]
- Tröeng, S.; Dutton, P.H.; Evans, D. Migration of hawksbill turtles Eretmochelys imbricate from Tortuguero, Costa Rica. Ecography 2005, 28, 394–402. [Google Scholar] [CrossRef]
- Blumenthal, J.M.; Solomon, J.L.; Bell, C.D.; Austin, T.J.; Ebanks-Petrie, G.; Coyne, M.S.; Broderick, A.C.; Godley, B.J. Satellite tracking highlights the need for international cooperation in marine turtle management. Endanger. Species Res. 2006, 2, 51–61. [Google Scholar] [CrossRef]
- Horrocks, J.A.; Krueger, B.H.; Fastigi, M.; Pemberton, E.L.; Eckert, K.L. International movements of adult female hawksbill turtles (Eretmochelys imbricata): First results from the Caribbean’s Marine Turtle Tagging Centre. Chelonian Conserv. Biol. 2011, 10, 18–25. [Google Scholar] [CrossRef]
- Hawkes, L.A.; Tomás, J.; Revuelta, O.; León, Y.M.; Blumenthal, J.M.; Broderick, A.C.; Fish, M.; Raga, J.A.; Witt, M.J.; Godley, B.J. Migratory patterns in hawksbill turtles described by satellite tracking. Mar. Ecol. Prog. Ser. 2012, 461, 223–232. [Google Scholar] [CrossRef]
- Revuelta, O.; Hawkes, L.A.; León, Y.M.; Godley, B.J.; Raga, J.A.; Tomás, J. Evaluating the importance of Marine Protected Areas for the conservation of hawksbill turtles Eretmochelys imbricata nesting in the Dominican Republic. Endanger. Species Res. 2015, 27, 169–180. [Google Scholar] [CrossRef]
- Becking, L.E.; Christianen, M.J.A.; Nava, M.I.; Miller, N.; Willis, S.; van Dam, R.P. Post-breeding migration routes of marine turtles from Bonaire and Klein Bonaire, Caribbean Netherlands. Endanger. Species Res. 2016, 30, 117–127. [Google Scholar] [CrossRef]
- Lagueux, C.J.; Campbell, C.L.; Strindberg, S. Artisanal green turtle, Chelonia mydas, fishery of Caribbean Nicaragua: I. Catch rates and trends, 1991–2011. PLoS ONE 2014, 9, e94667. [Google Scholar] [CrossRef]
- Mortimer, J.A.; Meylan, P.A.; Donnelly, M. Whose turtles are they, anyway? Mol. Ecol. 2007, 16, 17–18. [Google Scholar] [CrossRef]
- Foley, A.M.; Schroeder, B.A.; Hardy, R.; MacPherson, S.L.; Nicholas, M.; Coyne, M.S. Postnesting migratory behavior of loggerhead sea turtles Caretta caretta from three Florida rookeries. Endanger. Species Res. 2013, 21, 129–142. [Google Scholar] [CrossRef][Green Version]
- Eggleston, D.B.; Lipcius, R.N.; Miller, D.L.; Coba-Cetina, L. Shelter scaling regulates survival of juvenile Caribbean spiny lobster Panulirus argus. Mar. Ecol. Prog. Ser. 1990, 62, 79–88. [Google Scholar] [CrossRef]
- Aleska, K.T.; Sasso, C.R.; Nero, R.W.; Evans, D.R. Movements of leatherback turtles (Dermochelys coriacea) in the Gulf of Mexico. Mar. Biol. 2018, 165, 158. [Google Scholar] [CrossRef]
- Kraus, R.T.; Wells, R.J.D.; Rooker, J.R. Horizontal movements of Atlantic blue marlin (Makaira nigricans) in the Gulf of Mexico. Mar. Biol. 2011, 158, 699–713. [Google Scholar] [CrossRef]
- Hueter, R.E.; Tyminski, J.P.; de la Parra, R. Horizontal movements, migration patterns, and population structure of whale sharks in the Gulf of Mexico and northwestern Caribbean Sea. PLoS ONE 2013, 8, e71883. [Google Scholar] [CrossRef]
- Cuevas-Gómez, G.A. Identificación de Áreas de Crianza para el Tiburón Martillo Sphyrna lewini (Griffith y Smith, 1834) en el sur del GOLFO de México Usando Datos Dependientes de la Pesca. Master’s Thesis, El Colegio de la Frontera Sur, Campeche, Mexico, 2018. [Google Scholar]
- Sea Turtle Conservancy. Available online: https://www.conserveturtles.org/sea-turtle-tracking-list-of-sea-turtle-tracking-projects/ (accessed on 11 April 2021).
- Girard, C.; Tucker, A.; Calmettes, B. Post-nesting migrations of loggerhead sea turtles in the Gulf of Mexico: Dispersal in highly dynamic conditions. Mar. Biol. 2009, 156, 1827–1839. [Google Scholar] [CrossRef]
- Dunn, D.C.; Harrison, A.L.; Curtice, C.; DeLand, S.; Donnelly, B.; Fujioka, E.; Heywood, E.; Kot, C.Y.; Poulin, S.; Whitten, M.; Åkesson, S.; et al. The importance of migratory connectivity for global ocean policy. Proc. R. Soc. B 2019, 286, 20191472. [Google Scholar] [CrossRef]
- Eckert, K.; Azanza-Ricardo, J.; Barrientos-Muñoz, K.G.; Barrios-Garrido, H.; Bevan, E.; Baumbach, D.; Berkel, J.; Campbell, C.L.; Cuevas, E.; Charles, K.; et al. Sea Turtles of the Caribbean. State of the World’s Sea Turtle (SWOT). Available online: https://www.seaturtlestatus.org/articles/2020/2/25/sea-turtles-of-the-caribbean (accessed on 31 April 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uribe-Martínez, A.; Liceaga-Correa, M.d.l.A.; Cuevas, E. Critical In-Water Habitats for Post-Nesting Sea Turtles from the Southern Gulf of Mexico. J. Mar. Sci. Eng. 2021, 9, 793. https://doi.org/10.3390/jmse9080793
Uribe-Martínez A, Liceaga-Correa MdlA, Cuevas E. Critical In-Water Habitats for Post-Nesting Sea Turtles from the Southern Gulf of Mexico. Journal of Marine Science and Engineering. 2021; 9(8):793. https://doi.org/10.3390/jmse9080793
Chicago/Turabian StyleUribe-Martínez, Abigail, María de los Angeles Liceaga-Correa, and Eduardo Cuevas. 2021. "Critical In-Water Habitats for Post-Nesting Sea Turtles from the Southern Gulf of Mexico" Journal of Marine Science and Engineering 9, no. 8: 793. https://doi.org/10.3390/jmse9080793
APA StyleUribe-Martínez, A., Liceaga-Correa, M. d. l. A., & Cuevas, E. (2021). Critical In-Water Habitats for Post-Nesting Sea Turtles from the Southern Gulf of Mexico. Journal of Marine Science and Engineering, 9(8), 793. https://doi.org/10.3390/jmse9080793




