Asset Tracking Whales—First Deployment of a Custom-Made GPS/GSM Suction Cup Tag on Migrating Humpback Whales
Abstract
1. Introduction
2. Materials and Methods
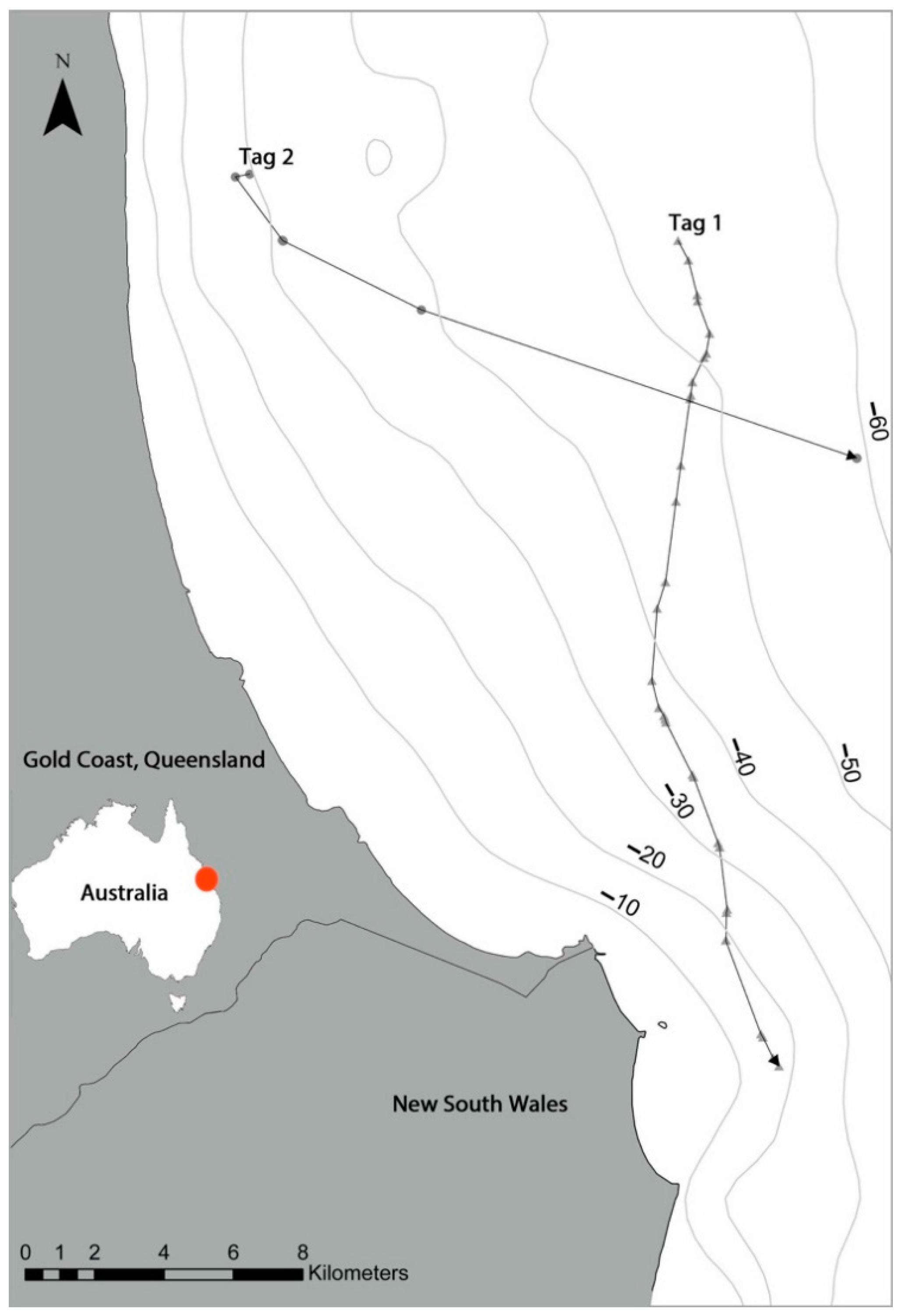
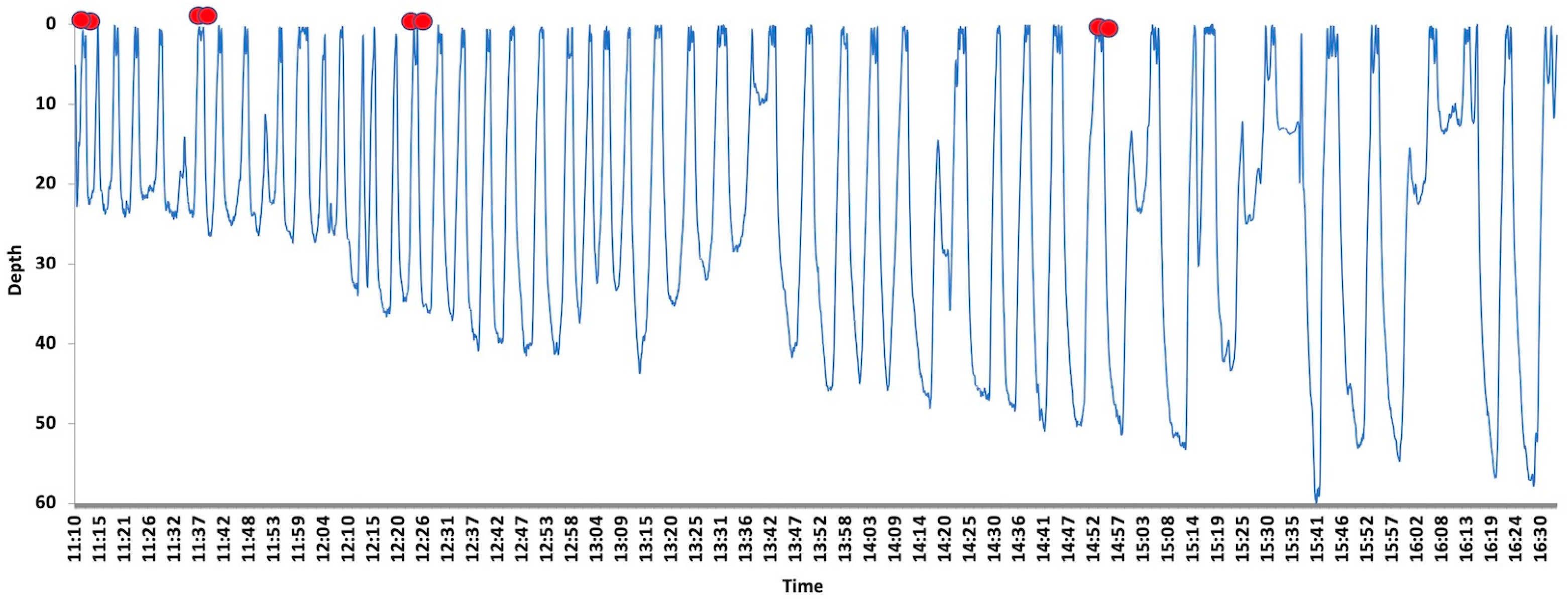
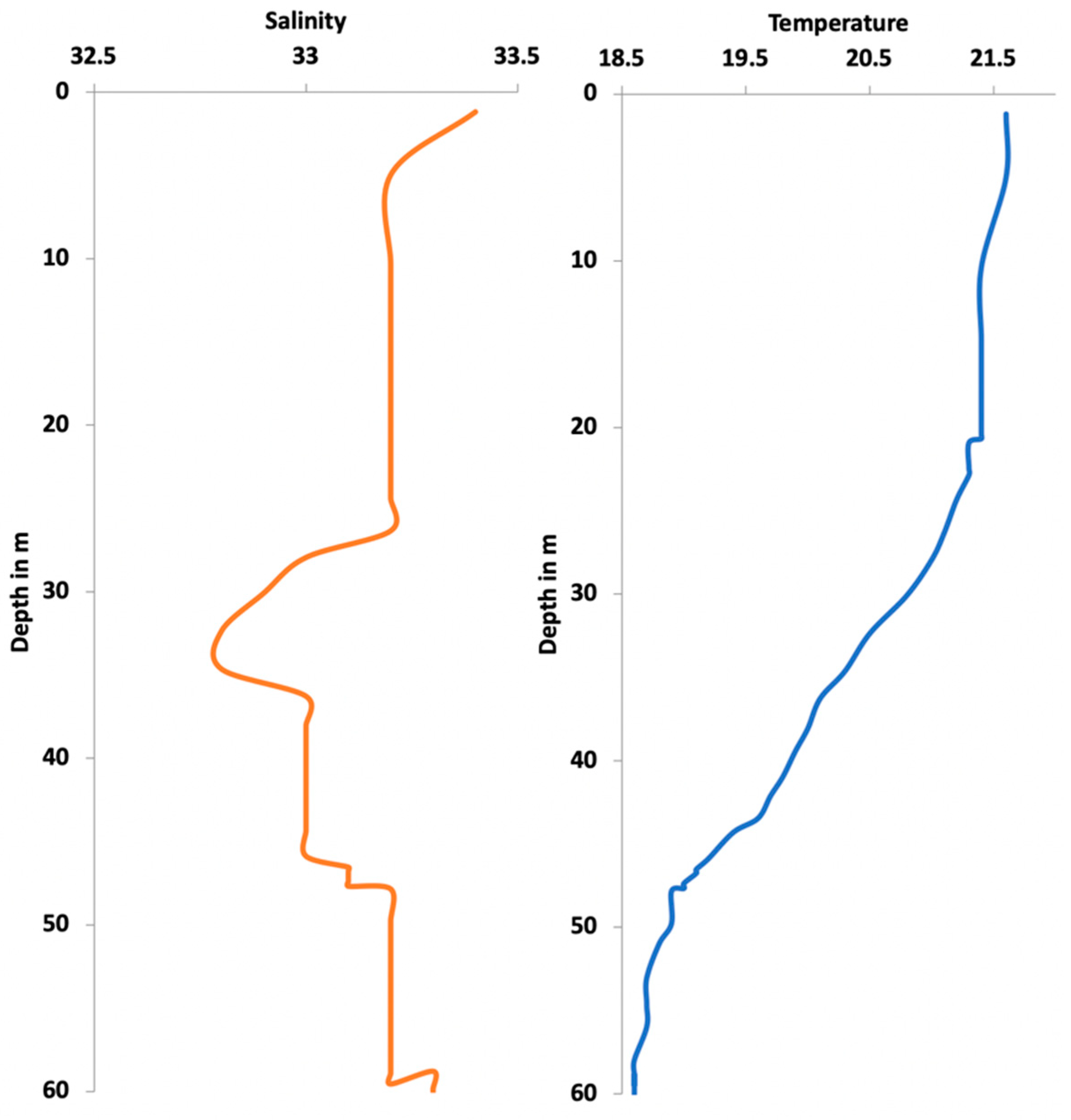
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Heylen, B.C.; Nachtsheim, D.A. Bio-telemetry as an Essential Tool in Movement Ecology and Marine Conservation. In YOUMARES 8—Oceans across Boundaries: Learning from Each Other; Springer: Cham, Switzerland, 2018; pp. 83–107. [Google Scholar]
- Watkins, W.A.; Daher, M.A.; Dimarzio, N.A.; Samuels, A.; Wartzok, D.; Fristrup, K.M.; Gannon, D.P.; Howey, P.W.; Maiefski, R.R.; Spradlin, T.R. Sperm Whale surface activity from tracking by radio and satellite tags. Mar. Mammal Sci. 1999, 15, 1158–1180. [Google Scholar] [CrossRef]
- Stewart, B.S.; Leatherwood, S.; Yochem, P.K.; Heide-Jørgensen, M.-P. Harbor seal tracking and telemetry by satellite. Mar. Mammal Sci. 1989, 5, 361–375. [Google Scholar] [CrossRef]
- Bograd, S.J.; Block, B.A.; Costa, D.P.; Godley, B.J. Biologging technologies: New tools for conservation. Introduction. Endanger. Species Res. 2010, 10, 1–7. [Google Scholar] [CrossRef]
- Stone, G.; Schubel, J.; Tausig, H. Electronic marine animal tagging: New frontier in ocean science. Oceanography 1999, 12, 24–27. [Google Scholar] [CrossRef][Green Version]
- Scott, M.D.; Wells, R.S.; Irvine, A.B.; Mate, B.R. Tagging and marking studies on small cetaceans. In The Bottlenose Dolphin; Reeves, R.R., Leatherwood, S., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 489–514. [Google Scholar]
- Guzman, H.M.; Capella, J.J. Short-term recovery of humpback whales after percutaneous satellite tagging. J. Wildl. Manag. 2017, 81, 728–733. [Google Scholar] [CrossRef]
- Szesciorka, A.R.; Calambokidis, J.; Harvey, J.T. Testing tag attachments to increase the attachment duration of archival tags on baleen whales. Anim. Biotelem. 2016, 4, 18. [Google Scholar] [CrossRef]
- Baumgartner, M.F.; Hammar, T.; Robbins, J. Development and assessment of a new dermal attachment for short-term tagging studies of baleen whales. Methods Ecol. Evol. 2015, 6, 289–297. [Google Scholar] [CrossRef]
- Tackaberry, J.E.; Cade, D.E.; Goldbogen, J.A.; Wiley, D.N.; Friedlaender, A.S.; Stimpert, A.K. From a calf’s perspective: Humpback whale nursing behavior on two US feeding grounds. PeerJ 2020, 8, e8538. [Google Scholar] [CrossRef]
- Tyson, R.B.; Friedlaender, A.S.; Ware, C.; Stimpert, A.K.; Nowacek, D.P. Synchronous mother and calf foraging behaviour in humpback whales Megaptera novaeangliae: Insights from multi-sensor suction cup tags. Mar. Ecol. Prog. Ser. 2012, 457, 209–220. [Google Scholar] [CrossRef]
- Hazen, E.L.; Friedlaender, A.S.; Thompson, M.A.; Ware, C.R.; Weinrich, M.T.; Halpin, P.N.; Wiley, D.N. Fine-scale prey aggregations and foraging ecology of humpback whales Megaptera novaeangliae. Mar. Ecol. Prog. Ser. 2009, 395, 75–89. [Google Scholar] [CrossRef]
- Goldbogen, J.A.; Calambokidis, J.; Friedlaender, A.S.; Francis, J.; DeRuiter, S.L.; Stimpert, A.K.; Falcone, E.; Southall, B.L. Underwater acrobatics by the world’s largest predator: 360° rolling manoeuvres by lunge-feeding blue whales. Biol. Lett. 2013, 9, 20120986. [Google Scholar] [CrossRef] [PubMed]
- Nelms, S.E.; Alfaro-Shigueto, J.; Arnould, J.P.Y.; Avila, I.C.; Bengtson Nash, S.; Campbell, E.; Carter, M.I.D.; Collins, T.; Currey, R.J.C.; Domit, C.; et al. Marine mammal conservation: Over the horizon. Endanger. Species Res. 2021, 44, 291–325. [Google Scholar] [CrossRef]
- Cartwright, R.; Venema, A.; Hernandez, V.; Wyels, C.; Cesere, J.; Cesere, D. Fluctuating reproductive rates in Hawaii’s humpback whales, Megaptera novaeangliae, reflect recent climate anomalies in the North Pacific. R. Soc. Open Sci. 2019, 6, 181463. [Google Scholar] [CrossRef] [PubMed]
- Garrigue, C.; Clapham, P.J.; Geyer, Y.; Kennedy, A.S.; Zerbini, A.N. Satellite tracking reveals novel migratory patterns and the importance of seamounts for endangered South Pacific humpback whales. R. Soc. Open Sci. 2015, 2, 150489. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.S.; Zerbini, A.N.; Vásquez, O.V.; Gandilhon, N.; Clapham, P.J.; Adam, O. Local and migratory movements of humpback whales (Megaptera novaeangliae) satellite-tracked in the North Atlantic Ocean. Can. J. Zool. 2013, 92, 9–18. [Google Scholar] [CrossRef]
- Stimpert, A.K.; Mattila, D.; Nosal, E.M.; Au, W.W.L. Tagging young humpback whale calves: Methodology and diving behavior. Endanger. Species Res. 2012, 19, 11–17. [Google Scholar] [CrossRef][Green Version]
- Wiley, D.; Ware, C.; Bocconcelli, A.; Cholewiak, D.; Friedlaender, A.; Thompson, M.; Weinrich, M. Underwater components of humpback whale bubble-net feeding behaviour. Behaviour 2011, 148, 575–602. [Google Scholar]
- Guzman, H.M.; Capella, J.J.; Valladares, C.; Gibbons, J.; Condit, R. Humpback whale movements in a narrow and heavily-used shipping passage, Chile. Mar. Policy 2020, 118, 103990. [Google Scholar] [CrossRef]
- Bejder, L.; Videsen, S.; Hermannsen, L.; Simon, M.; Hanf, D.; Madsen, P.T. Low energy expenditure and resting behaviour of humpback whale mother-calf pairs highlights conservation importance of sheltered breeding areas. Sci. Rep. 2019, 9, 771. [Google Scholar] [CrossRef]
- Calambokidis, J.; Fahlbusch, J.A.; Szesciorka, A.R.; Southall, B.L.; Cade, D.E.; Friedlaender, A.S.; Goldbogen, J.A. Differential Vulnerability to Ship Strikes between Day and Night for Blue, Fin, and Humpback Whales Based on Dive and Movement Data From Medium Duration Archival Tags. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Garrigue, C.; Zerbini, A.N.; Geyer, Y.; Heide-JØrgensen, M.-P.; Hanaoka, W.; Clapham, P. Movements of satellite-monitored humpback whales from New Caledonia. J. Mammal. 2010, 91, 109–115. [Google Scholar] [CrossRef][Green Version]
- Garrigue, C.; Derville, S.; Bonneville, C.; Baker, C.S.; Cheeseman, T.; Millet, L.; Paton, D.; Steel, D. Searching for humpback whales in a historical whaling hotspot of the Coral Sea, South Pacific. Endanger. Species Res. 2020, 42, 67–82. [Google Scholar] [CrossRef]
- Weinstein, B.G.; Double, M.; Gales, N.; Johnston, D.W.; Friedlaender, A.S. Identifying overlap between humpback whale foraging grounds and the Antarctic krill fishery. Biol. Conserv. 2017, 210, 184–191. [Google Scholar] [CrossRef]
- Cerchio, S.; Trudelle, L.; Zerbini, A.N.; Charrassin, J.B.; Geyer, Y.; Mayer, F.X.; Andrianarivelo, N.; Jung, J.L.; Adam, O.; Rosenbaum, H.C. Satellite telemetry of humpback whales off Madagascar reveals insights on breeding behavior and long-range movements within the southwest Indian Ocean. Mar. Ecol. Prog. Ser. 2016, 562, 193–209. [Google Scholar] [CrossRef]
- Reinke, J.; Lemckert, C.; Meynecke, J.-O. Coastal fronts utilized by migrating humpback whales, Megaptera novaeangliae, on the Gold Coast, Australia. J. Coast. Res. 2016, 75, 552–557. [Google Scholar] [CrossRef]
- Hazel, J. Evaluation of fast-acquisition GPS in stationary tests and fine-scale tracking of green turtles. J. Exp. Mar. Biol. Ecol. 2009, 374, 58–68. [Google Scholar] [CrossRef]
- Costa, D.P.; Robinson, P.W.; Arnould, J.P.Y.; Harrison, A.-L.; Simmons, S.E.; Hassrick, J.L.; Hoskins, A.J.; Kirkman, S.P.; Oosthuizen, H.; Villegas-Amtmann, S.; et al. Accuracy of ARGOS Locations of Pinnipeds at-Sea Estimated Using Fastloc GPS. PLoS ONE 2010, 5, e8677. [Google Scholar] [CrossRef]
- Hazen, E.L.; Maxwell, S.M.; Bailey, H.; Bograd, S.J.; Hamann, M.; Gaspar, P.; Godley, B.J.; Shillinger, G.L. Ontogeny in marine tagging and tracking science: Technologies and data gaps. Mar. Ecol. Prog. Ser. 2012, 457, 221–240. [Google Scholar] [CrossRef]
- Dujon, A.M.; Lindstrom, R.T.; Hays, G.C. The accuracy of Fastloc-GPS locations and implications for animal tracking. Methods Ecol. Evol. 2014, 5, 1162–1169. [Google Scholar] [CrossRef]
- Wilson, K.; Littnan, C.; Halpin, P.; Read, A. Integrating multiple technologies to understand the foraging behavior of Hawaiian monk seals. R. Soc. Open Sci. 2017, 4, 160703. [Google Scholar] [CrossRef] [PubMed]
- McConnell, B.; Beaton, R.; Bryant, E.; Hunter, C.; Lovell, P.; Hall, A. Phoning home—A new GSM mobile phone telemetry system to collect mark-recapture data. Mar. Mammal Sci. 2004, 20, 274–283. [Google Scholar] [CrossRef]
- Senko, J.F.; Megill, W.M.; Brooks, L.B.; Templeton, R.P.; Koch, V. Developing low-cost tags: Assessing the ecological impacts of tethered tag technology on host species. Endanger. Species Res. 2019, 39, 255–268. [Google Scholar] [CrossRef]
- Goodyear, I. A new radio tag; the Remora, and behavior of a humpback whale(Megaptcra novaeangliae). Cetacean Res. 1981, 2, 1–2. [Google Scholar]
- Johnson, M.; Tyack, P.; Nowacek, D.; Shorter, A. A digital acoustic recording tag for measuring the response of marine mammals to sound. J. Acoust. Soc. Am. 2000, 108, 2582–2583. [Google Scholar] [CrossRef]
- Friedlaender, A.S.; Goldbogen, J.A.; Nowacek, D.P.; Read, A.J.; Johnston, D.; Gales, N. Feeding rates and under-ice foraging strategies of the smallest lunge filter feeder, the Antarctic minke whale (Balaenoptera bonaerensis). J. Exp. Biol. 2014, 217, 2851. [Google Scholar] [CrossRef]
- Baird, R. Studying diving behavior of whales and dolphins using suction-cup attached tags. Whalewatcher 1998, 31, 3–7. [Google Scholar]
- Johnson, M.P.; Tyack, P.L. A digital acoustic recording tag for measuring the response of wild marine mammals to sound. IEEE J. Oceanic Eng. 2003, 28, 3–12. [Google Scholar] [CrossRef]
- Goldbogen, J.A.; Friedlaender, A.S.; Calambokidis, J.; McKenna, M.F.; Simon, M.; Nowacek, D.P. Integrative Approaches to the Study of Baleen Whale Diving Behavior, Feeding Performance, and Foraging Ecology. BioScience 2013, 63, 90–100. [Google Scholar] [CrossRef]
- Meynecke, J.O.; Vindenes, S.; Teixeira, D. Monitoring humpback whale (Megaptera novaeangliae) behaviour in a highly urbanised coastline: Gold Coast, Australia. In Global Challenges in Integrated Coastal Zone Management; Moksness, E., Dahl, E., Støttrup, J., Eds.; Wiley: Chichester, UK, 2013; pp. 101–113. [Google Scholar]
- Torre-Williams, L.; Martinez, E.; Meynecke, J.O.; Reinke, J.; Stockin, K.A. Presence of newborn humpback whale (Megaptera novaeangliae) calves in Gold Coast Bay, Australia. Mar. Freshwater Behav. Physiol. 2019, 52, 199–216. [Google Scholar] [CrossRef]
- McCulloch, S.; Meynecke, J.O.; Franklin, T.; Franklin, W.; Chauvenet, A.L.M. Humpback whale (Megaptera novaeangliae) behaviour determines habitat use in two Australian bays. Mar. Freshwater Res. 2021. [Google Scholar] [CrossRef]
- Cade, D.E.; Seakamela, S.M.; Findlay, K.P.; Fukunaga, J.; Kahane-Rapport, S.R.; Warren, J.D.; Calambokidis, J.; Fahlbusch, J.A.; Friedlaender, A.S.; Hazen, E.L.; et al. Predator-scale spatial analysis of intra-patch prey distribution reveals the energetic drivers of rorqual whale super-group formation. Funct. Ecol. 2021. [Google Scholar] [CrossRef]
- Dulau, V.; Pinet, P.; Geyer, Y.; Fayan, J.; Mongin, P.; Cottarel, G.; Zerbini, A.; Cerchio, S. Continuous movement behavior of humpback whales during the breeding season in the southwest Indian Ocean: On the road again! Mov. Ecol. 2017, 5, 11. [Google Scholar] [CrossRef] [PubMed]
- Félix, F.; Novillo, J. Structure and dynamics of humpback whales competitive groups in Ecuador. Anim. Behav. Cogn. 2015, 2, 56–70. [Google Scholar] [CrossRef]
- Mul, E.; Blanchet, M.-A.; Biuw, M.; Rikardsen, A. Implications of tag positioning and performance on the analysis of cetacean movement. Anim. Biotelem. 2019, 7, 11. [Google Scholar] [CrossRef]
- Weinstein, B.G.; Friedlaender, A.S. Dynamic foraging of a top predator in a seasonal polar marine environment. Oecologia 2017, 185, 427–435. [Google Scholar] [CrossRef]
- Félix, F.; Botero-Acosta, N. Distribution and behaviour of humpback whale mother–calf pairs during the breeding season off Ecuador. Mar. Ecol. Prog. Ser. 2011, 426, 277–287. [Google Scholar] [CrossRef][Green Version]
- Harcourt, R.; Sequeira, A.M.M.; Zhang, X.; Roquet, F.; Komatsu, K.; Heupel, M.; McMahon, C.; Whoriskey, F.; Meekan, M.; Carroll, G.; et al. Animal-Borne Telemetry: An Integral Component of the Ocean Observing Toolkit. Front. Mar. Sci. 2019, 6, 326. [Google Scholar] [CrossRef]
- Andrews, R.D.; Baird, R.W.; Calambokidis, J.; Goertz, C.E.C.; Gulland, F.M.D.; Heide-Jorgensen, M.P.; Hooker, S.K.; Johnson, M.; Mate, B.; Mitani, Y.; et al. Best practice guidelines for cetacean tagging. J. Cetacean Res. Manag. 2019, 20, 27–66. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meynecke, J.-O.; Liebsch, N. Asset Tracking Whales—First Deployment of a Custom-Made GPS/GSM Suction Cup Tag on Migrating Humpback Whales. J. Mar. Sci. Eng. 2021, 9, 597. https://doi.org/10.3390/jmse9060597
Meynecke J-O, Liebsch N. Asset Tracking Whales—First Deployment of a Custom-Made GPS/GSM Suction Cup Tag on Migrating Humpback Whales. Journal of Marine Science and Engineering. 2021; 9(6):597. https://doi.org/10.3390/jmse9060597
Chicago/Turabian StyleMeynecke, Jan-Olaf, and Nikolai Liebsch. 2021. "Asset Tracking Whales—First Deployment of a Custom-Made GPS/GSM Suction Cup Tag on Migrating Humpback Whales" Journal of Marine Science and Engineering 9, no. 6: 597. https://doi.org/10.3390/jmse9060597
APA StyleMeynecke, J.-O., & Liebsch, N. (2021). Asset Tracking Whales—First Deployment of a Custom-Made GPS/GSM Suction Cup Tag on Migrating Humpback Whales. Journal of Marine Science and Engineering, 9(6), 597. https://doi.org/10.3390/jmse9060597




