Deep Eutectic Solvents Based Ultrasonic Extraction of Polysaccharides from Edible Brown Seaweed Sargassum horneri
Abstract
1. Introduction
2. Results and Discussion
2.1. Optimization of the DESs-Assisted Ultrasonic Extraction of Polysaccharides
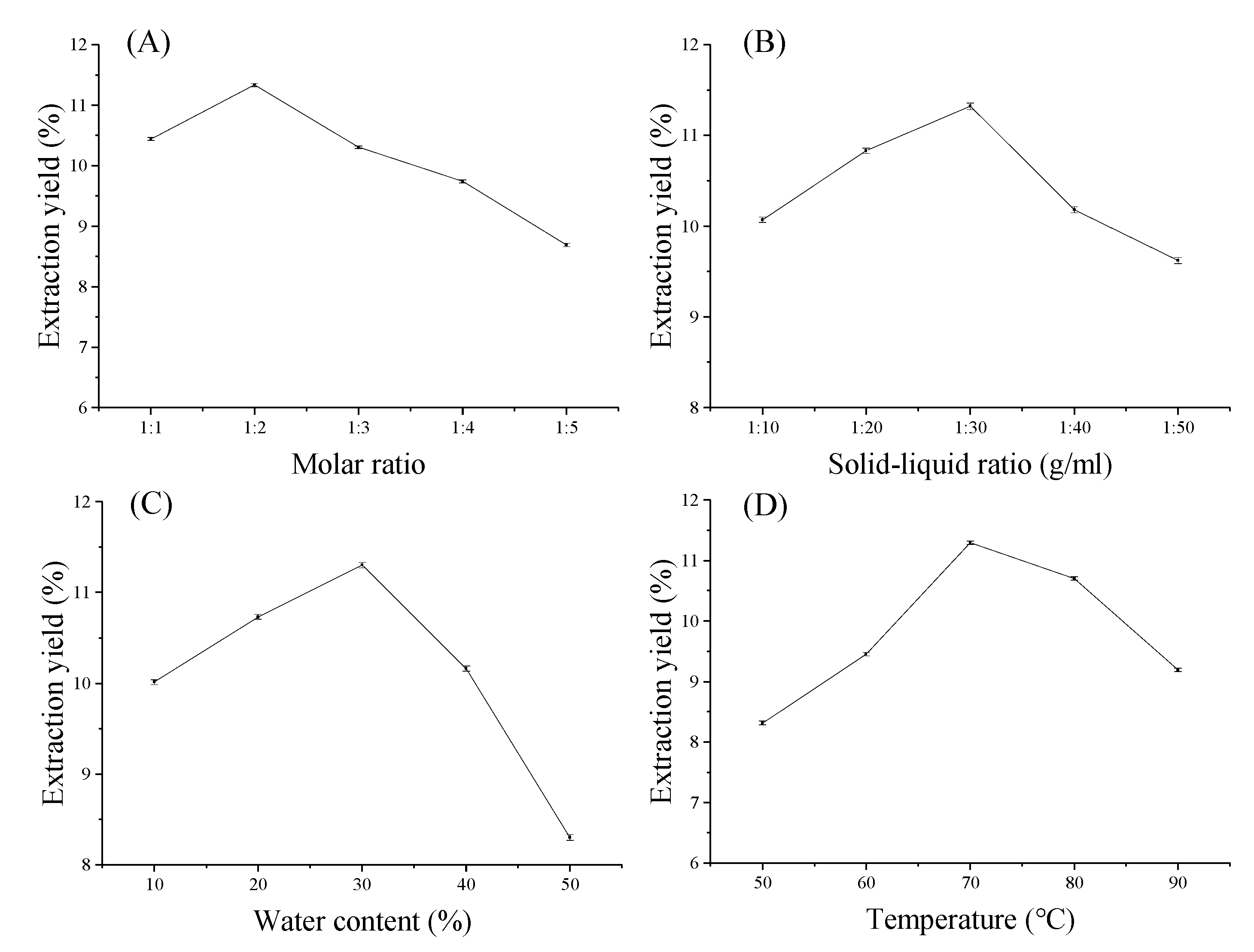
2.1.1. Effect of Molar Composition of Studied DESs
2.1.2. Effect of Solid–Liquid Ratio
2.1.3. Effect of Water Content
2.1.4. Effect of Temperature
2.1.5. Comparison with the Conventional Hot Water Extraction
2.2. Characterization of Extracted Polysaccharides
2.2.1. Infrared Spectrometry Analysis
2.2.2. X-Ray Diffraction Analysis
2.3. Antioxidant Activity of Extracted Polysaccharides
3. Materials and Methods
3.1. Materials
3.2. Ultrasonic-Assisted Extraction of Polysaccharides Using DESs
3.2.1. Preparation of DESs
3.2.2. Ultrasonic-Assisted Extraction of Polysaccharides by DESs
3.3. Conventional Hot Water Extraction Method
3.4. Characterization of Extracted Polysaccharides
3.5. Determination of Antioxidant Activity of Extracted Polysaccharides
3.5.1. DPPH Free Radical Clearance Yield Determination
3.5.2. ABTS Free Radical Scavenging Yield Determination
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Abdel-Fattah, A.F.; Hussein, M.D.; Salem, H.M. Some structural features of sargassan, a sulphated heteropolysaccharide from Sargassum linifolium. Carbohyd. Res. 1974, 12, 1995–1998. [Google Scholar] [CrossRef]
- Pujol, C.A.; Ray, S.; Ray, B.; Damonte, E.B. Antiviral activity against dengue virus of diverse classes of algal sulfated polysaccharides. Int. J. Biol. Macromol. 2012, 51, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sun, Y.; Huang, G. Preparation and antioxidant activities of important traditional plant polysaccharides. Int. J. Biol. Macromol. 2018, 111, 780–786. [Google Scholar] [CrossRef]
- Tang, W.Y.; Li, G.Z.; Chen, B.Q.; Zhu, T.; Kyung, H.R. Evaluating ternary deep eutectic solvents as novel media for extraction of flavonoids from Ginkgo biloba. Sep. Sci. Technol. 2017, 52, 91–99. [Google Scholar] [CrossRef]
- Bubalo, M.C.; Ćurko, N.; Tomašević, M.; Ganić, K.K.; Redovniković, I.R. Green extraction of grape skin phenolics by using deep eutectic solvents. Food. Chem. 2016, 200, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Ventura, S.P.M.; Silva, F.A.E.; Quental, M.V.; Mondal, D.; Freire, M.G.; Coutinho, J.A.P. Ionic-liquid-mediated extraction and separation processes for bioactive compounds: Past, present, and future trends. Chem. Rev. 2017, 117, 6984–7052. [Google Scholar] [CrossRef]
- Passos, H.; Freire, M.G.; Coutinho, J.A. Ionic liquid solutions as extractive solvents for value-added compounds from biomass. Green Chem. 2015, 46, 4786–4815. [Google Scholar] [CrossRef]
- Gao, F.; Liu, L.L.; Tang, W.Y.; Kyung, H.R.; Zhu, T. Optimization of chromatographic behaviors of quercetin using choline chloride-based deep eutectic solvents as HPLC mobile phase additives. Sep. Sci. Technol. 2018, 53, 397–403. [Google Scholar] [CrossRef]
- Hayyan, M.; Looi, C.Y.; Hayyan, A.; Wong, W.F.; Hashim, M.A. In vitro and in vivo toxicity profiling of ammonium-based deep eutectic solvents. PLoS ONE 2015, 10, e0117934. [Google Scholar] [CrossRef]
- Juneidi, I.; Hayyan, M.; Mohd Ali, O. Toxicity profile of choline chloride-based deep eutectic solvents for fungi andcyprinus carpiofish. Environ. Sci. Pollut. Res. 2016, 23, 7648–7659. [Google Scholar] [CrossRef]
- Abbott, A.P.; Ahmed, E.I.; Harris, R.C.; Ryder, K.S. Evaluating water miscible deep eutectic solvents (DESs) and ionic liquids as potential lubricants. Green Chem. 2014, 16, 4156–4161. [Google Scholar] [CrossRef]
- Kareem, M.A.; Mjalli, F.S.; Hashim, M.A.; Alnashef, I.M. Phosphonium-based ionic liquids analogues and their physical properties. J. Chem. Eng. Data 2010, 55, 4632–4637. [Google Scholar] [CrossRef]
- Zdanowicz, M.; Wilpiszewska, K.; Spychaj, T. Deep eutectic solvents for polysaccharides processing. A review. Carbohyd. Polym. 2018, 200, 361–380. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Dou, L.L.; Guo, L.; Li, P.; Liu, E. Comprehensive evaluation of deep eutectic solvents in extraction of bioactive natural products. Acs Sustain. Chem. Eng. 2016, 4, 2005–2411. [Google Scholar] [CrossRef]
- Das, A.K.; Sharma, M.; Mondal, D.; Prasad, K. Deep eutectic solvents as efficient solvent system for the extraction of κ-carrageenan from Kappaphycus alvarezii. Carbohyd. Polym. 2016, 136, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Duan, M.H.; Yao, X.H.; Zhang, Y.H.; Zhao, C.J.; Zu, Y.G. Green extraction of five target phenolic acids from Lonicerae japonicae flos with deep eutectic solvent. Sep. Purif. Technol. 2016, 157, 249–257. [Google Scholar] [CrossRef]
- Tan, T.; Zhang, M.; Wan, Y.; Qiu, H. Utilization of deep eutectic solvents as novel mobile phase additives for improving the separation of bioactive quaternary alkaloids. Talanta 2016, 149, 85–90. [Google Scholar] [CrossRef]
- Chen, C.; You, L.J.; Abbasi, A.M.; Fu, X.; Liu, R.H. Optimization for ultrasound extraction of polysaccharides from mulberry fruits with antioxidant and hyperglycemic activity in vitro. Carbohyd. Polym. 2015, 130, 122–132. [Google Scholar] [CrossRef]
- Lu, W.; Alam, M.A.; Pan, Y.; Wu, J.; Wang, Z.; Yuan, Z. A new approach of microalgal biomass pretreatment using deep eutectic solvents for enhanced lipid recovery for biodiesel production. Bioresour. Technol. 2016, 218, 123–128. [Google Scholar] [CrossRef]
- Dai, Y.; Van, S.J.; Witkamp, G.J.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents as new potential media for green technology. Anal. Chim. Acta 2013, 766, 61–68. [Google Scholar] [CrossRef]
- Wang, M.; Wang, J.; Zhang, Y.; Xia, Q.; Bi, W.; Yang, X. Fast environment-friendly ball mill-assisted deep eutectic solvent-based extraction of natural products. J. Chromatogr. A 2016, 1443, 262–266. [Google Scholar] [CrossRef]
- Gutiérrez, M.C.; Ferrer, M.L.; Mateo, C.R.; Del, M.F. Freeze-drying of aqueous solutions of deep eutectic solvents: A suitable approach to deep eutectic suspensions of self-assembled structures. Langmuir 2009, 25, 5509–5515. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Yuan, L.; Hang, D.; Liu, Z.; Li, S.; Yang, S. Optimization of ultrasonic extraction process of polysaccharides from Ornithogalum caudatum ait and evaluation of its biological activities. Ultrason. Sonochem. 2012, 19, 1160–1168. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Lee, K.Y.; Lee, H.G. Chemical composition and physicochemical properties of barley dietary fiber by chemical modification. Int. J. Biol. Macromol. 2013, 60, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Xie, Y.; Sun, S.; Sun, X.; Ren, F.; Shi, Q. Chemical analysis of astragalus mongholicus polysaccharides and antioxidant activity of the polysaccharides. Carbohyd. Polym. 2010, 82, 636–640. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, Z.W. Soluble dietary fiber from canna edulis ker by-product and its physicochemical properties. Carbohyd. Polym. 2013, 92, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Ifuku, S.; Nogi, M.; Abe, K.; Yoshioka, M.; Morimoto, M.; Saimoto, H. Preparation of chitin nanofibers with a uniform width as alpha-chitin from crab shells. Biomacromolecules 2009, 10, 1584–1588. [Google Scholar] [CrossRef]
- Zhu, Y.; Xiong, J.; Tang, Y.; Yu, Z. Eis study on failure process of two polyurethane composite coatings. Prog. Org. Coat. 2010, 69, 7–11. [Google Scholar] [CrossRef]
- Huang, W.C.; Zhao, D.; Guo, N.; Xue, C.; Mao, X. Green and facile production of chitin from crustacean shells using a natural deep eutectic solvent. J. Agric. Food. Chem. 2018, 66, 11897–11901. [Google Scholar] [CrossRef]
- French, A.D.; And, J.W.B. Computer Modeling of Carbohydrates. ACS Sym. Ser. 1990, 430, 20–30. [Google Scholar]
- Fenglin, H.; Ruili, L.; Bao, H.; Liang, M. Free radical scavenging activity of extracts prepared from fresh leaves of selected chinese medicinal plants. Fitoterapia 2004, 75, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Guan, X.; Liu, W.; Zhang, X.; Yan, W.; Yao, W. Characterization and antioxidant activity of a polysaccharide extracted from Sarcandra glabra. Carbohyd. Polym. 2012, 90, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food. Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Methods for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Wang, S.; Dong, X.; Tong, J. Optimization of enzyme-assisted extraction of polysaccharides from alfalfa and its antioxidant activity. Int. J. Biol. Macromol. 2013, 62, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Shao, P.; Chen, X.; Sun, P. Chemical characterization, antioxidant and antitumor activity of sulfated polysaccharide from Sargassum horneri. Carbohyd. Polym. 2014, 105, 260–269. [Google Scholar] [CrossRef]
- Segal, L.; Creely, J.J.; Martin, A.E.; Conrad, C.M. An empirical method for estimating the degree of crystallinity of native cellulose using the x-ray diffractometer. Text. Res. J. 1959, 29, 786–794. [Google Scholar] [CrossRef]
- Lee, B.W.; Jin, H.L.; Sang, W.G.; Moon, Y.H.; Park, K.H. Selective ABTS radical-scavenging activity of prenylated flavonoids from Cudrania tricuspidata. Biosci. Biotech. Biochem. 2006, 70, 427–432. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nie, J.; Chen, D.; Lu, Y. Deep Eutectic Solvents Based Ultrasonic Extraction of Polysaccharides from Edible Brown Seaweed Sargassum horneri. J. Mar. Sci. Eng. 2020, 8, 440. https://doi.org/10.3390/jmse8060440
Nie J, Chen D, Lu Y. Deep Eutectic Solvents Based Ultrasonic Extraction of Polysaccharides from Edible Brown Seaweed Sargassum horneri. Journal of Marine Science and Engineering. 2020; 8(6):440. https://doi.org/10.3390/jmse8060440
Chicago/Turabian StyleNie, Jinggui, Danting Chen, and Yanbin Lu. 2020. "Deep Eutectic Solvents Based Ultrasonic Extraction of Polysaccharides from Edible Brown Seaweed Sargassum horneri" Journal of Marine Science and Engineering 8, no. 6: 440. https://doi.org/10.3390/jmse8060440
APA StyleNie, J., Chen, D., & Lu, Y. (2020). Deep Eutectic Solvents Based Ultrasonic Extraction of Polysaccharides from Edible Brown Seaweed Sargassum horneri. Journal of Marine Science and Engineering, 8(6), 440. https://doi.org/10.3390/jmse8060440





