Review of the Scientific Literature on Biology, Ecology, and Aspects Related to the Fishing Sector of the Striped Venus (Chamelea gallina) in Northern Adriatic Sea
Abstract
:1. Introduction
2. Growth and Reproduction of Chamelea gallina
- -
- The analysis of the modal components in the frequency distribution linking an age to the different pseudocohorts identified in the size distributions and whose evolution was observed over time. This method is widely used in older studies in the Mediterranean and Atlantic [12,43]; it is currently supported by other methods to allow comparisons and validations;
- -
- Analysis of the outer growth rings of the shell. The formation of the outer rings occurs annually in winter, during a period of growth slowdown, and is considered by many authors to be rapidly applicable [14,26]. However, as mentioned above, this method could be flawed considering what Gizzi et al. [36] found regarding the influence of temperature on the development of the shell and consequently on its appearance;
- -
- Microscopic analysis of the deposition lines inside the shell with counting of the annual cracks [48,53,54]. This approach overcomes the interpretation difficulties of the previous method. However, even in this case, the influence of solar radiation on macroporosity and fragility of the shell must be taken into account [36,50].
3. Distribution and Habitat of Chamelea gallina
- -
- Reduction in the salinity of the estuarine water due to increased freshwater inflow, which may have altered the osmotic balance of molluscs;
- -
- Discharge of toxins by industry and factories (e.g., mills);
- -
- Leaching from agricultural lands and associated inputs of pesticides to water bodies;
- -
- Severe storms;
- -
- Oxygen deficiency (anoxia) or low oxygen concentration (hypoxia);
- -
- Temperature fluctuations;
- -
- Increased water turbidity due to elevated suspended sediment concentrations, a consequence of the presence of silt, a material of terrestrial origin whose transport to the sea via waterways increases significantly after abundant rainfall;
- -
- Increased turbidity of the water due to the resuspension of sediments by wave action;
- -
- Direction and intensity of ocean currents, which affect the transport and dispersal of species in the planktonic larval stage (i.e., veligers) [66].
4. Survival, Production Decline, Local Mass Mortality, and Fishery-Induced Mortality for Released Undersized Individuals
- -
- The stress to which the bivalves were subjected after capture, which in turn was divided into two categories (hydraulic bivalve dredges: specimens collected in the collection tank, marked “D”; hydraulic dredges plus sieve sorting: specimens collected before returning to the sea, marked “S”);
- -
- Temperature, checking survival at +12 °C and at +20 °C.
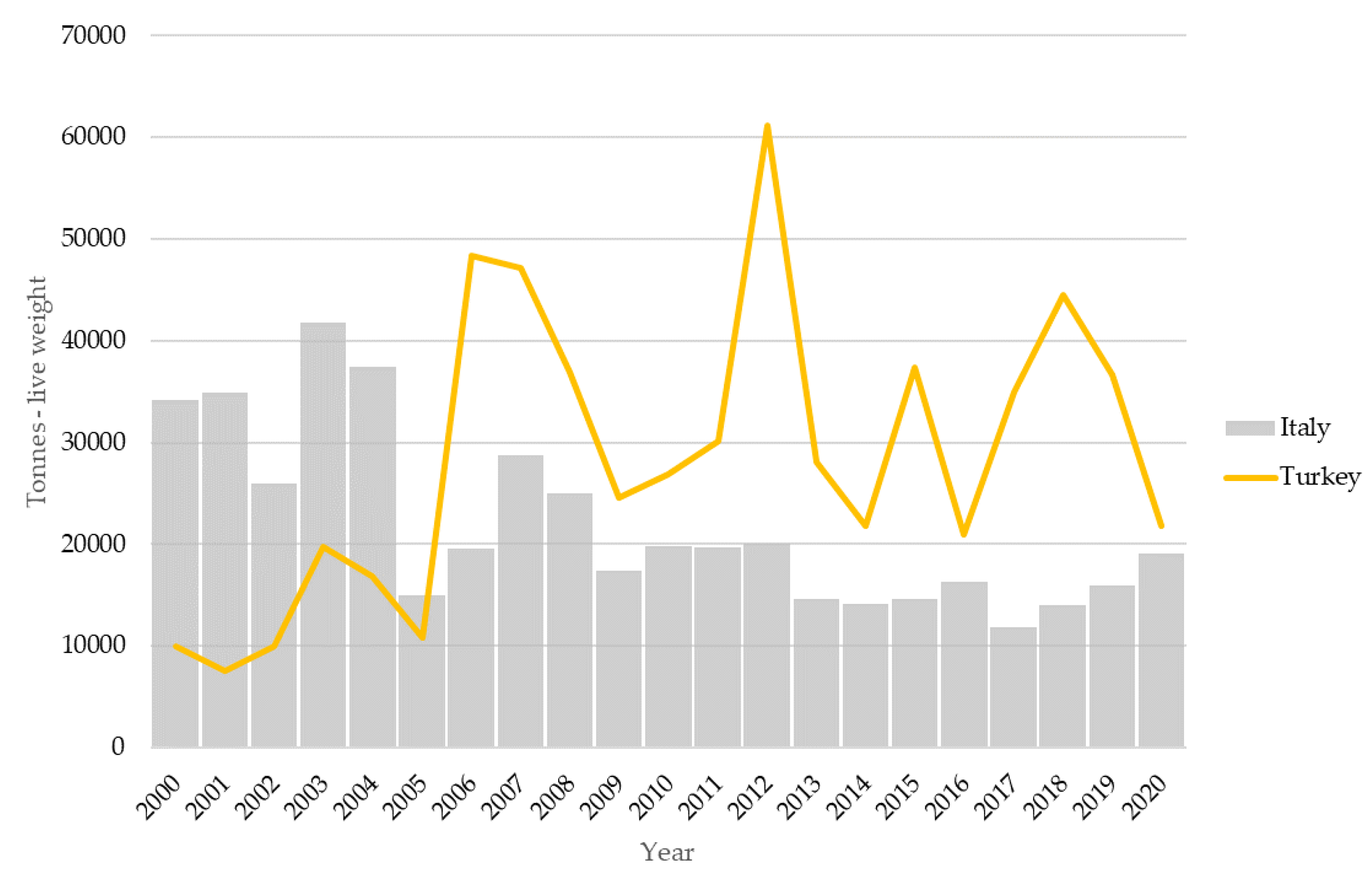
5. Fishing Methods and Fisheries
6. Status of the Chamelea gallina Stocks in North Adriatic
- -
- Class 0.1–1.5 cm (0+ specimens in I° year): 40.4%;
- -
- Class 1.6–1.9 cm (1+ specimens in II° year): 26.2%;
- -
- Class 2.0–2.4 cm (1+ specimens in II° year and subcategory of trade): 30.8%;
- -
- Class 2.5 cm (2+ specimens in III° year and tradeable): 2.6%.
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Turolla, E. La venericoltura in Italia. In Estado Actual Del Cultivo Y Manejo De Moluscos Bivalvos Y Su Proyecci On Futura. Factores Que Afectan Su Sustentabilidad En America Latina; Lovatelli, A., Farıas, A., Uriarte, I., Eds.; FAO: Roma, Italy, 2008; pp. 177–188. [Google Scholar]
- FAO. 2018. Available online: http://www.fao.org/in-action/globefish/market-reports/resource-detail/en/c/1156024/ (accessed on 15 July 2022).
- FAO. 2021. Available online: http://www.fao.org/fishery/facp/ITA/en (accessed on 15 July 2022).
- FAO. 2022. Available online: https://www.fao.org/fishery/en/culturedspecies/ruditapes_philippinarum (accessed on 15 July 2022).
- FAO. FishStatJ, a Tool for Fishery Statistics Analysis v. 4.02.06. 2022. Available online: https://www.fao.org/fishery/en/statistics/software/fishstatj/en (accessed on 15 July 2022).
- Turolla, E. L’allevamento della Vongola Verace nel Delta del Po; Grafiche Adriatica: Taglio di Po, Italy, 2008; pp. 1–111. [Google Scholar]
- Trevisan, G. Le vongole dell’Alto Adriatico tra Ambiente e Mercato; Franco Angeli: Milan, Italy, 2011; pp. 1–205. [Google Scholar]
- Petetta, A.; Herrmann, B.; Virgili, M.; Bargione, G.; Vasapollo, C.; Lucchetti, A. Dredge selectivity in a Mediterranean striped venus clam (Chamelea gallina) fishery. Fisher. Res. 2021, 238, 105895. [Google Scholar] [CrossRef]
- Fischer, W.; Bauchot, M.L.; Schneider, M. Fiche FAO D’identification des Especes Pour les Besoins de la Peche, Rev. 1 Mediterranee et Mer Noire (Zone de Peche 37); FAO: Rome, Italy, 1987. [Google Scholar]
- Bombace, G.; Lucchetti, A. Elementi di Biologia Della Pesca; Edizioni Edagricole, Il Sole 24 Ore; Edagricole: Milano, Italy, 2011; 383p. [Google Scholar]
- Rocha, V.P.; Matthews-Cascon, H. Comparative anatomy of Chioninae and Venerinae species (Mollusca: Bivalvia: Veneridae). Rev. Nord. Zool. 2019, 12, 123–146. [Google Scholar]
- Froglia, C. Clam fisheries with hydraulic dredges in the Adriatic Sea. In Marine Invertebrate Fisheries; Caddy, J., Ed.; Wiley: Boca Ration, NJ, USA, 1989; pp. 507–524. [Google Scholar]
- Romanelli, M.; Cordisco, C.A.; Giovanardi, O. The long-term decline of the Chamelea gallina L. (Bivalvia: Veneridae) clam fishery in the Adriatic Sea: Is a synthesis possible. Acta Adriat. 2009, 50, 171–205. [Google Scholar]
- Marano, G.; Casavola, N.; Saracino, C.; Rizzi, E. Riproduzione e crescita di Chamelea gallina (L.) e Venus verrucosa (L.) (Bivalvia: Veneridae) nel Basso Adriatico. Mem. Biol. Mar. Ocean. 1982, 12, 97–110. [Google Scholar]
- Bodoy, A. Croissance et variations saisonnières de la composition biochimique de Venus gallina dans le Golfe de Marseille (Méditerranée Occidentale). Tethys 1983, 11, 57–66. [Google Scholar]
- Ramon Herrero, M. Estudio de las poblaciones de Chamelea gallina (Linnaeus, 1758) y Donax trunculus (Linnaeus, 1758) (Mollusco: Bivalvia) en el Golfo di Valencia (Mediterraneo Occidental). Ph.D. Thesis, University of Barcelona, Barcelona, Spain, 1993. [Google Scholar]
- Erkan, M.; Sousa, M. Fine structural study of the spermatogenic cycle in Pitar rudis and Chamelea gallina (Mollusca, Bivalvia, Veneridae). Tissue Cell 2002, 34, 262–272. [Google Scholar] [CrossRef]
- Erkan, M. Ultrastructure of ovary and oogenesis in Chamelea gallina (Linné, 1758) (Bivalvia, Veneridae). Invert. Rep. Dev. 2009, 53, 201–209. [Google Scholar] [CrossRef]
- Dalgic, G.; Karayucel, S.; Okumus, I. Reproduction cycle of striped venus Chamelea gallina from the Black Sea coast of Turkey. J. An. Vet. Adv. 2009, 8, 2009–2012. [Google Scholar]
- Poggiani, L.; Piccinetti, C.; Piccinetti-Manfrin, G. Osservazioni sulla biologia dei molluschi bivalvi Venus gallina L. e Tapes aureus nell’Alto Adriatico. In Note del Laboratorio di Biologia Marina e Pesca 4; Fano: Rome, Italy, 1980; pp. 189–212. [Google Scholar]
- Salvatorelli, G. Osservazioni sul ciclo riproduttivo annuo di Venus gallina (Molluschi Lamellibranchi). Ann. Univ. Ferrara Anatom. Comp. 1967, 2, 15–22. [Google Scholar]
- De la Rua, A.R.; Prado, M.A.; Bruzon, M.A. The reproductive cycle of Chamelea gallina (L., 1758) (Mollusca: Bivalvia) in three populations from the Andalusian coast (southern Spain). Bol. Inst. Esp. Oceanogr. 2003, 19, 57. [Google Scholar]
- Delgado, M.; Silva, L.; Juárez, A. Aspects of reproduction of striped venus Chamelea gallina in the Gulf of Cádiz (SW Spain): Implications for fishery management. Fish. Res. 2013, 146, 86–95. [Google Scholar] [CrossRef]
- Gaspar, M.B.; Monteiro, C.C. Reproductive cycles of the razor clam Ensis siliqua and the clam Venus striatula off Vilamoura, Southern Portugal. J. Mar. Biol. Ass. UK 1998, 78, 1247–1258. [Google Scholar] [CrossRef]
- Joaquim, S.; Matias, D.; Matias, A.M.; Moura, P.; Roque, C.; Chícharo, L.; Gaspar, M.B. Biochemical and energy dynamics throughout the reproductive cycle of the striped venus Chamelea gallina (Mollusca, Bivalvia). Inv. Rep. Dev. Vol. 2014, 58, 284–293. [Google Scholar] [CrossRef]
- Gaspar, M.B.; Pereira, A.M.; Vasconcelos, P.; Monteiro, C.C. Age and growth of Chamelea gallina from the Algarve coast (southern Portugal): Influence of seawater temperature and gametogenic cycle on growth rate. J. Moll. Stud. 2004, 70, 371–377. [Google Scholar] [CrossRef]
- Baeta, M.; Solís, M.A.; Ballesteros, M.; Defeo, O. Long-term trends in striped venus clam (Chamelea gallina) fisheries in the western Mediterranean Sea: The case of Ebro Delta (NE Spain). Mar. Policy 2021, 134, 104798. [Google Scholar] [CrossRef]
- Froglia, C. Observations on the growth of Chamelea gallina (L.) and Ensis minor (Chenu) in the middle Adriatic. Quad. Lab Tecnol. Pesca 1975, 2, 37–48. [Google Scholar]
- MIPAAF. 2019. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/16994 (accessed on 15 July 2022).
- MIPAAF. 2019. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/13760 (accessed on 15 July 2022).
- MIPAAF. 2019. Available online: https://www.gazzettaufficiale.it/atto/serie_generale/caricaDettaglioAtto/originario?atto.dataPubblicazioneGazzetta=2020-02-21&atto.codiceRedazionale=20A00773&elenco30giorni=true (accessed on 15 July 2022).
- MIPAAF. 2019. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=CELEX:32020R0003 (accessed on 15 July 2022).
- FAO. 2019. Available online: https://leap.unep.org/countries/it/national-legislation/decree-27-december-2016-adoption-national-plan-management (accessed on 15 July 2022).
- ISPRA. 2018. Available online: https://www.isprambiente.gov.it/files2018/pubblicazioni/stato-ambiente/Rapporto_clima_2017.pdf (accessed on 15 July 2022).
- ISPRA. 2019. Available online: https://www.isprambiente.gov.it/files2020/pubblicazioni/stato-ambiente/rapporto_clima_2019-1.pdf (accessed on 15 July 2022).
- Gizzi, F.; Caccia, M.G.; Simoncini, G.A.; Mancuso, A.; Reggi, M.; Fermani, S.; Vrizi, L.; Fantazzini, P.; Stagioni, M.; Falini, G.; et al. Biochemical and energy dynamics throughout the reproductive cycle of the striped venus Chamelea gallina (Mollusca, Bivalvia). Sci. Rep. 2016, 6, 36420. [Google Scholar] [CrossRef]
- Bargione, G.; Donato, F.; Barone, G.; Virgili, M.; Penna, P.; Lucchetti, A. Chamelea gallina reproductive biology and Minimum Conservation Reference Size: Implications for fishery management in the Adriatic Sea. BMC Zool. 2021, 6, 1–16. [Google Scholar] [CrossRef]
- Vermeiji, G.J. A Natural History of Shells; Princeton University Press: Princeton, NJ, USA, 1995. [Google Scholar]
- Watson, S.A.; Peck, L.S.; Tyler, P.A.; Southgate, P.C.; Tan, K.S.; Day, R.W.; Morley, S.A. Marine invertebrate skeleton size varies with latitude, temperature and carbonate saturation: Implications for global change and ocean acidification. Glob. Chang. Biol. 2012, 18, 3026–3038. [Google Scholar] [CrossRef]
- Clarke, A. Temperature and extinction in the sea: A physiologist’s view. Paleobiology 1993, 19, 499–518. [Google Scholar] [CrossRef]
- Heilmayer, O.; Brey, T.; Pörtner, H.O. Growth efficiency and temperature in scallops: A comparative analysis of species adapted to different temperatures. Funct. Ecol. 2004, 18, 641–647. [Google Scholar] [CrossRef]
- Peck, L.S.; Powell, D.K.; Tyler, P.A. Very slow development in two Antarctic bivalve molluscs, the infaunal clam, Laternula elliptica and the scallop Adamussium colbecki. Mar. Biol. 2007, 150, 1191–1197. [Google Scholar] [CrossRef]
- Moschino, V.; Marin, M.G. Seasonal changes in physiological responses and evaluation of “well-being”in the Venus clam Chamelea gallina from the Northern Adriatic Sea. Comp. Biochem. Physiol. Part A 2006, 145, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Ramón, M.; Richardson, C.A. Age determination and shell growth of Chamelea gallina (Bivalvia: Veneridae) in the western Mediterranean. Mar. Ecol. Prog. Ser. 1992, 89, 15–23. [Google Scholar] [CrossRef]
- Häder, D.P.; Kumar, H.D.; Smith, R.C.; Worrest, R.C. Effects of solar UV radiation on aquatic ecosystems and interactions with climate change. Photochem. Photobiol. Sci. 2007, 6, 267–285. [Google Scholar] [CrossRef]
- Yukihira, H.; Klumpp, D.W.; Lucas, J.S. Comparative effects of microalgal species and food concentration on suspension feeding and energy budgets of the pearl oysters Pinctada margaritifera and P. maxima (Bivalvia: Pteriidae). Mar. Ecol. Prog. Ser. 1998, 171, 71–84. [Google Scholar] [CrossRef]
- Zavatarelli, M.; Raicich, F.; Bregant, D.; Russo, A.; Artegiani, A. Climatological biogeochemical characteristics of the Adriatic Sea. J. Mar. Syst. 1998, 18, 227–263. [Google Scholar] [CrossRef]
- Bressan, M.; Chinellato, A.; Munari, M.; Matozzo, V.; Manci, A.; Marčeta, T.; Finos, L.; Moro, I.; Pastore, P.; Badocco, D.; et al. Does seawater acidification affect survival, growth and shell integrity in bivalve juveniles? Mar. Environ. Res. 2014, 99, 136–148. [Google Scholar] [CrossRef]
- Richardson, C.A.; Seed, R.; Naylor, E. Use of internal growth bands for measuring individual and population growth rates in Mytilus edulis from offshore production platforms. Mar. Ecol. Prog. Ser. 1990, 66, 259–265. [Google Scholar] [CrossRef]
- Moura, P.; Gaspar, M.B.M.; Monteiro, C.C.C. Age determination and growth rate of a Callista chione population from the southwestern coast of Portugal. Aquat. Biol. 2009, 5, 97–106. [Google Scholar] [CrossRef]
- Mancuso, A.; Stagioni, M.; Prada, F.; Scarponi, D.; Piccinetti, C.; Goffredo, S. Environmental influence on calcification of the bivalve Chamelea gallina along a latitudinal gradient in the Adriatic Sea. Nat. Res. 2019, 9, 11198. [Google Scholar] [CrossRef] [PubMed]
- Keller, N.; Del Piero, D.; Longinelli, A. Isotopic composition, growth rates and biological behaviour of Chamelea gallina and Callista chione from the Gulf of Trieste (Italy). Mar. Biol. 2002, 140, 9–15. [Google Scholar]
- Schöne, B.R.; Giere, O. Growth increments and stable isotope variation in shells of the deep-sea hydrothermal vent bivalve mollusk Bathymodiolus brevior from the North Fiji Basin. Pacific Ocean. Deep. Res. Part Oceanogr. Res. Pap. 2005, 52, 1896–1910. [Google Scholar] [CrossRef]
- Bourget, E.; Brock, V. Short-term shell growth in bivalves: Individual, regional, and age-related variations in the rhythm of deposition of Cerastoderma (=Cardium) edule. Mar. Biol. 1990, 106, 103–108. [Google Scholar] [CrossRef]
- Cordisco, C.A.; Trotta, P.; Romanelli, M. Spawning plasticity of baby clam Chamela gallina, Linnaeus 1758. Biol. Mar. Medit. 2005, 12, 385–388. [Google Scholar]
- MEDAC. 2017. Available online: http://en.med-ac.eu/files/documentazione_pareri_lettere/2019/03/71_medac_opinion_discard_management_plan_chamelea_gallina_2019-1.pdf (accessed on 15 July 2022).
- FAO. 2017. Available online: https://leap.unep.org/countries/it/national-legislation/ministerial-decree-regulating-clam-fishing-maritime-compartments (accessed on 15 July 2022).
- Commission Delegated Regulation (EU) 2020/2237. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32020R2237 (accessed on 15 July 2022).
- PO FEAMP National Work Plan Alieutic Data Collection Manual for Sampling DRES-Dredgers Mollusc Surveys—Strategic Plan. Available online: https://datacollection.jrc.ec.europa.eu/documents/10213/1131890/Italy_WorkPlan_2017–2019.pdf/5be89aee-3ae0-4414-9c67-803778e49711?version=1.0 (accessed on 15 July 2022).
- Da Costa, M.E. Historia Naturalis Testaceorum Britanniæ, or, the British Conchology; Containing the Descriptions and Other Particulars of Natural History of the Shells of Great Britain and Ireland: Illustrated with figures. In English and French. Available online: https://www.biodiversitylibrary.org/page/13116783 (accessed on 15 July 2022).
- Backeljau, T.; Bouchet, P.; Gofas, S.; De Brunyn, L. Genetic variation, systematics and distribution of the venerid clam Chamelea Gall. J. Mar. Biol. Ass. UK 1994, 74, 211–223. [Google Scholar] [CrossRef]
- Rufino, M.M.; Gaspar, M.B.; Pereira, A.M.; Vasconcelos, P. Use of Shape to Distinguish Chamelea gallina and Chamelea striatula (Bivalvia: Veneridae): Linear and Geometric Morphometric Methods. J. Morphol. 2006, 267, 1433–1440. [Google Scholar] [CrossRef]
- Froglia, C. Clam fisheries with hydraulic dredges in the Adriatic sea. In Marine Invertebrate Fisheries: Their Assessment and Management; Caddy, J.F., Ed.; Wiley & Sons: Hoboken, NJ, USA, 1989; pp. 507–521. [Google Scholar]
- Péres, J.M.; Picard, J. New manual for benthic bionomics in the Mediterranean Sea. Trav. Stn. Marit. Endoum. 1964, 31, 1–137. [Google Scholar]
- Pampanin, D.M.; Ballarin, L.; Carotenuto, L.; Marin, M.G. Air exposure and functionality of Chamelea gallina haemocytes: Effects on haematocrit, adhesion, phagocytosis and enzyme contents. Comp. Biochem. Physiol. Part 2002, 131, 605–614. [Google Scholar] [CrossRef]
- Barillari, A.; Boldrin, A.; Mozzi, C.; Rabitti, S. Alcune relazioni tra natura dei sedimenti e presenza della vongola Chamelea gallina, nell’Alto Adriatico, presso Venezia. Att. Ist. Venet. Sci. Lett. Art. Cl Sci. Fi Mat. Nat. 1978, 137, 19–34. [Google Scholar]
- Angioni, S.A.; Giansante, C.; Ferri, N. Clam (Chamelea gallina): Evaluation of the effects of suspended solids in sea water in the bivalve mollusk Veter. It. 2010, 46, 93–99. [Google Scholar]
- Delgado, M.; Silva, L.; Moura, P.; Sánchez-Leal, R.F.; Miguel, B.G. Variation of growth performance of the striped venus clam Chamelea gallina (Mollusca: Bivalvia) (Linnaeus, 1758) in relation to environmental variables along the southern part of its geographic range. Vie Milieu Life Environ. 2015, 65, 1–10. [Google Scholar]
- Osservatorio Socio-Economico Della Pesca e dell’Acquacoltura. Available online: https://www.venetoagricoltura.org/2010/08/news (accessed on 15 July 2022).
- AGRI.TE.CO. Implementation of Urgent Nourishment Interventions on Beaches Damaged by Calamitous Events and Cleaning of the Beaches-Environmental Monitoring-Final Report for the Municipality of Grado. 2019. Available online: www.comunegrado.it (accessed on 15 July 2022).
- Morello, E.B.; Froglia, C.; Atkinson, R.J.A.; Moore, P.G. The effects of hydraulic dredging on the reburial of several molluscan species). Biol Mar. Mediterr. 2006, 13, 610–613. [Google Scholar]
- ISPRA. L’attività di Molluschicoltura in Italia. 2018. Available online: https://www.isprambiente.gov.it/files2018/eventi/cadeau/Giovanardi_ISPRA_MolluschicolturainItalia.pdf (accessed on 15 July 2022).
- Co.Ge.Mo. Monfalcone–AGRI.TE.CO. Monitoring of the Natural Banks of Bivalve Molluscs and Environmental Investigations and the Related Sedimentary Environment as Part of the Maintenance Works of the Seabed of the Terminal Stretch of the Tagliamento River and of the Lignano Canal for the Two-Year Period 2018–2019-Dredging Interventions at the Terminal Mouth of the Tagliamento River and of the Lignano Canal for the Years 2018–2019. 2018. Available online: https://www.legacoopfvg.it/cooperative/co-ge-mo-consorzio-gestione-pesca-comp-monfalcone/ (accessed on 15 July 2022).
- Co.Ge.Mo. Monfalcone–AGRI.TE.CO. Monitoring of the Natural Banks of Bivalve Molluscs and Environmental Investigations and of the Related Sedimentary Environment as Part of the Maintenance Works of the Seabed of the Terminal Stretch of the Tagliamento River and of the Lignano Canal for the Two-Year Period 2018–2020-Dredging Interventions at the Terminal Mouth of the Tagliamento River and of the Lignano Canal for the Years 2018–2019. 2019. Available online: https://www.legacoopfvg.it/cooperative/co-ge-mo-consorzio-gestione-pesca-comp-monfalcone/ (accessed on 15 July 2022).
- Currie, D.R.; Parry, G.D. Effects of scallop dredging on a soft sediment community: A large-scale experimental study. Mar. Ecol. Prog. Ser. 1996, 134, 131–150. [Google Scholar] [CrossRef]
- Kaiser, M.J.; Hill, A.S.; Ramsay, K.; Spencer, B.E.; Brand, A.R.; Veale, L.O.; Prudden, K.; Rees, E.I.S.; Munday, B.W.; Ball, B.; et al. Benthic disturbance by fishing gear in the Irish Sea: A comparison of beam trawling and scallop dredging. Aquat. Conserv. 1996, 6, 269–285. [Google Scholar] [CrossRef]
- Zajac, R.N.; Whitlatch, R.B. Community and population-level responses to disturbance in a sandflat community. J. Exp. Mar. Biol. Ecol. 2003, 294, 101–125. [Google Scholar] [CrossRef]
- ISPRA. Sustainable Use of Renewable Resources and Impact of Anthropic Activities in the Venice Lagoon and in the Upper Adriatic. 2008. Available online: https://www.isprambiente.gov.it/contentfiles/00004100/4125-attiv-antropiche-ve.pdf (accessed on 15 July 2022).
- Matozzo, V.; Monari, M.; Foschi, J.; Serrazanetti, G.P.; Cattani, O.; Marin, M.G. Effects of salinity on the clam Chamelea gallina. Part I: Alterations in immune responses. Mar. Biol. 2007, 151, 1051–1058. [Google Scholar] [CrossRef]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2000; X Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2002; pp. 1–311. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2001–2002; XI Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2003; pp. 1–61. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2004; XIII Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2005; pp. 1–55. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2005; XIV Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2006; pp. 1–56. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2006; XV Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2007; pp. 1–55. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2007; XVI Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2008; pp. 1–55. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2008; XVII Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2009; pp. 1–55. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2009; XVIII Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2010; pp. 1–55. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2010; XIX Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2011; pp. 1–55. [Google Scholar]
- IREPA. Economic Observatory on the Productive Structures of Sea Fishing in Italy 2011; XX Report; Irepa Ricerche Series; Franco Angeli: Milan, Italy, 2012; pp. 1–55. [Google Scholar]
- Biondi, S.; Del Piero, D. Survey on Chamelea gallina beds in the Lignano area (Gulf of Trieste, Adriatic Sea). Ann. Ser. Hist. Nat. 2021, 22, 35–44. [Google Scholar]
- Sala, A.; Brčić, J.; Herrmann, B.; Lucchetti, A.; Virgili, M. Assessment of size selectivity in hydraulic clam 1 dredge fisheries. Can. J. Fish. Aquat. Sci. 2017, 74, 339–348. [Google Scholar] [CrossRef]
- M.D. n. 44 of 12 January 1995. Available online: https://www.gazzettaufficiale.it/eli/id/1995/02/24/095G0070/sg (accessed on 15 July 2022).
- M.D. n. 515 of 1 December 1998. Available online: https://www.gazzettaufficiale.it/eli/id/1999/03/29/099G0131/sg (accessed on 15 July 2022).
- M.D. n. 22 of December 2000. Available online: https://www.gazzettaufficiale.it/atto/serie_generale/caricaDettaglioAtto/originario?atto.dataPubblicazioneGazzetta=2001-02-26&atto.codiceRedazionale=001A1900&elenco30giorni=false (accessed on 15 July 2022).
- M.D. n. 7 of February 2005. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/163 (accessed on 15 July 2022).
- M.D. n. 7 of February 2006. Available online: https://www.gazzettaufficiale.it/eli/id/2006/02/17/06A01528/sg (accessed on 15 July 2022).
- M.D. n. 7 of May 2012. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/5590 (accessed on 15 July 2022).
- M.D. n. 24 of July 2015. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/8949 (accessed on 15 July 2022).
- M.D. n. 27 of December 2016. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/10771 (accessed on 15 July 2022).
- Regulation (EC) n. 1967/2006. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2006:409:0009:0064:IT:PDF (accessed on 15 July 2022).
- Vaccarella, R.; Pastorelli, A.M.; Marano, G. Study on the efficiency of turbo-blowing dredges and their effect on benthic communities. Boll. Malacol. 1994, 30, 17–28. [Google Scholar]
- Morello, E.B.; Froglia, C.; Atkinson, R.J.A.; Moore, P.G. Hydraulic dredge discards of the clam (Chamelea gallina) fishery in the western Adriatic Sea, Italy. Fish. Res. 2005, 76, 430–444. [Google Scholar] [CrossRef]
- Papetti, C.; Schiavon, L.; Milan, M.; Lucassen, M.; Caccavo, J.A.; Paterno, M.; Boscari, E.; Marino, I.A.M.; Congiu, L.; Zane, L. Genetic variability of the striped Venus Chamelea gallina in the northern Adriatic Sea. Fish. Res. 2018, 201, 68–78. [Google Scholar] [CrossRef]
- M.D. n. 405 of July 2019. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/14271 (accessed on 15 July 2022).
- MIPAAF. Strategic Plan for Aquaculture in Italy 2014–2020. 2014. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/8752 (accessed on 15 July 2022).
- M.D. 24 July 2015. Available online: https://www.gazzettaufficiale.it/atto/serie_generale/caricaDettaglioAtto/originario?atto.dataPubblicazioneGazzetta=2015-08-13&atto.codiceRedazionale=15A06046 (accessed on 15 July 2022).
- Executive Decree n. 5850 of 11 March 2020. Available online: https://www.politicheagricole.it/flex/cm/pages/ServeBLOB.php/L/IT/IDPagina/15390 (accessed on 15 July 2022).
- CREA. Settore Ittico in Cifre. 2015. Available online: http://dspace.crea.gov.it/bitstream/inea/1297/1/Settore_ittico_cifre_2015_Pierangeli.pdf (accessed on 15 July 2022).
- Cardillo, C. 2015–The Community Fleet-in CREA The Fish Sector in Numbers. Available online: http://www.inea.it/pubbl/ (accessed on 23 August 2022).
- ISTAT. Statistiche Sulla Pesca in Italia, Uso Integrato di Indagini Campionarie e dati Amministrativi. 2020. Available online: https://www.istat.it/it/files/2020/05/IWP-4-2020.pdf (accessed on 15 July 2022).
- BTMI. Annuario del Settore Ittico. 2019. Available online: https://www.bmti.it/wp-content/uploads/2020/04/Le-principali-evidenze-per-il-settore-ittico-nel-2019.pdf (accessed on 15 July 2022).
- Pravoni, F.; Colla, S.; Valeri, P.; Monti, M.A. Present and future status of artisanal fisheries in the Adriatic Sea (western Mediterranean Sea). Ocean Coast. Manag. 2016, 122, 49–56. [Google Scholar]
- Reg. (CE) 853/2004. Available online: https://eur-lex.europa.eu/legal-content/IT/TXT/PDF/?uri=CELEX:32004R0853&from=HR (accessed on 15 July 2022).
- Reg. (CE) 854/2004. Available online: https://eur-lex.europa.eu/legal-content/IT/TXT/PDF/?uri=CELEX:32004R0854&from=GA (accessed on 15 July 2022).
- Arcangeli, G.; Dalla Pozza, M.; Mascarello, G.; Tiozzo, B. Bivalve Molluscs. Fruits of Our Sea. Science Notes-10, IZS Venezie. 2016. Available online: https://www.wiley.com/en-ie/Bivalve+Molluscs:+Biology,+Ecology+and+Culture-p-9780852382349 (accessed on 15 July 2022).
- Co.Ge.Mo. Monfalcone-AGRI.TE.CO. 2016. Available online: https://www.legacoopfvg.it/cooperative/co-ge-mo-consorzio-gestione-pesca-comp-monfalcone/ (accessed on 15 July 2022).
- Co.Ge.Vo. Venice and Chioggia_AGRI.TE.CO. ANNEX A DGR nr. 745 of 27 May 2016. Available online: https://bur.regione.veneto.it/BurvServices/pubblica/Download.aspx?name=745_AllegatoB_323907.pdf&type=9&storico=False (accessed on 15 July 2022).

| Survey Area | Site | Good Management | Critical Range | Prohibition of Fishing |
|---|---|---|---|---|
| g/m2 | ||||
| GSA17 | Upper-Middle Adriatic | >10.00 | 5.00–7.50 | <5.00 |
| GSA18 | Southern Adriatic | >8.00 | 4.00–6.00 | <4.00 |
| GSA9–10 | Tyrrhenian | >8.00 | 4.00–6.00 | <4.00 |
| 2002 | 2003 | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | |
|---|---|---|---|---|---|---|---|---|---|---|
| Tons | 998 | - | - | 903 | 1447 | 1245 | 1340 | 1019 | 632 | 529 |
| Revenue (M EUR) | 4.88 | - | - | 3.90 | 6.40 | 4.75 | 4.67 | 4.51 | 2.93 | 2.41 |
| 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | |
|---|---|---|---|---|---|---|
| Chamelea gallina | 201.7 | 205.3 | 130.1 | 62.6 | 2.5 | 14.6 |
| Ensis minor | 115.8 | 62.4 | 18.0 | 19.9 | 23.9 | 4.0 |
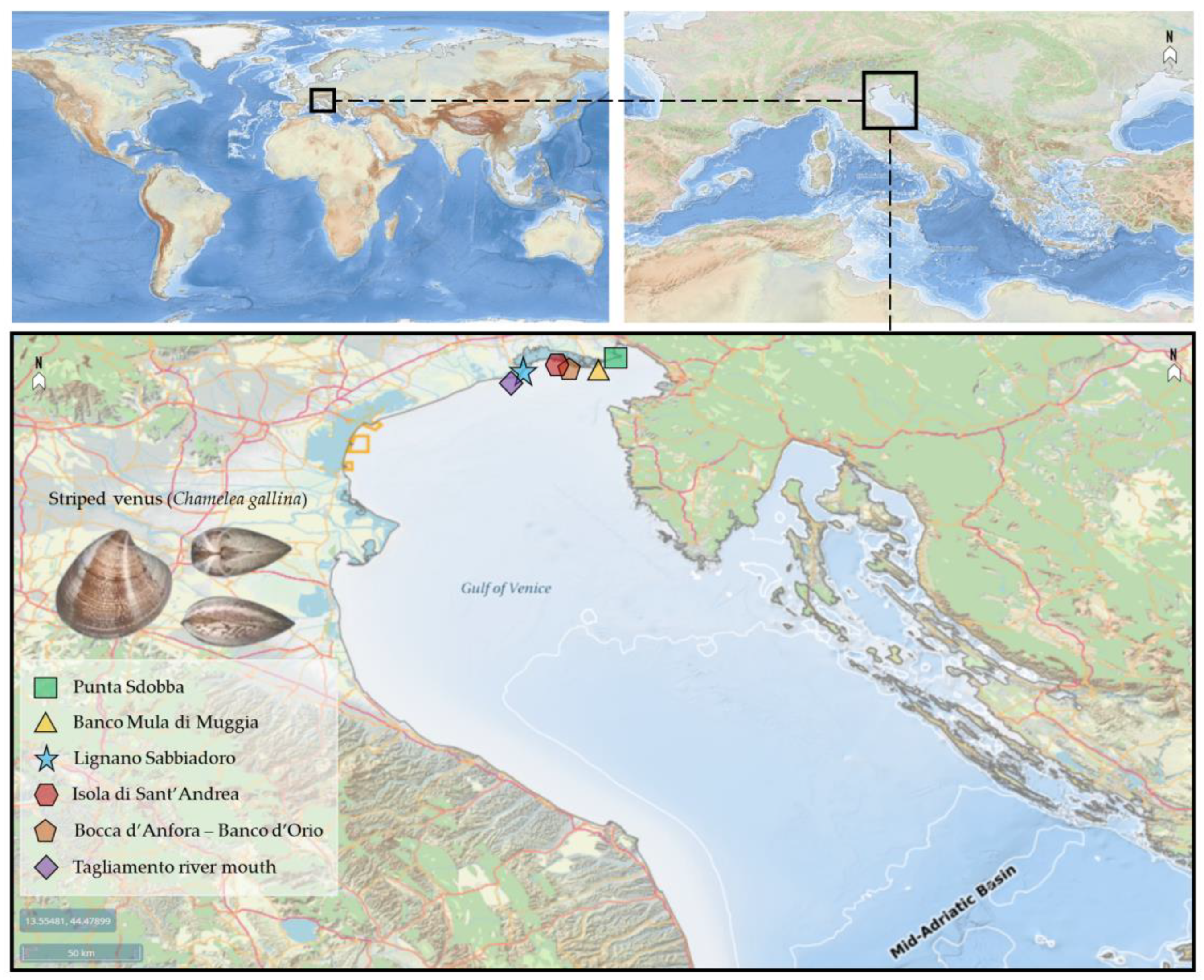
| Survey Area | Population Structure | Biomass Pre-Restocking | Biomass Post-Restocking | Biomass | Surviving Tests |
|---|---|---|---|---|---|
| Punta Sdobba | 2016 | 2016–2018 | 2016–2018 | - | - |
| Banco Mula di Muggia | 2016 | 2016–2018 | 2016–2018 | - | - |
| Banco d’Orio-Bocca d’Anfora | 2016 | 2016–2018 | 2016–2018 | - | - |
| Isola di Sant’Andrea | 2016 | 2016–2018 | 2016–2018 | - | - |
| Lignano Sabbiadoro | 2016 | 2016–2018 | 2016–2018 | 2018–2019 | 2018–2019 |
| Tagliamento river mouth | 2018–2019 | - | - | 2018–2019 | 2018–2019 |
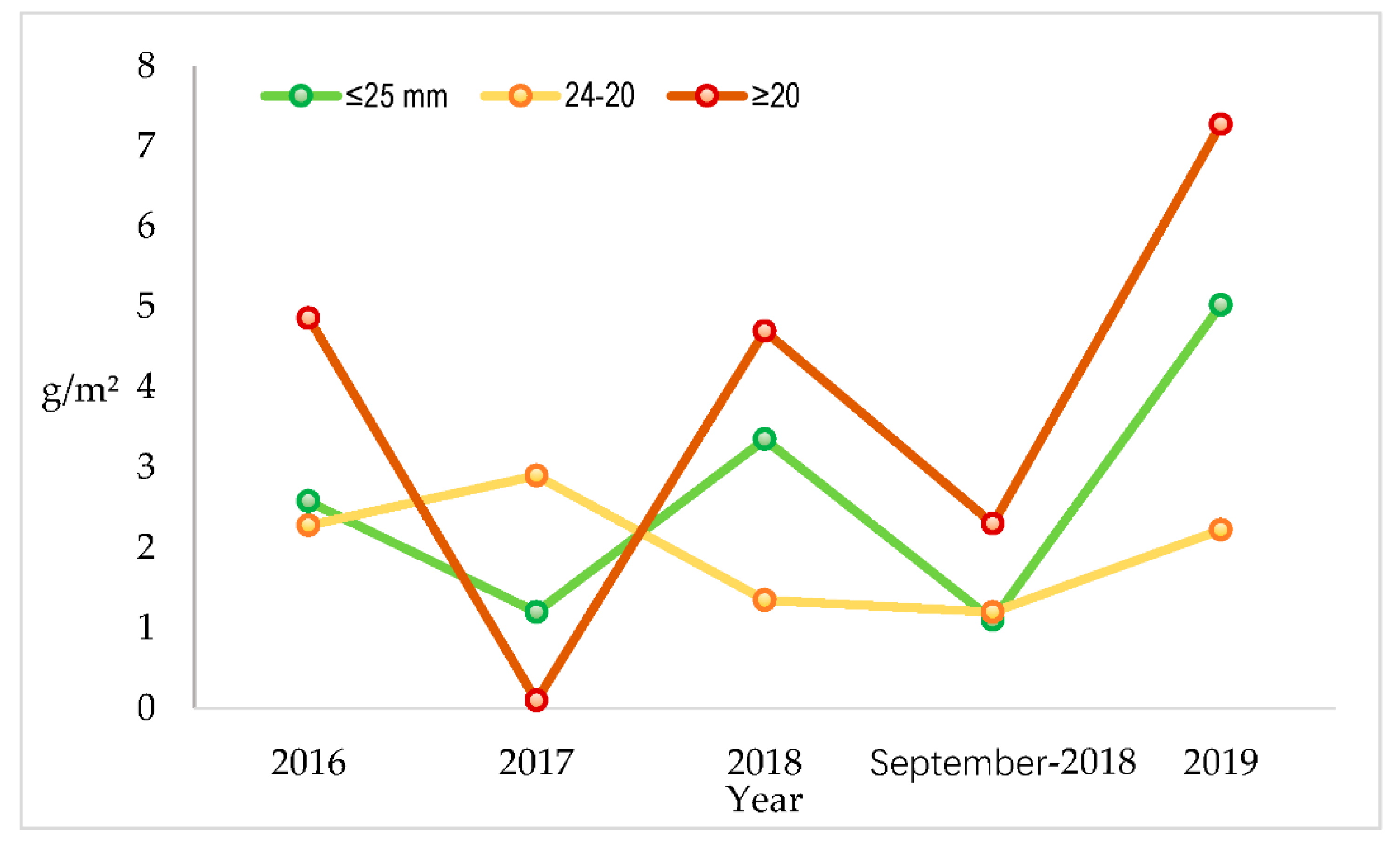
| Survey Area | February 2016 | 2017 | September 2018 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| ≥2.5 cm | 2.4–2.0 cm | ≤2.0 cm | ≥2.5 cm | 2.4–2.0 cm | ≤2.0 cm | ≥2.5 cm | 2.4–2.0 cm | ≤2.0 cm | |
| Punta Sdobba | 0.54 | 0.32 | 0.86 | n.d. | n.d. | n.d. | 0.00 | 0.00 | 0.00 |
| Banco Mula di Muggia | 0.10 | 4.42 | 4.51 | n.d. | n.d. | n.d. | 0.00 | 0.10 | 0.10 |
| Banco d‘Orio-Bocca d’Anfora | 0.26 | 0.95 | 1.21 | n.d. | n.d. | n.d. | 0.10 | 0.10 | 0.20 |
| Isola di Sant’Andrea | 1.53 | 7.52 | 9.05 | n.d. | n.d. | n.d. | 1.50 | 2.00 | 3.50 |
| Lignano Sabbiadoro | 2.58 | 2.28 | 4.86 | 1.20 | 2.90 | 0.10 | 1.10 | 1.20 | 2.30 |
| Survey Area | 2018 | 2019 | ||||
|---|---|---|---|---|---|---|
| ≥2.5 cm | 2.4–2.0 cm | ≤2.0 cm | ≥2.5 cm | 2.4–2.0 cm | ≤2.0 cm | |
| Punta Sdobba | 1.05 | 0.30 | 1.35 | 0.65 | 0.15 | 0.85 |
| Banco Mula di Muggia | 5.65 | 2.40 | 8.05 | 9.40 | 4.30 | 13.70 |
| Banco d’Orio-Bocca d’Anfora | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Isola di Sant’Andrea | 0.90 | 1.95 | 5.85 | 0.00 | 0.00 | 0.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grazioli, E.; Guerranti, C.; Pastorino, P.; Esposito, G.; Bianco, E.; Simonetti, E.; Rainis, S.; Renzi, M.; Terlizzi, A. Review of the Scientific Literature on Biology, Ecology, and Aspects Related to the Fishing Sector of the Striped Venus (Chamelea gallina) in Northern Adriatic Sea. J. Mar. Sci. Eng. 2022, 10, 1328. https://doi.org/10.3390/jmse10091328
Grazioli E, Guerranti C, Pastorino P, Esposito G, Bianco E, Simonetti E, Rainis S, Renzi M, Terlizzi A. Review of the Scientific Literature on Biology, Ecology, and Aspects Related to the Fishing Sector of the Striped Venus (Chamelea gallina) in Northern Adriatic Sea. Journal of Marine Science and Engineering. 2022; 10(9):1328. https://doi.org/10.3390/jmse10091328
Chicago/Turabian StyleGrazioli, Eleonora, Cristiana Guerranti, Paolo Pastorino, Giuseppe Esposito, Emanuele Bianco, Emilio Simonetti, Simona Rainis, Monia Renzi, and Antonio Terlizzi. 2022. "Review of the Scientific Literature on Biology, Ecology, and Aspects Related to the Fishing Sector of the Striped Venus (Chamelea gallina) in Northern Adriatic Sea" Journal of Marine Science and Engineering 10, no. 9: 1328. https://doi.org/10.3390/jmse10091328
APA StyleGrazioli, E., Guerranti, C., Pastorino, P., Esposito, G., Bianco, E., Simonetti, E., Rainis, S., Renzi, M., & Terlizzi, A. (2022). Review of the Scientific Literature on Biology, Ecology, and Aspects Related to the Fishing Sector of the Striped Venus (Chamelea gallina) in Northern Adriatic Sea. Journal of Marine Science and Engineering, 10(9), 1328. https://doi.org/10.3390/jmse10091328












