Effects of Salinity on Growth and In Vitro Ichthyotoxicity of Three Strains of Karenia mikimotoi
Abstract
:1. Introduction
2. Materials and Methods
2.1. Karenia Mikimotoi Strains and Cell Cutivation Conditions
2.2. DNA Extraction, PCR Conditions and Phylogenetic Analysis
2.3. Growth Curves
2.4. Effects of Salinity on Growth and Cell Width
2.5. Fish Gill Cell Line and Culturing Condition
2.6. Ichthyotoxicity of K. Mikimotoi Strains on Fish Gill Cells
2.7. Statistical Analyses
3. Results and Discussion
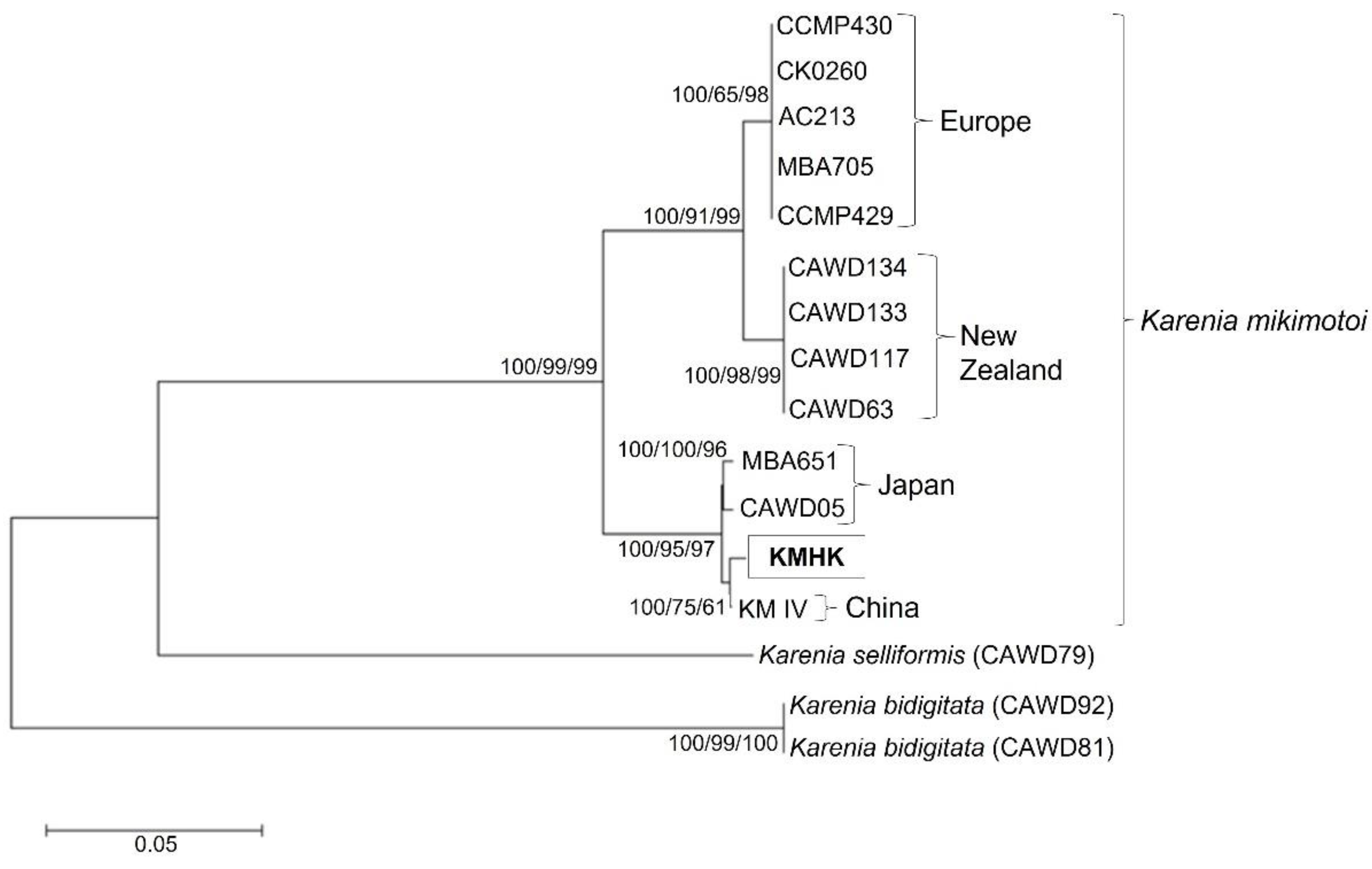
3.1. Phylogenetic Analysis of KMHK
3.2. Growth of K. Mikimotoi Strains
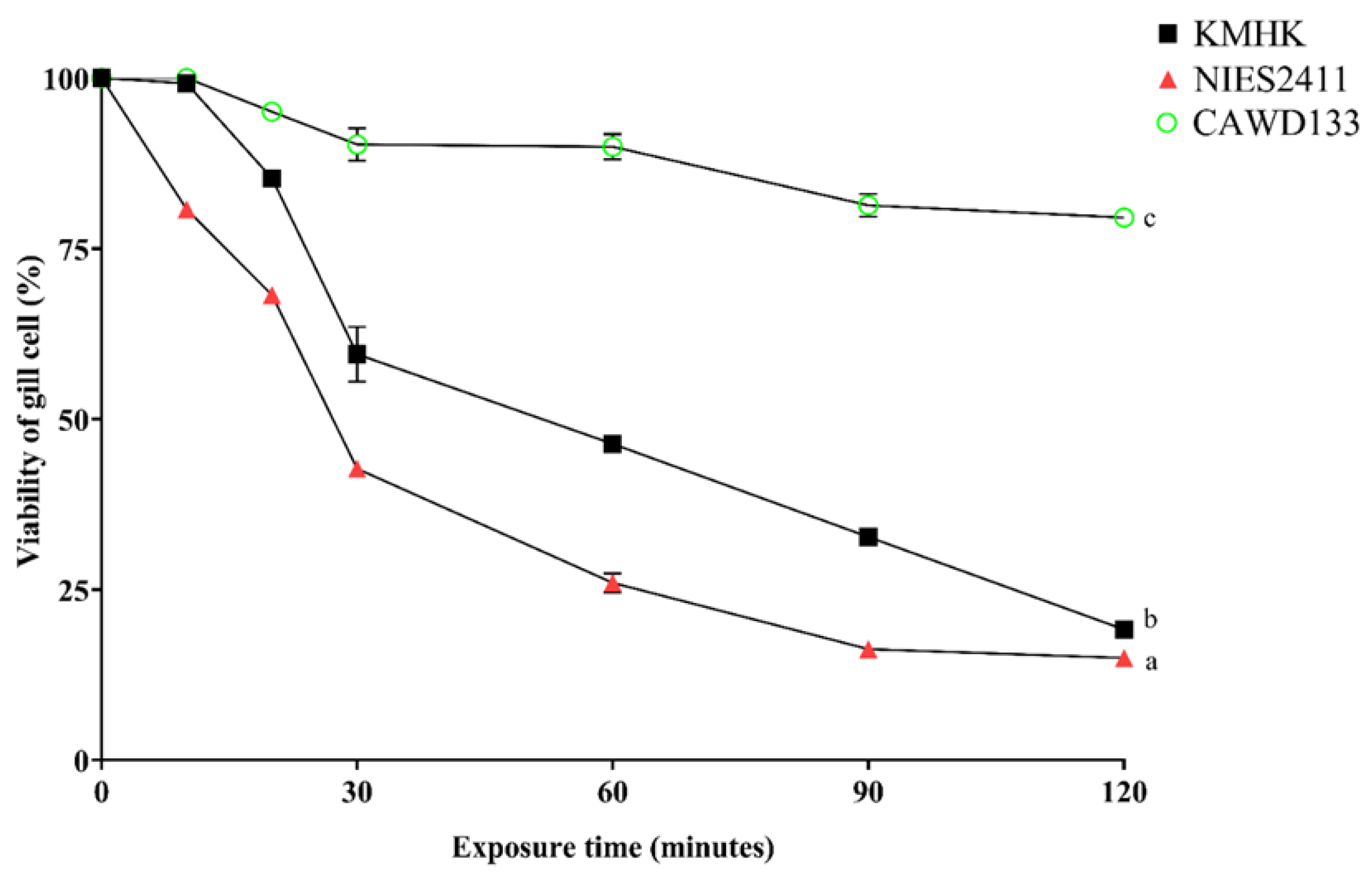
3.3. Ichthyotoxicity of K. Mikimotoi Strains
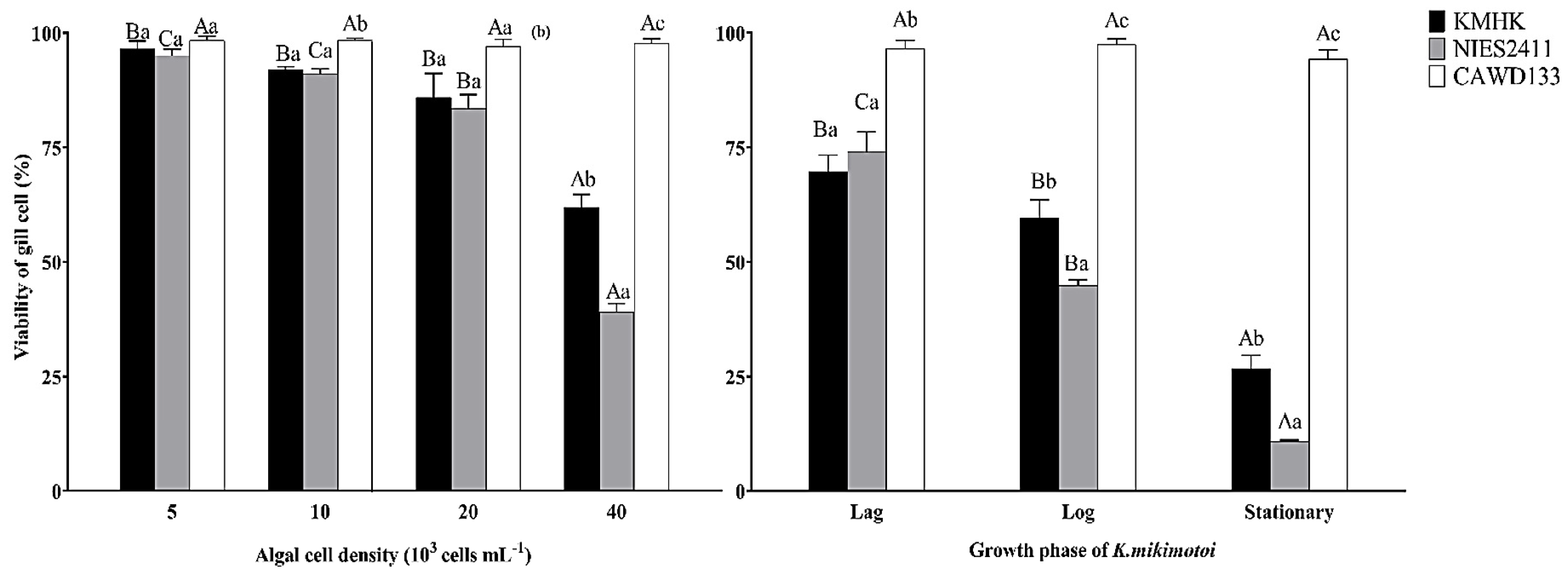
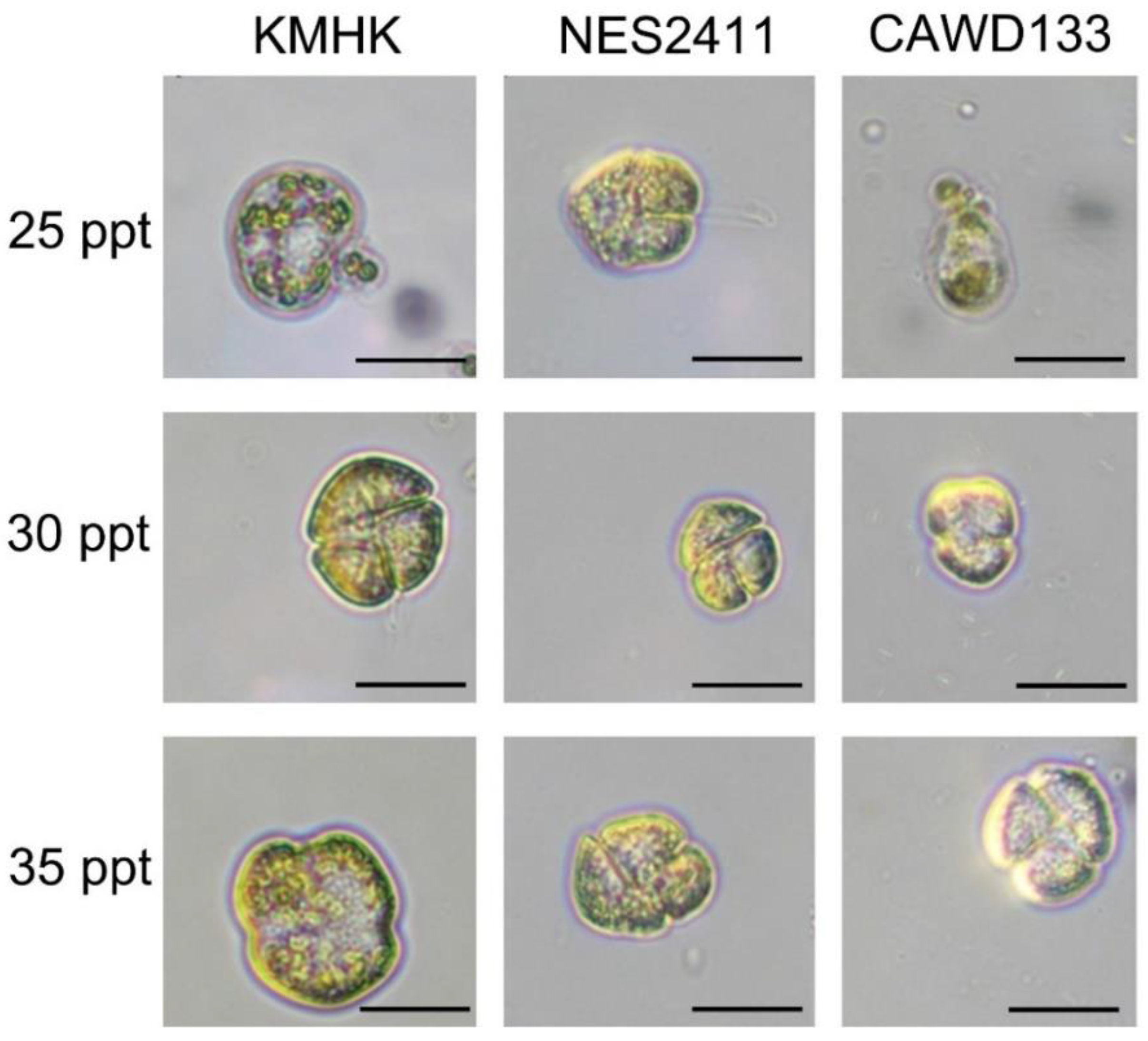
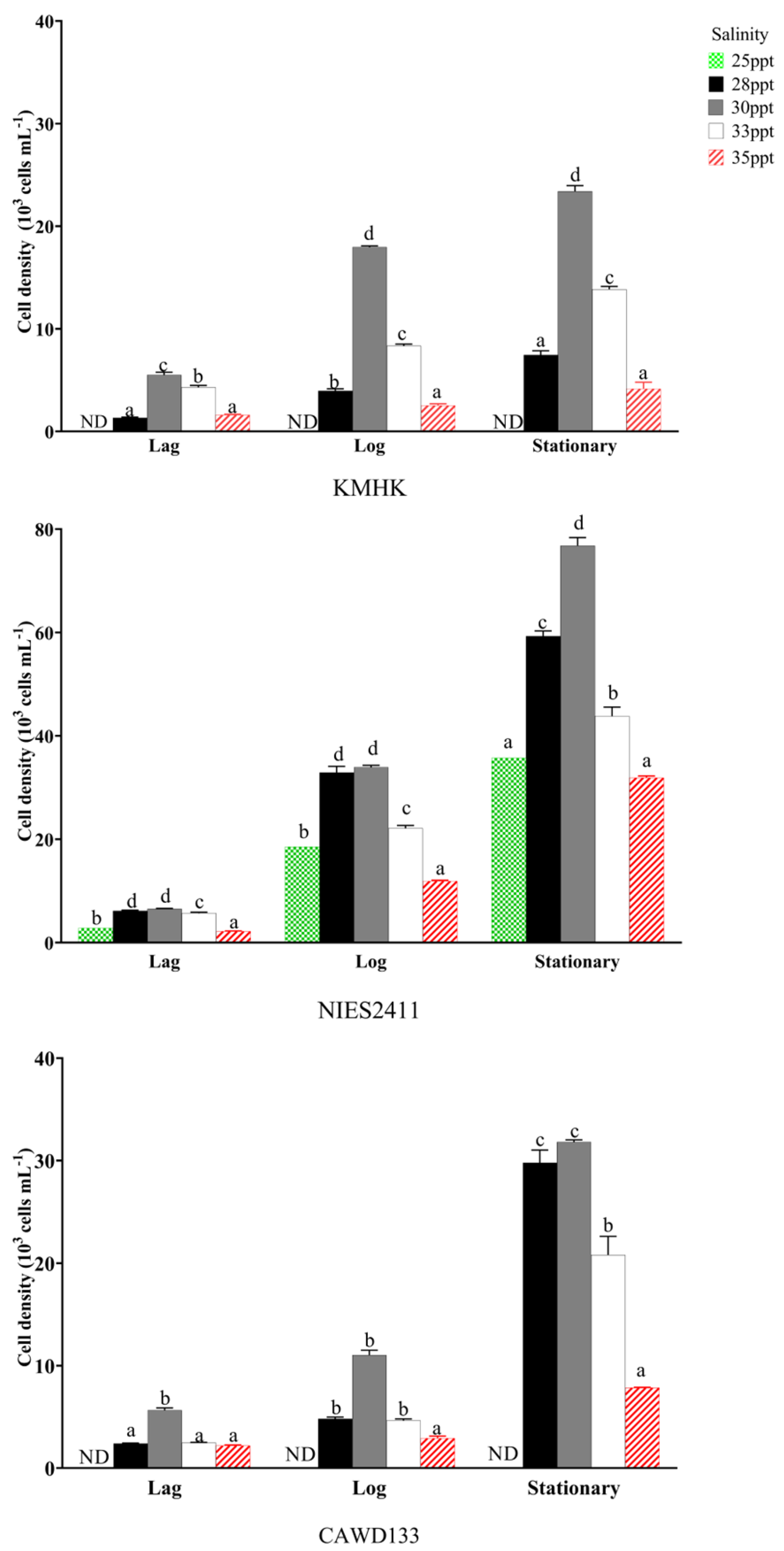
3.4. Effect of Salinity on Cell Size, and Growth of K. Mikimotoi Strains
3.5. Effect of Salinity on Ichthyotoxicity of K. Mikimotoi Strains
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Appendix

| Starting Date | Estimated Area (10−3 km2) | Estimated Cell Density (103 Cells mL−1) | Salinity(ppt) | |||
|---|---|---|---|---|---|---|
| One Month Before Algal Blooming | The Month with Algal Blooming | |||||
| Surface Layer | Middle Layer | Surface Layer | Middle Layer | |||
| December 2015 | 15.1 | 1–10 | 31.5 | 32 | 29.2–30.1 | 30–30.8 |
| October 2010 | 12.1 | 1–10 | 29.6 | 30.9 | 26.9 | 29.9 |
| June 2003 | 1.1 | 10–50 | 32.3 | 32.5 | 28.1 | 28.6 |
| July 2001 | 0.01 | 1–10 | 32.1 | 32.3 | 26.4–28.7 | 30.7–31.9 |
| March 1997 | 0.3 | 10–50 | NM | |||
| August 1988 | 0.12 | 1–10 | 33.5 | 34.3 | 29.6 | 31 |
| December 1986 | 0.3 | <1 | NM | NM | NM | NM |
| October 1986 | 0.3 | 1–10 | NM | NM | NM | NM |
| October 1983 | 0.3 | >50 | NM | NM | NM | NM |
| February 1981 | 0.012 | 10–50 | NM | NM | NM | NM |
| September 1980 | 0.3 | 10–50 | NM | NM | NM | NM |
| Growth Phase | KMHK | NIES2411 | CAWD133 |
|---|---|---|---|
| Lag | 4 | 7 | 4 |
| Log | 9 | 14 | 9 |
| Stationary | 13 | 21 | 13 |
| Group | Geographic Origin | GenBank Accession Number | I.D. Code | References |
|---|---|---|---|---|
| 1 | Hong Kong, China | KKMHK | N/A | The present study |
| China | KT733616 | KM IV | [58] | |
| Japan | U92247 | CAWD05 | [46] | |
| Japan | HM807312 | MBA561 | [24] | |
| 2 | New Zealand | U92249 | CAWD63 | [46] |
| New Zealand | HM807326 | CAWD117 | [24] | |
| New Zealand | HM807327 | CAWD133 | [24] | |
| New Zealand | HM807328 | CAWD134 | [24] | |
| 3 | Not reported | HM807333 | CCMP430 | [24] |
| UK | HM807318 | CCMP429 | [24] | |
| UK | HM807317 | MBA705 | [24] |
| Loci | Forward and Reverse Primer | Primer Sequence (5′–3′) | Annealing Temperature (°C) | Amplicon Size (bp) | References |
|---|---|---|---|---|---|
| LSU | D1R-F | ACCCGCTGAATTTAAGCATA | 60 | 760 | [21] |
| D2C-R | CCTTGGTCCGTGTTTCAAGA | ||||
| ITS | ITSA-F | CCGGATCCAAGCTTTCGTAACAAGGHTCCGTAGGT | 56 | 700 | [22] |
| ITSB-R | CCGGATCCGTCGACAKATGCTTAARTTCAGCRGG | ||||
| rbcL | rbcL 640-F | ATGATGAAAA(CT)ATTAATTCTCAACC | 56 | 619 | [24] |
| rbcL 1240-R | TG(AT)CC(AG)AT(AGT)GTACCACCACC |
References
- Hallegraeff, G.M. Harmful Algal Blooms: A Global Overview; Unesco Publishing: Paris, France, 2003. [Google Scholar]
- Sha, J.; Xiong, H.; Li, C.; Lu, Z.; Zhang, J.; Zhong, H.; Zhang, W.; Yan, B. Harmful algal blooms and their eco-environmental indication. Chemosphere 2021, 274, 129912. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yan, T.; Yu, R.; Zhou, M. A review of Karenia mikimotoi: Bloom events, physiology, toxicity and toxic mechanism. Harmful Algae 2019, 90, 101702. [Google Scholar] [CrossRef] [PubMed]
- Heil, C.A.; Glibert, P.M.; Al-Sarawi, M.A.; Faraj, M.; Behbehani, M.; Husain, M. First record of a fish-killing Gymnodinium sp. bloom in Kuwait Bay, Arabian Sea: Chronology and potential causes. Mar. Ecol. Prog. Ser. 2001, 214, 15–23. [Google Scholar] [CrossRef]
- Honjo, T. Karenia (formarly Gymnodinium) mikimotoi. In Red Tides; Okaichi, T., Ed.; Terra Scientific Publishing Company: Tokyo, Japan, 2004; pp. 345–356. [Google Scholar]
- Lin, J.N.; Yan, T.; Zhang, Q.C.; Wang, Y.F.; Liu, Q.; Zhou, M.J. The detrimental impacts of Karenia mikimotoi blooms on the abalone haliotis discus hannai in fujian province. Mar. Environ. Sci. 2016, 35, 27–34. [Google Scholar]
- Li, X.; Yan, T.; Lin, J.; Yu, R.; Zhou, M. Detrimental impacts of the dinoflagellate Karenia mikimotoi in Fujian coastal waters on typical marine organisms. Harmful Algae 2017, 61, 1–12. [Google Scholar] [CrossRef]
- Kwok, C.S.N.; Wan, W.W.; Chan, K.K.K.; Xu, S.J.L.; Lee, F.W.F.; Ho, K.C. Karenia mikimotoi, a rare species in Hong Kong waters, associated with a recent massive fish kill. Harmful Algae News 2016, 53, 4–5. [Google Scholar]
- AFCD. Hong Kong Red Tide Database. Available online: https://www.afcd.gov.hk/tc_chi/fisheries/hkredtide/database/database.html (accessed on 1 January 2021).
- Martens, H.; Van de Waal, D.B.; Brandenburg, K.M.; Krock, B.; Tillmann, U. Salinity effects on growth and toxin production in an Alexandrium ostenfeldii (Dinophyceae) isolate from The Netherlands. J. Plankton Res. 2016, 38, 1302–1316. [Google Scholar] [CrossRef]
- Gomes, M.P.; Juneau, P. Temperature and light modulation of herbicide toxicity on algal and cyanobacterial physiology. Front. Environ. Sci. 2017, 5, 50. [Google Scholar] [CrossRef]
- Hamasaki, K.; Horie, M.; Tokimitsu, S.; Toda, T.; Taguchi, S. Variability in toxicity of the dinoflagellate Alexandrium Tamarense isolated from Hiroshima Bay, western Japan, as a reflection of changing environmental conditions. J. Plankton Res. 2001, 23, 271–278. [Google Scholar] [CrossRef]
- Band-Schmidt, C.J.; Morquecho, L.; Lechuga-Devéze, C.H.; Anderson, D.M. Effects of growth medium, temperature, salinity and seawater source on the growth of Gymnodinium catenatum (Dinophyceae) from Bahía Concepción, Gulf of California, Mexico. J. Plankton Res. 2004, 26, 1459–1470. [Google Scholar] [CrossRef]
- Silveira, S.B.; Odebrecht, C. Effects of salinity and temperature on the growth, toxin production, and akinete germination of the cyanobacterium. Nodularia spumigena. Front. Environ. Sci. 2019, 6, 339. [Google Scholar] [CrossRef]
- Grzebyk, D.; Béchemin, C.; Ward, C.J.; Vérité, C.; Codd, G.A.; Maestrini, S.Y. Effects of salinity and two coastal waters on the growth and toxin content of the dinoflagellate Alexandrium minutum. J. Plankton Res. 2003, 25, 1185–1199. [Google Scholar] [CrossRef]
- Maier Brown, A.F.; Dortch, Q.; Dolah, F.M.V.; Leighfield, T.A.; Morrison, W.; Thessen, A.E.; Steidinger, K.; Richardson, B.; Moncreiff, C.A.; Pennock, J.R. Effect of salinity on the distribution, growth, and toxicity of Karenia spp. Harmful Algae 2006, 5, 199–212. [Google Scholar] [CrossRef]
- Leong, S.C.Y.; Nakazawa, M.; Taguchi, S. Physiological and optical responses of the harmful dinoflagellate Heterocapsa circularisquama to a range of salinity. Hydrobiologia 2006, 559, 149–159. [Google Scholar] [CrossRef]
- Bearon, R.; Grünbaum, D.; Cattolico, R.A. Effects of salinity structure on swimming behavior and harmful algal bloom formation in Heterosigma akashiwo, a toxic raphidophyte. Mar. Ecol. Prog. Ser. 2006, 306, 153–163. [Google Scholar] [CrossRef]
- Sandoval-Sanhueza, A.; Aguilera-Belmonte, A.; Basti, L.; Figueroa, R.I.; Molinet, C.; Álvarez, G.; Oyanedel, S.; Riobó, P.; Mancilla-Gutiérrez, G.; Díaz, P.A. Interactive effects of temperature and salinity on the growth and cytotoxicity of the fish-killing microalgal species Heterosigma akashiwo and Pseudochattonella verruculosa. Mar. Pollut. Bull. 2022, 174, 113234. [Google Scholar] [CrossRef]
- Lee, F.W.F.; Lo, S.C.L. Proteomic study of micro algae: Sample preparation for two dimensional gel electrophoresis and de novo peptide sequencing Using MALDI-TOF MS. Curr. Proteom. 2007, 4, 67–78. [Google Scholar] [CrossRef]
- Lenaers, G.; Maroteaux, L.; Michot, B.; Herzog, M. Dinoflagellates in evolution. A molecular phylogenetic analysis of large subunit ribosomal RNA. J. Mol. Evol. 1989, 29, 40–51. [Google Scholar] [CrossRef]
- Ho, K.C.; Lee, T.C.H.; Kwok, O.T.; Lee, F.W.F. Phylogenetic analysis on a strain of Alexandrium tamarense collected from Antarctic Ocean. Harmful Algae 2012, 15, 100–108. [Google Scholar] [CrossRef]
- Yoon, H.S.; Hackett, J.D.; Bhattacharya, D. A single origin of the peridinin- and fucoxanthin-containing plastids in dinoflagellates through tertiary endosymbiosis. Proc. Natl. Acad. Sci. USA 2002, 99, 11724–11729. [Google Scholar] [CrossRef]
- Al-Kandari, M.A.; Highfield, A.C.; Hall, M.J.; Hayes, P.; Schroeder, D.C. Molecular tools separate harmful algal bloom species, Karenia mikimotoi, from different geographical regions into distinct sub-groups. Harmful Algae 2011, 10, 636–643. [Google Scholar] [CrossRef]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Dayeh, V.R.; Schirmer, K.; Bols, N.C. Applying whole-water samples directly to fish cell cultures in order to evaluate the toxicity of industrial effluent. Water Res. 2002, 36, 3727–3738. [Google Scholar] [CrossRef]
- Mardones, J.I.; Fuenzalida, G.; Zenteno, K.; Alves-de-Souza, C.; Astuya, A.; Dorantes-Aranda, J.J. Salinity-growth response and ichthyotoxic potency of the Chilean Pseudochattonella verruculosa. Front. Mar. Sci. 2019, 6, 24. [Google Scholar] [CrossRef]
- Schirmer, K.; Chan, A.G.J.; Greenberg, B.M.; Dixon, D.G.; Bols, N.C. Methodology for demonstrating and measuring the hotocytotoxicity of fluoranthene to fish cells in culture. Toxicol. Vitr. 1997, 11, 107–113. [Google Scholar]
- Marshall, J.A.; Nichols, P.D.; Hamilton, B.; Lewis, R.J.; Hallegraeff, G.M. Ichthyotoxicity of Chattonella marina (Raphidophyceae) to damselfish (Acanthochromis polycanthus): The synergistic role of reactive oxygen species and free fatty acid. Harmful Algae 2003, 2, 273–281. [Google Scholar] [CrossRef]
- Kim, D.; Li, W.; Matsuyama, Y.; Matsuo, A.; Yagi, M.; Cho, K.; Yamasaki, Y.; Takeshita, S.; Yamaguchi, K.; Oda, T. Strain-dependent lethal effects on abalone and haemolytic activities of the dinoflagellate Karenia mikimotoi. Aquaculture 2020, 520, 734953. [Google Scholar] [CrossRef]
- Guan, W.; Li, P. Dependency of UVR-induced photoinhibition on atomic ratio of N to P in the dinoflagellate Karenia mikimotoi. Mar. Biol. 2017, 164, 1–9. [Google Scholar] [CrossRef]
- Shen, A.; Ma, Z.L.; Jiang, K.J.; Li, D.J. Effects of temperature on growth, photophysiology, Rubisco gene expression in Prorocentrum donghaiense and Karenia mikimotoi. Ocean Sci. J. 2016, 51, 581–589. [Google Scholar] [CrossRef]
- Stæhr, P.A.; Cullen, J.J. Detection of Karenia mikimotoi by spectral absorption signatures. J. Plankton Res. 2003, 25, 1237–1249. [Google Scholar] [CrossRef]
- Dorantes-Aranda, J.J.; Waite, T.D.; Godrant, A.; Rose, A.L.; Tovar, C.D.; Woods, G.M.; Hallegraeff, G.M. Novel application of a fish gill cell line assay to assess ichthyotoxicity of harmful marine microalgae. Harmful Algae 2011, 10, 366–373. [Google Scholar] [CrossRef]
- Grossmann, J. Molecular mechanisms of “detachment-induced apoptosis—Anoikis”. Apoptosis 2002, 7, 247–260. [Google Scholar] [CrossRef]
- Dorantes-Aranda, J.J.; Seger, A.; Mardones, J.I.; Nichols, P.D.; Hallegraeff, G.M. Progress in understanding algal bloom-mediated fish kills: The role of superoxide radicals, phycotoxins and fatty acids. PLoS ONE 2015, 10, e0133549. [Google Scholar] [CrossRef] [PubMed]
- Parker, N.S.; Negri, A.P.; Frampton, D.M.F.; Rodolfi, L.; Tredici, M.R.; Blackburn, S.I. Growth of the toxic dinoflagellate Alexandrium minutum (Dinophyceae) using high biomass culture systems. J. Appl. Phycol. 2002, 14, 313–324. [Google Scholar] [CrossRef]
- Lee, T.C.H.; Chan, P.L.; Xu, S.J.L.; Lee, F.W.F. Comparison of the growth and toxicity responses between a non toxic and a toxic strain of Prorocentrum hoffmannianum. Aquat. Biol. 2020, 29, 59–70. [Google Scholar] [CrossRef]
- Guerrini, F.; Ciminiello, P.; Dell’Aversano, C.; Tartaglione, L.; Fattorusso, E.; Boni, L.; Pistocchi, R. Influence of temperature, salinity and nutrient limitation on yessotoxin production and release by the dinoflagellate Protoceratium reticulatum in batch-cultures. Harmful Algae 2007, 6, 707–717. [Google Scholar] [CrossRef]
- Röder, K.; Hantzsche, F.M.; Gebühr, C.; Miene, C.; Helbig, T.; Krock, B.; Hoppenrath, M.; Luckas, B.; Gerdts, G. Effects of salinity, temperature and nutrients on growth, cellular characteristics and yessotoxin production of Protoceratium reticulatum. Harmful Algae 2012, 15, 59–70. [Google Scholar] [CrossRef]
- Sala-Pérez, M.; Alpermann, T.J.; Krock, B.; Tillmann, U. Growth and bioactive secondary metabolites of arctic Protoceratium reticulatum (Dinophyceae). Harmful Algae 2016, 55, 85–96. [Google Scholar] [CrossRef]
- Peter, C.; Krock, B.; Cembella, A. Effects of salinity variation on growth and yessotoxin composition in the marine dinoflagellate Lingulodinium polyedra from a Skagerrak fjord system (western Sweden). Harmful Algae 2018, 78, 9–17. [Google Scholar] [CrossRef]
- ØStergaard Jensen, M.; Moestrup, Ø. Autecology of the toxic dinoflagellate Alexandrium ostenfeldii: Life history and growth at different temperatures and salinities. Eur. J. Phycol. 1997, 32, 9–18. [Google Scholar] [CrossRef]
- Laabir, M.; Jauzein, C.; Genovesi, B.; Masseret, E.; Grzebyk, D.; Cecchi, P.; Vaquer, A.; Perrin, Y.; Collos, Y. Influence of temperature, salinity and irradiance on the growth and cell yield of the harmful red tide dinoflagellate Alexandrium catenella colonizing Mediterranean waters. J. Plankton Res. 2011, 33, 1550–1563. [Google Scholar] [CrossRef]
- Haywood, A.J.; Steidinger, K.A.; Truby, E.W.; Bergquist, P.R.; Bergquist, P.L.; Adamson, J.; Mackenzie, L. Comparative morphology and molecular phylogenetic analysis of three new species of the genus Karenia (dinophyceae) from new zealand. J. Phycol. 2004, 40, 165–179. [Google Scholar] [CrossRef]
- Zhao, T.; Tan, L.; Huang, W.; Wang, J. The interactions between micro polyvinyl chloride (mPVC) and marine dinoflagellate Karenia mikimotoi: The inhibition of growth, chlorophyll and photosynthetic efficiency. Environ. Pollut. 2019, 247, 883–889. [Google Scholar] [CrossRef]
- Kirst, G.O. Salinity tolerance of eukaryotic marine algae. Annu. Rev. Plant Physiol. 1990, 41, 21–53. [Google Scholar] [CrossRef]
- Shetty, P.; Gitau, M.M.; Maróti, G. Salinity stress responses and adaptation mechanisms in eukaryotic green microalgae. Cells 2019, 8, 1657. [Google Scholar] [CrossRef]
- Marañón, E.; Cermeño, P.; López-Sandoval, D.C.; Rodríguez-Ramos, T.; Sobrino, C.; Huete-Ortega, M.; Blanco, J.M.; Rodríguez, J. Unimodal size scaling of phytoplankton growth and the size dependence of nutrient uptake and use. Ecol. Lett. 2013, 16, 371–379. [Google Scholar] [CrossRef]
- Strom, S.L.; Harvey, E.L.; Fredrickson, K.A.; Menden-Deuer, S. Broad salinity tolerance as a refuge from predation in the harmful raphidophyte alga Heterosigma akashiwo (Raphidophyceae). J. Phycol. 2013, 49, 20–31. [Google Scholar] [CrossRef]
- Skarlato, S.; Filatova, N.; Knyazev, N.; Berdieva, M.; Telesh, I. Salinity stress response of the invasive dinoflagellate Prorocentrum minimum. Estuar. Coast. Shelf Sci. 2018, 211, 199–207. [Google Scholar] [CrossRef]
- Gilmour, D.J.; Hipkins, M.L.F.; Boney, A.D. The effect of decreasing the external salinity on the primary processes of photosynthesis in Dunaliella tertiolecta. J. Exp. Bot. 1984, 35, 28–35. [Google Scholar] [CrossRef]
- Salgado, P.; Riobó, P.; Rodríguez, F.; Franco, J.M.; Bravo, I. Differences in the toxin profiles of Alexandrium ostenfeldii (Dinophyceae) strains isolated from different geographic origins: Evidence of paralytic toxin, spirolide, and gymnodimine. Toxicon 2015, 103, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.W.; Arega, F. Eutrophication Dynamics of Tolo Harbour, Hong Kong. Mar. Pollut. Bull. 1999, 39, 187–192. [Google Scholar] [CrossRef]
- EPD. Marine Water Quality Data. Available online: https://cd.epic.epd.gov.hk/EPICRIVER/marine/?lang=en (accessed on 1 January 2021).
- Parkhill, J.P.; Cembella, A. Effects of salinity, light and inorganic nitrogen on growth and toxigenity of the marine dinoflagellate Alexandrium tamarense from northeastern Canada. J. Plankton Res. 1999, 21, 5. [Google Scholar] [CrossRef]
- Zhang, Q. Phylogenetic Analysis and Pigment Features of the Causative Species of the Dinoflagellate Bloom in Fujian in 2012; Key Laboratory of Marine Ecology and Environmental Sciences, Institute of Oceanology, Chinese Academy of Sciences: Qingdao, China, 2015. [Google Scholar]


| Assess Code | Geographic Locations | Duration of Lag, Log Phase in Days | Specific Growth Rates, in Day-1 | Maximum Cell Density, 104 Cells mL−1 | Salinity (ppt) | References |
|---|---|---|---|---|---|---|
| KMHK | Hong Kong, China | 4, 9 | 0.169 | 2.53 | 30 | This study |
| CAWD133 | New Zealand | 4, 3 | 0.271 | 3.25 | 30 | This study |
| NIES2411 | Japan | 4, 12 | 0.189 | 7.69 | 30 | This study |
| NIES2411 | Japan | 6, 14 | 0.146 | 11.6 | 25 | [31] |
| NGU04 | Japan | 10, 10 | 0.190 | 10.0 | 25 | [31] |
| SUO-1 | Japan | 4, 14 | 0.126 | 18.6 | 25 | [31] |
| -- | China | -- | 0.257 | -- | 34 | [32] |
| ECSFRI081109 | China | -- | 0.336 | -- | -- | [33] |
| K-0260 | Norway | -- | 0.150 | -- | -- | [34] |
| Salinity (ppt) | Cell Width (μm) | Relative Mobility | ||||
|---|---|---|---|---|---|---|
| KMHK | NIES2411 | CAWD133 | KMHK | NIES2411 | CAWD133 | |
| 25 | ND | 28.3 ± 4.0 b | ND | ND | ** | ND |
| 28 | 26.5 ± 3.3 b | 23.2 ± 2.6 a | 24.7 ± 4.1 b | *** | *** | *** |
| 30 | 23.7 ± 2.0 a | 22.7 ± 2.3 a | 21.9 ± 2.3 a | *** | *** | *** |
| 33 | 26.9 ± 2.1 b | 23.8 ± 2.8 a | 24.4 ± 2.2 b | *** | *** | *** |
| 35 | 29.0 ± 3.6 c | 28.0 ± 4.2 b | 24.7 ± 3.3 b | ** | ** | * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lam, W.; Cheung, E.M.-S.; Tam, N.F.-Y.; Lee, T.C.-H.; Kwok, C.S.-N.; Lai, K.K.-Y.; Xu, S.J.; Lee, F.W.-F. Effects of Salinity on Growth and In Vitro Ichthyotoxicity of Three Strains of Karenia mikimotoi. J. Mar. Sci. Eng. 2022, 10, 1236. https://doi.org/10.3390/jmse10091236
Lam W, Cheung EM-S, Tam NF-Y, Lee TC-H, Kwok CS-N, Lai KK-Y, Xu SJ, Lee FW-F. Effects of Salinity on Growth and In Vitro Ichthyotoxicity of Three Strains of Karenia mikimotoi. Journal of Marine Science and Engineering. 2022; 10(9):1236. https://doi.org/10.3390/jmse10091236
Chicago/Turabian StyleLam, Winnie, Emily Man-Shan Cheung, Nora Fung-Yee Tam, Thomas Chun-Hung Lee, Celia Sze-Nga Kwok, Kaze King-Yip Lai, Steven Jingliang Xu, and Fred Wang-Fat Lee. 2022. "Effects of Salinity on Growth and In Vitro Ichthyotoxicity of Three Strains of Karenia mikimotoi" Journal of Marine Science and Engineering 10, no. 9: 1236. https://doi.org/10.3390/jmse10091236
APA StyleLam, W., Cheung, E. M.-S., Tam, N. F.-Y., Lee, T. C.-H., Kwok, C. S.-N., Lai, K. K.-Y., Xu, S. J., & Lee, F. W.-F. (2022). Effects of Salinity on Growth and In Vitro Ichthyotoxicity of Three Strains of Karenia mikimotoi. Journal of Marine Science and Engineering, 10(9), 1236. https://doi.org/10.3390/jmse10091236







