Ocean Warming Amplifies the Effects of Ocean Acidification on Skeletal Mineralogy and Microstructure in the Asterinid Starfish Aquilonastra yairi
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design and Control of Seawater Chemistry
2.2. Skeletal Mineral Composition Analysis
2.3. Analysis of the Skeleton Microstructure
2.4. Statistical Analysis
3. Results
3.1. Elemental Composition of Skeletal Carbonate
3.2. Skeletal Microstructure
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Watson, A.J.; Schuster, U.; Shutler, J.D.; Holding, T.; Ashton, I.G.C.; Landschutzer, P.; Woolf, D.K.; Goddijn-Murphy, L. Revised estimates of ocean-atmosphere CO2 flux are consistent with ocean carbon inventory. Nat. Commun. 2020, 11, 4422. [Google Scholar] [CrossRef] [PubMed]
- Feely, R.A.; Sabine, C.L.; Lee, K.; Berelson, W.; Kleypas, J.; Fabry, V.J.; Millero, F.J. Impact of anthropogenic CO2 on the CaCO3 system in the oceans. Science 2004, 305, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Sabine, C.L.; Feely, R.A.; Gruber, N.; Key, R.M.; Lee, K.; Bullister, J.L.; Wanninkhof, R.; Wong, C.S.; Wallace, D.W.; Tilbrook, B.; et al. The oceanic sink for anthropogenic CO2. Science 2004, 305, 367–371. [Google Scholar] [CrossRef]
- Orr, J.C.; Fabry, V.J.; Aumont, O.; Bopp, L.; Doney, S.C.; Feely, R.A.; Gnanadesikan, A.; Gruber, N.; Ishida, A.; Joos, F.; et al. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 2005, 437, 681–686. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Bruno, J.F. The impact of climate change on the world’s marine ecosystems. Science 2010, 328, 1523–1528. [Google Scholar] [CrossRef] [PubMed]
- Dupont, S.; Ortega-Martinez, O.; Thorndyke, M. Impact of near-future ocean acidification on echinoderms. Ecotoxicology 2010, 19, 449–462. [Google Scholar] [CrossRef] [PubMed]
- Rodolfo-Metalpa, R.; Lombardi, C.; Cocito, S.; Hall-Spencer, J.M.; Gambi, M.C. Effects of ocean acidification and high temperatures on the bryozoan Myriapora truncata at natural CO2 vents. Mar. Ecol. 2010, 31, 447–456. [Google Scholar] [CrossRef]
- Matranga, V.; Bonaventura, R.; Costa, C.; Karakostis, K.; Pinsino, A.; Russo, R.; Zito, F. Echinoderms as Blueprints for Biocalcification: Regulation of Skeletogenic Genes and Matrices. In Molecular Biomineralization: Aquatic Organisms Forming Extraordinary Materials; Müller, W.E.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 225–248. [Google Scholar]
- Mann, S. Mineralization in Biological Systems; Springer: Berlin/Heidelberg, Germany, 1983; pp. 125–174. [Google Scholar]
- Killian, C.E.; Wilt, F.H. Molecular aspects of biomineralization of the echinoderm endoskeleton. Chem. Rev. 2008, 108, 4463–4474. [Google Scholar] [CrossRef]
- Gorzelak, P. Functional Micromorphology of the Echinoderm Skeleton; Cambridge University Press: Cambridge, UK, 2021; p. 44. [Google Scholar]
- Feng, Q. Principles of Calcium-Based Biomineralization. In Molecular Biomineralization: Aquatic Organisms Forming Extraordinary Materials; Müller, W.E.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 141–197. [Google Scholar]
- Gilbert, P.U.P.A.; Wilt, F.H. Molecular Aspects of Biomineralization of the Echinoderm Endoskeleton. In Molecular Biomineralization: Aquatic Organisms Forming Extraordinary Materials; Müller, W.E.G., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 199–223. [Google Scholar]
- Ries, J.B.; Ghazaleh, M.N.; Connolly, B.; Westfield, I.; Castillo, K.D. Impacts of seawater saturation state (ΩA = 0.4–4.6) and temperature (10, 25 °C) on the dissolution kinetics of whole-shell biogenic carbonates. Geochim. Cosmochim. Acta 2016, 192, 318–337. [Google Scholar] [CrossRef]
- Kawahata, H.; Fujita, K.; Iguchi, A.; Inoue, M.; Iwasaki, S.; Kuroyanagi, A.; Maeda, A.; Manaka, T.; Moriya, K.; Takagi, H.; et al. Perspective on the response of marine calcifiers to global warming and ocean acidification—Behavior of corals and foraminifera in a high CO2 world “hot house”. Prog. Earth Planet Sci. 2019, 6, 5. [Google Scholar] [CrossRef]
- Morse, J.W.; Andersson, A.J.; Mackenzie, F.T. Initial responses of carbonate-rich shelf sediments to rising atmospheric pCO2 and “ocean acidification”: Role of high Mg-calcites. Geochim. Cosmochim. Acta 2006, 70, 5814–5830. [Google Scholar] [CrossRef]
- Watson, S.A.; Peck, L.S.; Tyler, P.A.; Southgate, P.C.; Tan, K.S.; Day, R.W.; Morley, S.A. Marine invertebrate skeleton size varies with latitude, temperature and carbonate saturation: Implications for global change and ocean acidification. Glob. Chang. Biol. 2012, 18, 3026–3038. [Google Scholar] [CrossRef] [PubMed]
- Anand, M.; Rangesh, K.; Maruthupandy, M.; Jayanthi, G.; Rajeswari, B.; Priya, R.J. Effect of CO2 driven ocean acidification on calcification, physiology and ovarian cells of tropical sea urchin Salmacis virgulata—A microcosm approach. Heliyon 2021, 7, e05970. [Google Scholar] [CrossRef] [PubMed]
- Duquette, A.; McClintock, J.B.; Amsler, C.D.; Perez-Huerta, A.; Milazzo, M.; Hall-Spencer, J.M. Effects of ocean acidification on the shells of four Mediterranean gastropod species near a CO2 seep. Mar. Pollut. Bull. 2017, 124, 917–928. [Google Scholar] [CrossRef]
- Ries, J.B.; Cohen, A.L.; McCorkle, D.C. Marine calcifiers exhibit mixed responses to CO2-induced ocean acidification. Geology 2009, 37, 1131–1134. [Google Scholar] [CrossRef]
- Pörtner, H. Ecosystem effects of ocean acidification in times of ocean warming: A physiologist’s view. Mar. Ecol. Prog. Ser. 2008, 373, 203–217. [Google Scholar] [CrossRef]
- Weiner, S.; Dove, P.M. An overview of biomineralization processes and the problem of the vital effect. Rev. Mineral. Geochem. 2003, 54, 1–29. [Google Scholar] [CrossRef]
- Weber, J.N. Temperature dependence of magnesium in echinoid and asteroid skeletal calcite: A reinterpretation of its significance. J. Geol. 1973, 81, 543–556. [Google Scholar] [CrossRef]
- Chave, K.E. Aspects of the biogeochemistry of magnesium 1. Calcareous marine organisms. J. Geol. 1954, 62, 266–283. [Google Scholar] [CrossRef]
- Hermans, J.; Borremans, C.; Willenz, P.; André, L.; Dubois, P. Temperature, salinity and growth rate dependences of Mg/Ca and Sr/Ca ratios of the skeleton of the sea urchin Paracentrotus lividus (Lamarck): An experimental approach. Mar. Biol. 2010, 157, 1293–1300. [Google Scholar] [CrossRef]
- Duquette, A.; Vohra, Y.K.; McClintock, J.B.; Angus, R.A. Near-future temperature reduces Mg/Ca ratios in the major skeletal components of the common subtropical sea urchin Lytechinus variegatus. J. Exp. Mar. Biol. Ecol. 2018, 509, 1–7. [Google Scholar] [CrossRef]
- Lowenstam, H.A.; Weiner, S. On Biomineralization; Oxford University Press: New York, NY, USA, 1989. [Google Scholar]
- Gazeau, F.; Parker, L.M.; Comeau, S.; Gattuso, J.-P.; O’Connor, W.A.; Martin, S.; Pörtner, H.-O.; Ross, P.M. Impacts of ocean acidification on marine shelled molluscs. Mar. Biol. 2013, 160, 2207–2245. [Google Scholar] [CrossRef]
- Olson, I.C.; Kozdon, R.; Valley, J.W.; Gilbert, P.U. Mollusk shell nacre ultrastructure correlates with environmental temperature and pressure. J. Am. Chem. Soc. 2012, 134, 7351–7358. [Google Scholar] [CrossRef] [PubMed]
- Reynaud, S.; Ferrier-Pagès, C.; Meibom, A.; Mostefaoui, S.; Mortlock, R.; Fairbanks, R.; Allemand, D. Light and temperature effects on Sr/Ca and Mg/Ca ratios in the scleractinian coral Acropora sp. Geochim. Cosmochim. Acta 2007, 71, 354–362. [Google Scholar] [CrossRef]
- Toyofuku, T.; Kitazato, H.; Kawahata, H.; Tsuchiya, M.; Nohara, M. Evaluation of Mg/Ca thermometry in foraminifera: Comparison of experimental results and measurements in nature. Paleoceanography 2000, 15, 456–464. [Google Scholar] [CrossRef]
- Dissard, D.; Nehrke, G.; Reichart, G.J.; Bijma, J. The impact of salinity on the Mg/Ca and Sr/Ca ratio in the benthic foraminifera Ammonia tepida: Results from culture experiments. Geochim. Cosmochim. Acta 2010, 74, 928–940. [Google Scholar] [CrossRef]
- Bell, T.; Nishida, K.; Ishikawa, K.; Suzuki, A.; Nakamura, T.; Sakai, K.; Ohno, Y.; Iguchi, A.; Yokoyama, Y. Temperature-controlled culture experiments with primary polyps of coral Acropora digitifera: Calcification rate variations and skeletal Sr/Ca, Mg/Ca, and Na/Ca ratios. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2017, 484, 129–135. [Google Scholar] [CrossRef]
- Kısakürek, B.; Eisenhauer, A.; Böhm, F.; Garbe-Schönberg, D.; Erez, J. Controls on shell Mg/Ca and Sr/Ca in cultured planktonic foraminiferan, Globigerinoides ruber (white). Earth Planet Sci. Lett. 2008, 273, 260–269. [Google Scholar] [CrossRef]
- Rathburn, A.E.; De Deckker, P. Magnesium and strontium compositions of recent benthic foraminifera from the Coral Sea, Australia and Prydz Bay, Antarctica. Mar. Micropaleontol. 1997, 32, 231–248. [Google Scholar] [CrossRef]
- Dodd, J.R. Magnesium and strontium in calcareous skeletons: A review. J. Paleontol. 1967, 41, 1313–1329. [Google Scholar]
- Kolbuk, D.; Di Giglio, S.; M’Zoudi, S.; Dubois, P.; Stolarski, J.; Gorzelak, P. Effects of seawater Mg2+/Ca2+ ratio and diet on the biomineralization and growth of sea urchins and the relevance of fossil echinoderms to paleoenvironmental reconstructions. Geobiology 2020, 18, 710–724. [Google Scholar] [CrossRef] [PubMed]
- LaVigne, M.; Hill, T.M.; Sanford, E.; Gaylord, B.; Russell, A.D.; Lenz, E.A.; Hosfelt, J.D.; Young, M.K. The elemental composition of purple sea urchin (Strongylocentrotus purpuratus) calcite and potential effects of pCO2 during early life stages. Biogeosciences 2013, 10, 3465–3477. [Google Scholar] [CrossRef]
- Todgham, A.E.; Stillman, J.H. Physiological responses to shifts in multiple environmental stressors: Relevance in a changing world. Integr. Comp. Biol. 2013, 53, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Harvey, B.P.; Gwynn-Jones, D.; Moore, P.J. Meta-analysis reveals complex marine biological responses to the interactive effects of ocean acidification and warming. Ecol. Evol. 2013, 3, 1016–1030. [Google Scholar] [CrossRef] [PubMed]
- Kroeker, K.J.; Kordas, R.L.; Crim, R.; Hendriks, I.E.; Ramajo, L.; Singh, G.S.; Duarte, C.M.; Gattuso, J.-P. Impacts of ocean acidification on marine organisms: Quantifying sensitivities and interaction with warming. Glob. Chang. Biol. 2013, 19, 1884–1896. [Google Scholar] [CrossRef] [PubMed]
- Brahmi, C.; Chapron, L.; Le Moullac, G.; Soyez, C.; Beliaeff, B.; Lazareth, C.E.; Gaertner-Mazouni, N.; Vidal-Dupiol, J. Effects of elevated temperature and pCO2 on the respiration, biomineralization and photophysiology of the giant clam Tridacna maxima. Conserv. Physiol. 2021, 9, coab041. [Google Scholar] [CrossRef]
- Knights, A.M.; Norton, M.J.; Lemasson, A.J.; Stephen, N. Ocean acidification mitigates the negative effects of increased sea temperatures on the biomineralization and crystalline ultrastructure of Mytilus. Front. Mar. Sci. 2020, 7. [Google Scholar] [CrossRef]
- Li, S.; Liu, C.; Huang, J.; Liu, Y.; Zheng, G.; Xie, L.; Zhang, R. Interactive effects of seawater acidification and elevated temperature on biomineralization and amino acid metabolism in the mussel Mytilus edulis. J. Exp. Biol. 2015, 218, 3623–3631. [Google Scholar] [CrossRef]
- Byrne, M.; Smith, A.M.; West, S.; Collard, M.; Dubois, P.; Graba-landry, A.; Dworjanyn, S.A. Warming influences Mg2+ content, while warming and acidification influence calcification and test strength of a sea urchin. Environ. Sci. Technol. 2014, 48, 12620–12627. [Google Scholar] [CrossRef]
- Byrne, M. Impact of ocean warming and ocean acidification on marine invertebrate life history stages: Vulnerabilities and potential for persistence in a changing ocean. In Oceanography and Marine Biology: An Annual Review; Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Smith, I.P., Hughes, D.J., Eds.; Taylor & Francis: Boca Raton, FL, USA, 2011; pp. 1–42. [Google Scholar]
- Politi, Y.; Arad, T.; Klein, E.; Weiner, S.; Addadi, L. Sea urchin spine calcite forms via a transient amorphous calcium carbonate phase. Science 2004, 306, 1161–1164. [Google Scholar] [CrossRef]
- Kokorin, A.I.; Mirantsev, G.V.; Rozhnov, S.V. General features of echinoderm skeleton formation. Paleontol. J. 2015, 48, 1532–1539. [Google Scholar] [CrossRef]
- Borremans, C.; Hermans, J.; Baillon, S.; André, L.; Dubois, P. Salinity effects on the Mg/Ca and Sr/Ca in starfish skeletons and the echinoderm relevance for paleoenvironmental reconstructions. Geology 2009, 37, 351–354. [Google Scholar] [CrossRef]
- Dubois, P. The skeleton of postmetamorphic echinoderms in a changing world. Biol. Bull. 2014, 226, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Weiner, S. Organic matrixlike macromolecules associated with the mineral phase of sea urchin skeletal plates and teeth. J. Exp. Zool. 1985, 234, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Killian, C.E.; Wilt, F.H. Characterization of the proteins comprising the integral matrix of Strongylocentrotus purpuratus embryonic spicules. J. Biol. Chem. 1996, 271, 9150–9159. [Google Scholar] [CrossRef]
- Addadi, L.; Weiner, S. Control and design principles in biological mineralization. Angew. Chem. Int. Ed. 1992, 31, 153–169. [Google Scholar] [CrossRef]
- Hermans, J.; Andre, L.; Navez, J.; Pernet, P.; Dubois, P. Relative influences of solution composition and presence of intracrystalline proteins on magnesium incorporation in calcium carbonate minerals: Insight into vital effects. J. Geophys. Res. 2011, 116, G01001. [Google Scholar] [CrossRef]
- Ries, J.B. A physicochemical framework for interpreting the biological calcification response to CO2-induced ocean acidification. Geochim. Cosmochim. Acta 2011, 75, 4053–4064. [Google Scholar] [CrossRef]
- Mackenzie, F.T.; Bischoff, W.D.; Bishop, F.C.; Loijens, M.; Schoonmaker, J.; Wollast, R. Magnesian Calcites: Low Temperature Occurrence, Solubility and Solid Solution Behavior. In Carbonates: Mineralogy and Chemistry, Reviews in Mineralogy; Richard, J.R., Ed.; Mineralogical Society of America: Stony Brook, NY, USA, 1983; Volume 11, pp. 97–144. [Google Scholar]
- Knoll, A.H. Biomineralization and evolutionary history. Rev. Mineral. Geochem. 2003, 54, 329–356. [Google Scholar] [CrossRef]
- Byrne, M.; Fitzer, S. The impact of environmental acidification on the microstructure and mechanical integrity of marine invertebrate skeletons. Conserv. Physiol. 2019, 7, coz062. [Google Scholar] [CrossRef]
- Andersson, A.J.; Mackenzie, F.T.; Bates, N.R. Life on the margin: Implications of ocean acidification on Mg-calcite, high latitude and cold-water marine calcifiers. Mar. Ecol. Prog. Ser. 2008, 373, 265–273. [Google Scholar] [CrossRef]
- O’Loughlin, P.M.; Rowe, F.W.E. A systematic revision of the asterinid genus Aquilonastra O’Loughlin, 2004 (Echinodermata: Asteroidea). Mem. Mus. Vic. 2006, 63, 257–287. [Google Scholar] [CrossRef]
- Menge, B.A.; Sanford, E. Ecological role of sea stars from populations to meta ecosystems. In Starfish: Biology and Ecology of the Asteroidea; Lawrence, J.M., Ed.; The Johns Hopkins University Press: Baltimore, MD, USA, 2013; pp. 67–80. [Google Scholar]
- Balogh, R.; Byrne, M. Developing in a warming intertidal, negative carry over effects of heatwave conditions in development to the pentameral starfish in Parvulastra exigua. Mar. Environ. Res. 2020, 162, 105083. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.D.; Byrne, M. Early benthic juvenile Parvulastra exigua (Asteroidea) are tolerant to extreme acidification and warming in its intertidal habitat. J. Exp. Mar. Biol. Ecol. 2014, 453, 36–42. [Google Scholar] [CrossRef]
- McElroy, D.J.; Nguyen, H.D.; Byrne, M. Respiratory response of the intertidal seastar Parvulastra exigua to contemporary and near-future pulses of warming and hypercapnia. J. Exp. Mar. Biol. Ecol. 2012, 416–417, 1–7. [Google Scholar] [CrossRef]
- Khalil, M.; Doo, S.S.; Stuhr, M.; Westphal, H. Leibniz Centre for Tropical Marine Research (ZMT), Bremen, Germany. 2022, Unpublished Work.
- Dickson, A.G.; Sabine, C.L.; Christian, J.R. (Eds.) Guide to Best Practices for Ocean CO2 Measurements; PICES Special Publication 3; North Pacific Marine Science Organization: British Columbia, Canada, 2007; Volume 3, p. 191. [Google Scholar]
- Bray, L.; Pancucci-Papadopoulou, M.A.; Hall-Spencer, J.M. Sea urchin response to rising pCO2 shows ocean acidification may fundamentally alter the chemistry of marine skeletons. Mediterr. Mar. Sci. 2014, 15, 510–519. [Google Scholar] [CrossRef][Green Version]
- Ehrlich, H.; Elkin, Y.N.; Artoukov, A.A.; Stonik, V.A.; Safronov, P.P.; Bazhenov, V.V.; Kurek, D.V.; Varlamov, V.P.; Born, R.; Meissner, H.; et al. Simple method for preparation of nanostructurally organized spines of sand dollar Scaphechinus mirabilis (Agassiz, 1863). Mar. Biotechnol. 2011, 13, 402–410. [Google Scholar] [CrossRef]
- Russell, A.D.; Hönisch, B.; Spero, H.J.; Lea, D.W. Effects of seawater carbonate ion concentration and temperature on shell U, Mg, and Sr in cultured planktonic foraminifera. Geochim. Cosmochim. Acta 2004, 68, 4347–4361. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022. [Google Scholar]
- Shapiro, S.S.; Wilk, M.B. An analysis of variance test for normality (complete samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Levene, H. Robust tests for equality of variances. In Contributions to Probability and Statistics; Olkin, I., Ed.; Stanford University Press: Palo Alto, CA, USA, 1960; pp. 278–292. [Google Scholar]
- De Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research. R Package Version 1.3-5. 2021. Available online: https://CRAN.R-project.org/package=agricolae (accessed on 19 January 2022).
- Weber, J.N. The incorporation of magnesium into the skeletal calcites of echinoderms. Am. J. Sci. 1969, 267, 537–566. [Google Scholar] [CrossRef]
- Rosenthal, Y.; Boyle, E.A.; Slowey, N. Temperature control on the incorporation of magnesium, strontium, fluorine, and cadmium into benthic foraminiferal shells from Little Bahama Bank: Prospects for thermocline paleoceanography. Geochim. Cosmochim. Acta 1997, 61, 3633–3643. [Google Scholar] [CrossRef]
- Purgstaller, B.; Mavromatis, V.; Goetschl, K.E.; Steindl, F.R.; Dietzel, M. Effect of temperature on the transformation of amorphous calcium magnesium carbonate with near-dolomite stoichiometry into high Mg-calcite. CrystEngComm 2021, 23, 1969–1981. [Google Scholar] [CrossRef]
- Radha, A.V.; Fernandez-Martinez, A.; Hu, Y.; Jun, Y.-S.; Waychunas, G.A.; Navrotsky, A. Energetic and structural studies of amorphous Ca1−xMgxCO3·nH2O (0 ≤ x ≤ 1). Geochim. Cosmochim. Acta 2012, 90, 83–95. [Google Scholar] [CrossRef]
- Di Lorenzo, F.; Rodríguez-Galán, R.M.; Prieto, M. Kinetics of the solvent-mediated transformation of hydromagnesite into magnesite at different temperatures. Mineral. Mag. 2018, 78, 1363–1372. [Google Scholar] [CrossRef]
- Stumpp, M.; Hu, M.Y. pH Regulation and Excretion in Echinoderms. In Acid-Base Balance and Nitrogen Excretion in Invertebrates; Weihrauch, D., O’Donnell, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 261–273. [Google Scholar]
- Miles, H.; Widdicombe, S.; Spicer, J.I.; Hall-Spencer, J. Effects of anthropogenic seawater acidification on acid-base balance in the sea urchin Psammechinus miliaris. Mar. Pollut. Bull. 2007, 54, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Asnaghi, V.; Mangialajo, L.; Gattuso, J.-P.; Francour, P.; Privitera, D.; Chiantore, M. Effects of ocean acidification and diet on thickness and carbonate elemental composition of the test of juvenile sea urchins. Mar. Environ. Res. 2014, 93, 78–84. [Google Scholar] [CrossRef]
- Lorens, R.B. Sr, Cd, Mn and Co distribution coefficients in calcite as a function of calcite precipitation rate. Geochim. Cosmochim. Acta 1981, 45, 553–561. [Google Scholar] [CrossRef]
- Lea, D.W.; Mashiotta, T.A.; Spero, H.J. Controls on magnesium and strontium uptake in planktonic foraminifera determined by live culturing. Geochim. Cosmochim. Acta 1999, 63, 2369–2379. [Google Scholar] [CrossRef]
- Stoll, H.M.; Rosenthal, Y.; Falkowski, P. Climate proxies from Sr/Ca of coccolith calcite: Calibrations from continuous culture of Emiliania huxleyi. Geochim. Cosmochim. Acta 2002, 66, 927–936. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Tesoriero, A.J.; Pankow, J.F. Solid solution partitioning of Sr2+, Ba2+, and Cd2+ to calcite. Geochim. Cosmochim. Acta 1996, 60, 1053–1063. [Google Scholar] [CrossRef]
- Mucci, A.; Morse, J.W. The incorporation of Mg2+ and Sr2+ into calcite overgrowths: Influences of growth rate and solution composition. Geochim. Cosmochim. Acta 1983, 47, 217–233. [Google Scholar] [CrossRef]
- Zeebe, R.E.; Wolf-Gladrow, D. CO2 in Seawater: Equilibrium, Kinetics, Isotopes; Elsevier: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Dubois, P.; Chen, C.P. Calcification in echinoderms. In Echinoderm Studies; Jangoux, M., Lawrence, J.M., Eds.; AA Balkema: Rotterdam, The Netherlands, 1989; Volume 3, pp. 109–178. [Google Scholar]
- Melzner, F.; Mark, F.C.; Seibel, B.A.; Tomanek, L. Ocean acidification and coastal marine invertebrates: Tracking CO2 effects from seawater to the cell. Ann. Rev. Mar. Sci. 2020, 12, 499–523. [Google Scholar] [CrossRef] [PubMed]
- Feely, R.; Doney, S.; Cooley, S. Ocean acidification: Present conditions and future changes in a high-CO₂ world. Oceanography 2009, 22, 36–47. [Google Scholar] [CrossRef]
- Challener, R.C.; McClintock, J.B.; Makowsky, R. Effects of reduced carbonate saturation state on early development in the common edible sea urchin Lytechinus variegatus: Implications for land-based aquaculture. J. Appl. Aquac. 2013, 25, 154–175. [Google Scholar] [CrossRef]
- McClintock, J.B.; Amsler, M.O.; Angus, R.A.; Challener, R.C.; Schram, J.B.; Amsler, C.D.; Mah, C.L.; Cuce, J.; Baker, B.J. The Mg-Calcite composition of antarctic echinoderms: Important implications for predicting the impacts of ocean acidification. J. Geol. 2011, 119, 457–466. [Google Scholar] [CrossRef]
- Johnson, R.; Harianto, J.; Thomson, M.; Byrne, M. The effects of long-term exposure to low pH on the skeletal microstructure of the sea urchin Heliocidaris erythrogramma. J. Exp. Mar. Biol. Ecol. 2020, 523, 151250. [Google Scholar] [CrossRef]
- Hazan, Y.; Wangensteen, O.S.; Fine, M. Tough as a rock-boring urchin: Adult Echinometra sp. EE from the Red Sea show high resistance to ocean acidification over long-term exposures. Mar. Biol. 2014, 161, 2531–2545. [Google Scholar] [CrossRef]
- Gibbin, E.M.; Massamba N’Siala, G.; Chakravarti, L.J.; Jarrold, M.D.; Calosi, P. The evolution of phenotypic plasticity under global change. Sci. Rep. 2017, 7, 17253. [Google Scholar] [CrossRef]
- Stumpp, M.; Hu, M.Y.; Melzner, F.; Gutowska, M.A.; Dorey, N.; Himmerkus, N.; Holtmann, W.C.; Dupont, S.T.; Thorndyke, M.C.; Bleich, M. Acidified seawater impacts sea urchin larvae pH regulatory systems relevant for calcification. Proc. Natl. Acad. Sci. USA 2012, 109, 18192–18197. [Google Scholar] [CrossRef]
- Wood, H.L.; Spicer, J.I.; Widdicombe, S. Ocean acidification may increase calcification rates, but at a cost. Proc. Biol. Sci. 2008, 275, 1767–1773. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, G.; Lewis, E.; Wallace, D. CO2SYS.xls: A calculator for the CO2 System in Seawater for Microsoft Excel/VBA. 2007. Available online: https://www.baltex-research.eu/baltic2009/downloads/Lectures/Bernd_Schneider/co2sys_ver14.xls (accessed on 19 January 2022).
- Hansson, I. A new set of acidity constants for carbonic acid and boric acid in sea water. Deep Sea Res. Oceanogr. Abstr. 1973, 20, 461–478. [Google Scholar] [CrossRef]
- Mehrbach, C.; Culberson, C.H.; Hawley, J.E.; Pytkowicx, R.M. Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnol. Oceanogr. 1973, 18, 897–907. [Google Scholar] [CrossRef]
- Dickson, A.; Millero, F. A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep Sea Res. Part A Oceanogr. Res. Pap. 1987, 34, 1733–1743. [Google Scholar] [CrossRef]
- Dickson, A.G. Standard potential of the reaction: AgCl(s) + 1/2H2(g) = Ag(s) + HCl(aq), and the standard acidity constant of the ion HSO4- in synthetic seawater from 273.15 to 318.15 K. J. Chem. Thermodyn. 1990, 22, 113–127. [Google Scholar] [CrossRef]
- Dickson, A.; Riley, J. The estimation of acid dissociation constants in seawater media from potentionmetric titrations with strong base. I. The ionic product of water—Kw. Mar. Chem. 1979, 7, 89–99. [Google Scholar] [CrossRef]
- Uppström, L.R. The boron/chlorinity ratio of deep-sea water from the Pacific Ocean. Deep Sea Res. Oceanogr. Abstr. 1974, 21, 161–162. [Google Scholar] [CrossRef]
- Mucci, A. The solubility of calcite and aragonite in seawater at various salinities, temperatures, and one atmosphere total pressure. Am. J. Sci. 1983, 283, 780–799. [Google Scholar] [CrossRef]
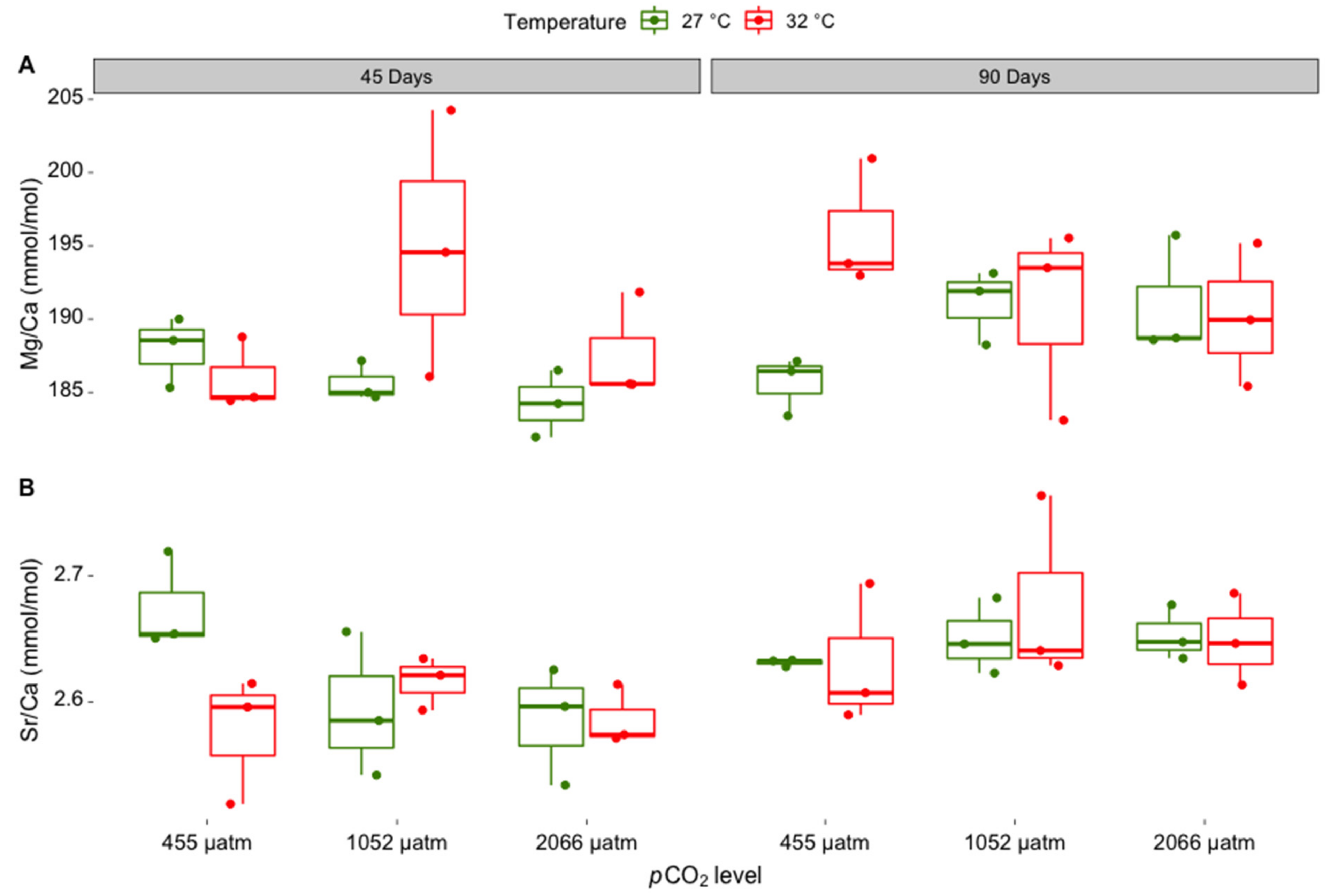
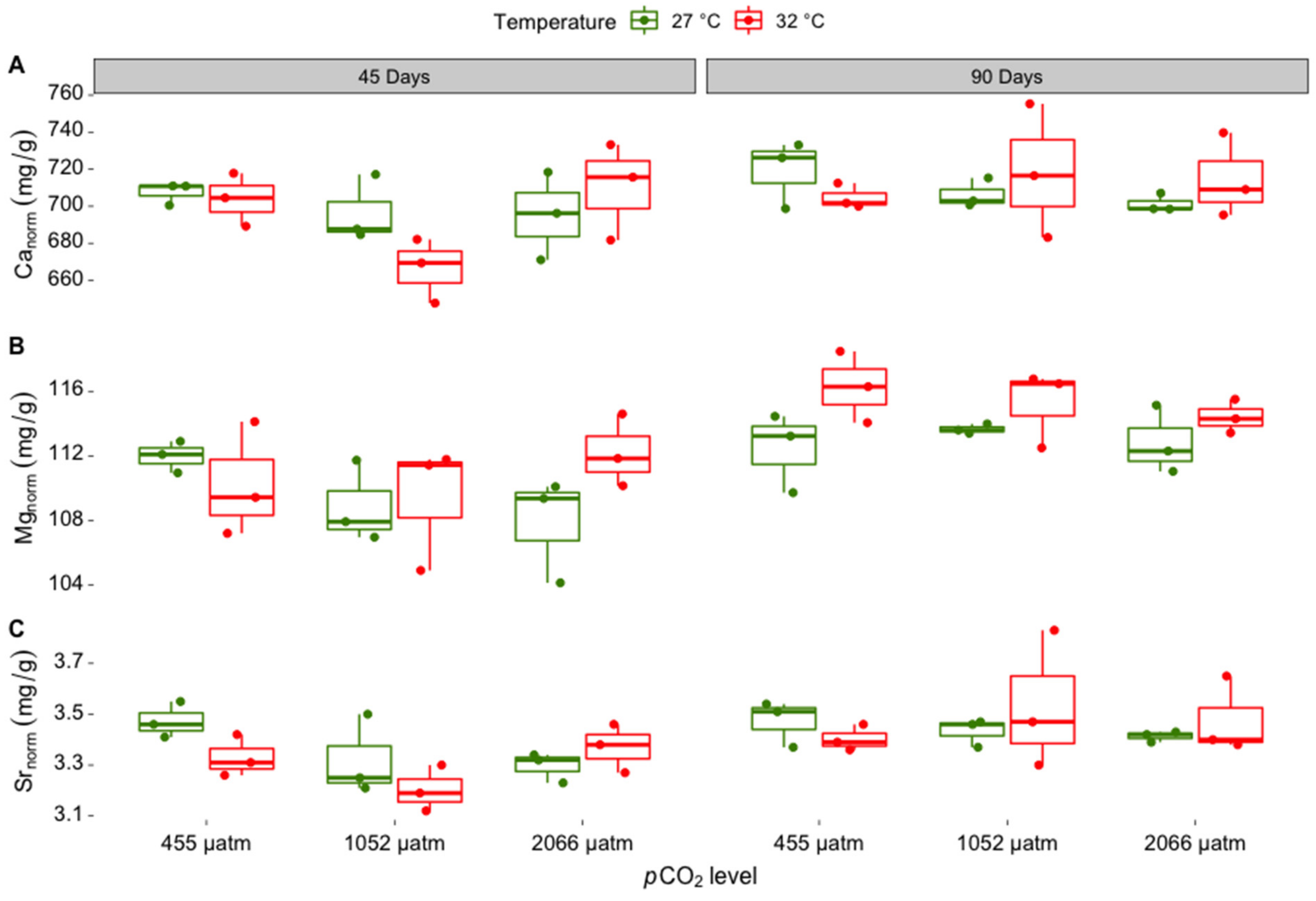
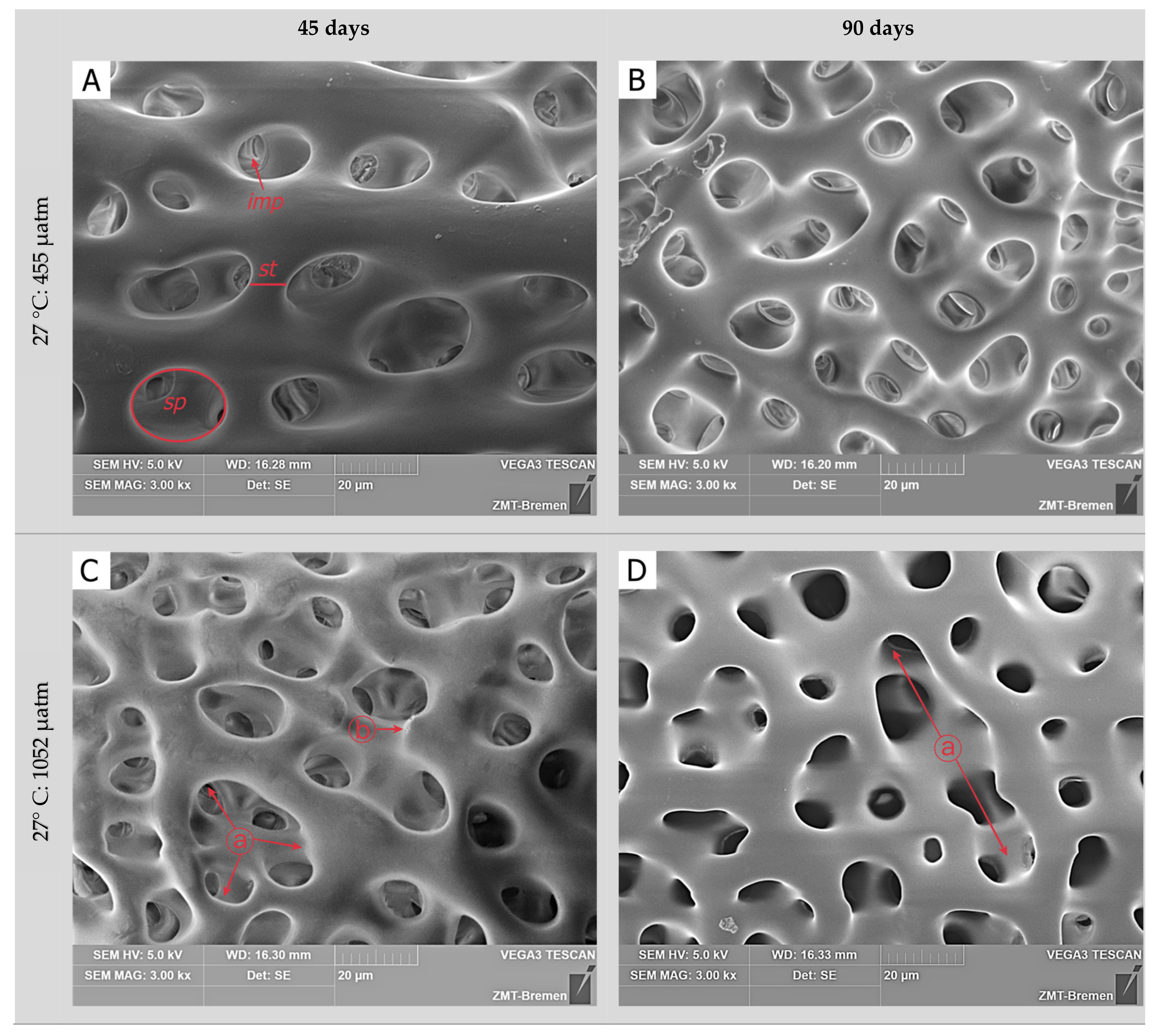

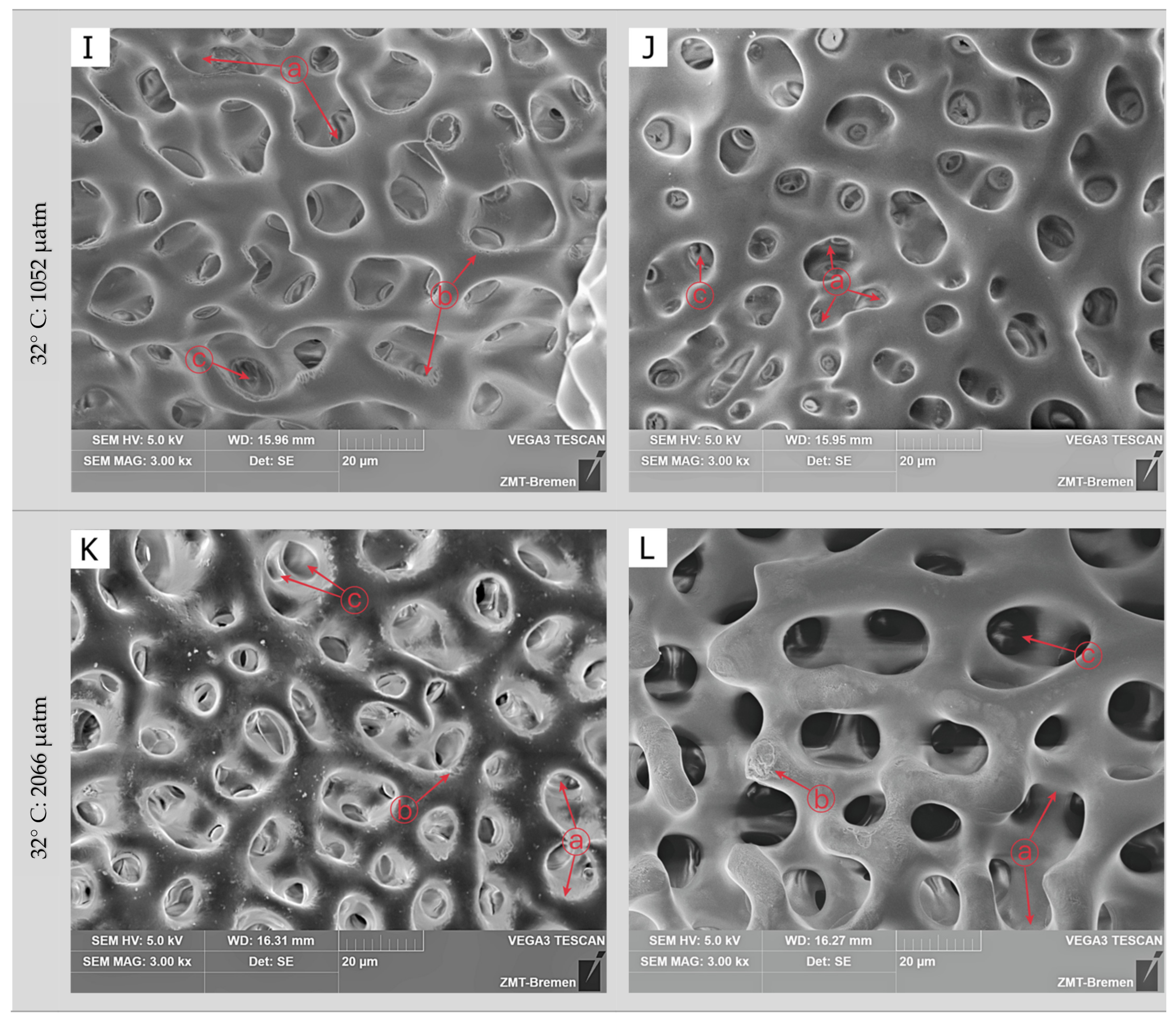
| Treatment | Measured Parameters | |||||
| Salinity (PSU) | Temperature (°C) | pH(NBS scale) | pH(total scale) | AT (µmol/kg-SW) | DIC (µmol/kg-SW | |
| 27 °C: 455 µatm | 34.56 ± 0.12 | 27.48 ± 0.06 | 8.13 ± 0.00 | 8.00 ± 0.00 | 2504.42 ± 15.33 | 2168.86 ± 15.23 |
| 27 °C: 1052 µatm | 34.73 ± 0.06 | 27.23 ± 0.04 | 7.87 ± 0.01 | 7.74 ± 0.01 | 2514.45 ± 16.78 | 2340.99 ± 11.62 |
| 27 °C: 2066 µatm | 34.75 ± 0.05 | 27.34 ± 0.03 | 7.60 ± 0.01 | 7.47 ± 0.01 | 2539.83 ± 38.86 | 2479.40 ± 36.58 |
| 32 °C: 455 µatm | 34.65 ± 0.08 | 32.03 ± 0.05 | 8.13 ± 0.00 | 8.00 ± 0.00 | 2510.27 ± 47.18 | 2134.38 ± 38.40 |
| 32 °C: 1052 µatm | 34.78 ± 0.05 | 32.10 ± 0.04 | 7.87 ± 0.00 | 7.74 ± 0.00 | 2532.19 ± 40.05 | 2325.20 ± 40.02 |
| 32 °C: 2066 µatm | 34.76 ± 0.02 | 32.20 ± 0.08 | 7.60 ± 0.01 | 7.47 ± 0.01 | 2584.38 ± 41.93 | 2493.30 ± 37.87 |
| Treatment | Calculated Parameters | |||||
| pCO2 (µatm) | [CO32−] (µmol/kg-SW) | [HCO3−] (µmol/kg-SW) | [CO2] (µmol/kg-SW) | ΩCa | ΩAr | |
| 27 °C: 455 µatm | 456.13 ± 8.24 | 245.64 ± 2.52 | 1911.05 ± 14.94 | 12.17 ± 0.22 | 5.96 ± 0.06 | 3.96 ± 0.04 |
| 27 °C: 1052 µatm | 1059.58 ± 32.04 | 138.12 ± 4.64 | 2178.74 ± 9.58 | 28.42 ± 0.86 | 3.35 ± 0.11 | 2.22 ± 0.07 |
| 27 °C: 2066 µatm | 2075.40 ± 30.99 | 81.18 ± 2.35 | 2342.71 ± 34.29 | 55.51 ± 0.82 | 1.97 ± 0.06 | 1.31 ± 0.04 |
| 32 °C: 455 µatm | 453.78 ± 6.51 | 273.82 ± 8.23 | 1849.66 ± 30.77 | 10.90 ± 0.16 | 6.71 ± 0.20 | 4.52 ± 0.14 |
| 32 °C: 1052 µatm | 1045.15 ± 44.00 | 162.68 ± 4.67 | 2147.47 ± 38.59 | 25.05 ± 1.06 | 3.98 ± 0.11 | 2.69 ± 0.08 |
| 32 °C: 2066 µatm | 2057.31 ± 74.42 | 99.46 ± 4.64 | 2344.63 ± 34.94 | 49.20 ± 1.78 | 2.44 ± 0.11 | 1.64 ± 0.08 |
| Skeletal Mineral Ratio | df | F | Pr (<F) | Post-Hoc Test Result |
|---|---|---|---|---|
| Mg/Ca ratio | ||||
| Incubation time | 1 | 4.319 | 0.049 | 90 days > 45 days |
| pCO2 | 2 | 0.915 | 0.414 | |
| Temperature | 1 | 5.144 | 0.033 | 32 °C > 27 °C |
| Incubation time: pCO2 | 2 | 0.715 | 0.500 | |
| Incubation time:temperature | 1 | 0.039 | 0.845 | |
| pCO2: temperature | 2 | 0.478 | 0.626 | |
| Incubation time: pCO2: temperature | 2 | 5.143 | 0.014 | n.s., electronic Supplementary Materials Table S1 |
| Residuals | 24 | |||
| Sr/Ca ratio | ||||
| Incubation time | 1 | 9.027 | 0.006 | 90 days > 45 days |
| pCO2 | 2 | 0.405 | 0.671 | |
| Temperature | 1 | 0.481 | 0.495 | |
| Incubation time: pCO2 | 2 | 1.794 | 0.188 | |
| Incubation time: temperature | 1 | 1.519 | 0.230 | |
| pCO2: temperature | 2 | 2.016 | 0.155 | |
| Incubation time: pCO2: temperature | 2 | 1.132 | 0.339 | |
| Residuals | 24 | |||
| Canorm ratio | ||||
| Incubation time | 1 | 4.951 | 0.036 | 90 days > 45 days |
| pCO2 | 2 | 1.242 | 0.307 | |
| Temperature | 1 | 0.043 | 0.838 | |
| Incubation time: pCO2 | 2 | 1.711 | 0.202 | |
| Incubation time: temperature | 1 | 0.59 | 0.450 | |
| pCO2: temperature | 2 | 1.471 | 0.250 | |
| Incubation time: pCO2: temperature | 2 | 1.65 | 0.213 | |
| Residuals | 24 | |||
| Mgnorm ratio | ||||
| Incubation time | 1 | 24.523 | 0.001 | 90 days > 45 days |
| pCO2 | 2 | 0.582 | 0.566 | |
| Temperature | 1 | 4.206 | 0.051 | 32 °C > 27 °C |
| Incubation time: pCO2 | 2 | 0.616 | 0.548 | |
| Incubation time: temperature | 1 | 0.621 | 0.438 | |
| pCO2: temperature | 2 | 0.609 | 0.552 | |
| Incubation time: pCO2: temperature | 2 | 2.133 | 0.140 | |
| Residuals | 24 | |||
| Srnorm ratio | ||||
| Incubation time | 1 | 9.814 | 0.005 | 90 days > 45 days |
| pCO2 | 2 | 0.500 | 0.613 | |
| Temperature | 1 | 0.156 | 0.696 | |
| Incubation time: pCO2 | 2 | 1.862 | 0.177 | |
| Incubation time: temperature | 1 | 1.405 | 0.248 | |
| pCO2: temperature | 2 | 1.655 | 0.212 | |
| Incubation time: pCO2: temperature | 2 | 0.707 | 0.503 | |
| Residuals | 24 |
| Incubation Time | Temperature | pCO2 | Skeletal Surface | Stereom Pores | Inner Matrix Pores |
|---|---|---|---|---|---|
| 45 days | 27 °C | 455 µatm | ND | ES | EP |
| 1052 µatm | DS | US | EP | ||
| 2066 µatm | DS | US | UP | ||
| 32 °C | 455 µatm | ND | ES | EP | |
| 1052 µatm | DS | US | UP | ||
| 2066 µatm | HD | HU | UP | ||
| 90 days | 27 °C | 455 µatm | ND | ES | EP |
| 1052 µatm | DS | US | EP | ||
| 2066 µatm | DS | US | UP | ||
| 32 °C | 455 µatm | ND | ES | EP | |
| 1052 µatm | DS | US | UP | ||
| 2066 µatm | HD | HU | UP |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalil, M.; Doo, S.S.; Stuhr, M.; Westphal, H. Ocean Warming Amplifies the Effects of Ocean Acidification on Skeletal Mineralogy and Microstructure in the Asterinid Starfish Aquilonastra yairi. J. Mar. Sci. Eng. 2022, 10, 1065. https://doi.org/10.3390/jmse10081065
Khalil M, Doo SS, Stuhr M, Westphal H. Ocean Warming Amplifies the Effects of Ocean Acidification on Skeletal Mineralogy and Microstructure in the Asterinid Starfish Aquilonastra yairi. Journal of Marine Science and Engineering. 2022; 10(8):1065. https://doi.org/10.3390/jmse10081065
Chicago/Turabian StyleKhalil, Munawar, Steve S. Doo, Marleen Stuhr, and Hildegard Westphal. 2022. "Ocean Warming Amplifies the Effects of Ocean Acidification on Skeletal Mineralogy and Microstructure in the Asterinid Starfish Aquilonastra yairi" Journal of Marine Science and Engineering 10, no. 8: 1065. https://doi.org/10.3390/jmse10081065
APA StyleKhalil, M., Doo, S. S., Stuhr, M., & Westphal, H. (2022). Ocean Warming Amplifies the Effects of Ocean Acidification on Skeletal Mineralogy and Microstructure in the Asterinid Starfish Aquilonastra yairi. Journal of Marine Science and Engineering, 10(8), 1065. https://doi.org/10.3390/jmse10081065









