Waste to Energy Onboard Cruise Ships: A New Paradigm for Sustainable Cruising
Abstract
:1. Introduction
2. Materials and Methods
2.1. Incineration System
- a primary combustion chamber, in which a preliminary semi-pyrolytic decomposition is achieved in oxygen deficiency and in a low turbulence atmosphere, to contain the entrainment of dust. A thin layer of solid waste is deposited on a grid, which moves it horizontally and slowly towards the discharge area, where it is finally disposed of as ash with low organic content. The chamber is completed with a burner that allows us to reach the operating temperature (950 °C) during the start-up phases, and to sustain the combustion during ordinary operation. Furthermore, an atomizing nozzle introduces into the chamber the exhausted oil, which must be disposed of;
- a secondary post-combustion chamber, designed to achieve the complete oxidation of the unburnt gas with a residence time of the fumes of at least 2 s; fresh air with high turbulence is admitted into the chamber to ensure optimal mixing. To maintain the temperature above the 850 °C required by the regulations, a burner fed with used fuel oil is installed;
- the third chamber allows us to attain the prescribed minimum transit time of the fumes and to achieve the sedimentation of coarse dust, as a consequence of the sudden impact of the fumes with a solid surface, caused by abrupt path deviation.
- efficiency of the combustion chamber (taking into account heat losses through isolation, ∼0.97);
- lower calorific value of combustion waste materials (kJ/kg);
- quantity of exhaust gases per 1 kg of waste burned (Nm/kg);
- specific heat power of the exhaust gas (kJ/Nm °C);
- temperature of the air entering the combustion chamber (°C).
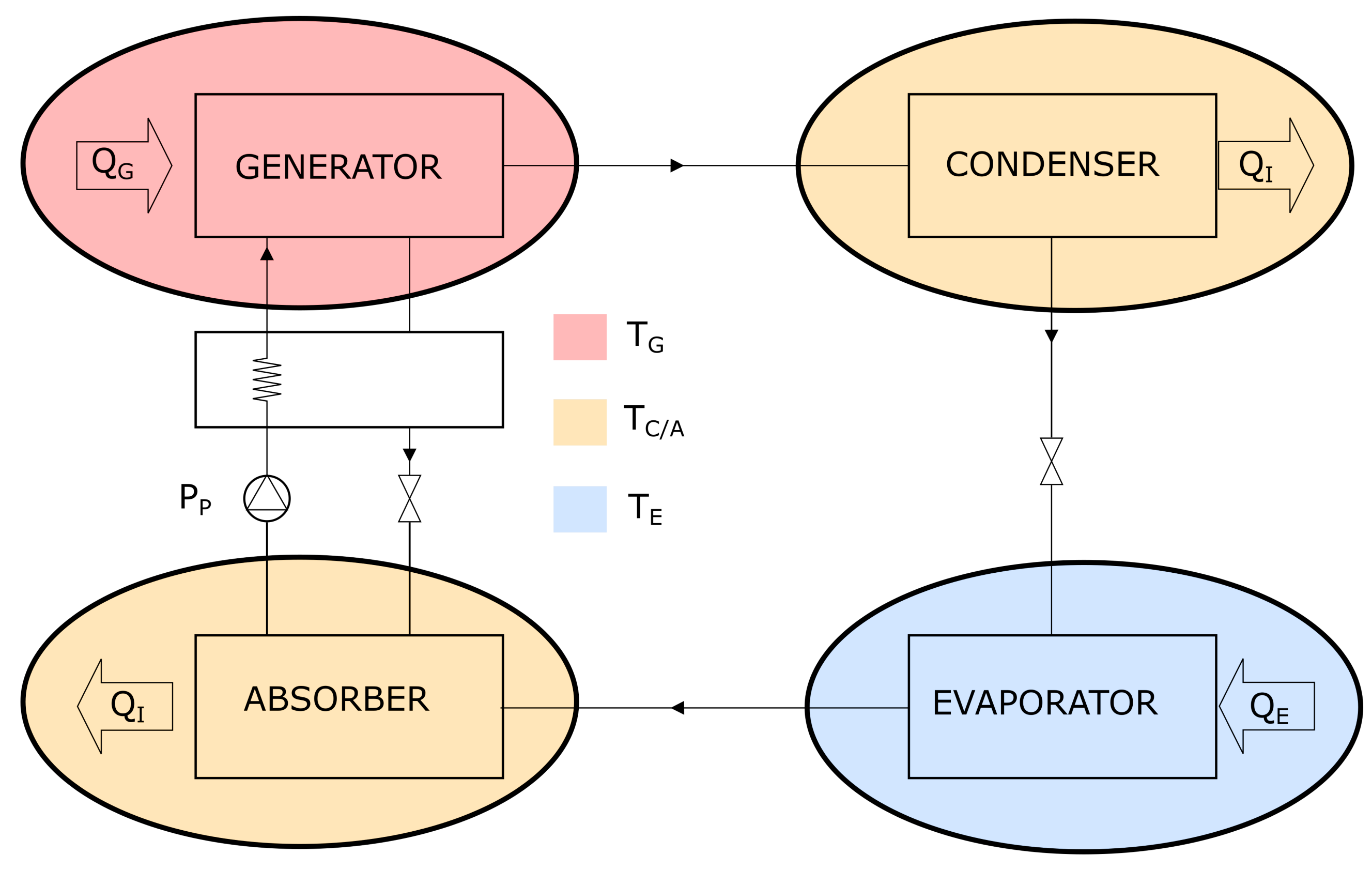
2.2. Absorption Cycles
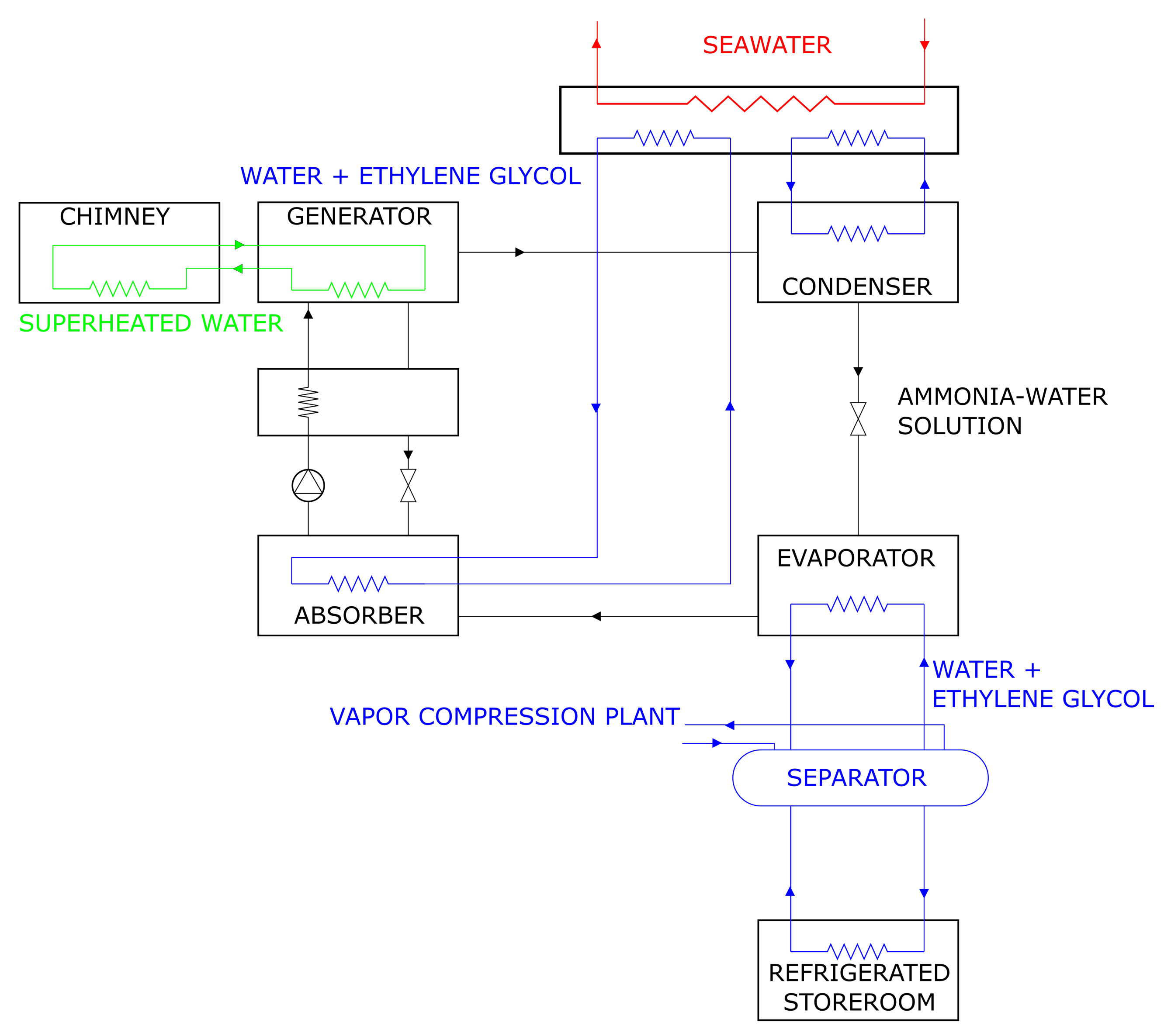
- In the generator, ammonia separates from a lean (i.e., with reduced concentration in ammonia) water-ammonia solution. The reaction is endothermic: the required heat is transferred from the exhaust gas leaving the incinerator, through an intermediate pressurized-water heat transfer circuit (IPWHTC). Leaving the generator, the evaporated, rich (in ammonia) solution enters a distillation column, where it is further concentrated via an endothermic process, still deriving the necessary heat from the exhaust gas of the incinerator. The concentrated vapor leaving the distillation column is referred to as ammonia vapor.
- The ammonia vapor returns to the state of saturated liquid in the condenser, transferring heat () to a flow of seawater through an intermediate pressurized-water heat transfer circuit.;
- The condensed ammonia is forced through a lamination valve, which lowers the pressure. Part of the ammonia evaporates, resulting in a liquid-vapor mixture of ammonia with a high liquid fraction.
- In the evaporator, the mixture absorbs heat () from the ambient to be refrigerated via an IPWHTC and turns to saturated or superheated vapor.
- The vapor enters the absorber and is absorbed by the concentrated (in ) water-ammonia solution residing in it. The dissolution process is exothermic: the rejected heat is transferred to the seawater via an IPWHTC.
- A pump transfers the concentrated solution from the absorber to the generator. In the process, the concentrated solution is pre-heated in a heat exchanger by the warm flow of lean solution being transferred from the generator to the absorber.
- heat absorbed in the evaporator;
- heat supplied to the generator;
- mechanical energy supplied to the pump.
- the temperature of the condenser and absorber, , which reject heat to the seawater via an IPWHTC;
- the temperature of the generator, , imposed by the energy that can be extracted from the exhaust gases leaving the incinerator at a given temperature;
- the evaporator temperature, , set by the end user according to the needs (cold cell, freezing department, etc.).
2.3. Possible Energy Exploitation
3. Results
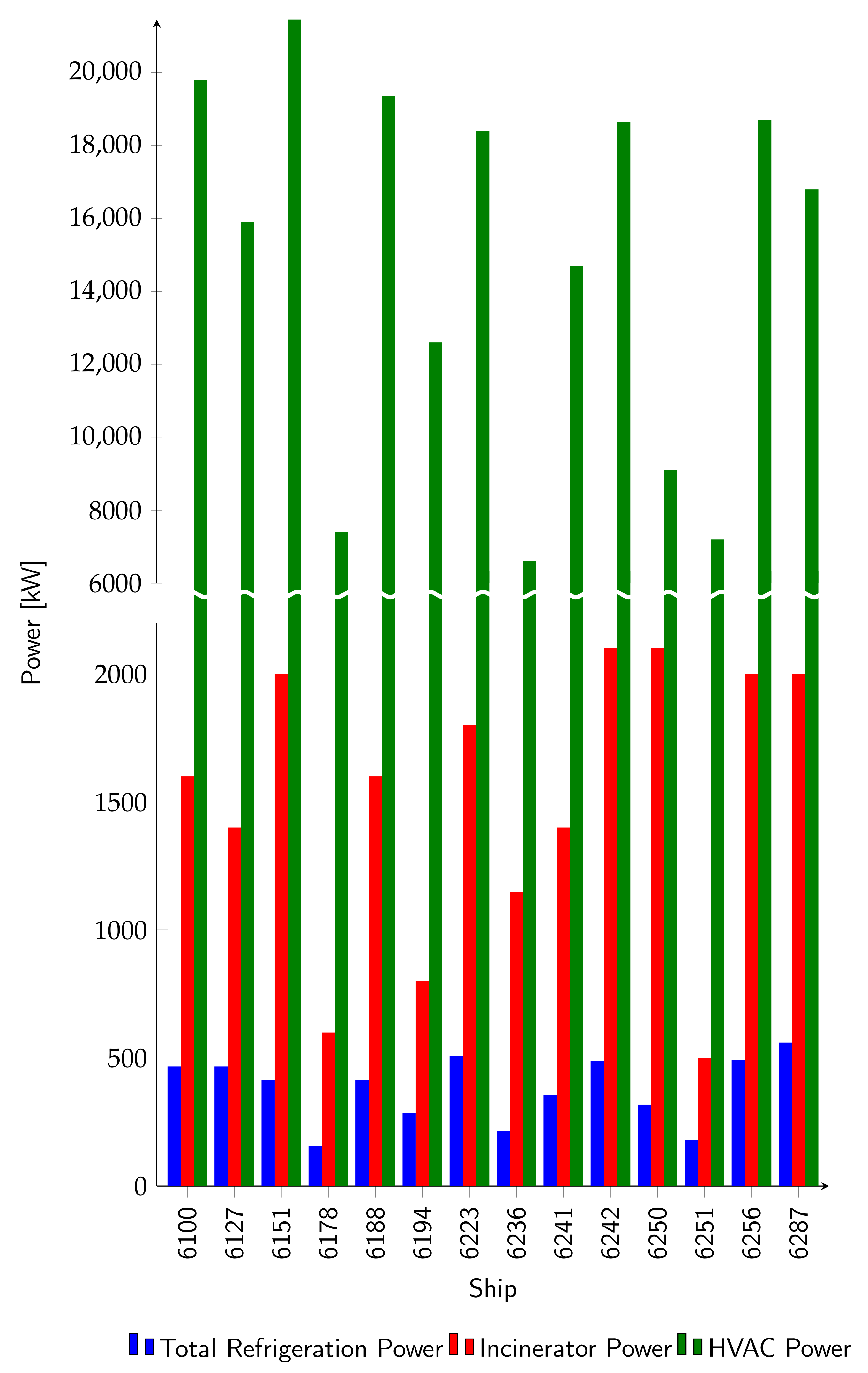
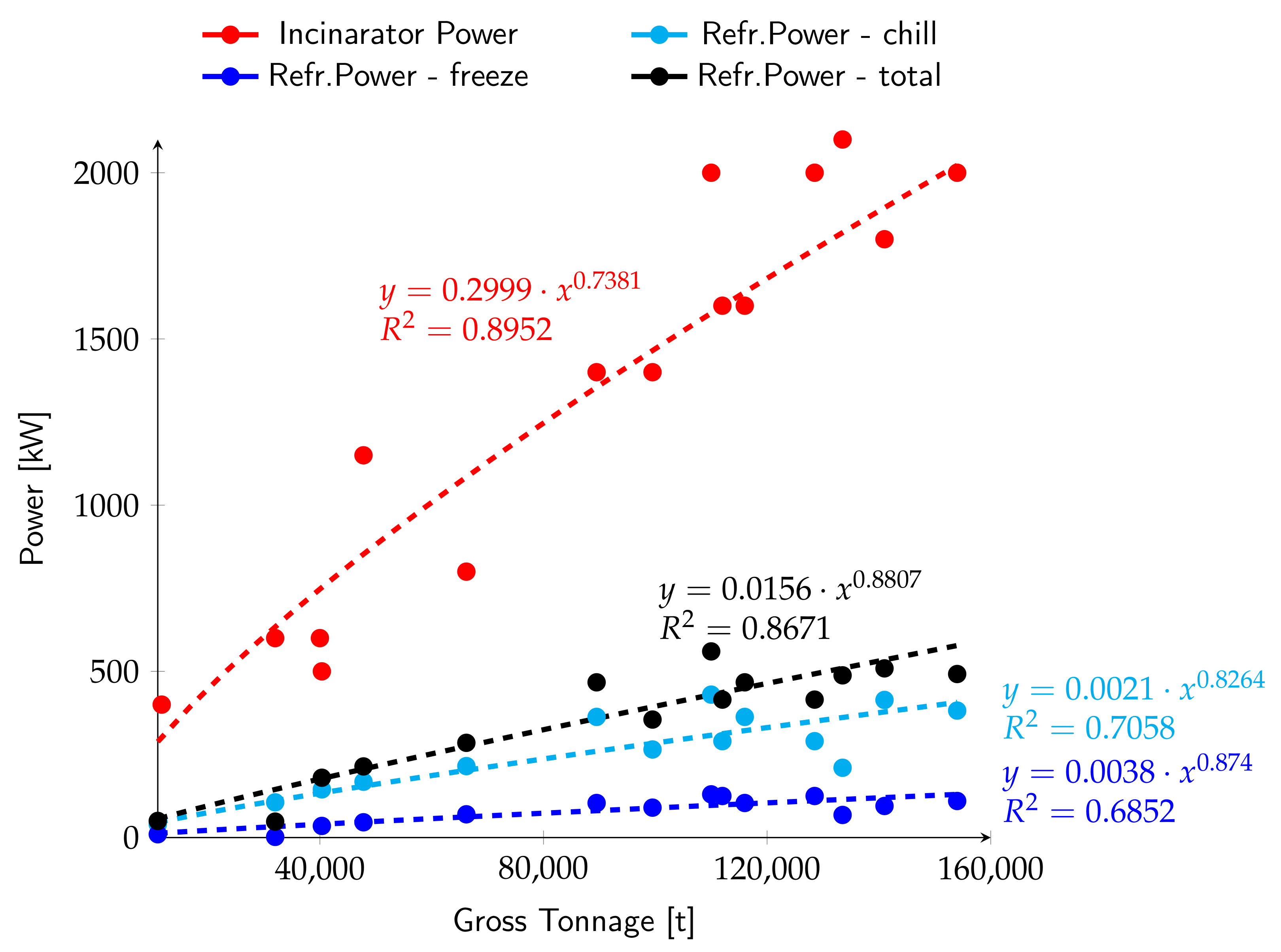
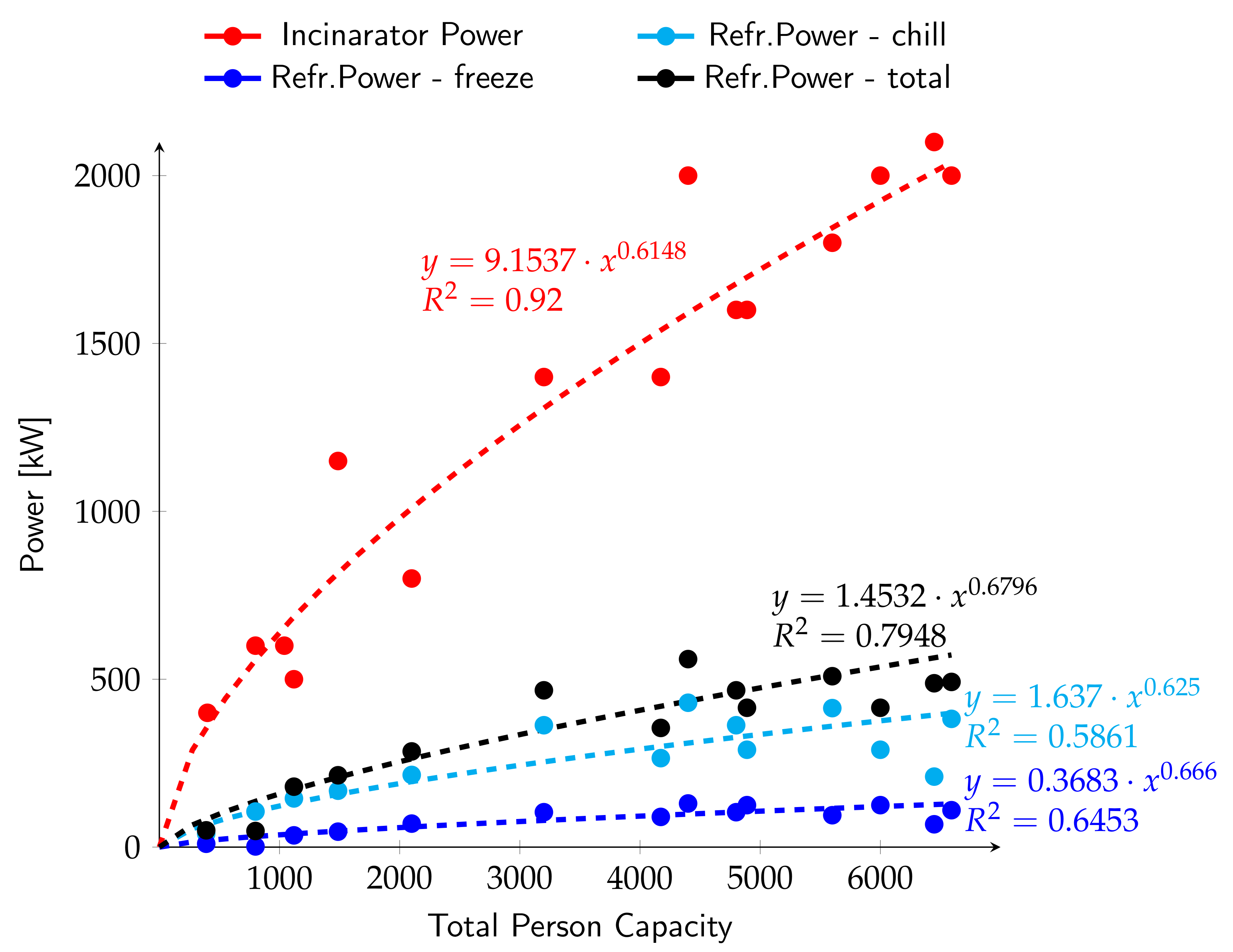
3.1. Correlation between Generated/Absorbed Power, Ship Tonnage and Person Capacity
3.2. General Considerations about the Sizing of the Absorption Plant
- CHILL: cooling power 414 kW, mechanical power 195.5 kW and COP 2.12;
- FREEZE: cooling power 95 kW, mechanical power 106 kW and COP 0.90.
3.3. Expected Energy Savings
4. Discussion
- conversion of thermal energy into electricity;
- direct or semi-direct exploitation of thermal energy.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABS | absorber |
| ARS | absorption refrigeration system |
| CND | condenser |
| COP | coefficient of performance |
| DC | distillation column |
| DE | direct expansion |
| EV | evaporator |
| GEN | generator |
| GRT | gross tonnage |
| HEX | heat exchange circuits |
| HVAC | heating, ventilation and air conditioning |
| IE | indirect expansion |
| IP | incineration plant |
| IPWHTC | intermediate pressurized-water heat transfer circuit |
| TPC | total person capacity |
| VOC | volatile organic compounds |
| IHX | recuperative heat exchanger |
| TV | throttling valve |
Appendix A. Description of the Absorption Refrigeration Cycle (ARC)
Appendix A.1. System Structure
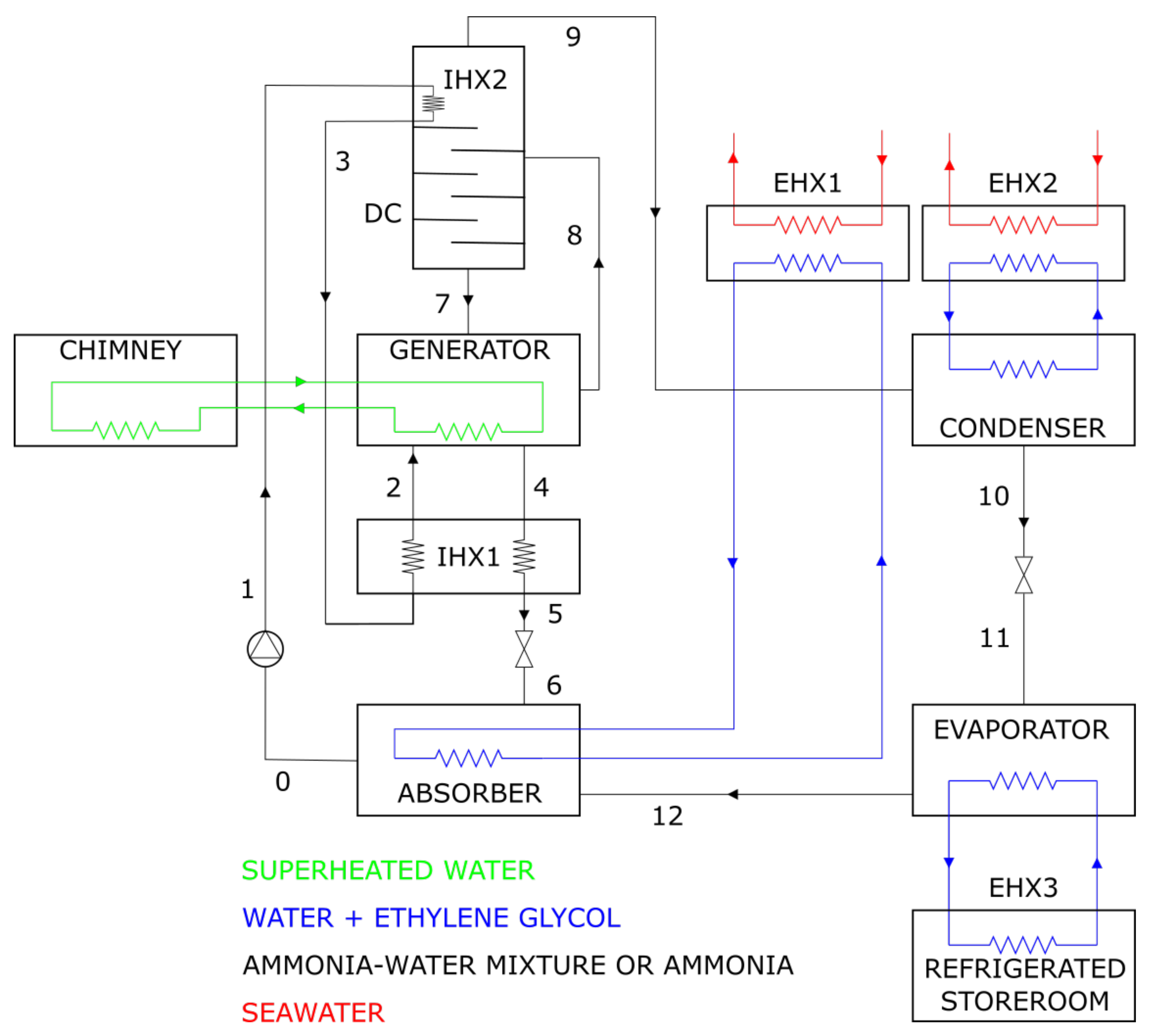
Appendix A.2. Assumptions and Design Parameters
- The system runs under steady-state conditions, and changes in the kinetic and potential energies are ignored.
- The flow is adiabatic through the pump PU and iso-enthalpic through the throttling valves TV1 and TV2.
- The flow through the connecting pipes and through the components of the ARC is free of frictional losses and of the ensuing pressure drops.
- The refrigerants at the EV and CND outlets are at the saturated vapor state and saturated liquid state, respectively. The vapor at the DC outlet contains only ammonia.
- The liquid stream of lean mixture transferred from GEN to ABS enters the ABS in saturated conditions at the evaporation pressure (which is present also in the ABS). Notice that the concentration of in the aforementioned stream remains constant as it is transferred (in liquid state) from GEN to ABS.
- Only the liquid phase of the water-ammonia mixture is withdrawn from the ABS and transferred to the GEN.
- Only the vapor phase of the water-ammonia mixture is withdrawn from the GEN and transferred to the DC.
- The stream of condensed water-ammonia mixture leaving the DC is saturated at the conditions present in the GEN.
- The inlet temperature of the water-glycol mixture in EHX1 is and , while its temperature is increased by flowing through EHX1.
- The temperature of seawater, used as a refrigerant in the EHX2 and EHX3 heat exchangers, is raised by when flowing through this components. The outlet temperature of the water-glycol solution from EHX2 and EHX3 is above the maximum seawater temperature and its temperature drop is .
- The hydraulic efficiency of the pump is .
- The desired refrigerating power , transferred from the cold room to the refrigerant fluid in the evaporator.
- The temperature of the mixture within the generator: .
- The temperature of the storage room, the temperature difference between the storage room and the water-glycol mixture in the intermediate heat exchange circuit, the temperature difference between the evaporating refrigerant fluid and the water-glycol mixture in the intermediate heat exchange circuit. Using these input parameters, the evaporation temperature can be derived.
- The seawater temperature, the admissible temperature increase for seawater, the temperature differences between the seawater and the water-glycol mixture in the intermediate heat exchange circuits. Using these input parameters, the condensation temperature and the temperature within the absorber can be calculated.
References
- Butt, N. The impact of cruise ship generated waste on home ports and ports of call: A study of Southampton. Mar. Policy 2007, 31, 591–598. [Google Scholar] [CrossRef]
- Commoy, J.; Polytika, C.A.; Nadel, R.; Bulkley, J.W. The Environmental Impact of Cruise Ships. In Impacts of Global Climate Change; American Society of Civil Engineers: Reston, VA, USA, 2005; pp. 1–12. [Google Scholar] [CrossRef]
- Klein, R.A. The cruise sector and its environmental impact. Bridg. Tour. Theory Pract. 2010, 3, 113–130. [Google Scholar] [CrossRef]
- International Maritime Organization. International Convention for the Prevention of Pollution from Ships (MARPOL); International Maritime Organiztion: London, UK, 1973. [Google Scholar]
- Batista, W.R.; Fernandes, F.C.; Lopes, C.C.; Lopes, R.S.C.; Miller, W.; Ruiz, G. Which Ballast Water Management System Will You Put Aboard? Remnant Anxieties: A Mini-Review. Environments 2017, 4, 54. [Google Scholar] [CrossRef] [Green Version]
- Jang, P.G.; Hyun, B.; Shin, K. Ballast Water Treatment Performance Evaluation under Real Changing Conditions. J. Mar. Sci. Eng. 2020, 8, 817. [Google Scholar] [CrossRef]
- Lu, K.T.; Lui, H.K.; Chen, C.T.A.; Liu, L.L.; Yang, L.; Dong, C.D.; Chen, C.W. Using Onboard-Produced Drinking Water to Achieve Ballast-Free Management. Sustainability 2021, 13, 7648. [Google Scholar] [CrossRef]
- Abuhasel, K.; Kchaou, M.; Alquraish, M.; Munusamy, Y.; Jeng, Y.T. Oily Wastewater Treatment: Overview of Conventional and Modern Methods, Challenges, and Future Opportunities. Water 2021, 13, 980. [Google Scholar] [CrossRef]
- Capodici, M.; Cosenza, A.; Di Trapani, D.; Mannina, G.; Torregrossa, M.; Viviani, G. Treatment of Oily Wastewater with Membrane Bioreactor Systems. Water 2017, 9, 412. [Google Scholar] [CrossRef] [Green Version]
- Öz, Ç.; Çetin, E. Treatment of bilge water by fenton oxidation followed by granular activated carbon adsorption. Water 2021, 13, 2792. [Google Scholar] [CrossRef]
- Mustapha, S.N.A.; Amran, N.A.; Roslan, I.L.; Chandra Segaran, R.; Samsuri, S. Potential Efficient Separation of Oil from Bilgewater and Kitchen Wastewater by Fractional Freezing Process. Crystals 2021, 11, 685. [Google Scholar] [CrossRef]
- Guilbaud, J.; Massé, A.; Andrs, Y.; Combe, F.; Jaouen, P. Laundry water recycling in ship by direct nanofiltration with tubular membranes. Resour. Conserv. Recycl. 2010, 55, 148–154. [Google Scholar] [CrossRef]
- Guilbaud, J.; Massé, A.; Andrès, Y.; Combe, F.; Jaouen, P. Influence of operating conditions on direct nanofiltration of greywaters: Application to laundry water recycling aboard ships. Resour. Conserv. Recycl. 2012, 62, 64–70. [Google Scholar] [CrossRef]
- Sanches, V.; Aguiar, M.; de Freitas, M.; Pacheco, E. Management of cruise ship-generated solid waste: A review. Mar. Pollut. Bull. 2020, 151, 110785. [Google Scholar] [CrossRef] [PubMed]
- Strazza, C.; Borghi, A.D.; Gallo, M.; Manariti, R.; Missanelli, E. Investigation of green practices for paper use reduction onboard a cruise ship—A life cycle approach. Int. J. Life Cycle Assess. 2015, 20, 982–993. [Google Scholar] [CrossRef]
- Toneatti, L.; Deluca, C.; Fraleoni-Morgera, A.; Pozzetto, D. Rationalization and optimization of waste management and treatment in modern cruise ships. Waste Manag. 2020, 118, 209–218. [Google Scholar] [CrossRef]
- American Bureau of Shipping. ABS Garbage Management Manual; ABS Group: Spring, TX, USA, 2012. [Google Scholar]
- Iannello, A.; Bertagna, S.; Pozzetto, D.; Toneatti, L.; Zamarini, R.; Bucci, V. Technical and Economic and Environmental Feasibility of an Innovative Integrated System of Management and Treatment of Waste on Board. In Technology and Science for the Ships of the Future; IOS Press: Amsterdam, The Netherlands, 2018; pp. 762–769. [Google Scholar] [CrossRef]
- Kotrikla, A.M.; Zavantias, A.; Kaloupi, M. Waste generation and management onboard a cruise ship: A case study. Ocean Coast. Manag. 2021, 212, 105850. [Google Scholar] [CrossRef]
- Prelec, Z.; Mrakovcic, T.; Dragicevic, V. Technical and Environmental Aspects of Shipboard Incinerators Design. Pomorski Zbornik 2005, 43, 207–217. [Google Scholar]
- Avellaneda, P.M.; Englehardt, J.D.; Olascoaga, J.; Babcock, E.A.; Brand, L.; Lirman, D.; Rogge, W.F.; Solo-Gabriele, H.; Tchobanoglous, G. Relative risk assessment of cruise ships biosolids disposal alternatives. Mar. Pollut. Bull. 2011, 62, 2157–2169. [Google Scholar] [CrossRef] [PubMed]
- Gonsalves, F.; Pasdeloup, B.; Billot, R.; Meyer, P.; Jacques, A.; Lorang, M. New Insights into the Propulsion Power Prediction of Cruise Ships. In Proceedings of the 2021 IEEE 33rd International Conference on Tools with Artificial Intelligence (ICTAI), Washington, DC, USA, 1–3 November 2021; pp. 846–850. [Google Scholar] [CrossRef]
- Wei, P.; Vogt, S.; Wang, D.; Gonzalez, R.E.; Yurdakul, O.; Albayrak, S. A novel energy management system for cruise ships including forecasting via LSTM. In Proceedings of the 2020 IEEE PES Innovative Smart Grid Technologies Europe (ISGT-Europe), The Hague, The Netherlands, 26–28 October 2020; pp. 1050–1054. [Google Scholar] [CrossRef]
- Firouzmakan, P.; Homayie, S.; Hooshmand, R.A. Optimal power management of electrical energy storage system, CHP, conventional and heat-only units considering both electrical and thermal loads for assessment of all-electric ship’s system. IET Electr. Syst. Transp. 2020, 10, 213–223. [Google Scholar] [CrossRef]
- Fang, S.; Fang, Y.; Wang, H.; Liu, L. Optimal Heterogeneous Energy Storage Management for Multienergy Cruise Ships. IEEE Syst. J. 2020, 14, 4754–4764. [Google Scholar] [CrossRef]
- Zhang, Q.; Luo, Z.; Zhao, Y.; Pavel, S. Thermodynamic analysis and multi-objective optimization of a transcritical CO2 waste heat recovery system for cruise ship application. Energy Convers. Manag. 2021, 227, 113612. [Google Scholar] [CrossRef]
- Gnes, P.; Pinamonti, P.; Reini, M. Bi-level optimization of the energy recovery system from internal combustion engines of a cruise ship. Appl. Sci. 2020, 10, 6917. [Google Scholar] [CrossRef]
- Alklaibi, A.; Lior, N. Waste heat utilization from internal combustion engines for power augmentation and refrigeration. Renew. Sustain. Energy Rev. 2021, 152, 111629. [Google Scholar] [CrossRef]
- Ouyang, T.; Wang, Z.; Wang, G.; Zhao, Z.; Xie, S.; Li, X. Advanced thermo-economic scheme and multi-objective optimization for exploiting the waste heat potentiality of marine natural gas engine. Energy 2021, 236, 121440. [Google Scholar] [CrossRef]
- Ouyang, T.; Huang, G.; Lu, Y.; Liu, B.; Hu, X. Multi-criteria assessment and optimization of waste heat recovery for large marine diesel engines. J. Clean. Prod. 2021, 309, 127307. [Google Scholar] [CrossRef]
- Aprhornratana, S.; Eames, I.W. Thermodynamic analysis of absorption refrigeration cycles using the second law of thermodynamics method. Int. J. Refrig. 1995, 18, 244–252. [Google Scholar] [CrossRef]
- Boccaletti, C. Aspetti Termodinamici e di Scambio Termico nel Funzionamento delle Macchine Frigorifere ad Assorbimento—Possibilità di Sviluppo di Macchine Innovative ad Acqua Ammoniaca. Ph.D. Thesis, Sapienza Università di Roma, Rome, Italy, 1995. [Google Scholar]
- Kang, Y.T.; Kunugi, Y.; Kashiwagi, T. Review of advanced absorption cycles: Performance improvement and temperature lift enhancement. Int. J. Refrig. 2000, 23, 388–401. [Google Scholar] [CrossRef]
- Toneatti, L.; Piller, M.; Pozzetto, D.; Padoano, E.; Boscolo, M. Hydraulic and thermal characterization of a family of thermo-hydraulic separators. Appl. Therm. Eng. 2020, 179, 115701. [Google Scholar] [CrossRef]
- Nasa Earth Observations—Sea Surface Temperature. 2021. Available online: https://earthobservatory.nasa.gov/global-maps/MYD28M (accessed on 28 June 2021).
- Winnes, H.; Fridell, E. Particle Emissions from Ships: Dependence on Fuel Type. J. Air Waste Manag. Assoc. 2012, 59, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Schumüller, K.; Weichgrebe, D.; Köster, S. Biogas potential of organic waste onboard cruise ships—A yet untapped energy source. Biomass Convers. Biorefinery 2021. [Google Scholar] [CrossRef]
- Pátek, J.; Klomfar, J. Simple functions for fast calculations of selected thermodynamic properties of the ammonia-water system. Int. J. Refrig. 1995, 18, 228–234. [Google Scholar] [CrossRef]
- Fernàndez-Seara, J.; Sieres, J. The importance of the ammonia purification process in ammonia-water absorption systems. Energy Convers. Manag. 2006, 47, 1975–1987. [Google Scholar] [CrossRef]
- Zavaleta-Aguilar, E.; Simoes Moreira, J. Thermal design of a tray-type distillation column of an ammonia/water absorption refrigeration cycle. Appl. Therm. Eng. 2012, 41, 52–60. [Google Scholar] [CrossRef]
- Clerx, M.; Trezek, G. Performance of an Aqua-Ammonia Absorption Solar Refrigerator at Sub-Freezing Evaporator Conditions. Sol. Energy 1987, 39, 379–389. [Google Scholar] [CrossRef]
- White, S.; O’Neill, B. Analysis of an improved aqua-ammonia absorption refrigeration cycle employing evaporator blowdown to provide rectifier reflux. Appl. Energy 1995, 50, 323–337. [Google Scholar] [CrossRef]

| Type of Waste | Total Mass Daily Production [kg/day] | Lower Heating Value [MJ/kg] | Total Recoverable Energy [MJ/day] |
|---|---|---|---|
| Plastic | 1188 | 36 | 42,768 |
| Paper and cardboard | 5360 | 14.3 | 76,707 |
| Food waste | 10,800 | 5.7 | 61,535 |
| Glass | 3672 | - | - |
| Aluminum | 108 | - | - |
| Total | 21,128 | - | 181,010 |
| Parameter | Value | Description | |
|---|---|---|---|
| 414 | kW | Cooling power exchanged to the evaporator | |
| 917 | kW | Thermal power required from the generator | |
| 1329 | kW | Thermal power rejected to the seawater | |
| 0.45 | Coefficient of performance | ||
| 22.7 | kg/s | Mass flow rate of the water-ethylene glycol solution | |
| 0.41 | kg/s | Mass flow rate of the ammonia | |
| 899 | kW | Thermal power exchanged by the absorber | |
| 0.237 | kg/kg | Titer of the poor concentration solution | |
| 0.318 | kg/kg | Titer of the rich concentration solution | |
| Parameter | Value | Description | ||
|---|---|---|---|---|
| Theoretical | Company | |||
| 414 | 414 | kW | Cooling power exchanged to the evaporator | |
| 917 | 880 | kW | Thermal power required from the generator | |
| 1329 | 1315 | kW | Thermal power to be dissipated in the sea | |
| 0.45 | 0.47 | Coefficient of performance | ||
| Utilization | Fuel Savings | CO Savings | CO Savings |
|---|---|---|---|
| Factor | [kg/h of Navigation] | [kg/day of Cruise] | [tonnes/year (305 Cruise Days)] |
| 25% | 10.25 | 348 | 106 |
| 50% | 20.50 | 695 | 212 |
| 75% | 30.75 | 1043 | 318 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toneatti, L.; Deluca, C.; Fraleoni Morgera, A.; Piller, M.; Pozzetto, D. Waste to Energy Onboard Cruise Ships: A New Paradigm for Sustainable Cruising. J. Mar. Sci. Eng. 2022, 10, 480. https://doi.org/10.3390/jmse10040480
Toneatti L, Deluca C, Fraleoni Morgera A, Piller M, Pozzetto D. Waste to Energy Onboard Cruise Ships: A New Paradigm for Sustainable Cruising. Journal of Marine Science and Engineering. 2022; 10(4):480. https://doi.org/10.3390/jmse10040480
Chicago/Turabian StyleToneatti, Luca, Claudio Deluca, Alessandro Fraleoni Morgera, Marzio Piller, and Dario Pozzetto. 2022. "Waste to Energy Onboard Cruise Ships: A New Paradigm for Sustainable Cruising" Journal of Marine Science and Engineering 10, no. 4: 480. https://doi.org/10.3390/jmse10040480
APA StyleToneatti, L., Deluca, C., Fraleoni Morgera, A., Piller, M., & Pozzetto, D. (2022). Waste to Energy Onboard Cruise Ships: A New Paradigm for Sustainable Cruising. Journal of Marine Science and Engineering, 10(4), 480. https://doi.org/10.3390/jmse10040480






