Protein Elicitor PeaT1 Efficiently Controlled Barley Yellow Dwarf Virus in Wheat
Abstract
:1. Introduction
2. Material and Method
2.1. Materials
2.2. Bioassay
2.3. Determination of Wheat Height and Chlorophyll Content
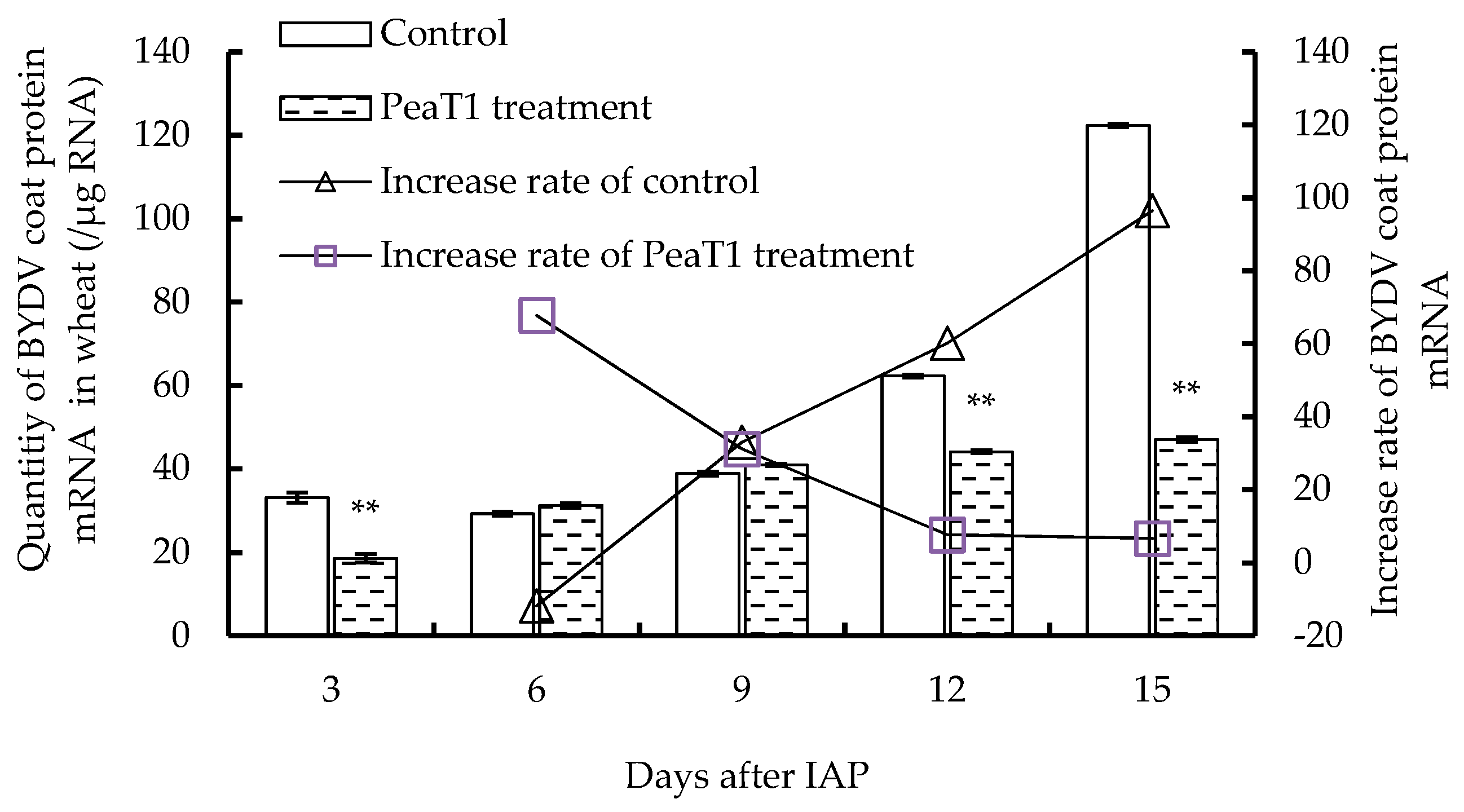
2.4. Quantity Determination of BYDV Coat Protein mRNA by Quantitative Real Time Polymerase Chain Reaction(Q-RT-PCR)
2.5. Detection of Apterous Viruliferous Schizaphis graminum Feeding Behavior
2.6. Data Analysis
3. Results
3.1. Control Efficiency of PeaT1 on BYDV Indoor
3.2. Effect of PeaT1 on BYDV Coat Protein mRNA
3.3. Effect of PeaT1 on Transmission Vector Schizaphis graminum Feeding Behavior
3.4. PeaT1 Weakened the Symptom of Wheat Caused by BYDV
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Domier, L.L.; D’Arcy, C.J. Luteoviruses. In Encyclopedia of Virology, 3rd ed.; Mahy, B.W.J., Van Regenmortel, M.H.V., Eds.; Academic Press: Oxford, UK, 2008; pp. 231–238. [Google Scholar]
- Harris, J. Chemical Pesticide Markets, Health Risks and Residues; CABI Publishing: Wallingford, UK, 2000. [Google Scholar]
- MacKenzie, T.D.B.; Arju, I.; Poirier, R.; Singh, M. A genetic survey of pyrethroid insecticide resistance in aphids in New Brunswick, Canada, with particular emphasis on aphids as vectors of potato virus Y. J. Econ. Entomol. 2018, 111, 1361–1368. [Google Scholar] [CrossRef]
- Pascher, K.; Gollmann, G. Ecological risk assessment of transgenic plant releases: An Austrian perspective. Biodivers. Conserv. 1999, 8, 1139–1158. [Google Scholar] [CrossRef]
- Mauch-Mani, B.; Baccelli, I.; Luna, E.; Flors, V. Defense priming: An adaptive part of induced resistance. Annu. Rev. Plant Biol. 2017, 68, 485–512. [Google Scholar] [CrossRef]
- Hael, C.V.; Perato, S.M.; Arias, M.E.; Martínez Zamora, M.G.; Di, P.P.; Martos, G.G.; Castagnaro, A.P.; Diaz-Ricci, J.C.; Chalfoun, N.R. The elicitor protein AsES induces a SAR response accompanied by systemic microbursts and micro-HRs in Fragaria ananassa. Mol. Plant-Microbe Interact. 2017, 31, 46–60. [Google Scholar] [CrossRef]
- Zhang, W.; Yang, X.; Qiu, D.; Guo, L.; Zeng, H.; Mao, J.; Gao, Q. PeaT1-induced systemic acquired resistance in tobacco follows salicylic acid-dependent pathway. Mol. Biol. Rep. 2011, 38, 2549–2556. [Google Scholar] [CrossRef]
- Silva, H.S.A.; Romeiro, R.S.; Carrer Filho, R.; Pereira, J.L.A.; Mizubuti, E.S.G.; Mounteer, A. Induction of systemic resistance by Bacillus cereus against tomato foliar diseases under field conditions. J. Phytopathol. 2004, 152, 371–375. [Google Scholar] [CrossRef]
- Kouzai, Y.; Kimura, M.; Watanabe, M.; Kusunoki, K.; Osaka, D.; Suzuki, T.; Matsui, H.; Yamamoto, M.; Ichinose, Y.; Toyoda, K.; et al. Salicylic acid-dependent immunity contributes to resistance against Rhizoctonia solani, a necrotrophic fungal agent of sheath blight, in rice and Brachypodium distachyon. New Phytol. 2018, 217, 771–783. [Google Scholar] [CrossRef]
- Sanches, P.A.; Santos, F.; Penaflor, M.; Bento, J.M.S. Direct and indirect resistance of sugarcane to Diatraea saccharalis induced by jasmonic acid. Bull. Entomol. Res. 2017, 107, 828–838. [Google Scholar] [CrossRef]
- Wang, N.; Liu, M.; Guo, L.; Yang, X.; Qiu, D. A novel protein elicitor (PeBA1) from Bacillus amyloliquefaciens NC6 induces systemic resistance in tobacco. Int. J. Biol. Sci. 2016, 12, 757–767. [Google Scholar] [CrossRef]
- Dodds, P.N.; Rathjen, J.P. Plant immunity: Towards an integrated view of plant-pathogen interactions. Nat. Rev. Genet. 2010, 11, 539–548. [Google Scholar] [CrossRef]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef] [Green Version]
- Kulye, M.; Liu, H.; Zhang, Y.; Zeng, H.; Yang, X.; Qiu, D. Hrip1, a novel protein elicitor from necrotrophic fungus, Alternaria tenuissima, elicits cell death, expression of defence-related genes and systemic acquired resistance in tobacco. Plant Cell Environ. 2012, 35, 2104–2120. [Google Scholar] [CrossRef]
- Li, L.; Wang, S.; Yang, X.; Francis, F.; Qiu, D. Protein elicitor PeaT1 enchanced resistance against aphid (Sitobion avenae) in wheat. Pest Manag. Sci. 2019. [Google Scholar] [CrossRef]
- Peng, X.C.; Qiu, D.W.; Zeng, H.M.; Guo, L.H.; Yang, X.F.; Liu, Z. Inducible and constitutive expression of an elicitor gene Hrip1 from Alternaria tenuissima enhances stress tolerance in Arabidopsis. Transgenic Res. 2015, 24, 135–145. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar]
- Wang, Y.; Zhou, K.; Wang, X.; Zhou, G. Studies on the movement of Barley yellow dwarf virus-GAV in oat plant by RT-PCR (in Chinese). Acta Phytopathol. Sin. 2009, 39, 249–253. [Google Scholar]
- Li, L.; Zhao, C.; Li, H.; Li, W.; Zhang, L.; Xu, D.; Wang, J.; Li, H. Establishment of the plasmid standard curve generation method for absolute quantification PCR (in Chinese). J. Agric. Biotechnol. 2011, 19, 1157–1162. [Google Scholar]
- Zhang, Y.; Fan, J.; Francis, F.; Chen, J. Watery saliva secreted by the grain aphid Sitobion avenae stimulates aphid resistance in wheat. J. Agric. Food Chem. 2017, 65, 8798–8805. [Google Scholar] [CrossRef]
- Tjallingii, W.F.; Esch, T.H. Fine structure of aphid stylet routes in plant tissues in correlation with EPG signals. Physiol. Entomol. 1993, 18, 317–328. [Google Scholar] [CrossRef]
- Sarria, E.; Cid, M.; Garzo, E.; Fereres, A. Excel workbook for automatic parameter calculation of EPG data. Comput. Electron. Agric. 2009, 67, 35–42. [Google Scholar] [CrossRef]
- Ma, Z.; Li, W.; Wang, H.; Zhang, X. Induced resistance to tobacco mosaic virus (TMV) on tobacco by botanical-derived VFB. Acta Phytopathol. Sin. 2010, 40, 419–425. [Google Scholar]
- Wang, B.; Wang, S.; Tan, B.; Qiu, D.; Yang, X. Systemic acquired resistance to tobacco mosaic virus (TMV) induced by protein elicitor from Vericillium dahliae (PevD1) and its mechanisms in tobacco. J. Agric. Biotechnol. 2012, 20, 188–195. [Google Scholar]
- Shi, F.; Dong, Y.; Zhang, Y.; Yang, X.; Qiu, D. Overexpression of the PeaT1 elicitor gene from Alternaria tenuissima improves drought tolerance in rice plants via interaction with a Myo-inositol oxygenase. Front. Plant Sci. 2017, 8, 970. [Google Scholar] [CrossRef]
- Gray, S.; Gildow, F.E. Luteovirus-aphid interactions. Annu. Rev. Phytopathol. 2003, 41, 539–566. [Google Scholar] [CrossRef]
- Prado, E.; Tjallingii, W.F. Aphid activities during sieve element punctures. Entomol. Exp. Appl. 1994, 72, 157–165. [Google Scholar] [CrossRef]
- Givovich, A.; Niemeyer, H.M. Hydroxamic acids affecting barley yellow dwarf virus transmission by the aphid Rhopalosiphum padi. Entomol. Exp. Appl. 1991, 59, 79–85. [Google Scholar] [CrossRef]
- Rong, W.; Wang, X.; Wang, X.; Massart, S.; Zhang, Z. Molecular and Ultrastructural Mechanisms Underlying Yellow Dwarf Symptom Formation in Wheat after Infection of Barley Yellow Dwarf Virus. Int. J. Mol. Sci. 2018, 19, 1187. [Google Scholar] [CrossRef]
- Sheng, S.; Zhou, Q.; Qiu, D.; Yang, X. Effects and mechanism of disease resistance and yield improvement induced by plant immune protein preparation Ataling in wheat. Chin. J. Biol. Control 2017, 33, 213–218. [Google Scholar]

| Disease Grade | Grading Standard |
|---|---|
| 0 | Health leaf without chlorosis |
| 1 | Top end of leaf is chlorosis |
| 2 | The margin of 1–2 cm top of leaf is light yellow |
| 3 | 1–2 cm top of leaf is yellow |
| 4 | 1/4 of leaf is yellow, other part is light chlorosis |
| 5 | 1/3 of leaf is yellow, other part is light chlorosis |
| 6 | Top end of leaf is dried up, 1/2 of leaf is yellow, other part is light chlorosis |
| 7 | Top end of leaf is dried up, over 1/2 of leaf is yellow, other part is light chlorosis |
| 8 | Top end of leaf is dried up, whole leaf is chlorosis or yellow nonuniformly |
| 9 | Top end of leaf is dried up, whole leaf is yellow |
| 10 | whole leaf is yellow or dried up |
| Time after IAP (Day) | Disease Index | Control Efficiency (%) | |
|---|---|---|---|
| Control | PeaT1 Treatment | ||
| 14 | 27.47 ± 2.22 | 17.92 ± 1.44 ** | 35.34 ± 2.20 |
| 21 | 38.93 ± 0.70 | 24.58 ± 3.45 ** | 36.77 ± 9.59 |
| Parameters of EPG | Control (n = 22) | PeaT1 Treatment (n = 22) | p Value |
|---|---|---|---|
| Time to 1st probe from start of EPG (min) | 4.20 ± 0.64 | 7.25 ± 0.87 * | 0.04 |
| Duration of nonprobe period before the 1st E1 (min) | 18.11 ± 2.22 | 27.69 ± 3.94 * | 0.05 |
| Time from 1st probe to 1st E1 (h) | 1.80 ± 0.14 | 2.50 ± 0.28 * | 0.04 |
| Time from start of EPG to 1st E1 (h) | 1.86 ± 0.14 | 2.53 ± 0.28 * | 0.05 |
| Mean duration of E1 (min) | 2.28 ± 0.28 | 2.90 ± 0.49 | 0.32 |
| Time after IAP (Day) | Content of Chlorophyll a&b (μg/g × FW) | Wheat Height (cm) | ||
|---|---|---|---|---|
| Control | PeaT1 Treatment | Control | PeaT1 Treatment | |
| 14 | 144.29 ± 4.58 | 194.23 ± 7.05 ** | 26.88 ± 1.3 | 33.35 ± 1.56 ** |
| 21 | 106.06 ± 7.56 | 172.64 ± 5.29 ** | 27.59 ± 1.82 | 33.41 ± 2.54 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Wang, S.; Yang, X.; Francis, F.; Qiu, D. Protein Elicitor PeaT1 Efficiently Controlled Barley Yellow Dwarf Virus in Wheat. Agriculture 2019, 9, 193. https://doi.org/10.3390/agriculture9090193
Li L, Wang S, Yang X, Francis F, Qiu D. Protein Elicitor PeaT1 Efficiently Controlled Barley Yellow Dwarf Virus in Wheat. Agriculture. 2019; 9(9):193. https://doi.org/10.3390/agriculture9090193
Chicago/Turabian StyleLi, Lin, Shuangchao Wang, Xiufen Yang, Frederic Francis, and Dewen Qiu. 2019. "Protein Elicitor PeaT1 Efficiently Controlled Barley Yellow Dwarf Virus in Wheat" Agriculture 9, no. 9: 193. https://doi.org/10.3390/agriculture9090193







