Aflatoxins in Mozambique: Etiology, Epidemiology and Control
Abstract
:1. Introduction
2. Major Aflatoxin Studies in Mozambique
3. Etiology and Commodities
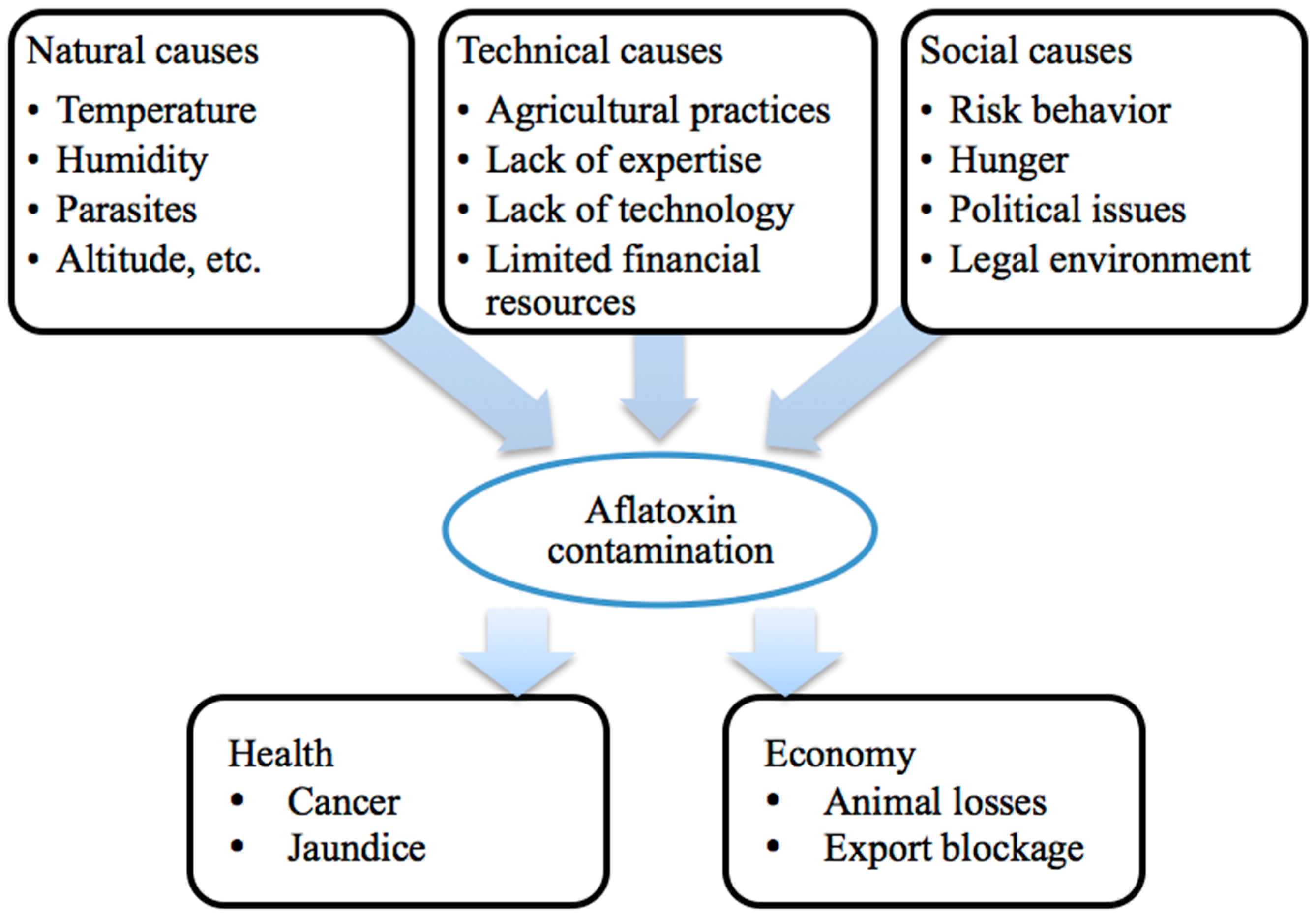
3.1. Etiology
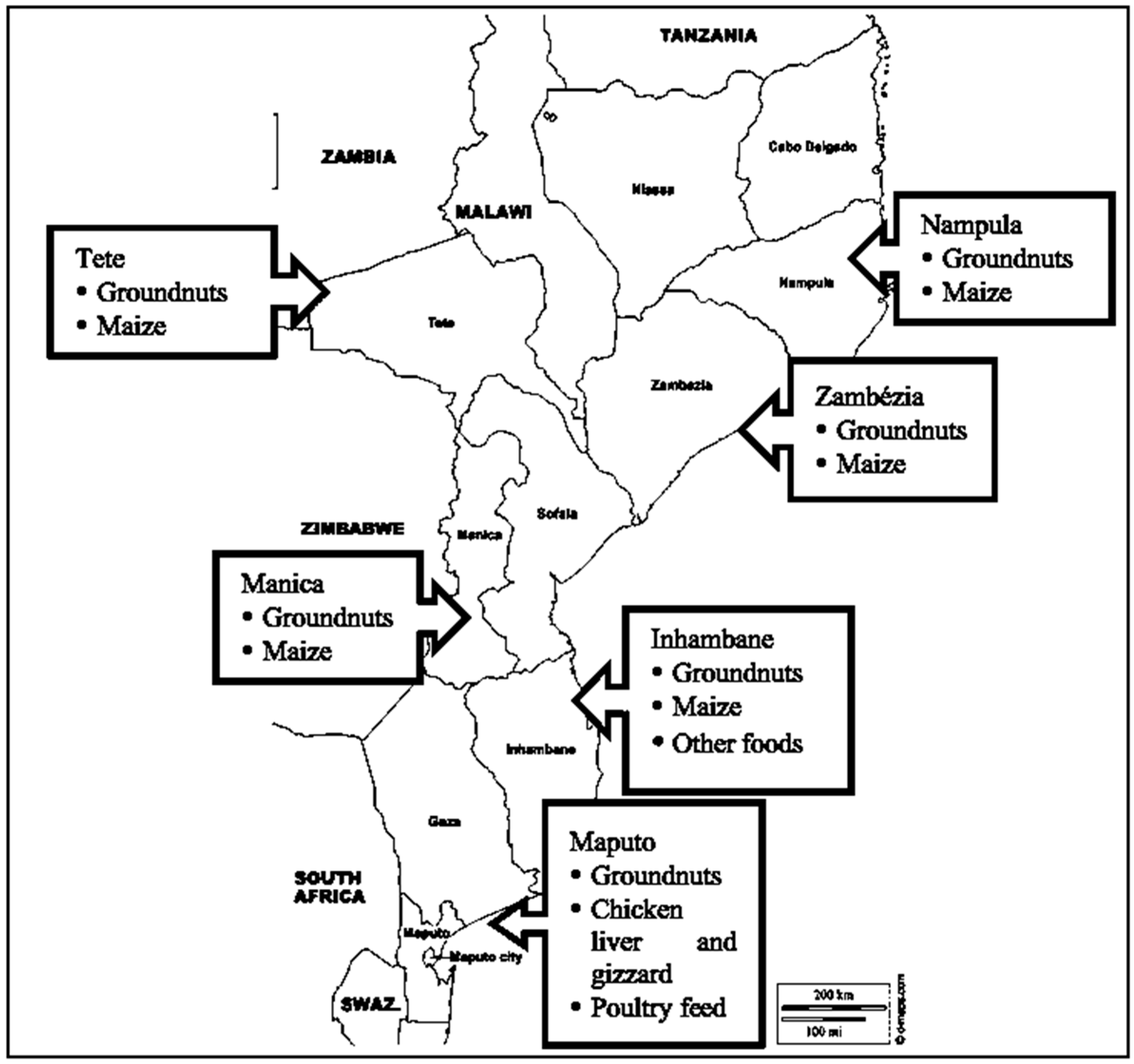
3.2. Commodities
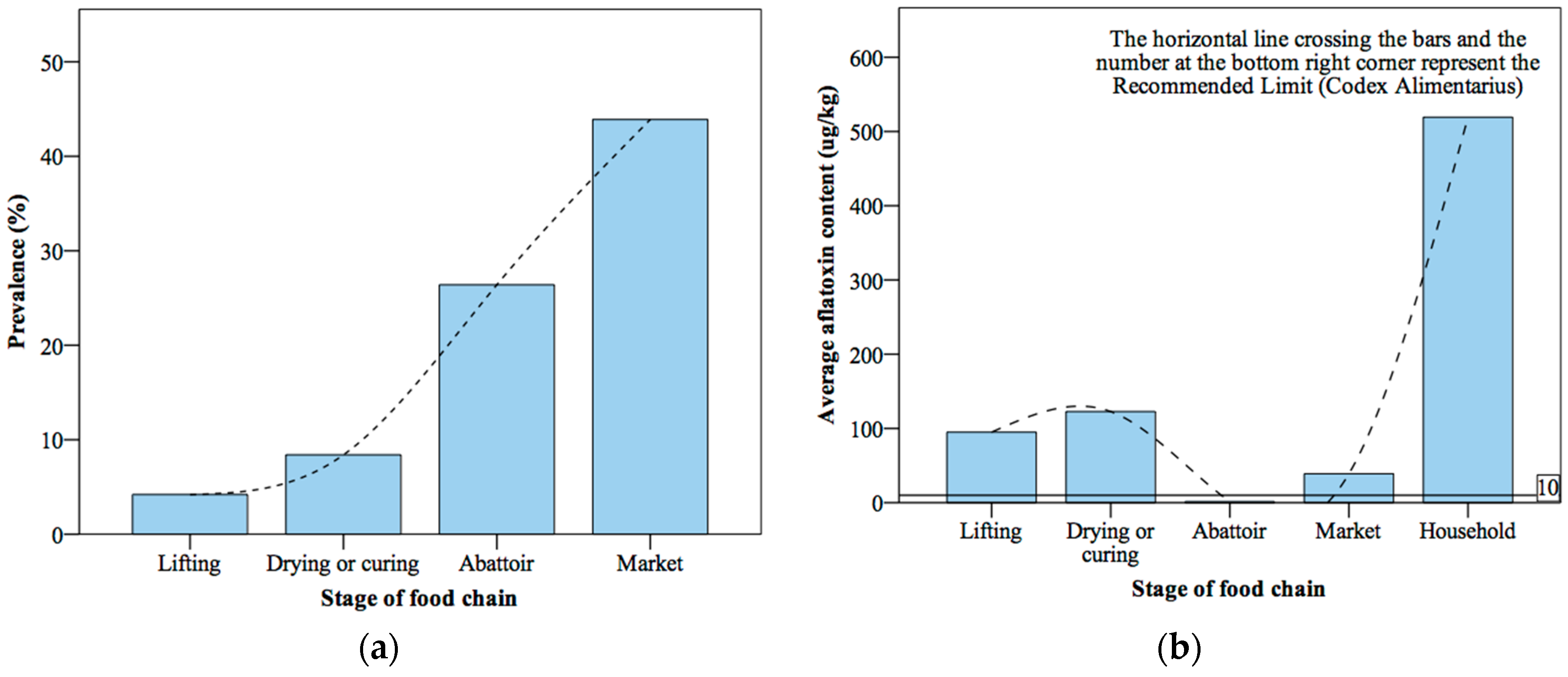
3.3. Circumstances of Contamination
4. Geographic Distribution of Aflatoxins and Related Diseases
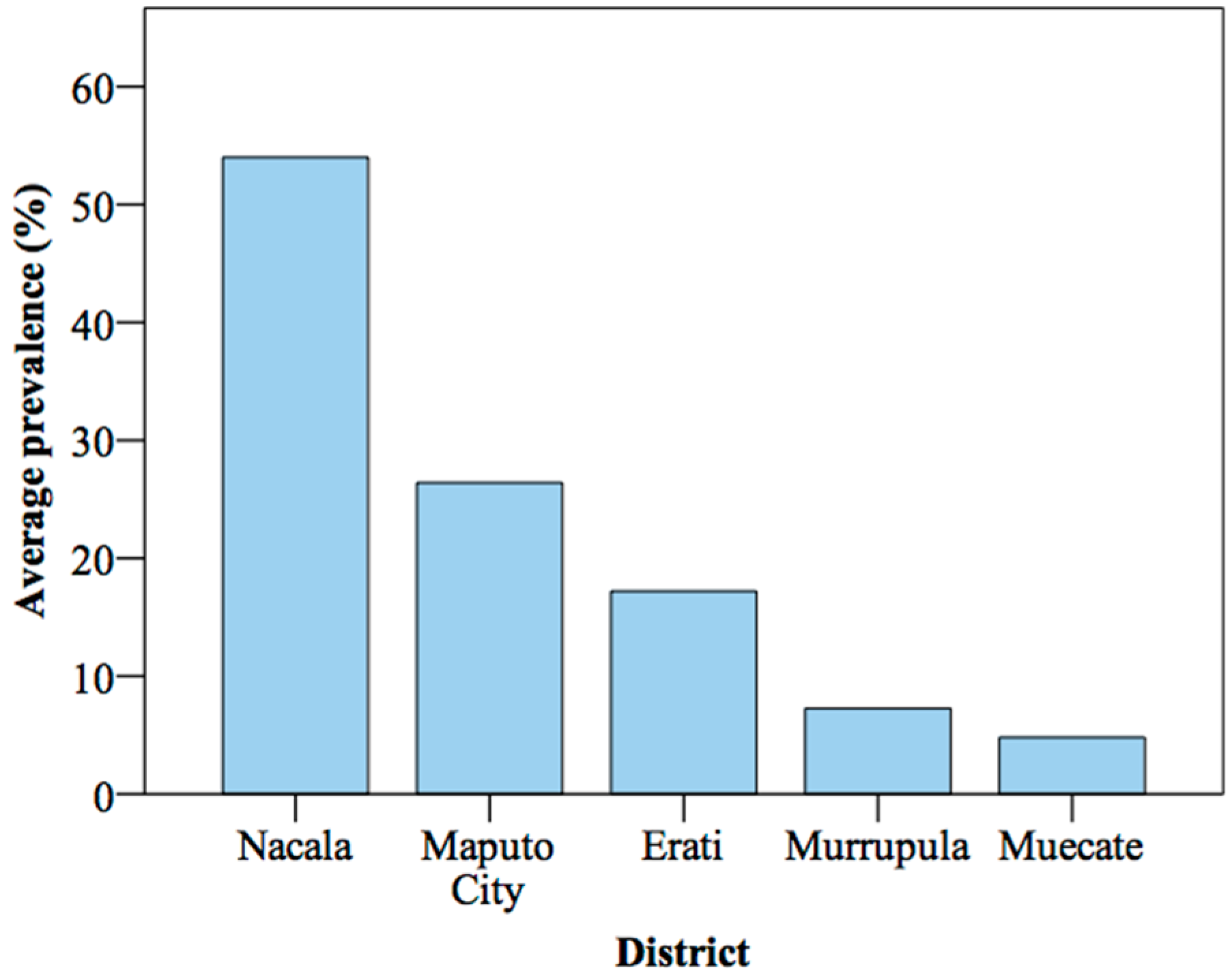
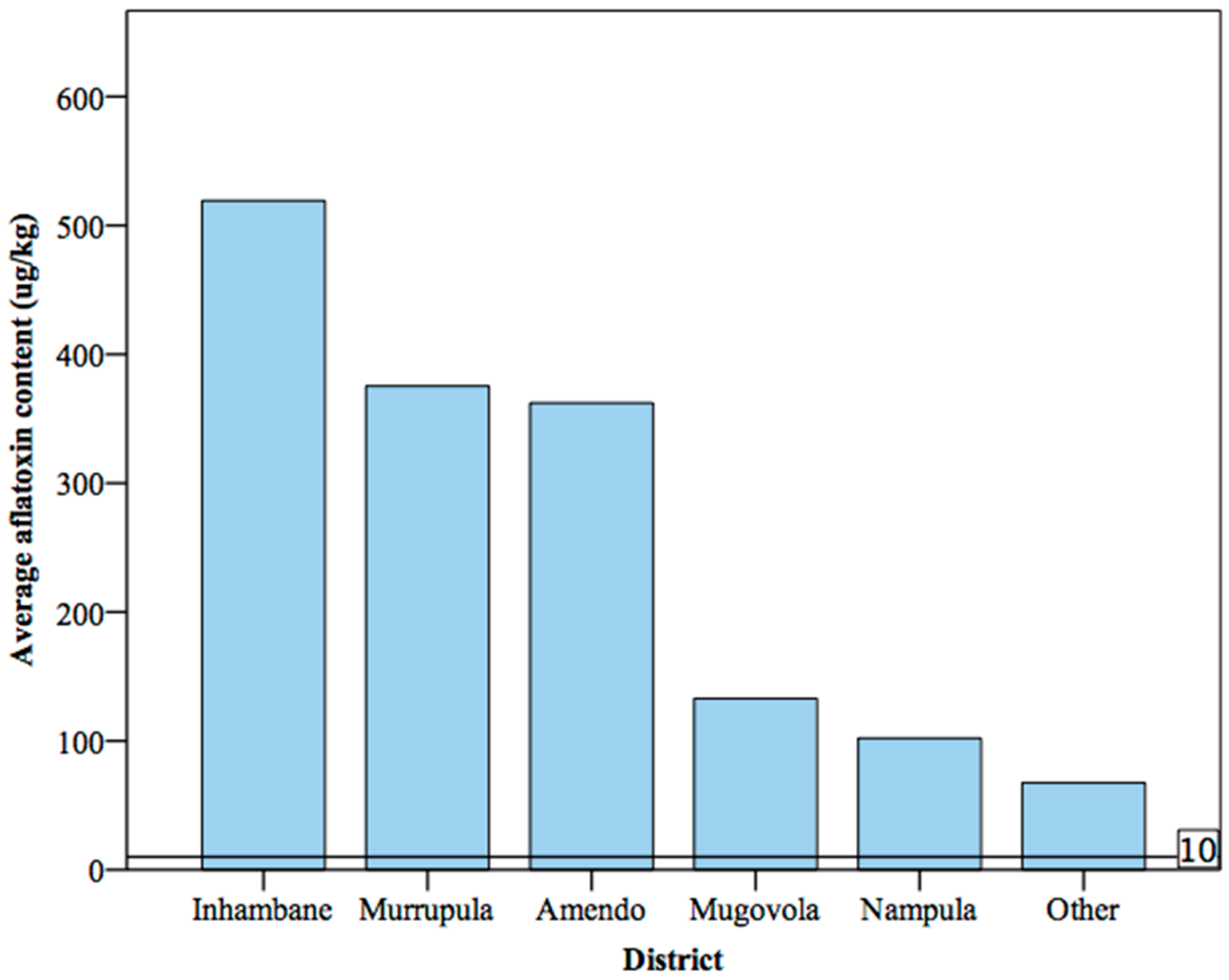
4.1. Geographic Distribution
4.2. Susceptible Groups
5. Detection, Prevention and Control
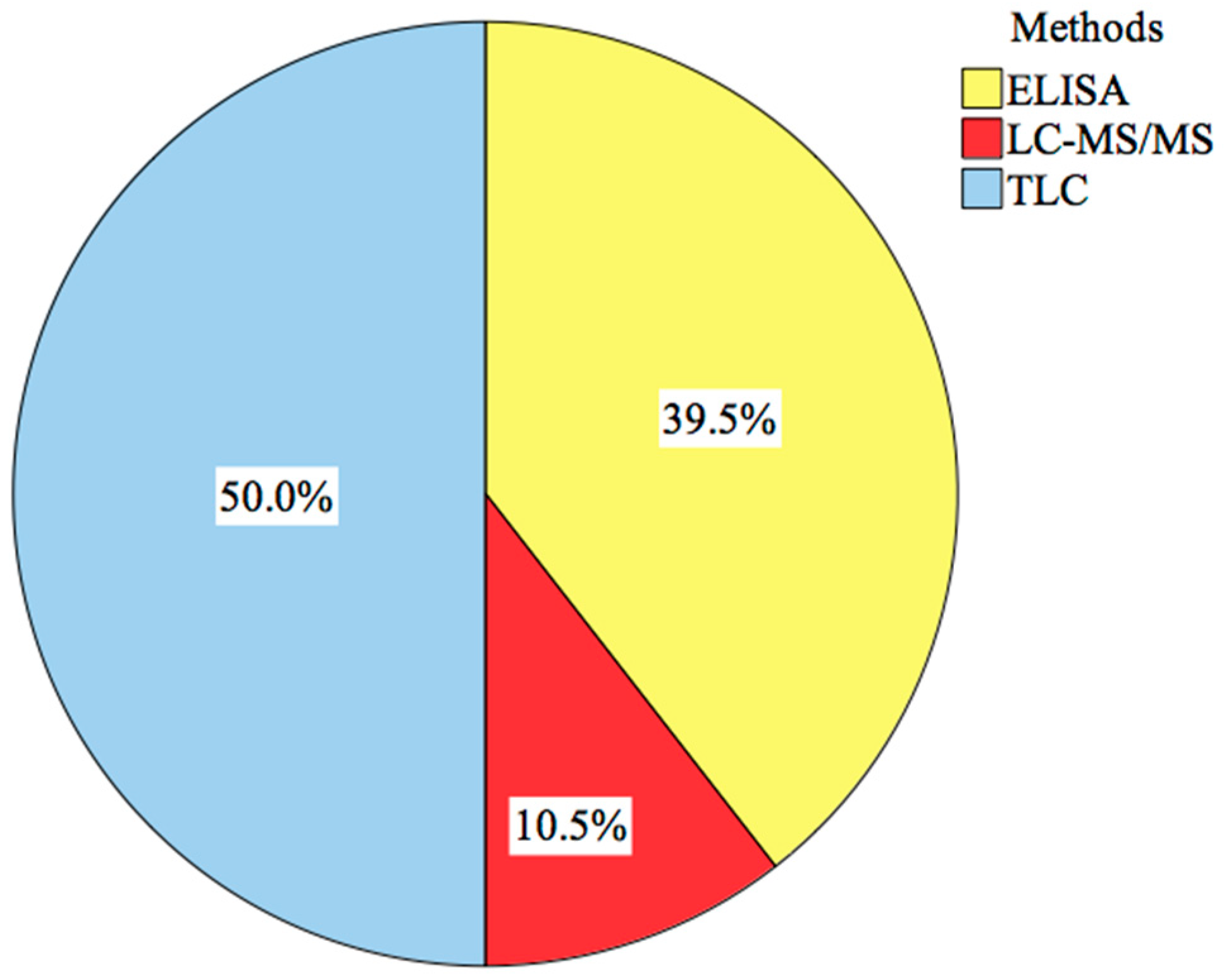
5.1. Detection Methods
5.2. Prevention
5.3. Control
6. Recommendations
Funding
Conflicts of Interest
References
- Van Wyk, P.; Van der Merwe, P.; Subrahmanyam, P.; Boughton, D. Aflatoxin contamination of groundnuts in mozambique. Int. Arachis Newsl. 1999, 19, 25–27. [Google Scholar]
- Warth, B.; Parich, A.; Atehnkeng, J.; Bandyopadhyay, R.; Schuhmacher, R.; Sulyok, M.; Krska, R. Quantitation of mycotoxins in food and feed from burkina faso and mozambique using a modern lc-ms/ms multitoxin method. J. Agric. Food Chem. 2012, 60, 9352–9363. [Google Scholar] [CrossRef] [PubMed]
- Ferrão, J.; Bell, V.; Fernandes, T.H. Mycotoxins, food safety and security in sub-saharan africa. SM J. Food Nutr. Disord. 2017, 3, 1–9. [Google Scholar]
- Harmsen, J.; Bremmer, J.; Maria, R.M. Pre Evaluation of a Soil and Plant Laboratory in Mozambique; Alterra: Wageningen, The Netherlands, 2012; p. 60. [Google Scholar]
- Zuza, E.; Mondjana, A.; Muitia, A.; Amane, M. Effects of harvesting date on aflatoxin contamination in groundnuts in northern mozambique. In The Fifth RUFORUM Biennial Conference and African Higher Education Week; Nampala, M.P., Egeru, A., Tusiime, G., Osiru, M., Mensah, S., Adipala, E., Eds.; Regional Universities Forum for Capacity Building in Agriculture (RUFORUM): Cape Town, South Africa, 2016; Volume 14, pp. 167–172. [Google Scholar]
- Casadei, E. Os contaminantes nos alimentos. In Mocambique: Águas, Alimentos e Ambiente; Molisv: Rome, Italy, 1980. [Google Scholar]
- Van Rensburg, S.J.; Cook-Mozaffari, P.; Van Schalkwyk, D.J.; Van der Watt, J.J.; Vincent, T.J.; Purchase, I.F. Hepatocellular carcinoma and dietary aflatoxin in mozambique and transkei. Br. J. Cancer 1985, 51, 713–726. [Google Scholar] [CrossRef] [PubMed]
- Sineque, A.R.; Macuamule, C.L.; Dos Anjos, F.R. Aflatoxin b1 contamination in chicken livers and gizzards from industrial and small abattoirs, measured by elisa technique in maputo, mozambique. Int. J. Environ. Res. Public Health 2017, 14, 951. [Google Scholar] [CrossRef] [PubMed]
- Codex Alimentarius Commission. Codex General Standard for Contaminants and Toxins in Food and Feed; Codex Stan 193, 1995; Codex Alimentarius Commission: Omaha, NE, USA, 2013. [Google Scholar]
- Augusto, J.; Atehnkeng, J.; Akello, J.; Cotty, P.; Bandyopadhyay, R. Prevalence and distribution of aspergillus section flavi in maize and groundnut fields and aflatoxin contamination in mozambique. In Proceedings of the 2014 APS-CPS Joint Meeting, Minneapolis, MN, USA, 9–13 August 2014; The American Phytopathological Society: Minneapolis, MN, USA, 2014. [Google Scholar]
- Baquete, E.; Freire, M. Present Status and Perspectives of Aflatoxin Research in Mozambique. Aflatoxin Contam. Groundn. 1989, pp. 93–94. Available online: http://oar.icrisat.org/502/1/RA_00132.pdf#page=263 (accessed on 10 May 2005).
- Abbas, H.; Zablotowicz, R.; Locke, M. Spatial variability of aspergillus flavus soil populations under different crops and corn grain colonization and aflatoxins. Can. J. Bot. 2004, 82, 1768–1775. [Google Scholar] [CrossRef]
- Andrade, P.D.; Caldas, E.D. Aflatoxins in cereals: Worldwide occurrence and dietary risk assessment. World Mycotoxin J. 2015, 8, 415–431. [Google Scholar] [CrossRef]
- Liu, Z.; Gao, J.; Yu, J. Aflatoxins in stored maize and rice grains in liaoning province, China. J. Stored Prod. Res. 2006, 42, 468–479. [Google Scholar] [CrossRef]
- Mota, T.P.; Lourenço, M.C. A farinha de mandioca de moçambique. Agronomia Mocambicana 1974, 8, 47–59. [Google Scholar]
- Essers, A.A.; Nout, M.R. The safety of dark, moulded cassava flour compared with white-a comparison of traditionally dried cassava pieces in north-east mozambique. J. Food Technol. Afr. 2000, 5, 19–21. [Google Scholar] [CrossRef]
- Bandyopadhyay, R.; Dubois, T. Aflatoxin control projects launched in southern Africa. In IIAPPS Newsletter; 2012; pp. 2–4. [Google Scholar]
- Mussagy, I.H.; Babatunde, M.A. Government Spending on Education and Economic Growth in Mozambique: A Cointegration Approach. Revista Electrónica de Investigação e Desenvolvimento. 2015, pp. 1–18. Available online: http://reid.ucm.ac.mz/index.php/reid/article/view/68/84 (accessed on 12 May 2005).
- Chiona, M.; Ntawuruhunga, P.; Benesi, I.; Matumba, L.; Moyo, C. Aflatoxins contamination in processed cassava in malawi and zambia. Afr. J. Food Agric. Nutr. Dev. 2014, 14, 3. [Google Scholar]
- Mondlane, I.A.P.; Capece, B.P.S.; Parruque, A.F. Relação Entre a Ocorrência de Fungos e a Presença de Aflatoxinas b1 em Rações para aves Fabricadas em Maputo; Instituto de Investigação Agrária de Moçambique: Maputo, Mozambique, 2005. [Google Scholar]
- Walker, T.; Pitoro, R.; Tomo, A.; Sitoe, I.; Salência, C.; Mahanzule, R.; Donovan, C.; Mazuze, F. Priority Setting for Public-Sector Agricultural Research in Mozambique with the National Agricultural Survey Data; Directorate of Training, Documentation, and Technology Transfer, Institute of Agricultural Research of Mozambique: Maputo, Mozambique, 2006; p. 80. [Google Scholar]
- Anjos, F.D.; Ledoux, D.; Rottinghaus, G.; Chimonyo, M. Efficacy of mozambican bentonite and diatomaceous earth in reducing the toxic effects of aflatoxins in chicks. World Mycotoxin J. 2016, 9, 63–72. [Google Scholar] [CrossRef]
- Carlson, M.P.; Ensley, S.M. Sampling and Analyzing Feed for Fungal (Mold) Toxins (Mycotoxins); Cooperative Extension, Institute of Agriculture and Natural Resources, University of Nebraska-Lincoln: Lincoln, NE, USA, 2003. [Google Scholar]
- Guo, B.; Sobolev, V.; Holbrook, C.; Lynch, R. Impact of phytoalexins and lesser cornstalk borer damage on resistance to aflatoxin contamination. In American Peanut Research and Education Society Proceedings; American Peanut Research and Education Society: Tifton, GA, USA, 2004. [Google Scholar]
- Sachs, J.D.; McArthur, J.W. The millennium project: A plan for meeting the millennium development goals. Lancet 2005, 365, 347–353. [Google Scholar] [CrossRef]
- Griggs, D.; Stafford-Smith, M.; Gaffney, O.; Rockström, J.; Öhman, M.C.; Shyamsundar, P.; Steffen, W.; Glaser, G.; Kanie, N.; Noble, I. Policy: Sustainable development goals for people and planet. Nature 2013, 495, 305. [Google Scholar] [CrossRef] [PubMed]
- Stevens, A.; Saunders, C.; Spence, J.; Newham, A. Investigations into “diseases” of turkey poults. Vet. Rec. 1960, 72, 627–628. [Google Scholar]
- Ozturk, M. P53 mutation in hepatocellular carcinoma after aflatoxin exposure. Lancet 1991, 338, 1356–1359. [Google Scholar] [PubMed]
- Strosnider, H.; Azziz-Baumgartner, E.; Banziger, M.; Bhat, R.V.; Breiman, R.; Brune, M.N.; DeCock, K.; Dilley, A.; Groopman, J.; Hell, K.; et al. Workgroup report: Public health strategies for reducing aflatoxin exposure in developing countries. Environ. Health Perspect. 2006, 114, 1898–1903. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). Aflatoxins. In IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2002; Volume 82, pp. 171–300. [Google Scholar]
- D-Maps. Mozambique/República de Moçambique: Boundaries, Provinces, Names (White). Available online: http://d-maps.com/carte.php?num_car=3537&lang=en (accessed on 15 May 2018).
- Mendonça, I.N.D. Urban mobility in maputo metropolitan area: Analysis of the management bodies of urban planning and mobility, institutional arrangements and inputs for effective articulation. J. Transp. Lit. 2014, 8, 244–270. [Google Scholar] [CrossRef]
- Arias, F.J.; Libombo, M. Groundnut evaluation in mozambique: Preliminary results for the 1993/94 season in maputo province. In Sustainable Groundnut Production in Southern and Eastern Africa, Proceedings of a Workshop, Mbabane, Swaziland, 5–7 July 1994; Ndunguru, B.J., Hildebrand, G.L., Subrahmanyam, P., Eds.; International Crops Research Institute for the Semi-Arid Tropics: Mbabane, Swaziland, 1994. [Google Scholar]
- Shephard, G.S. Risk assessment of aflatoxins in food in africa. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2008, 25, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Van Rensburg, S.J.; Kirsipuu, A.; Coutinho, L.P.; Van Der Watt, J.J. Circumstances associated with the contamination of food by aflatoxin in a high primary liver cancer area. S. Afr. Med. J. 1975, 49, 877–883. [Google Scholar] [PubMed]
- Linsell, C.A.; Peers, F.G. Aflatoxin and liver cell cancer. Trans. R. Soc. Trop. Med. Hyg. 1977, 71, 471–473. [Google Scholar] [CrossRef]
- Kaaya, A.N.; Warren, H.L.; Kyamanywa, S.; Kyamuhangire, W. The effect of delayed harvest on moisture content, insect damage, moulds and aflatoxin contamination of maize in mayuge district of uganda. J. Sci. Food Agric. 2005, 85, 2595–2599. [Google Scholar] [CrossRef]
- Jones, R.; Duncan, H.; Hamilton, P. Planting date, harvest date, and irrigation effects on infection and aflatoxin production by aspergillus flavus in field corn. Development 1981, 19, 32. [Google Scholar]
- Bulaong, S.S. Fungal population, aflatoxin and free fatty acid contents of peanuts packed in different bag types. BIOTROPIA Southeast Asian J. Trop. Biol. 2002. [Google Scholar] [CrossRef]
- Essono, G.; Ayodele, M.; Akoa, A.; Foko, J.; Filtenborg, O.; Olembo, S. Aflatoxin-producing aspergillus spp. And aflatoxin levels in stored cassava chips as affected by processing practices. Food Control 2009, 20, 648–654. [Google Scholar] [CrossRef]
- Cardwell, K.; Cotty, P.; Bandyopadhyay, R.; Mutegi, C.; Nelson, F.; Marwa, M.; Manyong, V. Biocontrol for Aflatoxin; International Institute for Tropical Agriculture (IITA): Dar es Salaam, Tanzania, 2015; p. 50. [Google Scholar]

| Author | Year | Product | Sample Size (n) | Prevalence (%) | Average (µg/kg) | Median (µg/kg) |
|---|---|---|---|---|---|---|
| Casadei, E. [6] | 1980 | Beans | 54 | 17.0 | - | 6.0 |
| Beer | 15 | 93.0 | 1.1 | 1.0 | ||
| Cassava flour | 12 | 25.0 | 28.1 | 40.0 | ||
| Corn | 32 | 87.0 | 22.6 | 17.0 | ||
| Corn flour | 31 | 52.0 | 18.3 | 10.0 | ||
| Dry cassava | 10 | 10.0 | - | - | ||
| Forage | 25 | 32.0 | - | 50.0 | ||
| Groundnut | 37 | 92.0 | - | 49.0 | ||
| Maize | 18 | 67.0 | 22.0 | 21.0 | ||
| Rice | 56 | 63.0 | 16.2 | 11.0 | ||
| Sesame | 6 | 33.0 | - | - | ||
| Sorghum | 11 | 9.0 | 6.5 | 5.0 | ||
| Wheat | 13 | 22.0 | 8.5 | 9.0 | ||
| Van Rensburg, S.J. et al. [7] | 1985 | Groundnut | 153 | - | 1036.0 | - |
| Maize | 168 | - | 2.4 | - | ||
| Van Wyk, P. et al. [1] | 1999 | Groundnut | 4 | 8.3 | - | 92.6 |
| Warth, B. et al. [2] | 2012 | Feed waste | 1 | - | - | 433.0 |
| Groundnut | 23 | - | - | 3.4 | ||
| Maize | 13 | - | - | 69.9 | ||
| Millet | 2 | - | - | 4.0 | ||
| Sineque, A.R. et al. [8] | 2017 | Chicken gizzard | 80 | 13.8 | 1.1 | - |
| Chicken liver | 100 | 39.0 | 1.7 | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cambaza, E.; Koseki, S.; Kawamura, S. Aflatoxins in Mozambique: Etiology, Epidemiology and Control. Agriculture 2018, 8, 87. https://doi.org/10.3390/agriculture8070087
Cambaza E, Koseki S, Kawamura S. Aflatoxins in Mozambique: Etiology, Epidemiology and Control. Agriculture. 2018; 8(7):87. https://doi.org/10.3390/agriculture8070087
Chicago/Turabian StyleCambaza, Edgar, Shigenobu Koseki, and Shuso Kawamura. 2018. "Aflatoxins in Mozambique: Etiology, Epidemiology and Control" Agriculture 8, no. 7: 87. https://doi.org/10.3390/agriculture8070087
APA StyleCambaza, E., Koseki, S., & Kawamura, S. (2018). Aflatoxins in Mozambique: Etiology, Epidemiology and Control. Agriculture, 8(7), 87. https://doi.org/10.3390/agriculture8070087






