A Discussion on Mehlich-3 Phosphorus Extraction from the Perspective of Governing Chemical Reactions and Phases: Impact of Soil pH
Abstract
1. Introduction
2. Materials and Methods
2.1. Laboratory Analysis
2.2. Statistical Analysis
3. Results and Discussion
3.1. General Soil Characteristics
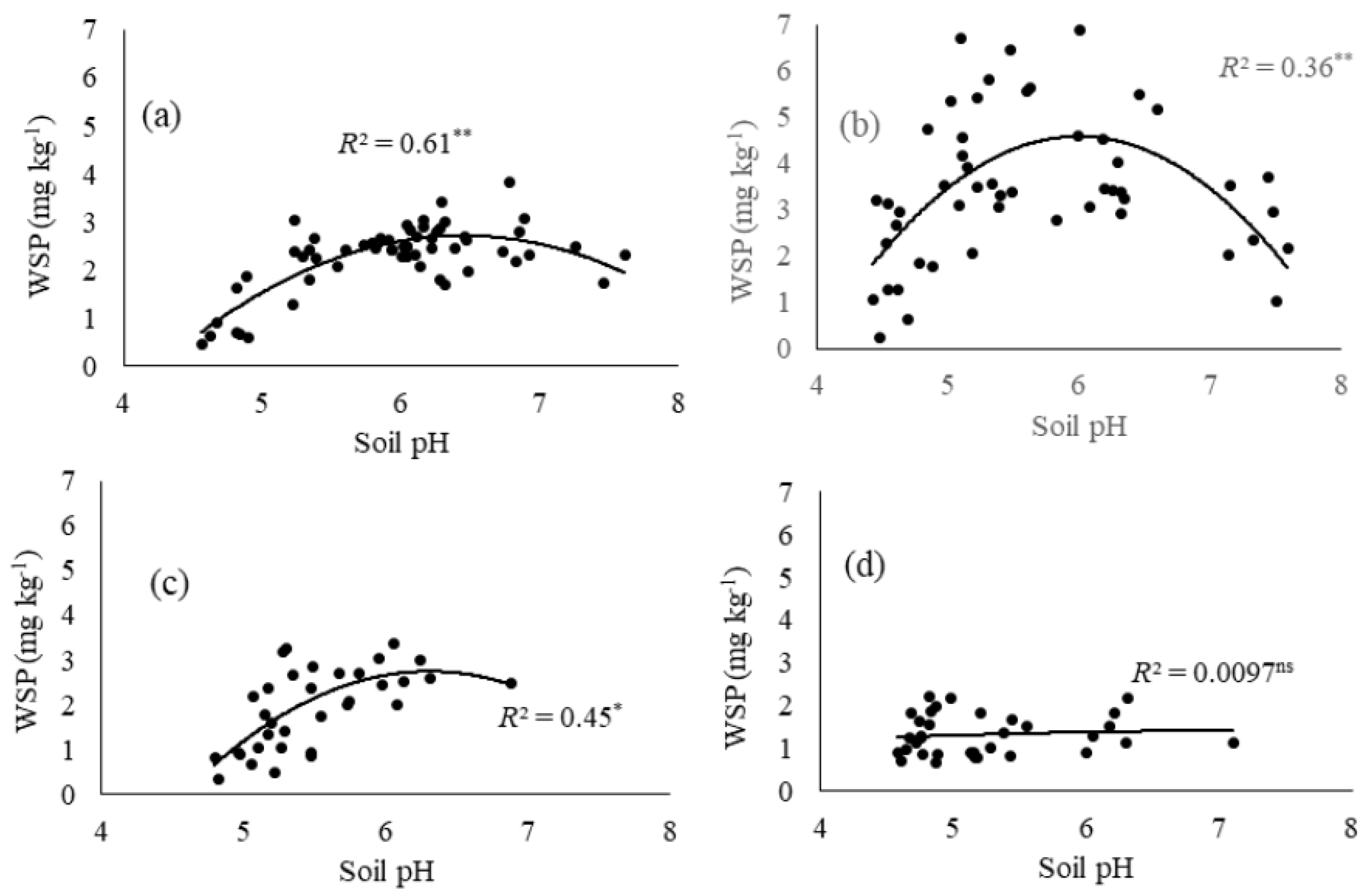
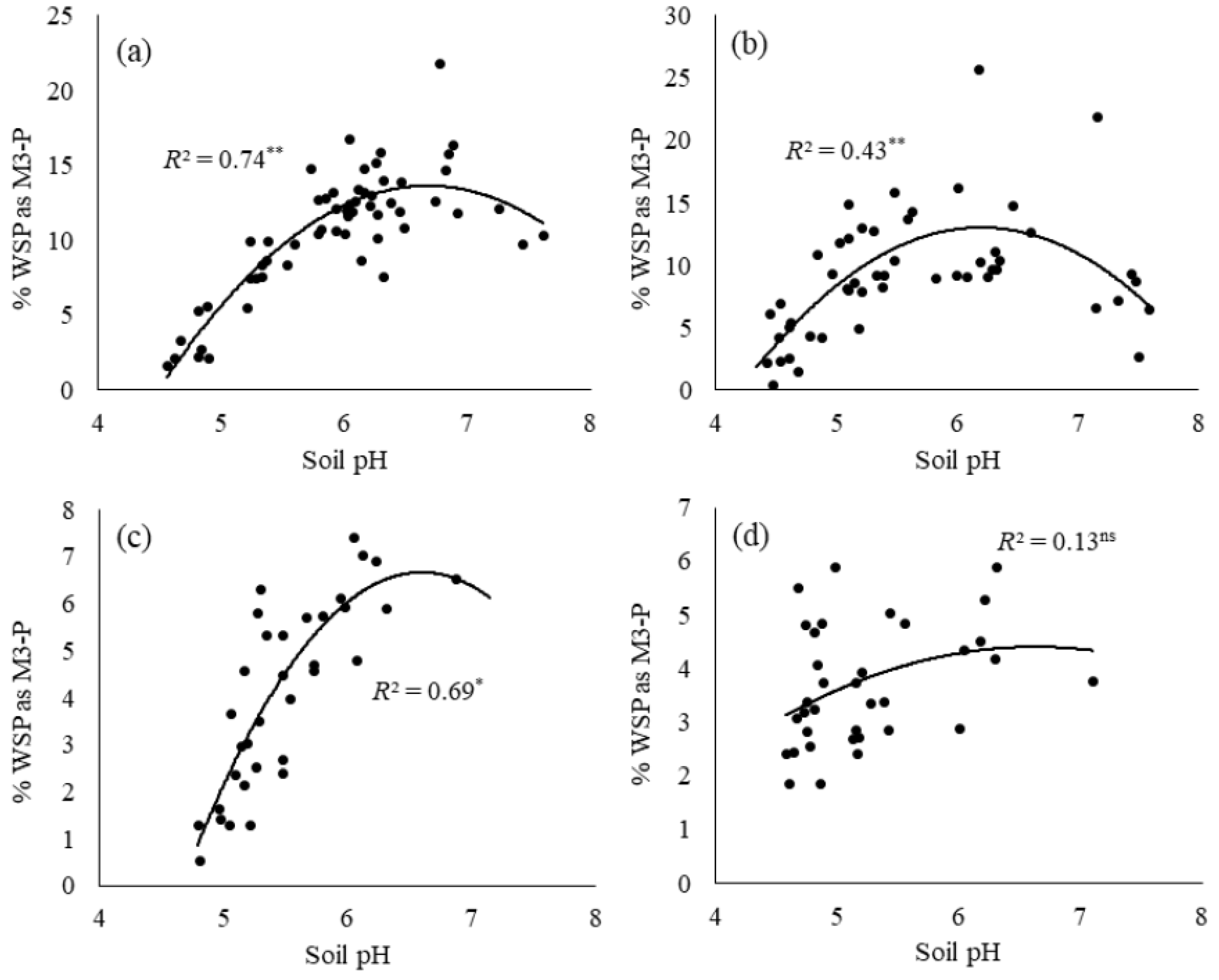
3.2. Impact of Soil pH on Water-Soluble Phosphorus
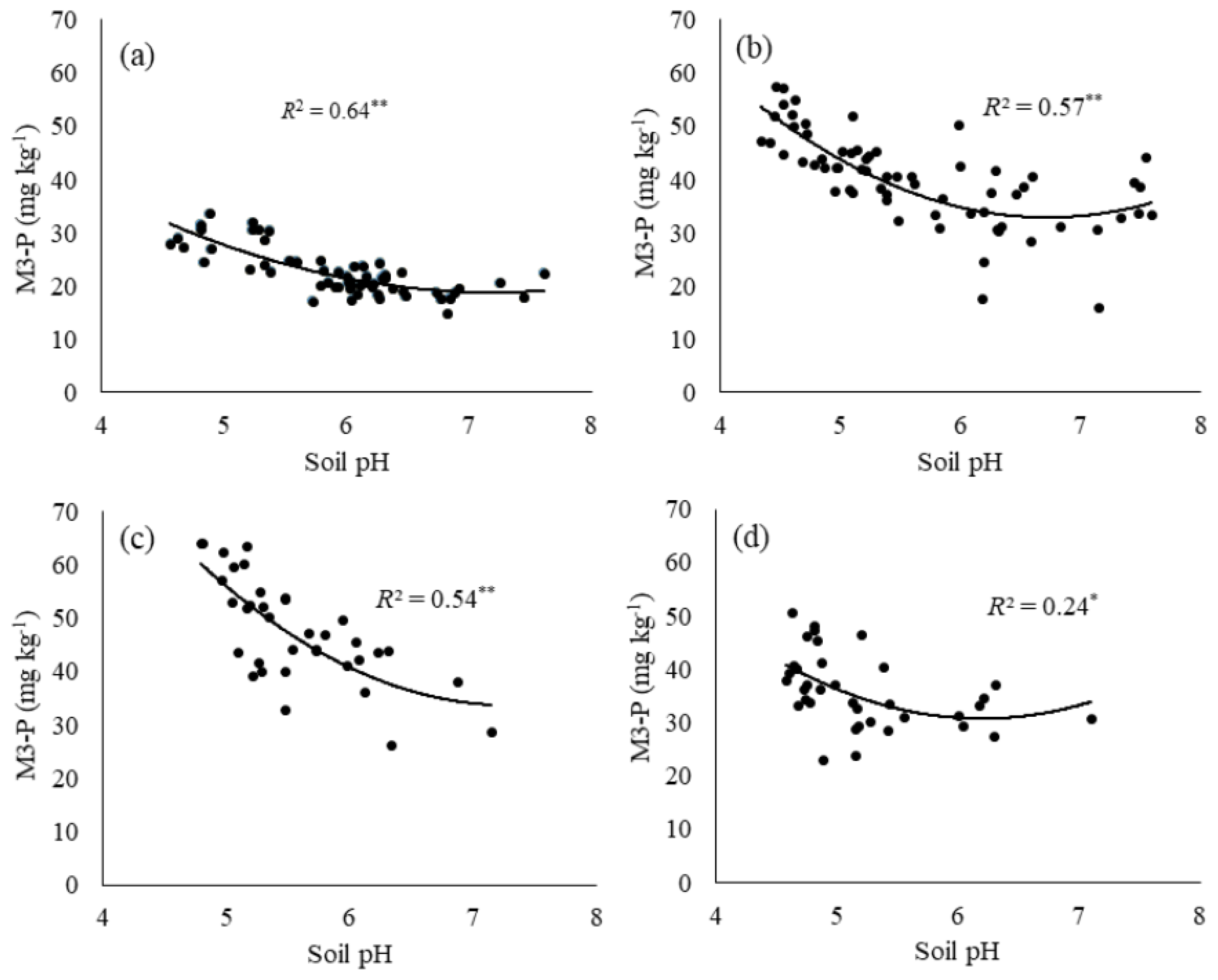
3.3. Impact of Soil pH on Mehlich-3 Extractable Phosphorus
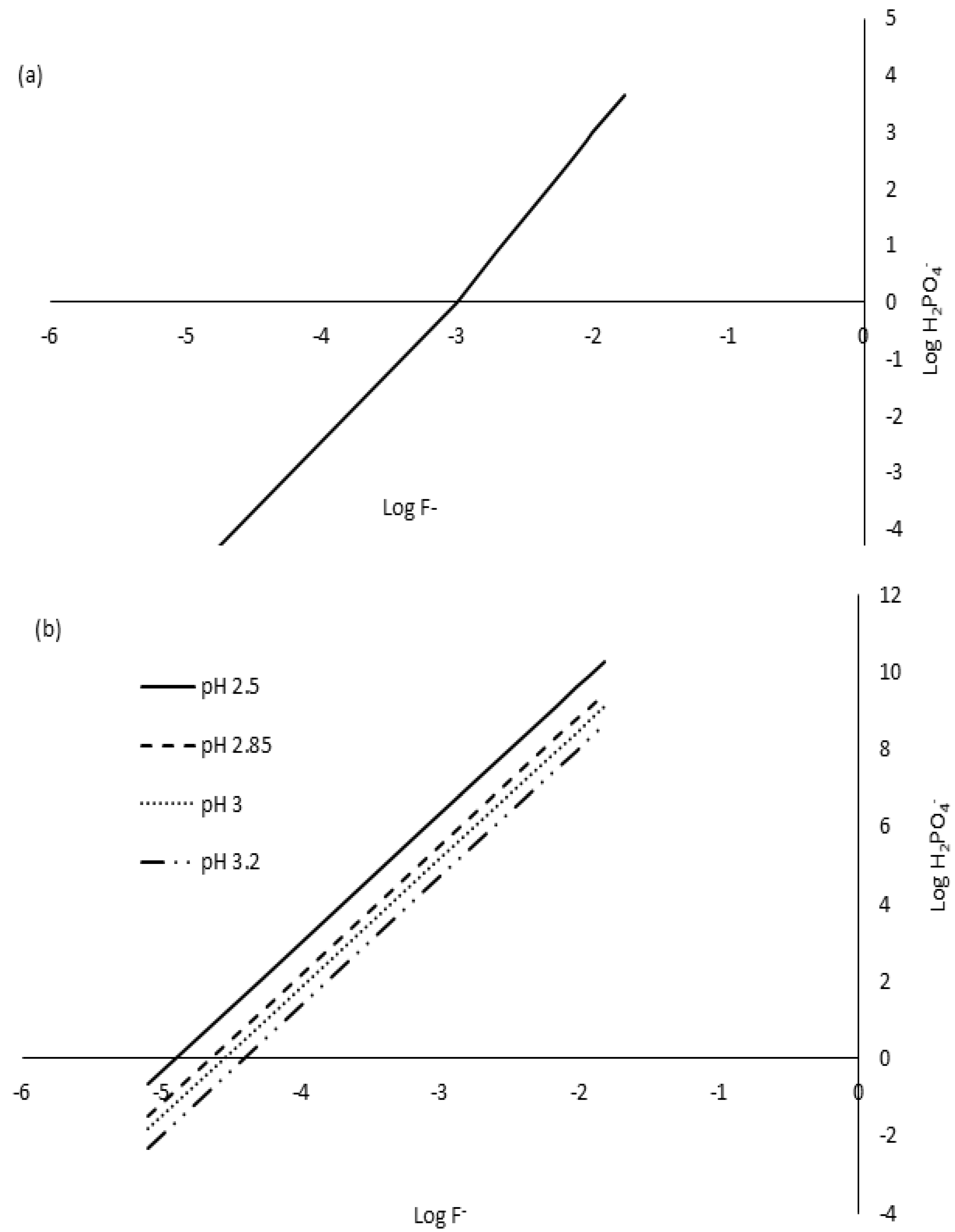
3.3.1. Mehlich-3 Phosphorus Extraction in Low pH Soils (<5.5)
3.3.2. Mehlich-3 Phosphorus Extraction in Medium pH Soils (5.5–7.0)
3.3.3. Mehlich-3 Phosphorus Extraction in High pH Soils (>7.0)
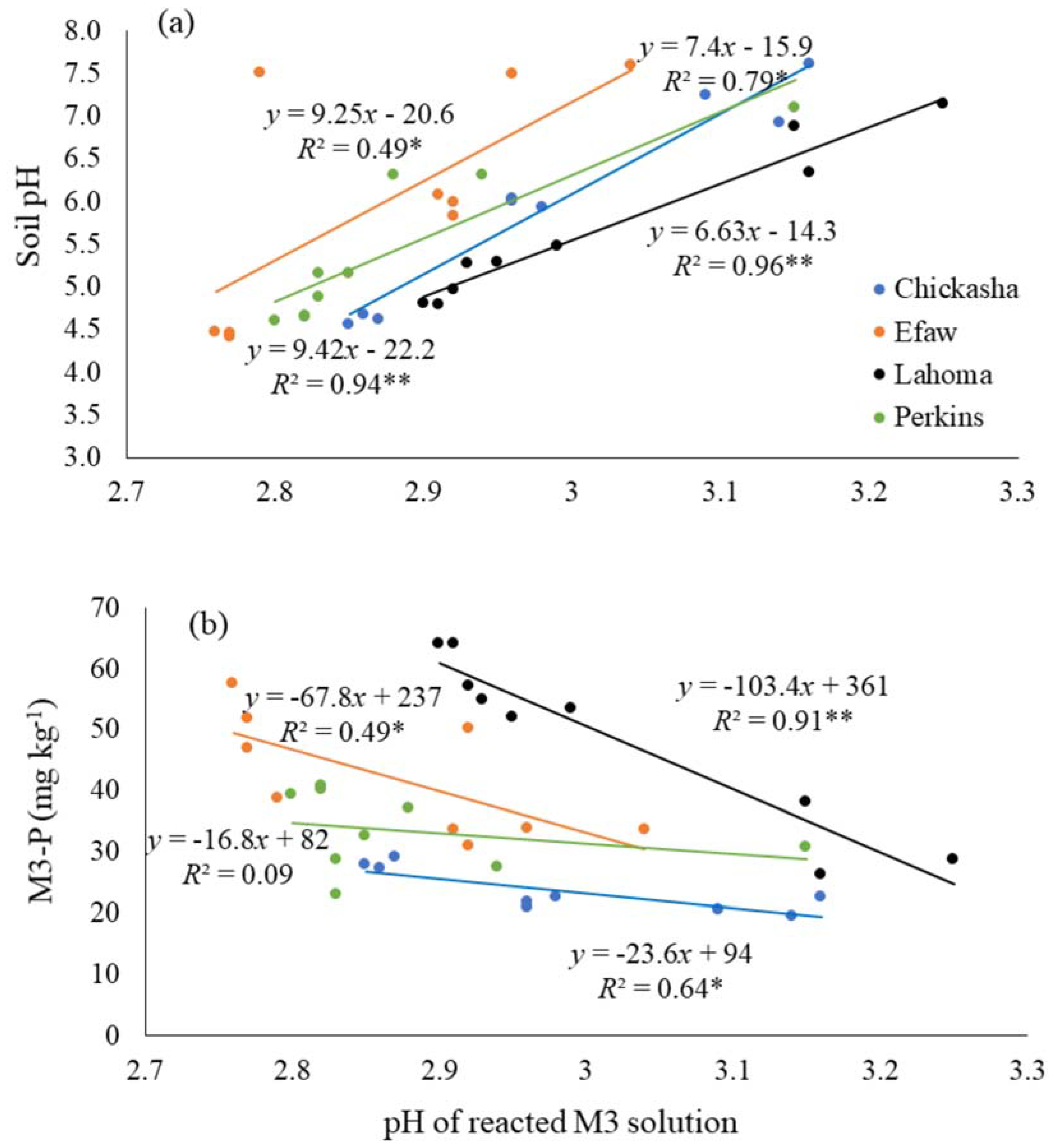
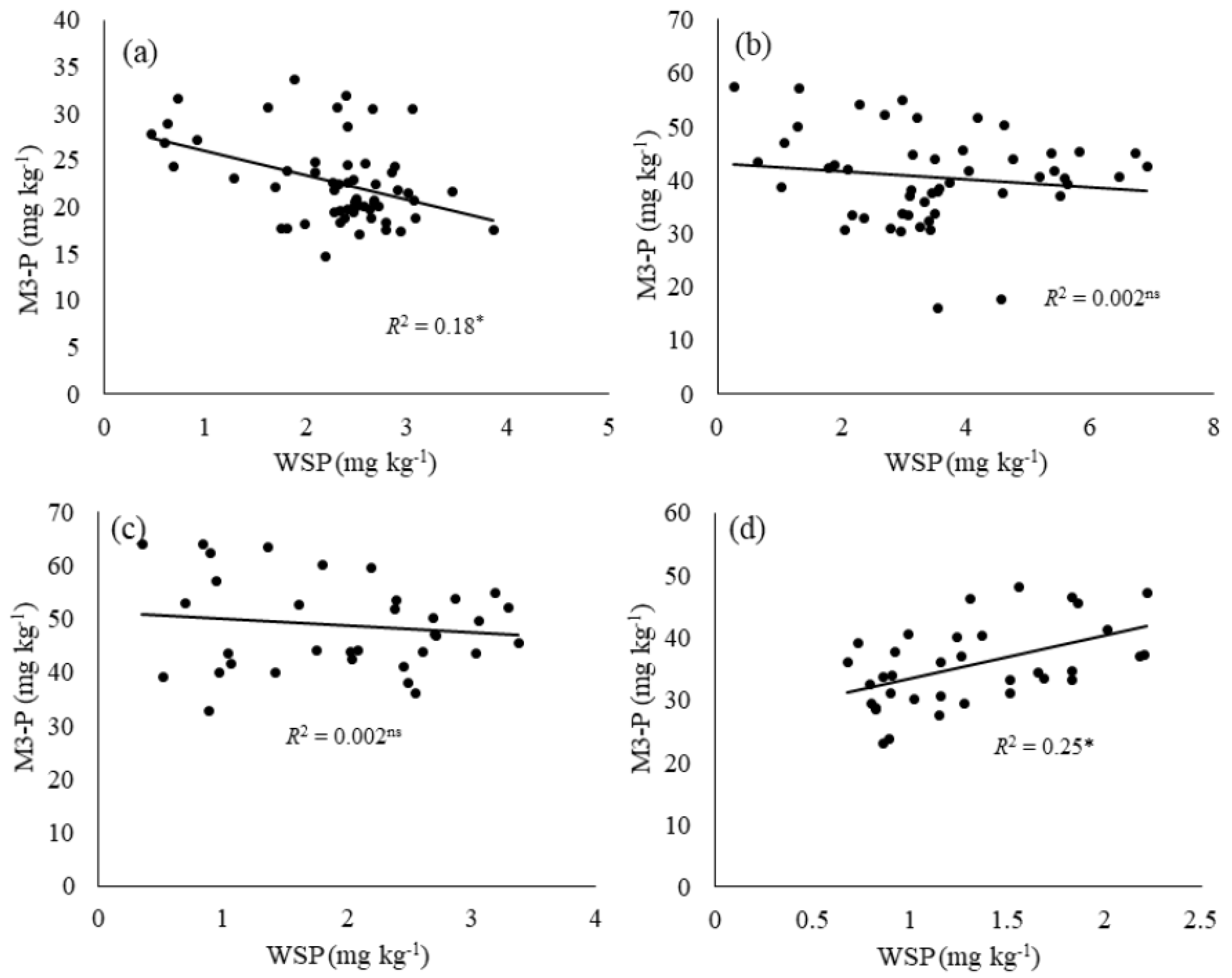
3.4. Relationship between Mehlich-3 and Water-Soluble Phosphorus
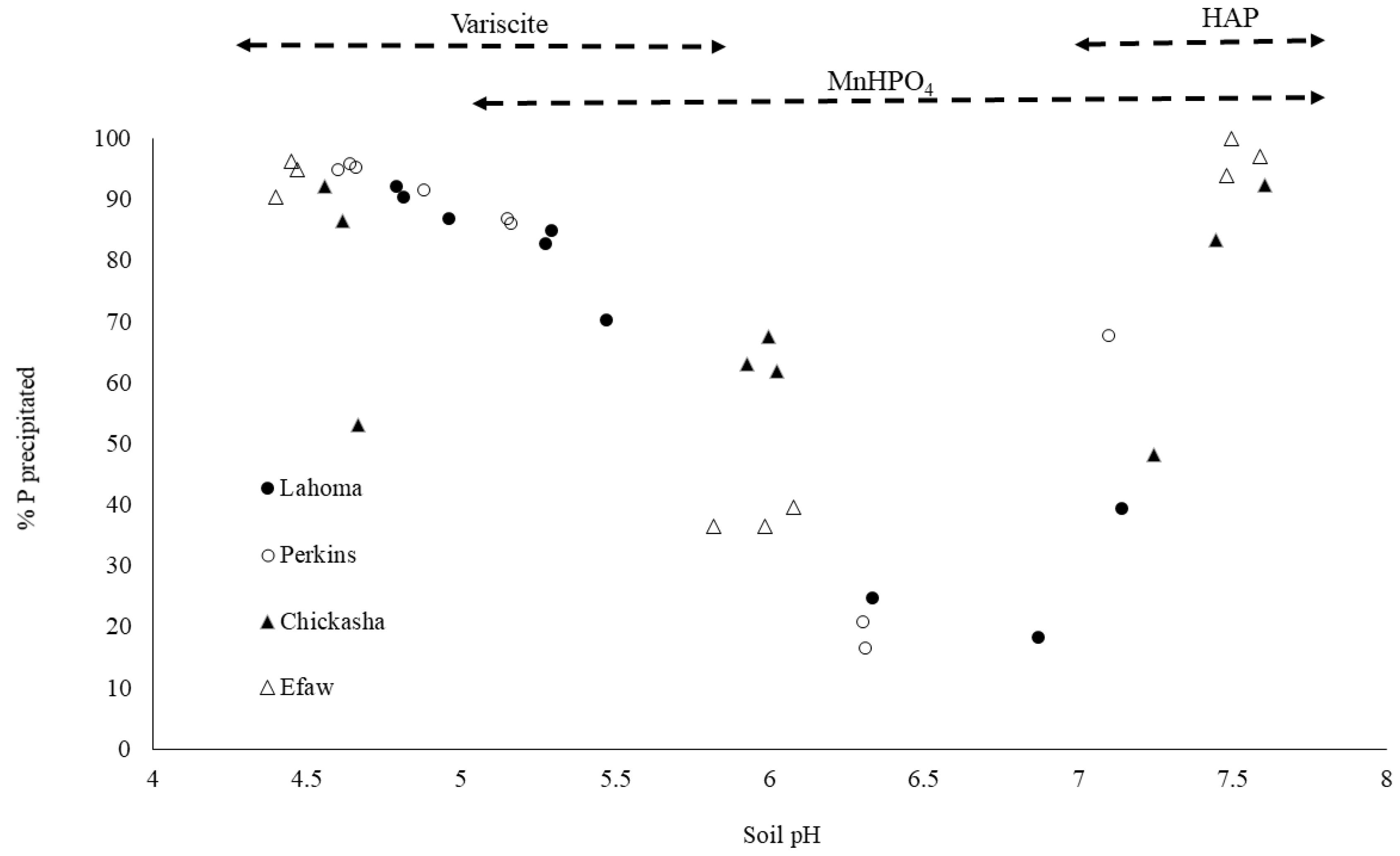
3.5. Soil Phosphorus Forms with Changes in pH
4. Conclusions and Implications
Author Contributions
Funding
Conflicts of Interest
References
- Mehlich, A. Mehlich 3 soil test extractant: A modification of Mehlich 2 extractant. Commun. Soil Sci. Plant Anal. 1984, 15, 1409–1416. [Google Scholar] [CrossRef]
- Sims, J.T.; Maguire, R.O.; Leytem, A.B.; Gartley, K.L.; Pautler, M.C. Evaluation of Mehlich 3 as an agri-environmental soil phosphorus test for the Mid-Atlantic United States of America. Soil Sci. Soc. Am. J. 2002, 66, 2016–2032. [Google Scholar] [CrossRef]
- Osmond, D.; Bolster, C.; Sharpley, A.; Cabrera, M.; Feagley, S.; Forsberg, A.; Mitchell, C.; Mylavarapu, R.; Oldham, J.L.; Radcliffe, D.E. Southern Phosphorus Indices, water quality data, and modeling (APEX, APLE, and TBET) results: A comparison. J. Environ. Qual. 2017, 46, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Lins, I.D.G.; Cox, F.R. Effect of extractant and selected soil properties on predicting the optimum phosphorus fertilizer rate for growing soybeans under field conditions 1. Commun. Soil Sci. Plant Anal. 1989, 20, 319–333. [Google Scholar] [CrossRef]
- Lins, I.D.G.; Cox, F.R. Effect of extractant and selected soil properties on predicting the correct phosphorus fertilization of soybean. Soil Sci. Soc. Am. J. 1989, 53, 813–816. [Google Scholar] [CrossRef]
- Cox, F.R.; Lins, I.D.G. A phosphorus soil test interpretation for corn grown on acid soils varying in crystalline clay content 1. Commun. Soil Sci. Plant Anal. 1984, 15, 1481–1491. [Google Scholar] [CrossRef]
- Michaelson, G.J.; Ping, C.L. Extraction of phosphorus from the major agricultural soils of Alaska 1. Commun. Soil Sci. Plant Anal. 1986, 17, 275–297. [Google Scholar] [CrossRef]
- Fuhrman, J.K.; Zhang, H.; Schroder, J.L.; Davis, R.L.; Payton, M.E. Water-Soluble Phosphorus as Affected by Soil to Extractant Ratios, Extraction Times, and Electrolyte. Commun. Soil Sci. Plant Anal. 2005, 36, 925–935. [Google Scholar] [CrossRef]
- McDowell, R.W.; Sharpley, A.N. Approximating phosphorus release from soils to surface runoff and subsurface drainage. J. Environ. Qual. 2001, 30, 508–520. [Google Scholar] [CrossRef] [PubMed]
- Maguire, R.O.; Sims, J.T. Soil testing to predict phosphorus leaching. J. Environ. Qual. 2002, 31, 1601–1609. [Google Scholar] [CrossRef] [PubMed]
- Penn, C.J.; Mullins, G.L.; Zelazny, L.W. Mineralogy in relation to phosphorus sorption and dissolved phosphorus losses in runoff. Soil Sci. Soc. Am. J. 2005, 69, 1532–1540. [Google Scholar] [CrossRef]
- Penn, C.J.; Mullins, G.L.; Zelazny, L.W.; Sharpley, A.N. Estimating dissolved phosphorus concentrations in runoff from three physiographic regions of Virginia. Soil Sci. Soc. Am. J. 2006, 70, 1967–1974. [Google Scholar] [CrossRef]
- Marschner, H. Marschner’s Mineral Nutrition of Higher Plants; Academic Press: London, UK, 2011; ISBN 0123849063. [Google Scholar]
- Butchee, K.; Arnall, D.B.; Sutradhar, A.; Godsey, C.; Zhang, H.; Penn, C. Determining critical soil pH for grain sorghum production. Int. J. Agron. 2012, 2012, 1–6. [Google Scholar] [CrossRef]
- Sutradhar, A.; Lollato, R.P.; Butchee, K.; Arnall, D.B. Determining critical soil pH for sunflower production. Int. J. Agron. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Lollato, R.P. Limits to Winter Wheat (Triticum aestivum L.) Productivity and Resource-Use Efficiency in the Southern Great Plains; Oklahoma State University: Stillwater, OK, USA, 2015. [Google Scholar]
- U.S. Environmental Protection Agency (USEPA). Method 3050B: Acid Digestion of Sediments, Sludges, and Soils; U.S. Environmental Protection Agency: Washington, DC, USA, 1996.
- Day, P.R. Particle fractionation and particle-size analysis. Methods Soil Anal. 1965, 9, 545–567. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon and organic matter. In Methods of Soil Analysis. Part 3. Chemical methods; Sparks, D.L., Ed.; American Society of Agronomy: Madison, WI, USA, 1996. [Google Scholar]
- Kovar, J.L.; Pierzynski, G.M. Methods of phosphorus analysis for soils, sediments, residuals, and waters second edition. South. Coop. Ser. Bull. 2009, 408, 29–33. [Google Scholar]
- Allison, J.D.; Brown, D.S.; Novo-Gradac, K.J. MINTEQA2/PRODEFA2, A Geochemical Assessment model for Environmental Systems: Version 3.0 User’s Manual; U.S. Environmental Protection Agency: Washington, DC, USA, 1991.
- Lindsay, W.L. Chemical Equilibria in Soils; John Wiley and Sons Ltd.: New York, NY, USA, 1979; ISBN 0471027049. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://cran.r-project.org/manuals.html (accessed on 28 June 2018).
- SAS Inst. Inc. Guide, S.U. Version 9.1; SAS Inst. Inc.: Cary, NC, USA, 2003. [Google Scholar]
- Natural Resources Conservation Service (NRCS). Keys to Soil Taxonomy; USDA Natural Resources Conservation Service: Washington, DC, USA, 2010.
- Vitosh, M.L.; Johnson, J.W.; Mengel, D.B. TH-State Fertilizer Recommendations for Corn, Soybeans, Wheat and Alfalfa; Purdue University: West Lafayette, IN, USA, 1995. [Google Scholar]
- Jackman, R.H.; Black, C.A. Solubility of iron, aluminum, calcium, and magnesium inositol phosphates at different pH values. Soil Sci. 1951, 72, 179–186. [Google Scholar] [CrossRef]
- He, L.M.; Zelazny, L.W.; Martens, D.C.; Baligar, V.C.; Ritchey, K.D. Ionic strength effects on sulfate and phosphate adsorption on γ-alumina and kaolinite: Triple-layer model. Soil Sci. Soc. Am. J. 1997, 61, 784–793. [Google Scholar] [CrossRef]
- Van Raij, B.; Quaggio, J.A. Extractable phosphorus availability indexes as affected by liming. Commun. Soil Sci. Plant Anal. 1990, 21, 1267–1276. [Google Scholar] [CrossRef]
- Sarker, A.; Kashem, M.A.; Osman, K.T.; Hossain, I.; Ahmed, F. Evaluation of available phosphorus by soil test methods in an acidic soil incubated with different levels of lime and phosphorus. Open J. Soil Sci. 2014, 4, 103. [Google Scholar] [CrossRef]
- Sharpley, A.N.; Jones, C.A.; Gray, C. Relationships among soil p test values for soils of differing pedogenesis 1. Commun. Soil Sci. Plant Anal. 1984, 15, 985–995. [Google Scholar] [CrossRef]
- Thomas, G.W.; Peaslee, D.E. Testing soils for phosphorus. Soil Test. Plant Anal. 1973, 1, 115–132. [Google Scholar]
- Mehlich, A. Influence of fluoride, sulfate and acidity on extractable phosphorus, calcium, magnesium and potassium. Commun. Soil Sci. Plant Anal. 1978, 9, 455–476. [Google Scholar] [CrossRef]
- Swenson, R.M.; Cole, C.V.; Sieling, D.H. Fixation of phosphate by iron and aluminum and replacement by organic and inorganic ions. Soil Sci. 1949, 67, 3–22. [Google Scholar] [CrossRef]
- Chang, S.C.; Jackson, M.L. Fractionation of soil phosphorus. Soil Sci. 1957, 84, 133–144. [Google Scholar] [CrossRef]
- Essington, M.E. Soil and Water Chemistry: An Integrative Approach; CRC Press: Boca Raton, FL, USA; London, UK; New York, NY, USA; Washington, DC, USA, 2004. [Google Scholar]
- Warren, J.G.; Penn, C.J.; McGrath, J.M.; Sistani, K. The impact of alum addition on organic P transformations in poultry litter and litter-amended soil. J. Environ. Qual. 2008, 37, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Dao, T.H. Ligands and phytase hydrolysis of organic phosphorus in soils amended with dairy manure. Agron. J. 2004, 96, 1188–1195. [Google Scholar] [CrossRef]
- He, Z.; Honeycutt, C.W.; Zhang, T.; Bertsch, P.M. Preparation and FT-IR characterization of metal phytate compounds. J. Environ. Qual. 2006, 35, 1319–1328. [Google Scholar] [CrossRef] [PubMed]
- Nathan, M.V.; Mallarino, A.P.; Eliason, R.; Miller, R. ICP vs. colorimetric determination of Mehlich III extractable phosphorus. Commun. Soil Sci. Plant Anal. 2002, 33, 2432–2433. [Google Scholar]
- McGrath, J.M.; Sims, J.T.; Maguire, R.O.; Saylor, W.W.; Angel, C.R.; Turner, B.L. Broiler diet modification and litter storage. J. Environ. Qual. 2005, 34, 1896–1909. [Google Scholar] [CrossRef] [PubMed]
- Shang, C.; Zelazny, L.W.; Berry, D.F.; Maguire, R.O. Orthophosphate and phytate extraction from soil components by common soil phosphorus tests. Geoderma 2013, 209, 22–30. [Google Scholar] [CrossRef]
- Sparks, D.L. Environmental Soil Chemistry; Elsevier: London, UK, 2003; ISBN 0080494803. [Google Scholar]
- Penn, C.J.; Warren, J.G. Investigating phosphorus sorption onto kaolinite using isothermal titration calorimetry. Soil Sci. Soc. Am. J. 2009, 73, 560–568. [Google Scholar] [CrossRef]
- Robarge, W.P.; Corey, R.B. Adsorption of phosphate by hydroxy-aluminum species on a cation exchange resin. Soil Sci. Soc. Am. J. 1979, 43, 481–487. [Google Scholar] [CrossRef]
- Smillie, G.W.; Syers, J.K. Calcium fluoride formation during extraction of calcareous soils with fluoride: II. Implications to the Bray P-1 test. Soil Sci. Soc. Am. J. 1972, 36, 25–30. [Google Scholar] [CrossRef]
- Williams, J.D.H.; Syers, J.K.; Harris, R.F.; Armstrong, D.E. Fractionation of inorganic phosphate in calcareous lake sediments. Soil Sci. Soc. Am. J. 1971, 35, 250–255. [Google Scholar] [CrossRef]
- Ebeling, A.M.; Bundy, L.G.; Kittell, A.W.; Ebeling, D.D. Evaluating the Bray P1 test on alkaline, calcareous soils. Soil Sci. Soc. Am. J. 2008, 72, 985–991. [Google Scholar] [CrossRef]
- Mallarino, A.P. Interpretation of soil phosphorus tests for corn in soils with varying pH and calcium carbonate content. J. Prod. Agric. 1997, 10, 163–167. [Google Scholar] [CrossRef]
- Penn, C.J.; Bryant, R.B. Incubation of dried and sieved soils can induce calcium phosphate precipitation/adsorption. Commun. Soil Sci. Plant Anal. 2006, 37, 1437–1449. [Google Scholar] [CrossRef]
- McDowell, R.W.; Condron, L.M.; Mahieu, N.; Brookes, P.C.; Poulton, P.R.; Sharpley, A.N. Analysis of potentially mobile phosphorus in arable soils using solid state nuclear magnetic resonance. J. Environ. Qual. 2002, 31, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Schwab, A.P.; Lindsay, W.L. The effect of redox on the solubility and availability of manganese in a calcareous soil. Soil Sci. Soc. Am. J. 1983, 47, 217–220. [Google Scholar] [CrossRef]
- Schwab, A.P. Stability of Fe Chelates and the Availability of Fe and Mn to Plants Affected by Redox. Ph.D. Thesis, Colorado State University, Fort Collins, CO, USA, 1981. [Google Scholar]
- Boyle, F.W.; Lindsay, W.L. Manganese phosphate equilibrium relationships in soils. Soil Sci. Soc. Am. J. 1986, 50, 588–593. [Google Scholar] [CrossRef]
- Baker, D.B.; Confesor, R.; Ewing, D.E.; Johnson, L.T.; Kramer, J.W.; Merryfield, B.J. Phosphorus loading to Lake Erie from the Maumee, Sandusky and Cuyahoga rivers: The importance of bioavailability. J. Gt. Lakes Res. 2014, 40, 502–517. [Google Scholar] [CrossRef]
- Barber, S.A. A diffusion and mass-flow concept of soil nutrient availability. Soil Sci. 1962, 93, 39–49. [Google Scholar] [CrossRef]
- Edwards, A.C. Soil acidity and its interactions with phosphorus availability for a range of different crop types. In Plant-Soil Interactions at Low pH; Springer: Dordrecht, The Netherlands, 1991; pp. 299–305. [Google Scholar]
- Luz, J.M.Q; Queiroz, A.A.; Borges, M.; Oliveira, R.C.; Leite, S.S.; Cardoso, R.R. Influence of phosphate fertilization on phosphorus levels in foliage and tuber yield of the potato cv. Ágata. Semina Ciências Agrárias 2013, 34, 649–656. [Google Scholar] [CrossRef]
| Location | Soil Series | Sand | Silt | Clay | OM | pH | Total P | WSP | M3-P | M3-Fe | M3-Al | M3-Ca | DPSM3 | DPSOx |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| g 100 g−1 | g kg−1 | mg kg−1 | % | |||||||||||
| Chickasha | Dale | 40 | 45 | 15 | 14.5 | 4.6–7.6 (5.9) | 283–336 (320) | 0.46–7.61 (2.29) | 15–34 (25) | 67–103 (82) | 458–1001 (643) | 762–1997 (1212) | 2.1–3.9 (3.1) | 5.9–7.7 (6.9) |
| Efaw | Easpur | 60 | 27.5 | 12.5 | 7.6 | 4.3–7.6 (5.64) | 174–210 (186) | 0.27–6.91 (3.48) | 16–58 (40) | 74–139 (107) | 370–704 (514) | 461–2016 (949) | 2.5–9.9 (6.9) | 12.8–16.9 (14.1) |
| Lahoma | Grant | 30 | 50 | 20 | 16.9 | 4.8–7.1 (5.6) | 246–302 (278) | 0.35–3.38 (1.91) | 26–64 (48) | 46–100 (81) | 590–1070 (842) | 760–2289 (1232) | 2.4–6.4 (5.0) | 6.1–12.6 (9.5) |
| Perkins | Teller | 65 | 25 | 10 | 9.6 | 4.6–7.1 (5.2) | 139–183 (163) | 0.68–2.22 (1.31) | 23–51 (36) | 70–120 (92) | 600–950 (754) | 180–1100 (496) | 2.9–5.2 (4.1) | 5.9–10.5 (7.7) |
| Location | Soil pH | Percent Precipitated | Predicted Solid Minerals | ||||
|---|---|---|---|---|---|---|---|
| Al | Ca | Fe | Mn | P | |||
| Lahoma | 4.79 | 99.6 | 0 | 100 | 0 | 92.2 | Var, Hem, Gib |
| Lahoma | 4.81 | 99.6 | 0 | 100 | 0 | 90.5 | Var, Hem, Gib |
| Lahoma | 4.96 | 99.8 | 0 | 100 | 0 | 86.9 | Var, Hem, Gib |
| Lahoma | 5.27 | 100 | 0 | 100 | 39.8 | 82.9 | Var, Hem, Gib, MnHPO4 |
| Lahoma | 5.29 | 100 | 0 | 100 | 55.1 | 85 | Var, Hem, Gib, MnHPO4 |
| Lahoma | 5.47 | 100 | 0 | 100 | 75.1 | 70.3 | Var, Hem, Gib, MnHPO4 |
| Lahoma | 6.33 | 100 | 0 | 100 | 94.1 | 24.9 | Hem, Gib, MnHPO4 |
| Lahoma | 6.87 | 100 | 0 | 100 | 99.2 | 18.4 | Hem, Gib, MnHPO4 |
| Lahoma | 7.14 | 100 | 1.5 | 100 | 97.3 | 39.4 | Hem, Gib, MnHPO4, HAP |
| Perkins | 4.66 | 99.9 | 0 | 100 | 0 | 95.3 | Var, Hem, Gib |
| Perkins | 4.64 | 99.9 | 0 | 100 | 0 | 95.8 | Var, Hem, Gib |
| Perkins | 4.6 | 99.9 | 0 | 100 | 0 | 94.8 | Var, Hem, Gib |
| Perkins | 4.88 | 99.9 | 0 | 100 | 0 | 91.6 | Var, Hem, Gib |
| Perkins | 5.15 | 100 | 0 | 100 | 1.4 | 86.8 | Var, Hem, Gib, MnHPO4 |
| Perkins | 5.16 | 100 | 0 | 100 | 0 | 86.1 | Var, Hem, Gib |
| Perkins | 6.31 | 100 | 0 | 100 | 98.9 | 16.5 | Hem, Gib, MnHPO4 |
| Perkins | 6.3 | 100 | 0 | 100 | 98.5 | 20.9 | Hem, Gib, MnHPO4 |
| Perkins | 7.1 | 100 | 6.9 | 100 | 98.5 | 67.7 | Hem, Gib, MnHPO4, HAP |
| Chickasha | 4.56 | 99.2 | 0 | 100 | 1.1 | 92.1 | Hem, Gib, MnHPO4 |
| Chickasha | 4.6 | 99.3 | 0 | 100 | 1.5 | 86.5 | Var, Hem, Gib |
| Chickasha | 4.67 | 99.5 | 0 | 100 | 0 | 53 | Var, Hem, Gib |
| Chickasha | 5.93 | 100 | 0 | 100 | 94.6 | 63 | Hem, Gib, MnHPO4 |
| Chickasha | 5.99 | 100 | 0 | 100 | 94.5 | 67.6 | Hem, Gib, MnHPO4 |
| Chickasha | 6.03 | 100 | 0 | 100 | 95.4 | 61.9 | Hem, Gib, MnHPO4 |
| Chickasha | 7.25 | 100 | 1.7 | 100 | 99 | 48.1 | Hem, Gib, MnHPO4, HAP |
| Chickasha | 7.44 | 100 | 4.8 | 100 | 95.9 | 83.3 | Hem, Gib MnHPO4, HAP |
| Chickasha | 7.61 | 100 | 5.4 | 100 | 89.9 | 92.3 | Hem, Gib MnHPO4, HAP |
| Efaw | 4.40 | 13 | 0 | 100 | 4.4 | 65.3 | Var, Hem, MnHPO4 |
| Efaw | 4.45 | 97.1 | 0 | 100 | 0 | 96.2 | Var, Hem, Gib |
| Efaw | 4.47 | 95.6 | 0 | 100 | 0 | 94.8 | Var, Hem, Gib |
| Efaw | 5.82 | 100 | 0 | 100 | 96.5 | 36.4 | Var, Hem, Gib, MnHPO4 |
| Efaw | 5.99 | 100 | 0 | 100 | 98.4 | 36.4 | Hem, Gib, MnHPO4 |
| Efaw | 6.08 | 100 | 0 | 100 | 97.4 | 39.5 | Hem, Gib, MnHPO4 |
| Efaw | 7.50 | 98.2 | 0 | 100 | 8.2 | 99.9 | Hem, Gib, MnHPO4 |
| Efaw | 7.48 | 100 | 6.5 | 100 | 95.8 | 93.9 | Hem, Gib, MnHPO4, HAP |
| Efaw | 7.59 | 100 | 4.5 | 100 | 87 | 97.1 | Hem, Gib, MnHPO4, HAP |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Penn, C.J.; Rutter, E.B.; Arnall, D.B.; Camberato, J.; Williams, M.; Watkins, P. A Discussion on Mehlich-3 Phosphorus Extraction from the Perspective of Governing Chemical Reactions and Phases: Impact of Soil pH. Agriculture 2018, 8, 106. https://doi.org/10.3390/agriculture8070106
Penn CJ, Rutter EB, Arnall DB, Camberato J, Williams M, Watkins P. A Discussion on Mehlich-3 Phosphorus Extraction from the Perspective of Governing Chemical Reactions and Phases: Impact of Soil pH. Agriculture. 2018; 8(7):106. https://doi.org/10.3390/agriculture8070106
Chicago/Turabian StylePenn, Chad J., E. Bryan Rutter, D. Brian Arnall, James Camberato, Mark Williams, and Patrick Watkins. 2018. "A Discussion on Mehlich-3 Phosphorus Extraction from the Perspective of Governing Chemical Reactions and Phases: Impact of Soil pH" Agriculture 8, no. 7: 106. https://doi.org/10.3390/agriculture8070106
APA StylePenn, C. J., Rutter, E. B., Arnall, D. B., Camberato, J., Williams, M., & Watkins, P. (2018). A Discussion on Mehlich-3 Phosphorus Extraction from the Perspective of Governing Chemical Reactions and Phases: Impact of Soil pH. Agriculture, 8(7), 106. https://doi.org/10.3390/agriculture8070106






