Differential Gene Expression in the Model Actinomycete Streptomyces coelicolor A3(2) Supports Nitrogen Mining Dependent on the Plant Carbon to Nitrogen Ratio
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil and Plant Sampling
2.2. Bacterial Strain and Culture Conditions
2.3. Soil Incubation
2.4. RNA Extraction, Sequencing, and DGE Analysis
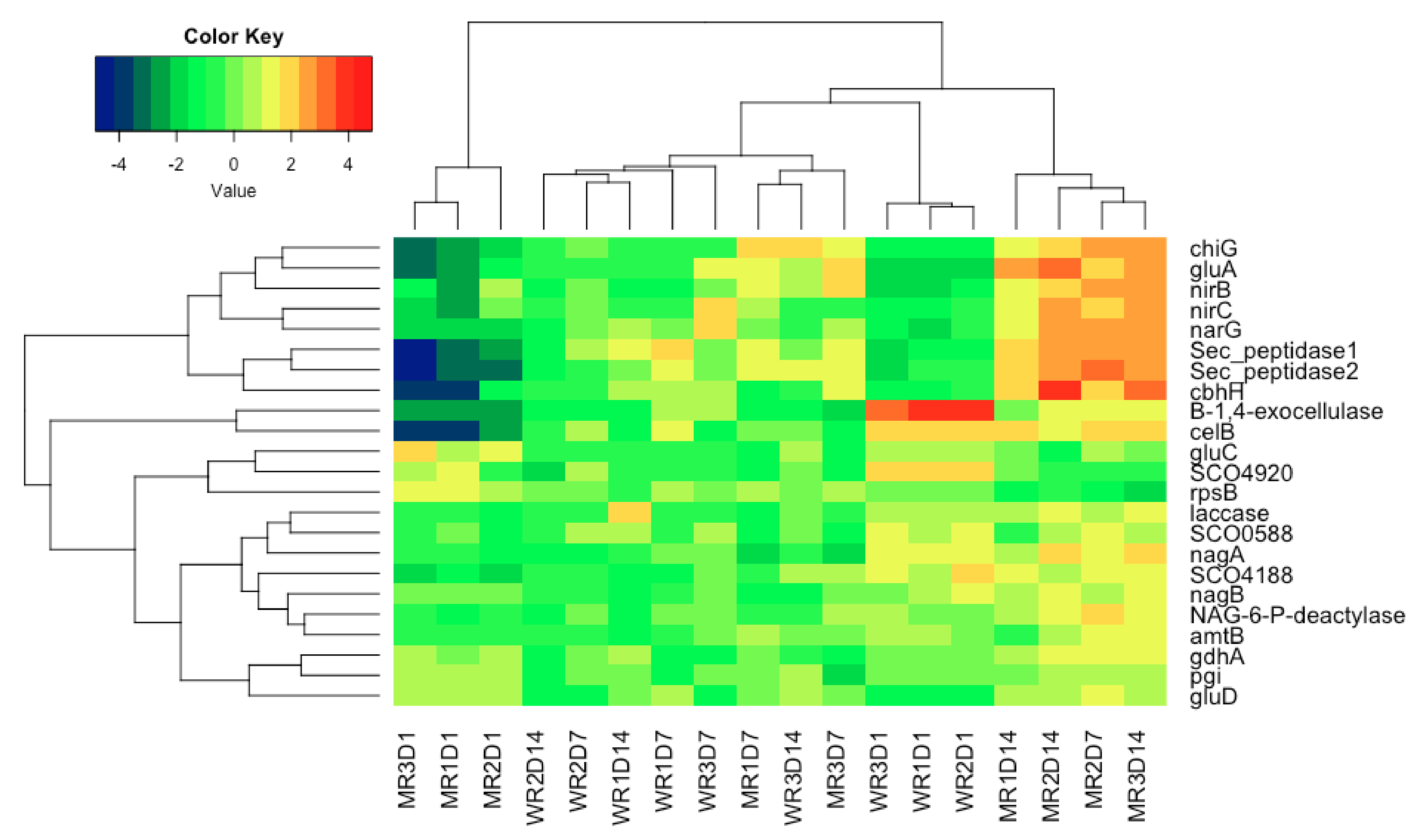
3. Results
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Murty, D.; Kirschbaum, M.U.F.; McMurtrie, R.E.; McGilvray, H. Does conversion of forest to agricultural land change soil carbon and nitrogen? A review of the literature. Glob. Chang. Biol. 2002, 17, 1658–1670. [Google Scholar] [CrossRef]
- Ciais, P.; Sabine, C.; Bala, G.; Bopp, L.; Brovkin, V.; Canadell, J.; Chhabra, A.; DeFries, R.; Galloway, J.; Heimann, M.; et al. Carbon and other biogeochemical cycles. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- FAO. Food and Agriculture Organization Chapter 5: Drivers of global soil changes. In Status of the World’s Soil Resources (SWSR); Food and Agriculture Organization: Rome, Italy, 2015. [Google Scholar]
- Hutchinson, J.J.; Campbell, C.A.; Desjardins, R.L. Some perspectives on carbon sequestration in agriculture. Agric. For. Meteorol. 2007, 142, 288–302. [Google Scholar] [CrossRef]
- Cotrufo, M.F.; Wallenstein, M.D.; Boot, C.M.; Denef, K.; Paul, E. The Microbial Efficiency-Matrix Stabilization (MEMS) framework integrates plant litter decomposition with soil organic matter stabilization: Do labile plant inputs form stable soil organic matter? Glob. Chang. Biol. 2013, 19, 988–995. [Google Scholar] [CrossRef]
- Schmidt, M.W.I.; Torn, M.S.; Abiven, S.; Dittmar, T.; Guggenberger, G.; Janssens, I.A.; Kleber, M.; Kogel-Knabner, I.; Lehmann, J.; Manning, D.A.C.; et al. Persistence of soil organic matter as an ecosystem property. Nature 2011, 478, 49–56. [Google Scholar] [CrossRef]
- Six, J.; Conant, R.T.; Paul, E.A.; Paustian, K. Stabilization mechanisms of soil organic matter: Implications for C-saturation of soils. Plant Soil 2002, 241, 155–176. [Google Scholar] [CrossRef]
- Craine, J.M.; Morrow, C.; Fierer, N. Microbial nitrogen limitation increases decomposition. Ecology 2007, 88, 2105–2113. [Google Scholar] [CrossRef]
- Moorhead, D.L.; Sinsabaugh, R.L. A theoretical model of litter decay and microbial interaction. Ecol. Monogr. 2006, 76, 151–174. [Google Scholar] [CrossRef]
- Zechmeister-Boltenstern, S.; Keiblinger, K.M.; Mooshammer, M.; Penuelas, J.; Richter, A.; Sardans, J.; Wanek, W. The application of ecological stoichiometry to plant-microbial-soil organic matter transformations. Ecol. Monogr. 2015, 85, 133–155. [Google Scholar] [CrossRef]
- Finn, D.; Page, K.; Catton, K.; Strounina, E.; Kienzle, M.; Robertson, F.; Armstrong, R.; Dalal, R. Effect of added nitrogen on plant litter decomposition depends on initial soil carbon and nitrogen stoichiometry. Soil Biol. Biochem. 2015, 91, 160–168. [Google Scholar] [CrossRef]
- Knicker, H. Soil organic N–An under-rated player for C sequestration in soils? Soil Biol. Biochem. 2011, 43, 1118–1129. [Google Scholar] [CrossRef]
- Fierer, N.; Lauber, C.L.; Ramirez, K.S.; Zaneveld, J.; Bradford, M.A.; Knight, R. Comparative metagenomic, phylogenetic and physiological analyses of soil microbial communities across nitrogen gradients. ISME J. 2012, 6, 1007–1017. [Google Scholar] [CrossRef] [PubMed]
- Mendes, L.W.; Brossi, M.J.D.; Kuramae, E.E.; Tsai, S.M. Land-use system shapes soil bacterial communities in Southeastern Amazon region. Appl. Soil Ecol. 2015, 95, 151–160. [Google Scholar] [CrossRef]
- Wessen, E.; Hallin, S.; Philippot, L. Differential responses of bacterial and archaeal groups at high taxonomical ranks to soil management. Soil Biol. Biochem. 2010, 42, 1759–1765. [Google Scholar] [CrossRef]
- Goodfellow, M.; Williams, S.T. Ecology of Actinomycetes. Ann. Rev. Microbiol. 1983, 37, 189–216. [Google Scholar] [CrossRef] [PubMed]
- Hopwood, D.A. Forty years of genetics with Streptomyces: From in vivo through in vitro to in silico. Microbiol. UK 1999, 145, 2183–2202. [Google Scholar] [CrossRef] [PubMed]
- Bentley, S.D.; Chater, K.F.; Cerdeno-Tarraga, A.M.; Challis, G.L.; Thomson, N.R.; James, K.D.; Harris, D.E.; Quail, M.A.; Kieser, H.; Harper, D.; et al. Complete genome sequence of the model actinomycete Streptomyces coelicolor A3(2). Nature 2002, 417, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, D.A. Primary metabolism and its control in Streptomycetes: A most unusual group of bacteria. Adv. Microb. Physiol. 2000, 42, 47–238. [Google Scholar] [PubMed]
- Nieselt, K.; Battke, F.; Herbig, A.; Bruheim, P.; Wentzel, A.; Jakobsen, O.M.; Sletta, H.; Alam, M.T.; Merlo, M.E.; Moore, J.; et al. The dynamic architecture of the metabolic switch in Streptomyces coelicolor. BMC Genom. 2010, 11, 10. [Google Scholar] [CrossRef]
- Rayment, G.; Lyons, D. Soil Chemical Methods–Australasia; CSIRO Publishing: Clayton, Australia, 2011. [Google Scholar]
- Cassel, D.K.; Nielsen, D.R. Field capacity and available water capacity. In Methods of Soil Analysis, Part 1. Physical and Mineralogical Methods–Agronomy Monograph No. 9, 2nd ed.; Klute, A., Ed.; American Society of Agronomy–Soil Science Society of America: Madison, WI, USA, 1986; pp. 901–926. [Google Scholar]
- Bank, T.L.; Kukkadapu, R.K.; Madden, A.S.; Ginder-Vogel, M.A.; Baldwin, M.E.; Jardine, P.M. Effects of gamma-sterilization on the physico-chemical properties of natural sediments. Chem. Geol. 2008, 251, 1–7. [Google Scholar] [CrossRef]
- Berns, A.E.; Philipp, H.; Narres, H.D.; Burauel, P.; Vereecken, H.; Tappe, W. Effect of gamma-sterilization and autoclaving on soil organic matter structure as studied by solid state NMR, UV and fluorescence spectroscopy. Eur. J. Soil Sci. 2008, 59, 540–550. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017; ISBN 3-900051-07-0. [Google Scholar]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed]
- Venables, W.N.; Ripley, B.D. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, USA, 2002; ISBN 0-387-95457-0. [Google Scholar]
- Ulanova, D.; Kitani, S.; Fukusaki, E.; Nihira, T. SdrA, a New DeoR Family Regulator Involved in Streptomyces avermitilis Morphological Development and Antibiotic Production. Appl. Environ. Microbiol. 2013, 79, 7916–7921. [Google Scholar] [CrossRef]
- Castro-Melchor, M.; Charaniya, S.; Karypis, G.; Takano, E.; Hu, W.S. Genome-wide inference of regulatory networks in Streptomyces coelicolor. BMC Genom. 2010, 11, 578. [Google Scholar] [CrossRef]
- Hoskisson, P.A.; Rigali, S.; Fowler, K.; Findlay, K.C.; Buttner, M.J. DevA, a GntR-like transcriptional regulator required for development in Streptomyces coelicolor. J. Bacteriol. 2006, 188, 5014–5023. [Google Scholar] [CrossRef]
- Tiffert, Y.; Supra, P.; Wurm, R.; Wohlleben, W.; Wagner, R.; Reuther, J. The Streptomyces coelicolor GlnR regulon: Identification of new GlnR targets and evidence for a central role of GlnR in nitrogen metabolism in Actinomycetes. Mol. Microbiol. 2008, 67, 861–880. [Google Scholar] [CrossRef]
- Reitzer, L.; Schneider, B.L. Metabolic context and possible physiological themes of sigma(54)-dependent genes in Escherichia coli. Microbiol. Mol. Biol. Rev. 2001, 65, 422–444. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Jiang, W.; Lu, Y. A novel two component system, GluR-K, involved in glutamate sensing and uptake in Streptomyces coelicolor. J. Bacteriol. 2017. [Google Scholar] [CrossRef]
- Gil, R.; Silva, F.J.; Peretó, J.; Moya, A. Determination of the core of a minimal bacterial gene set. Microbiol. Mol. Biol. Rev. 2004, 68, 518–537. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.J.; Baldock, J.A.; Dalal, R.C.; Moody, P.W. Decomposition dynamics of plant materials in relation to nitrogen availability and biochemistry determined by NMR and wet chemical analysis. Soil Biol. Biochem. 2004, 36, 2045–2058. [Google Scholar] [CrossRef]
- Manzoni, S.; Taylor, P.; Richter, A.; Porporato, A.; Agren, G.I. Environmental and stoichiometric controls on microbial carbon-use efficiency in soils. New Phytol. 2012, 196, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Hessen, D.O.; Ågren, G.I.; Anderson, T.R.; Elser, J.J.; de Ruiter, P.C. Carbon sequestration in ecosystems: The role of stoichiometry. Ecology 2004, 85, 1179–1192. [Google Scholar] [CrossRef]
- Berg, B.; Matzner, E. Effect of N deposition on decomposition of plant litter and soil organic matter in forest systems. Environ. Rev. 1997, 5, 1–25. [Google Scholar] [CrossRef]
- Fog, K. The effect of added nitrogen on the rate of decomposition of organic matter. Biol. Rev. 1988, 63, 433–462. [Google Scholar] [CrossRef]
- Ramirez, K.S.; Craine, J.M.; Fierer, N. Consistent effects of nitrogen amendments on soil microbial communities and processes across biomes. Glob. Chang. Biol. 2012, 18, 1918–1927. [Google Scholar] [CrossRef]
- Allison, S.D.; Vitousek, P.M. Responses of extracellular enzymes to simple and complex nutrient inputs. Soil Biol. Biochem. 2005, 37, 937–944. [Google Scholar] [CrossRef]
- Sinsabaugh, R.L.; Carreiro, M.M.; Repert, D.A. Allocation of extracellular enzymatic activity in relation to litter composition, N deposition, and mass loss. Biogeochemistry 2002, 60, 1–24. [Google Scholar] [CrossRef]
- Carreiro, M.M.; Sinsabaugh, R.L.; Repert, D.A.; Parkhurst, D.F. Microbial enzyme shifts explain litter decay responses to simulated nitrogen deposition. Ecology 2000, 81, 2359–2365. [Google Scholar] [CrossRef]
- DeForest, J.L.; Zak, D.R.; Pregitzer, K.S.; Burton, A.J. Atmospheric nitrate deposition and the microbial degradation of cellobiose and vanillin in a northern hardwood forest. Soil Biol. Biochem. 2004, 36, 965–971. [Google Scholar] [CrossRef]
- Saiya-Cork, K.R.; Sinsabaugh, R.L.; Zak, D.R. The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol. Biochem. 2002, 34, 1309–1315. [Google Scholar] [CrossRef]
- Haynes, R.J. Labile organic matter fractions and aggregate stability under short-term, grass-based leys. Soil Biol. Biochem. 1999, 31, 1821–1830. [Google Scholar] [CrossRef]
- Kirkby, C.A.; Richardson, A.E.; Wade, L.J.; Batten, G.D.; Blanchard, C.; Kirkegaard, J.A. Carbon-nutrient stoichiometry to increase soil carbon sequestration. Soil Biol. Biochem. 2013, 60, 77–86. [Google Scholar] [CrossRef]
- Powlson, D.S.; Whitmore, A.P.; Goulding, K.W.T. Soil carbon sequestration to mitigate climate change: A critical re-examination to identify the true and the false. Eur. J. Soil Sci. 2011, 62, 42–55. [Google Scholar] [CrossRef]
- Conant, R.T.; Paustian, K.; Elliott, E.T. Grassland management and conversion into grassland: Effects on soil carbon. Ecol. Appl. 2001, 11, 343–355. [Google Scholar] [CrossRef]
- Lee, Z.M.; Schmidt, T.M. Bacterial growth efficiency varies in soils under different land management practices. Soil Biol. Biochem. 2014, 69, 282–290. [Google Scholar] [CrossRef]
- Dungait, J.A.; Hopkins, D.W.; Gregory, A.S.; Whitmore, A.P. Soil organic matter turnover is governed by accessibility not recalcitrance. Glob. Chang. Biol. 2012, 18, 1781–1796. [Google Scholar] [CrossRef]
- Finn, D.; Page, K.; Catton, K.; Kienzle, M.; Robertson, F.; Armstrong, R.; Dalal, R. Ecological stoichiometry controls the transformation and retention of plant-derived organic matter to humus in response to nitrogen fertilisation. Soil Biol. Biochem. 2016, 99, 117–127. [Google Scholar] [CrossRef]

| Day 1 | Day 7 | Day 14 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Category | Gene | Wheat | Mung Bean | Average LFC | Wheat | Mung Bean | Average LFC | Wheat | Mung Bean | Average LFC |
| Regulation | SCO4920 deoR-family reg | 2.07 ± 0.2 | 0.71 ± 0.4 | 1.36 * | −0.15 ± 0.9 | −1.05 ± 0.3 | 0.9 | −0.61 ± 0.9 | −0.46 ± 0.4 | 0.14 |
| SCO4188 gntR-family reg | 1.25 ± 0.4 | −1.77 ± 0.2 | 3.01 ** | −0.63 ± 0.6 | 0.97 ± 0.6 | 1.6 * | −0.48 ± 0.9 | 1.25 ± 0.4 | 1.73 | |
| SCO0588 sensor kinase | 1 ± 0.46 | −0.72 ± 0.7 | 1.72 * | 0 ± 0.5 | −1.47 ± 0.1 | 1.47 * | −0.02 ± 0.6 | 0.27 ± 0.8 | 0.29 | |
| Membrane | SCO5583 amtB | 0.36 ± 0.3 | −0.38 ± 0 | 0.74 * | −0.51 ± 0.6 | 0.52 ± 0.4 | 1.03 | −0.7 ± 0.4 | 0.38 ± 1 | 1.07 |
| SCO2831 gluA | −1.8 ± 0.2 | −2.39 ± 1 | 0.58 | −0.42 ± 0.1 | 1.5 ± 0.4 | 1.92 ** | −0.11 ± 0.6 | 2.76 ± 0.4 | 2.87 ** | |
| SCO5775 gluC | 0.69 ± 0.1 | 1.29 ± 0.5 | 0.61 | −0.56 ± 0.1 | −1.01 ± 0 | 0.45 | −0.26 ± 0.5 | 0.67 ± 0.1 | 0.19 | |
| SCO5774 gluD | −1.28 ± 0.1 | 0.67 ± 0.1 | 1.94 *** | −1.11 ± 0.1 | 0.42 ± 0.7 | 1.53 * | −0.31 ± 0.7 | 0.6 ± 0.3 | 0.92 | |
| Extracellular | SCO7176 Sec peptidase1 | −1.39 ± 0.4 | −3.53 ± 1.3 | 2.14 | 0.8 ± 0.9 | 1.7 ± 0.8 | 0.9 | −0.06 ± 1 | 2.48 ± 0.5 | 2.54 * |
| Enzymes | SCO7188 Sec peptidase2 | −1.08 ± 0.8 | −3.53 ± 0.7 | 2.45 * | −0.44 ± 0.4 | 2 ± 0.8 | 2.44 *** | 0.16 ± 1.2 | 2.34 ± 0.5 | 2.19 |
| SCO1187 celB | 1.86 ± 0.2 | −3.23 ± 0.7 | 5.1 ** | 1 ± 0.5 | −0.73 ± 0.7 | 1.73 * | −0.71 ± 0.4 | 1.6 ± 0.3 | 2.3 ** | |
| SCO6548 put beta-1,4-exocellulase | 3.72 ± 0.4 | −2.76 ± 0.2 | 6.49 *** | 0.54 ± 0 | −1.36 ± 0.4 | 1.9 *** | −1.29 ± 0.3 | 0.85 ± 0.8 | 2.14 * | |
| SCO7069 cbhH | −0.84 ± 0.3 | −3.14 ± 1.4 | 2.29 | 0.46 ± 0.8 | 1.6 ± 0.6 | 1.14 | −0.13 ± 0.9 | 2.93 ± 1.2 | 3.06 * | |
| SCO0481 chiG | −1.32 ± 0.3 | −2.7 ± 0.4 | 1.38 * | −0.37 ± 0.1 | 1.94 ± 0.6 | 2.31 ** | 0.49 ± 1.5 | 1.96 ± 0.9 | 1.49 | |
| SCO2758 nagA | 1.41 ± 0.1 | −0.88 ± 0.3 | 2.29 ** | 0.2 ± 0 | −2.03 ± 0.1 | 2.23 *** | −0.89 ± 0.4 | 1.49 ± 0.7 | 2.39 * | |
| SCO6712 laccase | −0.74 ± 0.4 | 0.63 ± 0.2 | 1.36 * | −0.88 ± 0.1 | −0.97 ± 0.5 | 0.09 | 1.1 ± 0.6 | 0.3 ± 1 | 0.8 | |
| Core | SCO4284 NAG-6-P deacetylase | 0.27 ± 0.2 | −0.92 ± 0.1 | 1.19 *** | 0.03 ± 0.2 | 0.67 ± 1 | 0.64 | −0.93 ± 0.3 | 0.88 ± 0.3 | 1.8 ** |
| Metabolism | SCO5236 nagB | 0.88 ± 0.1 | −0.02 ± 0.1 | 0.9 *** | −0.86 ± 0.1 | 0.47 ± 0.3 | 1.33 ** | −1.04 ± 0.2 | 1.14 ± 0.3 | 2.17 ** |
| SCO6535 narG | −1.34 ± 0.5 | −2.01 ± 0.1 | 0.67 | 0.6 ± 1 | 1.03 ± 1 | 0.43 | −0.44 ± 0.8 | 2.17 ± 0.5 | 2.61 * | |
| SCO2487 nirB | −1.9 ± 0.3 | −1.01 ± 1.6 | 0.89 | −0.59 ± 0.5 | 2.03 ± 0.7 | 2.62 ** | −0.55 ± 0.9 | 1.72 ± 0.6 | 2.27 * | |
| SCO2488 nirC | −1.29 ± 0.3 | −2.16 ± 0.3 | 0.87 * | −0.33 ± 0 | 1.14 ± 0.6 | 1.47 * | −0.44 ± 0.1 | 2.05 ± 0.9 | 2.49 * | |
| SCO4683 gdhA | −0.08 ± 0.2 | 0.34 ± 0.1 | 0.42 * | −0.94 ± 0.8 | −0.57 ± 0.5 | 0.37 | −1.06 ± 0.6 | 1.02 ± 0.4 | 2.09 * | |
| SCO1942 pgi | 0.16 ± 0.1 | 0.61 ± 0.2 | 0.46 * | −0.78 ± 0.7 | 0.22 ± 0.1 | 1 | −0.14 ± 1 | 0.38 ± 0.5 | 0.52 | |
| SCO5624 rpsB | −0.02 ± 0.2 | 1.1 ± 0.1 | 1.08 ** | 0.27 ± 0.2 | 0.71 ± 0.2 | 0.44 | −1.3 ± 0.6 | −0.4 ± 0.1 | 0.9 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finn, D.; Catton, K.; Heenan, M.; Kopittke, P.M.; Ouwerkerk, D.; Klieve, A.V.; Dalal, R.C. Differential Gene Expression in the Model Actinomycete Streptomyces coelicolor A3(2) Supports Nitrogen Mining Dependent on the Plant Carbon to Nitrogen Ratio. Agriculture 2018, 8, 192. https://doi.org/10.3390/agriculture8120192
Finn D, Catton K, Heenan M, Kopittke PM, Ouwerkerk D, Klieve AV, Dalal RC. Differential Gene Expression in the Model Actinomycete Streptomyces coelicolor A3(2) Supports Nitrogen Mining Dependent on the Plant Carbon to Nitrogen Ratio. Agriculture. 2018; 8(12):192. https://doi.org/10.3390/agriculture8120192
Chicago/Turabian StyleFinn, Damien, Kerrilyn Catton, Marijke Heenan, Peter M. Kopittke, Diane Ouwerkerk, Athol V. Klieve, and Ram C. Dalal. 2018. "Differential Gene Expression in the Model Actinomycete Streptomyces coelicolor A3(2) Supports Nitrogen Mining Dependent on the Plant Carbon to Nitrogen Ratio" Agriculture 8, no. 12: 192. https://doi.org/10.3390/agriculture8120192
APA StyleFinn, D., Catton, K., Heenan, M., Kopittke, P. M., Ouwerkerk, D., Klieve, A. V., & Dalal, R. C. (2018). Differential Gene Expression in the Model Actinomycete Streptomyces coelicolor A3(2) Supports Nitrogen Mining Dependent on the Plant Carbon to Nitrogen Ratio. Agriculture, 8(12), 192. https://doi.org/10.3390/agriculture8120192






