1. Introduction
European agriculture is currently facing the problem of the accumulation of large amounts of slurry and manure, particularly in regions with high livestock densities, for example northwest Germany, Flanders and the Netherlands. Slurry and manure contain considerable amounts of important plant nutrients, including phosphorus (P) and nitrogen (N). It has been estimated that if the Netherlands applied its manure up to the allowed amount of phosphate on all its agricultural land, in 2015 there would have still been excess manure containing 40–60 million kg of phosphate [
1]. Dealing with these manure and nutrient excesses is becoming an increasingly urgent challenge, and is heightened by the trend towards larger farm sizes as a consequence of increasing economic pressure. Manure storage is not only cost-intensive but is also associated with nutrient losses [
2], leading to environmental problems such as air pollution (gaseous N emissions in the form of ammonia and nitrous oxide) and groundwater contamination (nitrate leaching).
Today, large livestock producers often buy a substantial proportion of their animal feed instead of growing it on their own farm. Most protein feed used in Europe, for example, is soybean meal, which has to be imported from South America
. Farmers are no longer limited by regional feed supply and availability of arable land. Nutrients are imported along with the feed and remain in surplus on the farm within the manure. The livestock farms have too small a land area for the environmentally friendly field application of the accumulating nutrient load without exceeding the legal limits set by the European Union (EU) Nitrates Directive [
3] and the EU Water Framework Directive [
4]. Consequently, manure is considered a waste rather than a valuable resource. The situation is aggravated by the lack of regionally available, environmentally sound manure treatment solutions and the high costs of storage and disposal. As an example, Dutch farmers pay between €5 and €20 per tonne for the transport of surplus manure to other locations within the Netherlands [
5] or even abroad.
By contrast, in other regions nutrients are needed—for example, at sites where arable farming is predominant and animal feed is produced for export. However, the high water content (>90%, [
6]) makes long-distance transportation of manure neither profitable nor ecological. As a consequence, soil organic matter contents are depleted at these sites and nutrient deficits replaced through synthetic (N) or mineral (P, and potassium, K) fertilizers [
7], which considerably interferes with the global P cycle [
8].
Synthetic N fertilizers are mainly produced through the Haber–Bosch process. This process uses N from the air (thus unlimited in availability), but also consumes high amounts of natural gas and energy [
9]. In contrast, mineral fertilizers are mainly derived from fossil resources and are, as such, limited. This is especially true for fossil P sources.
As a vital component of DNA and ATP, P is essential for all living organisms. Thus, it is one of the main nutrients needed for crop nutrition. The goal of achieving food security for a growing world population, the increasing use of biomass for biofuel production and the progressive degradation of arable land have all led to P fertilizer becoming more important for agricultural production than ever before.
In 2013/14, annual phosphate fertilizer consumption in Germany was 284,000 t [
10]. In 2011, total EU phosphate consumption (fertilizer and industrial use) stood at approx. 4.6 million t per year. This represents 10% of global phosphate demand [
11].
Phosphate fertilizer used in agriculture is mainly produced from rock phosphate (RP). However, RP is a finite resource, as with all mined resources. For this reason, in 2014, the EC added it to the list of critical raw materials [
11]. Contrary to assertions in previous studies, there are still sufficient supplies of RP, but its extraction is very complex and not (yet) economically viable [
12]. In addition, mined RP is increasingly contaminated by uranium and cadmium [
13]. As 82% of the phosphorus extracted is used for fertilizers, these pollutants end up in the environment [
11].
For this reason, prudent management of available P resources is of paramount importance. Exploiting “fresh” RP resources is one option. Another is the recycling of already “exploited” P, for example from livestock manure.
Livestock manure contains highly plant-available forms of P (inorganic) and N (ammonium) [
14]. As such, it is a valuable organic fertilizer and a promising resource for P and N recovery. The manure excreted in EU-27 every year contains 1.8 million t of P, which corresponds to 150% of the amount of P used annually in fertilizers in Europe [
2]. Thus, P recovery from manure could theoretically more than meet the entire demand for P fertilizer in Europe—providing the fertilizing effect of the recovered product is comparable.
The EC-funded research project “BioEcoSIM” (“An innovative bio-economy solution to valorise livestock manure into a range of stabilised soil improving materials for environmental sustainability and economic benefit for European agriculture”; grant No. 308637) has succeeded in developing an innovative technology at pilot-scale to recover P and N from pig manure. In a first step, manure is pretreated, so that the P completely dissolves. Subsequently, the manure is separated into a solid and a liquid fraction. The solid fraction is dried and then pyrolyzed to biochar. The P is recovered from the liquid fraction by precipitation and filtered off as a mixture of calcium phosphate (hydroxyapatite), magnesium phosphate and magnesium ammonium phosphate (MAP, struvite). The raw manure contains sufficient magnesium (1.7% dry matter) to allow struvite formation; no additional magnesium source is necessary. In this study, the obtained product is referred to as phosphate salts or “P-Salt”.
This innovative technology has several advantages. It contributes to an environmentally friendly solution to the problem of manure disposal. It addresses the unfavourable nutrient ratio of manure, which often leads to an oversupply of P, as the amount of manure used in fertilization is usually calculated based solely on its N content. This also avoids the accompanying negative environmental consequences, such as P accumulation in soil, surface runoff and eutrophication of waterbodies. As the nutrients P and N are recovered separately, they can be used to create customized fertilizers as transportable and marketable products. This allows the fertilization of crops according to their respective requirements and the balancing of disrupted nutrient cycles. The technology could also reduce the EU’s dependency on P imports. The improvement in P-use efficiency could help to conserve fossil P resources and reduce energy consumption in mining.
Struvite has been shown to be a highly effective, slow-releasing P fertilizer [
15,
16]. Several studies have found that struvite recovered from different materials can improve the yields of various crops compared to untreated controls [
17,
18,
19]. Struvite recovered from swine wastewater has been shown to increase the biomass yield of maize more than commercial P fertilizer [
20].
However, the plant availability of P in recovered products is often low, or at least unpredictable [
21]. The assessment of fertilizers based on analytical results alone is not sufficient, because the predicted and actual availability and uptake of P by plants can differ substantially. Johnston and Richards [
22] as well as Römer [
23] confirmed that some P fertilizers ensure relatively good P availability and supply despite the small amounts contained in water-soluble form. Cabeza et al. [
17] concluded that the dissolution of P in soil is a much more accurate indicator of the fertilizing effectiveness of recycled P products than their solubility in water or citric acid. Thus, plant experiments are crucial to evaluate the actual efficacy of the P-Salt in terms of P-fertilizing performance.
Biochar is produced from the solid manure fraction in the BioEcoSIM process and can serve as a potential soil improver. Biochar made from different substrates was reported to have beneficial effects on crop yield, soil quality and soil biological activity [
24]. It can be used as an amendment to increase the water and nutrient retention capacity of light soils [
25,
26], thus aiding the sustainable production of food, feed and energy crops on progressively degrading soils—one measure to help meet the demand of an increasing world population. It also functioned as a means of carbon sequestration in soil [
27,
28] and has been shown to contribute to the mitigation of greenhouse gas emissions [
29,
30]. However, the use of biochar as a soil-improving substance is controversial and some studies have found biochar application to have no effect or even adverse effects on crop yield [
31,
32]. A meta-analysis review concluded that biochar application had a small, but statistically significant influence on crop productivity [
33]. In this study, the biochar produced is used together with the recovered P-Salt, underlining the integrated concept of the project.
The combined application of P-Salt and biochar recovered from the same material has not been tested before. Based on results from the use of biochar in combination with conventional fertilizer [
34,
35,
36], we assume that biochar prevents the leaching of nutrients contained in the P-Salt and increases crop yield. Biochar application may promote root development [
37] through improved soil structure, resulting in more efficient nutrient uptake from the P-Salt and thus better crop development [
38].
There are only a few studies [
15,
39,
40] on the use of P fertilizer recovered from pig manure that used a comparable technique and none of these tested and compared its fertilizing effect on different crop types.
For that reason, this study aimed to test the fertilizing effect of the manure-based P-Salt on two crop types and assess its competitiveness with conventional superphosphate. A further objective was to determine whether the combined application of P-Salt and biochar improves the fertilizing effect through synergy effects. A third objective was to assess whether there are differences in the uptake efficiency of recovered and synthetic nutrients between different crop types.
Based on these objectives, the following hypotheses were set up for the study:
P-Salts recovered as struvite from pig manure work equally well as or better than mineral P fertilizer.
There is a synergetic effect/an interaction between P-Salt and biochar application with regard to improved soil productivity and biomass yield.
Different crop types (cereals/legumes) react differently to P-Salt treatment, and this is also influenced by soil.
These hypotheses were tested by means of pot experiments with spring barley and faba beans. However, an important prerequisite for the use of novel products (in this case P-Salt and biochar) as fertilizers is that they do not have any undesirable effects on plants or soil biota. For this reason, a comprehensive chemical analysis and two bioassays were carried out on the products prior to the pot experiments.
2. Materials and Methods
The experimental part of this study included (1) the comprehensive determination of the chemical composition of P-Salt and biochar; (2) two bioassays to detect any eco-toxic effects on seed germination and crop development; and (3) two pot experiments to assess the fertilizing and soil-improving performance of the products.
This three-stage approach enabled detection of both desired and undesired impacts of the products on plants and soil biota at an early stage of the research project and, if necessary, the adaptation of the production process towards ecologically sound fertilizer products. Manure does not usually contain excessive amounts of problematic substances, such as heavy metals or organic pollutants. The bioassays were performed to determine whether these contaminants are concentrated in the products during the recovery process and to ensure that they do not affect crops.
2.1. Chemical Characterization
The P-Salt used in this study is a complex of struvite, magnesium phosphate and calcium phosphate obtained via the BioEcoSIM process. Pig manure was collected at a farm in Kupferzell (Germany). It was acidified with sulfuric acid to pH 5 and subsequently separated by coarse filtration into a solid and a liquid fraction. The solid fraction was dried and pyrolyzed in a superheated steam atmosphere (45 min at 450 °C). The P-Salt was recovered from the liquid manure fraction by precipitation and then filtered off. It serves as a potential source of P, but also contains N (
Table 1). Contents of additional macro- and micronutrients as well as heavy metals are provided in
Table A1.
2.2. Toxicity Studies
Preliminary testing in petri dishes showed the germination capacity of barley to be 98% and that of faba beans to be 100%.
Two bioassays were then carried out on the P-Salt and biochar to detect any inhibiting effects on seed germination and early crop growth (
Table 2 and
Table 3). Both tests employed a direct exposure approach. The P-Salt and biochar were applied to cress and barley at five different levels. The P-Salt applications ranged from 50% to 200% of the optimal P supply (=100%) of 150 mg P per kg substrate. The biochar application rates were calculated based on mass percentage of the cultivation substrate, not nutrient content. Both products were mixed with the substrate and filled into pots. The cress seeds were sown on top of the substrate and lightly covered. The barley seeds were sown at a depth of approximately 1 cm. The pots for the germination test were placed in a climate chamber and taken out regularly to count the number of germinated seeds. The pots for the growth test were placed on tables in a greenhouse. At the end of the test, the crops were cut 0.5 cm above the soil surface, weighed and dried at 60 °C for 48 h. Dry weight was determined and dry matter content calculated.
2.3. Pot Experiments
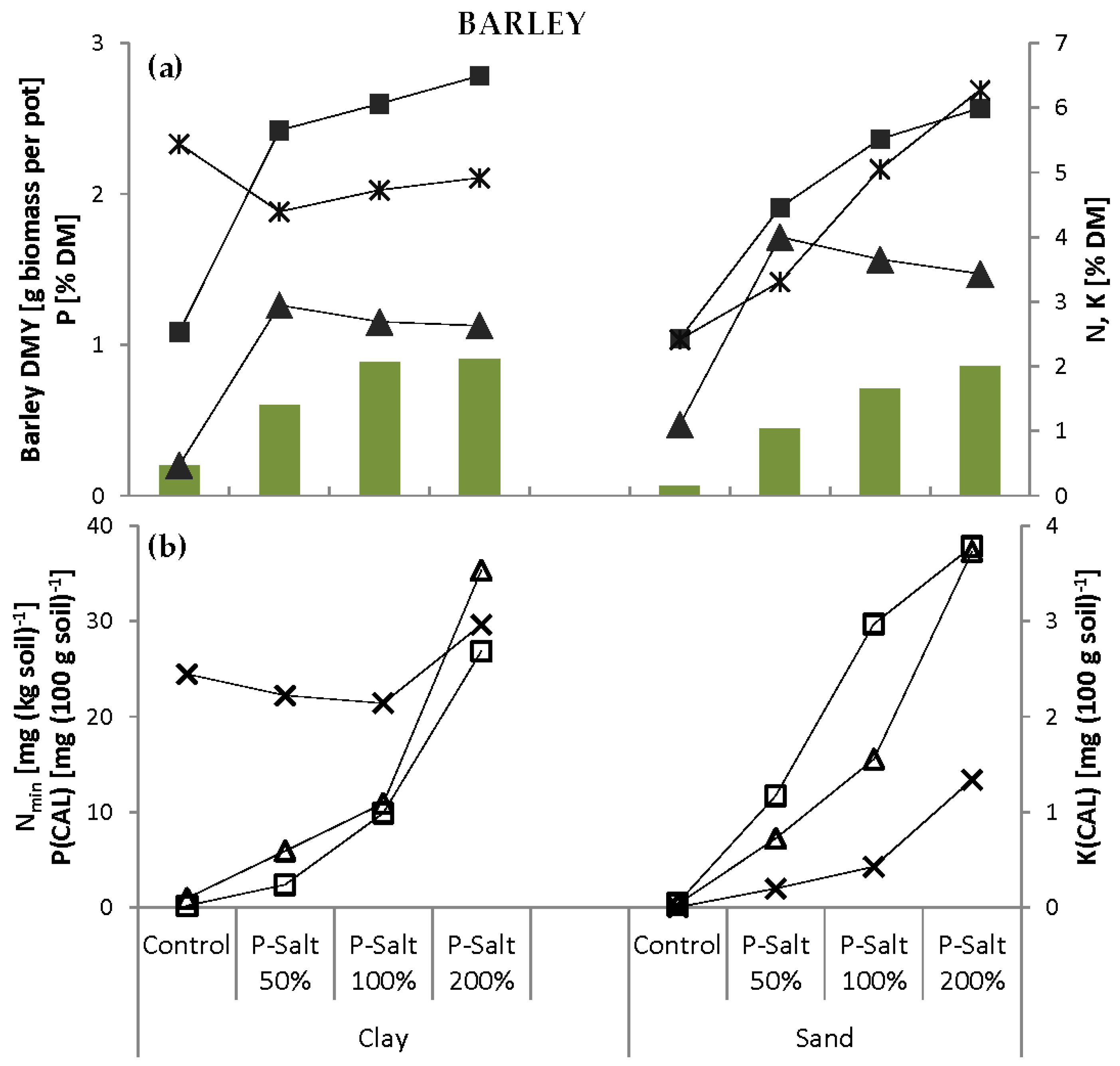
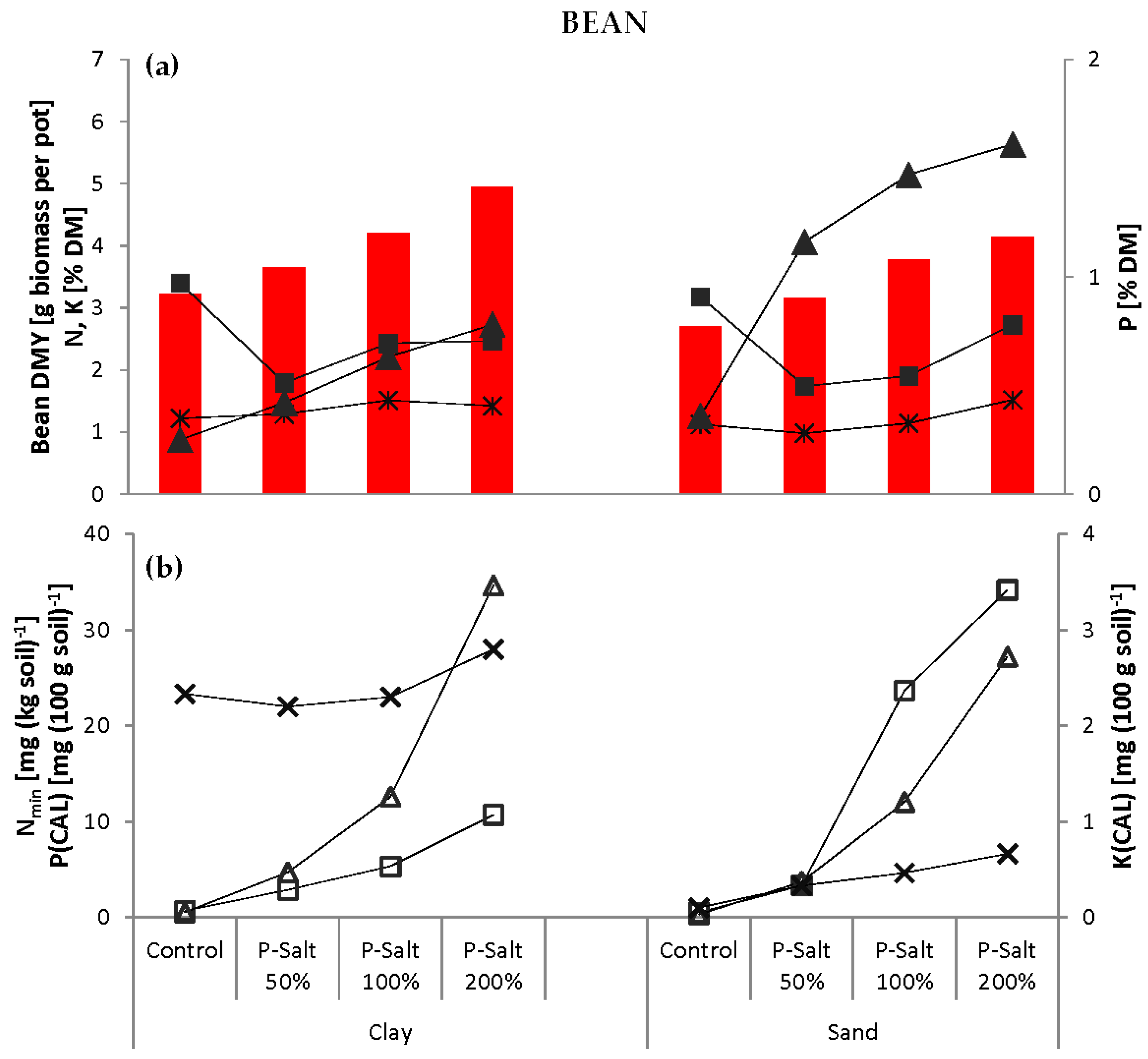
The pot experiments were carried out using two soil substrates. Clay and sand were chosen due to their low concentration and plant availability of P. The P content measured by calcium-acetate-lactate extraction (P(CAL)) in both soils is classified as very low according to Association of German Agricultural Analytic and Research Institutes (VDLUFA,
Table 4). Additionally, the clay soil had a high phosphate immobilization potential due to a high concentration of carbonates. The N mineralization potential was low in both soils. Both soils were of low fertility and thus not representative of agricultural soils. The clay soil had good water retention properties, but became very hard when dry and warmed only slowly. The sand soil had zero water retention capacity; water immediately flowed to the bottom of the pots.
The two soils were mixed with varying amounts of P-Salt, P-Salt in combination with biochar or conventional fertilizer (
Table 5). The application rates of the P-Salt were calculated based on its total P content. Optimal P supply was defined as 150 mg total P per kg·soil [
41], i.e., 0.225 g P or 4.5 g P-Salt pot
−1, and is referred to as 100%. A reduced dose (50%) to simulate nutrient shortage and an elevated dose (200%) were included. Levels higher than 200% were not considered reasonable and thus not tested.
The performance of the P-Salt was compared to conventional mineral fertilization with ammonium nitrate NH
4NO
3 (35% N) and calcium dihydrogen phosphate Ca(H
2PO
4)
2, (24.6% P). Mineral N and P were applied in the same amount as in the P-Salt (
Table 5). Other main plant nutrients (K, Mg, Ca) and trace elements were not considered in this experiment.
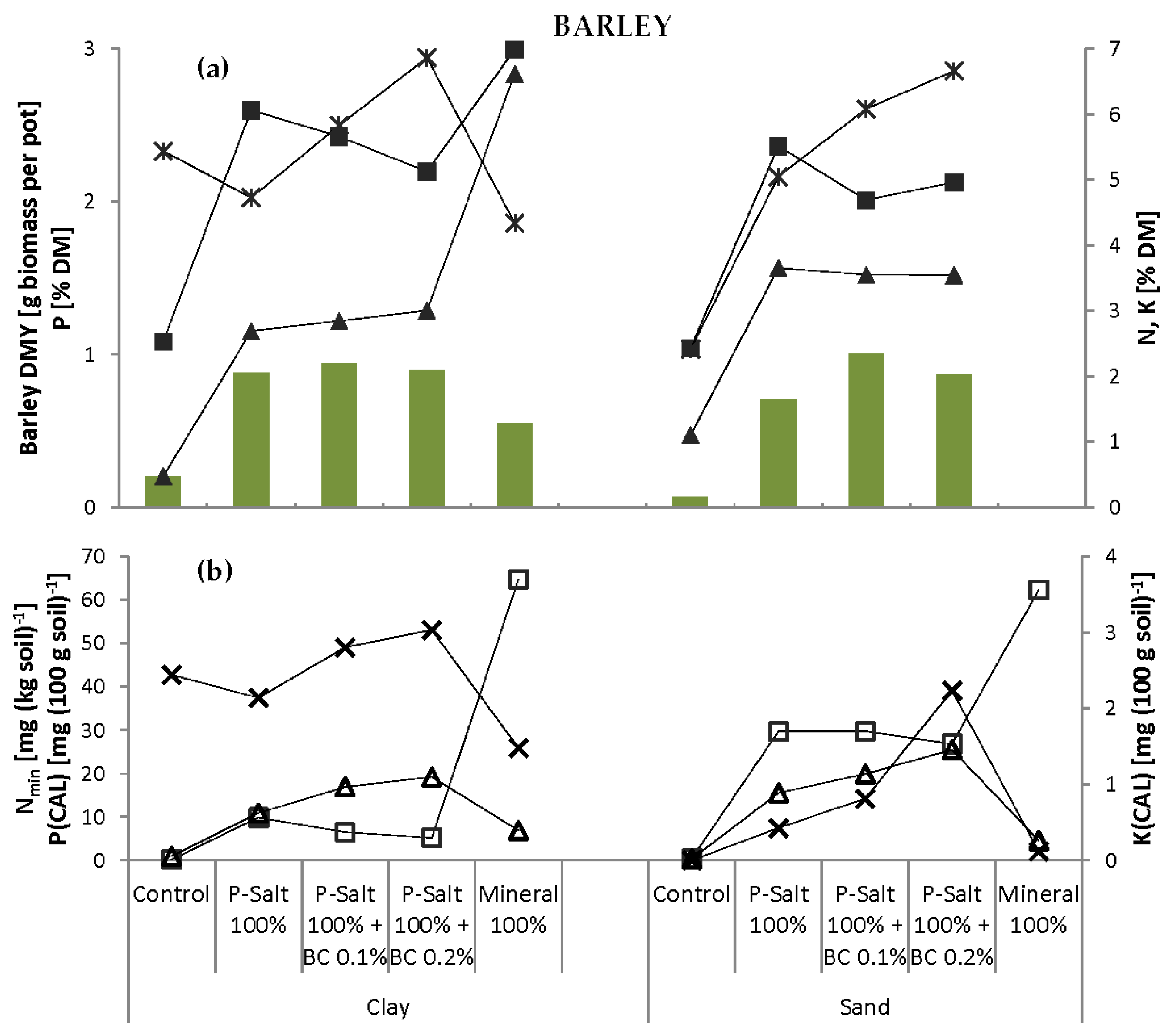
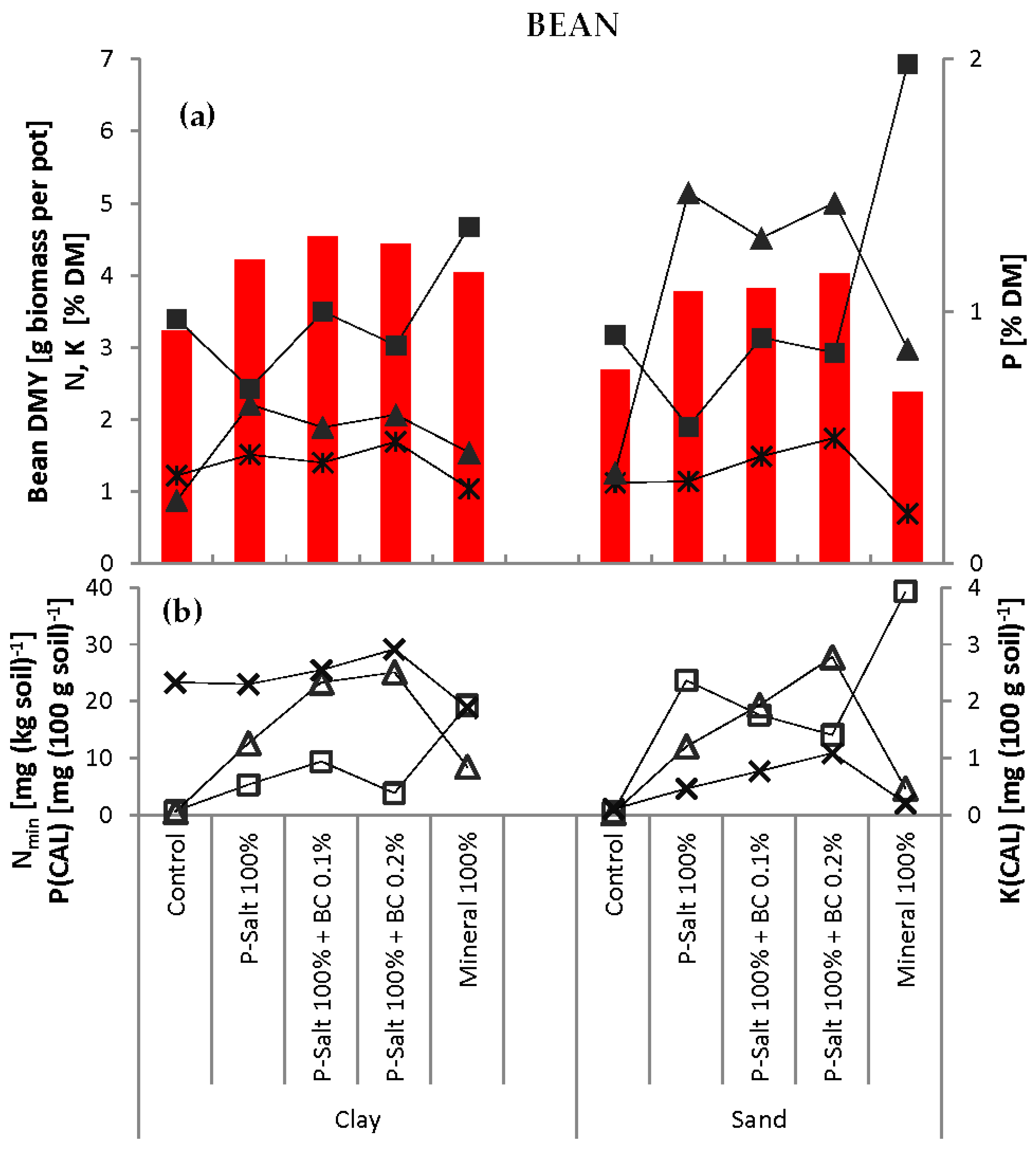
Biochar (BC) was applied in two concentrations (0.1% and 0.2%, equivalent to 1.5 and 3.0 g·pot
−1) in combination with the 100% level of P-Salt (
Table 5). The experiment also included control pots that remained completely unfertilized. The pot experiments were carried out first with barley, then with faba beans, and with both soils for each test crop.
The required amounts of P-Salt and biochar were mixed thoroughly with 1.5 kg·soil and filled into polypropylene pots (13 × 13 × 13 cm3, Goettinger). The conventional fertilizers (analytical grade NH4NO3 and Ca(H2PO4)2) were dissolved in water to ensure exact dosage of the small amounts and then added to the soil. Pots were initially watered with 300 mL deionized water each.
The prepared pots were sown with either ten seeds of spring barley (Hordeum vulgare L. var. ‘Grace’) or eight seeds of faba bean (Vicia faba L. var. minor var. ‘Isabell’). All pots were set up on a table in a greenhouse with no additional lighting in a randomized complete block design with four replications. After germination, plants were reduced to five per pot. The pots were watered from above with deionized water when necessary to keep the moisture near field capacity. Any leachates were collected and returned to the pots. Air temperature in the greenhouse was approx. 20 °C during the day and 16 °C at night.
The barley plants were treated once against powdery mildew with a combination of propiconazol, tebuconazol and fenpropidin. The bean plants were sprayed once against black bean aphids with Lambda-Cyhalothrin. Both treatments were carried out according to the manufacturer’s (Syngenta Agro GmbH, Maintal, Germany) instructions for the respective crop.
After six weeks (barley BBCH 29/31, faba beans BBCH 39/51), the shoots were cut 0.5 cm above the soil surface, weighed and then dried at 60 °C for 48 h. Dry weight was determined and dry matter content calculated. Soil samples were taken from each individual pot. Roots were washed and dried at 60 °C for 48 h to determine the root dry weight.
2.4. Sample Analyses
The dried shoots were ground in a mixer mill (duration 40 s, frequency 30 min−1; Retsch GmbH, Haan, Germany). Total N concentration in the biomass was determined according to Dumas (DIN EN 13654-2). Concentrations of P, K, Ca and Mg were determined using microwave digestion followed by ICP-OES measurement (DIN EN ISO 11885). All samples were analysed in duplicate. Plant P uptake was calculated from dry matter yield (DMY) and P concentration.
The soil samples were used to determine plant-available N (NO3 and NH4; referred to as Nmin) in fresh soil using CaCl2 extraction followed by FIA (Flow injection analysis) measurement (DIN ISO 14255:1998-11). Plant-available P and K were then determined in air-dried soil using CAL extraction followed by flame photometer or FIA measurement, respectively (OENORM L 1087:2012-12-01). Soil pH was measured using a glass electrode after CaCl2 extraction (DIN ISO 10390:2005).
2.5. Statistical Analysis
Data analysis was performed using SAS software version 9.3 PROC MIXED (SAS Institute Inc., Cary, NC, USA). Soil and treatment as well as their interaction were handled as fixed effects with DMY and nutrients in plant and soil samples as dependent variables. Data were log transformed where necessary. The graphs shown here were plotted with untransformed data. As large differences in biomass development were expected for the two soils, the treatments were compared separately for each soil. The level of significance was α = 0.05. Standard errors (SE) given in tables were calculated as pooled standard error of the mean.
5. Conclusions and Recommendations
This study explored the potential use of a P-Salt recovered from pig manure as a replacement for conventional mineral P fertilizer.
The P-Salt was found to have the same or even better effects than mineral fertilizer on growth in both crops in both soils. Thus, firstly, the recovered product can replace conventional mineral P in terms of the fertilizing effect for the two crop types tested here. Secondly, and perhaps more importantly, the demand for P fertilizer in European agriculture could theoretically be met by P recycling from manure alone. Ideally, this would render the extraction of “new” P from rock phosphate for fertilizer production superfluous in the medium to long term.
This study did not consider the potential fertilizer replacement value of the P-Salt. Organic products are usually applied in higher amounts in order to compensate for the slower release and lower plant availability of nutrients than with conventional products. If the amounts applied had been adjusted accordingly, the P-Salt would have certainly led to considerably better results than those obtained in this study. In addition, the P-Salt can supply plants and soil with additional microelements and a small amount of organic matter. These aspects render P-Salt recovered from manure by the BioEcoSIM process even more advantageous than conventional fertilizers.
However, the acceptance of such recycled fertilizers by agriculture and horticulture is currently fairly low. One constraint is certainly the reliability of the novel product. The combination of P-Salt and conventional products could serve as a convincing solution for users/farmers: conventional fertilizer provides readily available, water-soluble P in the early growth stages, whereas the slow-releasing P-Salt ensures a continuous supply during the entire growth period. This would allow the entire P fertilizer amount to be administered in one application without the risk of P deficiency in heavy soils with high P immobilization potential (e.g., clay) of water-soluble P. P-Salt also has a strong advantage in light soils with low buffer capacity (e.g., sand) where the slow release of P prevents its leaching or surface runoff. The fertilizing effect of P-Salt can be enhanced by combined application with biochar, which is also a product of the manure recycling process in which P-Salts are extracted.
The results indicate that biochar improves the soil status of sand, suggesting that biochar can be a valuable addition to sandy or degraded soils. However, no significant benefit was seen in the clay soil.
Granulation or pelletizing of finely ground P-Salt and biochar can considerably simplify their handling and turn them into marketable products. A reduction in N content of the P-Salt would avoid the accompanying N application, thus increasing flexibility. The next steps will be a detailed assessment of how the properties of the raw manure influence the emerging products and validation of the presented findings in field-scale experiments.