Aflatoxins, Fumonisins and Zearalenone Contamination of Maize in the Southeastern and Central Highlands Provinces of Vietnam
Abstract
:1. Introduction
2. Results and Discussion
| Samples | Aflatoxins | Fumonisins | Zearalenone | ||||||
|---|---|---|---|---|---|---|---|---|---|
| DN + BP a | DN + DL a | Total | DN + BP a | DN + DL a | Total | DN + BP a | DN + DL a | Total | |
| Analyzed (n) | 47 | 50 | 97 | 47 | 50 | 97 | 47 | 50 | 97 |
| Positive (n) | 29 | 25 | 54 | 26 | 39 | 65 | 5 | 22 | 27 |
| Positive (%) | 61.7 | 50.0 | 55.7 | 55.3 | 78.0 | 67.0 | 10.6 | 44.0 | 27.8 |
| Mycotoxins | Region | DN + BP a | DN + DL a | Prob. | |
|---|---|---|---|---|---|
| AFB1 b | n | 29 | 25 | ||
| Mean | 104.7 | 170.2 | 0.221 | ||
| Median | 81 | 28 | |||
| Range | 1–450 | 2–844 | |||
| AFB2 b | n | 25 | 15 | ||
| Mean | 10.5 | 20.7 | 0.071 | ||
| Median | 5.5 | 11 | |||
| Range | 1–33 | 2–86 | |||
| AFG1 b | n | 5 | 4 | ||
| Mean | 12.8 | 208 | 0.306 | ||
| Median | 2 | 9.5 | |||
| Range | 2–54 | 3–810 | |||
| FB1 b | n | 26 | 39 | ||
| Mean | 740 | 1757 | 0.042 * | ||
| Median | 390.5 | 871 | |||
| Range | 102–2747 | 160–10799 | |||
| FB2 b | n | 20 | 35 | ||
| Mean | 357.9 | 751 | 0.102 | ||
| Median | 229.5 | 367 | |||
| Range | 108–834 | 102–5051 | |||
| ZEA b | n | 5 | 22 | ||
| Mean | 48.8 | 299.5 | 0.367 | ||
| Median | 44 | 98 | |||
| Range | 35–80 | 36–2409 |
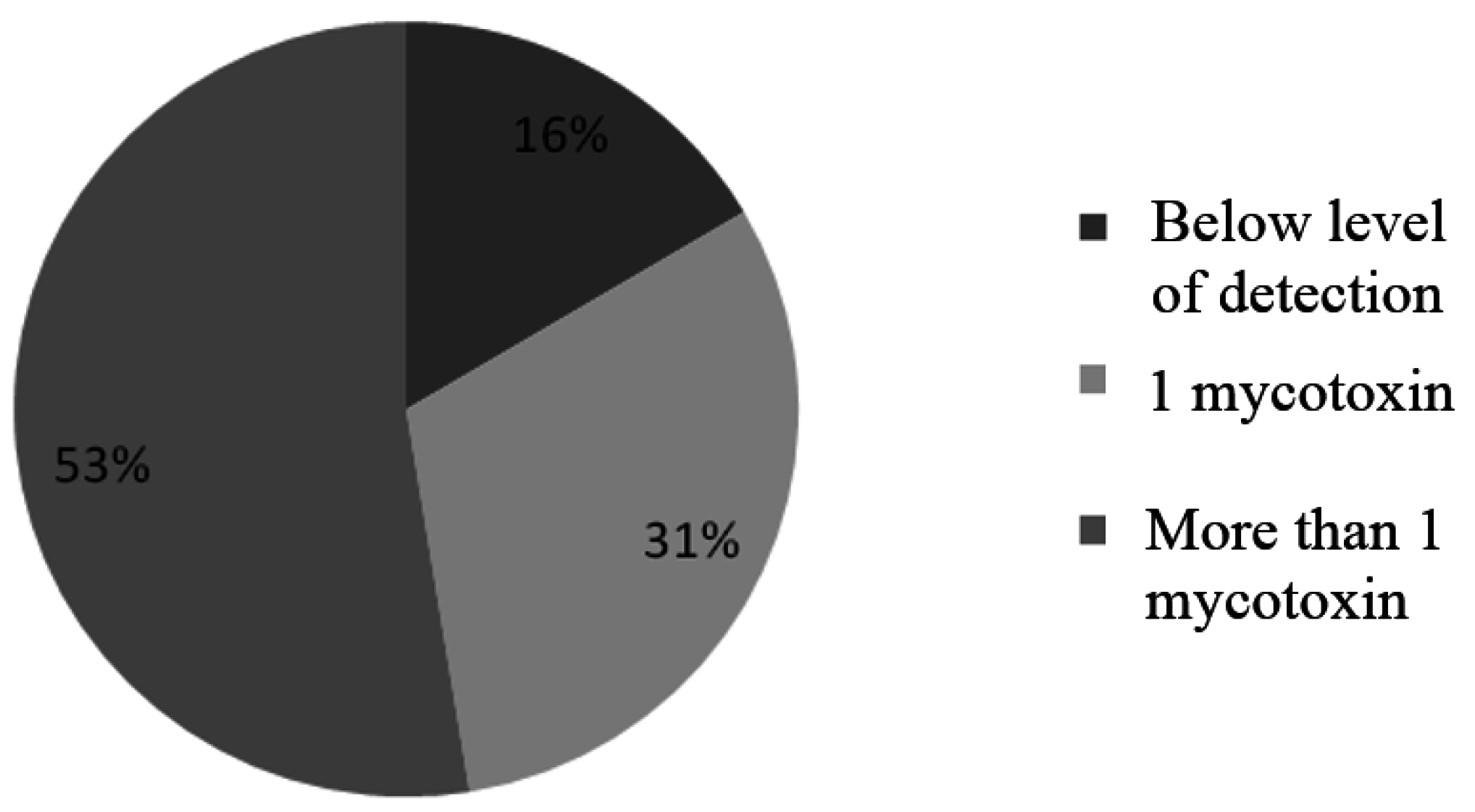
| Aflatoxins (μg/kg) | Frequency | % of Samples |
|---|---|---|
| 0 ≤ 20 | 17 | 31.5 |
| 21–100 | 14 | 25.9 |
| 101–300 | 15 | 27.8 |
| >300 | 8 | 14.8 |
| Fumonisins (μg/kg) | Frequency | % of Samples |
|---|---|---|
| 0 ≤ 4000 | 59 | 90.8 |
| 4001–8000 | 3 | 4.6 |
| 8001–12,000 | 1 | 1.5 |
| >12,000 | 2 | 3.1 |
| Zearalenone (µg/kg) | Frequency | % of Samples |
|---|---|---|
| 0 ≤ 100 | 17 | 63.0 |
| <100–500 | 7 | 25.9 |
| 500–1000 | 1 | 3.7 |
| >1000 | 2 | 7.4 |
3. Experimental Section
3.1. Site Description
3.1.1. Dong Nai and Binh Phuoc—Warm Region
3.1.2. Dak Lak and Dak Nong—Cool Region
3.2. Sampling Method
3.3. Mycotoxin Analysis Methods
3.4. Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Richard, J.L. Some major mycotoxins and their mycotoxicoses—An overview. Int. J. Food Microbiol. 2007, 119, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Bryden, W.L. Mycotoxins and mycotoxicoses: Significance, occurrence and mitigation in the food chain. In General and, Applied Toxicology, 3rd ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2009; pp. 3529–3553. [Google Scholar]
- CAST. Mycotoxins: Risks in Plant, Animal, and Human Systems; Council for Agricultural Science and Technology: Ames, IA, USA, 2003. [Google Scholar]
- Pettersson, H. Controlling mycotoxins in animal feed. In Mycotoxins in Food; Magan, N., Olsen, M., Eds.; Woodhead Publishing: Cambridge, UK, 2004; pp. 262–304. [Google Scholar]
- Abbas, H.K. (Ed.) Aflatoxin and Food Safety; CRC Press: Boca Raton, FL, USA, 2005; pp. 13–28.
- Reddy, K.R.N.; Abbas, H.K.; Abel, C.A.; Shier, W.T.; Oliveira, C.A.F.; Raghavender, C.R. Mycotoxin contamination of commercially important agricultural commodities. Toxin Rev. 2009, 28, 154–168. [Google Scholar] [CrossRef]
- Voss, K.A.; Smith, G.W.; Haschek, W.M. Fumonisins: Toxicokinetics, mechanism of action and toxicity. Anim. Feed Sci. Tech. 2007, 137, 299–325. [Google Scholar] [CrossRef]
- Gelineau-van Waes, J.; Starr, L.; Maddox, J.; Aleman, F.; Voss, K.A.; Wilberding, J.; Riley, R.T. Maternal fumonisin exposure and risk for neural tube defects: Mechanisms in an in vivo mouse model. Birth Defects Res. A Clin. Mol. Teratol. 2005, 73, 487–497. [Google Scholar] [CrossRef] [PubMed]
- Marasas, W.O. Fumonisins: History, world-wide occurrence and impact. In Fumonisins in Food; Jackson, L., DeVries, J., Bullerman, L., Eds.; Springer US: New York, NY, USA, 1996; Volume 392, pp. 1–17. [Google Scholar]
- CTA. Mycotoxins in Grain; CTA: Wageningen, The Netherlands, 1997. [Google Scholar]
- Soriano, J.M.; Dragacci, S. Occurrence of fumonisins in food. Food Res. Int. 2004, 37, 985–1000. [Google Scholar] [CrossRef]
- Binder, E.M.; Tan, L.M.; Chin, L.J.; Handl, J.; Richard, J. Worldwide occurrence of mycotoxins in commodities, feeds and feed ingredients. Anim. Feed Sci. Tech. 2007, 137, 265–282. [Google Scholar] [CrossRef]
- Summerell, B.A.; Leslie, J.F.; Backhouse, D.; Bryden, W.L.; Burgess, L.W. (Eds.) Fursarium: Paul E. Nelson Memorial Symposium; APS Press: St Paul, MN, USA.
- Desjardins, A.E. Fusarium Mycotoxins: Chemistry Genetics and Biology; APS Press: St Paul, MN, USA, 2005. [Google Scholar]
- Fink-Gremmels, J.; Malekinejad, H. Clinical effects and biochemical mechanisms associated with exposure to the mycoestrogen zearalenone. Anim. Feed Sci. Tech. 2007, 137, 326–341. [Google Scholar] [CrossRef]
- Miller, J.D. Mycotoxins in small grains and maize: Old problems, new challenges. Food Add. Contam. A 2008, 25, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Dinh Thao, T.; Tri Khiem, N.; Xuan Trieu, M.; Gerpacio, R.V.; Pingali, P.L. Maize in Vietnam: Production Systems, Constraints, and Research Priorities; CIMMYT: Mexico, D.F., Mexico, 2004. [Google Scholar]
- Rodrigues, I.; Naehrer, K. A three-year survey on the worldwide occurrence of mycotoxins in feedstuffs and feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Miraglia, M.; van Egmond, H.P.; Brera, C.; Gilbert, J. Mycotoxins and Phytotoxins. In IX International Iupac Symposium on Mycotoxins and Phycotoxins; Alaken, Inc.: Fort Collins, CO, USA, 1998. [Google Scholar]
- Trung, T.; Tabuc, C.; Bailly, S.; Querin, A.; Guerre, P.; Bailly, J. Fungal mycoflora and contamination of maize from Vietnam with aflatoxin B1 and fumonisin B1. World Mycotoxin J. 2008, 1, 87–94. [Google Scholar] [CrossRef]
- Silva, C.M.G.; Vargas, E.A. A survey of zearalenone in corn using Romer Mycosep™ 224 column and high performance liquid chromatography. Food Add. Contam. 2001, 18, 39–45. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phuong, N.H.; Thieu, N.Q.; Ogle, B.; Pettersson, H. Aflatoxins, Fumonisins and Zearalenone Contamination of Maize in the Southeastern and Central Highlands Provinces of Vietnam. Agriculture 2015, 5, 1195-1203. https://doi.org/10.3390/agriculture5041195
Phuong NH, Thieu NQ, Ogle B, Pettersson H. Aflatoxins, Fumonisins and Zearalenone Contamination of Maize in the Southeastern and Central Highlands Provinces of Vietnam. Agriculture. 2015; 5(4):1195-1203. https://doi.org/10.3390/agriculture5041195
Chicago/Turabian StylePhuong, Nguyen Hieu, Nguyen Quang Thieu, Brian Ogle, and Hans Pettersson. 2015. "Aflatoxins, Fumonisins and Zearalenone Contamination of Maize in the Southeastern and Central Highlands Provinces of Vietnam" Agriculture 5, no. 4: 1195-1203. https://doi.org/10.3390/agriculture5041195
APA StylePhuong, N. H., Thieu, N. Q., Ogle, B., & Pettersson, H. (2015). Aflatoxins, Fumonisins and Zearalenone Contamination of Maize in the Southeastern and Central Highlands Provinces of Vietnam. Agriculture, 5(4), 1195-1203. https://doi.org/10.3390/agriculture5041195





