Determination of Volatile Compounds in Blossoms of Rosa spinosissima, Rosa pendulina, Rosa gallica, and Their Cultivars
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Identification and Semi-Quantification of Volatile Compounds
2.3. Statistical Analyses
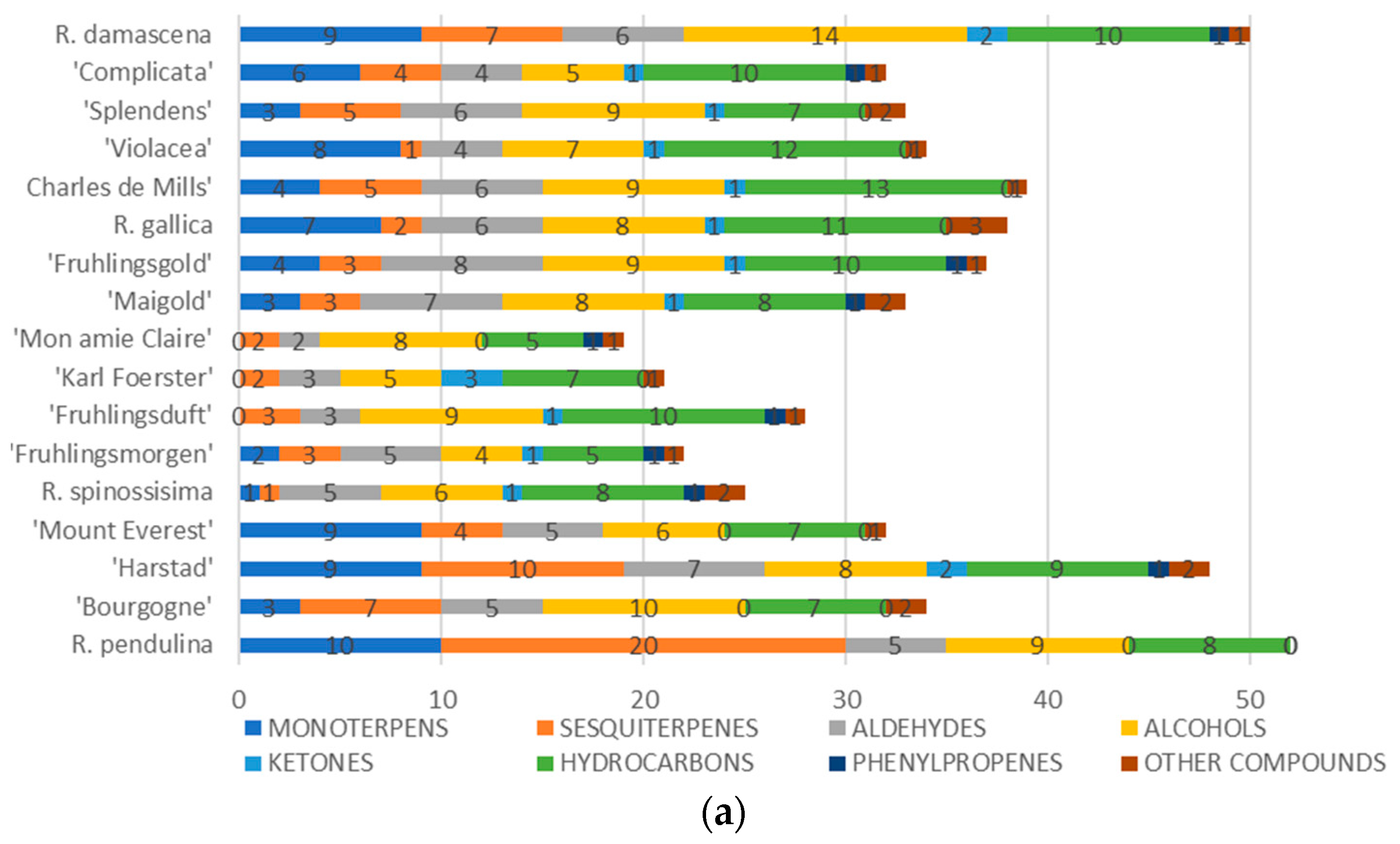
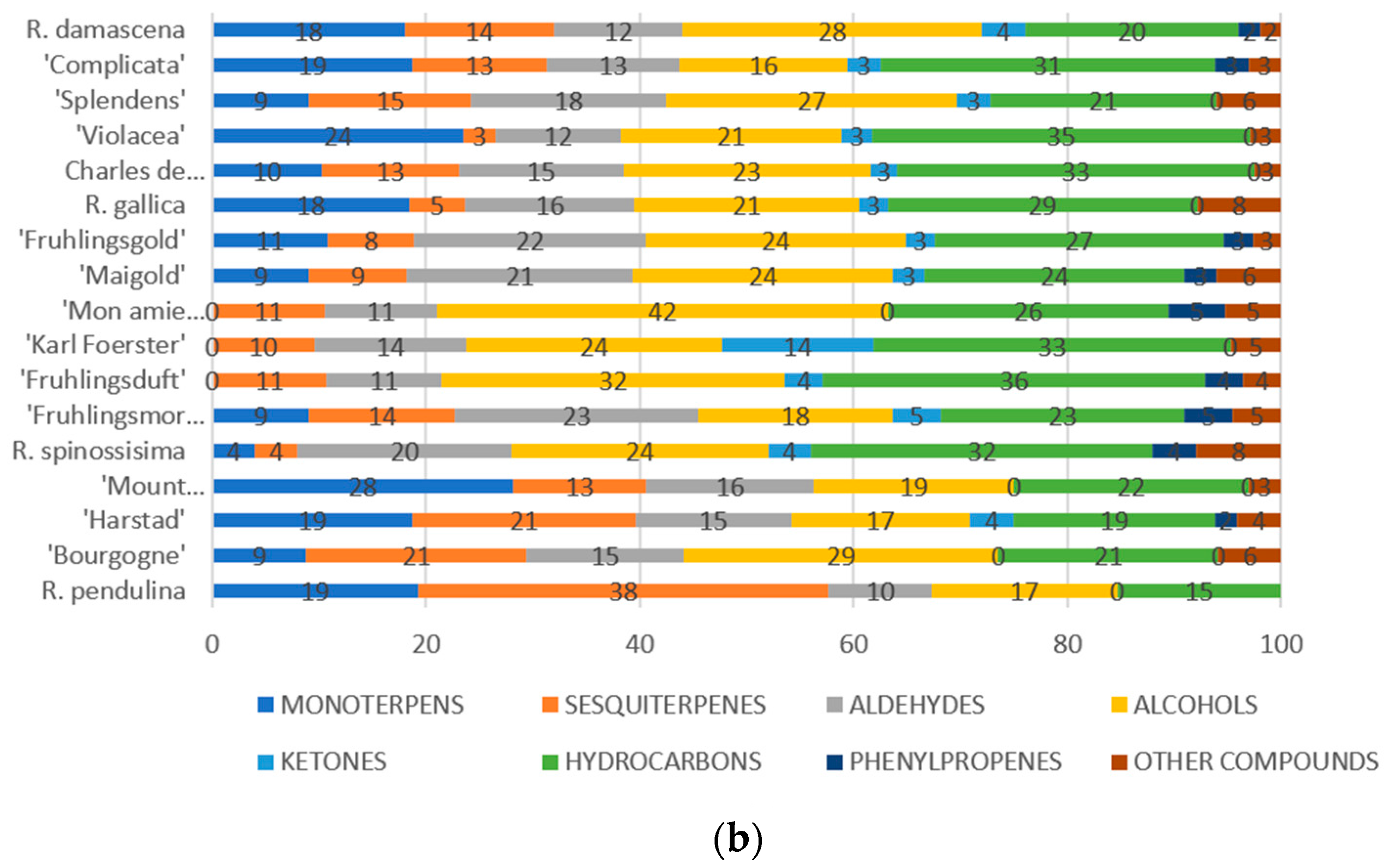
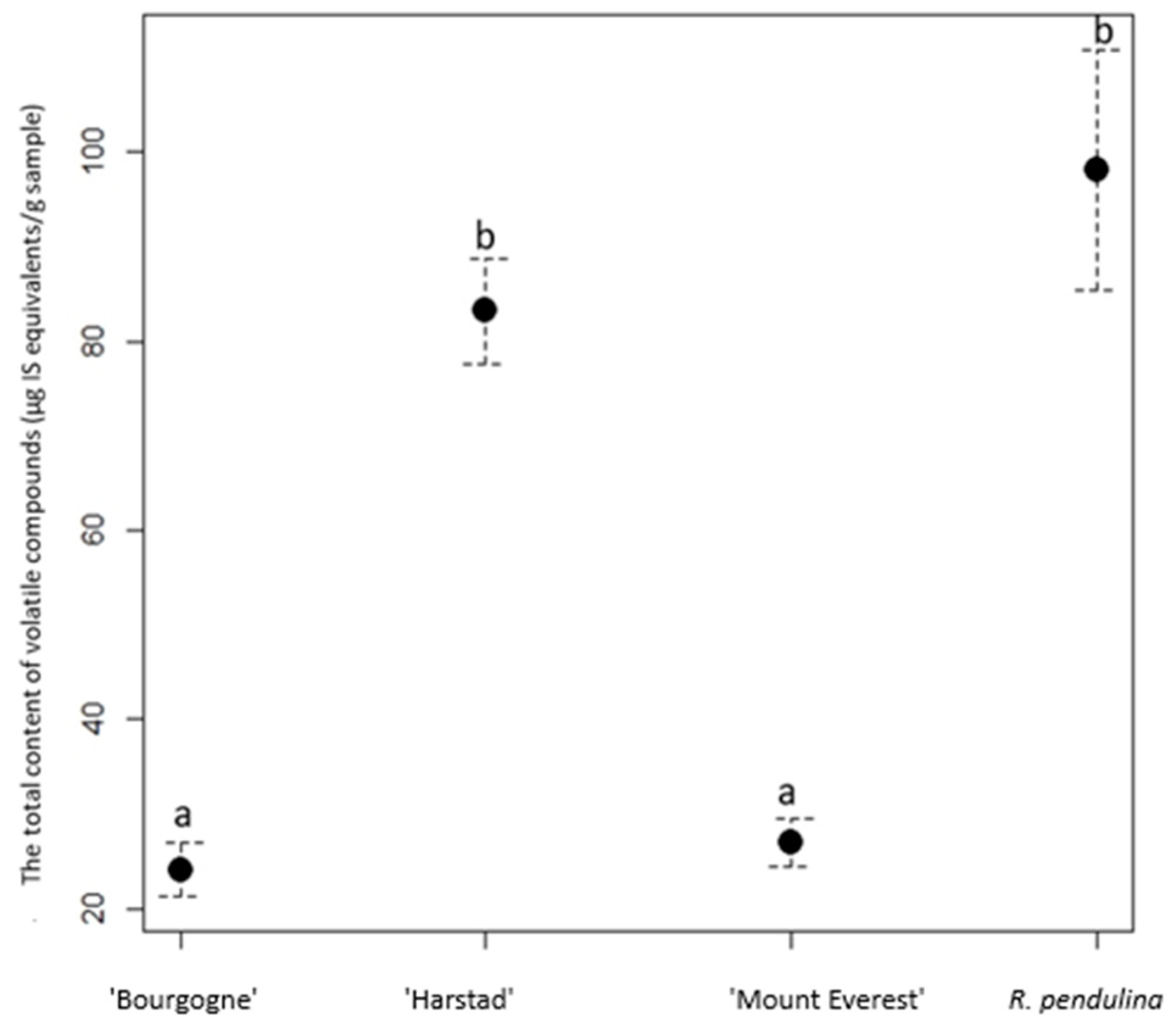
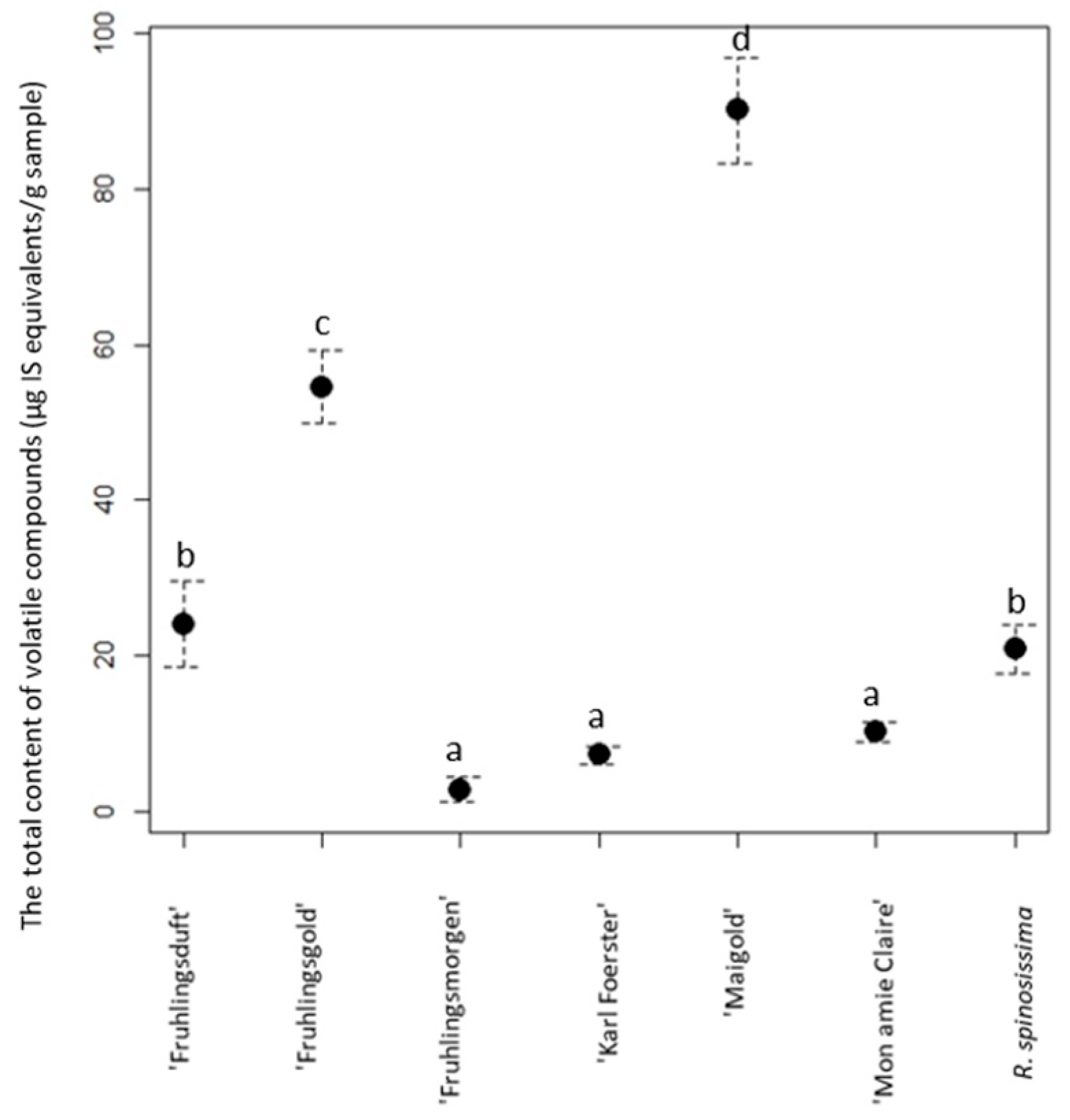
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Raymond, O.; Gouzy, J.; Just, J.; Badouin, H.; Verdenaud, M.; Lemainque, A.; Vergne, P.; Moja, S.; Choisne, N.; Pont, C.; et al. The Rosa genome provides new insights into the domestication of modern roses. Nat. Genet. 2018, 50, 772–777. [Google Scholar] [CrossRef]
- Shamspur, T.; Mostafavi, A. Chemical composition of the volatile oil of Rosa kazanlik and Rosa gallica from Kerman province in Iran. J. Essent. Oil-Bear. Plants 2010, 13, 78–84. [Google Scholar] [CrossRef]
- Cottini, P. Vrtnice, sorte in način gojenja (izbira, sajenje, nega, obrezovanje). Rože in vrt 2003, 1–34. [Google Scholar]
- Gudin, S. Rose: Genetics and Breeding. Plant Breed. Rev. 2000, 17, 319–334. [Google Scholar] [CrossRef]
- Pal, P.K. Evaluation, genetic diversity, recent development of distillation method, challenges and opportunities of Rosa damascena: A review. J. Essent. Oil-Bear. Plants 2013, 16, 1–10. [Google Scholar] [CrossRef]
- Antonelli, A.; Fabbri, C.; Giorgioni, M.E.; Bazzocchi, R. Characterization of 24 old garden roses from their volatile compositions. J. Agric. Food Chem. 1997, 45, 4435–4439. [Google Scholar] [CrossRef]
- Maffei, M. Sites of synthesis, biochemistry and functional role of plant volatiles. S. Afr. J. Bot. 2010, 76, 612–631. [Google Scholar] [CrossRef]
- Bianchi, G.; Nuzzi, M.; Avitabile Leva, A.; Rizzolo, A. Development of a headspace-solid phase micro extraction method to monitor changes in volatile profile of rose (Rosa hybrida, cv David Austin) petals during processing. J. Chromatogr. A 2007, 1150, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Lijun, Z.; Chao, Y.; Cheng, B.; Wan, H.; Le, L.; Huitanga, P.; Qixianga, Z. Volatile compound analysis and aroma evaluation of tea-scented roses in China. Ind. Crops Prod. 2020, 155, 112735. [Google Scholar] [CrossRef]
- Loghmani-Khouzani, H.; Fini, O.S.; Safari, J. Essential oil composition of Rosa damascena Mill. cultivated in Central Iran. Iran J. Sci. Technol. Trans. A Sci. 2007, 14, 316–319. [Google Scholar]
- Demir, N.; Yildiz, O.; Alpaslan, M.; Hayaloglu, A.A. Evaluation of volatiles, phenolic compounds and antioxidant activites of rose hip (Rosa L.) fruits in Turkey. LWT Food Sci. Technol. 2013, 57, 126–133. [Google Scholar] [CrossRef]
- Abudayeh, Z.; Karpiuk, U.; Armoon, N.; Abualassal, Q.; Mallah, E.; Hassouneh, D.L.; Aldalahmeh, Y. Phytochemical, Physiochemical, macroscopic, and microscopic analysis of Rosa damascena flower petals and buds. J. Food Qual. 2022, 2022, 5079964. [Google Scholar] [CrossRef]
- Kunc, N.; Hudina, M.; Bavcon, J.; Vreš, B.; Luthar, Z.; Gostinčar, K.; Mikulič-Petkovšek, M.; Osterc, G.; Ravnjak, B. Characterization of the Slovene autochthonous rose hybrid Rosa pendulina × spinosissima (Rosa reversa Waldst. and Kit) using biochemical patterns of the plant blossoms. Plants 2023, 12, 505. [Google Scholar] [CrossRef] [PubMed]
- Baydar, N.; Baydar, H. Phenolic compounds, antiradical activity and antioxidant capacity of oil-bearing rose (Rosa damascena Mill.) extracts. Ind. Crops Prod. 2013, 41, 375–380. [Google Scholar] [CrossRef]
- Boskabady, M.H.; Shafei, M.N.; Saberi, Z.; Amini, S. Pharmacological effects of Rosa damascena. Iran. J. Basic Med. Sci. 2011, 14, 295–307. [Google Scholar] [CrossRef]
- Hongratanaworakit, T. Relaxing effect of rose oil on humans. Nat. Prod. Commun. 2009, 4, 2. [Google Scholar] [CrossRef]
- Li, M.; Xie, J.; Bai, X. Anti-aging potential, anti-tyrosinase and antibacterial activities of extracts and compounds isolated from Rosa chinensis cv. ‘JinBian’. Ind. Crops Prod. 2021, 159, 113059. [Google Scholar] [CrossRef]
- Younis, I.; El-Hawary, S.; Eldahshan, O.; Abdel-Aziz, M.; Ali, Z. Green synthesis of magnesium nanoparticles mediated from Rosa floribunda charisma extract and its antioxidant, antiaging and antibiofilm activities. Sci. Rep. 2021, 11, 16868. [Google Scholar] [CrossRef]
- Özdemir, N.; Budak, N. Bioactive compounds and volatile aroma compounds in rose (Rosa damascena Mill.) vinegar during the aging period. Food Biosci. 2022, 50, 102062. [Google Scholar] [CrossRef]
- Rubtsova, O.; Kolesnichenko, O.; Shumyk, M.; Shynder, O.; Chyzhankova, V.; Dzyba, A.; Hrysiuk, S. Scots roses in Northern Ukraine. Hortic. Sci. 2021, 48, 144–148. [Google Scholar] [CrossRef]
- Gudin, S. Rose breeding technologies. Acta Hortic. 2001, 547, 23–33. [Google Scholar] [CrossRef]
- Datta, S.K. Breeding of new ornamental varieties: Rose. Curr. Sci. 2018, 114, 1194–1206. [Google Scholar] [CrossRef]
- Meier, U.; Graf, H.; Hack, H.; Hess, M.; Kennel, W.; Klose, R.; Mappes, D.; Seipp, D.; Stauß, R.; Streif, J.; et al. Phanologische Entwicklungsstadien des Kernobstes (Malus domestica Borkh. und Pyrus communis L.), des Steinobstes (Prunus-Arten), der Johannisbeere (Ribes-Arten) und der Erdbeere (Fragaria × ananassa Duch.). Nachr. Dtsch. Pflanzenschutzd. 1994, 46, 141–153. [Google Scholar]
- Baluszynska, U.B.; Licznar-Malanczuk, M.; Medic, A.; Veberic, R.; Grohar, M.C. Influence of living mulch and nitrogen dose on yield and fruit quality parameters of Malus domestica Borkh. cv. ‘Sampion’. Agriculture 2023, 13, 921. [Google Scholar] [CrossRef]
- Interplant. 2023. Available online: https://www.interplantroses.nl/company (accessed on 10 September 2023).
- Kordes. 2023. Available online: https://southamptonrose.org/wp-content/uploads/2012/pdf/Brochure_Kordes.pdf (accessed on 10 September 2023).
- Jang, S.S.; Choi, J.S.; Kim, J.H.; Kim, N.; Ference, E.H. Discordance between subjective and objective measures of smell and taste in US adults. Otolaryngol.–Head Neck Surg. 2022, 166, 572–579. [Google Scholar] [CrossRef]
- Ryu, J.; Lyu, J.I.; Kim, D.-G.; Kim, J.-M.; Jo, Y.D.; Kang, S.-Y.; Kim, J.-B.; Ahn, J.-W.; Kim, S.H. Comparative analysis of volatile compounds of gamma-irradiated mutants of rose (Rosa hybrida). Plants 2020, 9, 1221. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Cheng, X.; Lu, Y.; Wang, H.; Chen, D.; Luo, C.; Liu, H.; Gao, S.; Lei, T.; Huang, C.; et al. Gas chromatography-mass spectrometry analysis of floral fragrance-related compounds in scented rose (Rosa hybrida) varieties and a subsequent evaluation on the basis of the analytical hierarchy process. Plant Physiol. Biochem. 2022, 185, 368–377. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.C.; Miranda, M.C.; Schäfer, A.; Pham, M.N.; Greaney, A.; Arunachalam, P.S.; Navarro, M.J.; Tortorici, M.A.; Rogers, K.; O’Connor, M.A.; et al. Elicitation of broadly protective sarbecovirus immunity by receptor-binding domain nanoparticle vaccines. Plant Cell 2021, 184, 5432–5447.e16. [Google Scholar] [CrossRef] [PubMed]
- Guterman, I.; Shalit, M.; Menda, N.; Piestun, D.; Dafny-Yelin, M.; Shalev, G.; Weiss, D. Rose scent genomics approach to discovering novel foral fragrance-related genes. Plant Cell 2002, 14, 2325–2338. [Google Scholar] [CrossRef] [PubMed]
- Bohlmann, J.; Meyer-Gauen, G.; Croteau, R. Plant terpenoid synthases: Molecular biology and phylogenetic analysis. Proc. Nat. Acad. Sci. USA 1998, 95, 4126–4133. [Google Scholar] [CrossRef] [PubMed]
- Degenhardt, J.; Köllner, T.G.; Gershenzon, J. Monoterpene and sesquiterpene synthases and the origin of terpene skeletal diversity in plants. Phytochemistry 2009, 70, 1621–1637. [Google Scholar] [CrossRef]
- Magnard, J.L.; Roccia, A.; Caissard, J.C.; Vergne, P.; Sun, P.; Hecquet, R.; Baudino, S. Biosynthesis of monoterpene scent compounds in roses. Science 2015, 349, 81–83. [Google Scholar] [CrossRef]
- Sun, P.; Schuurink, R.C.; Caissard, J.C.; Hugueney, P.; Baudino, S. My way: Noncanonical biosynthesis pathways for plant volatiles. Trends Plant Sci. 2016, 21, 884–894. [Google Scholar] [CrossRef] [PubMed]
- Dani, K.G.S.; Fineschi, S.; Michelozzi, M.; Trivellini, A.; Pollastri, S.; Loreto, F. Diversification of petal monoterpene profiles during floral development and senescence in wild roses: Relationships among geraniol content, petal colour, and floral lifespan. Oecologia 2021, 197, 957–969. [Google Scholar] [CrossRef] [PubMed]
- Sparinska, A.; Rostoks, N. Volatile organic compounds of hybrid Rugosa roses in Latvia. Proc. Latv. Acad. Sci. Sect. B. Nat. Exact Appl. Sci. 2015, 69, 57–61. [Google Scholar] [CrossRef]
- Karani, L.W.; Tolo, F.M.; Karanja, S.M.; Khayeka−Wandabwa, C. Safety of Prunus africana and Warburgia ugandensis in Asthma Treatment. S. Afr. J. Bot. 2013, 88, 183–190. [Google Scholar] [CrossRef]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Mai, J.; Li, W.; Ledesma-Amaro, R.; Ji, X.J. Engineering plant sesquiterpene synthesis into yeasts: A review. J. Agric. Food Chem. 2021, 69, 9498–9510. [Google Scholar] [CrossRef] [PubMed]
- Abbas, F.; Zhou, Y.; O’Neill Rothenberg, D.; Alam, I.; Ke, Y.; Wang, H.-C. Aroma components in horticultural crops: Chemical diversity and usage of metabolic engineering for industrial applications. Plants 2023, 12, 1748. [Google Scholar] [CrossRef] [PubMed]
- Libre Text. 2023. Available online: https://chem.libretexts.org/Bookshelves/General_Chemistry/ChemPRIME_(Moore_et_al.)/08%3A_Properties_of_Organic_Compounds/8.09%3A_Aromatic_Hydrocarbons (accessed on 10 September 2023).
- Bergougnoux, V.; Caissard, J.C.; Jullien, F.; Magnard, J.L.; Scalliet, G.; Cock, J.M.; Hugueney, P.; Baudino, S. Both the adaxial and abaxial epidermal layers of the rose petal emit volatile scent compounds. Planta 2007, 226, 853–866. [Google Scholar] [CrossRef]
- Riu-Aumatell, M.; Castellari, M.; López-Tamames, E.; Galassi, S.; Buxaderas, S. Characterisation of volatile compounds of fruit juices and nectars by HS/SPME and GC/MS. Food Chem. 2004, 87, 627–637. [Google Scholar] [CrossRef]
- Chemical Book. 2023. Available online: https://m.chemicalbook.com/ (accessed on 10 September 2023).
- Mostafa, S.; Wang, Y.; Zeng, W.; Jin, B. Floral scents and fruit aromas: Functions, compositions, biosynthesis, and regulation. Front. Plant Sci. 2022, 13, 860157. [Google Scholar] [CrossRef]
- Tan, K.H.; Nishida, R. Methyl eugenol: Its occurrence, distribution, and role in nature, especially in relation to insect behavior and pollination. J. Insect Sci. 2012, 12, 56. [Google Scholar] [CrossRef] [PubMed]
- Rusanov, K.; Kovacheva, N.; Rusanova, M.; Atanassov, I. Low variability of flower volatiles of Rosa damascena Mill. plants from rose plantations along the Rose Valley, Bulgaria. Ind. Crops Prod. 2012, 37, 6–10. [Google Scholar] [CrossRef]
- Mileva, M.; Ilieva, Y.; Jovtchev, G.; Gateva, S.; Zaharieva, M.M.; Georgieva, A.; Dimitrova, L.; Dobreva, A.; Angelova, T.; Vilhelmova-Ilieva, N.; et al. Rose flowers-a delicate perfume or a natural healer? Biomolecules 2021, 11, 127. [Google Scholar] [CrossRef] [PubMed]
- Staudt, M.; Bertin, M. Light and temperature dependence of emission of cyclic and acyclic monoterpenes from holm oak (Quercus ilex L.) leaves. Plant Cell Environ. 1998, 21, 385–395. [Google Scholar] [CrossRef]


| Cultivar | Species of Origin | Breeding Company | Year of the Origin |
|---|---|---|---|
| ‘Violacea’ | R. gallica | unknown (The Netherlands) | 1795 |
| ‘Splendens’ | R. gallica | unknown | 1583 |
| ‘Charles de Mills’ | R. gallica | unknown | 1790 |
| ‘Complicata’ | R. gallica | unknown | before 1873 |
| ‘Karl Foerster’ | R. spinosissima | Kordes (Germany) | 1930 |
| ‘Fruhlingsduft’ | R. spinosissima | Kordes (Germany) | 1941 |
| ‘Fruhlingsmorgen’ | R. spinosissima | Kordes (Germany) | 1941 |
| ‘Fruhlingsgold’ | R. spinosissima | Kordes (Germany) | 1937 |
| ‘Mon amie Claire’ | R. spinosissima | Ivan Louette (Belgium) | 2005 |
| ‘Maigold’ | R. spinosissima | Kordes (Germany) | 1953 |
| ‘Harstad’ | R. pendulina | unknown | unknown (old cultivar) |
| ‘Mount Everest’ | R. pendulina | Interplant (Netherland) | 1956 |
| ‘Bourgogne’ | R. pendulina | Interplant (Netherland) | 1983 |
| MONOTERPENES | SESQUITERPENES | ALDEHYDES | ALCOHOLS | KETONES | HYDROCARBONS | PHENYLPROPENES | OTHER COMPOUNDS | Total | |
|---|---|---|---|---|---|---|---|---|---|
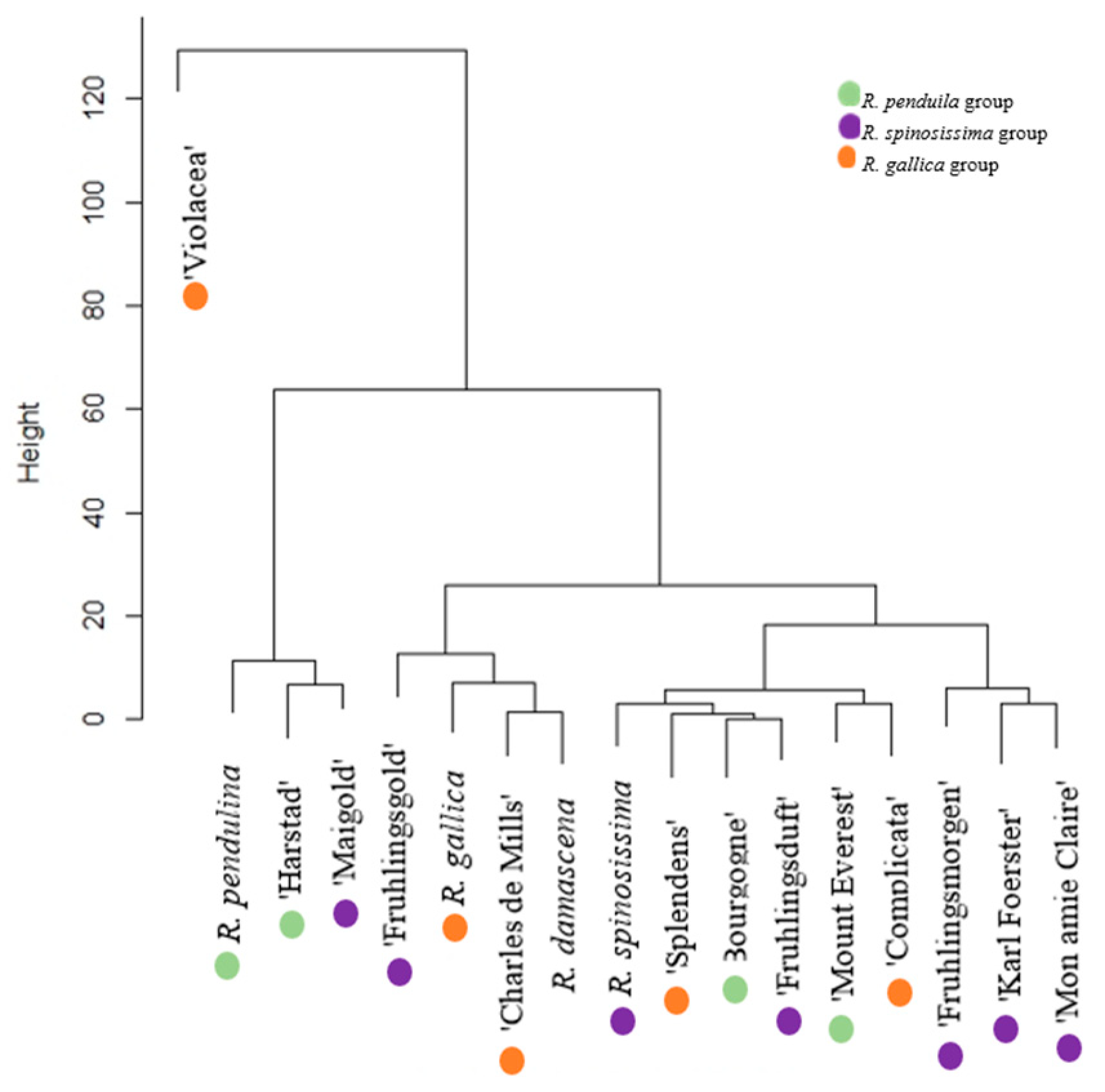
| R. pendulina | 59.15 ± 5.322 e | 13.04 ± 2.543 c | 15.44 ± 1.092 bd | 4.44 ± 1.935 ade | - | 6.17 ± 0.876 ce | - | - | 98.24 ± 11.768 e |
| ‘Bourgogne’ | 0.28 ± 0.07 a | 1.24 ± 0.216 a | 17.01 ± 1.501 bd | 1.32 ± 0.215 ab | - | 2.04 ± 0.51 ab | - | 2.19 ± 0.38 c | 24.08 ± 2.892 ac |
| ‘Harstad’ | 46.09 ± 3.049 d | 5.00 ± 0.627 b | 23.40 ± 1.87 de | 2.74 ± 0.193 ade | 0.09 ± 0.007 ac | 4.48 ± 0.404 abe | 0.04 ± 0.006 a | 1.40 ± 0.17 b | 83.24 ± 6.272 de |
| ‘Mount Everest’ | 2.04 ± 0.596 a | 0.54 ± 0.075 a | 22.04 ± 1.263 de | 0.96 ± 0.42 a | - | 1.32 ± 0.208 a | - | 0.10 ± 0.004 a | 27.00 ± 2.548 ac |
| R. spinosissima | 0.08 ± 0.02 a | 0.03 ± 0.007 a | 12.79 ± 1.53 bc | 1.50 ± 0.41 ab | 0.01 ± 0.01 ac | 5.81 ± 1.05 bce | 0.28 ± 0.04 e | 0.12 ± 0.02 a | 20.62 ± 3.087 ac |
| ‘Fruhlingsmorgen’ | 0.23 ± 0.03 a | 0.24 ± 0.043 a | 19.30 ± 1.26 be | 2.23 ± 0.42 ad | 0.08 ± 0.01 c | 3.65 ± 0.283 acd | 0.03 ± 0.003 a | 0.03 ± 0.005 a | 2.579 ± 2.054 a |
| ‘Fruhlingsduft’ | - | 0.61 ± 0.05 a | 1.04 ± 0.16 a | 9.74 ± 2.113 f | 0.19 ± 0.01 d | 12.30 ± 1.874 f | 0.11 ± 0.03 bd | 0.09 ± 0.01 a | 24.08 ± 4.247 ac |
| ‘Karl Foerster’ | - | 0.51 ± 0.022 a | 1.07 ± 0.15 a | 1.47 ± 0.302 ab | 0.52 ± 0.017 a | 3.45 ± 0.362 acd | - | 0.03 ± 0.002 a | 7.05 ± 0.855 ab |
| ‘Mon amie Claire’ | - | 0.28 ± 0.02 a | 0.30 ± 0.03 a | 5.95 ± 0.655 cdf | - | 3.32 ± 0.43 acd | 0.15 ± 0.02 d | 0.11 ± 0.01 a | 10.11 ± 1.165 ab |
| ‘Maigold’ | 2.98 ± 0.40 ab | 0.62 ± 0.02 a | 69.57 ± 3.20 g | 6.38 ± 1.356 df | 0.19 ± 0.003 bc | 8.60 ± 0.864 e | 0.05 ± 0.03 ab | 1.76 ± 0.227 bc | 90.15 ± 6.10 de |
| ‘Fruhlingsgold’ | 1.12 ± 0.27 a | 0.34 ± 0.027 a | 40.96 ± 1.612 f | 6.04 ± 1.70 df | 0.15 ± 0.008 a | 5.22 ± 0.437 abe | 0.06 ± 0.007 ab | 0.12 ± 0.01 a | 54.40 ± 4.071 cd |
| R. gallica | 5.26 ± 1.204 ab | 0.03 ± 0.005 a | 21.84 ± 1.27 cde | 2.45 ± 0.423 ad | 0.09 ± 0.003 ac | 6.98 ± 0.828 de | - | 0.14 ± 0.007 a | 36.84 ± 3.745 ac |
| ‘Charles de Mills’ | 3.60 ± 1.28 ab | 0.21 ± 0.032 a | 28.8 ± 3.521 e | 5.47 ± 1.203 bde | 0.10 ± 0.008 ac | 4.91 ± 0.544 abe | - | 0.03 ± 0.02 a | 43.12 ± 6.59 bc |
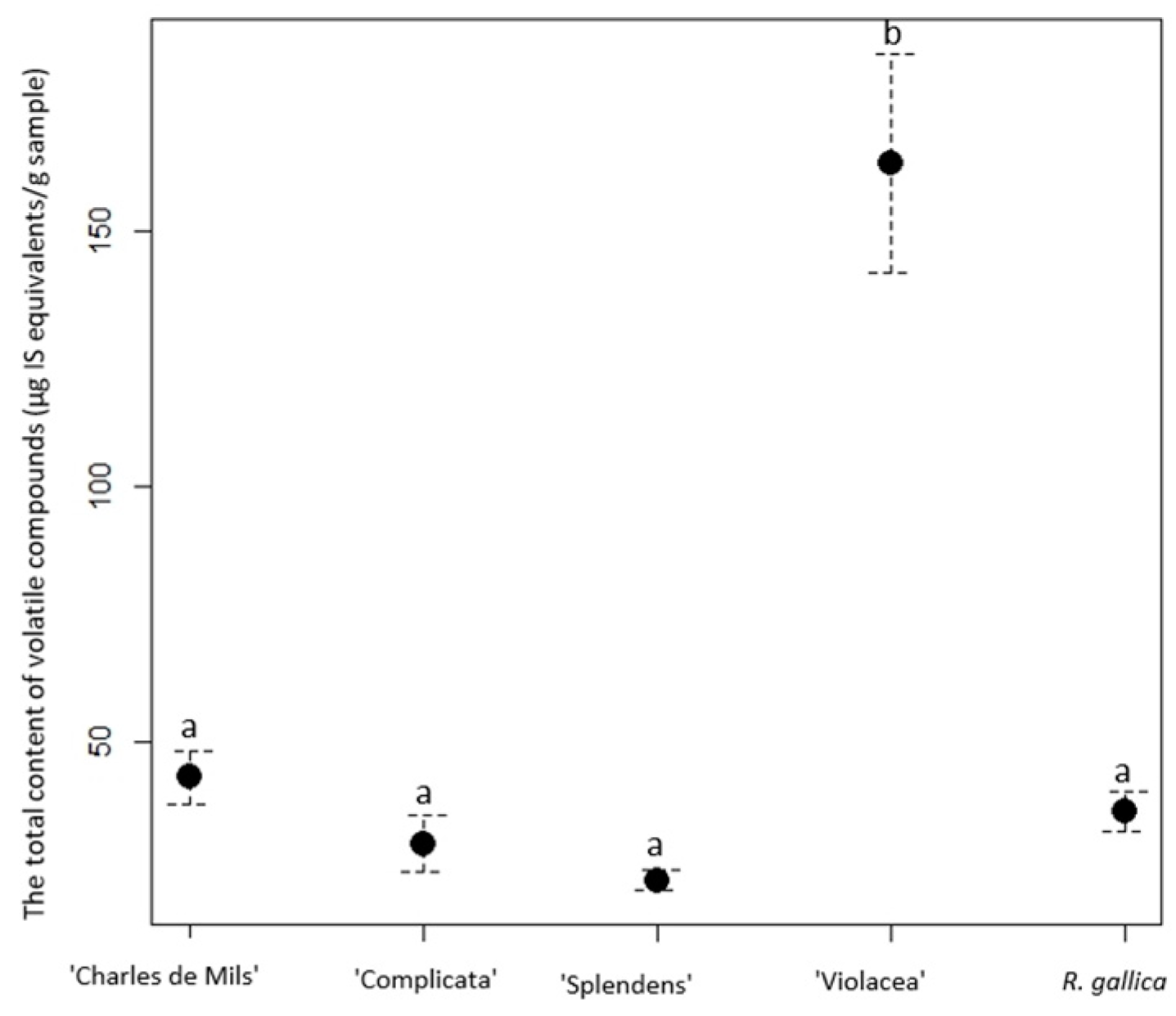
| ‘Violacea’ | 126.32 ± 19.24 f | 0.08 ± 0.01 a | 20.13 ± 2.605 cde | 4.45 ± 1.75 ade | 0.11 ± 0.01 ac | 17.01 ± 2.86 g | - | 0.14 ± 0.02 a | 168.24 ± 26.495 f |
| ‘Splendens’ | 0.58 ± 0.10 a | 0.48 ± 0.05 a | 18.00 ± 1.91 bd | 1.60 ± 0.199 abc | 0.11 ± 0.009 ac | 1.93 ± 0.097 ab | - | 0.40 ± 0.043 a | 23.10 ± 2.408 ac |
| ‘Complicata’ | 14.75 ± 3.22 bc | 0.18 ± 0.026 a | 10.74 ± 1.43 b | 1.45 ± 0.486 ab | 0.05 ± 0.008 ab | 2.91 ± 0.234 ac | 0.11 ± 0.02 cd | 0.07 ± 0.014 a | 30.26 ± 5.438 ac |
| R. damascena | 18.07 ± 2.71 c | 0.66 ± 0.16 a | 16.64 ± 3.164 bd | 6.76 ± 1.231 ef | 0.19 ± 0.03 c | 2.09 ± 0.302 ab | 0.06 ± 0.01 abc | 0.08 ± 0.002 a | 44.55 ± 7.609 bc |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kunc, N.; Hudina, M.; Osterc, G.; Grohar, M.C. Determination of Volatile Compounds in Blossoms of Rosa spinosissima, Rosa pendulina, Rosa gallica, and Their Cultivars. Agriculture 2024, 14, 253. https://doi.org/10.3390/agriculture14020253
Kunc N, Hudina M, Osterc G, Grohar MC. Determination of Volatile Compounds in Blossoms of Rosa spinosissima, Rosa pendulina, Rosa gallica, and Their Cultivars. Agriculture. 2024; 14(2):253. https://doi.org/10.3390/agriculture14020253
Chicago/Turabian StyleKunc, Nina, Metka Hudina, Gregor Osterc, and Mariana Cecilia Grohar. 2024. "Determination of Volatile Compounds in Blossoms of Rosa spinosissima, Rosa pendulina, Rosa gallica, and Their Cultivars" Agriculture 14, no. 2: 253. https://doi.org/10.3390/agriculture14020253
APA StyleKunc, N., Hudina, M., Osterc, G., & Grohar, M. C. (2024). Determination of Volatile Compounds in Blossoms of Rosa spinosissima, Rosa pendulina, Rosa gallica, and Their Cultivars. Agriculture, 14(2), 253. https://doi.org/10.3390/agriculture14020253







