Comparison of PCR Techniques in Adulteration Identification of Dairy Products
Abstract
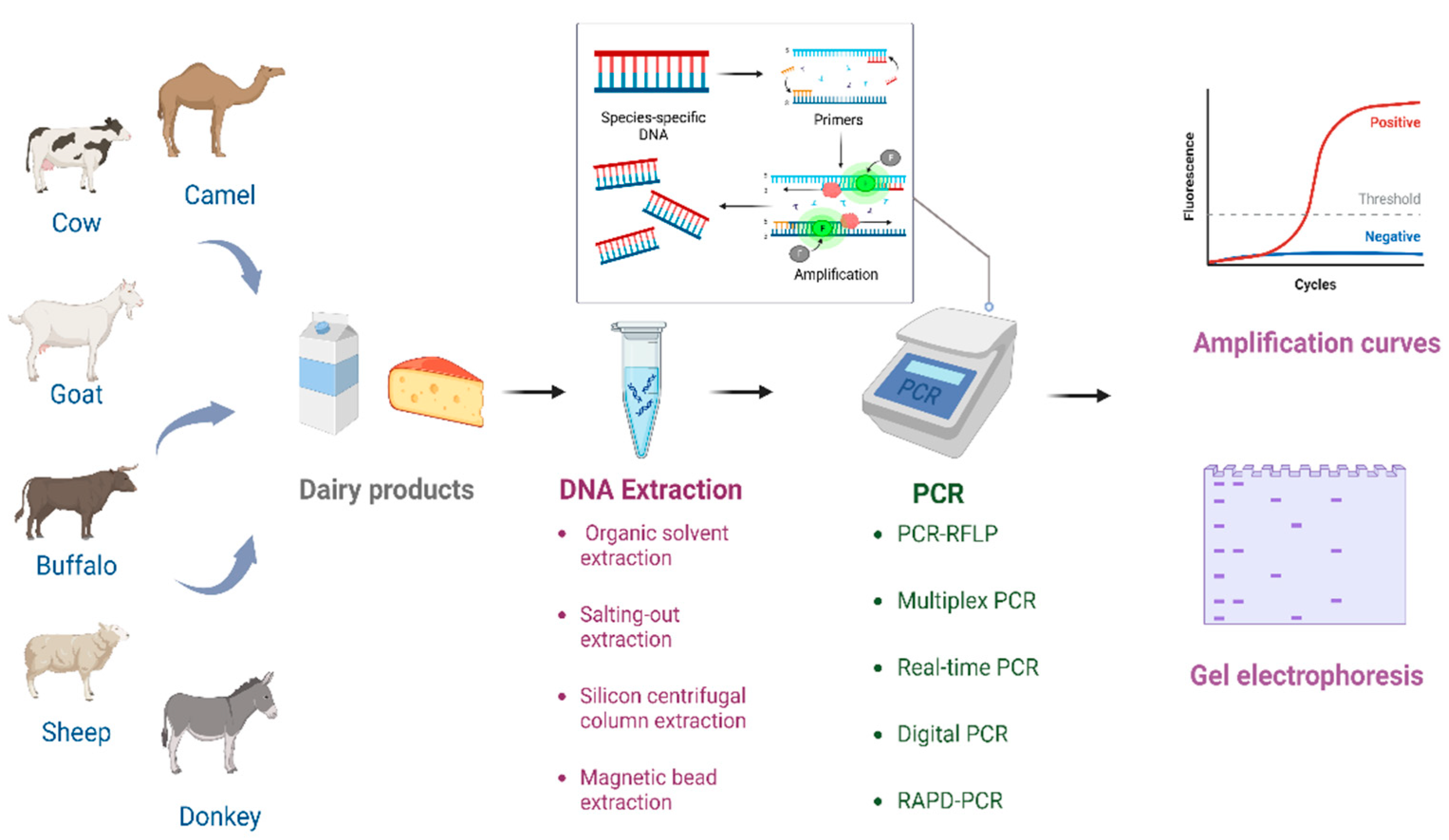
1. Introduction
2. DNA Extraction Methods
2.1. Organic Solvent Extraction Method
2.2. Salting-Out Extraction Method
2.3. Silicon Centrifugal Column Extraction Method
2.4. Magnetic Bead Extraction Method
3. PCR-Based Dairy Product Authenticity Testing
3.1. PCR-RFLP
3.2. Multiplex PCR
3.3. Real-Time PCR
3.4. Digital PCR (dPCR)
3.5. Randomly Amplified Polymorphic DNA PCR (RAPD-PCR)
4. Factors Affecting PCR-Based Dairy Product Authenticity Testing
4.1. Disruption of DNA Integrity by Food Processing
4.2. PCR Inhibitors
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Moreira, M.J.; Garcia-Diez, J.; de Almeida, J.M.M.M.; Saraiva, C. Consumer Knowledge about Food Labeling and Fraud. Foods 2021, 10, 1095. [Google Scholar] [CrossRef]
- Boosting knowledge on food fraud. Food Sci. Technol. 2020, 34, 24–25. [CrossRef]
- Committee on the Environment, Public Health and Food Safety. Draft Report the Food Crisis, Fraud in the Food Chain and the Control Thereof (2013/2091(INI)). Available online: https://www.europarl.europa.eu/doceo/document/ENVI-PR-519759_EN.pdf?redirect (accessed on 28 March 2023).
- Smith, N.W.; Fletcher, A.J.; Hill, J.P.; McNabb, W.C. Modeling the Contribution of Milk to Global Nutrition. Front. Nutr. 2021, 8, 716100. [Google Scholar] [CrossRef]
- Wu, W.; Zhang, D.; Wang, T.; Liang, X.; Cai, Z.; Yang, X. Study on the concepts of economically motivated food adulteration. J. Food Saf. Qual. 2020, 11, 3339–3347. [Google Scholar]
- Gonçalves, J.; Pereira, F.; Amorim, A.; van Asch, B. New method for the simultaneous identification of cow, sheep, goat, and water buffalo in dairy products by analysis of short species-specific mitochondrial DNA targets. J. Agric. Food Chem. 2012, 60, 10480–10485. [Google Scholar] [CrossRef]
- Di Pinto, A.; Terio, V.; Marchetti, P.; Bottaro, M.; Mottola, A.; Bozzo, G.; Bonerba, E.; Ceci, E.; Tantillo, G. DNA-based approach for species identification of goat-milk products. Food Chem. 2017, 229, 93–97. [Google Scholar] [CrossRef]
- Zhang, H.; Abdallah, M.F.; Zhang, J.; Yu, Y.; Zhao, Q.; Tang, C.; Qin, Y.; Zhang, J. Comprehensive quantitation of multi-signature peptides originating from casein for the discrimination of milk from eight different animal species using LC-HRMS with stable isotope labeled peptides. Food Chem. 2022, 390, 133126. [Google Scholar] [CrossRef]
- Lu, Y.; Dai, J.; Zhang, S.; Qiao, J.; Lian, H.; Mao, L. Identification of Characteristic Peptides of Casein in Cow Milk Based on MALDI-TOF MS for Direct Adulteration Detection of Goat Milk. Foods 2023, 12, 1519. [Google Scholar] [CrossRef]
- Scarpone, R.; Kimkool, P.; Ierodiakonou, D.; Leonardi-Bee, J.; Garcia-Larsen, V.; Perkin, M.R.; Boyle, R.J. Timing of Allergenic Food Introduction and Risk of Immunoglobulin E–Mediated Food Allergy: A Systematic Review and Meta-analysis. JAMA Pediatr. 2023, 177, 489–497. [Google Scholar] [CrossRef]
- Nelis, J.L.D.; Dawson, A.L.; Bose, U.; Anderson, A.; Colgrave, M.L.; Broadbent, J.A. Safe food through better labelling; a robust method for the rapid determination of caprine and bovine milk allergens. Food Chem. 2023, 417, 135885. [Google Scholar] [CrossRef]
- Baptista, M.; Cunha, J.T.; Domingues, L. DNA-based approaches for dairy products authentication: A review and perspectives. Trends Food Sci. Technol. 2021, 109, 386–397. [Google Scholar] [CrossRef]
- Dawan, J.; Ahn, J. Application of DNA barcoding for ensuring food safety and quality. Food Sci. Biotechnol. 2022, 31, 1355–1364. [Google Scholar] [CrossRef] [PubMed]
- Mafra, I.; Honrado, M.; Amaral, J.S. Animal Species Authentication in Dairy Products. Foods 2022, 11, 1124. [Google Scholar] [CrossRef] [PubMed]
- Grazina, L.; Amaral, J.S.; Mafra, I. Botanical origin authentication of dietary supplements by DNA-based approaches. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1080–1109. [Google Scholar] [CrossRef]
- Vishnuraj, M.R.; Aravind Kumar, N.; Vaithiyanathan, S.; Barbuddhe, S.B. Authentication issues in foods of animal origin and advanced molecular techniques for identification and vulnerability assessment. Trends Food Sci. Technol. 2023, 138, 164–177. [Google Scholar] [CrossRef]
- Sun, P.; Wang, R.; Wu, H.; Zhao, J. Analyses the Authenticity Identification Methods of Milk Products. China Dairy Cattle 2022, 12, 54–57. [Google Scholar]
- Chen, Y.; Abbas, Z.; Hu, L.; Kang, L.; Tan, X.; Xu, Q.; Wang, Y. Extraction and Elevation of Cell-Free DNA under Mastitis and Heat Stress in Dairy Cattle. Animals 2023, 13, 1487. [Google Scholar] [CrossRef]
- Liao, J.; Gao, J.; Ku, T.; Liu, Y. Assessment of milk quality during storage based on DNA extracted from milk. CyTA-J. Food 2018, 16, 786–792. [Google Scholar] [CrossRef]
- Dal Bosco, C.; Panero, S.; Navarra, M.A.; Tomai, P.; Curini, R.; Gentili, A. Screening and Assessment of Low-Molecular-Weight Biomarkers of Milk from Cow and Water Buffalo: An Alternative Approach for the Rapid Identification of Adulterated Water Buffalo Mozzarellas. J. Agric. Food Chem. 2018, 66, 5410–5417. [Google Scholar] [CrossRef]
- Wijenayake, S.; Eisha, S.; Tawhidi, Z.; Pitino, M.A.; Steele, M.A.; Fleming, A.S.; McGowan, P.O. Comparison of methods for pre-processing, exosome isolation, and RNA extraction in unpasteurized bovine and human milk. PLoS ONE 2021, 16, e0257633. [Google Scholar] [CrossRef]
- Burke, N.; Zacharski, K.; Adley, C.C.; Southern, M. A comparison of analytical test methods in dairy processing. Food Control 2021, 121, 107637. [Google Scholar] [CrossRef]
- Azevedo, B.T.; Vercesi Filho, A.E.; Gutmanis, G.; Verissimo, C.J.; Katiki, L.M.; Okino, C.H.; Cristina de Sena Oliveira, M.; Giglioti, R. New sensitive methods for fraud detection in buffalo dairy products. Int. Dairy J. 2021, 117, 105013. [Google Scholar] [CrossRef]
- Psifidi, A.; Dovas, C.I.; Banos, G. A comparison of six methods for genomic DNA extraction suitable for PCR-based genotyping applications using ovine milk samples. Mol. Cell. Probes 2010, 24, 93–98. [Google Scholar] [CrossRef]
- Yalçınkaya, B.; Yumbul, E.; Mozioğlu, E.; Akgoz, M. Comparison of DNA extraction methods for meat analysis. Food Chem. 2017, 221, 1253–1257. [Google Scholar] [CrossRef] [PubMed]
- Liao, J. Study on the Variation of DNA in Milk Processing and Storage and Its Application in Adulteration Identification. Master’s Thesis, Shaanxi Normal University, Xi’an, China, 2018. [Google Scholar]
- Chen, X.; Ke, Z. Research on Multiplex RT-PCR Detection Method of Animal and Plant Derived Ingredients in Goat Milk. Food Res. Dev. 2017, 38, 156–162. [Google Scholar]
- d’Angelo, F.; Santillo, A.; Sevi, A.; Albenzio, M. Technical Note: A Simple Salting-Out Method for DNA Extraction from Milk Somatic Cells: Investigation into the Goat CSN1S1 Gene. J. Dairy Sci. 2007, 90, 3550–3552. [Google Scholar] [CrossRef]
- Dieki, R.; Nsi Emvo, E.; Akue, J.P. Comparison of six methods for Loa loa genomic DNA extraction. PLoS ONE 2022, 17, e0265582. [Google Scholar] [CrossRef]
- Kovačević, N. Magnetic Beads Based Nucleic Acid Purification for Molecular Biology Applications. In Sample Preparation Techniques for Soil, Plant, and Animal Samples; Micic, M., Ed.; Springer Protocols Handbooks; Humana: New York, NY, USA, 2016; pp. 53–67. [Google Scholar]
- Díaz, I.L.-C.; Alonso, I.G.; Fajardo, V.; Martín, I.; Hernández, P.; Lacarra, T.G.; de Santos, R.M. Application of a polymerase chain reaction to detect adulteration of ovine cheeses with caprine milk. Eur. Food Res. Technol. 2007, 225, 345–349. [Google Scholar] [CrossRef]
- Xiao, X.; Zhang, Y.; Zhang, K.; Chen, H.; Yin, R. Research on Rapid DNA Extraction Method of Meat Products Based on Magnetic Bead Method. China Food Saf. Mag. 2022, 17, 65–67. [Google Scholar]
- Xu, P.; Zheng, C.; Sun, Z.; Yu, L.; Feng, K. Rapid extraction of genomic DNA by magnetic beads method. China J. Bioinform. 2018, 16, 190–195. [Google Scholar]
- Pinto, A.D.; Forte, V.; Guastadisegni, M.C.; Martino, C.; Schena, F.P.; Tantillo, G. A comparison of DNA extraction methods for food analysis. Food Control 2007, 18, 76–80. [Google Scholar] [CrossRef]
- Liu, Y.F.; Gao, J.L.; Yang, Y.F.; Ku, T.; Zan, L.S. Novel extraction method of genomic DNA suitable for long-fragment amplification from small amounts of milk. J. Dairy Sci. 2014, 97, 6804–6809. [Google Scholar] [CrossRef] [PubMed]
- Lusk, T.S.; Strain, E.; Kase, J.A. Comparison of six commercial DNA extraction kits for detection of Brucella neotomae in Mexican and Central American-style cheese and other milk products. Food Microbiol. 2013, 34, 100–105. [Google Scholar] [CrossRef]
- Yang, B.; Liu, B.; Chen, Z. DNA Extraction with TRIzol Reagent Using a Silica Column. Anal. Sci. 2021, 37, 1033–1037. [Google Scholar] [CrossRef]
- Gupta, N. DNA Extraction and Polymerase Chain Reaction. J. Cytol. 2019, 36, 116–117. [Google Scholar] [CrossRef]
- Yao, B.; Sun, D.; Ren, Y.; Wang, M. Introducing Theoretical Principles of Semi-, Relative, and Absolute Quantification via Conventional, Real-Time, and Digital PCR to Graduate and Senior Undergraduate Students of Chemistry. J. Chem. Educ. 2022, 99, 603–611. [Google Scholar] [CrossRef]
- Tan, M.; Liao, C.; Liang, L.; Yi, X.; Zhou, Z.; Wei, G. Recent advances in recombinase polymerase amplification: Principle, advantages, disadvantages and applications. Front. Cell. Infect. Microbiol. 2022, 12, 1744. [Google Scholar] [CrossRef] [PubMed]
- Plath, A.; Krause, I.; Einspanier, R. Species identification in dairy products by three different DNA-based techniques. Z. Für Leb. Und-Forsch. A 1997, 205, 437–441. [Google Scholar] [CrossRef]
- Abdel-Rahman, S.M.; Ahmed, M.M.M. Rapid and sensitive identification of buffalo’s, cattle’s and sheep’s milk using species-specific PCR and PCR–RFLP techniques. Food Control 2007, 18, 1246–1249. [Google Scholar] [CrossRef]
- Lanzilao, I.; Burgalassi, F.; Fancelli, S.; Settimelli, M.; Fani, R. Polymerase Chain Reaction-Restriction Fragment Length Polymorphism Analysis of Mitochondrial cytb Gene from Species of Dairy Interest. J. AOAC Int. 2019, 88, 128–135. [Google Scholar] [CrossRef]
- Vafin, R.R.; Galstyan, A.G.; Tyulkin, S.V.; Gilmanov, K.K.; Yurova, E.A.; Semipyatniy, V.K.; Bigaeva, A.V. Species identification of ruminant milk by genotyping of the κ-casein gene. J. Dairy Sci. 2022, 105, 1004–1013. [Google Scholar] [CrossRef] [PubMed]
- Bottero, M.T.; Civera, T.; Nucera, D.; Rosati, S.; Sacchi, P.; Turi, R.M. A multiplex polymerase chain reaction for the identification of cows’, goats’ and sheep’s milk in dairy products. Int. Dairy J. 2003, 13, 277–282. [Google Scholar] [CrossRef]
- Tsirigoti, E.; Katsirma, Z.; Papadopoulos, A.I.; Samouris, G.; Ekateriniadou, L.V.; Boukouvala, E. Application of triplex-PCR with an innovative combination of 3 pairs of primers for the detection of milk’s animal origin in cheese and yoghurt. J. Dairy Res. 2020, 87, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Hebert, P.D.N.; Cywinska, A.; Ball, S.L.; deWaard, J.R. Biological identifications through DNA barcodes. Proc. R. Soc. 2003, 270, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Calleja, I.; Gonzalez, I.; Fajardo, V.; Martin, I.; Hernandez, P.E.; Garcia, T.; Martin, R. Real-time TaqMan PCR for quantitative detection of cows’ milk in ewes’ milk mixtures. Int. Dairy J. 2007, 17, 729–736. [Google Scholar] [CrossRef]
- Cottenet, G.; Blancpain, C.; Golay, P.A. Simultaneous detection of cow and buffalo species in milk from China, India, and Pakistan using multiplex real-time PCR. J. Dairy Sci. 2011, 94, 3787–3793. [Google Scholar] [CrossRef]
- Deng, L.; Li, A.; Gao, Y.; Shen, T.; Yue, H.; Miao, J.; Li, R.; Yang, J. Detection of the Bovine Milk Adulterated in Camel, Horse, and Goat Milk Using Duplex PCR. Food Anal. Methods 2020, 13, 560–567. [Google Scholar] [CrossRef]
- Guo, L.; Qian, J.-P.; Guo, Y.-S.; Hai, X.; Liu, G.-Q.; Luo, J.-X.; Ya, M. Simultaneous identification of bovine and equine DNA in milks and dairy products inferred from triplex TaqMan real-time PCR technique. J. Dairy Sci. 2018, 101, 6776–6786. [Google Scholar] [CrossRef]
- Guo, L.; Ya, M.; Hai, X.; Guo, Y.-S.; Li, C.-D.; Xu, W.-L.; Liao, C.-S.; Feng, W.; Cai, Q. A simultaneous triplex TaqMan real-time PCR approach for authentication of caprine and bovine meat, milk and cheese. Int. Dairy J. 2019, 95, 58–64. [Google Scholar] [CrossRef]
- Guo, L.; Yu, Y.; Xu, W.-L.; Li, C.-D.; Liu, G.-Q.; Qi, L.; Luo, J.-X.; Guo, Y.-S. Simultaneous detection of ovine and caprine DNA in meat and dairy products using triplex TaqMan real-time PCR. Food Sci. Nutr. 2020, 8, 6467–6476. [Google Scholar] [CrossRef]
- Hai, X.; Liu, G.-Q.; Luo, J.-X.; Guo, Y.-S.; Qian, J.-P.; Ya, M.; Guo, L. Triplex real-time PCR assay for the authentication of camel-derived dairy and meat products. J. Dairy Sci. 2020, 103, 9841–9850. [Google Scholar] [CrossRef] [PubMed]
- Agrimonti, C.; Pirondini, A.; Marmiroli, M.; Marmiroli, N. A quadruplex PCR (qxPCR) assay for adulteration in dairy products. Food Chem. 2015, 187, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Reed, G.H.; Kent, J.O.; Wittwer, C.T. High-resolution DNA melting analysis for simple and efficient molecular diagnostics. Pharmacogenomics 2007, 8, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Wittwer, C.T. High-Resolution DNA Melting Analysis: Advancements and Limitations. Hum. Mutat. 2009, 30, 857–859. [Google Scholar] [CrossRef] [PubMed]
- Druml, B.; Cichna-Markl, M. High resolution melting (HRM) analysis of DNA—Its role and potential in food analysis. Food Chem. 2014, 158, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Ganopoulos, I.; Sakaridis, I.; Argiriou, A.; Madesis, P.; Tsaftaris, A. A novel closed-tube method based on high resolution melting (HRM) analysis for authenticity testing and quantitative detection in Greek PDO Feta cheese. Food Chem. 2013, 141, 835–840. [Google Scholar] [CrossRef]
- Vynck, M.; Chen, Y.; Gleerup, D.; Vandesompele, J.; Trypsteen, W.; Lievens, A.; Thas, O.; De Spiegelaere, W. Digital PCR Partition Classification. Clin. Chem. 2023, hvad063. [Google Scholar] [CrossRef]
- Saiki, R.K.; Gelfand, D.H.; Stoffel, S.; Scharf, S.J.; Higuchi, R.; Horn, G.T.; Mullis, K.B.; Erlich, H.A. Primer-Directed Enzymatic Amplification of DNA with a Thermostable DNA Polymerase. Science 1988, 239, 487–491. [Google Scholar] [CrossRef]
- Vogelstein, B.; Kinzler, K.W. Digital PCR. Proc. Natl. Acad. Sci. USA 1999, 96, 9236–9241. [Google Scholar] [CrossRef]
- Tiwari, A.; Ahmed, W.; Oikarinen, S.; Sherchan, S.P.; Heikinheimo, A.; Jiang, G.; Simpson, S.L.; Greaves, J.; Bivins, A. Application of digital PCR for public health-related water quality monitoring. Sci. Total Environ. 2022, 837, 155663. [Google Scholar] [CrossRef]
- Cutarelli, A.; Fulgione, A.; Fraulo, P.; Serpe, F.P.; Gallo, P.; Biondi, L.; Corrado, F.; Citro, A.; Capuano, F. Droplet Digital PCR (ddPCR) Analysis for the Detection and Quantification of Cow DNA in Buffalo Mozzarella Cheese. Animals 2021, 11, 1270. [Google Scholar] [CrossRef]
- Du, M.H.; Li, J.W.; Liu, Q.J.; Wang, Y.F.; Chen, E.N.; Kang, F.Y.; Tu, C.X. Rapid detection of trace Salmonella in milk using an effective pretreatment combined with droplet digital polymerase chain reaction. Microbiol. Res. 2021, 251, 126838. [Google Scholar] [CrossRef] [PubMed]
- Porcellato, D.; Narvhus, J.; Skeie, S.B. Detection and quantification of Bacillus cereus group in milk by droplet digital PCR. J. Microbiol. Methods 2016, 127, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yang, J.J.; Gai, Z.T.; Huo, S.N.; Zhu, J.H.; Li, J.; Wang, R.R.; Xing, S.; Shi, G.S.; Shi, F.; et al. Comparison between digital PCR and real-time PCR in detection of Salmonella typhimurium in milk. Int. J. Food Microbiol. 2018, 266, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.G.; Kubelik, A.R.; Livak, K.J.; Rafalski, J.A.; Tingey, S.V. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res. 1990, 18, 6531–6535. [Google Scholar] [CrossRef]
- Grover, A.; Sharma, P.C. Development and use of molecular markers: Past and present. Crit. Rev. Biotechnol. 2016, 36, 290–302. [Google Scholar] [CrossRef]
- Pottier, I.; Gente, S.; Vernoux, J.P.; Gueguen, M. Safety assessment of dairy microorganisms: Geotrichum candidum. Int. J. Food Microbiol. 2008, 126, 327–332. [Google Scholar] [CrossRef]
- Tabit, F.T. Advantages and limitations of potential methods for the analysis of bacteria in milk: A review. J. Food Sci. Technol. 2016, 53, 42–49. [Google Scholar] [CrossRef]
- Cunha, J.T.; Ribeiro, T.I.B.; Rocha, J.B.; Nunes, J.; Teixeira, J.A.; Domingues, L. RAPD and SCAR markers as potential tools for detection of milk origin in dairy products: Adulterant sheep breeds in Serra da Estrela cheese production. Food Chem. 2016, 211, 631–636. [Google Scholar] [CrossRef]
- Cunha, J.T.; Domingues, L. RAPD/SCAR Approaches for Identification of Adulterant Breeds’ Milk in Dairy Products. In PCR; Walker, J.M., Ed.; Methods in molecular biology; Springer: New York, NY, USA, 2017; Volume 1620, pp. 183–193. [Google Scholar]
- Abdel-Aziem, S.H.; Mahrous, K.F.; Abd El-Hafez, M.A.M.; Abdel Mordy, M. Genetic variability of myostatin and prolactin genes in popular goat breeds in Egypt. J. Genet. Eng. Biotechnol. 2018, 16, 89–97. [Google Scholar] [CrossRef]
- Tortorici, L.; Di Gerlando, R.; Tolone, M.; Mastrangelo, S.; Sardina, M.T. 12S rRNA mitochondrial gene as marker to trace Sicilian mono-species dairy products. Livest. Sci. 2016, 193, 39–44. [Google Scholar] [CrossRef]
- Feligini, M.; Alim, N.; Bonizzi, I.; Enne, G.; Aleandri, R. Detection of cow milk in water buffalo cheese by SYBR green real-time PCR: Sensitivity test on governing liquid samples. Pak. J. Nutr. 2007, 6, 94–98. [Google Scholar]
- Wajahat, W.; Azad, Z.R.A.A.; Nazir, S.; Nasir, G. Real Time-PCR coupled with melt curve analysis for detecting the authenticity of camel milk. J. Food Sci. Technol. 2022, 59, 1538–1548. [Google Scholar] [CrossRef]
- Liao, J.; Liu, Y.F.; Yang, L.; Li, F.P.; Sheppard, A.M. Development of a rapid mitochondrial DNA extraction method for species identification in milk and milk products. J. Dairy Sci. 2017, 100, 7035–7040. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Liu, Y.F.; Ku, T.; Liu, M.H.; Huang, Y. Qualitative and quantitative identification of adulteration of milk powder using DNA extracted with a novel method. J. Dairy Sci. 2017, 100, 1657–1663. [Google Scholar] [CrossRef] [PubMed]
- Feligini, M.; Bonizzi, I.; Curik, V.C.; Parma, P.; Greppi, G.F.; Enne, G. Detection of adulteration in Italian Mozzarella cheese using mitochondrial DNA templates as biomarkers. Food Technol. Biotechnol. 2005, 43, 91–95. [Google Scholar]
- Quigley, L.; O’Sullivan, O.; Beresford, T.P.; Ross, R.P.; Fitzgerald, G.F.; Cotter, P.D. A comparison of methods used to extract bacterial DNA from raw milk and raw milk cheese. J. Appl. Microbiol. 2012, 113, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Cordea, D.V.; Mihaiu, M. Authenticity and typicality markers to certify the traditional cheese on E.U. market. Manag. Chall. Contemp. Soc. Proc. 2016, 9, 18. [Google Scholar]
- Ponzoni, E.; Mastromauro, F.; Giani, S.; Breviario, D. Traceability of Plant Diet Contents in Raw Cow Milk Samples. Nutrients 2009, 1, 251–262. [Google Scholar] [CrossRef]
- Wilson, I.G. Inhibition and facilitation of nucleic acid amplification. Appl. Environ. Microbiol. 1997, 63, 3741–3751. [Google Scholar] [CrossRef]
- Nacham, O.; Clark, K.D.; Anderson, J.L. Extraction and Purification of DNA from Complex Biological Sample Matrices Using Solid-Phase Microextraction Coupled with Real-Time PCR. Anal. Chem. 2016, 88, 7813–7820. [Google Scholar] [CrossRef] [PubMed]
| Extraction Method | Advantages | Disadvantages | Refs. |
|---|---|---|---|
| Organic solvent extraction method | High purity; Large quantity; Intact fragments; Low cost | Use of toxic reagents; Poor reproducibility; Time-consuming | [25,26,35] |
| Salting-out extraction method | Simplicity; No use of toxic reagents; Highly efficient | Low yield; Less application range; Large quantities of raw material | [12,28,29] |
| Silicon centrifugal columns extraction method | Higher integrity and purity; Easy operation; Less time consumption | More pronounced shearing of genomic DNA; Higher cost; Lower concentration of DNA | [28,36,37] |
| Magnetic beads extraction method | Less time consumption; Easy operation; Efficient | Low yield | [33,34,36] |
| Techniques | Species Identification | Types of Dairy Products | DNA Target | Detection Limit | Ref. |
|---|---|---|---|---|---|
| PCR-RFLP | Identification of the milk sources from cow, sheep, and buffalo milk | Raw milk | cyt b gene | Qualitative analysis | [42] |
| Identification of the milk sources from cow, sheep, goat, and buffalo | Raw milk | cyt b gene | Qualitative analysis | [43] | |
| Identification of the milk sources from cow, sheep, and buffalo milk | Raw milk | κ-casein gene | Qualitative analysis | [44] | |
| Identification of the milk sources from three Egyptian goat breeds (Bariki, Damascus, and Zaraibl) | Blood sample | myostatin (MSTN) gene and prolactin (PRL) gene | Qualitative analysis | [74] | |
| Multiple PCR | Identification of the cheese sources from cow, sheep, and goat milk | Cheese | Mitochondrial 12S and 16S rRNA genes. | 0.5% detection limit | [45] |
| Identification of the milk sources from cow, goat, and sheep milk | Raw milk | cyt b genes (cow and sheep) D-loop range (goats) | 0.1% detection limit. | [46] | |
| Identification of the milk sources from camel, horse, and goat | Raw milk | 16S rRNA gene and D-LOOP range | 0.1% detection limit. | [50] | |
| Identification of the milk sources from cow, goat, sheep, and buffalo milk | Raw milk | cyt b gene and 12S rRNA | 0.1% detection limit. | [55] | |
| Identification of the milk sources from cow, sheep, and goat milk | Blood sample | 16S rRNA (sheep) and 12S RNA (cow, sheep, and goat) | 0.1% detection limit. | [75] | |
| Real-time TaqMan PCR | Identification of goat milk or cow milk from sheep milk | Raw milk | 12S rRNA | Linear dynamic range of 0.6–10% | [48] |
| Identification of the milk sources from cow and mare milk | Raw milk | 12S rRNA | 1 pg of cow DNA; 1 pg of mare DNA, | [51] | |
| Identification of the cheese, and milk sources from cow and goat milk | Cheese and raw milk | 12S rRNA | 0.005 ng/μL in milk and 0.01 ng/μL in cheese | [52] | |
| Identification of the cheese and milk sources of sheep and goat milk | Cheese and raw milk | 12S rRNA | 0.005 ng/μL in milk and 0.01 ng/μL in cheese | [53] | |
| Identification of the milk, yogurt, cheese, milk powder, and milk beverage sources from cow and camel milk | Milk, yogurt, cheese, milk powder, and milk beverage | 12S rRNA | 0.0025 to 0.001 ng/μL (milk); 0.5 to 0.001 ng/μL (yogurt); 1 to 0.05 ng/μL (cheese); 0.01 ng/μL (milk powder); 0.001 ng/μL (milk beverage) | [54] | |
| Real-time SYBR Green PCR | Identify the cheese source from cow and buffalo milk | Cheese | Cytochrome oxidase subunit 1 (coI) | - a | [47] |
| Identification of the milk sources from cow, and buffalo | Cheese | Cytochrome oxidase subunit 1 (coI) | 0.5 ng/μL | [76] | |
| Identification of the milk sources from camel, cow, and goat | Milk, milk powder, and milk soap | Follicle stimulating hormone receptor (FSHR) gene (camel and goat) and 12S RNA (cow) | Rang of 0.001–0.002% | [77] | |
| Digital PCR | Identify the cheese source from cow and buffalo milk | Cheese | cyt b genes | 0.1% detection limit. | [64] |
| RAPD-PCR | Identify the cheese source from four sheep breeds’ milk | Cheese | Sequence-characterized amplified region | Qualitative analysis | [73] |
| PCR Techniques | Cost | Detect Time | Analysis Procedure | Other |
|---|---|---|---|---|
| PCR-RFLP | Low | Fast | Not quantitative analysis. | Simple. |
| Multiple PCR | Low | Fast | Both qualitative and quantitative analyses. | High-throughput; False-positive result |
| Real-time PCR | Low | Fast | Both qualitative and quantitative analyses. | Excellent sensitivity; Specificity; High consistency; False-positive result |
| Digital PCR | High | Fast | Both qualitative and quantitative analyses. | Accurate; Highly reproducible; Narrow dynamic range |
| RAPD-PCR | Low | Fast | Not quantitative analysis. | Independent from Specific primers; Ease to use; Less interfering factors |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, B.; Yu, M.; Xu, W.; Chen, L.; Han, J. Comparison of PCR Techniques in Adulteration Identification of Dairy Products. Agriculture 2023, 13, 1450. https://doi.org/10.3390/agriculture13071450
Li B, Yu M, Xu W, Chen L, Han J. Comparison of PCR Techniques in Adulteration Identification of Dairy Products. Agriculture. 2023; 13(7):1450. https://doi.org/10.3390/agriculture13071450
Chicago/Turabian StyleLi, Baiyi, Mingxue Yu, Weiping Xu, Lu Chen, and Juan Han. 2023. "Comparison of PCR Techniques in Adulteration Identification of Dairy Products" Agriculture 13, no. 7: 1450. https://doi.org/10.3390/agriculture13071450
APA StyleLi, B., Yu, M., Xu, W., Chen, L., & Han, J. (2023). Comparison of PCR Techniques in Adulteration Identification of Dairy Products. Agriculture, 13(7), 1450. https://doi.org/10.3390/agriculture13071450








