Abstract
Grapevine virus A (GVA) is a phloem-restricted virus (genus Vitivirus, family Betaflexiviridae) that cause crop losses of 5–22% in grapevine cultivars, transmitted by different species of pseudococcid mealybugs, the mealybug Heliococcus bohemicus, and by the scale insect Neopulvinaria innumerabilis. In this work, we studied the genetic structure and molecular variability of GVA, ascertaining its presence and spread in different commercial vineyards of four Sicilian provinces (Italy). In total, 11 autochthonous grapevine cultivars in 20 commercial Sicilian vineyards were investigated, for a total of 617 grapevine samples. Preliminary screening by serological (DAS-ELISA) analysis for GVA detection were conducted and subsequently confirmed by molecular (RT-PCR) analysis. Results showed that 10 out of the 11 cultivars analyzed were positive to GVA, for a total of 49 out of 617 samples (8%). A higher incidence of infection was detected on ‘Nerello Mascalese’, ‘Carricante’, ‘Perricone’ and ‘Nero d’Avola’ cultivars, followed by ‘Alicante’, ‘Grecanico’, ‘Catarratto’, ‘Grillo’, ‘Nerello Cappuccio’ and ‘Zibibbo’, while in the ‘Moscato’ cultivar no infection was found. Phylogenetic analyses carried out on the coat protein (CP) gene of 16 GVA sequences selected in this study showed a low variability degree among the Sicilian isolates, closely related with other Italian isolates retrieved in GenBank, suggesting a common origin, probably due to the exchange of infected propagation material within the Italian territory.
Keywords:
grapevine disease; GVA; Vitivirus; Betaflexiviridae; DAS-ELISA; RT-PCR; phylogenetic analysis 1. Introduction
Grapevine (Vitis vinifera L.) has been cultivated for several millennia and is one of the most economically important and extensive crops worldwide. The global production of over 78 million tons covers an area of about 7 million ha, with more than 3 million ha located in the European continent, especially in the central and southern areas, according to the latest data available []. On the European continent, Italy has the highest number of grapevine cultivars (cvs), and every region has its own autochthonous cultivars, with a total production of over 8.1 million tons and an area of 722,000 ha [].
In particular, in the last 20 years the Sicily region, thanks to the favorable weather conditions, which allow the production of excellent wines obtained by autochthonous and imported cultivars, has become one of the most important Italian regions for the grapevine industry; the most common cultivated grapevine cultivars are ‘Catarratto’, ‘Grillo’, ‘Nero d’Avola’ and ‘Zibibbo’ [].
In order to obtain excellent products with a good yield in terms of production, it is important to consider all challenges related to the presence of several pests and pathogens in grapevine crops that play a main role, causing heavy losses, shortening the productive life and endangering the survival of affected grapevines []. The impacts of climate change on many aspects of the grapevine growing system and on pathogens and insect pests should also be taken into consideration [,]. Different diseases and disorders affect this crop, caused by fungi, such as Diplodia seriata, Lasiodiplodia sp., Neofusicoccum parvum and N. vitifusiforme [], but also by systemic pathogens, including bacteria, such as Agrobacterium tumefaciens [] and Xylophilus ampelinus [], and viruses. Grapevines host one of the largest number of viruses among cultivated crop species [], which are considered the most dangerous and damaging pathogens to viticulture worldwide. To date, a total of 86 species of grapevine viruses, belonging to 17 families and 34 genera, have been identified []. To be able to contain these dangerous pathogens, it is extremely fundamental to adopt a correct disease management and know their genetic diversity and spread [,,,].
The management of grapevine virus diseases depends primarily on preventive measures, in order to contain and suppress the viral inoculum in field and reduce their presence in propagation material, and also through clonal selection activities and phytosanitary measures [,].
For this reason, the most effective way to control grapevine viruses is the use of certified plant material, satisfying the conditions and requirements to be qualified as C.A.C. (Conformitas Agraria Communitatis) [], that involve screening for the presence of the five main grapevine viruses: Grapevine leafroll-associated virus 1—GLRaV-1 (family, Closteroviridae; genus, Ampelovirus); Grapevine leafroll-associated virus 3—GLRaV-3 (family, Closteroviridae; genus, Ampelovirus); Grapevine fanleaf virus—GFLV (family, Secoviridae; genus, Nepovirus); Arabis mosaic virus—ArMV (family, Secoviridae; genus, Nepovirus); Grapevine virus A—GVA (family, Betaflexiviridae; genus, Vitivirus; and Grapevine fleck virus—GFkV (family, Tymoviridae; genus, Maculavirus) only for rootstocks. The absence of these viruses in nursery must be confirmed through official inspections, as they are considered harmful pathogens by the European Commission directive (2005/43/EC) [].
Grapevine virus A, the type species of the genus Vitivirus [], is a phloem-restricted virus which multiplies in phloem parenchyma cells []. The GVA virions are constituted by helically flexuous filamentous particles, measuring approximately 800 × 12 nm, with a pitch of 3.6–4.0 nm and about 10 subunits per turn of the helix []. The genome of GVA consists of a single molecule of positive-sense single-stranded RNA (ssRNA+) of about 7.6 kb in size, capped at the 5′ terminus with m7G and polyadenylated at the 3′ terminus, containing five slightly overlapping open reading frames (ORFs) in the positive strand encoding, respectively, a 194-kDa polypeptide with conserved motifs of replication-related proteins (ORF 1), a 19 kDa product with unknown functions (ORF 2), the movement protein (ORF 3), the coat protein (ORF 4) and a 10 kDa product (ORF 5) with nucleotide-binding properties which, like in other viruses such as grapevine virus B (GVB), is suspected to be a suppressor of gene silencing [,,]. Regarding the molecular criteria for demarcation of Vitivirus genus, as reported by Adams and coworkers [], viral species belonging to this genus have the amino acid sequences of any gene product that differs by more than 10%, compared with other genera belonging to Betaflexiviridae family. GVA is widely distributed throughout the world, probably due to the inadvertent international dissemination of the virus in infected grapevine germplasm before it has been identified and its detection methods developed []. GVA is spread in almost all areas where grapevine is cultivated, including Africa, Asia, North and South America, Europe and Oceania []. In the last ten years, GVA has been observed also in North Macedonia [], Russia [] and Pakistan [].
In susceptible grapevine cultivars, GVA induces the following symptoms: stem pitting [,], Kober stem grooving [] and rugose wood [,]. GVA causes crop losses ranging from 5 to 22% in grapevine cultivars [] and decline and death of grapevines if co-infected by a causal agent of leafroll disease []. This virus is transmitted from infected to healthy grapevines in a non-circulative semi-persistent manner by different species of pseudococcid mealybugs (Pseudococcus longispinus, Ps. affinis [P. viburni], Planococcus citri and P. ficus) [,,,,]. Recently, GVA transmission by the mealybug Heliococcus bohemicus [] and by the scale insect Neopulvinaria innumerabilis was reported [].
The objective of this study was to evaluate the GVA presence and spread in different cultivars collected in twenty commercial vineyards in Sicily. Additional objectives were to ascertain its genetic structure and molecular variability.
2. Materials and Methods
2.1. Field Surveys and Samples Collection
During December 2020 and January–February 2021, a total of 617 grapevine samples were collected in different field surveys, in order to study the presence of GVA in Sicily and evaluate its genetic structure and molecular variability. Field surveys were carried out in 20 commercial vineyards in Sicily located in the provinces of Trapani, Agrigento, Caltanissetta and Ragusa. The hierarchical sampling scheme developed by Gottwald and Hughes [] was applied to randomly collect the grapevine materials, with a minor correction for adapting the scheme to grapevine plants; all collected samples were marked by GPS using the Planthology mobile application []. A total of eleven autochthonous cultivars were sampled: ‘Grillo’, ‘Zibibbo’, ‘Perricone’, ‘Catarratto’, ‘Nerello Mascalese’, ‘Nero d’Avola’, ‘Carricante’, ‘Nerello Cappuccio’, ‘Grecanico’, ‘Alicante’ and ‘Moscato’ (Table 1). In detail, ‘Grillo’, ‘Zibibbo’, ‘Perricone’ and ‘Catarratto’ are among the most widespread cultivars. For this reason, the sampling was made according to the major presence of the different cultivars in Sicily. Moreover, it is important to clarify that the higher number of samples for ‘Grillo’ and ‘Zibibbo’ cultivars is due to the major extension of the area of some commercial vineyards than others; in these cases, the samples number for each cultivar increased. To perform both serological and molecular analyses, each sample collected, consisting of 4 dormant cuttings, was split into two subsamples.

Table 1.
Number of samples for each cultivar analyzed by DAS-ELISA for GVA.
2.2. Preliminary Screening by Serological Analysis
The preliminary screening was performed by double-antibody sandwich enzyme-linked immunosorbent assay (DAS-ELISA) [], using capture polyclonal and conjugate monoclonal line F5 antibodies to GVA (Agritest srl, Valenzano, Italy). Five hundred mg of phloem tissue of each sample was mixed and homogenized with 5 mL extraction buffer (0.5 M Tris-HCl, 0,14 M NaCl, 2% PVP MW 24000, 1% PEG MW 6000, and 0.05% Tween 20 in 1 L of distilled water, pH 8.2), and a 1:10 dilution (w/v) of each sample was used for DAS-ELISA, following the manufacturer’s instructions. Lyophilized plant tissue infected by GVA and healthy plant tissue supplied in the commercial kit (Agritest srl, Valenzano, Italy) were resuspended in 2 mL of distilled water and used as positive and negative controls, respectively. The optical densities at 405 nm (OD405) were measured two hours after the addition of the p-nitro-phenylphosphate substrate, using an AMR-100 microplate reader (Hangzhou Allsheng Instruments, Hangzhou, China). As reported in the protocol supplied by Agritest srl, the sample was considered positive if its OD405 value was at least twice the negative control value.
2.3. Total RNA Extraction
All samples showing as positive by DAS-ELISA were used for the subsequent molecular analysis. In addition, all the samples from ‘Moscato’ cultivar, the only cultivar that was negative by DAS-ELISA assay, were also analyzed by molecular assay. Total RNA was extracted by using a GenUP Plant RNA kit (Biotechrabbit GmbH, Berlin, Germany) from 100 mg of phloem tissue of each sample homogenized as described above, according to the manufacturer’s instructions, and eluted in 50 µL RNase-free water. The total RNA concentration was measured twice with a UV–Vis Nanodrop 1000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA); dilutions were adjusted to approximately 50 ng/µL and stored at −20 °C until subsequent analyses.
2.4. Molecular Analyses
Reverse-transcription polymerase chain reaction (RT-PCR) tests were carried out on samples that were positive for GVA by DAS-ELISA and on all ‘Moscato’ samples. In detail, the full GVA coat protein (CP) gene and flanking regions (740 nt) were amplified by end-point RT-PCR, using the GVA-CPF6356/GVA-CPR7096 primer pair []. Two-step end-point RT-PCR was performed. The reverse transcription (RT) was performed in a final reaction volume of 20 µL, containing 3 µL of total RNA extract, 0.4 mM dNTPs, 4 µL of 5X First Strand Buffer [50 mM Tris-HCl (pH 8.3), 40 mM KCl, 6 mM MgCl2] (Thermo Fisher Scientific, Waltham, MA, USA), 1 µM of reverse primer (GVA-CPR7096), 20U of M-MLV reverse transcriptase (Thermo Fisher Scientific, Waltham, MA, USA) and RNase-free water to reach the final volume. The cycling conditions were as follows: initial incubation at 65 °C for 10 min, reverse transcription at 42 °C for 45 min, and enzyme inactivation at 95 °C for 10 min. The cDNA obtained was used for the subsequent PCR assay, performed in a final reaction volume of 25 µL, containing 2 µL of cDNA, 20 mM Tris–HCl (pH 8.4), 50 mM KCl, 3 mM MgCl2, 0.4 mM dNTPs, 1 µM of each primer, 2U of Taq DNA polymerase (Thermo Fisher Scientific, Waltham, MA, USA) and RNase-free water to reach the final volume. RT-PCR was carried out in a MultiGene OptiMax thermal cycler (Labnet International Inc., Edison, NJ, USA). The cycling conditions were as follows: initial denaturation at 95 °C for 5 min, followed by 40 cycles of 95 °C for 30 s, 55 °C for 45 s and 72 °C for 1 min, and a final elongation step at 72 °C for 10 min []. Total RNA derived from GVA-infected grapevine (Agritest srl, Valenzano, Italy) was used as positive control, while molecular-grade water and total RNA extracted from healthy grapevine plant (Agritest srl, Valenzano, Italy) were used as negative controls. The obtained RT-PCR products were electrophoresed on 1.5% agarose gel, stained with SYBR Safe (Thermo Fisher Scientific, Waltham, MA, USA), and visualized by UV light.
2.5. Sequence Analyses
A total of 16 out of 49 GVA obtained amplicons by RT-PCR, corresponding to ~33% of positive samples, were purified by using a QIAquick PCR purification kit (Qiagen, Hilden, Germany), following the manufacturer’s instructions. In detail, four obtained amplicons of ‘Nerello Mascalese’; two of ‘Carricante’, ‘Perricone’ and ‘Grillo’; and one of ‘Nero d’Avola’, ‘Grecanico’, ‘Catarratto’, ‘Alicante’, ‘Zibibbo’ and ‘Nerello Cappuccio’ cvs were selected. Since for each grapevine cultivar the positive samples were from the same field, it was possible to hypothesize a low genetic variability among the different isolates; for these reasons, sixteen samples were selected for sequencing. The selected amplicons were sequenced in both directions, using an ABI PRISM 3100 DNA sequence analyzer (Applied Biosystems, Foster City, CA, USA). The sequences obtained were trimmed to remove the flanking regions, leaving only 597 nt CP gene, and deposited in GenBank. The nucleotide sequences obtained were used for subsequent phylogenetic analyses, with other 31 sequences of GVA CP gene retrieved by GenBank from different countries (Japan, AB039841; Czech Republic, EU008560; Italy, from MG717810 to MG717813, and X75433; Greece, LK937679 and LK937680; Australia, MT070960 and MT070963; France, MK404722; China, JF754577, JF754578 and DQ911145; Macedonia, KF594435; South Africa, AF441235, DQ787959, DQ855084 and DQ855085; Croatia, MF979533; Brazil, AF494187, AY340581 and KX828703; Iran, MG977013 and MG977014; Israel, AF007415; Poland, JN565033 and JN861000; Canada, JX559641; USA, KF013763).
Multiple nucleotide sequence alignment was performed by using the CLUSTALW algorithm [], and a mathematical model was applied to estimate the number of nucleotide substitutions, considering nucleotide frequencies and instantaneous rate change. The model that fitted best was the Kimura 2-parameter (K2) [], modeled by using a discrete gamma distribution (+G) = 0.3774 with two rate categories.
Phylogenetic relationships were inferred by the maximum-likelihood method (ML), with 1000 bootstrap replicates to estimate the statistical significance of each node [], performed with the MEGA X software []. Initial trees for the heuristic search were obtained automatically by applying neighbor-joining and BIONJ algorithms to a matrix of pairwise distances estimated by using the maximum composite likelihood (MCL) approach, and then selecting the topology with superior log likelihood value. All analyses were performed by using MEGA X software [].
The presence of recombination events between the nucleotide sequences obtained from GVA Sicilian isolates was detected by the GENECONV, Bootscan, MaxChi, SiScan, 3Seq, and RDP algorithms [] using RDP4 software (v.4.39); RDP4 parameters were set as default values. Only concordant results of in silico analysis between different algorithms were considered as a positive result.
Nucleotide sequence diversity of GVA CP gene was estimated within and between different countries, which were considered as geographic populations, using the Jukes–Cantor model [].
The role of natural selection at the molecular level in the GVA Sicilian isolates was studied by evaluating the rate of synonymous substitutions per synonymous site (dS) and the rate of nonsynonymous substitutions per nonsynonymous site (dN), separately; these values were estimated by the Pamilo-Bianchi-Li method [], using the MEGA X software.
Lastly, the pairwise percent identities of GVA CP isolates were calculated within the GVA isolates from Sicily (Italy), and between the other reference isolates from the other countries, using the SDT v1.2 software [].
3. Results
3.1. Incidence of GVA Infection in Sicilian Vineyards
The presence of GVA infection was evaluated by a first screening using a DAS-ELISA assay in a total of 617 samples from 11 cultivars from commercial vineyards located in different Sicilian provinces (Trapani, Agrigento, Caltanissetta and Ragusa). Altogether, 49 out of 617 samples gave a positive result, representing 7.9% of the infection (Table 2). OD405 values of DAS-ELISA-positive samples are reported in Table S1. The samples that showed an OD405 value less than twice the value of the negative control were considered negative and therefore not reported, as suggested by the manufacturer’s instructions. The ‘Moscato’ cultivar was the unique cultivar that came back negative for GVA infection; in order to avoid possible false-negative results, all ‘Moscato’ samples were also analyzed by end-point RT-PCR.

Table 2.
Number of samples collected and incidence percentage for each cultivar tested by DAS-ELISA and end-point RT-PCR for Grapevine virus A.
3.2. Polymerase Chain Reaction and Sequencing
Based on the data obtained from the DAS-ELISA assay, a total of 49 positive samples were analyzed by end-point RT-PCR, using a specific primer pair for GVA detection. Moreover, ‘Moscato’ samples were analyzed to confirm the absence of GVA infection. All 49 samples gave positive results for GVA, obtaining the expected amplicon size of 740 nt (Table 2).
The highest percentage of positive samples was recorded in the cultivars ‘Nerello Mascalese’, ‘Carricante’, ‘Perricone’ and ‘Nero d’Avola’, with an incidence of 41.9%, 20.0%, 9.5% and 6.3%, respectively (Table 2). Meanwhile, a lower incidence was detected in the cultivars ‘Nerello Cappuccio’, ‘Grillo’, ‘Catarratto’, ‘Grecanico’ and ‘Alicante’, ranging from of 4.2% to 4.8% (Table 2). The lowest incidence was detected in the ‘Zibibbo’ cultivar, with a percentage of 0.9% in a total of 106 samples analyzed, while no ‘Moscato’ samples were positive for GVA infection by end-point RT-PCR, confirming the absence of GVA in this cultivar. The CP GVA sequences obtained were deposited in GenBank under the accession numbers from OL763357 to OL763372.
Regarding the GVA distribution in the four Sicilian provinces, the highest incidence was observed in Agrigento province (12.7%), followed by Trapani, Caltanissetta and Ragusa provinces (8.4%, 7.3% and 3.3%, respectively) (Table 3).

Table 3.
Number of samples collected and incidence percentage of Grapevine virus A for each vineyard.
All analyzed plots showed a variable range of GVA infection for each province. Specifically, in Trapani province, the infection percentage ranged from 16.7% (1T) to 6.4% (5T), while no infection was found in plot 4T. In Agrigento province, the 10A plot showed the highest percentage among all 20 plots analyzed (33.3%), followed by 7A plots and 8A (20.0% and 10.0%, respectively), while no infection was found in the 6A and 9A plots. In Ragusa province, infection rates ranged from 6.7% to 3.3%, with two plots (11R and 14R) without GVA infection. Finally, plots analyzed in Caltanissetta province showed a range from 26.7% (19C) to 3.3% (20C), while only two plots (17C and 18C) showed no GVA infection (Table 3).
3.3. Phylogenetic Analyses
In order to investigate the phylogenetic relationships between GVA isolates retrieved in this work and isolates from other countries, a total of 16 Sicilian GVA sequences obtained in this study were used for phylogenetic analyses on the CP gene, as well as another 5 Italian isolates; one from Czech Republic, France, North Macedonia, Croatia, Canada, USA, Japan and Israel; 2 from Greece, Poland, Australia and Iran; 3 from China and Brazil; and 4 from South Africa, retrieved from GenBank database. The phylogenetic tree reported in Figure 1 related to the evolutionary relationships among GVA sequences used in the present work showed that GVA isolates were separated into two statistically significant clusters: the first one (A) including isolates from different countries, probably suggesting a higher exchange of genetic material between these countries, and the second one (B) including one isolate from USA, Canada, China and South Africa. In detail, all Sicilian GVA isolates were grouped into the same subclade (I) within the first cluster (Figure 1) with other three Italian GVA isolates showing a low variability, while the isolates from other countries were grouped into another two subclades (II and III).
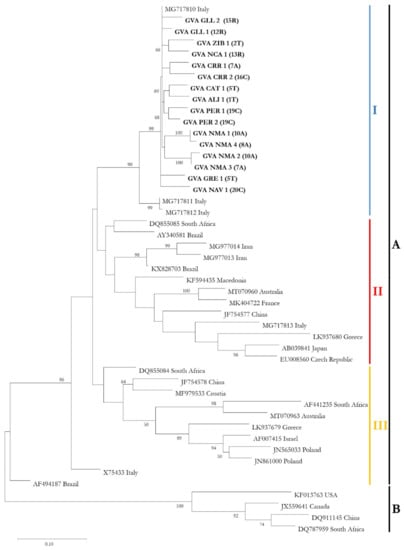
Figure 1.
Phylogenetic relationships between coat protein genes of Italian Grapevine virus A (GVA) isolates (16 Sicilian sequences obtained in the present work; 5 Italian isolates retrieved from GenBank) and isolates from other countries. The evolutionary history was inferred by using the maximum-likelihood method (ML) based on the Kimura 2-parameter model (K2) with bootstraps of 1000 replications, conducted with MEGA X program. Only bootstrap values ≥ 50% are indicated in the nodes. The sequences obtained in the present work are in bold. In brackets, the vineyard ID of origin (reported in Table 3). ‘A’ and ‘B’ letters indicate the two clusters obtained, while ‘I’, ‘II’ and ‘III’ indicate the three subclades within the cluster A.
The Sicilian isolates retrieved from all the cultivars that tested positive for GVA were grouped and closely related into the same subclade with three Italian isolates found in Vitis vinifera cv. Italia (MG717810, MG717811 and MG717812).
3.4. Recombination Analyses
Recombination analyses were performed to detect the presence of recombination events between the nucleotide sequences obtained from GVA Sicilian isolates, showing that no candidate recombinant events were detected in the Sicilian isolates by GENECONV, Bootscan, MaxChi, and RDP algorithms; however, SiScan and 3Seq algorithms identified putative recombination events among the Sicilian isolates analyzed. In detail, the SiScan algorithm detected one event in the GVA-PER-1 isolate, with a beginning breakpoint at position 137 (nt) and ending breakpoint at position 330 (nt) (major parent GVA-NAV-1 and minor parent GVA-NAV-4, with a 96.3% and 97.9% of similarity, respectively) and an average p-value of 2.380 × 10−02, while the 3Seq algorithm detected one recombination event in the GVA-CRR-1 isolate, with a beginning breakpoint at position 138 (nt) and ending breakpoint at position 505 (nt) (major parent GVA-NAV-1 and minor parent GVA-CRR-2, with a 98.7 and 97.8% of similarity, respectively) and an average p-value of 2.541 × 10−02. In both isolates, GVA-PER-1 and GVA-CRR-1, the major and minor parents, belonged to the Sicilian GVA isolates (Table 4).

Table 4.
Putative recombinant events detected among the Sicilian GVA isolates.
3.5. Nucleotide Diversity and Selection Pressure Analyses
The analysis of the nucleotide diversity showed a very low differentiation within Italian isolates (0.0681 ± 0.015) suggesting a common origin. A higher differentiation between isolates from Italy and those from Japan, Czech Republic, Greece, Australia, France, Macedonia, South Africa, Croatia, Brazil, Iran, Israel and Poland (0.1598 ± 0.0346, 0.1517 ± 0.0327, 0.1679 ± 0.0315, 0.1549 ± 0.0290, 0.1442 ± 0.0310, 0.1242 ± 0.0266, 0.1691 ± 0.0441, 0.1196 ± 0.0256, 0.1362 ± 0.0259, 0.1435 ± 0.0270, 0.1616 ± 0.0347, 0.1650 ± 0.0323, respectively) was observed. In addition, the highest level of differentiation was retrieved between Italian and Chinese, Canadian and American isolates (0.1847 ± 0.0515, 0.2665 ± 0.0568 and 0.2741 ± 0.0584, respectively) (Table S2). It was impossible to calculate the nucleotide diversity for Japan, Czech Republic, France, Macedonia, Croatia, Israel, Canada and USA, because only one sequence for each group was available in GenBank.
The role of natural selection at the molecular level was studied on the CP gene of the Sicilian GVA isolates, showing dN and dS values of 0.053 and 0.535, respectively, with a dN/dS ratio of 0.099. These values confirm the hypothesis of negative selection.
Finally, the pairwise percent identity was calculated to obtain the average percent identity over the alignment; values including the Sicilian CP sequences ranged from 93 to 100% (nt) (Figure 2), while when the CP sequences included both the Sicilian and reference isolates, the range was lower, from 76 to 100% (nt) (Figure 3).
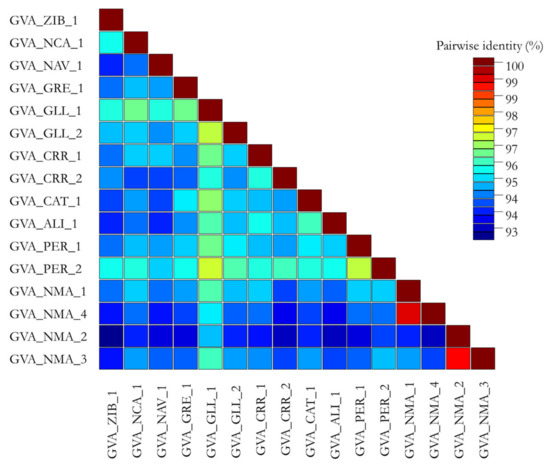
Figure 2.
Graphical representations of pairwise percent identity of nucleotides of the sequenced CP gene between the GVA isolates from Sicily (Italy) using Sequence Demarcation Tool v1.2 program. Each colored key represents a percentage of the identity score between two sequences.
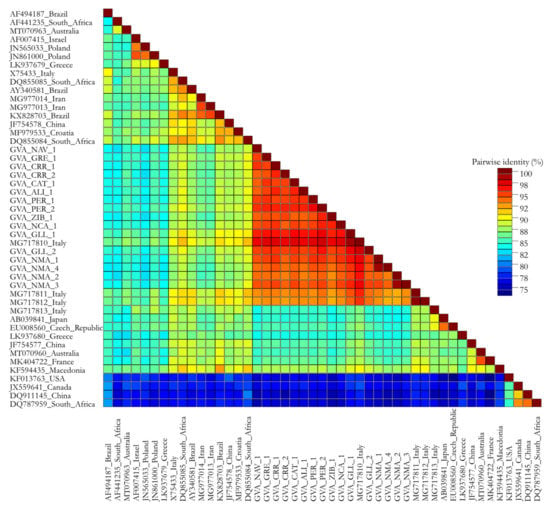
Figure 3.
Graphical representations of pairwise percent identity of nucleotides of the sequenced CP gene between the GVA isolates from Sicily (Italy) and reference isolates using Sequence Demarcation Tool v1.2 program. Each colored key represents a percentage of the identity score between two sequences.
4. Discussion
In recent decades, the most important tree and horticultural crops have suffered heavy losses caused by the emergence and/or recrudescence of new viral pathogens in one of the most important countries of the Mediterranean basin, such as Italy [,,,].
Grapevine virus A is one of the most regularly detected viruses in grapevine and have been reported in all major wine-producing regions of the world [].
The survey conducted in this study showed a different incidence of GVA infection in several grapevine cultivars, which represent the most widespread and important autochthonous species in Sicily.
In detail, a higher incidence of GVA presence was detected on ‘Nerello Mascalese’, (41.9%), ‘Carricante’, ‘Perricone’ and ‘Nero d’Avola’ cultivars; followed by ‘Alicante’, ‘Grecanico’, ‘Catarratto’, ‘Grillo’, ‘Nerello Cappuccio’; and lastly, ‘Zibibbo’ with the lowest incidence (0.9%); meanwhile, in ‘Moscato’ cultivar, no infection was found.
As regards the GVA incidence in each plot, a variable range of infection was detected for each province. The lower percentage of infection found in Ragusa province plots is probably due to a lower diffusion of grapevine cultivation than the other provinces analyzed, a reduced genetic drift and the ‘recent’ introduction of grapevine cultivation in this province, with an increased use of healthy propagation material.
The major GVA infection incidence observed in some cultivars could be explained by the fact that these cultivars can be more susceptible to GVA infection than others. In addition, it could be hypothesized that the absence of the pathogen in the ’Moscato’ cultivar is due to the different cultivation practices used for plant growth and/or a low introduction in Sicily of GVA-infected propagation material of this cultivar.
Moreover, the extensive use of Vitis rupestris Scheele in the last century as rootstock, imported from America, that allowed the eradication of phylloxera problems, and the use of uncertified propagative material within Italian territory, probably favored the wide spread of several viruses in almost all areas of grapevine cultivation, also explaining the presence of GVA infections in the autochthonous Sicilian cultivars [].
After establishing in Italy, also due to a strong deficiency of the certification program, GVA, like other viral pathogens, probably established and spread in the Sicilian territory. However, in recent decades, the introduction of the certification system (Conformitas Agraria Communitatis—C.A.C.) and the continuous and massive use of chemical treatments used for the control of the pseudococcid mealybugs, mealybug and scale insects, presumably could explain the low differentiation and the low spread of GVA in Sicily. In fact, since all the analyzed sequences are genetically close to the Italian sequences retrieved in GenBank and due to the low level of variability found among Sicilian isolates, regardless of the cultivar from which they were isolated and the absence of recombination events (except for two putative recombination events between the Sicilian isolates), we hypothesize a common origin.
As with other grapevine viruses, GVA can perpetuate as genetically diverse populations, defined as quasispecies []. However, GVA populations during their life cycle and perpetuation of its diversity, such as cell-to-cell movement, mixed virus infections, systemic movement within the host plant and during horizontal vector-mediated transmissions, can be affected by repeated bottleneck events [,,]. As a consequence of that, the overall genetic structure of GVA populations within an individual grapevine is dynamic and may be shaped during each growing season by these different variables. Although many of these mutant virus populations are suppressed from the source grapevine during pruning soon after dormancy, new variants can be generated anew during each growing season and spread to progeny plants (via cuttings used for new plantings); in this way, new generations of variants can be produced within each individual descendent [].
In this scenario, it is important to monitor the territory, the sanitary status of plant propagation material, the disease spread, the genetic diversity and molecular variability of these pathogens [,,], thus reducing the production for the livelihood of both the local grape and wine industries.
The spread of GVA, as with all viruses, can be countered through the application of preventive measures. Grapevine certification schemes, which require the GVA absence and other grapevine viruses in certified “virus-tested” and “virus-free” plants, are implemented and carried out by state-recognized certification services, which operate under government control through rules and standards established by legislation, preventing the wide spread of viral diseases that, in many cases, is favored by latent infections without any symptoms [].
Moreover, disease control and phytosanitary methods could be successfully achieved applying different techniques, such as heat therapy [], in vitro culture [], somatic embryogenesis using callus of anthers and ovaries [,] and cryopreservation [], obtaining virus-free plants. Since there is no natural resistance available for GVA, different attempts have also been made by inserting GVA sequences that encode for viral coat or movement proteins into the genomes of grapevine and/or herbaceous experimental hosts [,,,] to produce resistant transgenic plants. For vector controls, no field trials and reports for their chemical and/or biological control have been reported [], so it is extremely important that growers monitor the vectors and the symptomatic plants in order to avoid the establishment and spread of this virus, especially in the presence of co-infection with other grapevine viruses [].
The implementation of reliable diagnostic techniques for early GVA detection is extremely important to control its diffusion and provide technical support for the screening of GVA-free grapevine seedlings. Both serological (DAS-ELISA) and molecular (RT-PCR) tests give satisfactory and reproducible detection. In addition, quantitative RT-PCR and multiplex-PCR [,,,] can be adopted as more sensitive diagnostic techniques which, when combined with rapid and low-cost sample extraction methods reducing the processing time, allow the simultaneous analysis of multiple samples with reduced total cost for a single analysis []. Lastly, GVA rapid and reliable detection can be achieved with RT-LAMP (reverse-transcription loop-mediated isothermal amplification) assay [] with higher sensitivity over ELISA and PCR techniques, especially in the presence of inhibitors and low viral titer [].
5. Conclusions
Overall, this study represents the first extensive survey on GVA’s genetic structure and molecular variability in Sicily, providing useful data of most important red- and white-berried Sicilian cvs sanitary status.
In conclusion, our study revealed a significant presence of GVA in Sicilian vineyards; in order to clarify the GVA genetic population structure in the Sicily region and improve its containment and management, further analyses of more clones from single Sicilian GVA isolates would be useful to verify the presence of sequence variants and intra-isolate genetic diversity.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/agriculture12060835/s1, Table S1: Optical density (OD405) values of DAS-ELISA positive samples. Table S2: Nucleotide diversitya of Grapevine virus A (GVA) in different geographical populations.
Author Contributions
Conceptualization, S.D. and S.P.; methodology, A.G.C., S.B., S.D. and S.P.; software, A.G.C., S.B. and S.D.; validation, A.G.C., S.B., S.M., S.D. and S.P.; formal analysis, A.G.C., S.B. and A.R.; investigation, A.G.C., S.B., A.R., S.D. and S.P.; resources, S.P. and S.D.; data curation, A.G.C., S.D. and S.P.; writing—original draft preparation, A.G.C., S.D. and S.P.; writing—review and editing, A.G.C., S.B., A.R., S.M., S.D. and S.P.; visualization, A.G.C., S.B., A.R., S.M., S.D. and S.P.; supervision, S.D. and S.P.; project administration, S.D. and S.P.; funding acquisition, S.D. and S.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank “CON-0339—Analisi certificazione virus vite” project, for the technical support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Food and Agriculture Organization of the United Nations (FAO). Transforming Our World: The 2030 Agenda for Sustainable Development. Available online: http://www.fao.org/ (accessed on 8 January 2022).
- Istituto Nazionale di Statistica—ISTAT. Available online: www.dati.istat.it (accessed on 8 January 2022).
- Ministero delle Politiche Agricole, Alimentari e Forestali—Catalogo Nazionale delle Varietà di Vite. Available online: http://catalogoviti.politicheagricole.it/catalogo.php (accessed on 8 January 2022).
- Martelli, G.P. Directory of virus and virus-like diseases of the grapevine and their agents. J. Plant Pathol. 2014, 96, 1–136. [Google Scholar] [CrossRef]
- Bois, B.; Zito, S.; Calonnec, A. Climate vs grapevine pests and diseases worldwide: The first results of a global survey. OENO One 2017, 51, 133–139. [Google Scholar] [CrossRef]
- Reineke, A.; Thiéry, D. Grapevine insect pests and their natural enemies in the age of global warming. J. Pest Sci. 2016, 89, 313–328. [Google Scholar] [CrossRef]
- Mondello, V.; Lo Piccolo, S.; Conigliaro, G.; Alfonzo, A.; Torta, L.; Burruano, S. First report of Neofusiccoccum vitifusiforme and presence of other Botryosphaeriaceae species associated with Botryosphaeria dieback of grapevine in Sicily (Italy). Phytopathol. Mediterr. 2013, 52, 388–396. [Google Scholar]
- Peluso, R.; Raio, A.; Morra, F.; Zoina, A. Physiological, biochemical and molecular analyses of an Italian collection of Agrobacterium tumefaciens strains. Eur. J. Plant Pathol. 2003, 109, 291–300. [Google Scholar] [CrossRef]
- EFSA Panel on Plant Health (PLH). Scientific Opinion on the pest categorisation of Xylophilus ampelinus (Panagopoulos) Willems et al. EFSA J. 2014, 12, 3921. [Google Scholar] [CrossRef]
- Martelli, G.P. Where grapevine virology is heading to. In Proceedings of the 19th Congress of the International Council for the Study of Virus and Virus-like Diseases of the Grapevine (ICVG), Santiago, Chile, 9–12 April 2018. [Google Scholar]
- Fuchs, M. Grapevine viruses: A multitude of diverse species with simple but overall poorly adopted management solutions in the vineyard. J. Plant Pathol. 2020, 102, 643–653. [Google Scholar] [CrossRef]
- Davino, S.; Calari, A.; Davino, M.; Tessitori, M.; Bertaccini, A.; Bellardi, M.G. Virescence of tenweeks stock associated to phytoplasma infection in Sicily. Bull. Insectology 2007, 60, 279–280. [Google Scholar]
- Davino, S.; Panno, S.; Rangel, E.A.; Davino, M.; Bellardi, M.G.; Rubio, L. Population genetics of cucumber mosaic virus infecting medicinal, aromatic and ornamental plants from northern Italy. Arch. Virol. 2012, 157, 739–745. [Google Scholar] [CrossRef]
- Davino, S.; Panno, S.; Iacono, G.; Sabatino, L.; D’Anna, F.; Iapichino, G.; Olmos, A.; Scuderi, G.; Rubio, L.; Tomassoli, L.; et al. Genetic variation and evolutionary analysis of Pepino mosaic virus in Sicily: Insights into the dispersion and epidemiology. Plant Pathol. 2017, 66, 368–375. [Google Scholar] [CrossRef] [Green Version]
- Panno, S.; Caruso, A.G.; Troiano, E.; Luigi, M.; Manglli, A.; Vatrano, T.; Davino, S. Emergence of tomato leaf curl New Delhi virus in Italy: Estimation of incidence and genetic diversity. Plant Pathol. 2019, 68, 601–608. [Google Scholar] [CrossRef]
- Golino, D.; Fuchs, M.; Sim, S.; Farrar, K.; Martelli, G.P. Improvement of grapevine planting stocks through sanitary selection and pathogen elimination. In Grapevine Viruses: Molecular Biology, Diagnostics and Management; Meng, B., Martelli, G.P., Golino, D.A., Fuchs, M., Eds.; Springer: Cham, Switzerland, 2017; pp. 561–580. [Google Scholar]
- Golino, D.; Fuchs, M.; Al Rwahnih, M.; Farrar, K.; Schmidt, A.; Martelli, G.P. Regulatory aspects of grape viruses and virus diseases: Certification, quarantine and harmonization. In Grapevine Viruses: Molecular Biology, Diagnostics and Management; Meng, B., Martelli, G.P., Golino, D.A., Fuchs, M., Eds.; Springer: Cham, Switzerland, 2017; pp. 581–590. [Google Scholar]
- Crnogorac, A.; Panno, S.; Mandić, A.; Gašpar, M.; Caruso, A.G.; Noris, E.; Davino, S.; Matić, S. Survey of five major grapevine viruses infecting Blatina and Žilavka cultivars in Bosnia and Herzegovina. PLoS ONE 2021, 16, e0245959. [Google Scholar] [CrossRef] [PubMed]
- Sabella, E.; Pierro, R.; Luvisi, A.; Panattoni, A.; D’Onofrio, C.; Scalabrelli, G.; Nutricati, E.; Aprile, A.; De Bellis, L.; Materazzi, A. Phylogenetic analysis of viruses in Tuscan Vitis vinifera sylvestris (Gmeli) Hegi. PLoS ONE 2018, 13, e0200875. [Google Scholar] [CrossRef] [PubMed]
- Martelli, G.P.; Minafra, A.; Saldarelli, P. Vitivirus, a new genus of plant viruses. Arch. Virol. 1997, 142, 1929. [Google Scholar]
- Rosciglione, B.; Castellano, M.A.; Martelli, G.P.; Savino, V.; Cannizzaro, G. Mealybug transmission of grapevine virus A. Vitis 1983, 22, 331–347. [Google Scholar]
- Conti, M.; Milne, R.G.; Luisoni, E.; Boccardo, G. A closterovirus from a stem-pitting-diseased grapevine. Phytopathology 1980, 70, 394–399. [Google Scholar] [CrossRef]
- Galiakparov, N.; Tanne, E.; Sela, I.; Gafny, R. Functional analysis of the grapevine virus A genome. Virology 2003, 306, 42–50. [Google Scholar] [CrossRef] [Green Version]
- Turturo, C.; Dell’Orco, M.; Saldarelli, P.; Minafra, A.; Stavolone, L. RNA silencing is suppressed by Grapevine virus A infection. An overview of rugose wood-associated viruses: 2000–2003. In Proceedings of the 14th ICVG Conference, Locorotondo, Italy, 12–17 September 2003. [Google Scholar]
- Davino, S.; Ruiz-Ruiz, S.; Serra, P.; Forment, J.; Flores, R. Revisiting the cysteine-rich proteins encoded in the 3′-proximal open reading frame of the positive-sense single-stranded RNA of some monopartite filamentous plant viruses: Functional dissection of p15 from grapevine virus B. Arch. Virol. 2020, 165, 2229–2239. [Google Scholar] [CrossRef]
- Adams, M.J.; Antoniw, J.F.; Bar-Joseph, M.; Brunt, A.A.; Candresse, T.; Foster, G.D.; Martelli, G.P.; Milne, R.G.; Fauquet, C.M. Virology Division News: The new plant virus family Flexiviridae and assessment of molecular criteria for species demarcation. Arch. Virol. 2004, 149, 1045–1060. [Google Scholar] [CrossRef]
- Centre for Agriculture and Biosciences International—CABI. Available online: https://www.cabi.org/isc/datasheet/26189#todistribution (accessed on 8 January 2022).
- Kostadinovska, E.; Mitrev, S.; Bianco, P.A.; Casati, P.; Bulgari, D. First Report of Grapevine virus A and Grapevine fleck virus in the Former Yugoslav Republic of Macedonia. Plant Dis. 2014, 98, 1747. [Google Scholar] [CrossRef]
- Porotikova, E.V.; Dmitrenko, U.D.; Volodin, V.A.; Volkov, Y.A.; Gorislavets, S.M.; Stranishevskaya, E.P.; Risovannaya, V.I.; Kamionskaya, A.M.; Vinogradova, S.V. First report of Grapevine virus A in Russian grapevines. Plant Dis. 2016, 100, 2541. [Google Scholar] [CrossRef]
- Rasool, S.; Naz, S.; Rowhani, A.; Diaz-Lara, A.; Golino, D.A.; Farrar, K.D.; Al Rwahnih, M. Survey of grapevine pathogens in Pakistan. J. Plant Pathol. 2019, 101, 725–732. [Google Scholar] [CrossRef]
- Milne, R.G.; Conti, M.; Lesemann, D.E.; Stellmach, G.; Tanne, E.; Cohen, J. Clostero Virus-Like Particles of Two Types Associated with Diseased Grapevines. J. Phytopathol. 1984, 110, 360–368. [Google Scholar] [CrossRef]
- Conti, M.; Milne, R.G. Closterovirus associated with leafroll and stem pitting in grapevine. Phytopathol. Mediterr. 1985, 24, 110–113. [Google Scholar]
- Garau, R.; Prota, V.A.; Piredda, R.; Prota, U. Investigations on a stunting factor in Vitis vinifera L. transmissible by grafting to ‘Kober 5BB’. Phytopathol. Mediterr. 1994, 33, 113–118. [Google Scholar]
- Digiaro, M.; Bedzrob, M.P.; Bedrzob, M.P.; D’Onghia, A.M.; Boscia, D.; Savino, V. On the correlation between grapevine virus A and rugose wood. Phytopathol. Mediterr. 1994, 33, 187–193. [Google Scholar]
- Martelli, G.P.; Souchet, H.G.; Boscia, D.; Savino, V. Viruses of grapevine in Malta. EPPO Bull. 1992, 22, 607–612. [Google Scholar] [CrossRef]
- Garau, R.; Fiori, P.P.; Prota, V.A.; Tolu, G.; Fiori, M.; Prota, U. Effect of virus infection on own-rooted clones of different wine grapes cultivars from Sardinia. In Proceedings of the Extended Abstracts 12th Meeting ICVG, Lisbon, Portugal, 29 September–2 October 1997; pp. 171–172. [Google Scholar]
- Digiaro, M.; Boscia, D.; Simene, V.; Savino, V. Detrimental effects of filamentous viruses to table grape varieties newly introduced in southern Italy. In Proceedings of the XII Meeting of ICVG, Lisbon, Portugal, 29 September–2 October 1997; Volume 28, pp. 169–170. [Google Scholar]
- Engelbrecht, D.J.; Kasdorf, G.G.F. Transmission of grapevine leafroll disease and associated closteroviruses by the vine mealybug, Planococcus ficus. Phytophylactica 1990, 22, 341–346. [Google Scholar]
- Pedroso, E.I.; Sequeira, O.A.; Pinto, M.E.G.; Simoes, V. Assays of transmission of grapevine virus by pseudococcids. Ciência Tec. Vitivinic. 1991, 10, 39–46. [Google Scholar]
- Garau, R.; Prota, V.A.; Boscia, D.; Fiori, M.; Prota, U. Pseudococcus affinis Mask., new vector of Grapevine trichoviruses A and B. Vitis 1995, 34, 67–68. [Google Scholar]
- La Notte, P.; Buzkan, N.; Choueiri, E.; Minafra, A.; Martelli, G.P. Acquisition and transmission of grapevine virus A by the mealybug Pseudococcus longispinus. J. Plant Pathol. 1997, 79, 79–85. [Google Scholar]
- Zorloni, A.; Prati, S.; Bianco, P.A.; Belli, G. Transmission of Grapevine virus A and Grapevine leafroll-associated virus 3 by Heliococcus bohemicus. J. Plant Pathol. 2006, 88, 325–328. [Google Scholar]
- Fortusini, A.; Scattini, G.; Prati, S.; Cinquanta, S.; Belli, G. Transmission of Grapevine leafroll virus 1 (GLRaV-1) and Grapevine virus A (GVA) by scale insects. In Proceedings of the 12th Meeting of ICVG, Lisbon, Portugal, 29 September–2 October 1997; pp. 121–122. [Google Scholar]
- Gottwald, T.R.; Hughes, G. A new survey method for Citrus tristeza virus disease assessment. In Proceedings of the XIV International Organization of Citrus Virologists (IOCV), São Paulo, Brazil, 14–21 July 2000; pp. 77–87. [Google Scholar]
- Davino, S.; Panno, S.; Arrigo, M.; La Rocca, M.; Caruso, A.G.; Lo Bosco, G. Planthology: An application system for plant diseases management. Chem. Eng. Trans. 2017, 58, 619–624. [Google Scholar] [CrossRef]
- Clark, M.F.; Adams, A.N.; Graham, F.L.; Smiley, J.; Russell, W.C.; Nairn, R. Characteristics of the Microplate Method of Enzyme-Linked Immunosorbent Assay for the Detection of Plant Viruses. J. Gen. Virol. 1977, 34, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Alabi, O.J.; Rwahnih, M.A.; Mekuria, T.A.; Naidu, R.A. Genetic diversity of Grapevine virus A in Washington and California vineyards. Phytopathology 2014, 104, 548–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; Mc Gettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nei, M.; Kumar, S. Molecular Evolution and Phylogenetics; Oxford University Press: Oxford, UK, 2000; pp. 147–164. [Google Scholar]
- Efron, B.; Halloran, E.; Holmes, S. Bootstrap confidence levels for phylogenetic trees. Proc. Natl. Acad. Sci. USA 1996, 93, 7085–7090. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Martin, D.P.; Murrell, B.; Golden, M.; Khoosal, A.; Muhire, B. RDP4: Detection and analysis of recombination patterns in virus genomes. Virus Evol. 2015, 1, vev003. [Google Scholar] [CrossRef] [Green Version]
- Jukes, T.H.; Cantor, C.R. Evolution of Protein Molecules. In Mammalian Protein Metabolism; Munro, H., Ed.; Academic Press: New York, NY, USA, 1969; Volume III, Chapter 24; pp. 21–132. [Google Scholar]
- Pamilo, P.; Bianchi, N. Evolution of the Zfx and Zfy genes: Rates and interdependence between the genes. Mol. Biol. Evol. 1993, 10, 271–281. [Google Scholar] [CrossRef] [Green Version]
- Muhire, B.M.; Varsani, A.; Martin, D.P. SDT: A Virus Classification Tool Based on Pairwise Sequence Alignment and Identity Calculation. PLoS ONE 2014, 9, e108277. [Google Scholar] [CrossRef] [PubMed]
- Davino, S.; Willemsen, A.; Panno, S.; Davino, M.; Catara, A.; Elena, S.F.; Rubio, L. Emergence and Phylodynamics of Citrus tristeza virus in Sicily, Italy. PLoS ONE 2013, 8, e66700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panno, S.; Caruso, A.G.; Davino, S. The nucleotide sequence of a recombinant tomato yellow leaf curl virus strain frequently detected in Sicily isolated from tomato plants carrying the Ty-1 resistance gene. Arch. Virol. 2017, 163, 795–797. [Google Scholar] [CrossRef] [PubMed]
- Panno, S.; Caruso, A.; Blanco, G.; Davino, S. First report of Tomato brown rugose fruit virus infecting sweet pepper in Italy. New Dis. Rep. 2020, 41, 20. [Google Scholar] [CrossRef] [Green Version]
- Goszczynski, D.E.; Jooste, A.E.C. Identification of divergent variants of Grapevine virus A. Eur. J. Plant Pathol. 2003, 109, 397–403. [Google Scholar] [CrossRef]
- Panno, S.; Caruso, A.G.; Bertacca, S.; Pisciotta, A.; Lorenzo, R.D.; Marchione, S.; Matić, S.; Davino, S. Genetic Structure and Molecular Variability of Grapevine Fanleaf Virus in Sicily. Agriculture 2021, 11, 496. [Google Scholar] [CrossRef]
- Domingo, E.; Escarmís, C.; Menéndez-Arias, L.; Holland, J.J. Viral quasispecies and fitness variations. In Origin and Evolution of Viruses; Academic Press: Cambridge, MA, USA, 1999; pp. 141–161. [Google Scholar] [CrossRef]
- Ali, A.; Roossinck, M.J. Genetic bottlenecks. In Plant Virus Evolution; Roossinck, M.J., Ed.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 123–131. [Google Scholar]
- Li, H.Y.; Roossinck, M.J. Genetic bottlenecks reduce population variation in an experimental RNA virus population. J. Virol. 2004, 78, 10582–10587. [Google Scholar] [CrossRef] [Green Version]
- Panno, S.; Caruso, A.G.; Barone, S.; Lo Bosco, G.; Rangel, E.A.; Davino, S. Spread of Tomato Brown Rugose Fruit Virus in Sicily and Evaluation of the Spatiotemporal Dispersion in Experimental Conditions. Agronomy 2020, 10, 834. [Google Scholar] [CrossRef]
- Caruso, A.G.; Bertacca, S.; Ragona, A.; Matić, S.; Davino, S.; Panno, S. Epidemiological Survey of Grapevine Leafroll-Associated Virus 1 and 3 in Sicily (Italy): Genetic Structure and Molecular Variability. Agriculture 2022, 12, 647. [Google Scholar] [CrossRef]
- Guidoni, S.; Mannini, F.; Ferrandino, A.; Argamante, N.; Stefano, R.D. The effect of grapevine leafroll and rugose wood sanitation on agronomic performance and berry and leaf phenolic content of a Nebbiolo clone (Vitis vinifera L.). Am. J. Enol. Vitic. 1997, 48, 438–442. [Google Scholar]
- Bottalico, G.; Savino, V.; Campanale, A. Improvements in grapevine sanitation protocols. In Proceedings of the Extended Abstracts of the 13th Meeting of ICVG, Adelaide, Australia, 12–17 March 2000; p. 167. [Google Scholar]
- Goussard, P.G.; Wiid, J.; Kasdorf, G.G.F. The effectiveness of in vitro somatic embryogenesis in eliminating fanleaf virus and leafroll associated viruses from grapevines. S. Afr. J. Enol. Vitic. 1991, 12, 77–81. [Google Scholar] [CrossRef] [Green Version]
- Malenica, N.; Jagìc, M.; Pavletìc, B.; Bauer, N.; Vončina, D.; Zdunìc, G.; Leljak Levanìc, D. Somatic embryogenesis as a tool for virus elimination in croatian indigenous grapevine cultivars. Acta Bot. Croat. 2020, 79, 26–34. [Google Scholar] [CrossRef]
- Wang, Q.; Gafny, R.; Li, P.; Mawassi, M.; Sela, I.; Tanne, E. Elimination of Grapevine virus A by cryopreservation. In Proceedings of the Extended Abstracts of the 14th Meeting of ICVG, Locorotondo, Italy, 12–17 September 2003; p. 242. [Google Scholar]
- Radian-Sade, S.; Perl, A.; Edelbaum, O.; Kuznetsova, L.; Gafny, R.; Sela, I.; Tanne, E. Transgenic Nicotiana benthamiana and grapevine plants transformed with grapevine virus A (GVA) sequences. Phytoparasitica 2000, 28, 79–86. [Google Scholar] [CrossRef]
- Buzkan, N.; Minafra, A.; Saldarelli, P.; Castellano, A.; Dell’Orco, M.; Martelli, G.P.; Gölles, R.; Laimer da Camara Machado, M. Heterologous encapsidation in non-transgenic and transgenic Nicotiana plants infected by Grapevine viruses A and B. J. Plant Pathol. 2001, 83, 37–43. [Google Scholar]
- Martinelli, L.; Candioli, E.; Costa, D.; Minafra, A. Stable insertion and expression of the movement protein gene of Grapevine Virus A (GVA) in grape (Vitis rupestris S.). Vitis 2002, 41, 189–193. [Google Scholar]
- Muruganantham, M.; Moskovitz, Y.; Haviv, S.; Horesh, T.; Fenigstein, A.; du Preez, J.; Stephan, D.; Burger, J.T.; Mawassi, M. Grapevine virus A-mediated gene silencing in Nicotiana benthamiana and Vitis vinifera. J. Virol. Methods 2009, 155, 167–174. [Google Scholar] [CrossRef]
- Blaisdell, G.K.; Zhang, S.; Rowhani, A.; Klaassen, V.; Cooper, M.L.; Daane, K.M.; Almeida, R.P.P. Trends in vector-borne transmission efficiency from coinfected hosts: Grapevine leafroll-associated virus-3 and Grapevine virus A. Eur. J. Plant Pathol. 2020, 156, 1163–1167. [Google Scholar] [CrossRef]
- Ferriol, I.; Rubio, L.; Pérez-Panadés, J.; Carbonell, E.A.; Davino, S.; Belliure, B. Transmissibility of Broad bean wilt virus 1 by aphids: Influence of virus accumulation in plants, virus genotype and aphid species. Ann. Appl. Biol. 2013, 162, 71–79. [Google Scholar] [CrossRef]
- Pacifico, D.; Caciagli, P.; Palmano, S.; Mannini, F.; Marzachì, C. Quantitation of Grapevine leafroll associated virus-1 and -3, Grapevine virus A, Grapevine fanleaf virus and Grapevine fleck virus in field-collected Vitis vinifera L. ‘Nebbiolo’ by real-time reverse transcription-PCR. J. Virol. Methods 2011, 172, 1–7. [Google Scholar] [CrossRef]
- Osman, F.; Hodzic, E.; Omanska-Klusek, A.; Olineka, T.; Rowhani, A. Development and validation of a multiplex quantitative PCR assay for the rapid detection of Grapevine virus A, B and D. J. Virol. Methods 2013, 194, 138–145. [Google Scholar] [CrossRef]
- Panno, S.; Ferriol, I.; Rangel, E.A.; Olmos, A.; Han, C.; Martinelli, F.; Rubio, L.; Davino, S. Detection and identification of Fabavirus species by one-step RT-PCR and multiplex RT-PCR. J. Virol. Methods 2014, 197, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Panno, S.; Ruiz-Ruiz, S.; Caruso, A.G.; Alfaro-Fernandez, A.; San Ambrosio, M.I.F.; Davino, S. Real-time reverse transcription polymerase chain reaction development for rapid detection of Tomato brown rugose fruit virus and comparison with other techniques. PeerJ 2019, 7, e7928. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Xin, Y.; Li, G.; Qian, Y. Development of a RT-LAMP Assay for Detection of Grapevine virus A. Sci. Agric. Sin. 2016, 49, 103–109. [Google Scholar]
- Panno, S.; Matìc, S.; Tiberini, A.; Caruso, A.G.; Bella, P.; Torta, L.; Stassi, R.; Davino, S. Loop Mediated Isothermal Amplification: Principles and Applications in Plant Virology. Plants 2020, 9, 461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).