Revisiting Sulphur—The Once Neglected Nutrient: It’s Roles in Plant Growth, Metabolism, Stress Tolerance and Crop Production
Abstract
1. Introduction
2. Biological Functions of Sulphur (S) in Plants
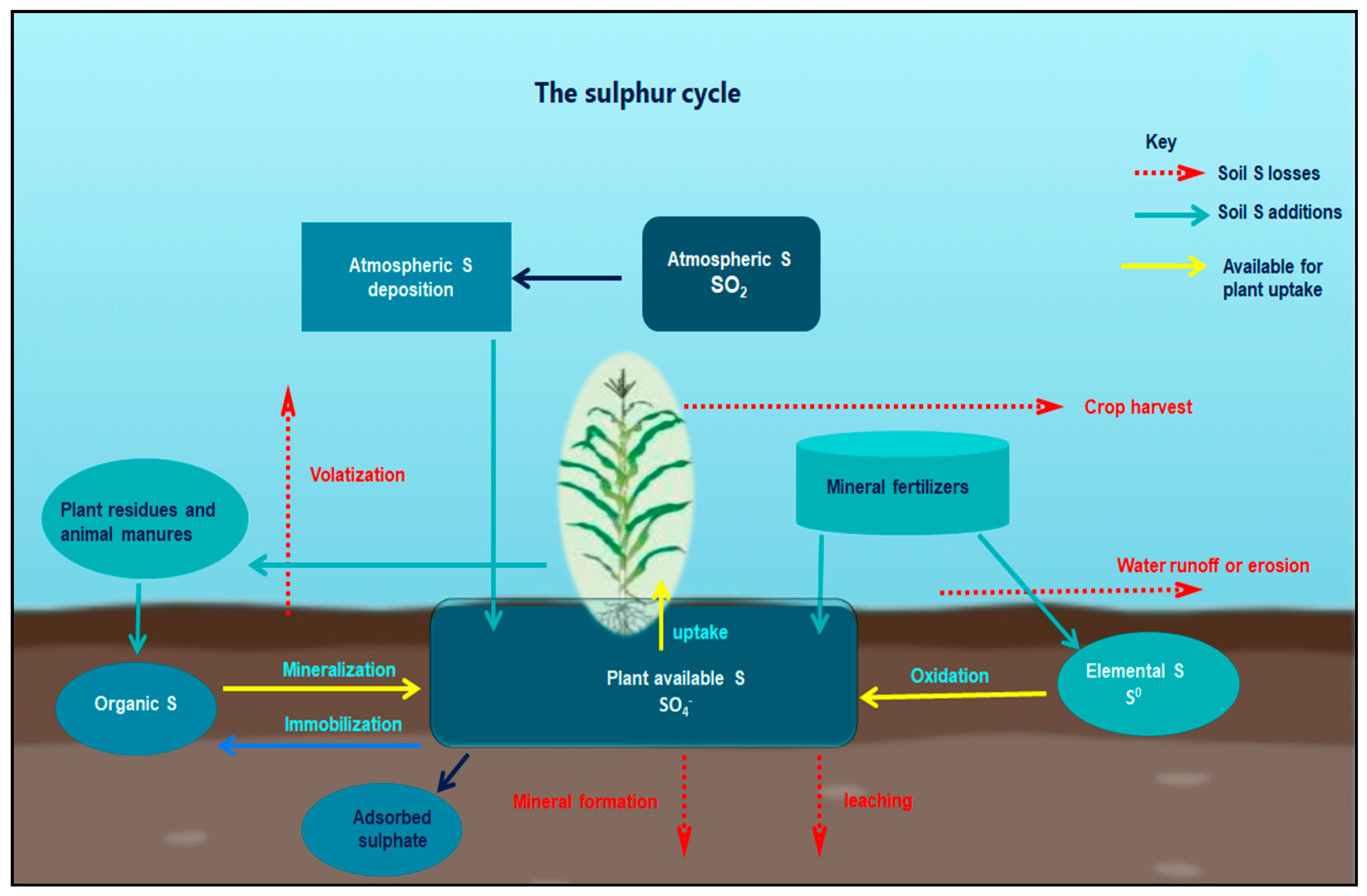
3. Sulphur in the Soil
4. Sulphur in the Plant System
4.1. Uptake, Assimilation and Metabolism of S by Plants
4.2. Physiological and Metabolic Alterations in Plants under S Deprivation
5. Interaction of S with Other Nutrients
5.1. S and Nitrogen (N) Interaction
5.2. Interaction of S with Non-N Nutrients
6. Sulphur Role in Stress Tolerance
7. Sulphur and Crop Production
7.1. Sulphur Requirements of Crops
7.2. Yield and Quality Effects of S in Different Cultivated Crop Species
7.3. Sulphur Deficiency Symptoms and Diagnosis Approaches
7.4. Crop S Sources and Supply (Fertilizer) Recommendations
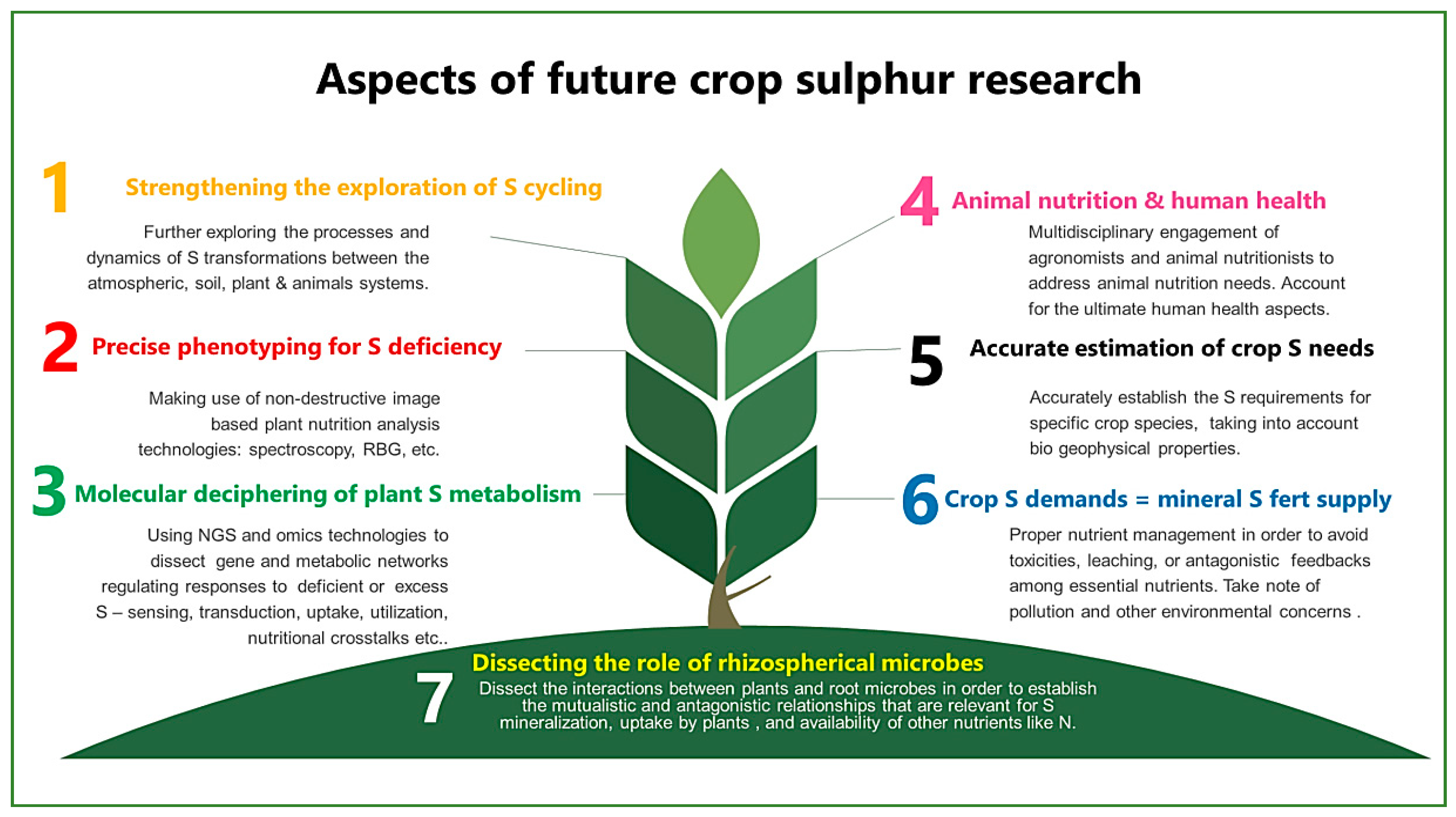
8. A View into the Future
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Food and Agricultural Organization of the United Nations (FAO); UNESCO. Global Agriculture towards 2050. High Level Expert Forum—How to Feed the World in 2050; FAO: Rome, Italy, 12–13 October 2009. [Google Scholar]
- Elferink, M.; Schierhorn, F. Global Demand for Food is Rising. Can we meet it. Harv. Bus. Rev. 2016, 7, 2–5. [Google Scholar]
- Marschner, P. Mineral Nutrition of Higher Plants, 3rd ed.; Elsevier: New York, NY, USA, 2012. [Google Scholar]
- Sahota, T.S. Importance of Sulphur in Crop Production. Ont. Farmer Northwest Link 2012, 46, 11–12. [Google Scholar] [CrossRef]
- Bouranis, D.L.; Chorianopoulou, S.N.; Siyiannis, V.F.; Protonotarios, V.E.; Koufos, C.; Maniou, P. Changes in nutrient allocation between roots and shoots of young maize plants during sulphate deprivation. J. Plant Nutr. Soil Sci. 2012, 175, 499–510. [Google Scholar] [CrossRef]
- Sienkiewicz-Cholewa, U.; Kieloch, R. Effect of sulphur and micronutrients fertilization yield and fat content in winter rape seeds (Brassica napus L.). Plant Soil Environ. 2015, 61, 164–170. [Google Scholar] [CrossRef]
- Kopriva, S.; Malagoli, M.; Takahashi, H. Sulfur nutrition: Impacts on plant development, metabolism, and stress responses. J. Exp. Bot. 2019, 70, 4069–4073. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Dong, S.; Hu, C.; Wang, K. The role of nitrogen and sulphur interaction in maize quality (Zea mays L.). Agric. Sci. China 2003, 2, 527–532. [Google Scholar]
- Tiwari, K.N.; Gupta, B.R. Sulphur for Sustainable High Yield Agriculture in Uttah Pradesh. Indian J. Fertil. 2006, 1, 37–52. [Google Scholar]
- Jarvan, M.; Edesi, L.; Adamson, A.; Lukme, L.; Akk, A. The effect of sulphur fertilization yield, quality of protein and baking properties of winter wheat. Agron. Res. 2008, 6, 459–469. [Google Scholar]
- Farhad, I.S.M.; Islam, M.N.; Hoque, S.; Bhuiyan, M.S.I. Role of Potassium and Sulphur on the Growth, Yield and Oil Content of Soybean (Glycine max L.). Acad. J. Plant Sci. 2010, 3, 99–103. [Google Scholar]
- Rasool, F.U.; Hassan, B.; Jahangir, I.A. Growth and yield of sunflower (Helianthus annus L.) as influenced by nitrogen, sulphur and farmyard manure under temperate conditions. SAARC J. Agric. 2013, 11, 81–89. [Google Scholar] [CrossRef]
- Ali, A.; Iqbal, Z.; Hassan, S.W.; Yasin, M.; Khaliq, T.; Ahmed, S. Effect of nitrogen and sulphur on phenology, growth and yield parameters of maize crop. Sci. Int. 2013, 25, 363–366. [Google Scholar]
- Nasreen, S.; Imamul Haq, S.M.; Hossain, M.A. Sulphur Effects on Growth, Responses and Yield of Onion. Asian J. Plant Sci 2003, 2, 897–902. [Google Scholar] [CrossRef]
- Ullah, M.H.; Huq, S.M.I.; Alam, M.D.U.; Rahman, M.A. Impacts of Sulphur Levels on Yield, Storability and Economic Return of Onion. Bangladesh J. Agric. Res. 2018, 33, 539–548. [Google Scholar] [CrossRef]
- Kopriva, S.; Calderwood, A.; Weckopp, S.C.; Koprivova, A. Plant sulphur and Big Data. Plant Sci. 2015, 241, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jamal, A.; Moon, Y.; Abdin, M.Z. Sulphur—A general overview and interaction with nitrogen. Aust. J. Crop Sci. 2010, 4, 523–529. [Google Scholar] [CrossRef]
- Nazar, R.; Iqbal, N.; Masood, A.; Syeed, S.; Khan, N.A. Understanding the significance of sulphur in improving salinity tolerance in plants. Environ. Exp. Bot. 2011, 70, 80–87. [Google Scholar] [CrossRef]
- Santisree, P.; Adimulam, S.S.; Bommineni, P.; Bhatnagar-Mathur, P.; Sharma, K.K. Hydrogen Sulfide in Plant Abiotic Stress Tolerance: Progress and Perspectives. In Reactive Oxygen, Nitrogen and Sulfur Species in Plants: Production, Metabolism, Signaling and Defense Mechanisms; John Wiley & Sons: New York, NY, USA, 2019; pp. 743–775. [Google Scholar]
- Anjum, N.A.; Gill, S.S.; Umar, S.; Ahmad, I.; Duarte, A.C.; Pereira, E. Improving Growth and Productivity of Oleiferous Brassicas Under Changing Environment: Significance of Nitrogen and Sulphur Nutrition, and Underlying Mechanisms. Sci. World J. Vol. 2012. Available online: http:www.hindawi.com/journals/tswj (accessed on 3 March 2020). [CrossRef] [PubMed]
- Giordano, M.; Raven, J.A. Nitrogen and Sulfur assimilation in plants and algae. Aquat. Bot. 2014, 118, 45–61. [Google Scholar] [CrossRef]
- HARSCO. Sustainable Management of Greens and Tees Under Abiotic Stress. In Cross Over—From Soil to Plant, Product Information Bulletin; HARSCO: Tampa, FL, USA, 2016; Available online: www.numeratortech.com (accessed on 10 October 2020).
- Edis, R.; Norton, R. Sulphur Nutrition and Fluid Fertilisers. 2012 Victorian Liquid Fertiliser Forum. 2012; p. 4. Available online: http://www.ipni.net/ (accessed on 12 February 2020).
- Bloem, E. The importance of sulphur in profitable crop production. In Proceedings of the Omnia Farmer’s Information Day, Hilton, KwaZulu-Natal, South Africa, 14 August 2015; Phillips, L., Ed.; Farmer’s Weekly. 2015. Available online: http://farmersweekly.co.za/article.aspx?id=782151 (accessed on 28 November 2019).
- Matula, J. Barley response to the soil reserve of sulphur and ammonium sulphate in short-term experiments under controlled conditions of cultivation. Plant Soil Environ. 2004, 50, 235–242. [Google Scholar] [CrossRef]
- Messick, D.L. World Sulphur Outlook; The World Sulphur Institute: Washington, DC, USA, 2013; Available online: http://www.firt.org (accessed on 18 April 2020).
- Chemical Economics Handbook. Sulfur. 2017. Available online: https://ihsmarkit.com/products/sulfur-chemical-economics-handbook.html (accessed on 27 July 2020).
- Stratistics Market Research Consulting (SMRC). Sulfur Fertilizers—Global Market Outlook (2017–2026) Report; SMRC: Gaithersburg, MD, USA, 2018; p. 173. [Google Scholar]
- MarketsandMarkets. Sulfur Fertilizers Market by Type (Sulfates, Elemental Sulfur, Liquid Sulfur Fertilizers), Crop Type (Oilseeds & Pulses, Cereals & Grains, Fruits & Vegetables, Others), Mode of Application, Form, Cultivation Type, and Region—Global Forecast to 2025; MarketsandMarkets: New York, NY, USA, 2020; p. 208. [Google Scholar]
- Allied Analytics LLP. Sulfur Fertilizers Market by Type, and Type of Agriculture: Global Opportunity Analysis and Industry Forecast, 2019–2026; Allied Analytics LLP: Pune, India, 2020; p. 269. [Google Scholar]
- Nickel, R. Price Spike in Sulfur Leaves Farmers, Fertilizer Makers Sour. Commodities. 20 December 2017. Reuters. Available online: https://www.reuters.com/article/us-fertilizers-sulphur-idUSKBN1EE07Q (accessed on 28 July 2020).
- Mazid, M.; Khan, Z.H.; Quddusi, S.; Khan, T.A.; Mohammad, F. Significance of sulphur nutrition against metal induced oxidative stress in plants. J. Stress Physiol. Biochem. 2011, 7, 165–184. [Google Scholar] [CrossRef]
- Mazid, M.; Khan, T.A.; Mohammad, F. Role of secondary metabolites in defense mechanisms of plants. Biol. Med. 2011, 3, 232–249. [Google Scholar]
- Till, A.R. Sulphur and Sustainable Agriculture, 1st ed.; International Fertilizer Industry Association: Paris, France, 2010; pp. 1–70. [Google Scholar]
- Wiesler, F.; Gerendias, J.; Sattelmacher, B. Influence of mineral fertilizers on nutritional quality of staple food crops. Impacts Agric. Hum. Health Nutr. 2010, 1, 1–36. [Google Scholar]
- Zhao, F.; Tausz, M.; De Kok, L.J. Role of Sulfur for Plant Production in Agricultural and Natural Ecosystems. In Sulfur Metabolism in Phototrophic Organisms; Hell, R., Dahl, C., Knaff, D., Leustek, T., Eds.; Advances in Photosynthesis and Respiration; Springer: Dordrecht, The Netherlands, 2008; Volume 27. [Google Scholar] [CrossRef]
- Gill, S.S.; Khan, N.A.; Anjum, N.A.; Tuteja, N. Amelioration of cadmium stress in crop plants by nutrients management: Morphological, physiological and biochemical aspects. Plant Stress 2011, 5, 1–23. [Google Scholar]
- Hawkesford, M.J.; Dekok, L.J. Managing sulphur metabolism in plants. Plant Cell Environ. 2006, 29, 382–395. [Google Scholar] [CrossRef] [PubMed]
- Koprivova, A.; Kopriva, S. Molecular mechanisms of regulation of sulfate assimilation: First steps on a long road. Front. Plant Sci. 2014, 5, 589. [Google Scholar] [CrossRef]
- Njira, K.O.W.; Nabwami, J. A review of effects of nutrients elements on crop quality. Afr. J. Food Agric. Nutr. Dev. 2015, 15, 9777–9793. [Google Scholar]
- Hell, R.; Khan, M.S.; Wirtz, M. Cellular biology of sulfur and its functions in plants. In Cell Biology of Metals and Nutrients; Hell, R., Mendel, R.-R., Eds.; Plant Cell Monographs; Springer: Berlin/Heidelberg, Germany, 2010; Volume 17, pp. 243–279. [Google Scholar] [CrossRef]
- Clarkson, D.T.; Hanson, J.B. The mineral nutrition of higher plants. Annu. Rev. Plant Physiol. 1980, 31, 239–298. [Google Scholar] [CrossRef]
- Aulakh, M.S. Crop responses to sulphur nutrition. In Sulphur in Plants; Abrol, Y.P., Ahmad, A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2003; pp. 341–358. [Google Scholar]
- Hassan, F.; Manaf, A.; Qadir, G.; Basra, S.M.A. Effects of Sulphur on Seed Yield, Oil, Protein and Glucosinolates of Canola Cultivars. Int. J. Agric. Biol. 2007, 9, 504–508. [Google Scholar]
- Scherer, H.W. Sulphur in crop production—Invited paper. Eur. J. Agron. 2001, 14, 81–111. [Google Scholar] [CrossRef]
- Kowalska, I. Effects of sulphate level in the nutrient solution plant growth and sulphur content in tomato plants. Folia Hortic. Ann. 2005, 17, 91–100. [Google Scholar] [CrossRef]
- El Sayed, H.E.; Hameda, E.A.; El-Morsy, H.A.E.; Hanan, S.A.O. The Role of Sulphur and Certain Foliar Spray Levels of Micro-nutrients on Garlic (Allium sativum L.) Plant. Int. J. Curr. Res. Biosci. Plant Biol. 2015, 2, 76–87. [Google Scholar]
- Smatanova, M.; Richter, R.; Hlušek, J. Spinach and pepper response to nitrogen and sulphur fertilization. Plant Soil Environ. 2004, 7, 303–308. [Google Scholar] [CrossRef]
- Singh, Y.; Singh, T.; Singh, U.N.; Rajput, T.K. Effect of nutrient management on yield, quality an economics of irrigated Indian mustard (Brassica juncea). Indian J. Agric. Sci. 2010, 80, 691–694. [Google Scholar] [CrossRef]
- Fuentes-Lara, L.O.; Medrano-Macías, J.; Pérez-Labrada, F.; Rivas-Martínez, E.N.; García-Enciso, E.L.; González-Morales, S.; Juárez-Maldonado, A.; Rincón-Sánchez, F.; Benavides-Mendoza, A. From Elemental Sulfur to Hydrogen Sulfide in Agricultural Soils and Plants. Molecules 2019, 24, 2282. [Google Scholar] [CrossRef]
- Eriksen, J. Chapter 2 Soil sulfur cycling in temperate agricultural systems. Adv. Agron. 2009, 102, 55–89. [Google Scholar] [CrossRef]
- Nikiforova, V.J.; Gakiere1, B.; Kempa, S.; Adamik, M.; Willmitzer, L.; Hesse, H.; Hoefgen, R. Towards dissecting nutrient metabolism in plants: A systems biology case study on sulphur metabolism. J. Exp. Bot. 2004, 55, 1861–1870. [Google Scholar] [CrossRef]
- Scherer, H.W. Sulfur in Soils. J. Plant Nutr. Soil Sci. 2009, 172, 326–335. [Google Scholar] [CrossRef]
- Landers, D.H.; David, M.B.; Mitchell, M.J. Analysis of Organic and Inorganic Sulfur Constituents in Sediments, Soils and Water. Int. J. Environ. Anal. Chem. 1983, 14, 245–256. [Google Scholar] [CrossRef]
- Peplow, M. Sulphur from soil. Nature 2004, 428, 136. Available online: www.nature.com/nature (accessed on 3 May 2019).
- Germida, J.J.; Siciliano, S.D. Sulfur Cycle in Soils. In Encyclopedia of Environmental Microbiology; Bitton, G., Ed.; John Wiley & Sons: New York, NY, USA, 2003. [Google Scholar] [CrossRef]
- Gunjal, A.B.; Waghmode, M.S.; Patil, N.N.; Nawani, N.N. Significance of soil enzymes in agriculture. In Smart Bioremediation Technologies; Academic Press: New York, NY, USA, 2019; pp. 159–168. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.A. Arylsulfatase activity of soils. Soil Sci. Soc. Am. Proc. 1970, 34, 225–229. [Google Scholar] [CrossRef]
- Brüser, T.; Lens, P.; Trüper, H.G. The biological sulfur cycle. In Environmental Technologies to Treat Sulfur Pollution; Lens, P., Pol, L.H., Eds.; IWA Publishing: London, UK, 2000; pp. 47–86. [Google Scholar]
- Westley, J.; Adler, H.; Westley, L.; Nishida, C. The sulfurtransferases. Fundam. Appl. Toxicol. 1984, 3, 377–382. [Google Scholar] [CrossRef]
- Buchner, P.; Stuiver, C.E.; Westerman, S.; Wirtz, M.; Hell, R.; Hawkesford, M.J.; De Kok, L.J. Regulation of sulfate uptake and expression of sulfate transporter genes in Brassica oleracea as affected by atmospheric H(2)S and pedospheric sulfate nutrition. Plant Physiol. 2004, 136, 3396–3408. [Google Scholar] [CrossRef] [PubMed]
- Droux, M. Sulfur assimilation and the role of sulfur in plant metabolism: A survey. Photosynth. Res. 2004, 79, 331–348. [Google Scholar] [CrossRef]
- Davidian, J.C.; Kopriva, S. Regulation of sulfate uptake and assimilation—The same or not the same? Mol. Plant 2010, 3, 314–325. [Google Scholar] [CrossRef]
- Kopriva, S.; Koprivova, A. Sulphate assimilation and glutathione synthesis in C4 plants. Photosynth. Res. 2005, 86, 363–372. [Google Scholar] [CrossRef]
- Khan, N.A.; Khan, M.I.R.; Asgher, M.; Fatma, M.; Masood, A.; Syeed, S. Salinity Tolerance in Plants: Revisiting the Role of Sulfur Metabolites. J. Plant Biochem. Physiol. 2014, 2. [Google Scholar] [CrossRef]
- Takahashi, H.; Yamazaki, M.; Sasakura, N.; Watanabe, A.; Leustek, T.; Engler, J.A.; Engler, G.; Van Montagu, M.; Saito, K. Regulation of sulfur assimilation in higher plants: A sulfate transporter induced in sulphate starved roots plays a central role in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 1997, 94, 11102–11107. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H. The roles of three functional sulphate transporters involved in uptake and translocation of sulphate in Arabidopsis thaliana. Plant J. 2000, 23, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Kopriva, S.; Giordano, M.; Saito, K.; Hell, R. Sulfur assimilation in photosynthetic organisms: Molecular functions and regulations of transporters and assimilatory enzymes. Annu. Rev. Plant Biol. 2011, 62, 157–184. [Google Scholar] [CrossRef]
- Saito, K. Sulfur Assimilatory Metabolism. The Long and Smelling Road. Update on Nutrient Metabolism. Plant Physiol. 2004, 136, 2443–2450. [Google Scholar] [CrossRef]
- Maruyama-Nakashita, A.; Inoue, E.; Watanabe-Takahashi, A.; Yamaya, T.; Takahashi, H. Transcriptome Profiling of Sulfur-Responsive Genes in Arabidopsis Reveals Global Effects of Sulfur Nutrition Multiple Metabolic Pathways. Plant Phys. 2003, 132, 597–605. [Google Scholar] [CrossRef]
- Prioretti, L.; Gontero, B.; Hell, R.; Giordano, M. Diversity and regulation of ATP sulfurylase in photosynthetic organisms. Front. Plant Sci. 2014, 5597, 1–12. [Google Scholar] [CrossRef]
- Kopriva, S.; Rennenberg, H. Control of sulphate assimilation and glutathione synthesis: Interaction with N and C metabolism. J. Exp. Bot. 2004, 55, 1831–1842. [Google Scholar] [CrossRef] [PubMed]
- Aubry, S.; Smith-Unna, R.D.; Boursnell, C.M.; Kopriva, S.; Hibberd, J.M. Transcript residency on ribosomes reveals a key role for the Arabidopsis thaliana bundle sheath in sulfur and glucosinolate metabolism. Plant J. 2014, 78, 659–673. [Google Scholar] [CrossRef] [PubMed]
- Lappartient, A.G.; Touraine, B. Demand-driven control of root ATP sulphurylase activity and SO2 4 uptake in intact canola. The role of phloem-translocated glutathione. Plant Physiol. 1996, 111, 147–157. [Google Scholar] [CrossRef]
- Leustek, T.; Martin, M.N.; Bick, J.A.; Davies, J.P. Pathways and regulation of sulfur metabolism revealed through molecular and genetic studies. Annu. Rev. Plant Phys. 2000, 51, 141–165. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Srivastava, R.; Verma, K.K. Biological sulfur acquisition: An update. Elixir Appl. Bot. 2013, 62, 17740–17747. [Google Scholar]
- Honsel, A.; Kojima, M.; Haas, R.; Frank, W.; Sakakibara, H.; Herschbach, C.; Rennenberg, H. Sulphur limitation and early sulphur deficiency responses in poplar: Significance of gene expression, metabolites, and plant hormones. J. Exp. Bot. 2012, 63, 1873–1893. [Google Scholar] [CrossRef]
- Kawashima, C.G.; Yoshimoto, N.; Maruyana-Nakashita, A.; Tsuchiya, Y.N.; Saito, K.; Takahashi, H.; Dalmayl, T. Sulphur starvation induces the expression of microRNA-395 and one of its target genes but in different cell types. Plant J. 2009, 57, 313–321. [Google Scholar] [CrossRef]
- Kaya, M.; Küçükyumuk, Z.; Erdal, I. Effects of elemental sulfur and sulfur-containing waste on nutrient concentrations and growth of bean and corn plants grown on calcareous soil. AJB 2009, 8, 4481–4489. [Google Scholar] [CrossRef][Green Version]
- Janzen, H.H.; Bettany, J.R. Sulfur nutrition of rapeseed: I. Influence of fertilizer nitrogen and sulfur rates. Soil Sci. Soc. Am. J. 1984, 48, 100–107. [Google Scholar] [CrossRef]
- Wardman, J. Don’t Forget about Sulphur. Yara. UK. Available online: https://www.yara.co.uk/crop-nutrition/agronomy-advice/dont-forget-about-sulphur/ (accessed on 25 August 2020).
- Dev, G.; Kumar, V. Secondary nutrients. In Review of Soil Research in India, Part I, 12th International Congress on Soil Science, (SS‘ 82); Indian Society of Soil Science: New Delhi, India, 1982; pp. 342–360. [Google Scholar]
- Potash Development Association. Sulphur as a Nutrient for Crops and Grass. 2017. Available online: https://www.pda.org.uk/sulphur-nutrient-crops-grass/ (accessed on 25 August 2020).
- Fismel, S.; Vong, P.C.; Guckert, A.; Frossard, E. ‘Influence of sulfur on apparent N-uses efficiency, yield and quality of oilseed rape (Brassica napus L.) grown on a calcareous soil. Eur. J. Agron. 2000, 12, 127–141. [Google Scholar] [CrossRef]
- McGrath, S.P.; Zhao, F.J. Sulphur uptake, yield responses and the interactions between nitrogen and sulphur in winter oilseed rape (Brassica napus). J. Agric. Sci. 1996, 126, 53–62. [Google Scholar] [CrossRef]
- Ahmad, A.; Abraham, G.; Gandotra, N.; Abrol, Y.P. Interactive effect of nitrogen and sulphur on growth and yield of rapeseed-mustard (Brassica juncea L. Czern and Coss. and Brassica compestris L.) genotypes. J. Agron. Crop Sci. 1998, 181, 193–199. [Google Scholar] [CrossRef]
- Jamal, A.; Fazil, I.S.; Ahmad, S.; Abdin, M.Z.; Song, J.Y. Effect of Sulphur and Nitrogen Application Growth Characteristics, Seed and Oil Yields of Soybean Cultivars. Korean J. Crop Sci. 2005, 50, 340–345. [Google Scholar]
- Jamal, A.; Fazil, I.S.; Ahmad, S.; Abdin, M.Z. Interactive Effect of Nitrogen and Sulphur on Yield and Quality of Groundnut (Arachis hypogea L.). Korean J. Crop Sci. 2006, 51, 519–522. [Google Scholar]
- Clarkson, D.T.; Saker, L.R.; Purves, J.V. Depression of nitrate and ammonium influx in barley plants with diminished sulphate-status. Evidence for co-regulation in nitrogen and sulphate intake. J. Exp. Bot. 1989, 40, 953–963. [Google Scholar] [CrossRef]
- Fazili, I.S.; Masoodi, M.; Ahmad, S.; Jamal, A.; Khan, J.S.; Abdin, M.Z. Interactive Effects of Sulfur and Nitrogen on Growth and Yield Attributes of Oilseed Crops (Brassica campestris L. and Eruca sativa Mill.) Differing in Yield Potential. J. Plant. Nutr. Soil Sci. 2010, 33, 1216–1228. [Google Scholar] [CrossRef]
- Salvagiotti, F.; Miralles, D.J. Radiation interception, biomass production and grain yield as affected by the interaction of nitrogen and sulfur fertilization in wheat. Eur. J. Agron. 2008, 28, 282–290. [Google Scholar] [CrossRef]
- Hocking, P.J.; Randal, P.J.; Pinkerton, A. Sulfur nutrition of sunflower (Helianthus annus) as affected by nitrogen supply. Effects on vegetative growth, development of yield components, and seed yield and quality. Field Crops Res. 1987, 16, 157–175. [Google Scholar] [CrossRef]
- Patra, P.; Pati, B.K.; Ghosh, G.K.; Mura, S.S.; Saha, A. Effect of Biofertilizers and Sulphur on Growth, Yield, and Oil Content of Hybrid Sunflower (Helianthus annuus L.) In a Typical Lateritic Soil. J. Bacteriol. Parasitol. 2013, 2, 603. [Google Scholar] [CrossRef]
- Raza, M.A.; Feng, L.Y.; Manaf, A.; Wasaya, A.; Ansar, M.; Hussain, A.; Khalid, M.H.; Iqbal, N.; Xi, Z.J.; Chen, Y.K.; et al. Sulphur application increases seed yield and oil content in sesame seeds under rainfed conditions. Field Crops Res. 2018, 218, 51–58. [Google Scholar] [CrossRef]
- Abdin, M.Z.; Ahmad, A.; Khan, N.; Khan, I.; Jamal, A.; Iqbal, M. Sulphur Interaction with Other Nutrients. In Sulphur Plants; Abrol, Y.P., Ahmad, A., Eds.; Springer: Dordrecht, The Netherlands, 2003; pp. 359–374. [Google Scholar] [CrossRef]
- Alam, A.K.M.M.; Goni, M.N.; Rahman, M.M.; Islam, M.N.; Hussain, M.S. Individual and interaction effect of macro and micronutrients on jute seed production. Int. J. Sustain. Crop. Prod. 2009, 4, 22–26. [Google Scholar]
- Yadav, B.K. Interaction Effect of Phosphorus and Sulphur on Yield and Quality of Clusterbean in Typic Haplustept. World J. Agric. Sci. 2011, 7, 556–560. [Google Scholar]
- Dash, A.K.; Singh, H.K.; Mahakud, T.; Pradhan, K.C.; Jena, D. Interaction Effect of Nitrogen, Phosphorus, Potassium with Sulphur, Boron and Zinc on Yield and Nutrient Uptake by Rice Under Rice—Rice Cropping System in Inceptisol of Coastal Odisha. Int. Res. J. Agric. Sci. Soil Sci. 2015, 5, 14–21. [Google Scholar]
- Anonymous. Phosphorus improves crop quality. Better Crops 1999, 83, 2829. [Google Scholar]
- Islam, M.; Ali, S.; Hayat, R. Effect of Integrated Application of Phosphorus and Sulphur on Yield and Micronutrient Uptake by Chickpea (Cicer arietinum). Int. J. Agric. Biol. 2009, 11, 33–38. [Google Scholar]
- Saha, B.; Saha, S.; Saha, R.; Hazra, G.C.; Mandal, B. Influence of Zn, B and S on the yield and quality of groundnut (Arachis Hypogea L.). Legume Res. 2015, 38, 832–836. [Google Scholar]
- Anjum, N.A.; Gill, R.; Kaushik, M.; Hasanuzzaman, M.; Pereira, E.; Ahmad, I.; Tuteja, N.; Gill, S.S. ATP-sulfurylase, sulfur-compounds, and plant stress tolerance. Front. Plant Sci. 2015, 6, 210. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef]
- Anjum, N.A.; Umar, S.; Chan, M.T. Ascorbate-Glutathione Pathway and Stress Tolerance in Plants; Springer: Dordrecht, The Netherlands, 2010. [Google Scholar] [CrossRef]
- Rais, L.; Masood, A.; Inam, A.; Khan, N. Sulfur and Nitrogen Co-ordinately Improve Photosynthetic Efficiency, Growth and Proline Accumulation in Two Cultivars of Mustard Under Salt Stress. J. Plant Biochem. Physiol. 2013, 1, 101. [Google Scholar] [CrossRef]
- Fatma, M.; Asgher, M.; Masood, A.; Khan, N.A. Excess sulfur supplementation improves photosynthesis and growth in mustard under salt stress through increased production of glutathione. Environ. Exp. Bot. 2014, 107, 55–63. [Google Scholar] [CrossRef]
- Bystrická, J.; Kavalcová, P.; Vollmannová, A.; Tomáš, J.; Orsák, M. The Role of Sulphur on the Content of Total Polyphenols and Antioxidant Activity in Onion (Allium cerpa L.). Potravin. Sci. J. Food Ind. 2014, 8, 284–289. [Google Scholar] [CrossRef]
- Noctor, G.; Arisi, A.-C.M.; Jouanin, L.; Kunert, K.J.; Rennenberg, H.; Foyer, C.H. Glutathione: Biosynthesis, metabolism and relationship to stress tolerance explored in transformed plants. J. Exp. Bot. 1998, 49, 623–647. [Google Scholar] [CrossRef]
- Khan, I.; Ahmad, A.; Iqbal, M. Modulation of antioxidant defence system for arsenic detoxification in Indian mustard. Ecotoxicol. Environ. Saf. 2009, 72, 626–634. [Google Scholar] [CrossRef]
- Anjum, N.A.; Aref, I.M.; Duarte, A.C.; Pereira, E.; Ahmad, I.; Iqbal, M. Glutathione and proline can coordinately make plants withstand the joint attack of metal (loid) and salinity stresses. Front. Plant Sci. 2014, 5, 662. [Google Scholar] [CrossRef] [PubMed]
- Talukdar, D.; Talukdar, T. Coordinated response of sulfate transport, cysteine biosynthesis, and glutathione-mediated antioxidant defense in lentil (Lens culinaris Medik.) genotypes exposed to arsenic. Protoplasma 2014, 251, 839–855. [Google Scholar] [CrossRef]
- Noji, M.; Saito, K. Metabolic engineering of sulphur assimilation in plants. In Applications of Plant Metabolic Engineering; Verpook, R., Alfermann, A.W., Johnson, T.S., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 297–309. [Google Scholar]
- Astolfi, S.; Zuchi, S. Adequate sulfur supply protects barley plants from adverse effects of salinity stress by increasing thiol contents. Acta Physiol. Plant 2013, 35, 175–181. [Google Scholar] [CrossRef]
- Romero, L.C.; Domínguez-Solís, J.R.; Gutiérrez-Alcalá, G.; Gotor, C. Salt regulation of O-acetylserine (thiol) lyase in Arabidopsis thaliana and increased tolerance in yeast. Plant Physiol. Biochem. 2001, 39, 643–647. [Google Scholar] [CrossRef]
- Fediuc, E.; Lips, S.H.; Erdei, L. O-actylserine (thiol) lyase activity in Phyragmites and Typha plants under cadium and NaCl stress conditions and the involvement of ABA in the stress response. J. Plant Physiol. 2005, 162, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.M.; Blumwald, E. Salinity induced glutathione synthesis in Brassica napus. Planta 2002, 214, 965–969. [Google Scholar] [CrossRef] [PubMed]
- Schnug, E. Physiological functions and environmental relevance of sulfur-containing secondary metabolites. In Sulfur Nutrition and Sulfur Assimilation in Higher Plants: Regulatory, Agricultural and Environmental Aspects; De Kok, L.J., Stulen, I., Rennenberg, H., Brunold, C., Rauser, W., Eds.; SPB Academic: The Hague, The Netherlands, 1993; pp. 179–190. [Google Scholar]
- Randle, W.M.; Lancaster, J.E. Sulphur compounds in Alliums in relation to flavour quality. In Allium Crop Science: Recent Advances; Rabinowitch, H.D., Currah, L., Eds.; CAB International Wallingford: Wallingford, UK, 2002; pp. 329–356. [Google Scholar]
- Lancaster, J.E.; Collin, H.A. Presence of alliinase in isolated vacuoles and of alkyl cysteine sulphoxides in the cytoplasm of bulbs of onion (Allium cepa). Plant Sci. Lett. 1981, 22, 169–176. [Google Scholar] [CrossRef]
- Hell, R.; Kruse, C. Sulfur in biotic interactions of plants. In Sulfur in Plants—An Ecological Perspective; Hawkesford, M.J., De Kok, L.J., Eds.; Springer: Cham, Switzerland, 2007; pp. 197–224. [Google Scholar]
- Khan, T.A.; Mazid, M. Nutritional significance of sulphur in pulse cropping system. Biol. Med. 2011, 3, 114–133. [Google Scholar] [CrossRef]
- Koca, Y.O.; Canavar, Ö.; Kaptan, M.A. Changes of seed yield and quality of maize (Zea mays L.) fertilized with sulphur in early and late sowing date. In Proceedings of the 9th International Soil Science Congress on ‘The Soul of Soil and Civilization’, Side, Antalya, Turkey, 14–16 October 2014; Soil Science Society of Turkey & Federation of Eurasian Soil Science Societies: Antalya, Turkey, 2014; pp. 1002–1007. [Google Scholar]
- Channabasamma, A.; Habsur, N.S.; Bangaremma, S.W.; Akshaya, M.C. Effect of Nitrogen and Sulphur Levels and Ratios on Growth and Yield of Maize. Mol. Plant Breed. 2013, 4, 292–296. [Google Scholar] [CrossRef]
- Jeet, S.; Singh, J.P.; Kumar, R.; Kumar, P.R.; Kumar, P.; Kumari, A.; Prakash, P. Effect of Nitrogen and Sulphur Levels on Yield, Economics and Quality of QPM Hybrids under Dryland Conditions of Eastern Uttar Pradesh, India. J. Agric. Sci. 2012, 4, 31–38. [Google Scholar] [CrossRef]
- Malavolta, E.; Vitti, G.C.; Rosolem, C.A.; Fageria, N.K.; Guimarães, P.T.G. Micronutrients. J. Plant Nutr. 1987, 10, 2153–2158. [Google Scholar] [CrossRef]
- Mathot, M.; Mertens, J.; Verlinden, G.; Lambert, R. Positive effect of sulphur fertilisation grasslands yields and quality in Belgium. Eur. J. Agron. 2008, 28, 655–658. [Google Scholar] [CrossRef]
- Zenda, T.; Yao, D.; Duan, H. Sulphur and chlorine effects on yield and quality in fresh corn. Int. J. Plant Soil Sci. 2017, 18, 1–10. [Google Scholar] [CrossRef]
- Tao, Z.; Chang, X.; Wang, D.; Wang, Y.; Ma, S.; Yang, Y.; Zhao, G. Effects of sulfur fertilization and short-term high temperature on wheat grain production and wheat flour proteins. Crop J. 2018, 6, 413–425. [Google Scholar] [CrossRef]
- Uchida, R. Essential Nutrients for Plant Growth: Nutrient Functions and Deficiency Symptoms. In Plant Nutrient Management in Hawaii’s Soils, Approaches for Tropical and Subtropical Agriculture; Silva, J.A., Uchida, R., Eds.; College of Tropical Agriculture and Human Resources, University of Hawaii at Manoa: Honolulu, HI, USA, 2000; pp. 1–55. [Google Scholar]
- Schnug, E.; Haneklaus, S. Diagnosis of Sulphur Nutrition. In Sulphur in Agroecosystems; Schnug, E., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1998; pp. 1–38. [Google Scholar] [CrossRef]
- Mikkelsen, R.; Norton, R. Soil and Fertilizer Sulfur. Better Crops 2013, 97, 7–9. [Google Scholar]
- Saskatchewan. Sulphur Fertilization in Crop Production. 2020. Available online: https://www.saskatchewan.ca/business/agriculture-natural-resources-and-industry/agribusiness-farmers-and-ranchers/crops-and-irrigation/soils-fertility-and-nutrients/sulphur-fertilization-in-crop-production (accessed on 2 September 2020).
- Tandon, H.L.S. Sulphur Research and Agricultural Production in India, 3rd ed.; The Sulphur Institute: Washington, DC, USA, 1991; pp. 257–343. [Google Scholar]
- Ohkama-Ohtsu, N.; Wasaki, J. Recent Progress in Plant Nutrition, Research: Cross-Talk Between Nutrients, Plant Physiology and Soil Microorganisms. Plant Cell Physiol. 2010, 51, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Li, C.; Yao, Y.; Li, M.; Liu, L. Modern imaging techniques in plant nutrition analysis: A review. Comput. Electron. Agric. 2020, 174, 105459. [Google Scholar] [CrossRef]
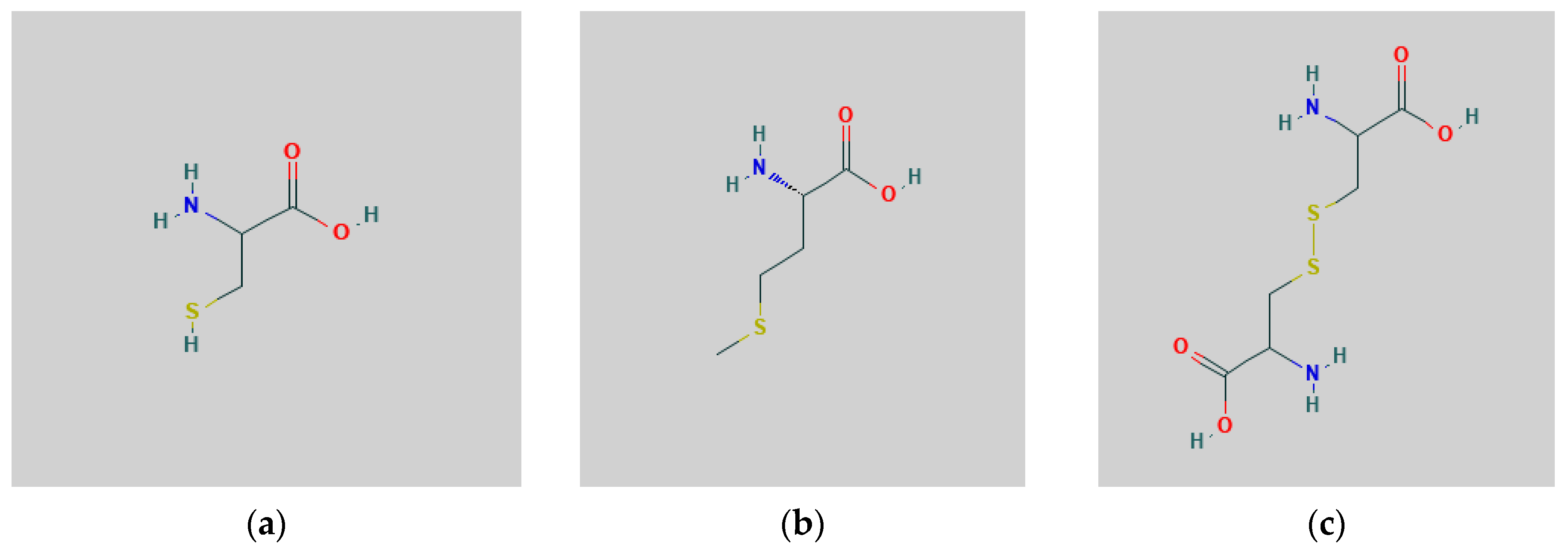

| Crop Species | N:S Ratio | Reference |
|---|---|---|
| Cereals | 10:1 | [81] |
| Maize | 15.6:1 | [82] |
| Wheat | 7.1:1 | [82] |
| Oil rapeseed | 5:1 | [81] |
| Oil rapeseed | 7:1 | [80] |
| Mustard | 3.1:1 | [81] |
| Grassland–grazing | 8–12:1 | [81] |
| Potato (Solanum tuberosum L.) | 10.1:1 | [81] |
| Potato | 12:1 | [83] |
| Groundnut | 14.8:1 | [82] |
| Crop Species | Optimum S Rate (kg ha−1) | References |
|---|---|---|
| Cereals | 24–40 | [43,133] |
| Cereals | 25–50 | [83] |
| Cereals | 30–40 | [9] |
| Winter cereals | 40–50 | [81] |
| Spring cereals | 20–25 | [81] |
| Oilseeds | 10–150 | [43,133] |
| Oilseeds | 30–60 | [9] |
| Oil rapeseed | 50–75 | [83] |
| Oil rapeseed | 75–100 | [81] |
| Oil rape seed | >58.3 | [34] |
| Pulses | 20–40 | [43,133] |
| Pulses | 30–45 | [9] |
| Peas | 25 | [83] |
| Fodder | 25–100 | [43,133] |
| Fodder | 40–60 | [9] |
| Grass for silage | 40 | [83] |
| Grassland -silage | 40 | [81] |
| Grass for grazing | 20–30/cut | [83] |
| Grassland -grazing | 10/month | [81] |
| Tubers | 25–60 | [43,133] |
| Potato | 40–80 | [9] |
| Onion and garlic | 40–50 | [9] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zenda, T.; Liu, S.; Dong, A.; Duan, H. Revisiting Sulphur—The Once Neglected Nutrient: It’s Roles in Plant Growth, Metabolism, Stress Tolerance and Crop Production. Agriculture 2021, 11, 626. https://doi.org/10.3390/agriculture11070626
Zenda T, Liu S, Dong A, Duan H. Revisiting Sulphur—The Once Neglected Nutrient: It’s Roles in Plant Growth, Metabolism, Stress Tolerance and Crop Production. Agriculture. 2021; 11(7):626. https://doi.org/10.3390/agriculture11070626
Chicago/Turabian StyleZenda, Tinashe, Songtao Liu, Anyi Dong, and Huijun Duan. 2021. "Revisiting Sulphur—The Once Neglected Nutrient: It’s Roles in Plant Growth, Metabolism, Stress Tolerance and Crop Production" Agriculture 11, no. 7: 626. https://doi.org/10.3390/agriculture11070626
APA StyleZenda, T., Liu, S., Dong, A., & Duan, H. (2021). Revisiting Sulphur—The Once Neglected Nutrient: It’s Roles in Plant Growth, Metabolism, Stress Tolerance and Crop Production. Agriculture, 11(7), 626. https://doi.org/10.3390/agriculture11070626






