Biogenic Selenium Nanoparticles in Animal Nutrition: A Review
Abstract
1. Introduction
2. The Importance of Selenium in Animal Nutrition
3. Selenium Supplementation
3.1. Selenium Absorption and Bioavailability
3.2. Selenium and Vitamin E
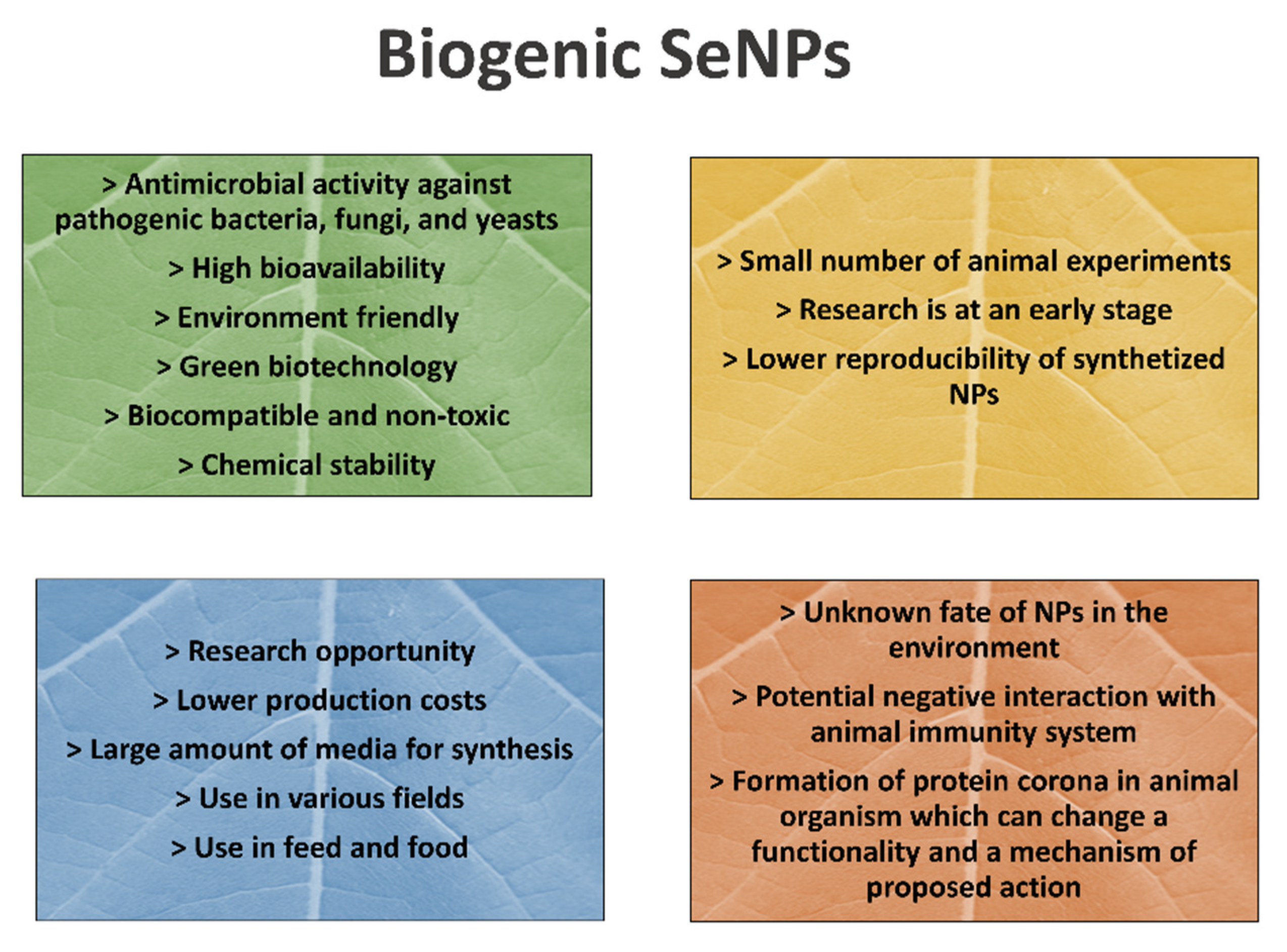
4. Selenium Nanoparticles in Animal Nutrition
4.1. Green Synthesis of SeNPs
4.2. Antimicrobial Potential of SeNp Produced via Green Synthesis
4.3. Synthesis by Plants and Microorganisms
4.4. Synthesis of SeNPs by Marine Algae and Microalgae
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kurokawa, S.; Berry, M.J. Selenium. Role of the essential metalloid in health. In Interrelations between Essential Metal Ions and Human Diseases; Springer: Cham, Switzerland, 2013; pp. 499–534. [Google Scholar]
- Moxon, A.L. Alkali Disease or Selenium Poisoning. Bulletins 1937, 311. [Google Scholar] [CrossRef]
- White, P.J. Selenium accumulation by plants. Ann. Bot. 2015, 117, 217–235. [Google Scholar] [CrossRef]
- Doj, R.; Khanal, D.; Knight, A. Selenium: Its Role in Livestock Health and Productivity. J. Agric. Environ. 2010, 11, 101–106. [Google Scholar] [CrossRef]
- Garousi, F. The essentiality of selenium for humans, animals, and plants, and the role of selenium in plant metabolism and physiology. Acta Univ. Sapientiae Aliment 2017, 10, 75–90. [Google Scholar] [CrossRef]
- James, L.F.; Shupe, J.L. Selenium poisoning in livestock. Rangelands 1984, 6, 64–67. [Google Scholar]
- Pilon-Smits, E.A.H. On the Ecology of Selenium Accumulation in Plants. Plants 2019, 8, 197. [Google Scholar] [CrossRef]
- Carlson, B.A.; Yoo, M.H.; Shrimali, R.K.; Irons, R.; Gladyshev, V.N.; Hatfield, D.L.; Park, J.M. Role of selenium-containing proteins in T-cell and macrophage function. Proc. Nutr. Soc. 2010, 69, 300–310. [Google Scholar] [CrossRef]
- Antonyak, H.; Iskra, R.; Panas, N.; Lysiuk, R. Selenium. In Trace Elements and Minerals in Health and Longevity. Healthy Ageing and Longevity; Malavolta, M., Mocchegiani, E., Eds.; Springer: Cham, Switzerland, 2018; Volume 8. [Google Scholar]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular pathways and physiological roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef]
- Horký, P.; Skladanka, J.; Nevrkla, P.; Slama, P. 16. Effect of Diet Supplemented with Antioxidants (Selenium, Copper, Vitamins E and C) on Antioxidant Status and Ejaculate Quality of reeding Boars. Ann. Anim. Sci. 2016, 16, 521–532. [Google Scholar] [CrossRef]
- Tóth, R.J.; Csapó, J. The role of selenium in nutrition—A review. Acta Univ. Sapientiae Aliment 2018, 11, 128–144. [Google Scholar] [CrossRef]
- Wachowicz, B.; Zbikowska, H.; Nowak, P. Selenium compounds in the environment; Their effect on human health. Cell. Mol. Biol. Lett. 2001, 6, 375–381. [Google Scholar]
- Skalickova, S.; Milosavljevic, V.; Cihalova, K.; Horky, P.; Richtera, L.; Adam, V. Selenium nanoparticles as a nutritional supplement. Nutrition 2017, 33, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Sharadamma, K.C.; Purushotham, B.; Radhakrishna, P.M.; Abhilekha, P.M.; Vagdevi, H.M. Role of Selenium in Pets Health and Nutrition: A Review. Asian J. Anim. Sci. 2011, 5, 64–70. [Google Scholar] [CrossRef][Green Version]
- Edens, F.W.; Sefton, A.E. Organic selenium in animal nutrition—Utilisation, metabolism, storage and comparison with other selenium sources. J. Appl. Anim. Nutr. 2016, 4, e9. [Google Scholar] [CrossRef]
- Shini, S.; Sultan, A.; Bryden, W.L. Selenium Biochemistry and Bioavailability: Implications for Animal Agriculture. Agriculture 2015, 5, 1277–1288. [Google Scholar] [CrossRef]
- Saha, U.K.; Fayiga, A.O.; Hancock, D.W.; Sonon, L.S. Selenium in Animal Nutrition: Deficiencies in Soils and Forages, Requirements, Supplementation and Toxicity. Int. J. Appl. Agric. Sci. 2016, 2, 112. [Google Scholar] [CrossRef]
- Mehdi, Y.; Dufrasne, I. Selenium in Cattle: A Review. Molecules 2016, 21, 545. [Google Scholar] [CrossRef]
- Sobiech, P.; Żarczyńska, K. The influence of selenium deficiency on chosen biochemical parameters and histopathological changes in muscles of goat kids. Pol. J. Vet. Sci. 2020, 23, 267–279. [Google Scholar] [CrossRef]
- Rodriguez, A.M.; Schild, C.O.; Cantón, G.J.; Riet-Correa, F.; Armendano, J.I.; Caffarena, R.D.; Brambilla, E.C.; García, J.A.; Morrell, E.L.; Poppenga, R.; et al. White muscle disease in three selenium deficient beef and dairy calves in Argentina and Uruguay. Ciência Rural 2018, 48. [Google Scholar] [CrossRef]
- Hosnedlova, B.; Kepinska, M.; Skalickova, S.; Fernandez, C.; Ruttkay-Nedecky, B.; Malevu, T.D.; Sochor, J.; Baron, M.; Melcova, M.; Zidkova, J.; et al. A Summary of New Findings on the Biological Effects of Selenium in Selected Animal Species—A Critical Review. Int. J. Mol. Sci. 2017, 18, 2209. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, D.; Zhang, J.; Bao, J.; Tang, C.; Zhang, Z. The role of necroptosis and apoptosis through the oxidative stress pathway in the liver of selenium-deficient swine. Metallomics 2020, 12, 607–616. [Google Scholar] [CrossRef]
- Xiao, J.; Khan, M.Z.; Ma, Y.; Alugongo, G.M.; Ma, J.; Chen, T.; Khan, A.; Cao, Z. The Antioxidant Properties of Selenium and Vitamin E.; Their Role in Periparturient Dairy Cattle Health Regulation. Antioxidants 2021, 10, 1555. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, Y.; Chen, B.; Zhao, B.; Gao, X.-J. Selenium Deficiency Promotes Oxidative Stress-Induced Mastitis via Activating the NF-κB and MAPK Pathways in Dairy Cow. Biol. Trace Elem. Res. 2021. [Google Scholar] [CrossRef]
- O’Brien, B.; Hennessy, D. Scientific appraisal of the Irish grass-based milk production system as a sustainable source of premium quality milk and dairy products. Irish J. Agric. Food Res. 2017, 56, 120–129. [Google Scholar] [CrossRef]
- Zust, J.; Hrovatin, B.; Simundić, B. Assessment of selenium and vitamin E deficiencies in dairy herds and clinical disease in calves. Vet. Rec. 1996, 139, 391–394. [Google Scholar] [CrossRef]
- Ademi, A.; Bernhoft, A.; Govasmark, E.; Bytyqi, H.; Sivertsen, T.; Singh, B.R. Selenium and other mineral concentrations in feed and sheep’s blood in Kosovo. Transl. Anim. Sci. 2017, 1, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.A.; Bobe, G.; Vorachek, W.R.; Hugejiletu; Gorman, M.E.; Mosher, W.D.; Pirelli, G.J. Effects of feeding selenium-enriched alfalfa hay on immunity and health of weaned beef calves. Biol. Trace Elem. Res. 2013, 156, 96–110. [Google Scholar] [CrossRef]
- Tolu, J.; Thiry, Y.; Bueno, M.; Jolivet, C.; Potin-Gautier, M.; Le Hécho, I. Distribution and speciation of ambient selenium in contrasted soils, from mineral to organic rich. Sci. Total Environ. 2014, 479–480, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Galgan, V.; Frank, A. Survey of bioavailable selenium in Sweden with the moose (Alces alces L.) as monitoring animal. Sci. Total Environ. 1995, 172, 37–45. [Google Scholar] [CrossRef]
- Alfthan, G.; Eurola, M.; Ekholm, P.; Venäläinen, E.R.; Root, T.; Korkalainen, K.; Hartikainen, H.; Salminen, P.; Hietaniemi, V.; Aspila, P.; et al. Effects of nationwide addition of selenium to fertilizers on foods, and animal and human health in Finland: From deficiency to optimal selenium status of the population. J. Trace Elem. Med. Biol. 2015, 31, 142–147. [Google Scholar] [CrossRef]
- Lemly, D. Aquatic selenium pollution is a global environmental safety issue. Ecotoxicol. Environ. Saf. 2004, 59, 44–56. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Nutrient Requirements of Beef Cattle; The National Research Council: Washington, DC, USA, 2016. [Google Scholar]
- Surai, P.F.; Kochish, I.I. Food for thought: Nano-selenium in poultry nutrition and health. Anim. Health Res. Rev. 2020, 21, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Mavromichalis, I. Formulating Pig Diets: Selenium Toxicity, Deficiency. Available online: https://www.wattagnet.com/articles/19843-formulating-pig-diets-selenium-toxicity-deficiency (accessed on 3 November 2021).
- National Research Council. Nutrient Requirements of Swine; National Academies Press: Washington, DC, USA, 2012. [Google Scholar]
- Olson, O.E. Selenium Toxicity in Animals with Emphasis on Man. J. Am. Coll. Toxicol. 1986, 5, 45–70. [Google Scholar] [CrossRef]
- Hall, J.O. Acute Selenium Toxicosis. In Merck Veterinary Manual; Utah State University: Logan, UT, USA, 2014. [Google Scholar]
- National Research Council. Nutrient Requirements of Small Ruminants: Sheep, Goats, Cervids, and New World Camelids; The National Academies Press: Washington, DC, USA, 2007; p. 384. [Google Scholar]
- Van Saun, R.J. Selenium nutrition and therapy in small ruminants. In Proceedings of the American Association of Bovine Practitioners Proceedings of the Annual Conference, Montreal, QC, Canada, 20–22 September 2012; pp. 148–154. [Google Scholar]
- Council, N.R. Nutrient Requirements of Horses: Sixth Revised Edition; The National Academies Press: Washington, DC, USA, 2007; p. 360. [Google Scholar]
- Arshad, M.A.; Ebeid, H.M.; Hassan, F.-U. Revisiting the Effects of Different Dietary Sources of Selenium on the Health and Performance of Dairy Animals: A Review. Biol. Trace Elem. Res. 2021, 199, 3319–3337. [Google Scholar] [CrossRef]
- McDowell. Minerals in Animal and Human Nutrition; Elsevier: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Sun, P.; Wang, J.; Liu, W.; Bu, D.P.; Liu, S.J.; Zhang, K.Z. Hydroxy-selenomethionine: A novel organic selenium source that improves antioxidant status and selenium concentrations in milk and plasma of mid-lactation dairy cows. J. Dairy Sci. 2017, 100, 9602–9610. [Google Scholar] [CrossRef]
- Singh, J.; Dutta, T.; Kim, K.-H.; Rawat, M.; Samddar, P.; Kumar, P. ‘Green’ synthesis of metals and their oxide nanoparticles: Applications for environmental remediation. J. Nanobiotechnol. 2018, 16, 84. [Google Scholar] [CrossRef]
- Bhattacharjee, A.; Basu, A.; Bhattacharya, S. Selenium nanoparticles are less toxic than inorganic and organic selenium to mice in vivo. Nucleus 2019, 62, 259–268. [Google Scholar] [CrossRef]
- Gangadoo, S.; Dinev, I.; Willson, N.L.; Moore, R.J.; Chapman, J.; Stanley, D. Nanoparticles of selenium as high bioavailable and non-toxic supplement alternatives for broiler chickens. Environ. Sci. Pollut. Res. Int. 2020, 27, 16159–16166. [Google Scholar] [CrossRef]
- Hosnedlova, B.; Kepinska, M.; Skalickova, S.; Fernandez, C.; Ruttkay-Nedecky, B.; Peng, Q.; Baron, M.; Melcova, M.; Opatrilova, R.; Zidkova, J.; et al. Nano-selenium and its nanomedicine applications: A critical review. Int. J. Nanomed. 2018, 13, 2107–2128. [Google Scholar] [CrossRef]
- Schlegel, P.; Durosoy, S.; Jongbloed, A.W. Trace Elements in Animal Production Systems; Wageningen Academic Publishers: Wageningen, The Netherlands, 2008. [Google Scholar]
- Mahima; Verma, A.; Kumar, A.; Rahal, A.; Sidhu, V.K.; Roy, D. Inorganic versus organic selenium supplementation: A Review. Pak. J. Biol. Sci. PJBS 2012, 15, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Schrauzer, G.N. The nutritional significance, metabolism and toxicology of selenomethionine. Adv. Food Nutr. Res. 2003, 47, 73–112. [Google Scholar] [CrossRef]
- Zoidis, E.; Seremelis, I.; Kontopoulos, N.; Danezis, G.P. Selenium-Dependent Antioxidant Enzymes: Actions and Properties of Selenoproteins. Antioxidants 2018, 7, 66. [Google Scholar] [CrossRef] [PubMed]
- Ghaderzadeh, S.; Mirzaei Aghjeh-Gheshlagh, F.; Nikbin, S.; Navidshad, B. Review on properties of selenium in animal nutrition. Iran. J. Appl. Anim. Sci. 2016, 6, 753–761. [Google Scholar]
- Rehman, A.; John, P.; Bhatti, A. Biogenic Selenium Nanoparticles: Potential Solution to Oxidative Stress Mediated Inflammation in Rheumatoid Arthritis and Associated Complications. Nanomaterials 2021, 11, 2005. [Google Scholar] [CrossRef] [PubMed]
- Stefanowicz, F.A.; Talwar, D.; O’Reilly, D.S.; Dickinson, N.; Atkinson, J.; Hursthouse, A.S.; Rankin, J.; Duncan, A. Erythrocyte selenium concentration as a marker of selenium status. Clin. Nutr. 2013, 32, 837–842. [Google Scholar] [CrossRef]
- Grossi, S.; Rossi, L.; De Marco, M.; Sgoifo Rossi, C.A. The Effect of Different Sources of Selenium Supplementation on the Meat Quality Traits of Young Charolaise Bulls during the Finishing Phase. Antioxidants 2021, 10, 596. [Google Scholar] [CrossRef]
- Mehdi, Y.; Hornick, J.-L.; Istasse, L.; Dufrasne, I. Selenium in the Environment, Metabolism and Involvement in Body Functions. Molecules 2013, 18, 3292–3311. [Google Scholar] [CrossRef]
- Surai, P.F.; Kochish, I.I.; Fisinin, V.I.; Velichko, O.A. Selenium in Poultry Nutrition: From Sodium Selenite to Organic Selenium Sources. J. Poult. Sci. 2018, 55, 79–93. [Google Scholar] [CrossRef]
- Sunde, R.A. Molecular Biology of Selenoproteins. Annu. Rev. Nutr. 1990, 10, 451–474. [Google Scholar] [CrossRef]
- Dalgaard, T.S.; Briens, M.; Engberg, R.M.; Lauridsen, C. The influence of selenium and selenoproteins on immune responses of poultry and pigs. Anim. Feed Sci. Technol. 2018, 238, 73–83. [Google Scholar] [CrossRef]
- Romero-Pérez, A.; García-García, E.; Zavaleta-Mancera, A.; Ramírez-Bribiesca, J.E.; Revilla-Vázquez, A.; Hernández-Calva, L.M.; López-Arellano, R.; Cruz-Monterrosa, R.G. Designing and evaluation of sodium selenite nanoparticles in vitro to improve selenium absorption in ruminants. Vet. Res. Commun. 2010, 34, 71–79. [Google Scholar] [CrossRef]
- Whanger, P.D. Selenocompounds in plants and animals and their biological significance. J. Am. Coll. Nutr. 2002, 21, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Kieliszek, M.; Błażejak, S.; Gientka, I.; Bzducha-Wróbel, A. Accumulation and metabolism of selenium by yeast cells. Appl. Microbiol. Biotechnol. 2015, 99, 5373–5382. [Google Scholar] [CrossRef] [PubMed]
- Vignola, G.; Lambertini, L.; Mazzone, G.; Giammarco, M.; Tassinari, M.; Martelli, G.; Bertin, G. Effects of selenium source and level of supplementation on the performance and meat quality of lambs. Meat Sci. 2009, 81, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Paiva, F.A.; Netto, A.S.; Corrêa, L.B.; Silva, T.H.; Guimarães, I.C.S.B.; Claro, G.R.d.; Cunha, J.A.; Zanetti, M.A. Organic selenium supplementation increases muscle selenium content in growing lambs compared to inorganic source. Small Rumin. Res. 2019, 175, 57–64. [Google Scholar] [CrossRef]
- Steen, A.; Strøm, T.; Bernhoft, A. Organic selenium supplementation increased selenium concentrations in ewe and newborn lamb blood and in slaughter lamb meat compared to inorganic selenium supplementation. Acta Vet. Scand. 2008, 50, 7. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.S.; Rodriguez, F.D.; Trettel, M.; Abal, R.T.; Lima, C.G.; Yoshikawa, C.Y.C.; Zanetti, M.A. Performance, carcass characteristics and meat quality of Nellore cattle supplemented with supranutritional doses of sodium selenite or selenium-enriched yeast. Animal 2020, 14, 215–222. [Google Scholar] [CrossRef]
- Hall, J.A.; Van Saun, R.J.; Bobe, G.; Stewart, W.C.; Vorachek, W.R.; Mosher, W.D.; Nichols, T.; Forsberg, N.E.; Pirelli, G.J. Organic and inorganic selenium: I. Oral bioavailability in ewes. J. Anim. Sci. 2012, 90, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Galbraith, M.L.; Vorachek, W.R.; Estill, C.T.; Whanger, P.D.; Bobe, G.; Davis, T.Z.; Hall, J.A. Rumen Microorganisms Decrease Bioavailability of Inorganic Selenium Supplements. Biol. Trace Elem. Res. 2016, 171, 338–343. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Garg, A.K.; Dass, R.S.; Chaturvedi, V.K.; Mudgal, V.; Varshney, V.P. Selenium supplementation influences growth performance, antioxidant status and immune response in lambs. Anim. Feed Sci. Technol. 2009, 153, 77–87. [Google Scholar] [CrossRef]
- Juniper, D.T.; Phipps, R.H.; Givens, D.I.; Jones, A.K.; Green, C.; Bertin, G. Tolerance of ruminant animals to high dose in-feed administration of a selenium-enriched yeast1. J. Anim. Sci. 2008, 86, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Surai, P. Selenium in poultry nutrition 1. Antioxidant properties, deficiency and toxicity. World’s Poult. Sci. J. 2002, 58, 333–347. [Google Scholar] [CrossRef]
- Faixová, Z.; Piešová, E.; Maková, Z.; Čobanová, K.; Faix, Š. Effect of dietary supplementation with selenium-enriched yeast or sodium selenite on ruminal enzyme activities and blood chemistry in sheep. Acta Vet. Brno 2016, 85, 185–194. [Google Scholar] [CrossRef]
- Pavlata, L.; Misurova, L.; Pechova, A.; Dvorak, R. The effect of inorganic and organically bound forms of selenium on glutathione peroxidase activity in the blood of goats. Vet. Med. 2011, 56, 75–81. [Google Scholar] [CrossRef]
- Koenig, K.M.; Rode, L.M.; Cohen, R.D.; Buckley, W.T. Effects of diet and chemical form of selenium on selenium metabolism in sheep. J. Anim. Sci. 1997, 75, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Heindl, J.; Ledvinka, Z.; Englmaierová, M.; Zita, L.; Tumová, E. The effect of dietary selenium sources and levels on performance, selenium content in muscle and glutathione peroxidase activity in broiler chickens. Czech J. Anim. Sci. 2010, 55, 572–578. [Google Scholar] [CrossRef]
- Wang, Z.-N.; Li, H.; Tang, H.; Zhang, S.-J.; Pauline, M.; Bi, C.-L. Short Communication: Effects of Dietary Selenium Supplementation on Selenium Deposition and Antioxidant Status in Postpartum Mice. Biol. Trace Elem. Res. 2021, 199, 1488–1492. [Google Scholar] [CrossRef]
- Novoselec, J.; Šperanda, M.; Klir, Ž.; Mioč, B.; Steiner, Z.; Antunović, Z. Blood biochemical indicators and concentration of thyroid hormones in heavily pregnant and lactating ewes depending on selenium supplementation. Acta Vet. Brno 2018, 86, 353–363. [Google Scholar] [CrossRef]
- Ortman, K.; Pehrson, B. Effect of selenate as a feed supplement to dairy cows in comparison to selenite and selenium yeast. J. Anim. Sci. 1999, 77, 3365–3370. [Google Scholar] [CrossRef]
- Petrera, F.; Calamari, L.; Bertin, G. Effect of either sodium selenite or Se–yeast supplementation on selenium status and milk characteristics in dairy goats. Small Rumin. Res. 2009, 82, 130–138. [Google Scholar] [CrossRef]
- Kachuee, R.; Abdi-Benemar, H.; Mansoori, Y.; Sánchez-Aparicio, P.; Seifdavati, J.; Elghandour, M.M.M.Y.; Guillén, R.J.; Salem, A.Z.M. Effects of Sodium Selenite, L-Selenomethionine, and Selenium Nanoparticles during Late Pregnancy on Selenium, Zinc, Copper, and Iron Concentrations in Khalkhali Goats and Their Kids. Biol. Trace Elem. Res. 2019, 191, 389–402. [Google Scholar] [CrossRef] [PubMed]
- Kouba, A.; Velíšek, J.; Stará, A.; Masojídek, J.; Kozák, P. Supplementation with sodium selenite and selenium-enriched microalgae biomass show varying effects on blood enzymes activities, antioxidant response, and accumulation in common barbel (Barbus barbus). BioMed Res. Int. 2014, 2014, 408270. [Google Scholar] [CrossRef] [PubMed]
- Doucha, J.; Lívanský, K.; Kotrbácek, V.; Zachleder, V. Production of Chlorella biomass enriched by selenium and its use in animal nutrition: A review. Appl. Microbiol. Biotechnol. 2009, 83, 1001–1008. [Google Scholar] [CrossRef]
- Marounek, M.; Dokoupilová, A.; Volek, Z.; Hoza, I. Quality of meat and selenium content in tissues of rabbits fed diets supplemented with sodium selenite, selenized yeast and selenized algae. World Rabbit Sci. 2009, 17, 207–212. [Google Scholar] [CrossRef][Green Version]
- Hassan, F.A.; Abdel-Azeem, N.; Abdel-Rahman, S.; Amin, H.F.; Abdel-Mawla, L.F. Effect of Dietary Organic Selenium Supplementation yfon Growth Performance, Carcass Characteristics and Antioxidative Status of Growing Rabbits. World Vet. J. 2019, 9, 16–25. Available online: www.wvj.science-line.com (accessed on 3 November 2021).
- Amer, F.; Hoballah, E.; Basyony, M.; El-Medany, S. Effect of dietary selenium enriched micro-algae supplementation on growth performance and anti-oxidative status of rabbits under high ambient temperature in summer season. Egypt. J. Nutr. Feeds 2015, 18, 229–244. [Google Scholar] [CrossRef]
- De Alcantara, S.; Lopes, C.C.; Wagener, K. Controlled introduction of selenium into Chlorella cells. Indian J. Exp. Biol. 1998, 36, 1286–1288. [Google Scholar]
- Neumann, P.M.; De Souza, M.P.; Pickering, I.J.; Terry, N. Rapid microalgal metabolism of selenate to volatile dimethylselenide. Plant Cell Environ. 2003, 26, 897–905. [Google Scholar] [CrossRef]
- Brummer, F.A.; Perelli, F.J.; Hall, J.A. Selenium Supplementation Strategies for Livestock in Oregon. 2014. Available online: https://catalog.extension.oregonstate.edu/sites/catalog/files/project/pdf/em9094.pdf (accessed on 3 November 2021).
- Smith, K.L.; Harrison, J.H.; Hancock, D.D.; Todhunter, D.A.; Conrad, H.R. Effect of vitamin E and selenium supplementation on incidence of clinical mastitis and duration of clinical symptoms. J. Dairy Sci. 1984, 67, 1293–1300. [Google Scholar] [CrossRef]
- Zahrazadeh, M.; Riasi, A.; Farhangfar, H.; Mahyari, S.A. Effects of close-up body condition score and selenium-vitamin E injection on lactation performance, blood metabolites, and oxidative status in high-producing dairy cows. J. Dairy Sci. 2018, 101, 10495–10504. [Google Scholar] [CrossRef] [PubMed]
- Hogan, J.S.; Weiss, W.P.; Smith, K.L. Role of vitamin E and selenium in host defense against mastitis. J. Dairy Sci. 1993, 76, 2795–2803. [Google Scholar] [CrossRef]
- Talib, A.; Bomboi, G.; Basilio, F. Does Vitamin E or Vitamin E plus Selenium improve reproductive performance of rams during hot weather? Ital. J. Anim. Sci. 2010, 8, 743–754. [Google Scholar] [CrossRef]
- Marin-Guzman, J.; Mahan, D.C.; Chung, Y.K.; Pate, J.L.; Pope, W.F. Effects of dietary selenium and vitamin E on boar performance and tissue responses, semen quality, and subsequent fertilization rates in mature gilts. J. Anim. Sci. 1997, 75, 2994–3003. [Google Scholar] [CrossRef]
- Kappel, L.C.; Ingraham, R.H.; Morgan, E.B.; Dixon, J.M.; Zeringue, L.; Wilson, D.; Babcock, D.K. Selenium concentrations in feeds and effects of treating pregnant Holstein cows with selenium and vitamin E on blood selenium values and reproductive performance. Am. J. Vet. Res. 1984, 45, 691–694. [Google Scholar]
- Dalia, A.M.; Loh, T.C.; Sazili, A.Q.; Jahromi, M.F.; Samsudin, A.A. Effects of vitamin E, inorganic selenium, bacterial organic selenium, and their combinations on immunity response in broiler chickens. BMC Vet. Res. 2018, 14, 249. [Google Scholar] [CrossRef]
- El-Hak, H.N.G.; Elaraby, E.E.; Hassan, A.K.; Abbas, O.A. Study of the toxic effect and safety of vitamin E supplement in male albino rats after 30 days of repeated treatment. Heliyon 2019, 5, e02645. [Google Scholar] [CrossRef]
- Hale, T.W.; Rais-Bahrami, K.; Montgomery, D.L.; Harkey, C.; Habersang, R.W. Vitamin E Toxicity in Neonatal Piglets. J. Toxicol. Clin. Toxicol. 1995, 33, 123–130. [Google Scholar] [CrossRef]
- Horky, P.; Ruttkay-Nedecky, B.; Nejdl, L.; Richtera, L.; Cernei, N.; Pohanka, M.; Kopel, P.; Skladanka, J.; Hloucalova, P.; Slama, P.; et al. Electrochemical Methods for Study of Influence of Selenium Nanoparticles on Antioxidant Status of Rats. Int. J. Electrochem. Sci. 2016, 11, 2799–2824. [Google Scholar] [CrossRef]
- Fratoddi, I. Hydrophobic and Hydrophilic Au and Ag Nanoparticles. Breakthroughs and Perspectives. Nanomaterials 2017, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.P.; Labhasetwar, V.; Walter, E.; Levy, R.J.; Amidon, G.L. The mechanism of uptake of biodegradable microparticles in Caco-2 cells is size dependent. Pharm. Res. 1997, 14, 1568–1573. [Google Scholar] [CrossRef] [PubMed]
- Corbo, C.; Molinaro, R.; Parodi, A.; Toledano Furman, N.E.; Salvatore, F.; Tasciotti, E. The impact of nanoparticle protein corona on cytotoxicity, immunotoxicity and target drug delivery. Nanomedicine (Lond.) 2016, 11, 81–100. [Google Scholar] [CrossRef]
- Abdelnour, S.; Alagawany, M.; Hashem, N.; Farag, M.; Alghamdi, E.; Hassan, F.-u.; Bilal, R.; Elnesr, S.; Dawood, M.; Nagadi, S.; et al. Nanominerals: Fabrication Methods, Benefits and Hazards, and Their Applications in Ruminants with Special Reference to Selenium and Zinc Nanoparticles. Animals 2021, 11, 1916. [Google Scholar] [CrossRef]
- Trang, H.D.N. Antibacterial properties of selenium nanoparticles and their toxicity to Caco-2 cells. Food Control 2017, 77, 17–24. [Google Scholar] [CrossRef]
- Shakibaie, M.; Forootanfar, H.; Golkari, Y.; Mohammadi-Khorsand, T.; Shakibaie, M.R. Anti-biofilm activity of biogenic selenium nanoparticles and selenium dioxide against clinical isolates of Staphylococcus aureus, Pseudomonas aeruginosa, and Proteus mirabilis. J. Trace Elem. Med. Biol. 2015, 29, 235–241. [Google Scholar] [CrossRef]
- Keyhani, A.; Zia-ali, N.; Shakibaie, M.; Kareshk, A.; Shojaee, S.; Asadi-Shekaari, M.; Sepahvand, M.; Mahmoudvand, H. Biogenic selenium nanoparticles target chronic toxoplasmosis with minimal cytotoxicity in a mouse model. J. Med. Microbiol. 2019, 69. [Google Scholar] [CrossRef] [PubMed]
- El-Zayat, M.M.; Eraqi, M.M.; Alrefai, H.; El-Khateeb, A.Y.; Ibrahim, M.A.; Aljohani, H.M.; Aljohani, M.M.; Elshaer, M.M. The Antimicrobial, Antioxidant, and Anticancer Activity of Greenly Synthesized Selenium and Zinc Composite Nanoparticles Using Ephedra aphylla Extract. Biomolecules 2021, 11, 470. [Google Scholar] [CrossRef] [PubMed]
- Lv, Q.; Liang, X.; Nong, K.; Gong, Z.; Qin, T.; Qin, X.; Wang, D.; Zhu, Y. Advances in Research on the Toxicological Effects of Selenium. Bull. Environ. Contam. Toxicol. 2021, 106, 715–726. [Google Scholar] [CrossRef]
- Baig, N.; Kammakakam, I.; Falath, W.S. Nanomaterials: A review of synthesis, properties, recent progress, and challenges. Mater. Adv. 2021, 2, 1821–1871. [Google Scholar] [CrossRef]
- Sabella, S.; Carney, R.P.; Brunetti, V.; Malvindi, M.A.; Al-Juffali, N.; Vecchio, G.; Janes, S.M.; Bakr, O.M.; Cingolani, R.; Stellacci, F.; et al. A general mechanism for intracellular toxicity of metal-containing nanoparticles. Nanoscale 2014, 6, 7052–7061. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, A. Toxicity of nanoparticles_ challenges and opportunities. Appl. Microsc. 2019, 49, 2. [Google Scholar] [CrossRef]
- Maharramov, A.M.; Hasanova, U.A.; Suleymanova, I.A.; Osmanova, G.E.; Hajiyeva, N.E. The engineered nanoparticles in food chain: Potential toxicity and effects. SN Appl. Sci. 2019, 1, 1362. [Google Scholar] [CrossRef]
- Elhardallou, S.; Babiker, W.; Sulieman, A.M.; Gobouri, A. Effect of Diet Supplementation with Food Industry By-Products on Diabetic Rats. Food Nutr. Sci. 2015, 6, 875–882. [Google Scholar] [CrossRef][Green Version]
- Boudreau, M.D.; Imam, M.S.; Paredes, A.M.; Bryant, M.S.; Cunningham, C.K.; Felton, R.P.; Jones, M.Y.; Davis, K.J.; Olson, G.R. Differential Effects of Silver Nanoparticles and Silver Ions on Tissue Accumulation, Distribution, and Toxicity in the Sprague Dawley Rat Following Daily Oral Gavage Administration for 13 Weeks. Toxicol. Sci. 2016, 150, 131–160. [Google Scholar] [CrossRef]
- Khan, F.H. Chemical hazards of nanoparticles to human and environment (a review). Orient. J. Chem. 2013, 29, 1399. [Google Scholar] [CrossRef]
- Buchman, J.T.; Hudson-Smith, N.V.; Landy, K.M.; Haynes, C.L. Understanding Nanoparticle Toxicity Mechanisms To Inform Redesign Strategies To Reduce Environmental Impact. Acc. Chem. Res. 2019, 52, 1632–1642. [Google Scholar] [CrossRef]
- Shi, L.; Xun, W.; Yue, W.; Zhang, C.; Ren, Y.; Qiang, L.; Wang, Q.; Shi, L. Effect of elemental nano-selenium on feed digestibility, rumen fermentation, and purine derivatives in sheep. Fuel Energy Abstr. 2011, 163, 136–142. [Google Scholar] [CrossRef]
- Kojouri, G.; Arbabi, F.; Mohebbi, A. The effects of selenium nanoparticles (SeNPs) on oxidant and antioxidant activities and neonatal lamb weight gain pattern. Comp. Clin. Pathol. 2020, 29, 369–374. [Google Scholar] [CrossRef]
- Sadeghian, S.; Kojouri, G.A.; Mohebbi, A. Nanoparticles of selenium as species with stronger physiological effects in sheep in comparison with sodium selenite. Biol. Trace Elem. Res. 2012, 146, 302–308. [Google Scholar] [CrossRef]
- Xun, W.; Shi, L.; Yue, W.; Zhang, C.; Ren, Y.; Qiang, L. Effect of High-Dose Nano-selenium and Selenium–Yeast on Feed Digestibility, Rumen Fermentation, and Purine Derivatives in Sheep. Biol. Trace Elem. Res. 2012, 150, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Yao, J.; Yang, Z.; Yue, W.; Ren, Y.; Zhang, C.; Liu, X.; Wang, H.; Zhao, X.; Yuan, S.; et al. Improved fetal hair follicle development by maternal supplement of selenium at nano size (Nano-Se). Livest. Sci. 2011, 142, 270–275. [Google Scholar] [CrossRef]
- Gangadoo, S.; Dinev, I.; Chapman, J.; Hughes, R.J.; Van, T.T.H.; Moore, R.J.; Stanley, D. Selenium nanoparticles in poultry feed modify gut microbiota and increase abundance of Faecalibacterium prausnitzii. Appl. Microbiol. Biotechnol. 2018, 102, 1455–1466. [Google Scholar] [CrossRef]
- Wang, Y. Differential effects of sodium selenite and nano-Se on growth performance, tissue se distribution, and glutathione peroxidase activity of avian broiler. Biol. Trace Elem. Res. 2009, 128, 184–190. [Google Scholar] [CrossRef]
- Gulyas, G.; Csosz, E.; Prokisch, J.; Javor, A.; Mezes, M.; Erdelyi, M.; Balogh, K.; Janaky, T.; Szabo, Z.; Simon, A.; et al. Effect of nano-sized, elemental selenium supplement on the proteome of chicken liver. J. Anim. Physiol. Anim. Nutr. (Berl.) 2017, 101, 502–510. [Google Scholar] [CrossRef]
- Kohshahi, A.J.; Sourinejad, I.; Sarkheil, M.; Johari, S.A. Dietary cosupplementation with curcumin and different selenium sources (nanoparticulate, organic, and inorganic selenium): Influence on growth performance, body composition, immune responses, and glutathione peroxidase activity of rainbow trout (Oncorhynchus mykiss). Fish Physiol. Biochem. 2019, 45, 793–804. [Google Scholar]
- Jahanbakhshi, A.; Pourmozaffar, S.; Adeshina, I.; Mahmoudi, R.; Erfanifar, E.; Ajdari, A. Selenium nanoparticle and selenomethionine as feed additives: Effects on growth performance, hepatic enzymes’ activity, mucosal immune parameters, liver histology, and appetite-related gene transcript in goldfish (Carassius auratus). Fish Physiol. Biochem. 2021, 47, 639–652. [Google Scholar] [CrossRef]
- Liu, G.; Yu, H.; Wang, C.; Li, P.; Liu, S.; Zhang, X.; Zhang, C.; Qi, M.; Ji, H. Nano-selenium supplements in high-fat diets relieve hepatopancreas injury and improve survival of grass carp Ctenopharyngodon Idella by reducing lipid deposition. Aquaculture 2021, 538, 736580. [Google Scholar] [CrossRef]
- Dawood, M.A. Nutritional immunity of fish intestines: Important insights for sustainable aquaculture. Rev. Aquac. 2021, 13, 642–663. [Google Scholar] [CrossRef]
- Ghazi, S.; Diab, A.M.; Khalafalla, M.M.; Mohamed, R.A. Synergistic Effects of Selenium and Zinc Oxide Nanoparticles on Growth Performance, Hemato-biochemical Profile, Immune and Oxidative Stress Responses, and Intestinal Morphometry of Nile Tilapia (Oreochromis niloticus). Biol. Trace Elem. Res. 2021, 200, 364–374. [Google Scholar] [CrossRef]
- Longbaf Dezfouli, M.; Ghaedtaheri, A.; Keyvanshokooh, S.; Salati, A.P.; Mousavi, S.M.; Pasha-Zanoosi, H. Combined or individual effects of dietary magnesium and selenium nanoparticles on growth performance, immunity, blood biochemistry and antioxidant status of Asian seabass (Lates calcarifer) reared in freshwater. Aquac. Nutr. 2019, 25, 1422–1430. [Google Scholar] [CrossRef]
- Zhang, T.; Yao, C.; Hu, Z.; Li, D.; Tang, R. Protective Effect of Selenium on the Oxidative Damage of Kidney Cells Induced by Sodium Nitrite in Grass Carp (Ctenopharyngodon idellus). Biol. Trace Elem. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.S.; El-gendy, G.M.; Ahmed, A.I.; Elharoun, E.R.; Hassaan, M.S. Nanoselenium versus bulk selenium as a dietary supplement: Effects on growth, feed efficiency, intestinal histology, haemato-biochemical and oxidative stress biomarkers in Nile tilapia (Oreochromis niloticus Linnaeus, 1758) fingerlings. Aquac. Res. 2021, 52, 5642–5655. [Google Scholar] [CrossRef]
- Dawood, M.A.O.; Basuini, M.F.E.; Yilmaz, S.; Abdel-Latif, H.M.R.; Kari, Z.A.; Abdul Razab, M.K.A.; Ahmed, H.A.; Alagawany, M.; Gewaily, M.S. Selenium Nanoparticles as a Natural Antioxidant and Metabolic Regulator in Aquaculture: A Review. Antioxidants 2021, 10, 1364. [Google Scholar] [CrossRef] [PubMed]
- Yaghmaie, P.; Ramin, A.; Asri-Rezaei, S.; Zamani, A. Evaluation of glutathion peroxidase activity, trace minerals and weight gain following administration of selenium compounds in lambs. Vet. Res. Forum 2017, 8, 133–137. [Google Scholar]
- Urbankova, L.; Skalickova, S.; Pribilova, M.; Ridoskova, A.; Pelcova, P.; Skladanka, J.; Horky, P. Effects of Sub-Lethal Doses of Selenium Nanoparticles on the Health Status of Rats. Toxics 2021, 9, 28. [Google Scholar] [CrossRef] [PubMed]
- Benko, I.; Nagy, G.; Tanczos, B.; Ungvari, E.; Sztrik, A.; Eszenyi, P.; Prokisch, J.; Banfalvi, G. Subacute toxicity of nano-selenium compared to other selenium species in mice. Environ. Toxicol. Chem. 2012, 31, 2812–2820. [Google Scholar] [CrossRef]
- He, Y.; Chen, S.; Liu, Z.; Cheng, C.; Li, H.; Wang, M. Toxicity of selenium nanoparticles in male Sprague-Dawley rats at supranutritional and nonlethal levels. Life Sci. 2014, 115, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; He, Y.; Xiao, Z.; Tao, W.; Zhu, J.; Wang, B.; Liu, Z.; Wang, M. Effects of Selenium Nanoparticles on Reproductive Performance of Male Sprague-Dawley Rats at Supranutritional and Nonlethal Levels. Biol. Trace Elem. Res. 2017, 180, 81–89. [Google Scholar] [CrossRef]
- Jin, Y.; He, Y.; Liu, L.; Tao, W.; Wang, G.; Sun, W.; Pei, X.; Xiao, Z.; Wang, H.; Wang, M. Effects of Supranutritional Selenium Nanoparticles on Immune and Antioxidant Capacity in Sprague-Dawley Rats. Biol. Trace Elem. Res. 2021, 199, 4666–4674. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; He, Y.; Liu, L.; Tao, W.; Wang, G.; Sun, W.; Pei, X.; Xiao, Z.; Jin, Y.; Wang, M. Prooxidation and Cytotoxicity of Selenium Nanoparticles at Nonlethal Level in Sprague-Dawley Rats and Buffalo Rat Liver Cells. Oxid. Med. Cell. Longev. 2020, 2020, 7680276. [Google Scholar] [CrossRef]
- Hadrup, N.; Loeschner, K.; Mandrup, K.; Ravn-Haren, G.; Frandsen, H.L.; Larsen, E.H.; Lam, H.R.; Mortensen, A. Subacute oral toxicity investigation of selenium nanoparticles and selenite in rats. Drug Chem. Toxicol. 2019, 42, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Qamar, N.; John, P.; Bhatti, A. Toxicological and Anti-Rheumatic Potential of Trachyspermum ammi Derived Biogenic Selenium Nanoparticles in Arthritic Balb/c Mice. Int. J. Nanomed. 2020, 15, 3497–3509. [Google Scholar] [CrossRef]
- Qiao, L.; Dou, X.; Yan, S.; Zhang, B.; Xu, C. Biogenic selenium nanoparticles by Lactobacillus casei ATCC 393 alleviate diquat-induced intestinal barrier dysfunction in C57BL/6 mice through their antioxidant activity. Food Funct. 2020, 11, 3020–3031. [Google Scholar] [CrossRef]
- Song, X.; Qiao, L.; Yan, S.; Chen, Y.; Dou, X.; Xu, C. Preparation, characterization, and in vivo evaluation of anti-inflammatory activities of selenium nanoparticles synthesized by Kluyveromyces lactis GG799. Food Funct. 2021, 12, 6403–6415. [Google Scholar] [CrossRef]
- Cai, S.J.; Wu, C.X.; Gong, L.M.; Song, T.; Wu, H.; Zhang, L.Y. Effects of nano-selenium on performance, meat quality, immune function, oxidation resistance, and tissue selenium content in broilers. Poult. Sci. 2012, 91, 2532–2539. [Google Scholar] [CrossRef]
- El-Deep, M.; Ijiri, D.; Ebeid, T.; Ohtsuka, A. Effects of Dietary Nano-Selenium Supplementation on Growth Performance, Antioxidative Status, and Immunity in Broiler Chickens under Thermoneutral and High Ambient Temperature Conditions. J. Poult. Sci. 2016, 53. [Google Scholar] [CrossRef]
- Ahmadi, M.; Ahmadian, A.; Seidavi, A. Effect of Different Levels of Nano-selenium on Performance, Blood Parameters, Immunity and Carcass Characteristics of BroilerChickens. Poult. Sci. J. 2018, 6, 99–108. [Google Scholar] [CrossRef]
- Bakhshalinejad, R.; Akbari Moghaddam Kakhki, R.; Zoidis, E. Effects of different dietary sources and levels of selenium supplements on growth performance, antioxidant status and immune parameters in Ross 308 broiler chickens. Br. Poult. Sci. 2018, 59, 81–91. [Google Scholar] [CrossRef]
- Ibrahim, D.; Kishawy, A.T.Y.; Khater, S.I.; Hamed Arisha, A.; Mohammed, H.A.; Abdelaziz, A.S.; Abd El-Rahman, G.I.; Elabbasy, M.T. Effect of Dietary Modulation of Selenium Form and Level on Performance, Tissue Retention, Quality of Frozen Stored Meat and Gene Expression of Antioxidant Status in Ross Broiler Chickens. Animals 2019, 9, 342. [Google Scholar] [CrossRef]
- Shabani, R.; Fakhraei, J.; Yarahmadi, H.; Seidavi, A. Effect of different sources of selenium on performance and characteristics of immune system of broiler chickens. Rev. Bras. Zootec. 2019, 48. [Google Scholar] [CrossRef]
- Popescu, M.; Velea, A.; Lőrinczi, A. Biogenic production of nanoparticles. Dig. J. Nanomater. Biostruct. 2010, 5, 1035–1040. [Google Scholar]
- Wadhwani, S.A.; Shedbalkar, U.U.; Singh, R.; Chopade, B.A. Biogenic selenium nanoparticles: Current status and future prospects. Appl. Microbiol. Biotechnol. 2016, 100, 2555–2566. [Google Scholar] [CrossRef]
- Kumar, N.; Krishnani, K.K.; Singh, N.P. Comparative study of selenium and selenium nanoparticles with reference to acute toxicity, biochemical attributes, and histopathological response in fish. Environ. Sci. Pollut. Res. 2018, 25, 8914–8927. [Google Scholar] [CrossRef]
- Li, H.; Zhang, J.; Wang, T.; Luo, W.; Zhou, Q.; Jiang, G. Elemental selenium particles at nano-size (Nano-Se) are more toxic to Medaka (Oryzias latipes) as a consequence of hyper-accumulation of selenium: A comparison with sodium selenite. Aquat. Toxicol. 2008, 89, 251–256. [Google Scholar] [CrossRef]
- Hassanin, K.M.; Abd El-Kawi, S.H.; Hashem, K.S. The prospective protective effect of selenium nanoparticles against chromium-induced oxidative and cellular damage in rat thyroid. Int. J. Nanomed. 2013, 8, 1713–1720. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, Y.; Gu, Q.; Li, W. Effect of different dietary selenium source (selenium nanoparticle and selenomethionine) on growth performance, muscle composition and glutathione peroxidase enzyme activity of crucian carp (Carassius auratus gibelio). Aquaculture 2009, 291, 78–81. [Google Scholar] [CrossRef]
- Sharma, D.; Kanchi, S.; Bisetty, K. Biogenic synthesis of nanoparticles: A review. Arab. J. Chem. 2019, 12, 3576–3600. [Google Scholar] [CrossRef]
- Duan, H.; Wang, D.; Li, Y. Green Chemistry for Nanoparticle Synthesis. Chem. Soc. Rev. 2015, 44, 5778–5792. [Google Scholar] [CrossRef] [PubMed]
- Al-khattaf, F.S. Gold and silver nanoparticles: Green synthesis, microbes, mechanism, factors, plant disease management and environmental risks. Saudi J. Biol. Sci. 2021, 28, 3624–3631. [Google Scholar] [CrossRef] [PubMed]
- Javed, R.; Zia, M.; Naz, S.; Aisida, S.O.; Ain, N.u.; Ao, Q. Role of capping agents in the application of nanoparticles in biomedicine and environmental remediation: Recent trends and future prospects. J. Nanobiotechnol. 2020, 18, 172. [Google Scholar] [CrossRef]
- Iravani, S.; Korbekandi, H.; Mirmohammadi, S.V.; Zolfaghari, B. Synthesis of silver nanoparticles: Chemical, physical and biological methods. Res. Pharm. Sci. 2014, 9, 385–406. [Google Scholar]
- Bhattarai, B.; Zaker, Y.; Bigioni, T.P. Green synthesis of gold and silver nanoparticles: Challenges and opportunities. Curr. Opin. Green Sustain. Chem. 2018, 12, 91–100. [Google Scholar] [CrossRef]
- Cardoso-Avila, P.E.; Patakfalvi, R.; Rodríguez-Pedroza, C.; Aparicio-Fernández, X.; Loza-Cornejo, S.; Villa-Cruz, V.; Martínez-Cano, E. One-pot green synthesis of gold and silver nanoparticles using Rosa canina L. extract. RSC Adv. 2021, 11, 14624–14631. [Google Scholar] [CrossRef]
- Hosny, M.; Fawzy, M.; Abdelfatah, A.M.; Fawzy, E.E.; Eltaweil, A.S. Comparative study on the potentialities of two halophytic species in the green synthesis of gold nanoparticles and their anticancer, antioxidant and catalytic efficiencies. Adv. Powder Technol. 2021, 32, 3220–3233. [Google Scholar] [CrossRef]
- Muthusamy, A.; Pottail, L. Rapid Green Synthesis of Gold and Silver Nanoparticles Using Ethanol Extract of Kedrostis Foetidissima (JACQ) COGN. And Its Anticancer Efficacy against A549 Human Lung Cancer Cell Lines. Indian Drugs 2021, 58, 30–40. Available online: http://www.indiandrugsonline.org/issuesarticle-details?id=MTE1OQ (accessed on 3 November 2021).
- Anu, K.; Singaravelu, G.; Murugan, K.; Benelli, G. Green-Synthesis of Selenium Nanoparticles Using Garlic Cloves (Allium sativum): Biophysical Characterization and Cytotoxicity on Vero Cells. J. Clust. Sci. 2017, 28, 551–563. [Google Scholar] [CrossRef]
- Kokila, K.; Elavarasan, N.; Sujatha, V. Diospyros montana leaf extract-mediated synthesis of selenium nanoparticles and their biological applications. New J. Chem. 2017, 41, 7481–7490. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, J.; Ding, D.; Zhang, L.; Muehlmann, L.A.; Deng, S.-e.; Wang, X.; Li, W.; Zhang, W. Synthesis and antioxidant properties of Lycium barbarum polysaccharides capped selenium nanoparticles using tea extract. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1463–1470. [Google Scholar] [CrossRef]
- Salem, S.S.; Fouda, M.M.; Fouda, A.; Awad, M.A.; Al-Olayan, E.M.; Allam, A.A.; Shaheen, T.I. Antibacterial, cytotoxicity and larvicidal activity of green synthesized selenium nanoparticles using Penicillium corylophilum. J. Clust. Sci. 2021, 32, 351–361. [Google Scholar] [CrossRef]
- Regulation (EC) No 1831/2003 of the European Parliament and of the Council of 22 September 2003 on Additives for Use in Animal Nutrition. 2003. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32003R1831 (accessed on 3 November 2021).
- Ramesh, M.; Anbuvannan, M.; Viruthagiri, G. Green synthesis of ZnO nanoparticles using Solanum nigrum leaf extract and their antibacterial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 136, 864–870. [Google Scholar] [CrossRef]
- Agarwal, H.; Venkat Kumar, S.; Rajeshkumar, S. A review on green synthesis of zinc oxide nanoparticles—An eco-friendly approach. Resour.-Effic. Technol. 2017, 3, 406–413. [Google Scholar] [CrossRef]
- Horky, P.; Skalickova, S.; Urbankova, L.; Baholet, D.; Kociova, S.; Bytesnikova, Z.; Kabourkova, E.; Lackova, Z.; Cernei, N.; Gagic, M. Zinc phosphate-based nanoparticles as a novel antibacterial agent: In vivo study on rats after dietary exposure. J. Anim. Sci. Biotechnol. 2019, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Cremonini, E.; Zonaro, E.; Donini, M.; Lampis, S.; Boaretti, M.; Dusi, S.; Melotti, P.; Lleo, M.M.; Vallini, G. Biogenic selenium nanoparticles: Characterization, antimicrobial activity and effects on human dendritic cells and fibroblasts. Microb. Biotechnol. 2016, 9, 758–771. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Awasthee, N.; Shekher, A.; Rai, L.C.; Gupta, S.C.; Dubey, S.K. Biogenic synthesis and characterization of selenium nanoparticles and their applications with special reference to antibacterial, antioxidant, anticancer and photocatalytic activity. Bioprocess Biosyst. Eng. 2021, 44, 2679–2696. [Google Scholar] [CrossRef]
- Yan, S.; Qiao, L.; Dou, X.; fan Song, X.; Chen, Y.; Zhang, B.; Xu, C. Biogenic Selenium nanoparticles by Lactobacillus casei ATCC 393 alleviate the intestinal permeability, mitochondrial dysfunction and mitophagy induced by oxidative stress. Food Funct. 2021. [Google Scholar] [CrossRef]
- Alvi, G.B.; Iqbal, M.S.; Ghaith, M.M.S.; Haseeb, A.; Ahmed, B.; Qadir, M.I. Biogenic selenium nanoparticles (SeNPs) from citrus fruit have anti-bacterial activities. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Kanchi, S.; Khan, A. Biogenic Synthesis of Selenium Nanoparticles with Edible Mushroom Extract: Evaluation of Cytotoxicity on Prostate Cancer Cell Lines and Their Antioxidant, and Antibacterial Activity. Biointerface Res. Appl. Chem. 2020, 10, 6629–6639. [Google Scholar]
- Vinu, D.; Govindaraju, K.; Vasantharaja, R.; Amreen Nisa, S.; Kannan, M.; Vijai Anand, K. Biogenic zinc oxide, copper oxide and selenium nanoparticles: Preparation, characterization and their anti-bacterial activity against Vibrio parahaemolyticus. J. Nanostruct. Chem. 2021, 11, 271–286. [Google Scholar] [CrossRef]
- Sonkusre, P.; Cameotra, S.S. Biogenic selenium nanoparticles induce ROS-mediated necroptosis in PC-3 cancer cells through TNF activation. J. Nanobiotechnol. 2017, 15, 43. [Google Scholar] [CrossRef]
- Lian, S.; Diko, C.S.; Yan, Y.; Li, Z.; Zhang, H.; Ma, Q.; Qu, Y. Characterization of biogenic selenium nanoparticles derived from cell-free extracts of a novel yeast Magnusiomyces ingens. 3 Biotech 2019, 9, 221. [Google Scholar] [CrossRef]
- Hu, D.; Yu, S.; Yu, D.; Liu, N.; Tang, Y.; Fan, Y.; Wang, C.; Wu, A. Biogenic Trichoderma harzianum-derived selenium nanoparticles with control functionalities originating from diverse recognition metabolites against phytopathogens and mycotoxins. Food Control 2019, 106, 106748. [Google Scholar] [CrossRef]
- Menon, S.; Devi, S.; Agarwal, H.; Kumar, V. Efficacy of Biogenic Selenium Nanoparticles from an Extract of Ginger towards Evaluation on Anti-Microbial and Anti-Oxidant Activities. Colloid Interface Sci. Commun. 2019, 29, 1–8. [Google Scholar] [CrossRef]
- Cittrarasu, V.; Kaliannan, D.; Dharman, K.; Maluventhen, V.; Easwaran, M.; Liu, W.C.; Balasubramanian, B.; Arumugam, M. Green synthesis of selenium nanoparticles mediated from Ceropegia bulbosa Roxb extract and its cytotoxicity, antimicrobial, mosquitocidal and photocatalytic activities. Sci. Rep. 2021, 11, 1032. [Google Scholar] [CrossRef]
- Abbas, H.S.; Abou Baker, D.H.; Ahmed, E.A. Cytotoxicity and antimicrobial efficiency of selenium nanoparticles biosynthesized by Spirulina platensis. Arch. Microbiol. 2021, 203, 523–532. [Google Scholar] [CrossRef]
- Medina Cruz, D.; Mi, G.; Webster, T.J. Synthesis and characterization of biogenic selenium nanoparticles with antimicrobial properties made by Staphylococcus aureus, methicillin-resistant Staphylococcus aureus (MRSA), Escherichia coli, and Pseudomonas aeruginosa. J. Biomed. Mater. Res. Part A 2018, 106, 1400–1412. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Holden, J.A.; Reynolds, E.C.; Heath, D.E.; O’Brien-Simpson, N.M.; O’Connor, A.J. Multifunctional Antimicrobial Polypeptide-Selenium Nanoparticles Combat Drug-Resistant Bacteria. ACS Appl. Mater. Interfaces 2020, 12, 55696–55709. [Google Scholar] [CrossRef]
- Zheng, S.; Su, J.; Wang, L.; Yao, R.; Wang, D.; Deng, Y.; Wang, R.; Wang, G.; Rensing, C. Selenite reduction by the obligate aerobic bacterium Comamonas testosteroni S44 isolated from a metal-contaminated soil. BMC Microbiol. 2014, 14, 204. [Google Scholar] [CrossRef]
- Ovais, M.; Khalil, A.T.; Ayaz, M.; Ahmad, I.; Nethi, S.K.; Mukherjee, S. Biosynthesis of Metal Nanoparticles via Microbial Enzymes: A Mechanistic Approach. Int. J. Mol. Sci. 2018, 19, 4100. [Google Scholar] [CrossRef]
- Mellinas, C.; Jiménez, A.; Garrigós, M.D.C. Microwave-Assisted Green Synthesis and Antioxidant Activity of Selenium Nanoparticles Using Theobroma cacao L. Bean Shell Extract. Molecules 2019, 24, 4048. [Google Scholar] [CrossRef] [PubMed]
- Fardsadegh, B.; Jafarizadeh, H. Aloe vera leaf extract mediated green synthesis of selenium nanoparticles and assessment of their In vitro antimicrobial activity against spoilage fungi and pathogenic bacteria strains. Green Process. Synth. 2019, 8, 399–407. [Google Scholar] [CrossRef]
- Singh, R.; Sadasivam, M.; Rakkiyappan, C. Ginger (Zingiber officinale) root extract: A source of silver NANOPARTICLES and their application. Int. J. Bio-Eng. Sci. Technol. 2021, 2, 75–80. [Google Scholar]
- Pyrzynska, K.; Sentkowska, A. Biosynthesis of selenium nanoparticles using plant extracts. J. Nanostruct. Chem. 2021. [Google Scholar] [CrossRef]
- Singh, R.; Shedbalkar, U.U.; Wadhwani, S.A.; Chopade, B.A. Bacteriagenic silver nanoparticles: Synthesis, mechanism, and applications. Appl. Microbiol. Biotechnol. 2015, 99, 4579–4593. [Google Scholar] [CrossRef]
- Yazhiniprabha, M.; Vaseeharan, B. In vitro and in vivo toxicity assessment of selenium nanoparticles with significant larvicidal and bacteriostatic properties. Mater. Sci. Eng. C 2019, 103, 109763. [Google Scholar] [CrossRef]
- Pandiyan, I.; Sakthi, S.D.; Indiran, M.A.; Rathinavelu, P.K.; Rajeshkumar, S. Mediated Selenium Nanoparticles, Characterization and its Antimicrobial Activity-An In Vitro Study. Thymus Vulgaris 2021, 7, 3516–3521. [Google Scholar]
- Eswayah, A.S.; Smith, T.J.; Gardiner, P.H. Microbial Transformations of Selenium Species of Relevance to Bioremediation. Appl. Environ. Microbiol. 2016, 82, 4848–4859. [Google Scholar] [CrossRef]
- Xu, X.; Cheng, W.; Liu, X.; You, H.; Wu, G.; Ding, K.; Tu, X.; Yang, L.; Wang, Y.; Li, Y.; et al. Selenate Reduction and Selenium Enrichment of Tea by the Endophytic Herbaspirillum sp. Strain WT00C. Curr. Microbiol. 2020, 77, 588–601. [Google Scholar] [CrossRef] [PubMed]
- Afzal, B.; Fatma, T. Selenium Nanoparticles: Green Synthesis and Exploitation. In Merging Technologies for Nanoparticle Manufacturing; Patel, J.K., Pathak, Y.V., Eds.; Springer: Cham, Switzerland, 2021. [Google Scholar]
- El-Saadony, M.T.; Saad, A.M.; Najjar, A.A.; Alzahrani, S.O.; Alkhatib, F.M.; Shafi, M.E.; Selem, E.; Desoky, E.M.; Fouda, S.E.E.; El-Tahan, A.M.; et al. The use of biological selenium nanoparticles to suppress Triticum aestivum L. crown and root rot diseases induced by Fusarium species and improve yield under drought and heat stress. Saudi J. Biol. Sci. 2021, 28, 4461–4471. [Google Scholar] [CrossRef] [PubMed]
- Sasidharan, S.; Balakrishnaraja, R. Comparison Studies on the Synthesis of Selenium Nanoparticles by Various Micro-organisms. Int. J. Pure Appl. Biosci. 2014, 2, 112–117. [Google Scholar]
- Afzal, B.; Yasin, D.; Husain, S.; Zaki, A.; Srivastava, P.; Kumar, R.; Fatma, T. Screening of cyanobacterial strains for the selenium nanoparticles synthesis and their anti-oxidant activity. Biocatal. Agric. Biotechnol. 2019, 21, 101307. [Google Scholar] [CrossRef]
- Hariharan, H.; Al-Harbi, N.; Karuppiah, P.; Rajaram, S.K. Microbial synthesis of selinium nanocomposite using Saccharomyces cerevisiae and its antimicrobial activity against pathogens causing nosocomial infection. Chalcogenide Lett. 2012, 9, 509–515. [Google Scholar]
- Shirsat, S.; Kadam, A.; Mane, R.; Jadhav, V.; Zate, M.; Highly Cited Researcher, M.; Kim, K. Protective Role of Biogenic Selenium Nanoparticles in Immunological and Oxidative Stress Generated by Enrofloxacin in Broiler Chicken. Dalton Trans. 2016, 45. [Google Scholar] [CrossRef]
- Hadrová, S.; Sedláková, K.; Krizova, L.; Malyugina, S. Alternative and Unconventional Feeds in Dairy Diets and Their Effect on Fatty Acid Profile and Health Properties of Milk Fat. Animals 2021, 11, 1817. [Google Scholar] [CrossRef]
- Singaravelu, G.; Arockiamary, J.; Kumar, G.; Govindaraju, K. A novel extracellular synthesis of monodisperse gold nanoparticles using marine alga, Sargassum wightii Greville. Colloids Surf. B Biointerfaces 2007, 57, 97–101. [Google Scholar] [CrossRef]
- Luangpipat, T.; Beattie, I.; Chisti, Y.; Haverkamp, R. Gold nanoparticles produced in a microalga. J. Nanopart. Res. 2011, 13, 6439–6445. [Google Scholar] [CrossRef]
- Senapati, S.; Syed, A.; Moeez, S.; Kumar, A.; Ahmad, A. Intracellular synthesis of gold nanoparticles using alga Tetraselmis kochinensis. Mater. Lett. 2012, 79, 116–118. [Google Scholar] [CrossRef]
- Castro, L.; Blázquez, M.L.; Muñoz, J.A.; González, F.; Ballester, A. Biological synthesis of metallic nanoparticles using algae. IET Nanobiotechnol. 2013, 7, 109–116. [Google Scholar] [CrossRef]
- ElSaied, B.; Diab, A.; Tayel, A.; Alghuthaymi, M.; Moussa, S. Potent antibacterial action of phycosynthesized selenium nanoparticles using Spirulina platensis extract. Green Process. Synth. 2021, 10, 49–60. [Google Scholar] [CrossRef]
- Mansouri-Tehrani, H.A.; Keyhanfar, M.; Behbahani, M.; Dini, G. Synthesis and characterization of algae-coated selenium nanoparticles as a novel antibacterial agent against Vibrio harveyi, a Penaeus vannamei pathogen. Aquaculture 2021, 534, 736260. [Google Scholar] [CrossRef]
- Venkatesan, J.; Anil, S.; Kim, S.-K.; Shim, M.S. Seaweed polysaccharide-based nanoparticles: Preparation and applications for drug delivery. Polymers 2016, 8, 30. [Google Scholar] [CrossRef]
- Chaudhary, R.; Nawaz, K.; Khan, A.K.; Hano, C.; Abbasi, B.H.; Anjum, S. An Overview of the Algae-Mediated Biosynthesis of Nanoparticles and Their Biomedical Applications. Biomolecules 2020, 10, 1498. [Google Scholar] [CrossRef]
| Animal Species | Selenium Daily Nutritional Requirement (μg/kg DM) | Se Dose (mg/kg Body Weight-BW) Inducing Acute Toxicity | Reference |
|---|---|---|---|
| Dairy cattle | 300 | 3.0 | [34] |
| Beef cattle | 100 | 3.0 | [34] |
| Sheep, goat | 100–300 | 0.5 | [40,41] |
| Swine | 150–300 | 1.2 | [37] |
| Horse | 100–200 | 3.0 | [42] |
| Selenium Sources | Description | Reference |
|---|---|---|
| Inorganic |
| [43] |
| Sodium selenite/selenate | Inorganic Se salts are frequently used as feed supplements because they are cost effective and relatively nontoxic; they are most commonly supplemented in the form of injections or mineral-salt complexes. However, they were reported to have lower bioavailability and lower transfer to animal products compared to organic Se and SeNPs. | |
| Organic |
| [44,45] |
| Selenomethionine (SeMet); Selenocysteine (SeCys) | SeMet is often supplemented in the form of SYs. Organic Se sources have shown better bioavailability and improved Se storage in animal tissues compared to inorganic. | |
| Selenium nanoforms |
| [46,47,48] |
| SeNPs prepared using a chemical/physical method; biogenic SeNPs prepared via green synthesis | Chemically synthesized SeNPs are less toxic to animals than inorganic and organic Se sources, but they are not environmentally friendly due to the toxic chemicals produced during the NPs’ preparation. Biogenic NPs are more stable, more eco-friendly, and less toxic. |
| Experimental Animals | Element | Dose | Toxicity | Major Effect | Reference |
|---|---|---|---|---|---|
| Dorset sheep | SeNPs | 0.3, 3, and 6 mg/kg DM fed for 75 days | No information | Improved Se content in blood and tissues and enhanced ruminal fermentation and feed utilization | [118] |
| Small tail Han sheep | SeNPs | 0.3, 3, and 6 mg/kg DM fed for 75 days | No information | Improved Se content in blood and tissues and enhanced ruminal fermentation and feed utilization | [118] |
| Tan sheep | SeNPs | 0.3, 3, and 6 mg/kg DM fed for 75 days | No information | Improved Se content in blood and tissues and enhanced ruminal fermentation and feed utilization | [118] |
| Neonatal lambs | Sans | 0.1 mg/kg DM fed for 60 days | No information | Improved weight gain on the 14th and 28th day; enhanced antioxidant parameters | [119] |
| Lori–Bakhtiari sheep | SeNPs | 1 mg/kg DM diet for 10 days | SeNPs were found to be less toxic than SS | Improved antioxidant parameters compared to the experimental group fed with sodium selenite (SS) | [120] |
| Sheep | SeNPs | 4 mg/kg DM | No information | Enhanced rumen fermentation and feed conversion efficiency compared with the group fed with 4 mg/kg DM selenized yeast (SY) | [121] |
| Cashmere goats | SeNPs | 0.5 mg/kg DM | No information | Improved the hair follicle development and promoted growth | [122] |
| Khalkhali goats | SeNPs | 0.5 mg per animal per day | No information | SeNP inclusion improved Se status in goats (increased Se in blood, colostrum, and milk) compared to SS and SeMet | [82] |
| Makuei sheep | SeNPs | 0.1 mg/kg of live weight | No information | Reduced oxidative stress and enhanced weight gain compared to the group supplemented with SS | [135] |
| Male rats | SeNPs | 0.5, 1.5, 3.0, and 5.0 mg Se/kg for 28 days | Damage of the liver parenchyma and intestinal epithelium in animal groups fed with 1.5, 3, and 5 mg Se/kg | Increased Se content in the blood compared to the control group | [136] |
| Mice | SeNPs | 4, 40, and 400 μg/kg of bw | Dietary SeNPs showed less toxicity compared to inorganic SS and sodium hydroselenite but was more toxic than SY; subacute toxicity was observed with administration of 400 μg/kg bw | Inorganic Se forms showed higher toxicity in comparison with SeNPs and organic SY | [137] |
| Swiss albino mice | SeNPs | 2 mg/kg of bw was administrated | Organic and inorganic Se forms showed less toxicity at the same dosage of 2 mg/kg of bw | SeNPs improved antioxidant protection of cells compared to inorganic forms | [47] |
| Male rats | SeNPs | 0.0, 0.2, 0.4, 0.8, 2.0, 4.0, or 8.0 mg Se/kg of bw were administrated for 14 consecutive days | Doses greater than 2.0 mg Se/kg of bw induced chronic toxicity | Supranutritional levels (0.2, 0.4, and 0.8) of SeNPs did not show toxic effect | [138] |
| Male Sprague–Dawley (SD) rats | SeNPs | 0.0, 0.2, 0.4, 0.8, 2.0, 4.0, or 8.0 mg Se/kg of bw were administrated for 2 weeks | Doses greater than 4.0 mg Se/kg of bw induced chronic toxicity, damaging effect | Supranutritional levels (0.2, 0.4, and 0.8) of SeNPs had a positive effect on reproductive function (promoted sperm motility) | [139] |
| Male SD rats | SeNPs | 0.0, 0.2, 0.4, or 0.8 mg Se/kg of bw were administrated for 2 weeks | No toxic effect was observed | Improved antioxidant capacity in the liver and kidney; beneficial effects on immune and antioxidant capacity (dose of 0.4 mg had the best response) | [140] |
| Male SD rats, Buffalo rats | SeNPs | 0.0, 2, 4, or 8 mg Se/kg of bw were administrated for 2 weeks | SeNP administration over 4.0 mg Se/kg bw caused a toxic effect on liver | Impaired the antioxidant capacity in serum with the administration of non-lethal doses | [141] |
| Female and male rats | SeNPs (20 nm) | 0.05, 0.5, or 4 mg Se/kg bw/day for 28 days for female rats; 4 mg Se/kg bw/day for male rats | Nanoparticle-specific toxicity of Se did not occur; no histological changes in the liver occurred | Lowered body weight at all doses of SeNPs | [142] |
| Male rats | SeNPs | 0.5, 1.5, 3.0, and 5.0 mg Se/kg were administered for 28 days | Tested doses did not have a significant toxic effect on liver, kidney, or spleen | Enhanced Se content in blood compared to the control group | [143] |
| Mice | Biogenic SeNPs 50–80 nm synthesized using Lactobacillus casei | - | No toxic effect occurred | Protected the intestinal barrier function against oxidative damage | [144] |
| Male mice | Biogenic SeNps 80–220 nm synthesized using Bacillus sp. MSh-1 | oral administration of 0, 2.5, 5 10, and 20 mg kg−1 of Se NPs for 14 consecutive days | A dose of 20 mg/kg showed toxicity | Less toxic effect compared to synthetic SeNPs | [107] |
| Male mice | Biogenic SeNPs produced using yeast strain Kluyveromyces lactis GG799 | 0.2, 0.6, and 6 mg/kg | No toxic effect occurred | Attenuation of oxidative stress, intestinal inflammation, and intestinal barrier disfunction | [145] |
| Ross 308 broiler male chicken | SeNPs | 0.3, 0.9, and 1.5 mg Se/kg were fed for 28 days | No toxic effect in tissues occurred | Improved the gut microflora environment; the best performance demonstrated a 0.9 mg/kg Se concentration | [123] |
| Broiler male chicken | SeNPs | 0.3, 0.9, and 1.5 mg Se/kg were fed for 28 days | No toxic effect in tissues occurred; no damaging effect on intestinal morphology | Dietary SeNP inclusion showed comparable results with organic Se at the best dose of 0.9 mg/kg (improved absorption in the duodenum) | [48] |
| male Arbor Acres broilers | SeNPs | 0.0, 0.3, 0.5, 1.0, or 2.0 mg/kg of diet | No toxic effect occurred | 0.3–0.5 mg/kg optimum doses improved meat quality, immune function, and antioxidant status. Liver and muscle Se contents increased with SeNP supplementation | [146] |
| Broiler chicken | SeNPs | 0.3 mg/kg of diet | No toxic effect occurred | Enhanced Se and vitamin E concentrations in breast muscles, improvement of antioxidant and immune properties | [147] |
| Broiler chicken | SeNPs | 4.25 mg/kg DM | No toxic effect occurred | Changes in proteome profile indicated dietary stress from SeNPs supplementation | [125] |
| Ross 308 broiler chicken | SeNPs | 0.1, 0.2, 0.3, 0.4, or 0.5 mg/kg of diet | No toxic effect occurred | Dietary SeNPs improved weight gain, feed conversion ratio, and growth performance | [148] |
| Ross 308 broiler male chicken | SeNPs | 0.1 or 0.4 mg/kg of diet | No toxic effect occurred | Improved antioxidant status, better production performance, and immune system response | [149] |
| Ross broiler chicken | SeNPs | 0.3, 0.45, or 0.6 mg/kg of diet | No toxic effect occurred | Increased body weight gain, improved feed conversion ratio, and meat quality | [150] |
| Broiler chicken | SeNPs | 0.5, 0.8, or 1.2 mg/kg of diet | No toxic effect occurred | SeNPs dietary inclusion improved performance and immune system better than diets included SeMet and vitamin E | [151] |
| Broiler chicken | Biogenic SeNPs produced using the bacteria Pantoea agglomerans | - | No toxic effect occurred | Protective effect against oxidative and immune stress | [152] |
| Biological Organism Used for NPs Synthesis | NPs Characterization (Size, Shape) | Applied Dose | Pathogens/Cells | Effect | Reference |
|---|---|---|---|---|---|
| Cyanobacteria Anabaena sp. | Spherical NPs 5–50 nm | 50 µg/mL | Gram-positive and Gram-negative strains of Staphylococcus aureus and Escherichia coli |
| [176] |
| Lactobacillus casei ATCC 393 | 50–80 nm | 8 μg Se/mL | The porcine jejunal epithelial cell line (IPEC-J2) |
| [177] |
| Citrus fruit (Citrus limon, Citrus paradise) extracts | 100–800 nm | 10 and 12 mM | E. coli, Micrococcus luteus, Bacillus subtilis, and Klebsiellapneumoniae |
| [178] |
| Mushroom extract | 8 nm | 0.5–1.5 μM | Gram-negative E.coli |
| [179] |
| Brown alga Sargassum swartzii | 21 nm | 10 mg/mL | V. parahaemolyticus |
| [180] |
| Bacillus licheniformis | 110 nm | 2 mg/mL | Cancer cells |
| [181] |
| Yeast Magnusiomyces ingens LH-F1 | Spherical; average size 87.82 nm | No information | Gram-positive Arthrobacter sp. W1, Gram-negative E. coli BL21 |
| [182] |
| Trichoderma harzianum JF309 | - | 400 μg/mL | Alternaria toxins, Fusarium verticillioide, and F. graminearum |
| [183] |
| Zingiber officinale | 100–150 nm | 250 μg/mL | E. coli, Klebsiella sp., Pseudomonas sp., Staphylococcus aureus, and Proteus sp. |
| [184] |
| Ceropegia bulbosa tuber’s aqueous extracts extract | 277.5 nm | 25, 50, 75, and 100 μL/mL | E. coli and B. subtilis |
| [185] |
| SeNPs synthesized by the green method (no information about organism used for preparation) | 60 nm | 0.3, 0.4, and 0.5 mg/mL | E. coli and Candida albicans |
| |
| The aqueous filtrate of Spirulina platensis | Average size 79.40 nm, spherical shape | 5 and 10 mM | C. albicans, Klebsiella pneumoniae, and Salmonella abony |
| [186] |
| Escherichia coli, Pseudomonas aeruginosa, Methicillin-resistant Staphylococcus aureus, and S. aureus | 90–150 nm | 25–250 µg/mL | E. coli and S. aureus |
| [187] |
| Gram-negative Stenotrophomonas maltophilia and Gram-positive Bacillus mycoides | 160.6 nm for G+; 170.6 nm for G− | 50, 100, and 250 µg/mL | C. albicans, C. parapsilosis, and Pseudomonas aeruginosa |
| [175] |
| SeNPs coated with the antimicrobial polypeptide (ε-poly-l-lysine) | 80 nm | 5, 10, 25, and 50 µg/mL | Enterococcus faecalis, E. coli, P. aeruginosa, S. aureus, K. pneumoniae, and K. |
| [188] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malyugina, S.; Skalickova, S.; Skladanka, J.; Slama, P.; Horky, P. Biogenic Selenium Nanoparticles in Animal Nutrition: A Review. Agriculture 2021, 11, 1244. https://doi.org/10.3390/agriculture11121244
Malyugina S, Skalickova S, Skladanka J, Slama P, Horky P. Biogenic Selenium Nanoparticles in Animal Nutrition: A Review. Agriculture. 2021; 11(12):1244. https://doi.org/10.3390/agriculture11121244
Chicago/Turabian StyleMalyugina, Svetlana, Sylvie Skalickova, Jiri Skladanka, Petr Slama, and Pavel Horky. 2021. "Biogenic Selenium Nanoparticles in Animal Nutrition: A Review" Agriculture 11, no. 12: 1244. https://doi.org/10.3390/agriculture11121244
APA StyleMalyugina, S., Skalickova, S., Skladanka, J., Slama, P., & Horky, P. (2021). Biogenic Selenium Nanoparticles in Animal Nutrition: A Review. Agriculture, 11(12), 1244. https://doi.org/10.3390/agriculture11121244








