Black Soldier Fly (Hermetia illucens) Meal as a Promising Feed Ingredient for Poultry: A Comprehensive Review
Abstract
:1. Introduction
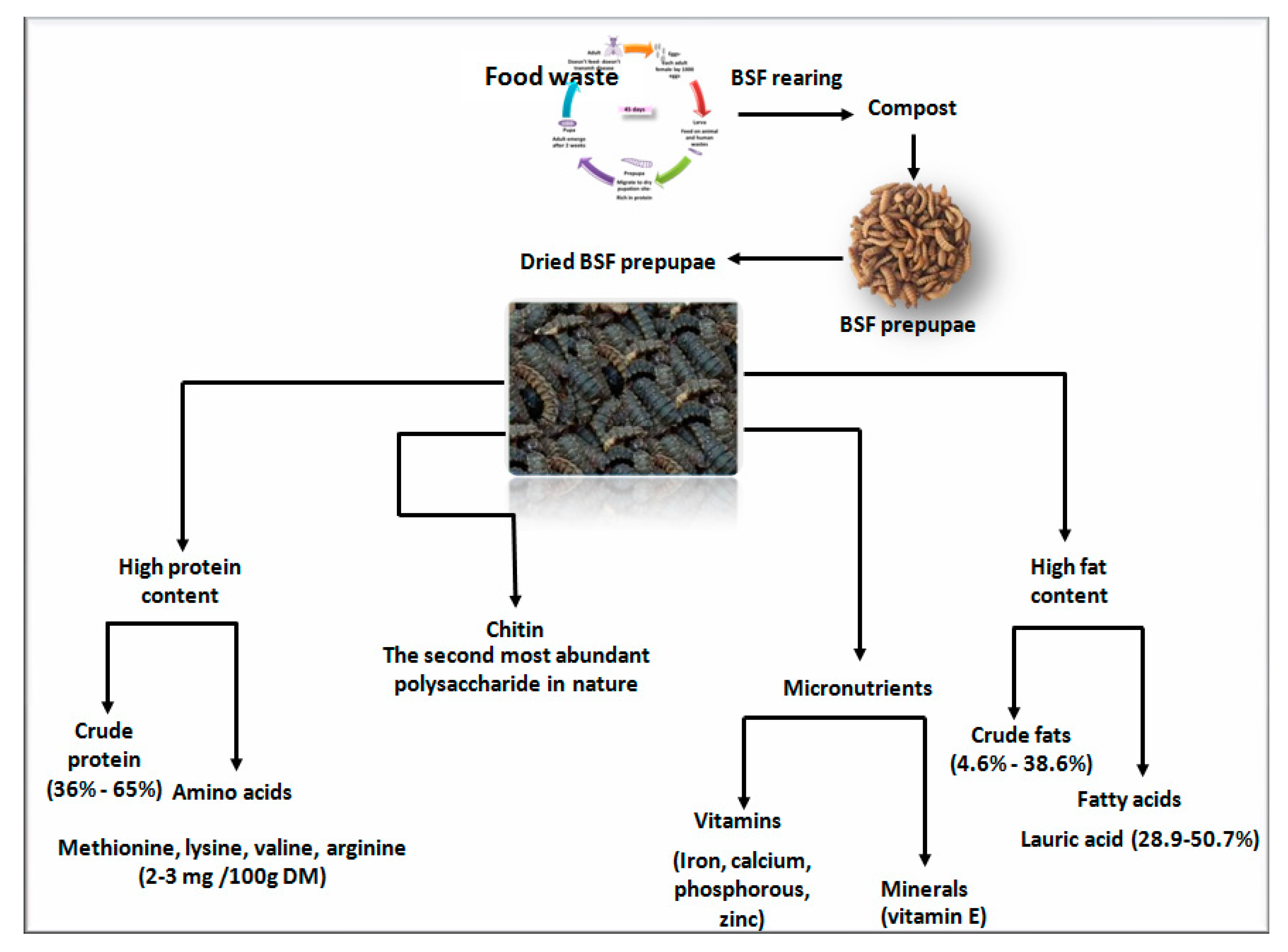
2. Nutritional Values
2.1. Crude Protein and Amino Acids
| References | CP % | CF % | Ash % | Amino Acids % DM | |||||
|---|---|---|---|---|---|---|---|---|---|
| LYS | MET | THR | ARG | VAL | ILE | ||||
| Spranghers et al. [13] | 39.9–43.1 | 21.8–38.6 | 2.7–19.7 | 2.34–2.57 | 0.71–0.87 | 1.54–1.68 | 1.99–2.03 | 2.41–2.82 | 1.72–1.91 |
| St-Hilaire et al. [18] | 43.6 | 33.1 | 15.5 | 2.62 | 0.74 | 1.78 | 2.65 | 2.79 | 2.03 |
| Barroso et al. [39] | 36.2 | 18 | 9.3 | 2.75 | 0.54 | 1.95 | 2.98 | 2.28 | 2.1 |
| Barroso et al. [39] | 40.7 | 15.6 | 19.7 | 2.9 | 1.3 | 2 | 3.27 | 2.6 | 2.17 |
| De Marco et al. [11] | 36.9 | 34.3 | 17.3 | 2.23 | 0.9 | 1.52 | 1.94 | 2.2 | 1.72 |
| De Marco et al. [11] | 55.3 | 18 | 9.9 | 2.1 | 0.65 | 1.7 | 2.2 | 2.7 | 1.9 |
| Cullere et al. [41] | 54.8 | 15.6 | 7.7 | 2.1 | 0.66 | 2.04 | 1.73 | 3.8 | 2.34 |
| De Marco et al. [11] | 65.5 | 4.6 | 9.3 | 2.5 | 0.86 | 2.2 | 2.7 | 3.5 | 2.4 |
| Marono et al. [42] | 62.7 | 4.7 | 8 | 4.14 | 1.33 | 2.37 | - | 5.13 | 3.18 |
| Mwaniki et al. [43] | 57.5 | 7 | - | 3.3 | 0.92 | 2.32 | 2.79 | 3.47 | 2.44 |
2.2. Amino Acids
2.3. Crude Fat and Fatty Acids
2.4. Micronutrients
2.5. Chitin
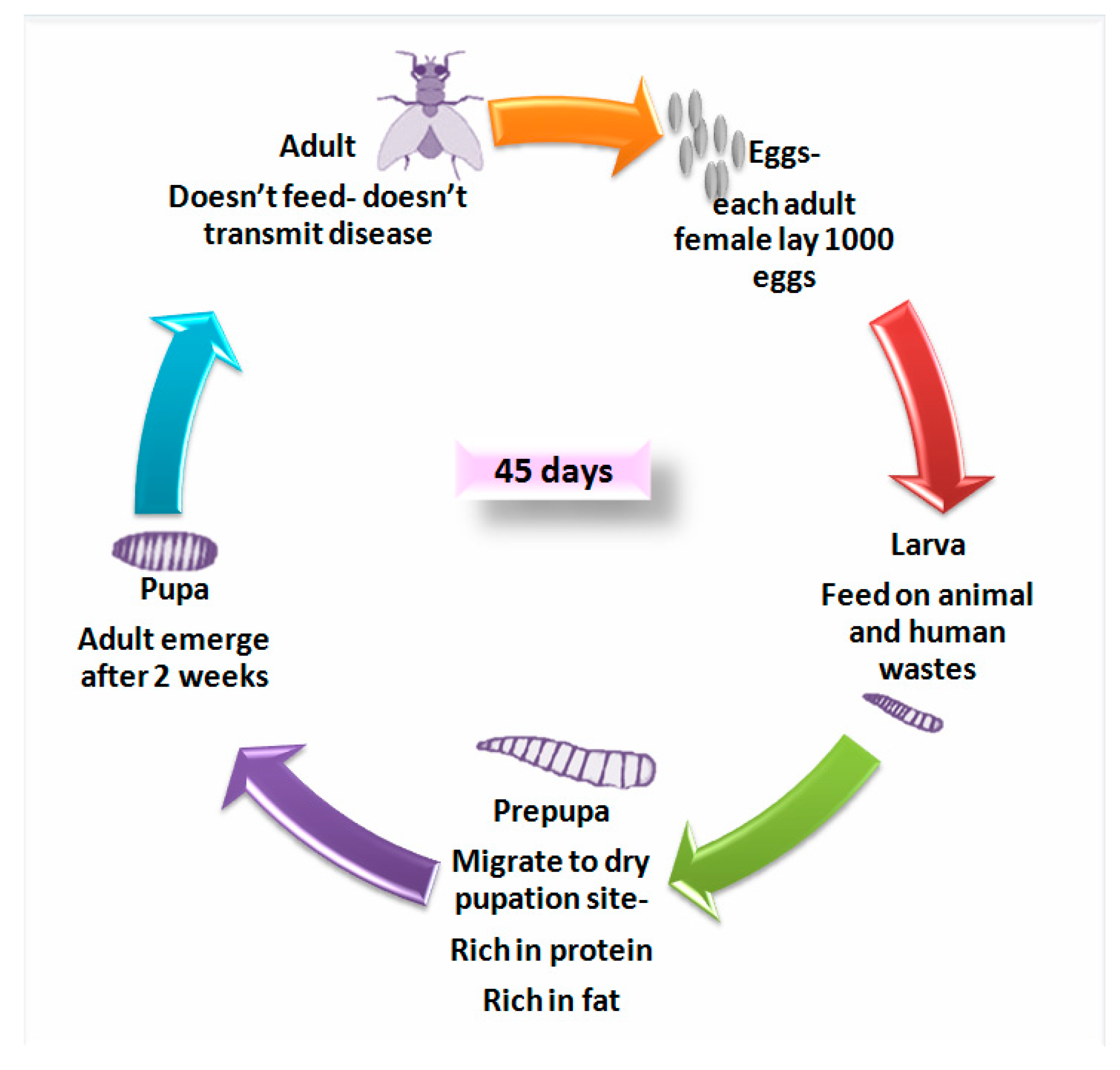
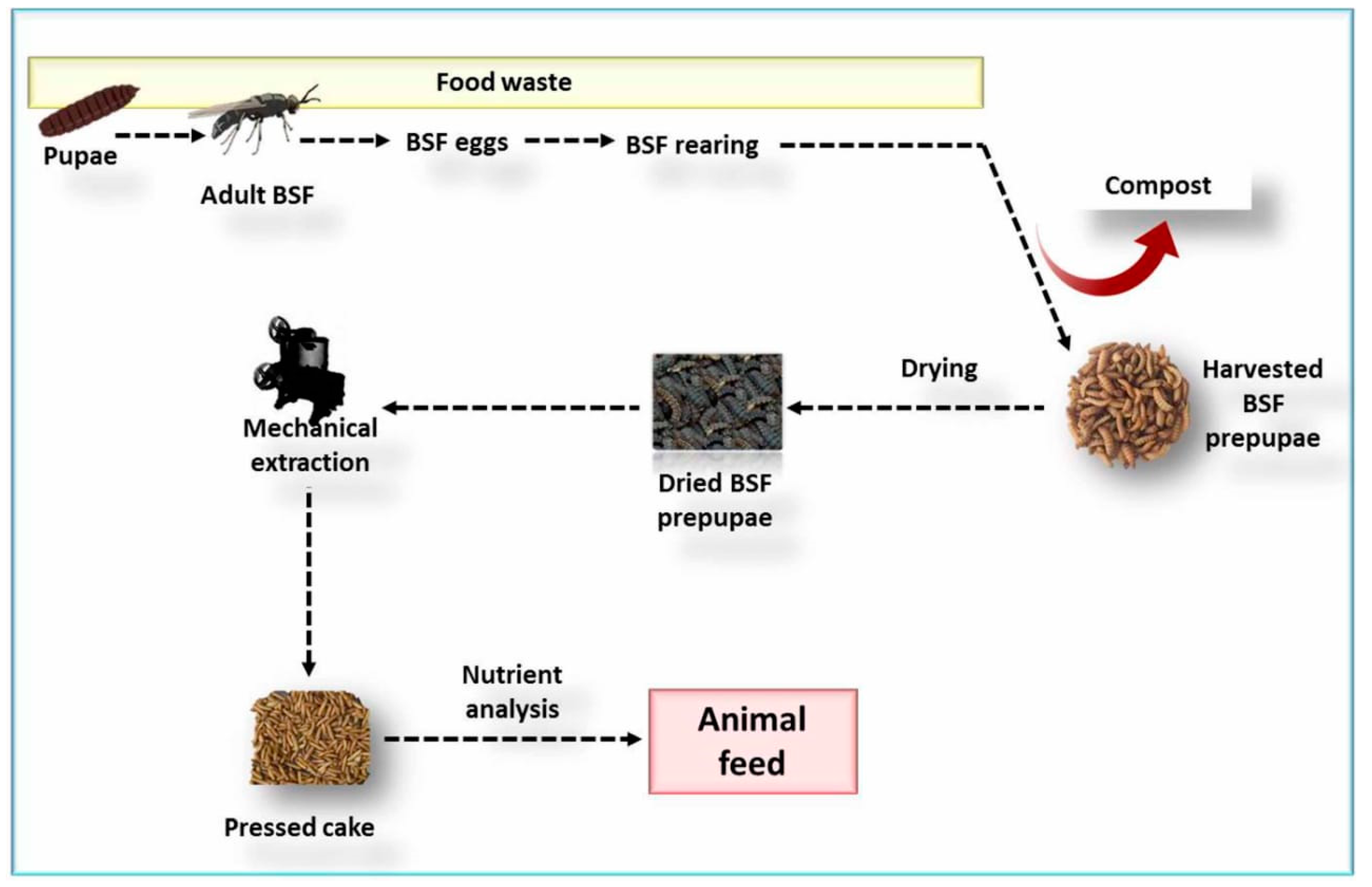
3. Production of Black Soldier Fly
3.1. Larvae Feed and Rearing Substrates
3.2. Growth Performance and Survival
3.3. Conversion Efficiency
3.4. Waste Reduction Index and Environmental Efficiency
4. Effects on Poultry Performance
4.1. Feed Intake
4.2. Daily Body Gain and Live Weight
4.3. Feed Conversion Ratio
4.4. Mortality and Diseases Signs
4.5. Apparent Digestibility
4.6. Apparent Metabolizable Energy (AME) and Nitrogen-Corrected Apparent Metabolizable Energy (AMEn)
4.7. Apparent Ileal Amino Acid Digestibility
4.8. Palatability
4.9. Blood Traits
4.10. Antioxidant Status
4.11. Gut Microbiota
4.12. Fatty Acids in the Ceca
4.13. Gut Morphometry and Histological Traits
4.14. Production Parameters of Layers
4.15. Egg Physico-Chemical Quality
4.16. Carcass Dissection
4.17. Meat Quality
4.18. Sensory Traits
5. Challenges of Using BSF in Poultry Feeding
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roberts, L. 9 billion? Science 2011, 333, 540–543. [Google Scholar] [CrossRef]
- Nijdam, D.; Rood, T.; Westhoek, H. The price of protein: Review of land use and carbon footprints from life cycle assessments of animal food products and their substitutes. Food Policy 2012, 37, 760–770. [Google Scholar] [CrossRef]
- Nkukwana, T.T. Global poultry production: Current impact and future outlook on the South African poultry industry. S. Afr. J. Anim. Sci. 2018, 48, 869–884. [Google Scholar] [CrossRef]
- Chadd, C. Future trends and developments in poultry nutrition. In Proceedings of the Poultry in the 21st century: Avian influenza and beyond. In Proceedings of the International Poultry Conference, Bangkok, Thailand, 5–7 November 2007. [Google Scholar]
- Foley, J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S.; Gerber, J.S.; Johnston, M.; Mueller, N.D.; O’Connell, C.; Ray, D.K.; West, P.C. Solutions for a cultivated planet. Nature 2011, 478, 337. [Google Scholar] [CrossRef] [Green Version]
- Makkar, H.P.S.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed Sci. Technol. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Kouřimská, L.; Adámková, A. Nutritional and sensory quality of edible insects. NFS J. 2016, 4, 22–26. [Google Scholar] [CrossRef] [Green Version]
- Van Huis, A. Potential of insects as food and feed in assuring food security. Annu. Rev. Entomol. 2013, 58, 563–583. [Google Scholar] [CrossRef] [PubMed]
- Bukkens, S.G.F. The nutritional value of edible insects. Ecol. Food Nutr. 1997, 36, 287–319. [Google Scholar] [CrossRef]
- Tabata, E.; Kashimura, A.; Wakita, S.; Ohno, M.; Sakaguchi, M.; Sugahara, Y.; Kino, Y.; Matoska, V.; Bauer, P.O.; Oyama, F. Gastric and intestinal proteases resistance of chicken acidic chitinase nominates chitin-containing organisms for alternative whole edible diets for poultry. Sci. Rep. 2017, 7, 6662. [Google Scholar] [CrossRef] [Green Version]
- De Marco, M.; Martínez, S.; Hernandez, F.; Madrid, J.; Gai, F.; Rotolo, L.; Belforti, M.; Bergero, D.; Katz, H.; Dabbou, S. Nutritional value of two insect larval meals (Tenebrio molitor and Hermetia illucens) for broiler chickens: Apparent nutrient digestibility, apparent ileal amino acid digestibility and apparent metabolizable energy. Anim. Feed Sci. Technol. 2015, 209, 211–218. [Google Scholar] [CrossRef]
- Nyakeri, E.M.; Ogola, H.J.; Ayieko, M.A.; Amimo, F.A. An open system for farming black soldier fly larvae as a source of proteins for smallscale poultry and fish production. J. Insects Food Feed 2017, 3, 51–56. [Google Scholar] [CrossRef]
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 2017, 97, 2594–2600. [Google Scholar] [CrossRef] [PubMed]
- Fitches, E.C.; Dickinson, M.; De Marzo, D.; Wakefield, M.E.; Charlton, A.C.; Hall, H. Alternative protein production for animal feed: Musca domestica productivity on poultry litter and nutritional quality of processed larval meals. J. Insects Food Feed 2019, 5, 77–88. [Google Scholar] [CrossRef]
- Vernooij, A.G.; Veldkamp, T.; Ndambi, A. Insects for Africa: Developing Business Opportunities for Insects in Animal Feed in Eastern Africa; Wageningen Livestock Research: Wageningen, The Netherlands, 2019. [Google Scholar]
- Henry, M.; Gasco, L.; Piccolo, G.; Fountoulaki, E. Review on the use of insects in the diet of farmed fish: Past and future. Anim. Feed Sci. Technol. 2015, 203, 1–22. [Google Scholar] [CrossRef]
- Veldkamp, T.; Van Duinkerken, G.; van Huis, A.; Lakemond, C.M.M.; Ottevanger, E.; Bosch, G.; Van Boekel, T. Insects as a Sustainable Feed Ingredient in Pig and Poultry Diets: A Feasibility Study = Insecten als Duurzame Diervoedergrondstof in Varkens-en Pluimveevoeders: Een Haalbaarheidsstudie; Wageningen UR Livestock Research: Wageningen, The Netherlands, 2012. [Google Scholar]
- St-Hilaire, S.; Sheppard, C.; Tomberlin, J.K.; Irving, S.; Newton, L.; McGuire, M.A.; Mosley, E.E.; Hardy, R.W.; Sealey, W. Fly prepupae as a feedstuff for rainbow trout, Oncorhynchus mykiss. J. World Aquac. Soc. 2007, 38, 59–67. [Google Scholar] [CrossRef]
- Newton, L.; Sheppard, C.; Watson, D.W.; Burtle, G.; Dove, R. Using the Black Soldier Fly, Hermetia Illucens, as a Value-Added Tool for the Management of Swine Manure; Animal and Poultry Waste Management Center, North Carolina State University: Raleigh, NC, USA, 2005; Volume 17. [Google Scholar]
- Newton, G.L.; Sheppard, D.C.; Watson, D.W.; Burtle, G.J.; Dove, C.R.; Tomberlin, J.K.; Thelen, E.E. The black soldier fly, Hermetia illucens, as a manure management/resource recovery tool. In Proceedings of the Symposium on the state of the science of Animal Manure and Waste Management, San Antonio, TX, USA, 15–18 January 2005; pp. 5–7. [Google Scholar]
- Sheppard, D.C.; Newton, G.L.; Thompson, S.A.; Savage, S. A value added manure management system using the black soldier fly. Bioresour. Technol. 1994, 50, 275–279. [Google Scholar] [CrossRef]
- Pimentel, D.; Dritschilo, W.; Krummel, J.; Kutzman, J. Energy and land constraints in food protein production. Science 1975, 190, 754–761. [Google Scholar] [CrossRef]
- Germond, J.; Philippossian, G.; Richli, U.; Bracco, I.; Arnaud, M.J. Rapid and complete urinary elimination of [14C]-5-hydroxymethyl-2-furaldehyde administered orally or intravenously to rats. J. Toxicol. Environ. Heal. Part A Curr. Issues 1987, 22, 79–89. [Google Scholar] [CrossRef]
- Sheppard, C. House fly and lesser fly control utilizing the black soldier fly in manure management systems for caged laying hens. Environ. Entomol. 1983, 12, 1439–1442. [Google Scholar] [CrossRef]
- Tingle, F.C.; Mitchell, E.R.; Copeland, W.W. Soldier fly, Hermetia illucens in poultry in north central Florida. J. Georg. Entomol. Soc. 1975, 10, 179–183. [Google Scholar]
- Wang, S.; Edens, F.W. Stress-induced heat-shock protein synthesis in peripheral leukocytes of turkeys, Meleagris gallopavo. Comp. Biochem. Physiol. Part B Comp. Biochem. 1993, 106, 621–628. [Google Scholar] [CrossRef]
- Payne, C.L.R.; Scarborough, P.; Rayner, M.; Nonaka, K. A systematic review of nutrient composition data available for twelve commercially available edible insects, and comparison with reference values. Trends Food Sci. Technol. 2016, 47, 69–77. [Google Scholar] [CrossRef]
- Newton, G.L.; Booram, C.V.; Barker, R.W.; Hale, O.M. Dried Hermetia illucens larvae meal as a supplement for swine. J. Anim. Sci. 1977, 44, 395–400. [Google Scholar] [CrossRef]
- Oluokun, J.A. Upgrading the nutritive value of full-fat soyabeans meal for broiler production with either fishmeal or black soldier fly larvae meal (Hermetia illucens). Niger. J. Anim. Sci. 2000, 3. [Google Scholar] [CrossRef]
- Solomon, J.M.; Rossi, J.M.; Golic, K.; McGarry, T.; Lindquist, S. Changes in hsp70 alter thermotolerance and heat-shock regulation in Drosophila. New Biol. 1991, 3, 1106–1120. [Google Scholar] [PubMed]
- Booth, D.C.; Sheppard, C. Oviposition of the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae): Eggs, masses, timing, and site characteristics. Environ. Entomol. 1984, 13, 421–423. [Google Scholar] [CrossRef]
- Akhtar, M.S.; Riffat, S. Field trial of Saussurea lappa roots against nematodes and Nigella sativa seeds against cestodes in children. J. Pak. Med. Assoc. 1991, 41, 185–187. [Google Scholar]
- Liu, X.; Chen, X.; Wang, H.; Yang, Q.; ur Rehman, K.; Li, W.; Cai, M.; Li, Q.; Mazza, L.; Zhang, J. Dynamic changes of nutrient composition throughout the entire life cycle of black soldier fly. PLoS ONE 2017, 12, e0182601. [Google Scholar] [CrossRef] [Green Version]
- Oonincx, D.; Dierenfeld, E.S. An investigation into the chemical composition of alternative invertebrate prey. Zoo Biol. 2012, 31, 40–54. [Google Scholar] [CrossRef]
- Oonincx, D.G.A.B.; Van Broekhoven, S.; Van Huis, A.; van Loon, J.J.A. Feed conversion, survival and development, and composition of four insect species on diets composed of food by-products. PLoS ONE 2015, 10, e0144601. [Google Scholar] [CrossRef] [Green Version]
- Veldkamp, T.; Bosch, G. Insects: A protein-rich feed ingredient in pig and poultry diets. Anim. Front. 2015, 5, 45–50. [Google Scholar]
- Schiavone, A.; De Marco, M.; Martínez, S.; Dabbou, S.; Renna, M.; Madrid, J.; Hernandez, F.; Rotolo, L.; Costa, P.; Gai, F. Nutritional value of a partially defatted and a highly defatted black soldier fly larvae (Hermetia illucens L.) meal for broiler chickens: Apparent nutrient digestibility, apparent metabolizable energy and apparent ileal amino acid digestibility. J. Anim. Sci. Biotechnol. 2017, 8, 51. [Google Scholar] [CrossRef]
- Sauvant, D.; Perez, J.-M.; Tran, G. Tables of Composition and Nutritional Value of Feed Materials: Pigs, Poultry, Cattle, Sheep, Goats, Rabbits, Horses and Fish; Wageningen Academic Publishers: Wageningen, The Netherlands, 2004; ISBN 9076998418. [Google Scholar]
- Barroso, F.G.; de Haro, C.; Sánchez-Muros, M.-J.; Venegas, E.; Martínez-Sánchez, A.; Pérez-Bañón, C. The potential of various insect species for use as food for fish. Aquaculture 2014, 422, 193–201. [Google Scholar] [CrossRef]
- Finke, M.D. Complete nutrient content of four species of feeder insects. Zoo Biol. 2013, 32, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Cullere, M.; Tasoniero, G.; Giaccone, V.; Miotti-Scapin, R.; Claeys, E.; De Smet, S.; Dalle Zotte, A. Black soldier fly as dietary protein source for broiler quails: Apparent digestibility, excreta microbial load, feed choice, performance, carcass and meat traits. Animal 2016, 10, 1923–1930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marono, S.; Loponte, R.; Lombardi, P.; Vassalotti, G.; Pero, M.E.; Russo, F.; Gasco, L.; Parisi, G.; Piccolo, G.; Nizza, S. Productive performance and blood profiles of laying hens fed Hermetia illucens larvae meal as total replacement of soybean meal from 24 to 45 weeks of age. Poult. Sci. 2017, 96, 1783–1790. [Google Scholar] [CrossRef] [PubMed]
- Mwaniki, Z.; Neijat, M.; Kiarie, E. Egg production and quality responses of adding up to 7.5% defatted black soldier fly larvae meal in a corn–soybean meal diet fed to Shaver White Leghorns from wk 19 to 27 of age. Poult. Sci. 2018, 97, 2829–2835. [Google Scholar] [CrossRef]
- Van Huis, A.; Van Itterbeeck, J.; Klunder, H.; Mertens, E.; Halloran, A.; Muir, G.; Vantomme, P. Edible Insects: Future Prospects for Food and Feed Security; Food and Agriculture Organization of the United Nations: Rome, Italy, 2013; ISBN 9251075964. [Google Scholar]
- Verkerk, M.C.; Tramper, J.; Van Trijp, J.C.M.; Martens, D.E. Insect cells for human food. Biotechnol. Adv. 2007, 25, 198–202. [Google Scholar] [CrossRef]
- Tran, G.; Heuzé, V.; Makkar, H.P.S. Insects in fish diets. Anim. Front. 2015, 5, 37–44. [Google Scholar]
- Elwert, C.; Knips, I.; Katz, P. A novel protein source: Maggot meal of the black soldier fly (Hermetia illucens) in broiler feed. Tag. Schweine-und Geflügelernährung 2010, 11, 140–142. [Google Scholar]
- Ravindran, V.; Hew, L.I.; Ravindran, G.; Bryden, W.L. A comparison of ileal digesta and excreta analysis for the determination of amino acid digestibility in food ingredients for poultry. Br. Poult. Sci. 1999, 40, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Tschirner, M.; Simon, A. Influence of different growing substrates and processing on the nutrient composition of black soldier fly larvae destined for animal feed. J. Insects Food Feed 2015, 1, 249–259. [Google Scholar] [CrossRef]
- NRC, National Research Council. Nutrient Requirements of Poultry: 1994; National Academies Press: Washington, DC, USA, 1994; ISBN 0309048923. [Google Scholar]
- Gasco, L.; Dabbou, S.; Trocino, A.; Xiccato, G.; Capucchio, M.T.; Biasato, I.; Dezzutto, D.; Birolo, M.; Meneguz, M.; Schiavone, A. Effect of dietary supplementation with insect fats on growth performance, digestive efficiency and health of rabbits. J. Anim. Sci. Biotechnol. 2019, 10, 4. [Google Scholar] [CrossRef] [PubMed]
- Gasco, L.; Dabbou, S.; Gai, F.; Brugiapaglia, A.; Schiavone, A.; Birolo, M.; Xiccato, G.; Trocino, A. Quality and consumer acceptance of meat from rabbits fed diets in which soybean oil is replaced with black soldier fly and yellow mealworm fats. Animals 2019, 9, 629. [Google Scholar] [CrossRef] [Green Version]
- McGuckin, M.A.; Lindén, S.K.; Sutton, P.; Florin, T.H. Mucin dynamics and enteric pathogens. Nat. Rev. Microbiol. 2011, 9, 265–278. [Google Scholar] [CrossRef]
- Alagawany, M.; Elnesr, S.S.; Farag, M.R.; El-Hack, A.; Mohamed, E.; Khafaga, A.F.; Taha, A.E.; Tiwari, R.; Yatoo, M.; Bhatt, P. Omega-3 and omega-6 fatty acids in poultry nutrition: Effect on production performance and health. Animals 2019, 9, 573. [Google Scholar] [CrossRef] [Green Version]
- Kawasaki, K.; Hashimoto, Y.; Hori, A.; Kawasaki, T.; Hirayasu, H.; Iwase, S.; Hashizume, A.; Ido, A.; Miura, C.; Miura, T. Evaluation of black soldier fly (Hermetia illucens) larvae and pre-pupae raised on household organic waste, as potential ingredients for poultry feed. Animals 2019, 9, 98. [Google Scholar] [CrossRef] [Green Version]
- Koletzko, B.; Baker, S.; Cleghorn, G.; Neto, U.F.; Gopalan, S.; Hernell, O.; Hock, Q.S.; Jirapinyo, P.; Lonnerdal, B.; Pencharz, P. Global standard for the composition of infant formula: Recommendations of an ESPGHAN coordinated international expert group. J. Pediatr. Gastroenterol. Nutr. 2005, 41, 584–599. [Google Scholar] [CrossRef] [Green Version]
- Michaelsen, K.F.; Hoppe, C.; Roos, N.; Kaestel, P.; Stougaard, M.; Lauritzen, L.; Mølgaard, C.; Girma, T.; Friis, H. Choice of foods and ingredients for moderately malnourished children 6 months to 5 years of age. Food Nutr. Bull. 2009, 30, S343–S404. [Google Scholar] [CrossRef] [Green Version]
- Liland, N.S.; Biancarosa, I.; Araujo, P.; Biemans, D.; Bruckner, C.G.; Waagbø, R.; Torstensen, B.E.; Lock, E.-J. Modulation of nutrient composition of black soldier fly (Hermetia illucens) larvae by feeding seaweed-enriched media. PLoS ONE 2017, 12, e0183188. [Google Scholar] [CrossRef]
- Moula, N.; Scippo, M.-L.; Douny, C.; Degand, G.; Dawans, E.; Cabaraux, J.-F.; Hornick, J.-L.; Medigo, R.C.; Leroy, P.; Francis, F.; et al. Performances of local poultry breed fed black soldier fly larvae reared on horse manure. Anim. Nutr. 2018, 4, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Da Silva, C.A.; Lee, J.-Y.; Hartl, D.; Elias, J.A. Chitin regulation of immune responses: An old molecule with new roles. Curr. Opin. Immunol. 2008, 20, 684–689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caligiani, A.; Marseglia, A.; Leni, G.; Baldassarre, S.; Maistrello, L.; Dossena, A.; Sforza, S. Composition of black soldier fly prepupae and systematic approaches for extraction and fractionation of proteins, lipids and chitin. Food Res. Int. 2018, 105, 812–820. [Google Scholar] [CrossRef]
- Gortari, M.C.; Hours, R.A. Biotechnological processes for chitin recovery out of crustacean waste: A mini-review. Electron. J. Biotechnol. 2013, 16, 14. [Google Scholar]
- Liaqat, F.; Eltem, R. Chitooligosaccharides and their biological activities: A comprehensive review. Carbohydr. Polym. 2018, 184, 243–259. [Google Scholar] [CrossRef] [PubMed]
- Muzzarelli, R. Chitins and chitosans as immunoadjuvants and non-allergenic drug carriers. Mar. Drugs 2010, 8, 292–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Benhabiles, M.S.; Salah, R.; Lounici, H.; Drouiche, N.; Goosen, M.F.A.; Mameri, N. Antibacterial activity of chitin, chitosan and its oligomers prepared from shrimp shell waste. Food Hydrocoll. 2012, 29, 48–56. [Google Scholar] [CrossRef]
- Sánchez, Á.; Mengíbar, M.; Rivera-Rodríguez, G.; Moerchbacher, B.; Acosta, N.; Heras, A. The effect of preparation processes on the physicochemical characteristics and antibacterial activity of chitooligosaccharides. Carbohydr. Polym. 2017, 157, 251–257. [Google Scholar] [CrossRef]
- Mateos-Aparicio, I.; Mengíbar, M.; Heras, A. Effect of chito-oligosaccharides over human faecal microbiota during fermentation in batch cultures. Carbohydr. Polym. 2016, 137, 617–624. [Google Scholar] [CrossRef]
- Fernandes, J.C.; Tavaria, F.K.; Soares, J.C.; Ramos, Ó.S.; Monteiro, M.J.; Pintado, M.E.; Malcata, F.X. Antimicrobial effects of chitosans and chitooligosaccharides, upon Staphylococcus aureus and Escherichia coli, in food model systems. Food Microbiol. 2008, 25, 922–928. [Google Scholar] [CrossRef]
- Laokuldilok, T.; Potivas, T.; Kanha, N.; Surawang, S.; Seesuriyachan, P.; Wangtueai, S.; Phimolsiripol, Y.; Regenstein, J.M. Physicochemical, antioxidant, and antimicrobial properties of chitooligosaccharides produced using three different enzyme treatments. Food Biosci. 2017, 18, 28–33. [Google Scholar] [CrossRef]
- Choi, B.-K.; Kim, K.-Y.; Yoo, Y.-J.; Oh, S.-J.; Choi, J.-H.; Kim, C.-Y. In vitro antimicrobial activity of a chitooligosaccharide mixture against Actinobacillusactinomycetemcomitans and Streptococcus mutans. Int. J. Antimicrob. Agents 2001, 18, 553–557. [Google Scholar] [CrossRef]
- Liu, P.; Piao, X.S.; Thacker, P.A.; Zeng, Z.K.; Li, P.F.; Wang, D.; Kim, S.W. Chito-oligosaccharide reduces diarrhea incidence and attenuates the immune response of weaned pigs challenged with Escherichia coli K88. J. Anim. Sci. 2010, 88, 3871–3879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira, E.N.; El Gueddari, N.E.; Moerschbacher, B.M.; Peter, M.G.; Franco, T.T. Growth of phytopathogenic fungi in the presence of partially acetylated chitooligosaccharides. Mycopathologia 2008, 166, 163–174. [Google Scholar] [CrossRef] [Green Version]
- Rahman, M.H.; Hjeljord, L.G.; Aam, B.B.; Sørlie, M.; Tronsmo, A. Antifungal effect of chito-oligosaccharides with different degrees of polymerization. Eur. J. Plant Pathol. 2015, 141, 147–158. [Google Scholar] [CrossRef]
- Mei, Y.; Dai, X.; Yang, W.; Xu, X.; Liang, Y. Antifungal activity of chitooligosaccharides against the dermatophyte Trichophyton rubrum. Int. J. Biol. Macromol. 2015, 77, 330–335. [Google Scholar] [CrossRef]
- Chirkov, S.N. The antiviral activity of Chitosan (review). Appl. Biochem. Microbiol. 2002, 38, 1–8. [Google Scholar] [CrossRef]
- Artan, M.; Karadeniz, F.; Karagozlu, M.Z.; Kim, M.-M.; Kim, S.-K. Anti-HIV-1 activity of low molecular weight sulfated chitooligosaccharides. Carbohydr. Res. 2010, 345, 656–662. [Google Scholar] [CrossRef]
- Cheung, R.C.F.; Wong, J.H.; Pan, W.L.; Chan, Y.S.; Yin, C.M.; Dan, X.L.; Wang, H.X.; Fang, E.F.; Lam, S.K.; Ngai, P.H.K.; et al. Antifungal and antiviral products of marine organisms. Appl. Microbiol. Biotechnol. 2014, 98, 3475–3494. [Google Scholar] [CrossRef] [Green Version]
- Niu, S.; Yang, L.; Zuo, H.; Zheng, J.; Weng, S.; He, J.; Xu, X. A chitinase from pacific white shrimp Litopenaeus vannamei involved in immune regulation. Dev. Comp. Immunol. 2018, 85, 161–169. [Google Scholar] [CrossRef]
- Gerez, J.R.; Buck, L.Y.; Marutani, V.H.B.; Calliari, C.M.; Cunha, L.S.; Bracarense, A.L. Effects of chito-oligosaccharide on piglet jejunal explants: An histological approach. Animal 2019, 13, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Nouri, A. Chitosan nano-encapsulation improves the effects of mint, thyme, and cinnamon essential oils in broiler chickens. Br. Poult. Sci. 2019, 60, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Swiatkiewicz, S.; Swiatkiewicz, M.; Arczewska-Wlosek, A.; Jozefiak, D. Chitosan and its oligosaccharide derivatives (chito-oligosaccharides) as feed supplements in poultry and swine nutrition. J. Anim. Physiol. Anim. Nutr. 2015, 99, 1–12. [Google Scholar] [CrossRef]
- Hu, S.; Wang, Y.; Wen, X.; Wang, L.; Jiang, Z.; Zheng, C. Effects of low-molecular-weight chitosan on the growth performance, intestinal morphology, barrier function, cytokine expression and antioxidant system of weaned piglets. BMC Vet. Res. 2018, 14, 215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khambualai, O.; Yamauchi, K.; Tangtaweewipat, S.; Cheva-Isarakul, B. Growth performance and intestinal histology in broiler chickens fed with dietary chitosan. Br. Poult. Sci. 2009, 50, 592–597. [Google Scholar] [CrossRef]
- Howell, L. Global Risks 2013; World Economic Forum: Cologny, Switzerland, 2013. [Google Scholar]
- Callan, E. Hermetia illucens (L.) (Diptera, Stratiomyidae), a cosmopolitan American species long established in Australia and New Zealand. Entomol. Mon. Mag. 1974, 109, 232–234. [Google Scholar]
- Leclercq, M. Dispersion et transport des insectes nuisibles: Hermetia illucens L.(diptera Stratiomyidae). Bull. Rech. Agron. Gembloux 1969, 1, 60–62. [Google Scholar]
- Diener, S.; Zurbrügg, C.; Tockner, K. Conversion of organic material by black soldier fly larvae: Establishing optimal feeding rates. Waste Manag. Res. 2009, 27, 603–610. [Google Scholar] [CrossRef]
- Sheppard, D.C.; Tomberlin, J.K.; Joyce, J.A.; Kiser, B.C.; Sumner, S.M. Rearing methods for the black soldier fly (Diptera: Stratiomyidae). J. Med. Entomol. 2002, 39, 695–698. [Google Scholar] [CrossRef] [Green Version]
- Tomberlin, J.K. Biological, Behavioral, and Toxicological Studies on the Black Soldier Fly (Diptera: Stratiomyidae). Ph.D. Thesis, University of Georgia, Athens, GA, USA, 2001. [Google Scholar]
- Salomone, R.; Saija, G.; Mondello, G.; Giannetto, A.; Fasulo, S.; Savastano, D. Environmental impact of food waste bioconversion by insects: Application of life cycle assessment to process using Hermetia illucens. J. Clean. Prod. 2017, 140, 890–905. [Google Scholar] [CrossRef]
- Tomberlin, J.K.; Sheppard, D.C. Lekking behavior of the black soldier fly (Diptera: Stratiomyidae). Florida Entomol. 2001, 84, 729. [Google Scholar] [CrossRef]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Ability of black soldier fly (Diptera: Stratiomyidae) larvae to recycle food waste. Environ. Entomol. 2015, 44, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Li, Q.; Zhang, J.; Yu, Z. Double the biodiesel yield: Rearing black soldier fly larvae, Hermetia illucens, on solid residual fraction of restaurant waste after grease extraction for biodiesel production. Renew. Energy 2012, 41, 75–79. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Dragone, G.; Roberto, I.C. Brewers’ spent grain: Generation, characteristics and potential applications. J. Cereal Sci. 2006, 43, 1–14. [Google Scholar] [CrossRef]
- Khare, S.K.; Jha, K.; Gandhi, A.P. Citric acid production from Okara (soy-residue) by solid-state fermentation. Bioresour. Technol. 1995, 54, 323–325. [Google Scholar] [CrossRef]
- Li, S.; Zhu, D.; Li, K.; Yang, Y.; Lei, Z.; Zhang, Z. Soybean curd residue: Composition, utilization, and related limiting factors. ISRN Ind. Eng. 2013, 2013, 423590. [Google Scholar] [CrossRef]
- Cammack, J.; Tomberlin, J. The impact of diet protein and carbohydrate on select life-history traits of the black soldier fly Hermetia illucens (L.) (Diptera: Stratiomyidae). Insects 2017, 8, 56. [Google Scholar] [CrossRef] [Green Version]
- Furman, D.P.; Young, R.D.; Catts, P.E. Hermetia illucens (Linnaeus) as a Factor in the natural control of musca domestica linnaeus. J. Econ. Entomol. 1959, 52, 917–921. [Google Scholar] [CrossRef]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Influence of resources on Hermetia illucens (Diptera: Stratiomyidae) larval development. J. Med. Entomol. 2013, 50, 898–906. [Google Scholar] [CrossRef] [Green Version]
- Scriber, J.M.; Slansky, F. The nutritional ecology of immature insects. Annu. Rev. Entomol. 1981, 26, 183–211. [Google Scholar] [CrossRef]
- Lundy, M.E.; Parrella, M.P. Crickets are not a free lunch: Protein capture from scalable organic side-streams via high-density populations of Acheta domesticus. PLoS ONE 2015, 10, e0118785. [Google Scholar] [CrossRef] [PubMed]
- Bradley, S.W.; Sheppard, D.C. House fly oviposition inhibition by larvae ofHermetia illucens, the black soldier fly. J. Chem. Ecol. 1984, 10, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Ghany, N.M. Pheromones and chemical communication in insects. In Pheromones; IntechOpen: London, UK, 2020. [Google Scholar]
- Liu, Q.; Tomberlin, J.K.; Brady, J.A.; Sanford, M.R.; Yu, Z. Black soldier fly (Diptera: Stratiomyidae) larvae reduce Escherichia coli in dairy manure. Environ. Entomol. 2008, 37, 1525–1530. [Google Scholar] [CrossRef] [PubMed]
- Erickson, M.C.; Islam, M.; Sheppard, C.; Liao, J.; Doyle, M.P. Reduction of escherichia coli O157: H7 and Salmonella enterica serovar enteritidis in chicken manure by larvae of the black soldier fly. J. Food Prot. 2004, 67, 685–690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muller, A.; Wiedmer, S.; Kurth, M. Risk evaluation of passive transmission of animal parasites by feeding of black soldier fly (Hermetia illucens) larvae and prepupae. J. Food Prot. 2019, 82, 948–954. [Google Scholar] [CrossRef] [PubMed]
- Lalander, C.; Diener, S.; Magri, M.E.; Zurbrügg, C.; Lindström, A.; Vinnerås, B. Faecal sludge management with the larvae of the black soldier fly (Hermetia illucens)—From a hygiene aspect. Sci. Total Environ. 2013, 458–460, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Parnés, A.; Lagan, K.M. Larval therapy in wound management: A review. Int. J. Clin. Pract. 2007, 61, 488–493. [Google Scholar] [CrossRef]
- Sherman, R.A. Maggot debridement in modern medicine. Infect. Med. 1998, 15, 651–656. [Google Scholar]
- Hoffmann, J.A.; Hetru, C. Insect defensins: Inducible antibacterial peptides. Immunol. Today 1992, 13, 411–415. [Google Scholar] [CrossRef]
- Banks, I.J.; Gibson, W.T.; Cameron, M.M. Growth rates of black soldier fly larvae fed on fresh human faeces and their implication for improving sanitation. Trop. Med. Int. Health 2014, 19, 14–22. [Google Scholar] [CrossRef]
- Maurer, V.; Holinger, M.; Amsler, Z.; Früh, B.; Wohlfahrt, J.; Stamer, A.; Leiber, F. Replacement of soybean cake by Hermetia illucens meal in diets for layers. J. Insects Food Feed 2016, 2, 83–90. [Google Scholar] [CrossRef] [Green Version]
- Hwangbo, J.; Hong, E.C.; Jang, A.; Kang, H.K.; Oh, J.S.; Kim, B.W.; Park, B.S. Utilization of house fly-maggots, a feed supplement in the production of broiler chickens. J. Environ. Biol. 2009, 30, 609–614. [Google Scholar] [PubMed]
- Borrelli, L.; Coretti, L.; Dipineto, L.; Bovera, F.; Menna, F.; Chiariotti, L.; Nizza, A.; Lembo, F.; Fioretti, A. Insect-based diet, a promising nutritional source, modulates gut microbiota composition and SCFAs production in laying hens. Sci. Rep. 2017, 7, 16269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jocken, J.W.E.; González Hernández, M.A.; Hoebers, N.T.H.; van der Beek, C.M.; Essers, Y.P.G.; Blaak, E.E.; Canfora, E.E. Short-chain fatty acids differentially affect intracellular lipolysis in a human white adipocyte model. Front. Endocrinol. 2018, 8, 372. [Google Scholar] [CrossRef] [Green Version]
- Byrne, C.S.; Chambers, E.S.; Alhabeeb, H.; Chhina, N.; Morrison, D.J.; Preston, T.; Tedford, C.; Fitzpatrick, J.; Irani, C.; Busza, A.; et al. Increased colonic propionate reduces anticipatory reward responses in the human striatum to high-energy foods. Am. J. Clin. Nutr. 2016, 104, 5–14. [Google Scholar] [CrossRef] [Green Version]
- Chambers, E.S.; Viardot, A.; Psichas, A.; Morrison, D.J.; Murphy, K.G.; Zac-Varghese, S.E.K.; MacDougall, K.; Preston, T.; Tedford, C.; Finlayson, G.S. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 2015, 64, 1744–1754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evans, E.W.; Beach, G.G.; Wunderlich, J.; Harmon, B.G. Isolation of antimicrobial peptides from avian heterophils. J. Leukoc. Biol. 1994, 56, 661–665. [Google Scholar] [CrossRef]
- Ruhnke, I.; Normant, C.; Campbell, D.L.M.; Iqbal, Z.; Lee, C.; Hinch, G.N.; Roberts, J. Impact of on-range choice feeding with black soldier fly larvae (Hermetia illucens) on flock performance, egg quality, and range use of free-range laying hens. Anim. Nutr. 2018, 4, 452–460. [Google Scholar] [CrossRef]
- Gariglio, M.; Dabbou, S.; Crispo, M.; Biasato, I.; Gai, F.; Gasco, L.; Piacente, F.; Odetti, P.; Bergagna, S.; Plachà, I. Effects of the dietary inclusion of partially defatted black soldier fly (Hermetia illucens) meal on the blood chemistry and tissue (spleen, liver, thymus, and bursa of fabricius) histology of muscovy ducks (cairina moschata domestica). Animals 2019, 9, 307. [Google Scholar] [CrossRef] [Green Version]
- Pretorius, Q. The Evaluation of Larvae of Musca Domestica (Common House Fly) as Protein Source for Broiler Production. Master’s Thesis, Stellenbosch University, Stellenbosch, South Afirica, 2011. [Google Scholar]
- Cousins, R.J. Absorption, transport, and hepatic metabolism of copper and zinc: Special reference to metallothionein and ceruloplasmin. Physiol. Rev. 1985, 65, 238–309. [Google Scholar] [CrossRef]
- Dabbou, S.; Gai, F.; Biasato, I.; Capucchio, M.T.; Biasibetti, E.; Dezzutto, D.; Meneguz, M.; Plachà, I.; Gasco, L.; Schiavone, A. Black soldier fly defatted meal as a dietary protein source for broiler chickens: Effects on growth performance, blood traits, gut morphology and histological features. J. Anim. Sci. Biotechnol. 2018, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Agunbiade, J.A.; Adeyemi, O.A.; Ashiru, O.M.; Awojobi, H.A.; Taiwo, A.A.; Oke, D.B.; Adekunmisi, A.A. Replacement of fish meal with maggot meal in cassava-based layers’ diets. J. Poult. Sci. 2007, 44, 278–282. [Google Scholar] [CrossRef] [Green Version]
- Koo, S.I.; Currin, T.A.; Johnson, M.G.; King, E.W.; Turk, D.E. The nutritional value and microbial content of dried face fly pupae (Musca autumnalis (De Geer)) when fed to chicks. Poult. Sci. 1980, 59, 2514–2518. [Google Scholar] [CrossRef]
- Atteh, J.O. The replacement value of maggots for groundnut cake in broiler diet. Cent. Point 1990, 2, 15–19. [Google Scholar]
- Sealey, W.M.; Gaylord, T.G.; Barrows, F.T.; Tomberlin, J.K.; McGuire, M.A.; Ross, C.; St-Hilaire, S. Sensory analysis of rainbow trout, Oncorhynchus mykiss, fed enriched black soldier fly prepupae, Hermetia illucens. J. World Aquac. Soc. 2011, 42, 34–45. [Google Scholar] [CrossRef]
- Koh, K.; Iwamae, S. Chitinolytic activity of mucosal enzymes in the different parts of the digestive tract in broilers. J. Poult. Sci. 2012, 50, 120054. [Google Scholar] [CrossRef] [Green Version]
- Kroeckel, S.; Harjes, A.-G.E.; Roth, I.; Katz, H.; Wuertz, S.; Susenbeth, A.; Schulz, C. When a turbot catches a fly: Evaluation of a pre-pupae meal of the Black Soldier Fly (Hermetia illucens) as fish meal substitute—Growth performance and chitin degradation in juvenile turbot (Psetta maxima). Aquaculture 2012, 364, 345–352. [Google Scholar] [CrossRef]
- Jeuniaux, C.; Cornelius, C. Distribution and activity of chitinolytic enzymes in the digestive tract of birds and mammals. Life Sci. 1997, 2, 544–548. [Google Scholar]
- Ramos-Elorduy, J.; González, E.A.; Hernández, A.R.; Pino, J.M. Use of Tenebrio molitor (Coleoptera: Tenebrionidae) to recycle organic wastes and as feed for broiler chickens. J. Econ. Entomol. 2002, 95, 214–220. [Google Scholar] [CrossRef]
- Han, B.K.; Lee, W.J.; Jo, D.H. Chitinolytic enzymes from the gizzard and the chyme of the broiler (Gallus gallus L.). Biotechnol. Lett. 1997, 19, 981–984. [Google Scholar] [CrossRef]
- Suzuki, M.; Fujimoto, W.; Goto, M.; Morimatsu, M.; Syuto, B.; Iwanaga, T. Cellular expression of gut chitinase mRNA in the gastrointestinal tract of mice and chickens. J. Histochem. Cytochem. 2002, 50, 1081–1089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tabata, E.; Kashimura, A.; Kikuchi, A.; Masuda, H.; Miyahara, R.; Hiruma, Y.; Wakita, S.; Ohno, M.; Sakaguchi, M.; Sugahara, Y. Chitin digestibility is dependent on feeding behaviors, which determine acidic chitinase mRNA levels in mammalian and poultry stomachs. Sci. Rep. 2018, 8, 1461. [Google Scholar] [CrossRef] [PubMed]
- Cutrignelli, M.I.; Messina, M.; Tulli, F.; Randazzo, B.; Olivotto, I.; Gasco, L.; Loponte, R.; Bovera, F. Evaluation of an insect meal of the Black Soldier Fly (Hermetia illucens) as soybean substitute: Intestinal morphometry, enzymatic and microbial activity in laying hens. Res. Vet. Sci. 2018, 117, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Payne, W.L.; Combs, G.F.; Kifer, R.R.; Snyder, D.G. Investigation of protein quality-ileal recovery of amino acids. In Proceedings of the Federation Proceedings, San Francisco, CA, USA, 9–11 December 1968; Volume 27, p. 1199. [Google Scholar]
- Loponte, R.; Nizza, S.; Bovera, F.; De Riu, N.; Fliegerova, K.; Lombardi, P.; Vassalotti, G.; Mastellone, V.; Nizza, A.; Moniello, G. Growth performance, blood profiles and carcass traits of Barbary partridge (Alectoris barbara) fed two different insect larvae meals (Tenebrio molitor and Hermetia illucens). Res. Vet. Sci. 2017, 115, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Hainida, E.; Ismail, A.; Hashim, N.; Mohd.-Esa, N.; Zakiah, A. Effects of defatted dried roselle (Hibiscus sabdariffa L.) seed powder on lipid profiles of hypercholesterolemia rats. J. Sci. Food Agric. 2008, 88, 1043–1050. [Google Scholar] [CrossRef]
- Ravindran, V.; Hew, L.I.; Ravindran, G.; Bryden, W.L. Apparent ileal digestibility of amino acids in dietary ingredients for broiler chickens. Anim. Sci. 2005, 81, 85–97. [Google Scholar] [CrossRef]
- Valencia, D.G.; Serrano, M.P.; Lázaro, R.; Jiménez-Moreno, E.; Mateos, G.G. Influence of micronization (fine grinding) of soya bean meal and full-fat soya bean on the ileal digestibility of amino acids for broilers. Anim. Feed Sci. Technol. 2009, 150, 238–248. [Google Scholar] [CrossRef]
- Sturkie, P.D.; Griminger, P. Body fluids: Blood. In Avian physiology; Springer: Cambridge, MA, USA, 1986; pp. 102–129. [Google Scholar]
- Elieh Ali Komi, D.; Sharma, L.; Dela Cruz, C.S. Chitin and its effects on inflammatory and immune responses. Clin. Rev. Allergy Immunol. 2018, 54, 213–223. [Google Scholar] [CrossRef] [Green Version]
- Gopalakannan, A.; Arul, V. Immunomodulatory effects of dietary intake of chitin, chitosan and levamisole on the immune system of Cyprinus carpio and control of Aeromonas hydrophila infection in ponds. Aquaculture 2006, 255, 179–187. [Google Scholar] [CrossRef]
- Lu, Y.; Fan, C.; Li, P.; Lu, Y.; Chang, X.; Qi, K. Short chain fatty acids prevent high-fat-diet-induced obesity in mice by regulating G protein-coupled receptors and gut microbiota. Sci. Rep. 2016, 6, 37589. [Google Scholar] [CrossRef] [Green Version]
- Hossain, S.M.; Blair, R. Chitin utilisation by broilers and its effect on body composition and blood metabolites. Br. Poult. Sci. 2007, 48, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Bedford, A.; Gong, J. Implications of butyrate and its derivatives for gut health and animal production. Anim. Nutr. 2018, 4, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Trinidad, T.P.; Wolever, T.M.; Thompson, L.U. Effect of acetate and propionate on calcium absorption from the rectum and distal colon of humans. Am. J. Clin. Nutr. 1996, 63, 574–578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delzenne, N.M.; Cani, P.D.; Everard, A.; Neyrinck, A.M.; Bindels, L.B. Gut microorganisms as promising targets for the management of type 2 diabetes. Diabetologia 2015, 58, 2206–2217. [Google Scholar] [CrossRef]
- Spranghers, T.; Michiels, J.; Vrancx, J.; Ovyn, A.; Eeckhout, M.; De Clercq, P.; De Smet, S. Gut antimicrobial effects and nutritional value of black soldier fly (Hermetia illucens L.) prepupae for weaned piglets. Anim. Feed Sci. Technol. 2018, 235, 33–42. [Google Scholar] [CrossRef]
- Skřivanová, E.; Marounek, M.; Dlouha, G.; Kaňka, J. Susceptibility of clostridium perfringens to C2–C18 fatty acids. Lett. Appl. Microbiol. 2005, 41, 77–81. [Google Scholar] [CrossRef]
- Srivastava, Y.; Semwal, A.D.; Sharma, G.K. Chapter 16—Virgin coconut oil as functional oil. In Probiotic, and Unconventional Foods; Grumezescu, A.M., Holban, A.M.B.T.-T., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 291–301. ISBN 978-0-12-814625-5. [Google Scholar]
- Ricke, S.C. Perspectives on the use of organic acids and short chain fatty acids as antimicrobials. Poult. Sci. 2003, 82, 632–639. [Google Scholar] [CrossRef]
- Chung, Y.-C.; Su, Y.-P.; Chen, C.-C.; Jia, G.; Wang, H.; Wu, J.C.G.; Lin, J.-G. Relationship between antibacterial activity of chitosan and surface characteristics of cell wall. Acta Pharmacol. Sin. 2004, 25, 932–936. [Google Scholar]
- Je, J.; Kim, S.-K. Chitosan derivatives killed bacteria by disrupting the outer and inner membrane. J. Agric. Food Chem. 2006, 54, 6629–6633. [Google Scholar] [CrossRef]
- Li, D.F.; Thaler, R.C.; Nelssen, J.L.; Harmon, D.L.; Allee, G.L.; Weeden, T.L. Effect of fat sources and combinations on starter pig performance, nutrient digestibility and intestinal morphology. J. Anim. Sci. 1990, 68, 3694–3704. [Google Scholar] [CrossRef]
- Schiavone, A.; Dabbou, S.; De Marco, M.; Cullere, M.; Biasato, I.; Biasibetti, E.; Capucchio, M.T.; Bergagna, S.; Dezzutto, D.; Meneguz, M. Black soldier fly larva fat inclusion in finisher broiler chicken diet as an alternative fat source. Animal 2018, 12, 2032–2039. [Google Scholar] [CrossRef] [PubMed]
- Zeitz, J.O.; Fennhoff, J.; Kluge, H.; Stangl, G.I.; Eder, K. Effects of dietary fats rich in lauric and myristic acid on performance, intestinal morphology, gut microbes, and meat quality in broilers. Poult. Sci. 2015, 94, 2404–2413. [Google Scholar] [CrossRef] [PubMed]
- Secci, G.; Bovera, F.; Nizza, S.; Baronti, N.; Gasco, L.; Conte, G.; Serra, A.; Bonelli, A.; Parisi, G. Quality of eggs from Lohmann Brown Classic laying hens fed black soldier fly meal as substitute for soya bean. Animal 2018, 12, 2191–2197. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.C.; Kien, C.L.; Bouthillier, L.; Levy, E. Short-chain fatty acids: Ready for prime time? Nutr. Clin. Pract. 2006, 21, 351–366. [Google Scholar] [CrossRef]
- Coudray, C.; Tressol, J.C.; Gueux, E.; Rayssiguier, Y. Effects of inulin-type fructans of different chain length and type of branching on intestinal absorption and balance of calcium and magnesium in rats. Eur. J. Nutr. 2003, 42, 91–98. [Google Scholar] [CrossRef]
- Schiavone, A.; Dabbou, S.; Petracci, M.; Zampiga, M.; Sirri, F.; Biasato, I.; Gai, F.; Gasco, L. Black soldier fly defatted meal as a dietary protein source for broiler chickens: Effects on carcass traits, breast meat quality and safety. Animal 2019, 13, 2397–2405. [Google Scholar] [CrossRef]
- Cullere, M.; Tasoniero, G.; Giaccone, V.; Acuti, G.; Marangon, A.; Dalle Zotte, A. Black soldier fly as dietary protein source for broiler quails: Meat proximate composition, fatty acid and amino acid profile, oxidative status and sensory traits. Animal 2018, 12, 640–647. [Google Scholar] [CrossRef]
- Cullere, M.; Schiavone, A.; Dabbou, S.; Gasco, L.; Dalle Zotte, A. Meat quality and sensory traits of finisher broiler chickens fed with black soldier fly (Hermetia Illucens, L.) larvae fat as alternative fat source. Animals 2019, 9, 140. [Google Scholar] [CrossRef] [Green Version]
- Schiavone, A.; Cullere, M.; De Marco, M.; Meneguz, M.; Biasato, I.; Bergagna, S.; Dezzutto, D.; Gai, F.; Dabbou, S.; Gasco, L. Partial or total replacement of soybean oil by black soldier fly larvae (Hermetia illucens L.) fat in broiler diets: Effect on growth performances, feed-choice, blood traits, carcass characteristics and meat quality. Ital. J. Anim. Sci. 2017, 16, 93–100. [Google Scholar] [CrossRef] [Green Version]
- Al-Qazzaz, M.F.A.; Ismail, D.; Akit, H.; Idris, L.H. Effect of using insect larvae meal as a complete protein source on quality and productivity characteristics of laying hens. Rev. Bras. Zootec. 2016, 45, 518–523. [Google Scholar] [CrossRef] [Green Version]
- Bejaei, M.; Cheng, K.M. The effect of including full-fat dried black soldier fly larvae in laying hen diet on egg quality and sensory characteristics. J. Insects Food Feed 2020, 6, 305–314. [Google Scholar] [CrossRef]
| Protein Source * | CP % | CF % | Amino Acids % DM | |||||
|---|---|---|---|---|---|---|---|---|
| LYS | MET | THR | ARG | VAL | ILE | |||
| BSF | 49.47 | 18.11 | 2.71 | 0.87 | 1.95 | 2.47 | 3.11 | 2.21 |
| Soybean meal | 44.0 | 0.80 | 1.00 | 0.60 | 1.05 | 2.30 | 1.60 | 1.00 |
| Herring fish meal | 72.3 | 10.0 | 5.47 | 2.16 | 3.07 | 4.21 | 3.90 | 3.23 |
| Corn gluten | 62.0 | 2.50 | 1.03 | 1.49 | 2.00 | 1.82 | 2.78 | 2.45 |
| Sunflower meal | 32.0 | 1.10 | 2.69 | 0.62 | 1.72 | 3.14 | 2.07 | 1.96 |
| Canola seed meal | 38.0 | 3.80 | 1.94 | 0.71 | 1.53 | 2.08 | 1.76 | 1.37 |
| Sesame seed meal | 43.8 | 6.50 | 0.91 | 1.22 | 1.40 | 4.68 | 1.91 | 1.51 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abd El-Hack, M.E.; Shafi, M.E.; Alghamdi, W.Y.; Abdelnour, S.A.; Shehata, A.M.; Noreldin, A.E.; Ashour, E.A.; Swelum, A.A.; Al-Sagan, A.A.; Alkhateeb, M.; et al. Black Soldier Fly (Hermetia illucens) Meal as a Promising Feed Ingredient for Poultry: A Comprehensive Review. Agriculture 2020, 10, 339. https://doi.org/10.3390/agriculture10080339
Abd El-Hack ME, Shafi ME, Alghamdi WY, Abdelnour SA, Shehata AM, Noreldin AE, Ashour EA, Swelum AA, Al-Sagan AA, Alkhateeb M, et al. Black Soldier Fly (Hermetia illucens) Meal as a Promising Feed Ingredient for Poultry: A Comprehensive Review. Agriculture. 2020; 10(8):339. https://doi.org/10.3390/agriculture10080339
Chicago/Turabian StyleAbd El-Hack, Mohamed E., Manal E. Shafi, Wed Y. Alghamdi, Sameh A. Abdelnour, Abdelrazeq M. Shehata, Ahmed E. Noreldin, Elwy A. Ashour, Ayman A. Swelum, Ahmed A. Al-Sagan, Mazen Alkhateeb, and et al. 2020. "Black Soldier Fly (Hermetia illucens) Meal as a Promising Feed Ingredient for Poultry: A Comprehensive Review" Agriculture 10, no. 8: 339. https://doi.org/10.3390/agriculture10080339






