Pig Organ Lesions Recorded in Different Abattoirs: A Statistical Approach to Assess the Comparability of Prevalence
Abstract
1. Introduction
2. Materials and Methods
2.1. Abattoir Data
2.2. Preliminary Examination of the Data Structure
2.3. Improving Data Quality and Structure
2.4. Statistical Model
2.5. Statistical Analysis
3. Results
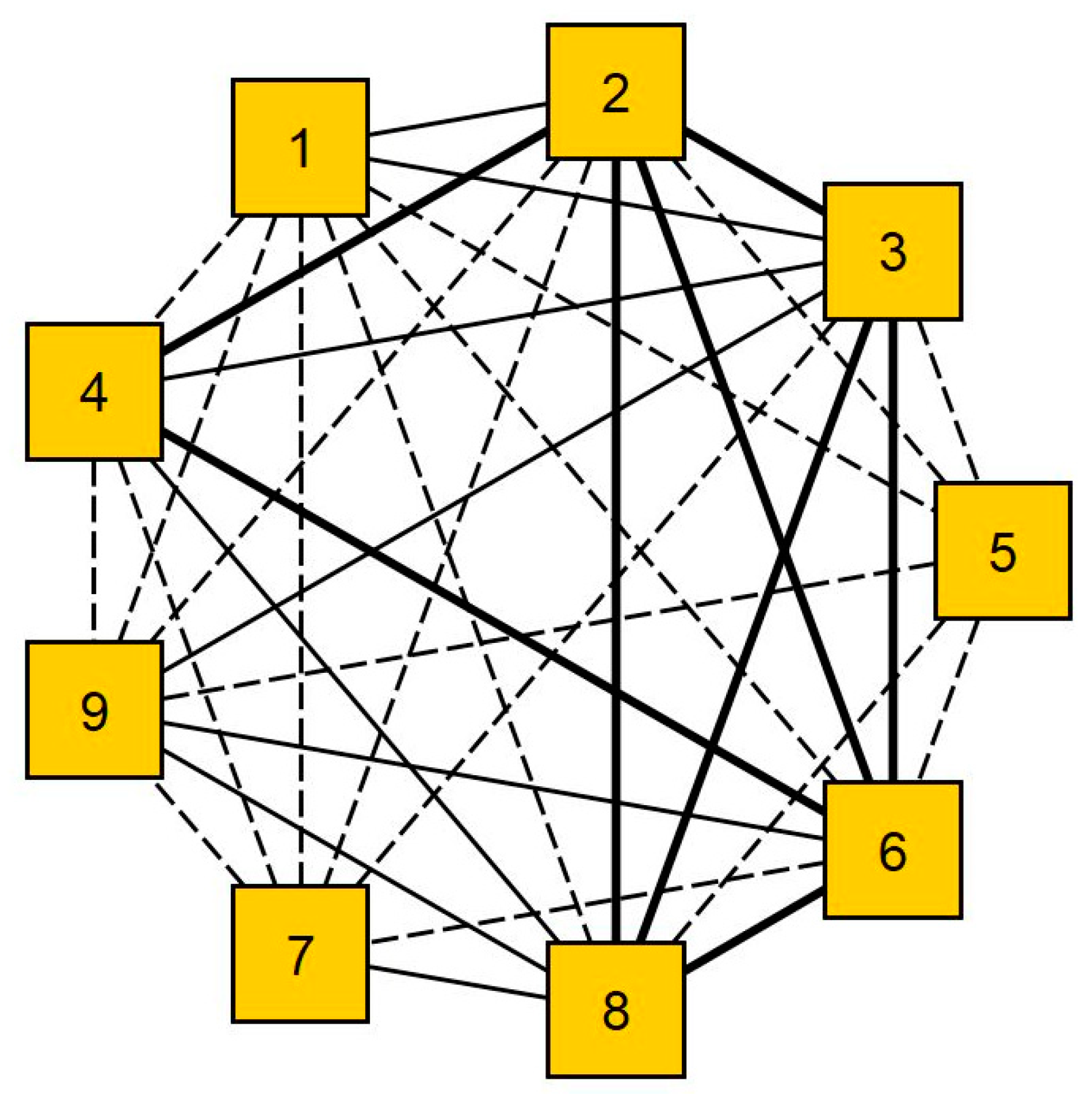
3.1. Network Analysis of the Data Structure
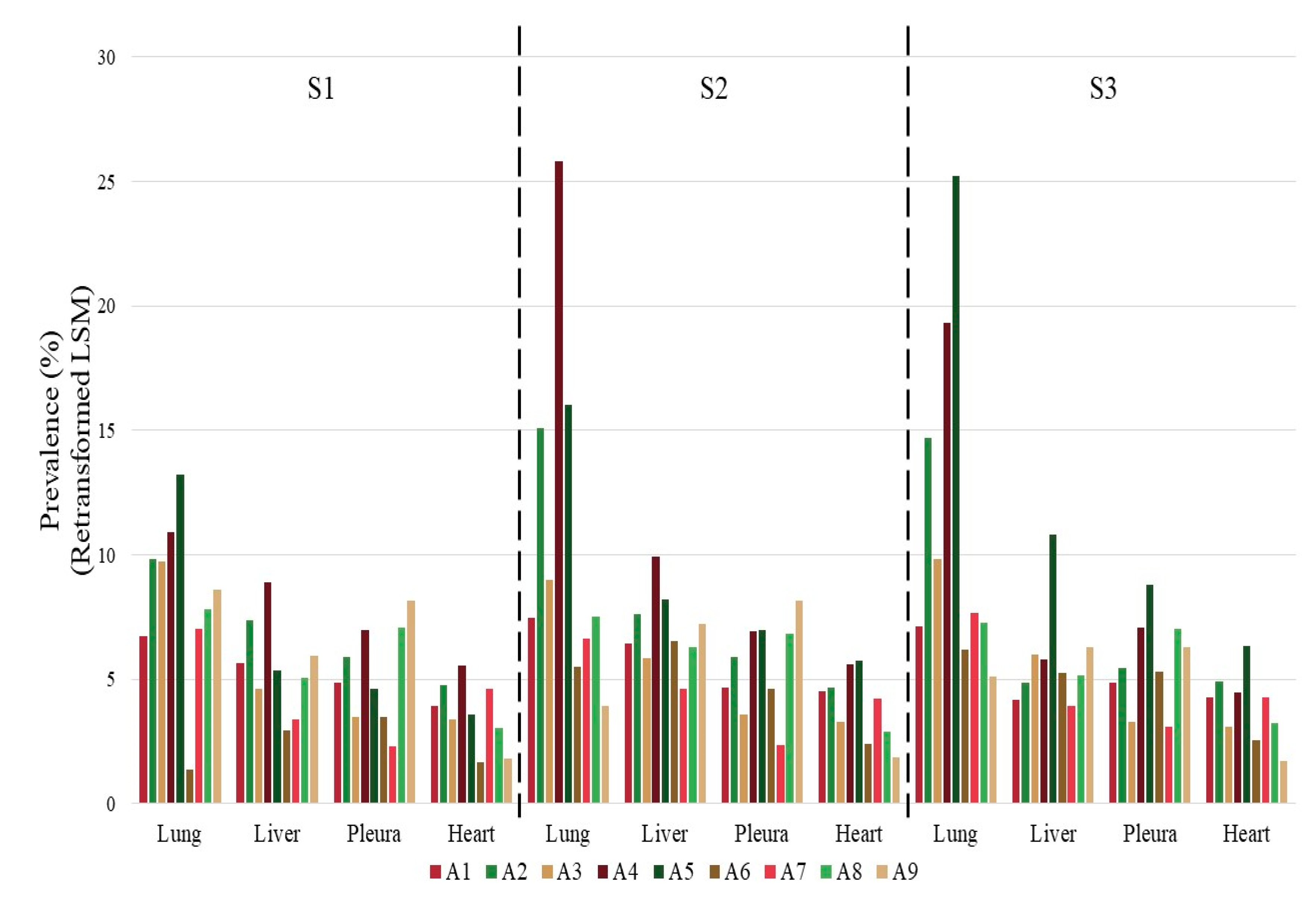
3.2. Intra-Abattoir Variation
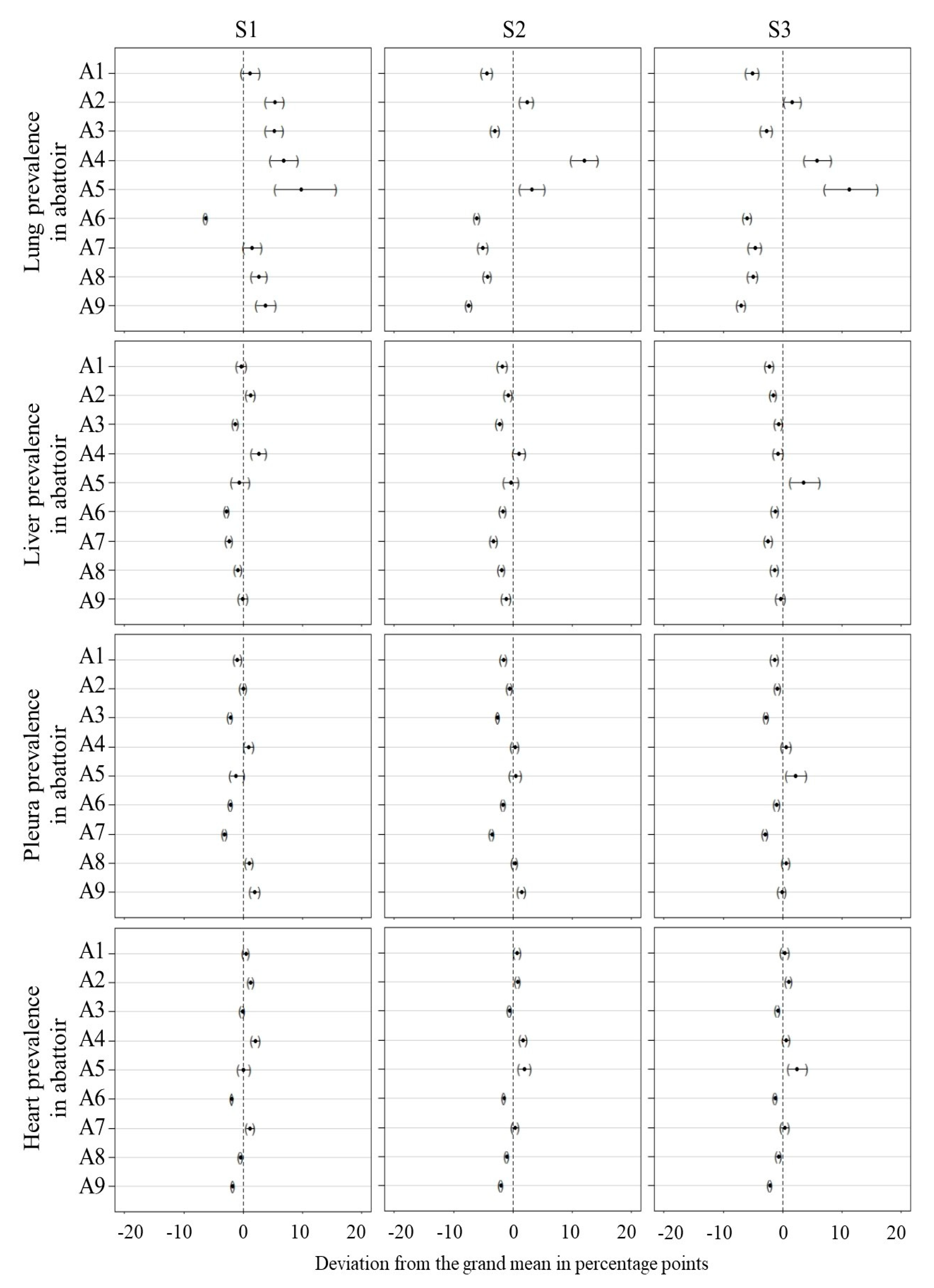
3.3. Inter-Abattoir Variation
3.4. Grand Mean Test of Significance
4. Discussion
4.1. Data Quality and Structure
4.2. Data Modeling
4.3. Intra-Abattoir Variation
4.4. Inter-Abattoir Variation
4.5. Grand Mean as Reference Value
4.6. Applicability of the Grand Mean Test
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Harley, S.; More, S.; Boyle, L.; Connell, N.O.; Hanlon, A. Good animal welfare makes economic sense: Potential of pig abattoir meat inspection as a welfare surveillance tool. Ir. Vet. J. 2012, 65, 11. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, M.; Bergman, P.; Fredriksson-Ahomaa, M.; Virtala, A.-M.; Munsterhjelm, C.; Valros, A.; Oliviero, C.; Peltoniemi, O.; Hälli, O. Sow mortality is associated with meat inspection findings. Livest. Sci. 2018, 208, 90–95. [Google Scholar] [CrossRef]
- Vial, F. Editorial: Slaughterhouses as Sources of Data for Animal Health Intelligence. Front. vet. sci. 2019, 5, 332. [Google Scholar] [CrossRef] [PubMed]
- Merks, J.W.M.; Mathur, P.K.; Knol, E.F. New phenotypes for new breeding goals in pigs. Animal 2012, 6, 535–543. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards (BIOHAZ), EFSA Panel on Contaminants in the Food Chain (CONTAM) and EFSA Panel on Animal Health and Welfare (AHAW). Scientific Opinion on the public health hazards to be covered by inspection of meat (swine). EFSA J. 2011, 9, 1–198. [Google Scholar]
- Horst, A.; Gertz, M.; Krieter, J. Challenges and opportunities of using meat inspection data to improve pig health traits by breeding: A review. Livest. Sci. 2019, 221, 155–162. [Google Scholar] [CrossRef]
- Dickhaus, C.P.; Meemken, D.; Blaha, T. Attempts to Quantify the Health Status of Pig Herds: Developing and validating a Herd Health Score (HHS). In Sustainable Animal Production: The challenges and Potential Developments for Professional Farming; Aland, A., Ed.; Wageningen Acad. Publ.: Wageningen, The Netherlands, 2009; pp. 191–201. [Google Scholar]
- van Staaveren, N.; Doyle, B.; Manzanilla, E.G.; Calderón Díaz, J.A.; Hanlon, A.; Boyle, L.A. Validation of carcass lesions as indicators for on-farm health and welfare of pigs. J. Anim. Sci. 2017, 95, 1528–1536. [Google Scholar] [CrossRef]
- Bundesministerium für Ernährung, Landwirtschaft und Verbraucherschutz. Allgemeine Verwaltungsvorschrift über die Durchführung der amtlichen Überwachung der Einhaltung von Hygienevorschriften für Lebensmittel und zum Verfahren zur Prüfung von Leitlinien für eine gute Verfahrenspraxis; AVV LmH: Berlin, Germany, 2009. [Google Scholar]
- Blaha, T. Stand der Umsetzung des Lebensmittelrechts in der Schweineproduktion. In 4. Leipziger Tierärztekongress, Schwerpunkt Schwein; Aschenbach, J.R., Gäbel, G., Daugschies, A.H., Eds.; Leipziger Blaue Hefte: Leipzig, Germany, 2008; pp. 549–553. [Google Scholar]
- Nienhaus, F.; Meemken, D.; Schoneberg, C.; Hartmann, M.; Kornhoff, T.; May, T.; Heß, S.; Kreienbrock, L.; Wendt, A. Health scores for farmed animals: Screening pig health with register data from public and private databases. PLoS ONE 2020, 15, e0228497. [Google Scholar] [CrossRef]
- Enøe, C.; Christensen, G.; Andersen, S.; Willeberg, P. The need for built-in validation of surveillance data so that changes in diagnostic performance of post-mortem meat inspection can be detected. Prev. Vet. Med. 2003, 57, 117–125. [Google Scholar]
- Hulsegge, B.; Greef de, K.H. A time-series approach for clustering farms based on slaughterhouse health aberration data. Prev. vet. med. 2018, 153, 64–70. [Google Scholar] [CrossRef]
- Alban, L.; Petersen, J.V.; Busch, M.E. A comparison between lesions found during meat inspection of finishing pigs raised under organic/free-range conditions and conventional, indoor conditions. Porcine Health Manag. 2015, 1. [Google Scholar] [CrossRef] [PubMed]
- Schleicher, C.; Scheriau, S.; Kopacka, I.; Wanda, S.; Hofrichter, J.; Köfer, J. Analysis of the variation in meat inspection of pigs using variance partitioning. Prev. Vet. Med. 2013, 111, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Scollo, A.; Gottardo, F.; Contiero, B.; Mazzoni, C.; Leneveu, P.; Edwards, S.A. Benchmarking of pluck lesions at slaughter as a health monitoring tool for pigs slaughtered at 170kg (heavy pigs). Prev. Vet. Med. 2017, 144, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Meyns, T.; van Steelant, J.; Rolly, E.; Dewulf, J.; Haesebrouck, F.; Maes, D. A cross-sectional study of risk factors associated with pulmonary lesions in pigs at slaughter. Vet. J. 2011, 187, 388–392. [Google Scholar] [CrossRef]
- Merialdi, G.; Dottori, M.; Bonilauri, P.; Luppi, A.; Gozio, S.; Pozzi, P.; Spaggiari, B.; Martelli, P. Survey of pleuritis and pulmonary lesions in pigs at abattoir with a focus on the extent of the condition and herd risk factors. Vet. J. 2012, 193, 234–239. [Google Scholar] [CrossRef]
- Blaha, T. Erfassung von Schlachttierbefunden für die Qualitätssicherung bei der Fleischproduktion. Deutsche tierärztliche Wochenschrift 1994, 101, 264–267. [Google Scholar]
- Bonde, M.; Toft, N.; Thomsen, P.T.; Sørensen, J.T. Evaluation of sensitivity and specificity of routine meat inspection of Danish slaughter pigs using Latent Class Analysis. Prev. Vet. Med. 2010, 94, 165–169. [Google Scholar] [CrossRef]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S.; Christensen, R.H.B.; Singmann, H.; Dai, B.; Scheipl, F.; Grothendieck, G.; Green, P.; et al. Package ′lme4′: Linear Mixed-Effects Models using ′Eigen′ and S4. Available online: https://cran.r-project.org/web/packages/lme4/lme4.pdf (accessed on 7 April 2020).
- Steinmann, T.; Blaha, T.; Meemken, D. A simplified evaluation system of surface-related lung lesions of pigs for official meat inspection under industrial slaughter conditions in Germany. BMC Vet. Res. 2014, 10, 98. [Google Scholar] [CrossRef]
- Correia-Gomes, C.; Smith, R.P.; Eze, J.I.; Henry, M.K.; Gunn, G.J.; Williamson, S.; Tongue, S.C. Pig Abattoir Inspection Data: Can It Be Used for Surveillance Purposes? PLoS ONE 2016, 11, e0161990. [Google Scholar] [CrossRef]
- Stärk, K.D.C.; Alonso, S.; Dadios, N.; Dupuy, C.; Ellerbroek, L.; Georgiev, M.; Hardstaff, J.; Huneau-Salaün, A.; Laugier, C.; Mateus, A.; et al. Strengths and weaknesses of meat inspection as a contribution to animal health and welfare surveillance. Food Control 2014, 39, 154–162. [Google Scholar] [CrossRef]
- Hoischen-Taubner, S.; Blaha, T.; Werner, C.; Sundrum, A. Zur Reproduzierbarkeit der Befunderfassung am Schlachthof für Merkmale der Tiergesundheit. Repeatability of anatomical-pathological findings at the abattoir for characteristics of animal health. J. Food Saf.Food Qual. 2011, 62, 82–87. [Google Scholar]
- Eckhardt, P.; Fuchs, K.; Kornberger, B.; Köfer, J. Untersuchungen über die Raliabilität der im Zuge der Fleischuntersuchung erhobenen Befunde von Schlachtschweinen. Wiener Tierärztliche Monatsschrift. Available online: https://center.ssi.at/smart_users/uni/user94/explorer/43/WTM/Archiv/2009/WTM_05-06-2009_Artikel_6.pdf (accessed on 1 November 2017).
- Teixeira, D.L.; Harley, S.; Hanlon, A.; O’Connell, N.E.; More, S.J.; Manzanilla, E.G.; Boyle, L.A. Study on the Association between Tail Lesion Score, Cold Carcass Weight, and Viscera Condemnations in Slaughter Pigs. Front. Vet. Sci. 2016, 3, 24. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Horst, A.; Gertz, M.; Hasler, M.; Krieter, J. Pig Organ Lesions Recorded in Different Abattoirs: A Statistical Approach to Assess the Comparability of Prevalence. Agriculture 2020, 10, 319. https://doi.org/10.3390/agriculture10080319
Horst A, Gertz M, Hasler M, Krieter J. Pig Organ Lesions Recorded in Different Abattoirs: A Statistical Approach to Assess the Comparability of Prevalence. Agriculture. 2020; 10(8):319. https://doi.org/10.3390/agriculture10080319
Chicago/Turabian StyleHorst, Ariane, Marvin Gertz, Mario Hasler, and Joachim Krieter. 2020. "Pig Organ Lesions Recorded in Different Abattoirs: A Statistical Approach to Assess the Comparability of Prevalence" Agriculture 10, no. 8: 319. https://doi.org/10.3390/agriculture10080319
APA StyleHorst, A., Gertz, M., Hasler, M., & Krieter, J. (2020). Pig Organ Lesions Recorded in Different Abattoirs: A Statistical Approach to Assess the Comparability of Prevalence. Agriculture, 10(8), 319. https://doi.org/10.3390/agriculture10080319




