Soil Fertilization with Urea Has Little Effect on Seed Quality but Reduces Soil N2O Emissions from a Hemp Cultivation
Abstract
1. Introduction
2. Materials and Methods
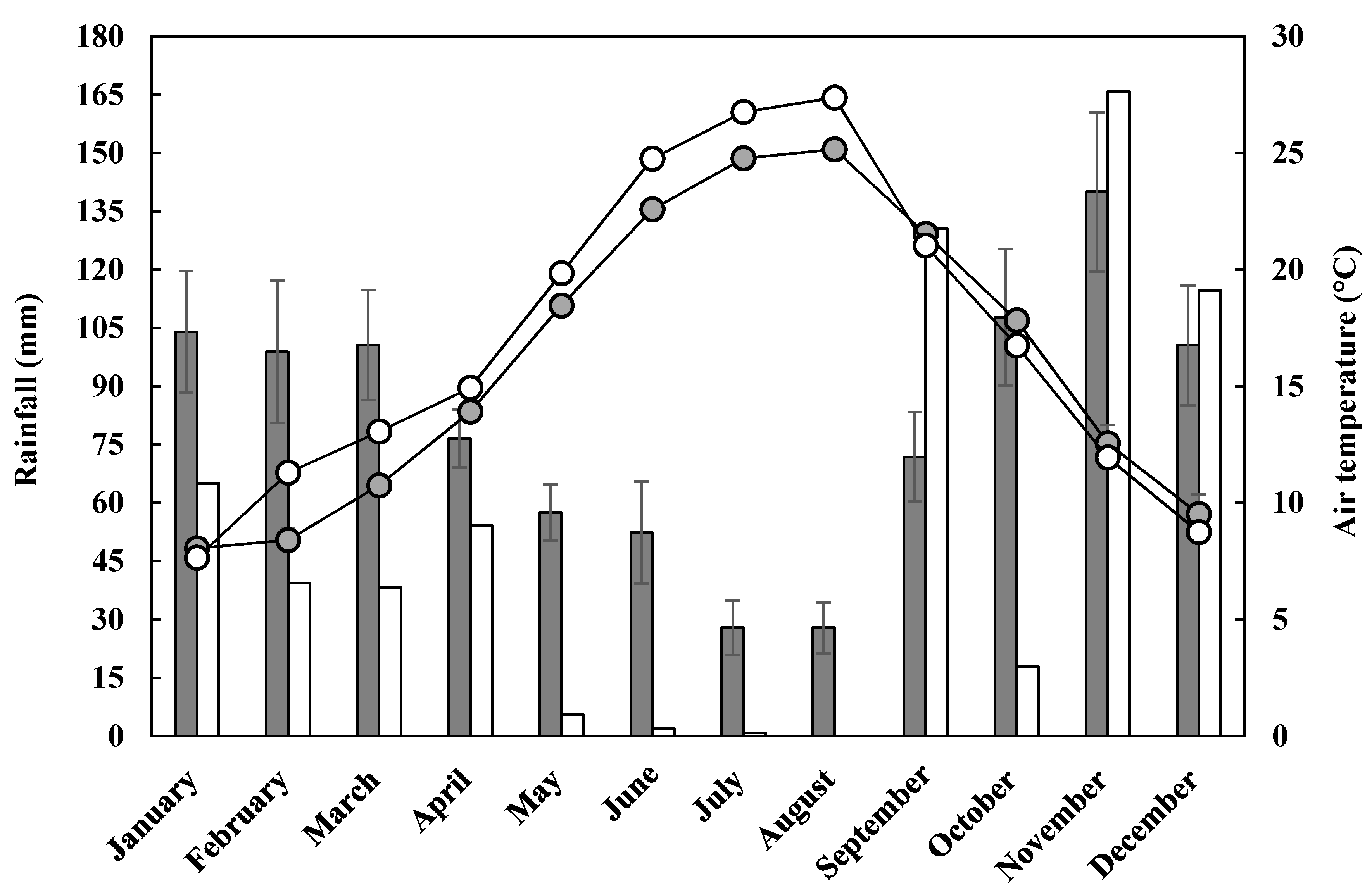
2.1. Experimental Site, Plant Material, and Crop Management
2.2. Seed Proximate Composition
2.3. Fatty Acid Methyl Esters (FAMEs) Preparation
2.4. FAMEs Gas Chromatographic Analysis
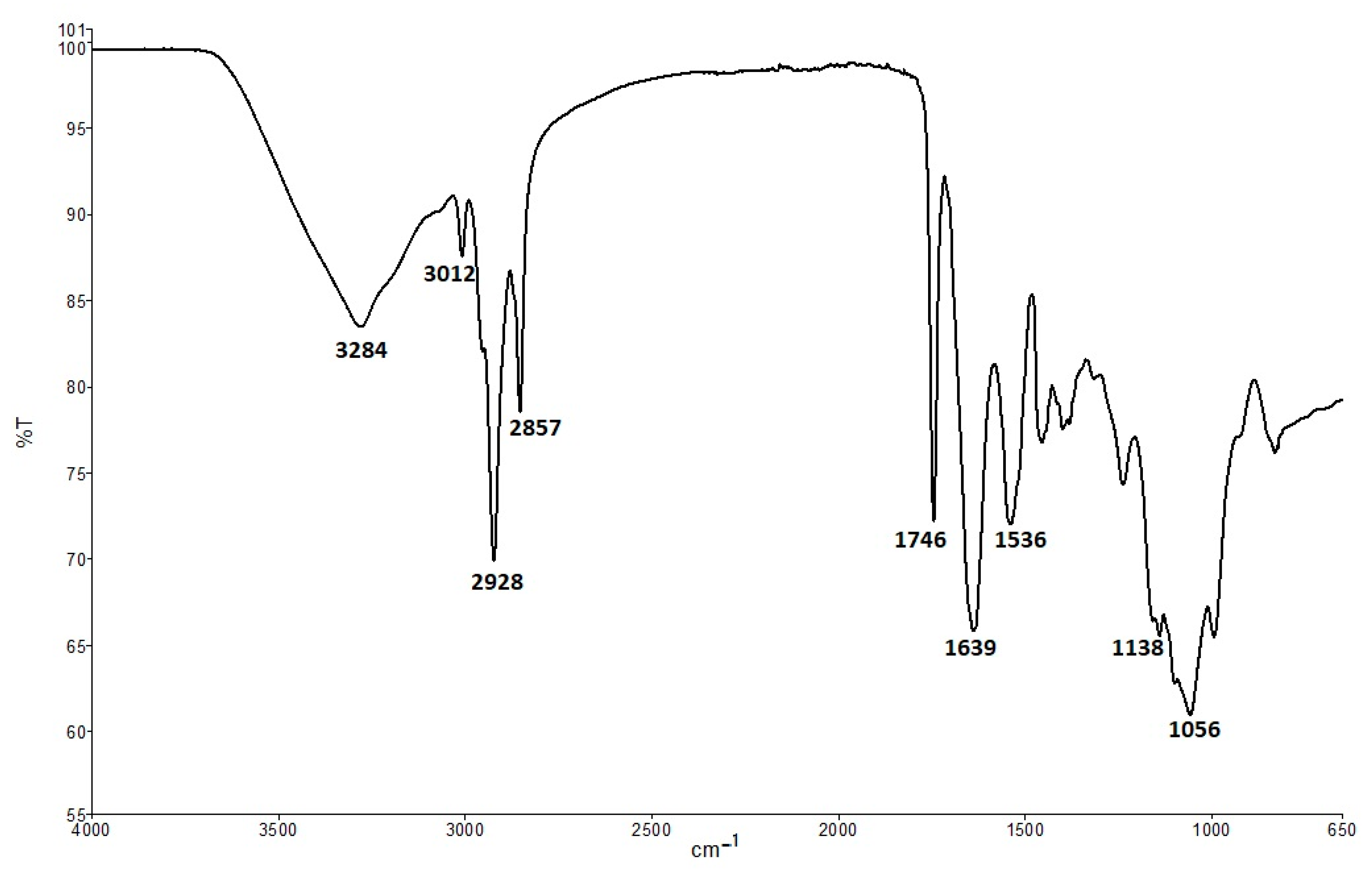
2.5. Attenuated Total Reflection-Fourier Transform Infrared (ATR-FTIR) Spectra
2.6. Biometrical Determinations
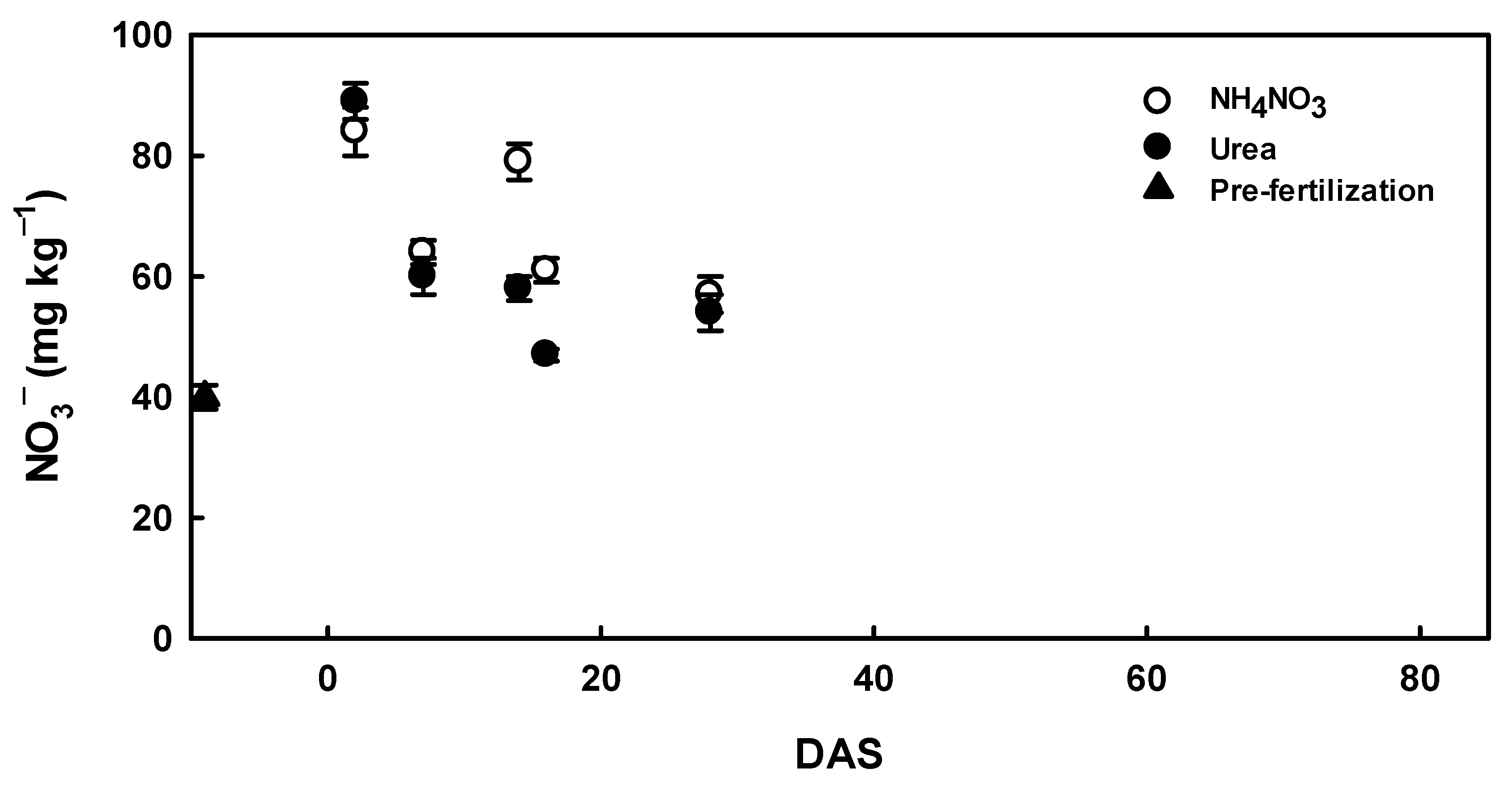
2.7. Soil and Gas Measurements
2.8. Statistical Analysis
3. Results
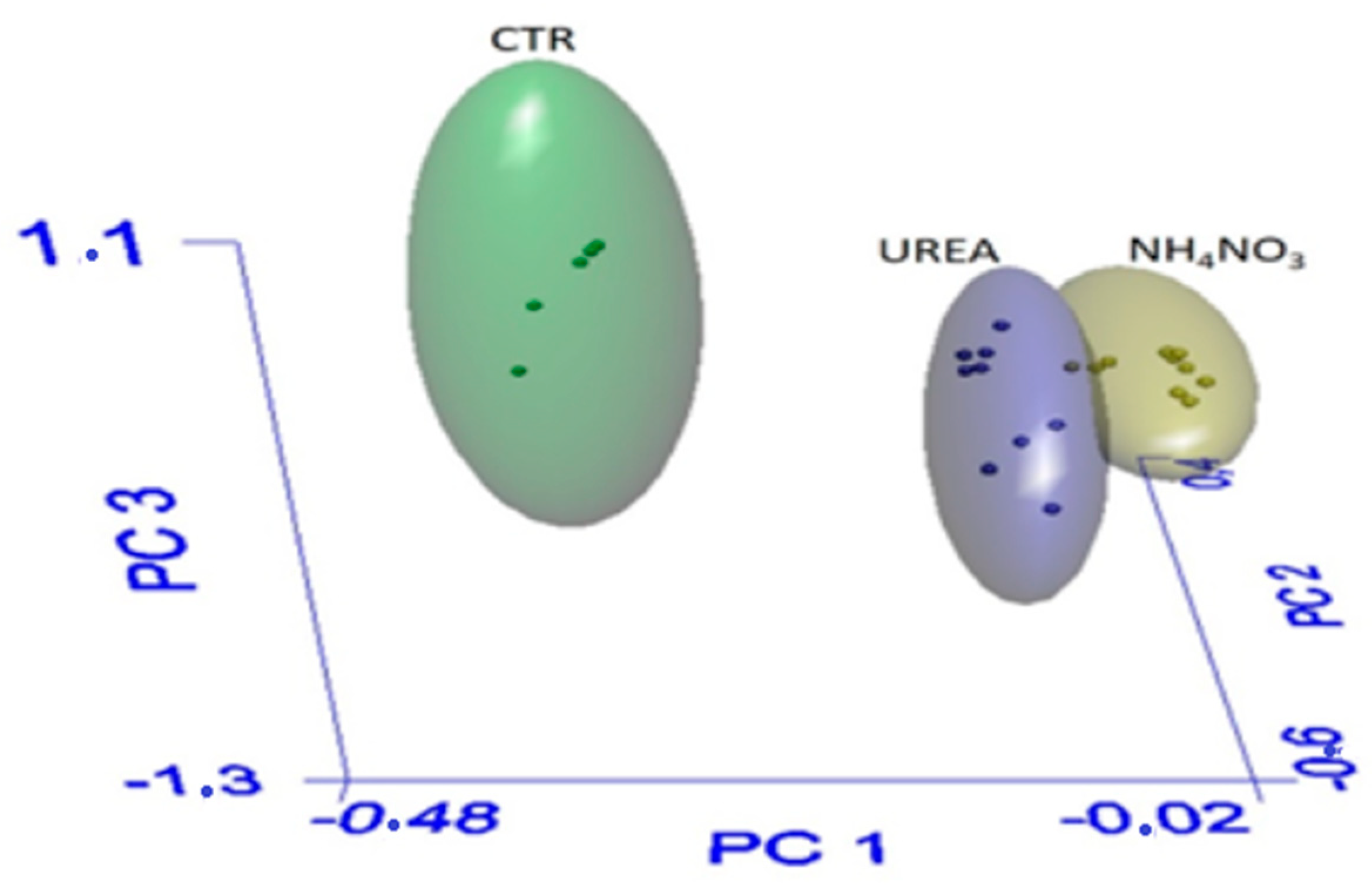
3.1. Seed Quality
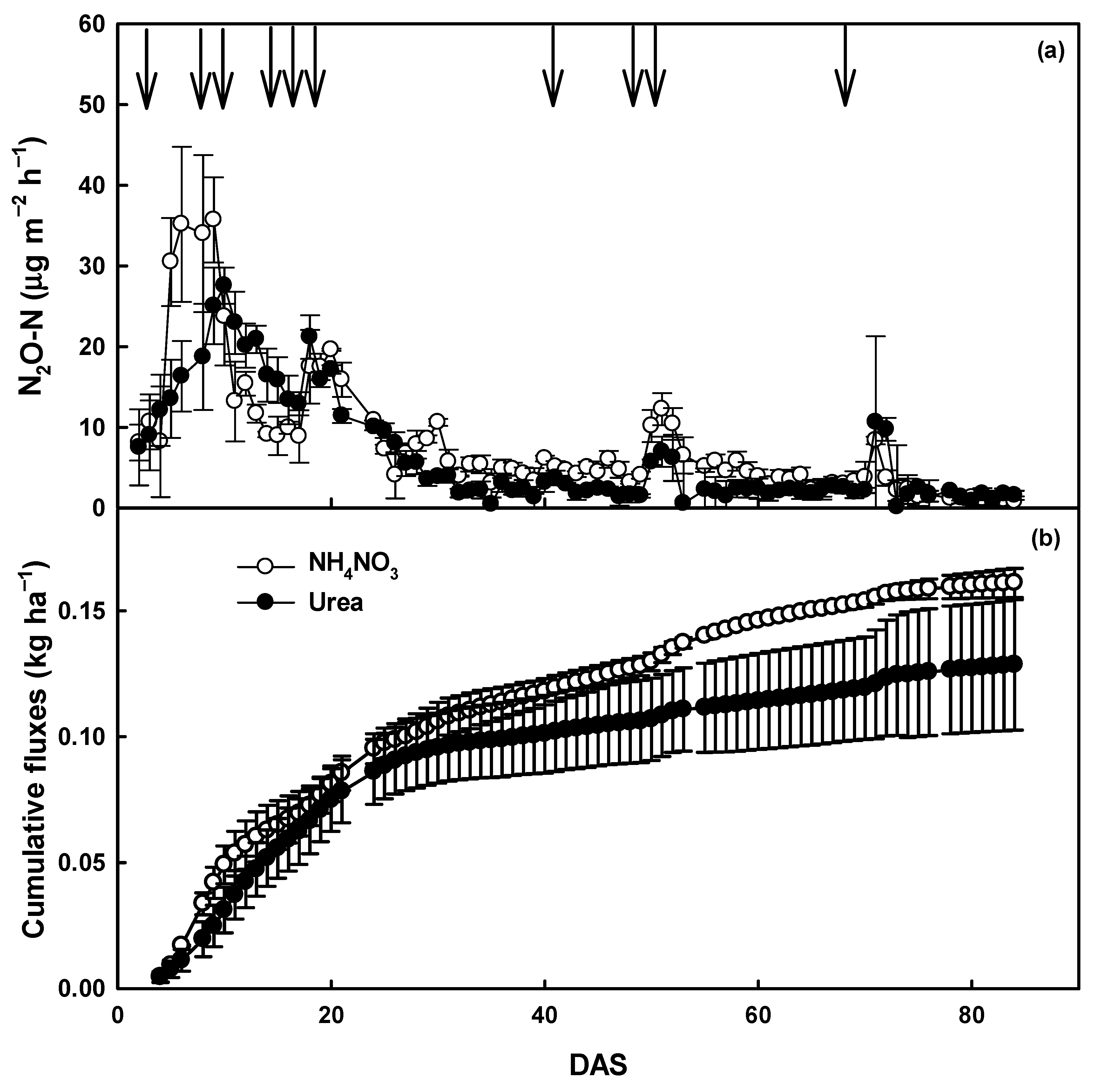
3.2. Agronimic Traits and Soil N2O Emission
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Salentijn, E.M.J.; Zhang, Q.; Amaducci, S.; Yang, M.; Trindade, L.M. New developments in fiber hemp (Cannabis sativa L.) breeding. Ind. Crops Prod. 2015, 68, 32–41. [Google Scholar] [CrossRef]
- Amaducci, S.; Scordia, D.; Liu, F.H.; Zhang, Q.; Guo, H.; Testa, G.; Cosentino, S.L. Key cultivation techniques for hemp in Europe and China. Ind. Crops Prod. 2015, 68, 2–16. [Google Scholar] [CrossRef]
- Tang, K.; Struik, P.C.; Yin, X.; Thouminot, C.; Bjelková, M.; Stramkale, V.; Amaducci, S. Comparing hemp (Cannabis sativa L.) cultivars for dual-purpose production under contrasting environments. Ind. Crops Prod. 2016, 87, 33–44. [Google Scholar] [CrossRef]
- Adesina, I.; Bhowmik, A.; Sharma, H.; Shahbazi, A. A Review on the Current State of Knowledge of Growing Conditions, Agronomic Soil Health Practices and Utilities of Hemp in the United States. Agriculture 2020, 10, 129. [Google Scholar] [CrossRef]
- Amaducci, S.; Venturi, G. Hemp Sys: A sustainable production system for hemp textiles. In Proceedings of the International South Europe Symposium “Non-Food Crops: From Agriculture to Industry”, Bologna, Italy, 16–17 May 2003; pp. 15–16. [Google Scholar]
- Baldini, M.; Ferfuia, C.; Piani, B.; Sepulcri, A.; Dorigo, G.; Zuliani, F.; Danuso, F.; Cattivello, C. The Performance and Potentiality of Monoecious Hemp (Cannabis sativa L.) Cultivars as a Multipurpose Crop. Agronomy 2018, 8, 162. [Google Scholar] [CrossRef]
- Siano, F.; Moccia, S.; Picariello, G.; Russo, G.L.; Sorrentino, G.; Di Stasio, M.; La Cara, F.; Volpe, M.G. Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L.). Molecules 2019, 24, 83. [Google Scholar] [CrossRef]
- Suriyong, S.; Krittigamas, N.; Pinmanee, S.; Punyalue, A.; Vearasilp, S. Influence of storage conditions on change of hemp seed quality. Agric. Agric. Sci. Procedia 2015, 5, 170–176. [Google Scholar] [CrossRef]
- Finnan, J.; Burke, B. Nitrogen fertilization to optimize the greenhouse gas balance of hemp crops grown for biomass. Glob. Chang. Biol. Bioenergy 2013, 5, 701–712. [Google Scholar] [CrossRef]
- IPCC. N2O emissions from managed soils and CO2 emissions from lime and urea applications. In IPCC Guidelines for National Greenhouse Gas Inventories; Eggleston, H.S., Buendia, L., Miwa, K., Ngara, T., Tanabe, K., Eds.; IGES: Kanagawa, Japan, 2006; pp. 11.1–11.54. [Google Scholar]
- Rafique, R.; Hennessy, D.; Kiely, G. Nitrous oxide emission from grazed grassland under different management systems. Ecosystems 2011, 14, 563–582. [Google Scholar] [CrossRef]
- Sainju, U.M.; Stevens, W.B.; Caesar-Tonthat, T.; Liebig, M.A. Soil greenhouse gas emissions affected by irrigation, tillage, crop rotation, and nitrogen fertilization. J. Environ. Qual. 2012, 41, 1774–1786. [Google Scholar] [CrossRef]
- Struik, P.C.; Amaducci, S.; Bullard, M.J.; Stutterheim, N.C.; Venturi, G.; Cromack, H.T.H. Agronomy of fibre hemp (Cannabis sativa L.) in Europe. Ind. Crops Prod. 2000, 11, 107–118. [Google Scholar] [CrossRef]
- Aubin, M.P.; Seguin, P.; Vanasse, A.; Tremblay, G.F.; Mustafa, A.F.; Charron, J.B. Industrial Hemp Response to Nitrogen, Phosphorus, and Potassium Fertilization. Crop Forage Turfgrass Manag. 2015, 1, 1–10. [Google Scholar] [CrossRef]
- Vitale, L.; Polimeno, F.; Ottaiano, L.; Maglione, G.; Tedeschi, A.; De Marco, A.; Di Tommasi, P.; Mori, M.; Magliulo, V. Fertilizer type influences tomato yield and soil N2O emissions. Plant. Soil Environ. 2017, 63, 105–110. [Google Scholar]
- Vitale, L.; Tedeschi, A.; Polimeno, F.; Ottaiano, L.; Maglione, G.; Arena, C.; De Marco, A.; Magliulo, V. Water regime affects soil N2O emission and tomato yield grown under different types of fertilisers. Ital. J. Agron. 2018, 13, 74–79. [Google Scholar] [CrossRef]
- Dane, J.H.; Topp, C.G. Part 4: Physical Methods. In Methods of Soil Analysis; Dane, J.H., Topp, C.G., Eds.; Soil Science Society of America: Madison, WI, USA, 2002. [Google Scholar]
- Sparks, D.L.; Page, A.L.; Helmke, P.A.; Loeppert, R.H.; Soltanpour, P.N.; Tabatabai, M.A.; Johnston, C.T.; Summer, M.E. Part 3 Chemical Methods, 5.3. In Methods of Soil Analysis; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Summer, M.E., Eds.; Soil Science Society of America, Inc.: Madison, WI, USA, 1996. [Google Scholar]
- Vera, C.L.; Malhi, S.S.; Raney, J.P.; Wang, Z.H. The effect of N and P fertilization on growth, seed yield and quality of industrial hemp in the Parkland region of Saskatchewan. Can. J. Plant. Sci. 2004, 84, 939–947. [Google Scholar] [CrossRef]
- AOAC International. Official methods of analysis of AOAC international, 20th ed.; Latimer, G.W.L., Jr., Ed.; AOAC International: Rockville, MD, USA, 2016. [Google Scholar]
- Siano, F.; Addeo, F.; Volpe, M.G.; Paolucci, M.; Picariello, G. Oxidative stability of pomegranate (Punica granatum L.) seed oil to simulated gastric conditions and thermal stress. J. Agric. Food Chem. 2016, 64, 8369–8378. [Google Scholar] [CrossRef]
- Van de Voort, F.R. Fourier transform infrared spectroscopy applied to food analysis. Food Res. Int. 1992, 25, 397–403. [Google Scholar] [CrossRef]
- Wilson, R.H.; Tapp, H.S. Mid-infrared spectroscopy for food analysis: Recent new applications and relevant developments in sample presentation methods. Trends Analyt. Chem. 1999, 18, 87–93. [Google Scholar] [CrossRef]
- IPCC. Summary for Policymakers. In Climate Change 2007: Mitigation. Contribution of Working Group III to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Metz, B., Davidson, O.R., Bosch, P.R., Dave, R., Meyer, L.A., Eds.; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Nikonenco, N.A.; Buslov, D.K.; Sushko, N.I.; Zhbankov, R.G. Investigation of stretching vibrations of glycosidic linkages in disaccharides and polysaccarides with use of IR spectra deconvolution. Biopolymers 2000, 57, 257–262. [Google Scholar] [CrossRef]
- Rathke, G.W.; Christen, O.; Diepenbrock, W. Effects of nitrogen source and rate on productivity and quality of winter oilseed rape (Brassica napus L.) grown in different crop rotations. Field Crop. Res. 2005, 94, 103–113. [Google Scholar] [CrossRef]
- El-Murtada Hassan Amin, M. Effect of different nitrogen sources on growth, yield and quality of fodder maize (Zea mays L.). J. Saudi Soc. Agric. Sci. 2011, 10, 17–23. [Google Scholar]
- De, M.; Moore, A.D.; Mikkelsen, R.L. In-season Accumulation and Partitioning of Macronutrients and Micronutrients in Irrigated Sugar Beet Production. J. Sugar Beet Res. 2019, 56, 54–78. [Google Scholar]
- Garnica, M.; Houdusse, F.; Yvin, J.C.; Garcia-Mina, J.M. Nitrate modifies urea root uptake and assimilation in wheat seedlings. J. Sci. Food Agric. 2009, 89, 55–62. [Google Scholar] [CrossRef]
- Zanin, L.; Zamboni, A.; Monte, R.; Tomasi, N.; Varanini, Z.; Cesco, S.; Pinton, R. Transcriptomic analysis highlights reciprocal interactions of urea and nitrate for nitrogen acquisition by maize roots. Plant. Cell Physiol. 2015, 56, 532–548. [Google Scholar] [CrossRef]
- Arkoun, M.; Sarda, X.; Jannin, L.; Laîné, P.; Etienne, P.; Garcia-Mina, J.M.; Yvin, J.C.; Ourry, A. Hydroponics versus field lysimeter studies of urea, ammonium and nitrate uptake by oilseed rape (Brassica napus L.). J. Exp. Bot. 2012, 63, 5245–5258. [Google Scholar] [CrossRef]
- Pinton, R.; Tomasi, N.; Zanin, L. Molecular and physiological interactions of urea and nitrate uptake in plants. Plant. Signal. Behav. 2016, 11, e1076603. [Google Scholar] [CrossRef]
- Snyder, C.S.; Bruulsema, T.W.; Jensen, T.L.; Fixen, P.E. Review of greenhouse gas emissions from crop production systems and fertilizer management effects. Agric. Ecosyst. Environ. 2009, 133, 247–266. [Google Scholar] [CrossRef]
- Senila, L.; Neag, E.; Cadar, O.; Kovacs, M.H.; Becze, A.; Senila, M. Chemical, Nutritional and Antioxidant Characteristics of Different Food Seeds. Appl. Sci. 2020, 10, 1589. [Google Scholar] [CrossRef]
- Orsavova, J.; Misurcova, L.; Ambrozova, J.V.; Vicha, R.; Mlcek, J. Fatty Acids Composition of Vegetable Oils and Its Contribution to Dietary Energy Intake and Dependence of Cardiovascular Mortality on Dietary Intake of Fatty Acids. Int. J. Mol. Sci. 2015, 16, 12871–12890. [Google Scholar] [CrossRef]
- Calder, P.C.; Yaqoob, P. Omega-3 polyunsaturated fatty acids and human health outcomes. Biofactors 2009, 35, 266–272. [Google Scholar] [CrossRef]
- Richard, D.; Kefi, K.; Barbe, U.; Bausero, P.; Visioli, F. Polyunsaturated fatty acids as antioxidants. Pharmacol. Res. 2008, 57, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Gülçin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef] [PubMed]
- Bilal, S.; Khan, A.L.; Waqas, M.; Shahzad, R.; Kim, I.-D.; Lee, I.-J.; Shin, D.-H. Biochemical constituents and in vitro antioxidant and anticholinesterase potential of seeds from Native Korean Persimmon Genotypes. Molecules 2016, 21, 893. [Google Scholar] [CrossRef]
- Deferne, J.L.; Pate, D.W. Hemp seed oil: A source of valuable essential fatty acids. J. Int. Hemp Assoc. 1996, 3, 1–7. [Google Scholar]
| Carbohydrates (%) * | Protein (%) * | Lipids (%) * | Ash (%) * | Moisture (%) | |
|---|---|---|---|---|---|
| CTR | 42.8 ± 2.5 | 22.1 ± 1.9 | 30.1 ± 1.6 | 5.0 ± 0.3 | 6.4 ± 0.6 |
| NH4NO3 | 69.9 ± 1.7 a | 16.2 ± 1.4 a | 6.9 ± 0.7 a | 7.0 ± 0.6 a | 8.1 ± 1.3 |
| Urea | 72.5 ± 1.5 a | 14.7 ± 1.3 a | 6.6 ± 0.7 a | 6.2 ± 0.4 a | 7.7 ± 1.2 |
| Fatty Acid, Area % | CTR | NH4NO3 | Urea |
|---|---|---|---|
| Palmitic, C16:0 | 6.54 ± 0.36 | 11.37 ± 0.77 a | 11.9 ± 0.75 a |
| Palmitoleic, C16:1 | 0.12 ± 0.03 | 0.22 ± 0.03 a | 0.24 ± 0.04 a |
| Stearic, C18:0 | 2.69 ± 0.25 | 2.23 ± 0.2 | 2.16 ± 0.28 |
| Oleic, C18:1 ω-9c | 9.85 ± 0.44 | 10.8 ± 0.63 | 10.46 ± 0.38 |
| C18:1 ω-7c | 0.88 ± 0.13 | 1.53 ± 0.21 a | 1.61 ± 0.17 a |
| Linoleic, C18:2 ω-6c | 55.36 ± 1.68 | 51.12 ± 2.02 a | 51.76 ± 1.5 a |
| Arachidic, C20:0 | 0.7 ± 0.14 | 1.06 ± 0.28 | 1.03 ± 0.38 |
| γ-Linolenic, C18:3 ω-6 | 3.86 ± 0.46 | 4.55 ± 0.58 | 4.96 ± 0.57 |
| cis-11-Eicosenoic, C20:1 | 0.32 ± 0.09 | 0.51 ± 0.19 | 0.48 ± 0.13 |
| α-Linolenic, C18:3 ω-3 | 17.34 ± 1.35 | 11.1 ± 0.92 a | 11.37 ± 1.25 a |
| cis-11,14-Eicosadienoic, C20:2 | 1.28 ± 0.27 | 1.31 ± 0.27 | 1.26 ± 0.28 |
| ω3/ω6 Ratio | 0.38 | 0.31 | 0.31 |
| Σ SFA | 9.93 ± 0.46 | 14.66 ± 0.84 | 15.09 ± 0.89 |
| Σ MUFA | 11.17 ± 0.47 | 13.06 ± 0.69 | 12.79 ± 0.44 |
| Σ PUFA | 78.84 ± 2.22 | 68.08 ± 2.31 | 69.35 ± 2.05 |
| Material | CTR | Urea | NH4NO3 |
|---|---|---|---|
| CTR | - | 6.53 | 10.70 |
| Urea | - | - | 3.03 |
| Treatments | Shoot (g m−2) | Root (g m−2) | NUE (kg kg−1 N) | NEI (kg CO2eq kg−1) | EF (%) |
|---|---|---|---|---|---|
| NH4NO3 | 775.1 ± 66.3 a | 169.7 ± 16 a | 118 ± 12 a | 0.0053 ± 0.0006 a | 0.2 ± 0.01 a |
| Urea | 979.4 ± 106 b | 232 ± 26.4 b | 151 ± 16 b | 0.0035 ± 0.0009 b | 0.16 ± 0.03 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tedeschi, A.; Volpe, M.G.; Polimeno, F.; Siano, F.; Maglione, G.; Di Tommasi, P.; Vasca, E.; Magliulo, V.; Vitale, L. Soil Fertilization with Urea Has Little Effect on Seed Quality but Reduces Soil N2O Emissions from a Hemp Cultivation. Agriculture 2020, 10, 240. https://doi.org/10.3390/agriculture10060240
Tedeschi A, Volpe MG, Polimeno F, Siano F, Maglione G, Di Tommasi P, Vasca E, Magliulo V, Vitale L. Soil Fertilization with Urea Has Little Effect on Seed Quality but Reduces Soil N2O Emissions from a Hemp Cultivation. Agriculture. 2020; 10(6):240. https://doi.org/10.3390/agriculture10060240
Chicago/Turabian StyleTedeschi, Anna, Maria Grazia Volpe, Franca Polimeno, Francesco Siano, Giuseppe Maglione, Paul Di Tommasi, Ermanno Vasca, Vincenzo Magliulo, and Luca Vitale. 2020. "Soil Fertilization with Urea Has Little Effect on Seed Quality but Reduces Soil N2O Emissions from a Hemp Cultivation" Agriculture 10, no. 6: 240. https://doi.org/10.3390/agriculture10060240
APA StyleTedeschi, A., Volpe, M. G., Polimeno, F., Siano, F., Maglione, G., Di Tommasi, P., Vasca, E., Magliulo, V., & Vitale, L. (2020). Soil Fertilization with Urea Has Little Effect on Seed Quality but Reduces Soil N2O Emissions from a Hemp Cultivation. Agriculture, 10(6), 240. https://doi.org/10.3390/agriculture10060240








