Landscape Features Associated with Damage to Maize (Zea mays) Fields in Central México: A Comparison of Wind and Wildlife Damage
Abstract
1. Introduction
2. Materials and Methods
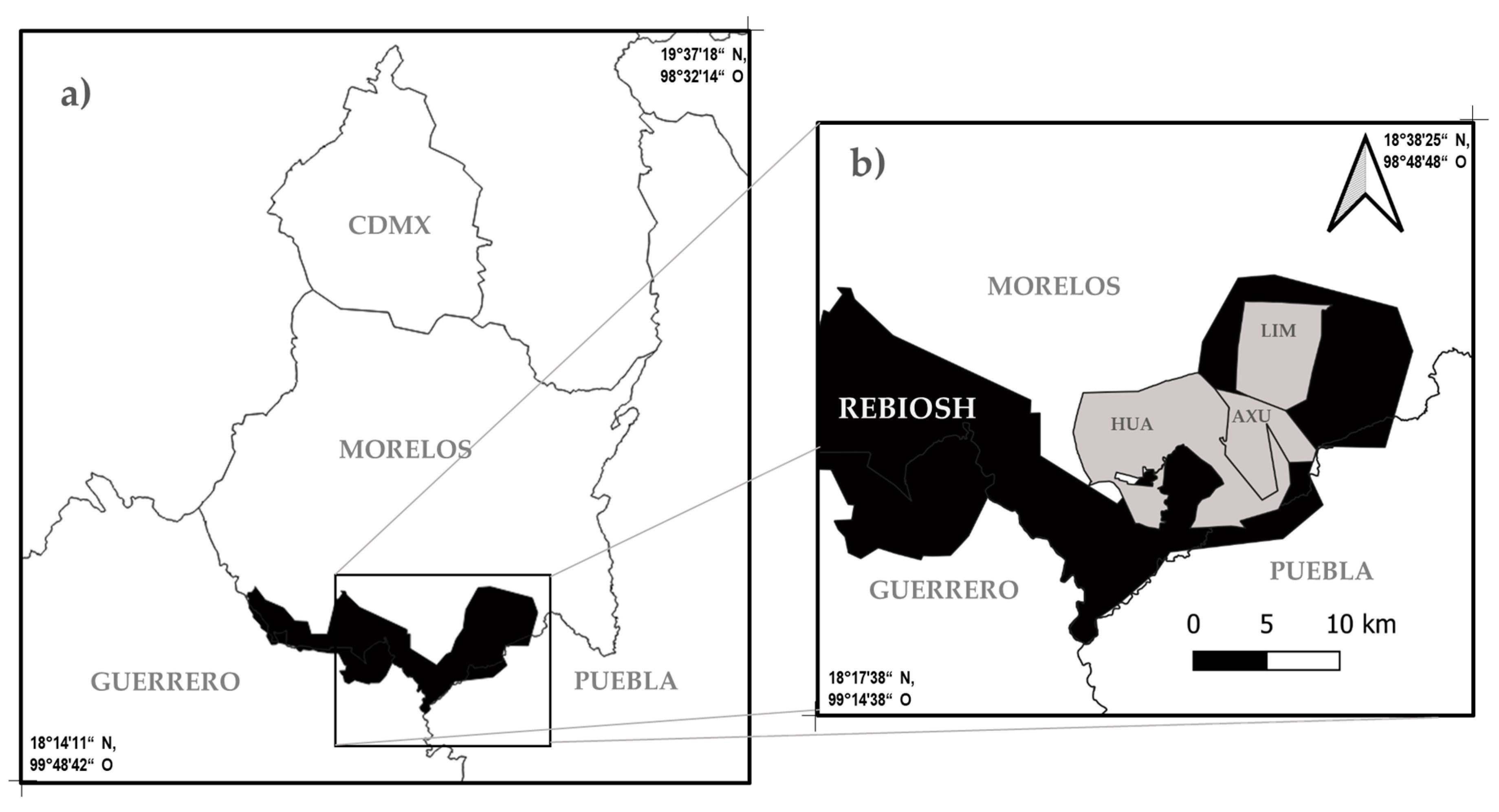
2.1. Study Area
2.2. Maize Damage
2.3. Landscape Features
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fahrig, L.; Baudry, J.; Brotons, L.; Burel, F.; Crist, T.O.; Fuller, R.J.; Sirami, C.; Siriwardena, G.M.; Martin, J.-L. Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol. Lett. 2010, 14, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Farina, A. Principles and Methods in Landscape Ecology, 1st ed.; Springer Science and Business Media: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- García-Marmolejo, G.; Chapa-Vargas, L.; Weber, M.; Sannwald, E.H. Landscape composition influences abundance patterns and habitat use of three ungulate species in fragmented secondary deciduous tropical forests, Mexico. Glob. Ecol. Conserv. 2015, 3, 744–755. [Google Scholar] [CrossRef]
- Green, B.; Simmons, E.; Woltjer, I. Landscape Conservation. Some Steps towards a New Conservation Dimension, a Draft Report of the IUCN-CESP Landscape Conservation Working Group; University of Kent: Canterbury, UK, 1996. [Google Scholar]
- Turner, M.; Donato, D.C.; Romme, W.H. Consequences of spatial heterogeneity for ecosystem services in changing forest landscapes: Priorities for future research. Landsc. Ecol. 2012, 28, 1081–1097. [Google Scholar] [CrossRef]
- Brennan, J.M.; Bender, D.J.; Contreras, T.A.; Fahrig, L. Focal patch landscape studies for wildlife management: Optimizing sampling effort across scales. In Integrating Landscape Ecology into Natural Resource Management; Liu, J., Taylor, W., Eds.; Cambridge University Press: Cambridge, UK, 2002; pp. 68–91. [Google Scholar]
- Devault, T.; MacGowan, B.; Beasley, J.; Humberg, L.; Retamosa, M.; Rhodes, O. Evaluation of maize and soybean damage by wildlife in northern Indiana. In Proceedings of the 12th Wildlife Damage Management Conference, Corpus Christi, TX, USA, 9–12 April 2007; pp. 563–570. [Google Scholar]
- Pedlar, J.H.; Fahrig, L.; Merriam, H.G. Raccoon Habitat Use at 2 Spatial Scales. J. Wildl. Manag. 1997, 61, 102. [Google Scholar] [CrossRef]
- Thies, C.; Tscharntke, T. Landscape Structure and Biological Control in Agroecosystems. Science 1999, 285, 893–895. [Google Scholar] [CrossRef]
- Turner, M.G. Landscape Ecology: What Is the State of the Science? Annu. Rev. Ecol. Evol. Syst. 2005, 36, 319–344. [Google Scholar] [CrossRef]
- Gross, J.E.; Zank, C.; Hobbs, N.T.; Spalinger, D.E. Movement rules for herbivores in spatially heterogeneous environments: Responses to small scale pattern. Landsc. Ecol. 1995, 10, 209–217. [Google Scholar] [CrossRef]
- Matschke, G.; de Calesta, D.; Harder, J. Crop damage and control. In White-Tailed Deer: Ecology and Management; Halls, L., Ed.; Stackpole Books: Harrisburg, PA, USA, 1984; pp. 647–654. [Google Scholar]
- Searle, K.; Vandervelde, T.; Hobbs, N.T.; Shipley, L.A.; Wunder, B.A. Spatial context influences patch residence time in foraging hierarchies. Oecologia 2006, 148, 710–719. [Google Scholar] [CrossRef]
- Dickman, A. Complexities of conflict: The importance of considering social factors for effectively resolving human-wildlife conflict. Anim. Conserv. 2010, 13, 458–466. [Google Scholar] [CrossRef]
- Lamarque, F.; Anderson, J.; Ferguson, R.; Lagrange, M.; Osei-Owusu, Y.; Bakker, L. Human-Wildlife Conflicts in Africa: Causes, Consequences and Management Strategies (No. 157); Forestry Paper; FAO: Roma, Italia, 2009. [Google Scholar]
- Madden, F. Creating Coexistence between Humans and Wildlife: Global Perspectives on Local Efforts to Address Human–Wildlife Conflict. Hum. Dimens. Wildl. 2004, 9, 247–257. [Google Scholar] [CrossRef]
- Nyhus, P.J. Human–Wildlife Conflict and Coexistence. Annu. Rev. Environ. Resour. 2016, 41, 143–171. [Google Scholar] [CrossRef]
- Marchini, S. Who’s in conflict with whom? Human dimensions of the conflicts involving wildlife. In Applied Ecology and Human Dimensions in Biological Conservation; Springer: Berlin/Heidelberg, Germany, 2014; pp. 189–209. [Google Scholar]
- Conover, M. Resolving Human-Wildlife Conflicts: The Science of Wildlife Damage Management; CRC Press LLC: Boca Raton, FL, USA, 2002. [Google Scholar]
- Hosey, G.; Melfi, V. Human-animal interactions relationship and bonds: A review and analysis of literature. Int. J. Comp. Psychol. 2014, 27, 117–142. [Google Scholar]
- Inskip, C.; Zimmermann, A. Human-felid conflict: A review of patterns and priorities worldwide. Oryx 2009, 43, 18. [Google Scholar] [CrossRef]
- IUCN. Benefits beyond boundaries. In Proceedings of the Vth IUCN World Parks Congress, Durban, South Africa, 8–17 September 2003. [Google Scholar]
- Morzillo, A.T.; De Beurs, K.M.; Martin-Mikle, C.J. A conceptual framework to evaluate human-wildlife interactions within coupled human and natural systems. Ecol. Soc. 2014, 19, 44. [Google Scholar] [CrossRef]
- Cupul-Magaña, F.; Rubio-Delgado, A.; Reyes-Núñez, C.; Torres-Campos, E.; Solis-Pecero, L. Ataques de cocodrilo de río (Crocodylus acutus) en Puerto Vallarta, Jalisco, México: Presentación de cinco casos. Cuad. Med. Forense 2010, 16, 153–160. [Google Scholar] [CrossRef]
- García-Grajales, J. El conflicto hombre-cocodrilo en México: Causas e implicaciones. Interciencia 2013, 38, 881–884. [Google Scholar]
- Peña-Mondragón, J. Daños Económicos al Ganado y Percepciones Sociales Sobre el jaguar (Panthera Onca Veraecrucis Nelson and Goldman, 1993) en la Gran Sierra Plegada, Nuevo León, México. Master’s Thesis, Universidad Nacional Autónoma de México, Morelia, México, 2011. [Google Scholar]
- Ponce-Campos, P. Un Nuevo Conflicto Hombre-Cocodrilo en Puerto Vallarta: Análisis del Caso, Hipótesis, Resultados y Recomendaciones (Informe de Caso); Bosque Tropical, AC: Puerto Vallarta, México, 2006. [Google Scholar]
- Treves, A.; Wallace, R.B.; Naughton-Treves, L.; Morales, A. Co-Managing Human–Wildlife Conflicts: A Review. Hum. Dimens. Wildl. 2006, 11, 383–396. [Google Scholar] [CrossRef]
- Gonzalez-Gallina, A.; Mihart, M.G.H. A Review of Road-killed Felids in Mexico. Therya 2018, 9, 147–159. [Google Scholar] [CrossRef]
- Arroyo-Rodríguez, V.; Fahrig, L. Why is a landscape perspective important in studies of primates? Am. J. Primatol. 2014, 76, 901–909. [Google Scholar] [CrossRef]
- Quinn, A.C.D.; Williams, D.M.; Porter, W.F. Landscape structure influences space use by white-tailed deer. J. Mammal. 2013, 94, 398–407. [Google Scholar] [CrossRef]
- Garmendia, A.; Arroyo-Rodríguez, V.; Estrada, A.; Naranjo, E.J.; Stoner, K.E. Landscape and patch attributes impacting medium- and large-sized terrestrial mammals in a fragmented rain forest. J. Trop. Ecol. 2013, 29, 331–344. [Google Scholar] [CrossRef]
- McGarigal, K.; McComb, W.C. Relationships between Landscape Structure and Breeding Birds in the Oregon Coast Range. Ecol. Monogr. 1995, 65, 235–260. [Google Scholar] [CrossRef]
- Retamosa, M.; Humberg, L.; Beasley, J.; Rhodes, O. Modeling wildlife damage to crops in northern Indiana. Hum. Wildl. Confl. 2008, 2, 225–239. [Google Scholar]
- Bleier, N.; Kovács, I.; Schally, G.; Szemethy, L.; Csányi, S. Spatial and temporal characteristics of the damage caused by wild ungulates in maize (Zea mays L.) crops. Int. J. Pest Manag. 2016, 63, 92–100. [Google Scholar] [CrossRef]
- Hernández-Sánchez, A.; Santos-Moreno, A.; Pérez-Irineo, G. Abundance of mesocarnivores in two vegetation types in the southeastern region of Mexico. Southwest. Nat. 2017, 62, 101–108. [Google Scholar] [CrossRef]
- Lima, M.; Peres, C.A.; Abrahams, M.I.; da Silva Junior, C.A.; de Medeiros Costa, G.; Dos Santos, R.C. The paradoxical situation of the white-lipped peccary (Tayassu pecari) in the state of Mato Grosso, Brazil. Perspect. Ecol. Conserv. 2019, 17, 36–39. [Google Scholar] [CrossRef]
- Halffter, G. Reservas de la Biosfera: Problemas y Oportunidades en México. Acta Zoológica Mex. 2011, 27, 177–189. [Google Scholar] [CrossRef]
- CONANP. Programa de Conservación y Manejo, Reserva de la Biosfera Sierra de Huautla, Primera Edición; Comisión Nacional de Áreas Naturales Protegidas: México, México, 2005. [Google Scholar]
- López-Medellín, X.; Vázquez, L.B.; Valenzuela-Galván, D.; Wehncke, E.; Maldonado-Almanza, B.; Durand-Smith, L. Percepciones de los habitantes de la Reserva de la Biosfera Sierra de Huautla: Hacia el desarrollo de nuevas estrategias de manejo participativo. Interciencia 2017, 42, 8–16. [Google Scholar]
- Juárez-Mondragón, A.; González-Rebeles, C.; Castillo, A.; García, E.; Ordoñez, M. La vida silvestre manejada como recurso de uso común: Estudio de caso en México. Trop. Subtrop. Agroecosyst. 2015, 18, 313–331. [Google Scholar]
- Velarde, S.; Cruz, A. La fauna silvestre y su relación con el bienestar de tres comunidades de la Reserva de la Biosfera Sierra de Huautla, Morelos. Etnobiología 2015, 13, 39–52. [Google Scholar]
- Heinonen, J.P.M.; Palmer, S.C.F.; Redpath, S.M.; Travis, J.M.J. Modelling Hen Harrier Dynamics to Inform Human-Wildlife Conflict Resolution: A Spatially-Realistic, Individual-Based Approach. PLoS ONE 2014, 9, e112492. [Google Scholar] [CrossRef] [PubMed]
- Sitati, N.W.; Walpole, M.; Smith, R.J.; Leader-Williams, N. Predicting spatial aspects of human–elephant conflict. J. Appl. Ecol. 2003, 40, 667–677. [Google Scholar] [CrossRef]
- Redpath, S.M.; Bhatia, S.; Young, J. Tilting at wildlife: Reconsidering human–wildlife conflict. Oryx 2014, 49, 222–225. [Google Scholar] [CrossRef]
- Romero-Balderas, K.; Naranjo, E.; Morales, H.; Nigh, R. Daños ocasionados por vertebrados silvestres al cultivo de maíz en la selva lacandona, Chiapas, México. Interciencia 2006, 31, 276–283. [Google Scholar]
- Schumacher, M.; Durán-Díaz, P.; Kurjenoja, A.; Gutiérrez-Juárez, E.; González-Rivas, D.A. Evolution and Collapse of Ejidos in Mexico—To What Extent Is Communal Land Used for Urban Development? Land 2019, 8, 146. [Google Scholar] [CrossRef]
- Rzendowski, J. La Vegetación de México; Editorial Limusa: Ciudad de México, México, 1978. [Google Scholar]
- SEMARNAT. Norma Oficial Mexicana. NOM-059-SEMARNAT-2010, Protección Ambiental-Especies Nativas de México de Flora y Fauna Silvestres-Categorías de Riesgo y Especificaciones Para su Inclusión, Exclusión o Cambio-Lista de Especies en Riesgo. Diario Oficial de la Federación. 30 de Diciembre de 2010; SEMARNAT: México City, México, 2010. [Google Scholar]
- IUCN. The IUCN Red List of Threatened Species. Version 2019-3. 2020. Available online: https://www.iucnredlist.org (accessed on 14 July 2020).
- Argote-Cortés, A.; Bueno, A.; Ramírez, J.E.; Pérez, J.E.; Ramírez, G.; Martínez, M.; Ferra, J.P.; Urbina, F. AICA 40: Sierra de Huautla. In Arizmendi y L. Márquez. Base de Datos de las AICAS; Benítez, H.C., Ed.; CIPAMEX, CONABIO, FMCN y CCA: México, México, 1999. [Google Scholar]
- Arriaga, L.; Espinoza, J.M.; Aguilar, C.; Martínez, E.; Gómez, L.; Loa, E. Regiones Terrestres Prioritarias de México; Comisión Nacional para el Conocimiento y uso de la Biodiversidad: México, México, 2000.
- Arriaga, L.; Aguilar, V.; Alcocer, J. Aguas Continentales y Diversidad Biológica de México; Comisión Nacional para el Conocimiento y Uso de la Biodiversidad: México, México, 2002.
- BirdLife International. Important Bird Areas Factsheet: Sierra de Huautla. 2019. Available online: http://www.birdlife.org (accessed on 30 September 2019).
- WWF (World Wildlife Fund). Balsas Dry Forest (NT0205); World Wildlife Found: Washington, DC, USA, 2001. [Google Scholar]
- Dorado, O. Sierra de Huautla-Cerro Frío, Morelos: Proyecto de Reserva de la Biosfera (No. Informe Final SNIB-CONABIO Proyecto No. Q025). Universidad Autónoma del Estado de Morelos; Centro de Investigación en Biodiversidad y Conservación: Cuernavaca, Morelos, México, 2001. [Google Scholar]
- García, A.; Valle, R.; Monroy, R. Traditional use of wild mammals in Pitzotlan, Morelos, México. Rev. Colomb. Cienc. Anim. 2018, 10, 111–123. [Google Scholar]
- Silva, D.A.H.; Díaz, E.C.; Ramírez, J.L.Z.; Hernández, P.A.M.; Bonilla, G.T.G.; Castañeda, B.R.; Sedas, D.A.H. Hábitat del venado cola blanca, en la Sierra de Huautla, Morelos, México. Acta Zool. Mex. 2011, 27, 47–66. [Google Scholar] [CrossRef]
- Sánchez-Martínez, L. Interacción Humano-Fauna Silvestre y el Daño Ocasionado a Los Cultivos en la Reserva de la Biósfera Sierra de Huautla: Percepciones y Actitudes. Bachelor’s Thesis, Facultad de Ciencias Biológicas, Universidad Autónoma del Estado de Morelos, Morelos, México, 2019; p. 66. [Google Scholar]
- Šálek, M.; Kučera, T.; Zimmermann, K.; Bartůšková, I.; Platek, M.; Grill, S.; Konvicka, M. Edges within farmland: Management implications of taxon specific species richness correlates. Basic Appl. Ecol. 2015, 16, 714–725. [Google Scholar] [CrossRef]
- Sorani, V.; Rodríguez, G.; Valenzuela-Galván, D. El cambio de uso de suelo en la Reserva de la Biosfera Sierra de Huautla. In La Diversidad Biológica en Morelos: Estudio del Estado. CONABIO/Secretaría de Desarrollo Sustentable—Gobierno del Estado de Morelos; Cuernavaca: Morelos, Mexico, 2020. [Google Scholar]
- Gallegos-Peña, A.; Bello-Gutiérrez, J.; De la Cruz, A. Cuantificación del daño ocasionado por mamíferos terrestres a cultivos de maíz en el ejido Oxolotán del municipio de Tacotalpa, Tabasco, México. In Uso y Manejo de Fauna Silvestre en el Norte de Mesoamérica; Guerra Roa, M.M., Calmé, S., Gallina Tessaro, S., Piñera, J.N., Eds.; Serie Hablemos de Ciencia y Tecnología; El Colegio de la Frontera Sur (ECOSUR); Secretaría de Educación del Gobierno del Estado de Veracruz; Instituto de Ecología A.C. (INECOL): Xalapa, Veracruz, México, 2010; pp. 297–311. [Google Scholar]
- Engeman, R.M.; Sterner, R.T. A comparison of potential labor-saving sampling methods for assessing large mammal damage in corn. Crop. Prot. 2002, 21, 101–105. [Google Scholar] [CrossRef]
- Kovács, I.; Tóth, B.; Schally, G.; Csányi, S.; Bleier, N. The assessment of wildlife damage estimation methods in maize with simulation in GIS environment. Crop. Prot. 2020, 127, 104971. [Google Scholar] [CrossRef]
- Bevanda, M.; Fronhofer, E.A.; Heurich, M.; Müller, J.; Reineking, B. Landscape configuration is a major determinant of home range size variation. Ecosphere 2015, 6, 195. [Google Scholar] [CrossRef]
- Valenzuela, D.; Macdonald, D.W. Home-range use by white-nosed coatis (Nasua narica): Limited water and a test of the resource dispersion hypothesis. J. Zool. 2002, 258, 247–256. [Google Scholar] [CrossRef]
- Peck, R.; Olsen, C.; Devore, J. Introduction to Statistics and Data Analysis, 3rd ed.; Brooks/Cole: Belmont, CA, USA, 2008. [Google Scholar]
- Wójcik-Gront, E. Variables influencing yield-scaled Global Warming Potential and yield of winter wheat production. Field Crop. Res. 2018, 227, 19–29. [Google Scholar] [CrossRef]
- Pesch, R.; Schmidt, G.; Schróder, W.; Weustermann, I. Application of CART in ecological landscape mapping: Two case studies. Ecol. Indic. 2011, 11, 115–122. [Google Scholar] [CrossRef]
- Kandel, S.L.; Smiley, R.W.; Garland-Campbell, K.; Elling, A.A.; Huggins, D.; Paulitz, T. Spatial distribution of root lesion nematodes (Pratylenchus spp.) in a long-term no-till cropping system and their relationship with soil and landscape properties. Eur. J. Plant Pathol. 2017, 150, 1011–1021. [Google Scholar] [CrossRef]
- Hartman, T.; Harbour, J.; Tharnish, B.; Van Meter, J.; Jackson-Ziems, T.A. Agronomic Factors Associated with Bacterial Leaf Streak Development Caused by Xanthomonas vasicola pv. vasculorum in Corn. Phytopathology 2020, 110, 1132–1138. [Google Scholar] [CrossRef]
- Subramanian, V.R. Classification and Regression Trees (CART). In Compendium Hands-on Training on “Statistical Tools and Database Management in Agriculture”; Dheri, G.S., Pal, S., Singh, V., Marwaha, S., Choudhary, O.P., Eds.; ICAR NAHEP-CAAST-SNRM Department of Soil Science Punjab Agricultural University: Ludhiana, Punjab, India, 2019; pp. 95–100. [Google Scholar]
- Gazzinelli, A.; Oliveira-Prado, R.; Matoso, L.F.; Veloso, B.M.; Andrade, G.; Kloos, H.; Bethony, J.M.; Assunção, R.M.; Corrêa-Oliveira, R. Schistosoma mansoni reinfection: Analysis of risk factors by classification and regression tree (CART) modeling. PLoS ONE 2017, 12, e0182197. [Google Scholar] [CrossRef]
- Breiman, L.; Friedman, J.; Olshen, R.; Stone, C. Classification and Regression Trees; CRC Press: Belmont, CA, USA, 1984. [Google Scholar]
- Crawley, M. The R Book, 2nd ed.; John Wiley & Sons: London, UK, 2013. [Google Scholar]
- De’ath, G.; Fabricius, K. Classification and regression trees: A powerful yet simple technique for ecological data analysis. Ecology 2000, 81, 3178–3192. [Google Scholar] [CrossRef]
- Andersen, M.; Watts, J.; Freilich, S.; Yool, G.; Wakefield, J.; McCauley, J.; Fahnestock, P. Regression-tree modeling of desert tortoise habitat in the central Mojave Desert. Ecol. Appl. 2000, 10, 890–900. [Google Scholar] [CrossRef]
- Jackson, R.D.; Bartolome, J.W. A state-transition approach to understanding nonequilibrium plant community dynamics in Californian grasslands. Plant Ecol. 2002, 162, 49–65. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Therneau, T.; Atkinson, B.; Ripley, B. Rpart: Recursive Partitioning and Regression Trees; R Package Version; R Foundation for Statistical Computing: Vienna, Austria, 2015; Volume 4, pp. 1–9. [Google Scholar]
- Can-Hernández, G.; Villanueva-García, C.; Gordillo-Chávez, E.J.; Pacheco-Figueroa, C.J.; Pérez-Netzahual, E.; García-Morales, R. Wildlife damage to crops adjacent to a protected area in southeastern México: Farmers’ perceptions versus actual impact. Hum. Wildl. Interact. 2019, 13, 423–438. [Google Scholar]
- Chávez, G. Determinación de las Relaciones Hombre Fauna Silvestre en una Zona Rural de Quintana Roo (Boletín Técnico No. 94); Instituto Nacional de Investigaciones Forestales: Distrito Federal, México, 1983. [Google Scholar]
- Hawthorne, W. Daños Provocados por Animales Silvestres y Técnicas de Control. In Manual de Técnicas de Gestión de Vida Silvestre; Schemnitz, S., Ed.; The Wildlife Society: Bethesda, MD, USA, 1987; pp. 431–462. [Google Scholar]
- Corona, P.; Gallina, S.; Contreras, A. El aprovechamiento del venado cola blanca en una UMA de la Sierra de Huautla, Morelos. In Uso y Manejo de La Fauna Silvestre En El Norte de Mesoamérica; Guerra, M., Calmé, S., Gallina, S., Naranjo, E., Eds.; Secretaría de Educación de Veracruz: Xalapa, México, 2010; pp. 263–298. [Google Scholar]
- López-Téllez, M.C.; Mandujano, S.; Yanes-Gómez, G. Evaluación poblacional del venado cola blanca en un bosque tropical seco de la mixteca poblana. Acta Zool. Mex. 2007, 23, 1–16. [Google Scholar] [CrossRef]
- Mandujano, S.; Yañez-Arenas, C.A.; González-Zamora, A.; Pérez-Arteaga, A. Habitat-population density relationship for the white-tailed deer Odocoileus virginianus during the dry season in a Pacific Mexican tropical dry forest. Mammalia 2013, 77, 381–389. [Google Scholar] [CrossRef]
- Ramos-Robles, M.; Gallina, S.; Mandujano, S. Habitat and Human Factors Associated with White-Tailed Deer Density in the Tropical Dry Forest of Tehuacán-Cuicatlán Biosphere Reserve, Mexico. Trop. Conserv. Sci. 2013, 6, 70–86. [Google Scholar] [CrossRef]
- Gardiner, B.; Berry, P.; Moulia, B. Review: Wind impacts on plant growth, mechanics and damage. Plant Sci. 2016, 245, 94–118. [Google Scholar] [CrossRef]
- Bellon, M.R. The ethnoecology of maize variety management: A case study from Mexico. Hum. Ecol. 1991, 19, 389–418. [Google Scholar] [CrossRef]
- Arroyo-Rodríguez, V.; Fahrig, L.; Tabarelli, M.; Watling, J.I.; Tischendorf, L.; Benchimol, M.; Cazetta, E.; Faria, D.; Leal, I.R.; Melo, F.P.L.; et al. Designing optimal human-modified landscapes for forest biodiversity conservation. Ecol. Lett. 2020, 23, 1404–1420. [Google Scholar] [CrossRef]
- Owens, J.; Lund, H.G. Forests and Forest Plant; UNESCO-EOLSS Publications: Oxford, UK, 2009; Volume II, p. 368. [Google Scholar]
- Morales, S.; Guzmán, E. Caracterización sociocultural de las milpas en dos ejidos del municipio de Tlaquiltenango. Etnobiología 2015, 13, 94–109. [Google Scholar]
- Hinton, G.C.; Strickland, B.K.; DeMarais, S.; Eubank, T.W.; Jones, P.D. Estimation of deer damage to soybean production in eastern Mississippi: Perception versus reality. Wildl. Soc. Bull. 2017, 41, 80–87. [Google Scholar] [CrossRef]
- Naughton-Treves, L. Predicting Patterns of Crop Damage by Wildlife around Kibale National Park, Uganda. Conserv. Boil. 1998, 12, 156–168. [Google Scholar] [CrossRef]
- Leslie, D.M.; Starkey, E.E.; Vavra, M. Elk and Deer Diets in Old-Growth Forests in Western Washington. J. Wildl. Manag. 1984, 48, 762. [Google Scholar] [CrossRef]
- Vangilder, L.D.; Torgerson, O.; Porath, W.R. Factors Influencing Diet Selection by White-Tailed Deer. J. Wildl. Manag. 1982, 46, 711. [Google Scholar] [CrossRef]
- Tejeda-Cruz, C.; Naranjo-Piñera, E.; Medina-Sanson, L.; Guevara-Hernández, F. Cacería de subsistencia en comunidades rurales de la selva Lacandona, Chiapas, México. Quehacer Cient. Chiapas 2014, 9, 59–73. [Google Scholar]
- Roseberry, J.; Woolf, A. Habitat-Population density relationships for White-tailed deer in Illinois. Wildl. Soc. Bull. 1998, 26, 252–258. [Google Scholar]
- Delfín-Alfonso, C.A.; Gallina-Tessaro, S.; López-González, C.A. Evaluación del hábitat del venado cola blanca utilizando modelos espaciales y sus implicaciones para el manejo en el centro de Veracruz, México. Trop. Conserv. Sci. 2009, 2, 215–228. [Google Scholar] [CrossRef]
- Villarreal-Espino, O.A.; Plata-Pérez, F.X.; Camacho-Ronquillo, J.C.; Hernández-Hernández, J.E.; Franco-Guerra, F.J.; Aguilar-Ortega, B.; Mendoza-Martínez, G.D. El Venado Cola Blanca en la mixteca poblana. Therya 2011, 2, 103–110. [Google Scholar] [CrossRef]
- Meinecke, L.; Soofi, M.; Riechers, M.; Khorozyan, I.G.; Hosseini, H.; Schwarze, S.; Waltert, M. Crop variety and prey richness affect spatial patterns of human-wildlife conflicts in Iran’s Hyrcanian forests. J. Nat. Conserv. 2018, 43, 165–172. [Google Scholar] [CrossRef]
- Hill, C.; Webber, A.D. Perceptions of nonhuman primates in human-wildlife conflict scenarios. Am. J. Primatol. 2010, 72, 919–924. [Google Scholar] [CrossRef]
- Nyirenda, V.R.; Myburg, W.J.; Reilly, B.K.; Phiri, A.I.; Chabwela, H.N. Wildlife crop damage valuation and conservation: Conflicting perception by local farmers in the Luangwa Valley, eastern Zambia. Int. J. Biodivers. Conserv. 2013, 5, 741–750. [Google Scholar]

| % of Total Damaged Maize Plants by Each Damaging Factor | ||||||
|---|---|---|---|---|---|---|
| % ALU | Total Maize Plants | Total Damaged Maize Plants | WTD | WNC | B&O | W |
| <25 | 15,422 | 1021 | 38.0% | 21.1% | 1.9% | 39.1% |
| 26–50 | 17,544 | 197 | 15.7% | 22.8% | 0.0% | 61.4% |
| 51–75 | 15,674 | 632 | 7.3% | 10.4% | 7.8% | 74.5% |
| >76 | 21,984 | 572 | 2.3% | 1.9% | 2.8% | 93.0% |
| % of Total Damaged Maize Cobs by Each Damaging Factor | ||||||
|---|---|---|---|---|---|---|
| % ALU | Total Maize Cobs | Total Damaged Maize Cobs | WTD | WNC | B&O | W |
| <25 | 10,294 | 597 | 71.0% | 20.4% | 1.0% | 7.5% |
| 26–50 | 8108 | 143 | 23.8% | 53.8% | 16.8% | 5.6% |
| 51–75 | 11,225 | 306 | 32.7% | 25.5% | 0.0% | 41.8% |
| >76 | 17,531 | 160 | 23.1% | 26.9% | 3.8% | 46.3% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-Armillas, V.H.; López-Medellín, X.; García Barrios, R.; MacGregor-Fors, I.; Valenzuela-Galván, D. Landscape Features Associated with Damage to Maize (Zea mays) Fields in Central México: A Comparison of Wind and Wildlife Damage. Agriculture 2020, 10, 460. https://doi.org/10.3390/agriculture10100460
Flores-Armillas VH, López-Medellín X, García Barrios R, MacGregor-Fors I, Valenzuela-Galván D. Landscape Features Associated with Damage to Maize (Zea mays) Fields in Central México: A Comparison of Wind and Wildlife Damage. Agriculture. 2020; 10(10):460. https://doi.org/10.3390/agriculture10100460
Chicago/Turabian StyleFlores-Armillas, Victor Hugo, Xavier López-Medellín, Raúl García Barrios, Ian MacGregor-Fors, and David Valenzuela-Galván. 2020. "Landscape Features Associated with Damage to Maize (Zea mays) Fields in Central México: A Comparison of Wind and Wildlife Damage" Agriculture 10, no. 10: 460. https://doi.org/10.3390/agriculture10100460
APA StyleFlores-Armillas, V. H., López-Medellín, X., García Barrios, R., MacGregor-Fors, I., & Valenzuela-Galván, D. (2020). Landscape Features Associated with Damage to Maize (Zea mays) Fields in Central México: A Comparison of Wind and Wildlife Damage. Agriculture, 10(10), 460. https://doi.org/10.3390/agriculture10100460





