1. Introduction
Systemic sclerosis (SSc) is a progressive autoimmune disease that affects multiple internal and external organs by pathogenetic mechanisms that mainly include microangiopathies and fibrosis [
1]. In spite of the high lethality and low quality of life (QOL) of the disease, to date no disease-modifying or definite curative modalities have been reported. Although recently updated treatment guidelines from the European League against Rheumatism (EULAR) covered approved treatment options based on literature reviews, only limited therapies received a high level of recommendation [
2].
Theoretically, every organ could be affected through the disease progress of SSc. Hand disability is one of the most frequently observed and QOL-influencing clinical features in patients with SSc, but remains a poorly solved issue in terms of therapeutic methods, as are other systemic involvements of SSc [
3]. Various phenotypes, such as Raynaud’s phenomenon, digital ulcers, skin fibrosis, and joint synovitis or contractures, are included in this category [
4]. Digital ulcers and skin fibrosis of hands are regarded as the main causes of hand pain and disability [
3]. Because microvascular injuries and fibrotic changes triggered by immunologic response are thought to be the biomechanics of such hand involvement, a number of pharmacologic or non-pharmacologic approaches have been suggested as treatment options [
4,
5]. Intravenous injection of iloprost, one of the synthetic prostanoids, is proved to have efficacy in the healing of digital ulcers, while bosentan, an endothelin receptor antagonist, reduces the additional development of digital ulcers in SSc patients [
2]. However, all these treatments have various adverse effects and limitations to maintaining persistent application for long periods. Moreover, at the present time, anti-fibrotic treatment to improve skin fibrosis and joint contracture of hands is barely available [
2].
To overcome such obstacles to management for hand involvement in SSc, several novel therapeutic modalities with clinical potency have been suggested. Since extraction and introduction from adipose tissue by Zuk et al. in 2002, stromal vascular fraction (SVF) consisting of multipotent cell populations, called adipose-derived stem cells (ASC), various growth factors and cytokines, has emerged as a potential therapeutic option, based on its regenerative and anti-inflammatory effects for the treatment of SSc [
6,
7]. According to the promising results of recent trials from French and Italian groups, long-term safety and efficacy of SVF from autologous adipose tissue were reported, improving the hand disability, QOL, and digital ulcers of SSc patients [
8,
9,
10,
11]. Moreover, a commercialized system enabling SVF to be expeditiously extracted from harvested adipose tissue makes it possible to perform whole procedures in an outpatient setting.
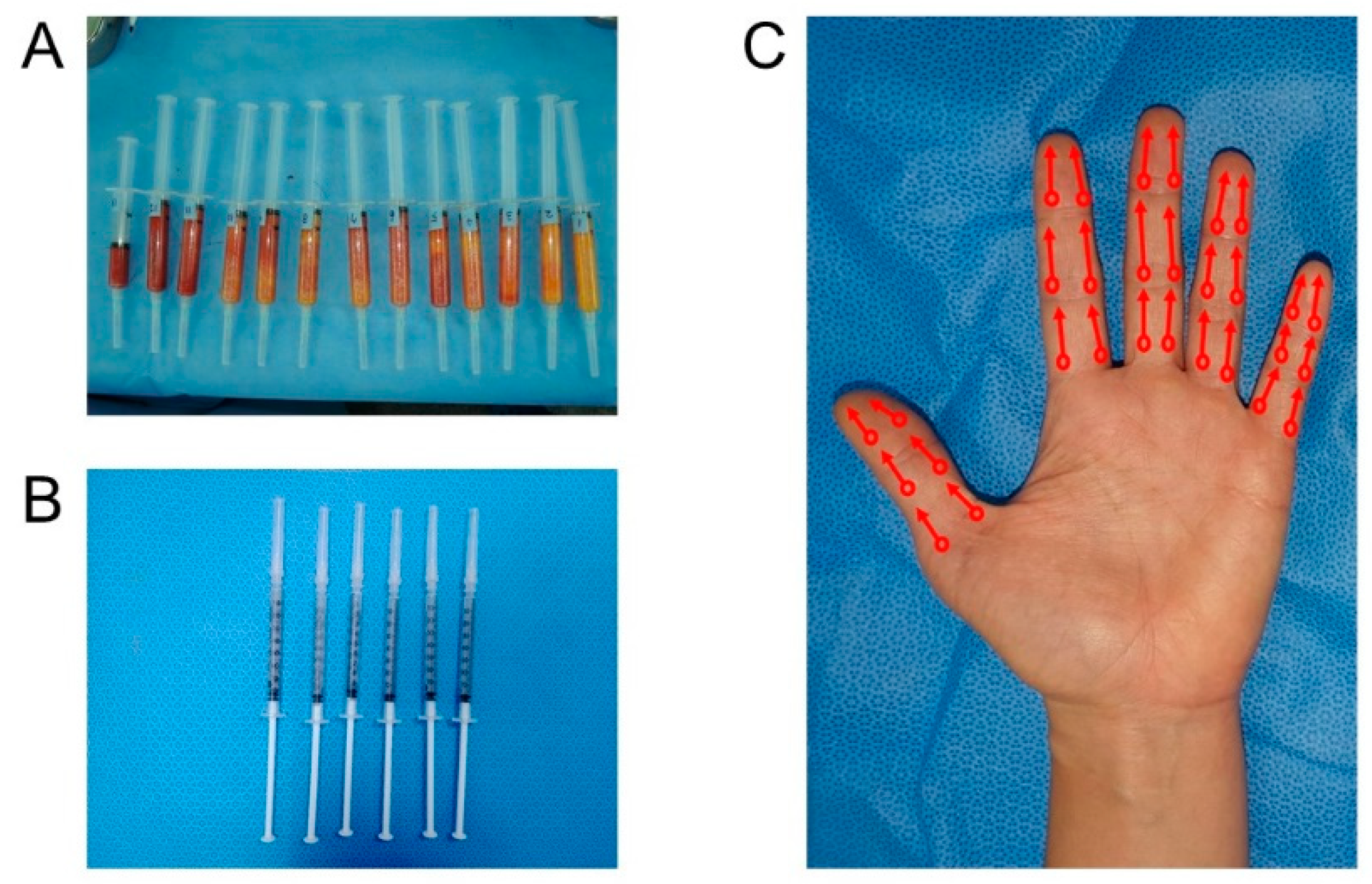
Herein, we report the results of an open-label proof-of-concept trial designed to investigate the tolerability and clinical outcomes of SVF administration into patients with SSc. The safety of SVF extraction from autologous adipose tissue and local injection in involved hands, and clinical efficacies in terms of SSc-related outcomes were assessed through a 24-week follow-up period.
4. Discussion
According to the results of the present trial, autologous fat tissue-derived SVF injection into the affected hands of SSc patients is well tolerated, and presents clinical efficacies in terms of improving skin fibrosis and QOL, healing digital ulcers, and ameliorating microangiopathies. Comparing with the results of the previous trials from France and Italy, the degree of improvements in microangiopathies, QOL and finger ulcers was relatively less but amelioration of skin fibrosis was more prominent in this trial. No single severe adverse event occurred, and several clinical improvements, such as anti-fibrotic effects and the recovery of active ulcers, occurred and did not get worse again during the period of the study.
Since the identification of undifferentiated and multipotent cell populations as its components in the early twentieth century, SVF is suggested as a new source of mesenchymal stem cells [
20]. Various therapeutic approaches have been attempted and revealed as effective in terms of regenerative medicine. Because SVF is a very complex mixture of cellular components and soluble factors, exact mechanisms, including autocrine or paracrine effects, are challenging to explore. Nevertheless, because of the prompt administration without further incubation, and the ease of its acquisition compared to other sources, such as bone marrow, SVF is a promising alternative. Commercialized kits for extracting SVF from adipose tissues facilitate application to broader spectra of diseases by reducing costs and the time required for procedures.
In the present study, improvement of fibrosis, represented by decreased mRSS and mean circumferences of fingers (
Figure 2, and
Table 3 and
Table 4), is the most prominent finding. Most past studies regarding ASC, as well as SVF, have mainly focused on their regenerative efficacy in poorly repaired wounds [
6,
9,
10]. According to the present study, SVF treatment considerably reduce mRSS, too. Although improvement of finger edema could be regarded as amelioration of skin fibrosis, mRSS, which is validated for measurement of skin thickness through other studies, showed meaningful changes under SVF treatment. The exact mechanism of SVF is scarcely elucidated, due to its heterogeneous cell populations and various mixtures of soluble factors. Nonetheless, ASC is regarded as the most likely key factor in operating the overall effects of local SVF injection, as well as the main cause of anti-fibrotic effect [
7]. Inhibiting the expression of transforming growth factor-beta is suggested as one of the modes of action of ASCs in anti-fibrosis, according to a previous study using mouse model with bleomycin-induced fibrosis [
21]. Intriguingly, local injection of SVF into both hands affects the degree of fibrosis of patient’s upper arms and faces, which are distant from the initial injection sites. Although several kinds of homing molecules, which are able to reach target organs, were reported in the past studies regarding the systemic infusion of mesenchymal stem cells (MSC), the mechanisms of how locally injected SVF effects systemically remain are in need of investigation in future study [
22].
Several reports in the literature have reported the regenerative potency of SVF. An Italian group performed a randomized controlled trial to compare the clinical effectiveness of adipose tissue grafting containing SVF in ischemic digital ulcers to placebo [
9]. Ulcers refractory to other medical treatments were well recovered, and the number of nailfold capillaries was gradually increased for 8 weeks of follow-up periods under a single injection of SVF. Granel et al. also reported positive results regarding the recovery of digital ulcers and nailfold capillary findings in SSc using SVF injection [
10]. Our results are also comparable with the previously mentioned data showing promising consequences in ischemic injuries and microscopic findings of the affected patients’ hands (
Table 5,
Table 6 and
Table 7). Although the stricter definition of ulcer healing, defined as full recovery of skin defect, and more detailed parameters in nailfold capillary microscopic findings might compel relatively reduced responses in the present trial compared to the previous studies, SVF is also proved to be effective considering the efficacy of conventional treatment for this complication. Although the exact biological mechanisms resulting in such effects are hard to derive, several possible candidates could be speculated through previous studies. ASCs contained in SVF could promote angiogenesis in ischemic tissues by paracrine mechanisms [
23]. Other cellular components, such as fibroblasts, pericytes, and several precursors cells in SVF also contribute to the regeneration of damaged tissues by secreting cytokines, and constitute the mechanical framework of repaired tissues [
24,
25].
Among efficacy outcomes, Raynaud’s condition scale, hand pain VAS, and CHFS showed rarely positive results in the present study. Although these variables presented significant differences, and were validated in the past studies, on the other hand, they are subjective indices that could be easily confounded by out-of-intervention factors. Seasonal variation of the severity of Raynaud’s phenomenon might influence the results of these subjective indices [
26]. Nevertheless, we observed several favorable outcomes in other objective parameters. Considering a recent transcriptomic report that supports that the angiogenic and repairing capacity of SVF from patients with SSc are not compromised compared to healthy populations, and another report that shows comparable efficacy with bone marrow-derived MSC, autologous SVF in SSc patients is a reasonable alternative source of MSC and soluble factors [
27,
28]. In some other views, collagenase used in enzymatic treatment of SVF could lower therapeutic potency by reducing intercellular communications and may even be forbidden in the future by policies. Future studies related to SVF application should be considered in relation to these points.
The present study has several limitations to be discussed. Although this trial involved more detailed outcome measures in some aspects and a newly introduced SVF isolation method, overall results presented limited novelty except in terms of skin fibrosis compared to the past studies. In addition, cell yields of SVF showed enormous inter-patient variability in this study. This point could limit proper assessment of clinical efficacies of SVF. In some patients, aspirated adipose tissues tended to be fibrotic and scattered, and resulted in extremely low cell yield. Although such low cell yield did not statistically indicate low clinical efficacies in the present study, proper reasons for why this phenomenon occurred in SSc patients should be clarified. Furthermore, considering the absence of control groups treated with placebo and the relatively small number of enrolled subjects in this trial, further studies are in need.