Sodium Hypochlorite Reduces Postoperative Discomfort and Painful Early Failure after Carious Exposure and Direct Pulp Capping—Initial Findings of a Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Ethics
2.3. Power Analysis
2.4. Patient Recruitment
2.5. Inclusion Criteria
2.6. Further Screening before Intervention
2.7. Clinical Procedures and Allocation to Treatment
2.8. Outcome Assessment
2.9. Data Collection, Monitoring and Statistical Analysis
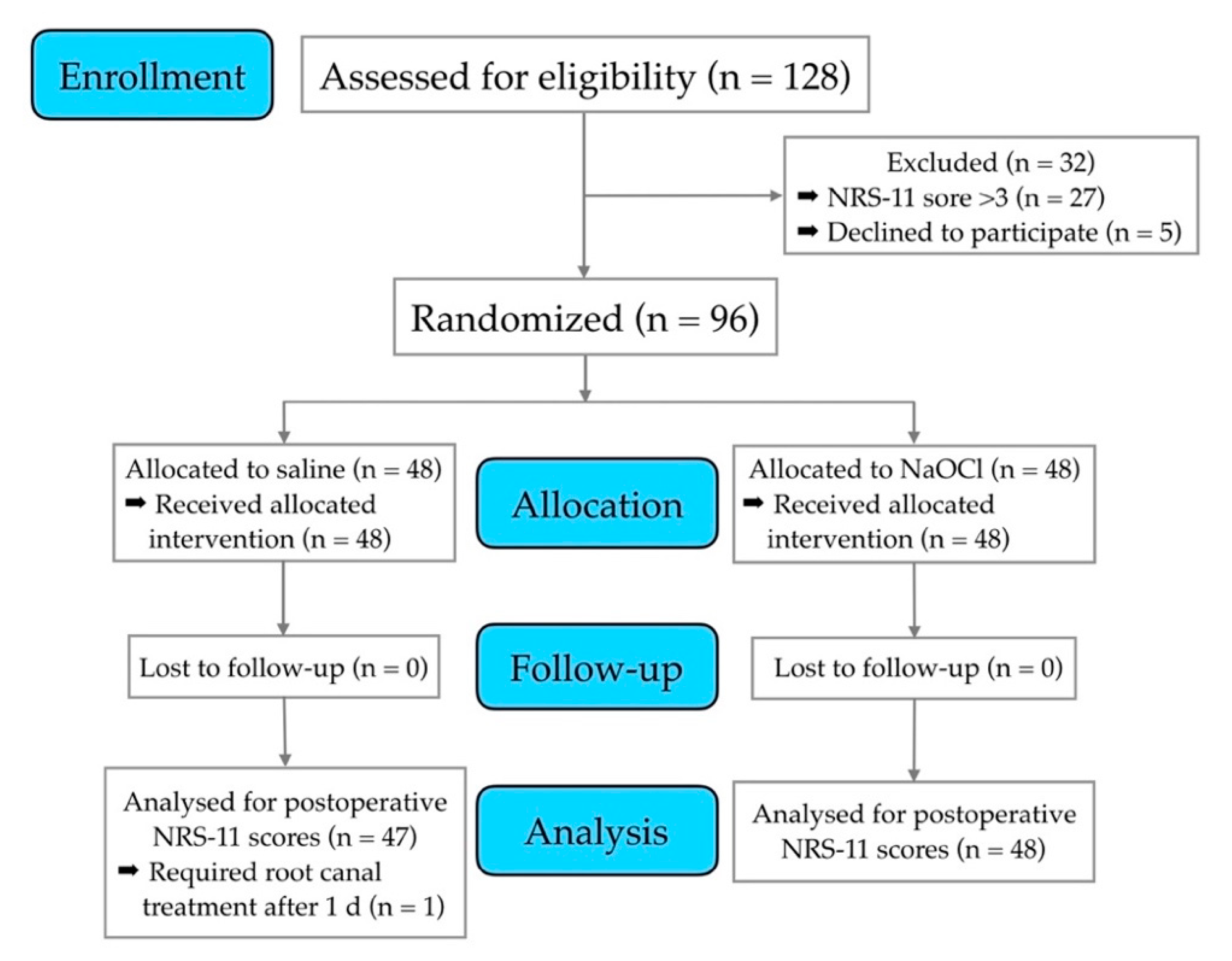
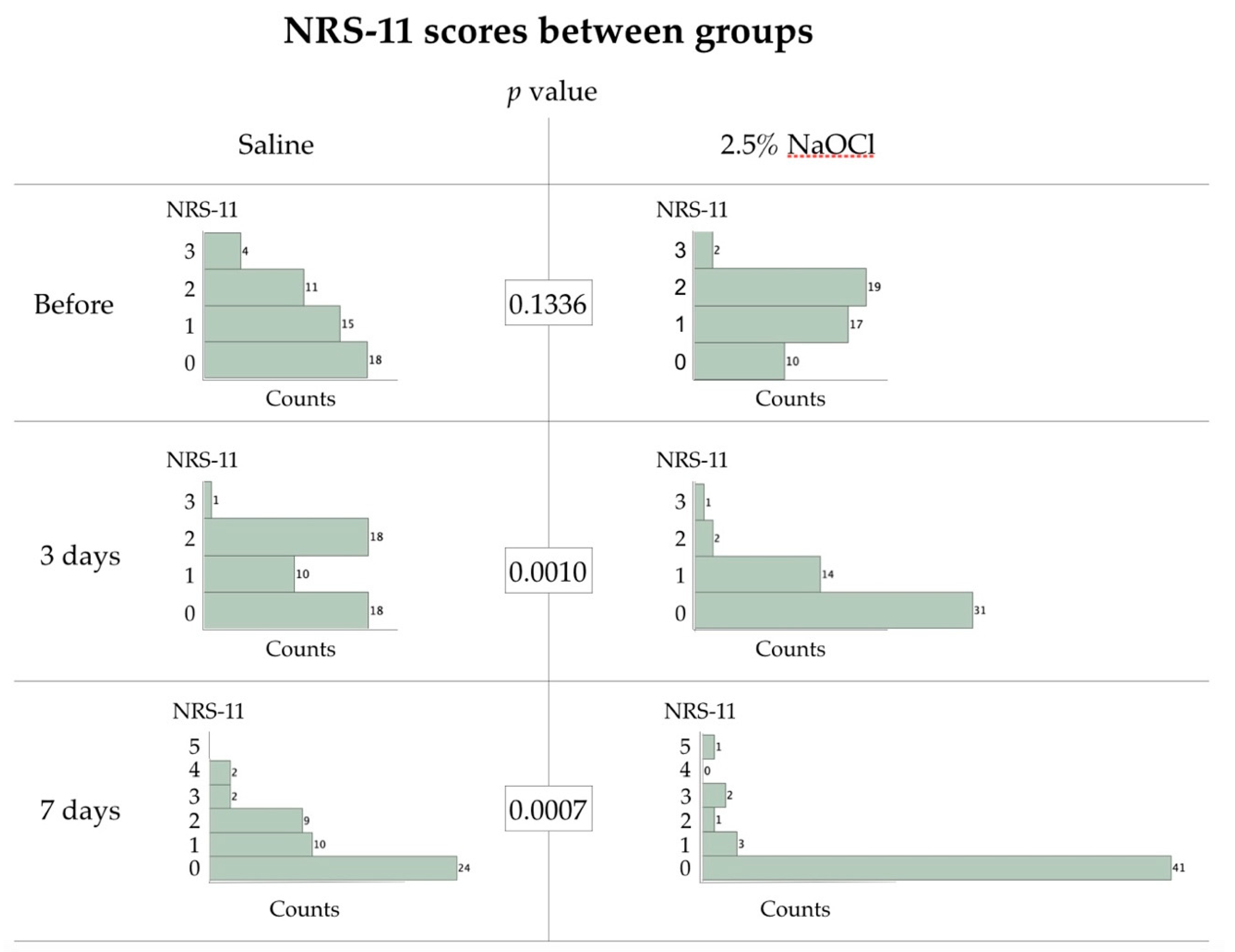
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Schwendicke, F.; Frencken, J.E.; Bjørndal, L.; Maltz, M.; Manton, D.J.; Ricketts, D.; Van Landuyt, K.; Banerjee, A.; Campus, G.; Doméjean, S.; et al. Managing Carious Lesions: Consensus Recommendations on Carious Tissue Removal. Adv. Dent. Res. 2016, 28, 58–67. [Google Scholar] [CrossRef]
- Bergenholtz, G.; Axelsson, S.; Davidson, T.; Frisk, F.; Hakeberg, M.; Kvist, T.; Norlund, A.; Petersson, A.; Portenier, I.; Sandberg, H.; et al. Treatment of pulps in teeth affected by deep caries—A systematic review of the literature. Singap. Dent. J. 2013, 34, 1–12. [Google Scholar] [CrossRef]
- Duncan, H.F.; Galler, K.M.; Tomson, P.L.; Simon, S.; El-Karim, I.; Kundzina, R.; Krastl, G.; Dammaschke, T.; Fransson, H.; Markvart, M.; et al. European Society of Endodontology position statement: Management of deep caries and the exposed pulp. Int. Endod. J. 2019, 52, 923–934. [Google Scholar] [PubMed]
- Aguilar, P.; Linsuwanont, P. Vital pulp therapy in vital permanent teeth with cariously exposed pulp: A systematic review. J. Endod. 2011, 37, 581–587. [Google Scholar] [CrossRef]
- Munir, A.; Zehnder, M.; Rechenberg, D.K. Wound Lavage in Studies on Vital Pulp Therapy of Permanent Teeth with Carious Exposures: A Qualitative Systematic Review. J. Clin. Med. 2020, 9, 984. [Google Scholar] [CrossRef]
- Levey, C.; Innes, N.; Schwendicke, F.; Lamont, T.; Göstemeyer, G. Outcomes in randomised controlled trials in prevention and management of carious lesions: A systematic review. Trials 2017, 18, 515. [Google Scholar] [CrossRef]
- Torabinejad, M.; Walton, R.E. Managing endodontic emergencies. J. Am. Dent. Assoc. 1991, 122, 99–103. [Google Scholar] [CrossRef]
- Bjørndal, L.; Reit, C.; Bruun, G.; Markvart, M.; Kjaeldgaard, M.; Nasman, P.; Thordrup, M.; Dige, I.; Nyvad, B.; Fransson, H.; et al. Treatment of deep caries lesions in adults: Randomized clinical trials comparing stepwise vs. direct complete excavation, and direct pulp capping vs. partial pulpotomy. Eur. J. Oral Sci. 2010, 118, 290–297. [Google Scholar] [CrossRef]
- Suhag, K.; Duhan, J.; Tewari, S.; Sangwan, P. Success of Direct Pulp Capping Using Mineral Trioxide Aggregate and Calcium Hydroxide in Mature Permanent Molars with Pulps Exposed during Carious Tissue Removal: 1-year Follow-up. J. Endod. 2019, 45, 840–847. [Google Scholar] [CrossRef]
- Wang, C.; Xu, P.; Ren, L.; Dong, G.; Ye, L. Comparison of post-obturation pain experience following one-visit and two-visit root canal treatment on teeth with vital pulps: A randomized controlled trial. Int. Endod. J. 2010, 43, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, L.; Lif Holgerson, P.; Esberg, A.; Johansson, I. Microbial Complexes and Caries in 17-Year-Olds with and without Streptococcus mutans. J. Dent. Res. 2018, 97, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Reeves, R.; Stanley, H.R. The relationship of bacterial penetration and pulpal pathosis in carious teeth. Oral Surg. Oral Med. Oral Pathol. 1966, 22, 59–65. [Google Scholar] [CrossRef]
- Ricucci, D.; Loghin, S.; Siqueira, J.F.J. Correlation between clinical and histologic pulp diagnoses. J. Endod. 2014, 40, 1932–1939. [Google Scholar] [CrossRef] [PubMed]
- Degnim, A.C.; Scow, J.S.; Hoskin, T.L.; Miller, J.P.; Loprinzi, M.; Boughey, J.C.; Jakub, J.W.; Throckmorton, A.; Patel, R.; Baddour, L.M. Randomized controlled trial to reduce bacterial colonization of surgical drains after breast and axillary operations. Ann. Surg. 2013, 258, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, R.; Griffiths, R.; Ussia, C. Effectiveness of solutions, techniques and pressure in wound cleansing. JBI Libr. Syst. Rev. 2004, 2, 1–55. [Google Scholar] [CrossRef]
- Dakin, H.D. On the use of certain antiseptic substances in treatment of infected wounds. Br. Med. J. 1915, 2, 318–320. [Google Scholar] [CrossRef]
- Austin, J.H.; Taylor, H.D. Behavior of hypochlorite and of chloramine-t solutions in contact with necrotic and normal tissues in vivo. J. Exp. Med. 1918, 27, 627–633. [Google Scholar] [CrossRef]
- Kundzina, R.; Stangvaltaite, L.; Eriksen, H.M.; Kerosuo, E. Capping carious exposures in adults: A randomized controlled trial investigating mineral trioxide aggregate versus calcium hydroxide. Int. Endod. J. 2017, 50, 924–932. [Google Scholar] [CrossRef]
- Hülsmann, M.; Hahn, W. Complications during root canal irrigation—Literature review and case reports. Int. Endod. J. 2000, 33, 186–193. [Google Scholar] [CrossRef]
- Hilton, T.J.; Ferracane, J.L.; Mancl, L. Comparison of CaOH with MTA for direct pulp capping: A PBRN randomized clinical trial. J. Dent. Res. 2013, 92, 16S–22S. [Google Scholar] [CrossRef]
- Hegde, S.; Sowmya, B.; Mathew, S.; Bhandi, S.H.; Nagaraja, S.; Dinesh, K. Clinical evaluation of mineral trioxide aggregate and biodentine as direct pulp capping agents in carious teeth. J. Conserv. Dent. 2017, 20, 91–95. [Google Scholar] [PubMed]
- Barthel, C.R.; Rosenkranz, B.; Leuenberg, A.; Roulet, J.F. Pulp capping of carious exposures: Treatment outcome after 5 and 10 years: A retrospective study. J. Endod. 2000, 26, 525–528. [Google Scholar] [CrossRef] [PubMed]
- American Association of Endodontists. AAE consensus conference recommended diagnostic terminology. J. Endod. 2009, 35, 1634. [Google Scholar] [CrossRef]
- Warren Grant Magnusson Clinical Center. Pain Intensity Instruments. 2003. Available online: http://www.mvltca.net/presentations/mvltca.pdf (accessed on 18 December 2018).
- Ferreira-Valente, M.A.; Pais-Ribeiro, J.L.; Jensen, M.P. Validity of four pain intensity rating scales. Pain 2011, 152, 2399–2404. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, V.; Singla, M.; Miglani, S.; Kohli, S. Efficacy of Articaine Versus Lidocaine Administered as Supplementary Intraligamentary Injection after a Failed Inferior Alveolar Nerve Block: A Randomized Double-blind Study. J. Endod. 2019, 45, 1–5. [Google Scholar] [CrossRef]
- Rauch, A.; Hahnel, S.; Schierz, O. Pain, Dental Fear, and Oral Health-related Quality of Life-Patients Seeking Care in an Emergency Dental Service in Germany. J. Contemp. Dent. Pract. 2019, 20, 3–7. [Google Scholar]
- Bjørndal, L.; Simon, S.; Tomson, P.L.; Duncan, H.F. Management of deep caries and the exposed pulp. Int. Endod. J. 2019, 52, 949–973. [Google Scholar] [CrossRef]
- Marending, M.; Attin, T.; Zehnder, M. Treatment options for permanent teeth with deep caries. Swiss Dent. J. 2016, 126, 1007–1027. [Google Scholar]
- Witton, R.; Henthorn, K.; Ethunandan, M.; Harmer, S.; Brennan, P.A. Neurological complications following extrusion of sodium hypochlorite solution during root canal treatment. Int. Endod. J. 2005, 38, 843–848. [Google Scholar] [CrossRef]
- Martin, D.E.; De Almeida, J.F.; Henry, M.A.; Khaing, Z.Z.; Schmidt, C.E.; Teixeira, F.B.; Diogenes, A. Concentration-dependent effect of sodium hypochlorite on stem cells of apical papilla survival and differentiation. J. Endod. 2014, 40, 51–55. [Google Scholar] [CrossRef]
- Mente, J.; Geletneky, B.; Ohle, M.; Koch, M.J.; Friedrich Ding, P.G.; Wolff, D.; Dreyhaupt, J.; Martin, N.; Staehle, H.J.; Pfefferle, T. Mineral trioxide aggregate or calcium hydroxide direct pulp capping: An analysis of the clinical treatment outcome. J. Endod. 2010, 36, 806–813. [Google Scholar] [CrossRef]
- Linsuwanont, P.; Wimonsutthikul, K.; Pothimoke, U.; Santiwong, B. Treatment Outcomes of Mineral Trioxide Aggregate Pulpotomy in Vital Permanent Teeth with Carious Pulp Exposure: The Retrospective Study. J. Endod. 2017, 43, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Janakiram, C.; Antony, B.; Joseph, J.; Ramanarayanan, V. Prevalence of dental caries in India among the WHO index age groups: A meta-analysis. J. Clin. Diagn. Res. 2018, 12, ZE08–ZE13. [Google Scholar] [CrossRef]
- Matsuo, T.; Nakanishi, T.; Shimizu, H.; Ebisu, S. A clinical study of direct pulp capping applied to carious-exposed pulps. J. Endod. 1996, 22, 551–556. [Google Scholar] [CrossRef]
- Jensen, M.P.; Karoly, P.; Braver, S. The measurement of clinical pain intensity: A comparison of six methods. Pain 1986, 27, 117–126. [Google Scholar] [CrossRef]
- Law, A.S.; Nixdorf, D.R.; Aguirre, A.M.; Reams, G.J.; Tortomasi, A.J.; Manne, B.D.; Harris, D.R.; National Dental PBRN Collaborative Group. Predicting severe pain after root canal therapy in the National Dental PBRN. J. Dent. Res. 2015, 94, 37S–43S. [Google Scholar] [CrossRef] [PubMed]
- Hasselgren, G.; Reit, C. Emergency pulpotomy: Pain relieving effect with and without the use of sedative dressings. J. Endod. 1989, 15, 254–256. [Google Scholar] [CrossRef]
- Deari, S.; Wegehaupt, F.J.; Tauböck, T.T.; Attin, T. Influence of Different Pretreatments on the Microtensile Bond Strength to Eroded Dentin. J. Adhes. Dent. 2017, 19, 147–155. [Google Scholar]
| Saline | 2.5% NaOCl | p Value | |
|---|---|---|---|
| Patient age (y) | 38 ± 12 | 30 ± 12 | 0.0011 * |
| Patient gender (f/m) | 32/16 | 23/25 | 0.0981 |
| Tooth type (pm/m) | 14/34 | 12/36 | 0.8187 |
| Cavity class (Black I/II) | 4/44 | 7/41 | 0.5235 |
| Jaw (maxilla/mandible) | 22/26 | 24/24 | 0.8383 |
| Operator (N.V.B/N.R./P.J.) | 19/13/16 | 16/17/15 | 0.6627 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ballal, N.V.; Duncan, H.F.; Rai, N.; Jalan, P.; Zehnder, M. Sodium Hypochlorite Reduces Postoperative Discomfort and Painful Early Failure after Carious Exposure and Direct Pulp Capping—Initial Findings of a Randomized Controlled Trial. J. Clin. Med. 2020, 9, 2408. https://doi.org/10.3390/jcm9082408
Ballal NV, Duncan HF, Rai N, Jalan P, Zehnder M. Sodium Hypochlorite Reduces Postoperative Discomfort and Painful Early Failure after Carious Exposure and Direct Pulp Capping—Initial Findings of a Randomized Controlled Trial. Journal of Clinical Medicine. 2020; 9(8):2408. https://doi.org/10.3390/jcm9082408
Chicago/Turabian StyleBallal, Nidambur Vasudev, Henry F. Duncan, Namith Rai, Prateek Jalan, and Matthias Zehnder. 2020. "Sodium Hypochlorite Reduces Postoperative Discomfort and Painful Early Failure after Carious Exposure and Direct Pulp Capping—Initial Findings of a Randomized Controlled Trial" Journal of Clinical Medicine 9, no. 8: 2408. https://doi.org/10.3390/jcm9082408
APA StyleBallal, N. V., Duncan, H. F., Rai, N., Jalan, P., & Zehnder, M. (2020). Sodium Hypochlorite Reduces Postoperative Discomfort and Painful Early Failure after Carious Exposure and Direct Pulp Capping—Initial Findings of a Randomized Controlled Trial. Journal of Clinical Medicine, 9(8), 2408. https://doi.org/10.3390/jcm9082408






