Feasibility of Using the Epidural Space Detecting Device (EPI-DetectionTM) for Interlaminar Cervical Epidural Injection
Abstract
1. Introduction
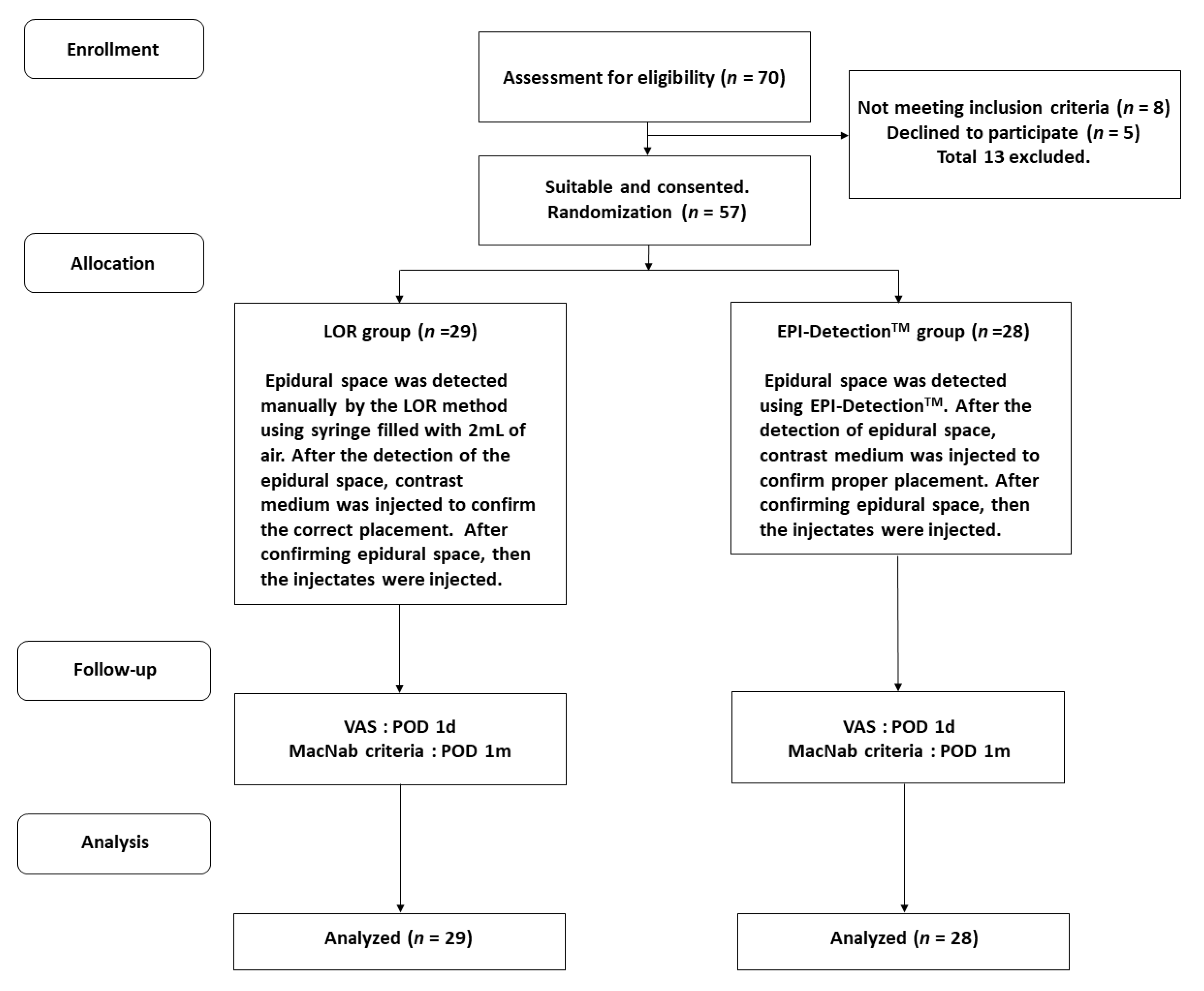
2. Materials and Methods
2.1. Study Design
2.2. Patient Selection and Blinding Strategy
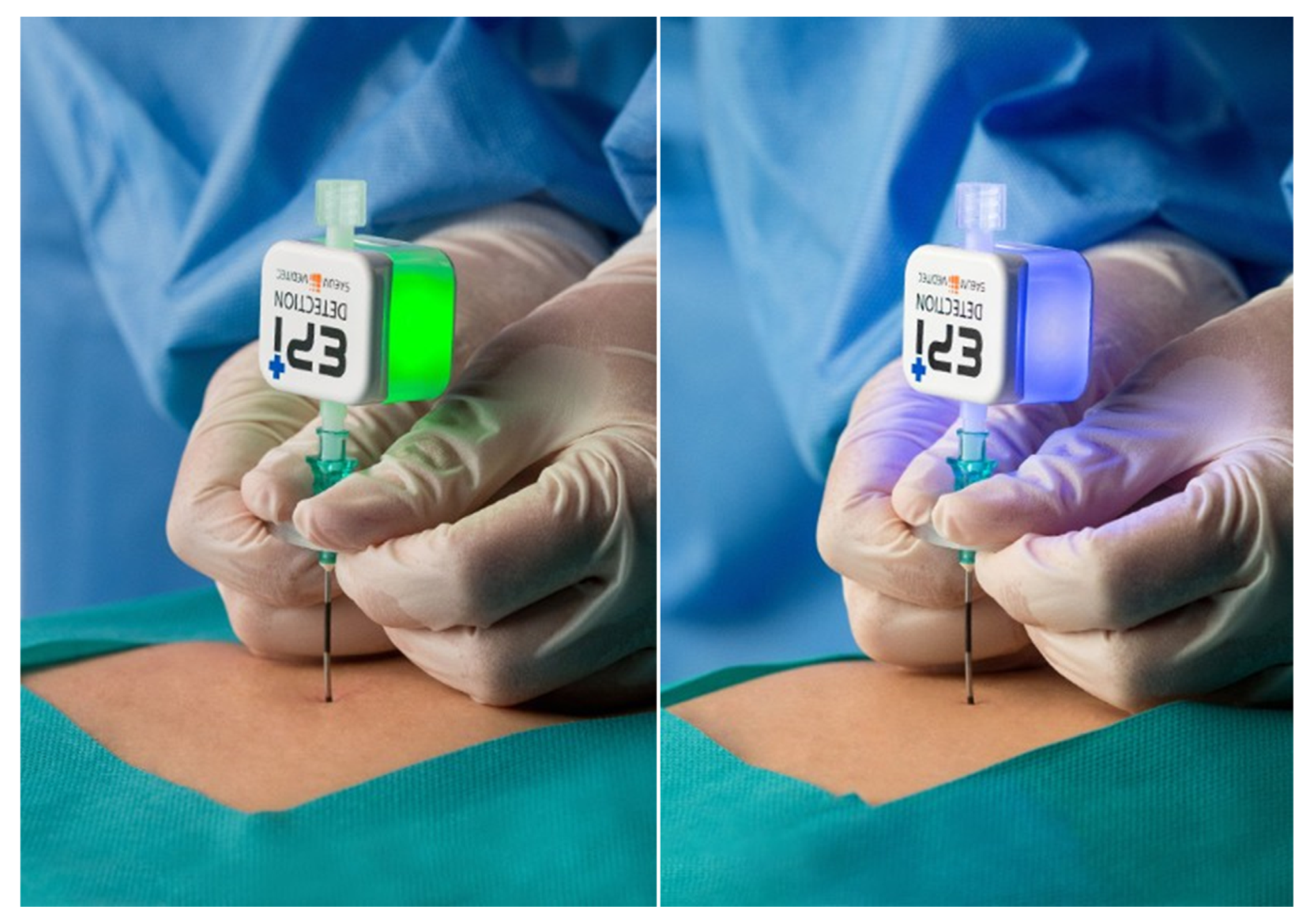
2.3. EPI-DetectionTM Device
2.4. Interlaminar Epidural Injection
2.5. EPI-DetectionTM Procedure
2.6. LOR Procedure
2.7. Outcomes Assessment
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Manchikanti, L.; Pampati, V.; Hirsch, J.A. Retrospective cohort study of usage patterns of epidural injections for spinal pain in the US fee-for-service Medicare population from 2000 to 2014. BMJ Open 2016, 6, e013042. [Google Scholar] [CrossRef]
- Stav, A.; Ovadia, L.; Sternberg, A.; Kaadan, M.; Weksler, N. Cervical epidural steroid injection for cervicobrachialgia. Acta Anaesthesiol. Scand. 1993, 37, 562–566. [Google Scholar] [CrossRef]
- Pasqualucci, A.; Varrassi, G.; Braschi, A.; Peduto, V.A.; Brunelli, A.; Marinangeli, F.; Gori, F.; Colò, F.; Paladini, A.; Mojoli, F. Epidural local anesthetic plus corticosteroid for the treatment of cervical brachial radicular pain: Single injection versus continuous infusion. Clin. J. Pain 2007, 23, 551–557. [Google Scholar] [CrossRef]
- Manchikanti, L.; Malla, Y.; Cash, K.A.; McManus, C.D.; Pampati, V. Fluoroscopic cervical interlaminar epidural injections in managing chronic pain of cervical postsurgery syndrome: Preliminary results of a randomized, double-blind, active control trial. Pain Physician 2012, 15, 13–26. [Google Scholar]
- Manchikanti, L.; Malla, Y.; Cash, K.A.; McManus, C.D.; Pampati, V. Fluoroscopic epidural injections in cervical spinal stenosis: Preliminary results of a randomized, double-blind, active control trial. Pain Physician 2012, 15, 59–70. [Google Scholar]
- McCormick, Z.L.; Nelson, A.; Bhave, M.; Zhukalin, M.; Kendall, M.; McCarthy, R.J.; Khan, D.; Nagpal, G.; Walega, D.R. A prospective randomized comparative trial of targeted steroid injection via epidural catheter versus standard C7-T1 interlaminar approach for the treatment of unilateral cervical radicular pain. Reg. Anesth. Pain Med. 2017, 42, 82–89. [Google Scholar] [CrossRef]
- Manchikanti, L.; Cash, K.A.; Pampati, V.; Wargo, B.W.; Malla, Y. A randomized, double-blind, active control trial of fluoroscopic cervical interlaminar epidural injections in chronic pain of cervical disc herniation: Results of a 2-year follow-up. Pain Physician 2013, 16, 465–478. [Google Scholar]
- Manchikanti, L.; Cash, K.A.; Pampati, V.; Malla, Y. Two-year follow-up results of fluoroscopic cervical epidural injections in chronic axial or discogenic neck pain: A randomized, double-blind, controlled trial. Int. J. Med. Sci. 2014, 11, 309–320. [Google Scholar] [CrossRef]
- Abbasi, A.; Malhotra, G.; Malanga, G.; Elovic, E.P.; Kahn, S. Complications of interlaminar cervical epidural steroid injections: A review of the literature. Spine 2007, 32, 2144–2151. [Google Scholar] [CrossRef]
- Waldman, S.D. Complications of cervical epidural nerve blocks steroids: A prospective study of 790 consecutive blocks. Reg. Anesth. Pain Med. 1989, 14, 149–151. [Google Scholar]
- Williams, K.; Jackowski, A.; Evans, P. Epidural haematoma requiring surgical decompression following repeated cervical epidural steroid injections for chronic pain. Pain 1990, 42, 197–199. [Google Scholar] [CrossRef]
- Hodges, S.D.; Castleberg, R.L.; Miller, T.; Ward, R.; Thornburg, C. Cervical epidural steroid injection with intrinsic spinal cord damage: Two case reports. Spine 1998, 23, 2137–2140. [Google Scholar] [CrossRef] [PubMed]
- Bose, B. Quadriparesis following cervical epidural steroid injections: Case report and review of the literature. Spine J. 2005, 5, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Muro, K.; O’shaughnessy, B.; Ganju, A. Infarction of the cervical spinal cord following multilevel transforaminal epidural steroid injection: Case report and review of the literature. J. Spinal Cord Med. 2007, 30, 385–388. [Google Scholar] [CrossRef]
- Sicard, J.; Forestier, J. Méthode radiologique d’exploration de la cavité épidurale par le lipiodol. Rev. Neurol. 1921, 37, 1264–1266. [Google Scholar]
- Hogan, Q.H. Epidural anatomy examined by cryomicrotome section: Influence of age, vertebral level, and disease. Reg. Anesth. Pain Med. 1996, 21, 395–406. [Google Scholar]
- Paech, M.; Godkin, R.; Webster, S. Complications of obstetric epidural analgesia and anaesthesia: A prospective analysis of 10 995 cases. Int. J. Obstet. Anesth. 1998, 7, 5–11. [Google Scholar] [CrossRef]
- Lorenz, I.; Keller, C.; Kirchmair, L.; Rieder, J.; Moriggl, B. Cervical and high thoracic ligamentum flavum frequently fails to fuse in the midline. Anesthesiology 2003, 99, 1387–1390. [Google Scholar]
- Kartal, S.; Kösem, B.; Kılınç, H.; Köşker, H.; Karabayırlı, S.; Çimen, N.; Demircioğlu, R. Comparison of Epidrum, Epi-Jet, and Loss of Resistance syringe techniques for identifying the epidural space in obstetric patients. Niger. J. Clin. Pract. 2017, 20, 992–997. [Google Scholar]
- Deighan, M.; Briain, D.O.; Shakeban, H.; O’Flaherty, D.; Abdulla, H.; Al-Jourany, A.; Ash, S.; Ahmed, S.; McMorrow, R. A randomised controlled trial using the Epidrum for labour epidurals. Ir. Med. J. 2015, 108, 73–75. [Google Scholar]
- Hanley, C.; Sweeney, K.; Kerin, M.; McDonnell, J. Successful use of ‘Epidrum’loss-of-resistance device for thoracic paravertebral blockade in patients undergoing breast cancer surgery: A case series. Eur. J. Anaesthesiol. 2014, 31, 648–649. [Google Scholar] [CrossRef] [PubMed]
- Sawada, A.; Kii, N.; Yoshikawa, Y.; Yamakage, M. Epidrum®: A new device to identify the epidural space with an epidural Tuohy needle. J. Anesth. 2012, 26, 292–295. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.; Park, S.S.; Ji, G.Y.; Kim, B.W.; Lee, C.K.; Kim, E.C.; Shin, D.A. Utility of an epidural pressure checker in the administration of trans-laminar epidural blocks. Asian J. Pain 2016, 2, 6–9. [Google Scholar] [CrossRef]
- House, L.M.; Barrette, K.; Mattie, R.; McCormick, Z.L. Cervical epidural steroid injection: Techniques and evidence. Phys. Med. Rehabil. Clin. N. Am. 2018, 29, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Rathmell, J.P.; Benzon, H.T.; Dreyfuss, P.; Huntoon, M.; Wallace, M.; Baker, R.; Riew, K.D.; Rosenquist, R.W.; Aprill, C.; Rost, N.S. Safeguards to prevent neurologic complications after epidural steroid injections: Consensus opinions from a multidisciplinary working group and national organizations. Anesthesiology 2015, 122, 974–984. [Google Scholar] [CrossRef]
- Fejer, R.; Kyvik, K.O.; Hartvigsen, J. The prevalence of neck pain in the world population: A systematic critical review of the literature. Eur. Spine J. 2006, 15, 834–848. [Google Scholar] [CrossRef]
- Huston, C.W. Cervical epidural steroid injections in the management of cervical radiculitis: Interlaminar versus transforaminal. A review. Curr. Rev. Musculoskelet. Med. 2009, 2, 30–42. [Google Scholar] [CrossRef]
- Carette, S.; Fehlings, M.G. Cervical radiculopathy. N. Engl. J. Med. 2005, 353, 392–399. [Google Scholar] [CrossRef]
- Kim, Y.H.; Seocho-gu, S. Assessment of clinical outcomes of cervical epidural neuroplasty using a Racz-catheter and predictive factors of efficacy in patients with cervical spinal pain. Pain Physician 2015, 18, 163–170. [Google Scholar]
- Bush, K.; Hillier, S. Outcome of cervical radiculopathy treated with periradicular/epidural corticosteroid injections: A prospective study with independent clinical review. Eur. Spine J. 1996, 5, 319–325. [Google Scholar] [CrossRef]
- Racz, G.B.; Heavner, J.E.; Raj, P.P. Epidural neuroplasty. In Seminars in Anesthesia, Perioperative Medicine and Pain; WB Saunders: Philadelphia, PA, USA, 1997; Volume 16, pp. 302–312. [Google Scholar]
- Liverpool, U.; Diwan, S.A. Effectiveness of cervical epidural injections in the management of chronic neck and upper extremity pain. Pain Physician 2012, 15, 405–434. [Google Scholar]
- Racz, G.B.; Heavner, J.E.; Noe, C.E.; Al-Kaisy, A.; Matsumoto, T.; Lee, S.C.; Nagy, L. Epidural lysis of adhesions and percutaneous neuroplasty. In Techniques of Neurolysis; Racz, G., Noe, C., Eds.; Springer: Cham, Switzerland, 2016; pp. 119–143. [Google Scholar]
- Oh, C.H.; Ji, G.Y.; Shin, D.A.; Cho, P.G.; Yoon, S.H. Clinical course of cervical percutaneous epidural neuroplasty in single-level cervical disc disease with 12-month follow-up. Pain Physician 2017, 20, 941–949. [Google Scholar]
- Slipman, C.W.; Lipetz, J.S.; DePalma, M.J.; Jackson, H.B. Therapeutic selective nerve root block in the nonsurgical treatment of traumatically induced cervical spondylotic radicular pain. Am. J. Phys. Med. Rehabil. 2004, 83, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Slipman, C.W.; Lipetz, J.S.; Jackson, H.B.; Plastaras, C.T.; Vresilovic, E. Outcomes of therapeutic selective nerve root blocks for whiplash induced cervical radicular pain. Pain Physician 2001, 4, 167–174. [Google Scholar]
- Castagnera, L.; Maurette, P.; Pointillart, V.; Vital, J.; Erny, P.; Senegas, J. Long-term results of cervical epidural steroid injection with and without morphine in chronic cervical radicular pain. Pain 1994, 58, 239–243. [Google Scholar] [CrossRef]
- Cicala, R.S.; Westbrook, L.; Angel, J.J. Side effects and complications of cervical epidural steroid injections. J. Pain Symptom Manag. 1989, 4, 64–66. [Google Scholar] [CrossRef]
- Simopoulos, T.; Peeters-Asdourian, C. Pneumocephalus after cervical epidural steroid injection. Anesth. Analg. 2001, 92, 1576–1577. [Google Scholar] [CrossRef]
- Reitman, C.A.; Watters, W., III. Subdural hematoma after cervical epidural steroid injection. Spine 2002, 27, 174–176. [Google Scholar] [CrossRef]
- Botwin, K.P.; Castellanos, R.; Rao, S.; Hanna, A.F.; Torres-Ramos, F.M.; Gruber, R.D.; Bouchlas, C.G.; Fuoco, G.S. Complications of fluoroscopically guided interlaminar cervical epidural injections. Arch. Phys. Med. Rehabil. 2003, 84, 627–633. [Google Scholar] [CrossRef]
- Talu, G.K.; Erdine, S. Complications of epidural neuroplasty: A retrospective evaluation. Neuromodulation 2003, 6, 237–247. [Google Scholar] [CrossRef]
- Derby, R.; Lee, S.-H.; Kim, B.-J.; Chen, Y.; Seo, K.S. Complications following cervical epidural steroid injections by expert interventionalists in 2003. Pain Physician 2004, 7, 445–450. [Google Scholar] [PubMed]
- Ludwig, M.A.; Burns, S.P. Spinal cord infarction following cervical transforaminal epidural injection: A case report. Spine 2005, 30, E266–E268. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, G.; Abbasi, A.; Rhee, M. Complications of transforaminal cervical epidural steroid injections. Spine 2009, 34, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.-J.; Park, H.-G.; Park, Y.-H.; Shin, M.-R.; Koo, G.-H.; Shin, H.-Y. Pneumocephalus during cervical transforaminal epidural steroid injections: A case report. Am. J. Phys. Med. Rehabil. 2015, 94, 63–69. [Google Scholar] [CrossRef]
- Kim, Y.-D.; Ham, H.-D.; Moon, H.-S.; Kim, S.-H. Delayed pneumocephalus following fluoroscopy guided cervical interlaminar epidural steroid injection: A rare complication and anatomical considerations. J. Korean Neurosurg. Soc. 2015, 57, 376–378. [Google Scholar] [CrossRef]
- Hirabayashi, T.; Kawamata, T.; Sakamoto, A.; Ikeno, S.; Minemura, H.; Ide, S.; Kurato, T.; Kawamata, M. Usefulness of Epidrum for teaching identification of the epidural space. Masui 2011, 60, 1078–1081. [Google Scholar]
- Kim, S.W.; Kim, Y.M.; Kim, S.H.; Chung, M.H.; Choi, Y.R.; Choi, E.M. Comparison of loss of resistance technique between Epidrum® and conventional method for identifying the epidural space. Korean J. Anesthesiol. 2012, 62, 322–326. [Google Scholar] [CrossRef]
- Lee, J.Y.; Lee, S.M.; Sim, W.S.; Ahn, H.J.; Park, M.H.; Lim, H.Y.; Lee, S.H.; Kim, Y.R.; Kim, Y.I.; Bang, Y.J. False loss of resistance in cervical epidural injection: The loss of resistance technique compared with the epidrum guidance in locating epidural space. Pain Physician 2016, 19, 131–138. [Google Scholar]
- Le Guen, M.; Charvet, A.; Leone, M.; Fischler, M. Epidrum is an unreliable device for identifying the thoracic epidural space. Eur. J. Anaesthesiol. 2018, 35, 716–717. [Google Scholar] [CrossRef]
- Zarzur, E. Genesis of the ‘true’negative pressure in the lumbar epidural space: A new hypothesis. Anaesthesia 1984, 39, 1101–1104. [Google Scholar] [CrossRef]
- Mittal, A.K.; Goel, N.; Chowdhury, I.; Shah, S.B.; Singh, B.P.; Jakhar, P. Acoustic puncture assist device versus loss of resistance technique for epidural space identification. Indian J. Anaesth. 2016, 60, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Al-Mokaddam, M.; Al-Harbi, M.; El-Jandali, S.; Al-Zahrani, T. Acoustic puncture assist device: A novel technique to identify the epidural space. Saudi J. Anaesth. 2016, 10, 179–181. [Google Scholar] [PubMed]
- Usubiaga, J.E.; Wikinski, J.A.; Usubiaga, L.E. Epidural pressure and its relation to spread of anesthetic solutions in epidural space. Anesth. Analg. 1967, 46, 440–446. [Google Scholar] [CrossRef] [PubMed]

| LOR (n = 29) | EPI-DetectionTM (n = 28) | p-Value | |
|---|---|---|---|
| Sex (M:F) | 22:7 | 17:11 | 0.219 |
| Age (years) | 54.2 ± 9.5 | 53.6 ± 11.2 | 0.848 |
| Height (cm) | 167.2 ± 8.7 | 166.6 ± 9.6 | 0.887 |
| Weight (kg) | 68.0 ± 11.5 | 67.6 ± 11.1 | 0.784 |
| BMI | 24.2 ± 2.8 | 24.2 ± 2.1 | 0.967 |
| BMI > 25 (yes: no) | 9:20 | 13:25 | 0.233 |
| Preoperative diagnosis | |||
| Herniated cervical disc (%) | 65.5% (19) | 67.9% (19) | 0.851 |
| Chronic neck pain (%) | 34.5% (10) | 32.1% (9) | |
| Treated level | |||
| C4–5 (%) | 41.4% (12) | 32.1% (9) | 0.609 |
| C5–6 (%) | 34.5% (10) | 32.1% (9) | |
| C6–7 (%) | 24.1% (7) | 35.7% (10) |
| LOR (n = 29) | EPI-DetectionTM (n = 28) | p-Value | |
|---|---|---|---|
| Operation Profile | |||
| Operation time (min) | 6.9 ± 2.1 | 4.6 ± 1.2 | 0.000 * |
| Radiation dose (mGy·cm2) | 380.3 ± 340.9 | 223.2 ± 206.7 | 0.040 * |
| Failure of epidural puncture on the first try (%) | 4 (13.8) | 1 (3.6) | 0.173 |
| Outcome Scale | |||
| VAS score on POD 1 | 2.4 ± 1.3 | 1.9 ± 1.8 | 0.153 |
| MacNab Scale score | |||
| Excellent | 18 | 20 | 0.454 |
| Good | 11 | 8 | |
| Complications (%) | 0 (0) | 0 (0) | 1.000 |
| B | ß | 95% CI | p-Value | |
|---|---|---|---|---|
| Age | −0.570 | −0.046 | −4.012, 2.873 | 0.741 |
| Sex (female) | 39.159 | 0.145 | −206.029, −88.784 | 0.399 |
| Weight | 0.033 | 0.003 | −4.502, 4.569 | 0.988 |
| Height | 0.807 | 0.058 | −5.629, 7.242 | 0.802 |
| EPI-DetectionTM (Yes) | −147.406 | −0.589 | −206.019, −88.784 | 0.000 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, J.; Park, S.S.; Kim, C.H.; Kim, E.C.; Kim, H.C.; Jeon, H.; Kim, K.H.; Shin, D.A. Feasibility of Using the Epidural Space Detecting Device (EPI-DetectionTM) for Interlaminar Cervical Epidural Injection. J. Clin. Med. 2020, 9, 2355. https://doi.org/10.3390/jcm9082355
Kang J, Park SS, Kim CH, Kim EC, Kim HC, Jeon H, Kim KH, Shin DA. Feasibility of Using the Epidural Space Detecting Device (EPI-DetectionTM) for Interlaminar Cervical Epidural Injection. Journal of Clinical Medicine. 2020; 9(8):2355. https://doi.org/10.3390/jcm9082355
Chicago/Turabian StyleKang, Jiin, Sam Sun Park, Chul Hwan Kim, Eui Chul Kim, Hyung Cheol Kim, Hyungseok Jeon, Kyung Hyun Kim, and Dong Ah Shin. 2020. "Feasibility of Using the Epidural Space Detecting Device (EPI-DetectionTM) for Interlaminar Cervical Epidural Injection" Journal of Clinical Medicine 9, no. 8: 2355. https://doi.org/10.3390/jcm9082355
APA StyleKang, J., Park, S. S., Kim, C. H., Kim, E. C., Kim, H. C., Jeon, H., Kim, K. H., & Shin, D. A. (2020). Feasibility of Using the Epidural Space Detecting Device (EPI-DetectionTM) for Interlaminar Cervical Epidural Injection. Journal of Clinical Medicine, 9(8), 2355. https://doi.org/10.3390/jcm9082355




