Diabetes Mellitus and Vitamin D Deficiency: Comparable Effect on Survival and a Deadly Association after a Myocardial Infarction
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Design
2.3. Definitions and End-points
2.4. Statistical Analysis
3. Results
3.1. Patients Characteristics
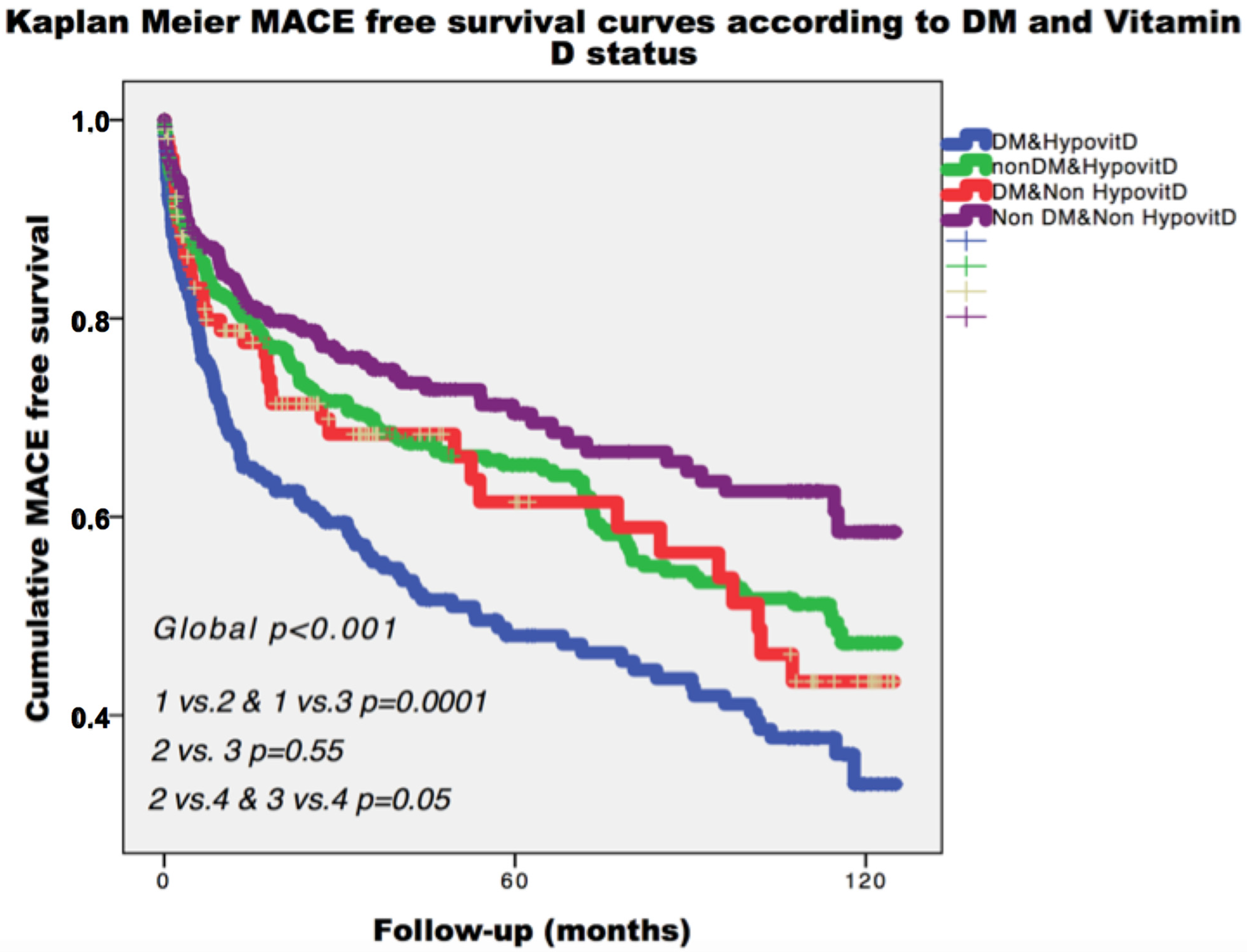
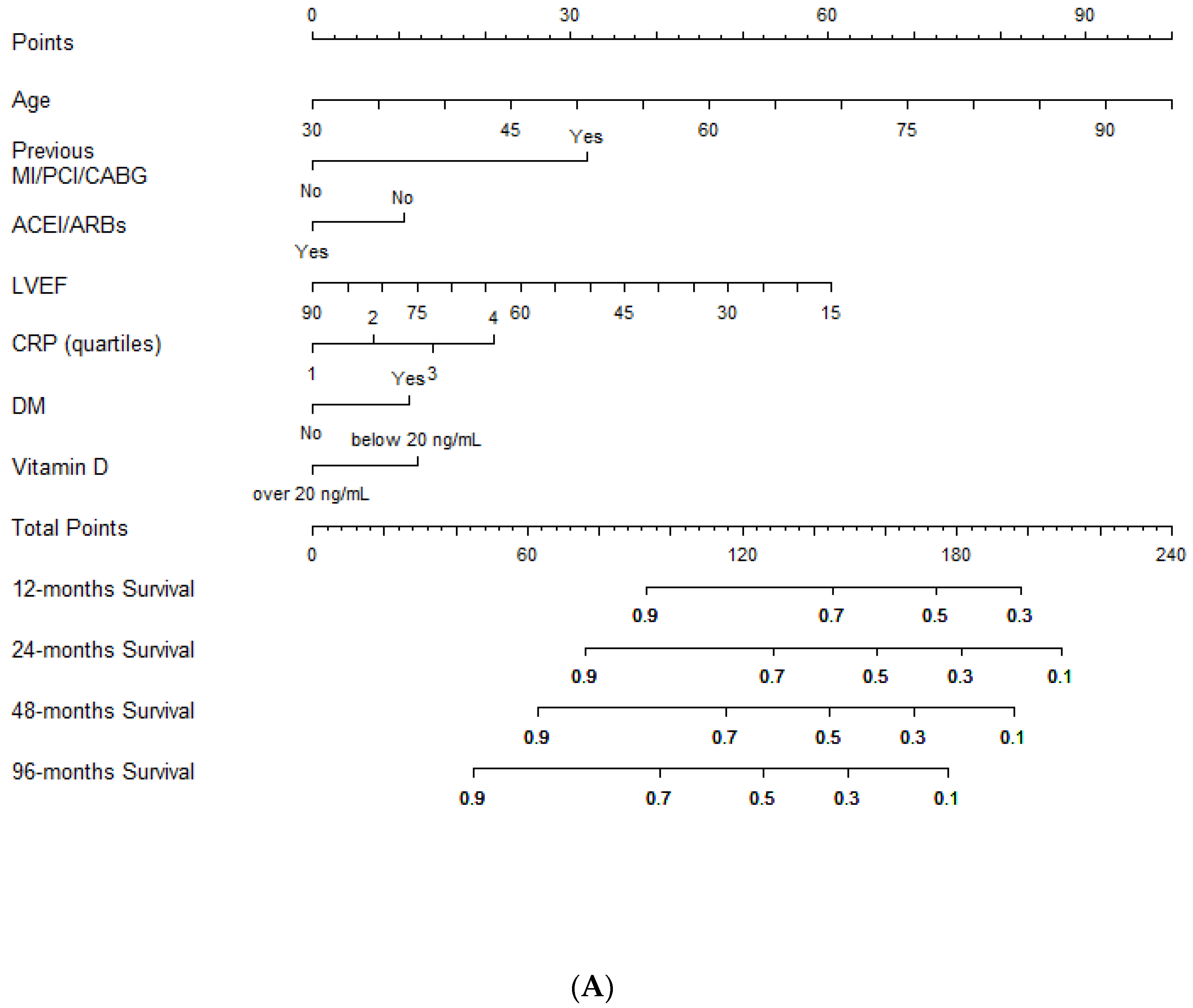
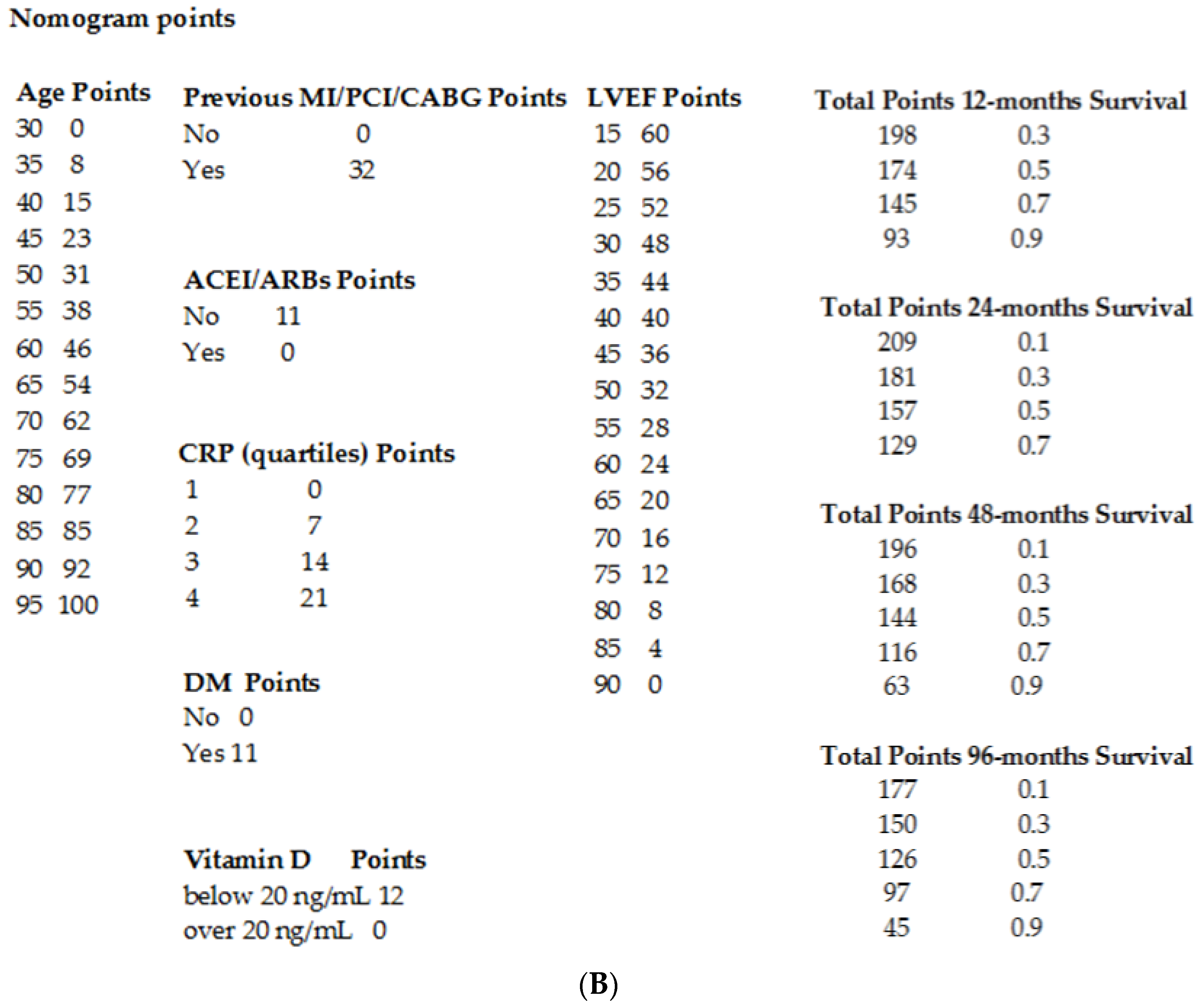
3.2. Clinical Outcome
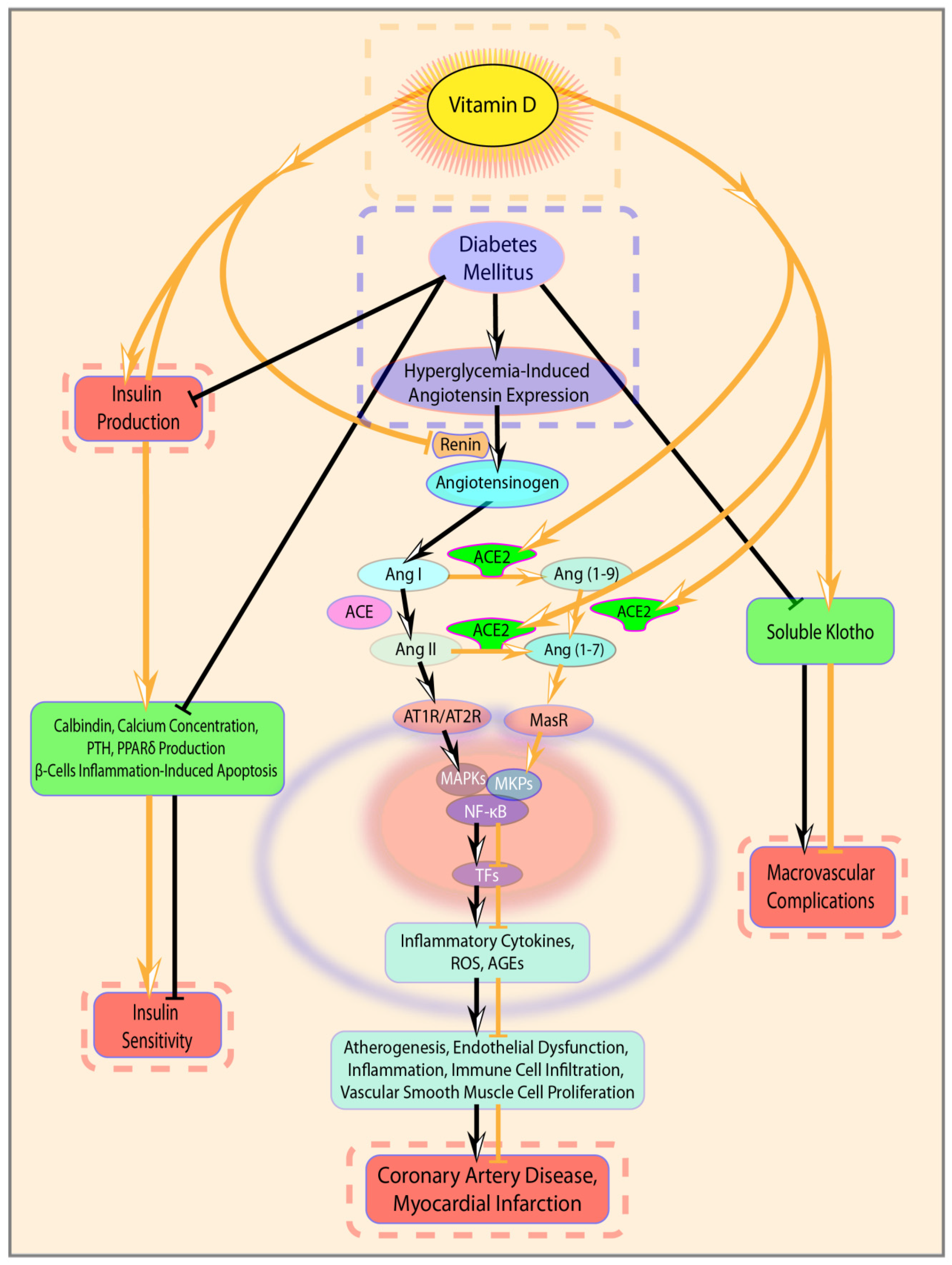
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mozaffarian, D.; Benjamin, E.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; De Ferranti, S.; Després, J.-P.; Fullerton, H.J.; Howard, V.J.; et al. Heart disease and stroke statistics—2015 update: A report from the American Heart Association. Circulation 2015, 131, e29–e322. [Google Scholar] [CrossRef]
- The Emerging Risk Factors Collaboration; Emerging Risk Factors Collaboration; Sarwar, N.; Gao, P.; Seshasai, S.R.K.; Gobin, R.; Kaptoge, S.; Di Angelantonio, E.; Ingelsson, E.; Lawlor, D.A.; et al. Diabetes mellitus, fasting blood glucose concentration, and risk of vascular disease: A collaborative meta-analysis of 102 prospective studies. Lancet 2010, 375, 2215–2222. [Google Scholar] [CrossRef]
- Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.E.; Grobbee, D.; Hansen, T.B.; et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur. Heart J. 2019, 41, 255–323. [Google Scholar] [CrossRef]
- Pan, A.; Wang, Y.; Talaei, M.; Hu, F.B. Relation of smoking with total mortality and cardiovascular events among patients with diabetes mellitus: A meta-analysis and systematic review. Circulation 2015, 132, 1795–1804. [Google Scholar] [CrossRef]
- Dziedzic, E.A.; Gąsior, J.S.; Pawłowski, M.; Dąbrowski, M. Association of Vitamin D deficiency and degree of coronary artery disease in cardiac patients with Type 2 diabetes. J. Diabetes Res. 2017, 2017, 3929075. [Google Scholar] [CrossRef]
- Aleksova, A.; Madeddu, P.; Belfiore, R.; Barbati, G.; Di Nucci, M.; Scapol, S.; De Paris, V.; Carriere, C.; Sinagra, G. U-shaped relationship between vitamin D levels and long-term outcome in large cohort of survivors of acute myocardial infarction. Int. J. Cardiol. 2016, 223, 962–966. [Google Scholar] [CrossRef]
- Nardin, M.; Verdoia, M.; Schaffer, A.; Barbieri, L.; Marino, P.; De Luca, G. Vitamin D status, diabetes mellitus and coronary artery disease in patients undergoing coronary angiography. Atherosclerosis 2016, 250, 114–121. [Google Scholar] [CrossRef]
- Jayashri, R.; Venkatesan, U.; Rani, C.S.S.; Deepa, M.; Anjana, R.M.; Mohan, V.; Pradeepa, R. Prevalence of vitamin D deficiency in urban south Indians with different grades of glucose tolerance. Br. J. Nutr. 2020, 124, 209–216. [Google Scholar] [CrossRef]
- Joergensen, C.; Gall, M.-A.; Schmedes, A.; Tarnow, L.; Parving, H.-H.; Rossing, P. Vitamin D levels and mortality in type 2 diabetes. Diabetes Care 2010, 33, 2238–2243. [Google Scholar] [CrossRef]
- Afarid, M.; Ghattavi, N.; Johari, M.K. Serum Levels of Vitamin D in Diabetic Patients with and without Retinopathy. J. Ophthalmic Vis. Res. 2020, 15, 172–177. [Google Scholar] [CrossRef]
- George, P.; Pearson, E.R.; Witham, M.D. Effect of vitamin D supplementation on glycaemic control and insulin resistance: A systematic review and meta-analysis. Diabet. Med. 2012, 29, e142–e150. [Google Scholar] [CrossRef] [PubMed]
- Cui, C.; Xu, P.; Li, G.; Qiao, Y.; Han, W.; Geng, C.; Liao, D.; Yang, M.; Chen, D.; Jiang, P. Vitamin D receptor activation regulates microglia polarization and oxidative stress in spontaneously hypertensive rats and angiotensin II-exposed microglial cells: Role of renin-angiotensin system. Redox Biol. 2019, 26, 101295. [Google Scholar] [CrossRef] [PubMed]
- Padoan, L.; Madeddu, P.; Stenner, E.; Beleù, A.; Ruscio, M.; Sinagra, G.; Aleksova, A. Left ventricular adverse remodeling after myocardial infarction and its association with vitamin D levels. Int. J. Cardiol. 2019, 277, 159–165. [Google Scholar] [CrossRef]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef]
- Lips, P.; Cashman, K.D.; Lamberg-Allardt, C.; Bischoff-Ferrari, H.A.; Obermayer-Pietsch, B.R.; Bianchi, M.L.; Stepan, J.; Fuleihan, G.E.-H.; Bouillon, R.; Lamberg-Allardt, C. Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: A position statement of the European Calcified Tissue Society. Eur. J. Endocrinol. 2019, 180, P23–P54. [Google Scholar] [CrossRef]
- Ibanez, B.; James, S.; Agewall, S.; Antunes, M.J.; Bucciarelli-Ducci, C.; Bueno, H.; Caforio, A.L.P.; Crea, F.; Goudevenos, J.A.; Halvorsen, S.; et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur. Heart J. 2018, 39, 119–177. [Google Scholar] [CrossRef] [PubMed]
- Roffi, M.; Patrono, C.; Collet, J.-P.; Mueller, C.; Valgimigli, M.; Andreotti, F.; Bax, J.J.; Borger, M.A.; Brotons, C.; Chew, D.P.; et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur. Heart J. 2016, 37, 267–315. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; González-Juanatey, J.R.; Harjola, V.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar]
- Gray, R. A Class of K-Sample Tests for Comparing the Cumulative Incidence of a Competing Risk. Ann. Stat. 1988, 16, 1141–1154. [Google Scholar] [CrossRef]
- Harrel, F.E. Regression Modeling Strategies with Applications to Linear Model, Logistic Regression and Survival Analysis; Springer: New York, NY, USA, 2001. [Google Scholar]
- Roth, D.E.; Abrams, S.A.; Aloia, J.; Bergeron, G.; Bourassa, M.W.; Brown, K.H.; Calvo, M.S.; Cashman, K.D.; Combs, G.; De-Regil, L.M.; et al. Global prevalence and disease burden of vitamin D deficiency: A roadmap for action in low- and middle-income countries. Ann. N. Y. Acad. Sci. 2018, 1430, 44–79. [Google Scholar] [CrossRef]
- WHO. Global Report on Diabetes; World Health Organizatio: Geneva, Switzerland, 2018. [Google Scholar]
- Syal, S.K.; Kapoor, A.; Bhatia, E.; Sinha, A.; Kumar, S.; Tewari, S.; Garg, N.; Goel, P.K. Vitamin D deficiency, coronary artery disease, and endothelial dysfunction: Observations from a coronary angiographic study in Indian patients. J. Invasive Cardiol. 2012, 24, 385–389. [Google Scholar] [PubMed]
- Cigolini, M.; Iagulli, M.P.; Miconi, V.; Galiotto, M.; Lombardi, S.; Targher, G. Serum 25-hydroxyvitamin D3 concentrations and prevalence of cardiovascular disease among type 2 diabetic patients. Diabetes Care 2006, 29, 722–724. [Google Scholar] [CrossRef] [PubMed]
- Forouhi, N.G.; Luan, J.; Cooper, A.; Boucher, B.J.; Wareham, N.J. Baseline serum 25-hydroxy vitamin d is predictive of future glycemic status and insulin resistance: The Medical Research Council Ely Prospective Study 1990–2000. Diabetes 2008, 57, 2619–2625. [Google Scholar] [CrossRef] [PubMed]
- Afzal, S.; Bojesen, S.E.; Nordestgaard, B.G. Low 25-hydroxyvitamin D and risk of type 2 diabetes: A prospective cohort study and metaanalysis. Clin. Chem. 2013, 59, 381–391. [Google Scholar] [CrossRef]
- Zella, J.B.; DeLuca, H.F. Vitamin D and autoimmune diabetes. J. Cell. Biochem. 2003, 88, 216–222. [Google Scholar] [CrossRef]
- Takiishi, T.; Ding, L.; Baeke, F.; Spagnuolo, I.; Sebastiani, G.; Laureys, J.; Verstuyf, A.; Carmeliet, G.; Dotta, F.; Van Belle, T.L.; et al. Dietary supplementation with high doses of regular vitamin D3 safely reduces diabetes incidence in NOD mice when given early and long term. Diabetes 2014, 63, 2026–2036. [Google Scholar] [CrossRef]
- Maestro, B.; Molero, S.; Bajo, S.; Davila, N.; Calle, C. Transcriptional activation of the human insulin receptor gene by 1,25-dihydroxyvitamin D3. Cell Biochem. Funct. 2002, 20, 227–232. [Google Scholar] [CrossRef]
- Maestro, B.; Dávila, N.; Carranza, M.C.; Calle, C. Identification of a Vitamin D response element in the human insulin receptor gene promoter. J. Steroid Biochem. Mol. Biol. 2003, 84, 223–230. [Google Scholar] [CrossRef]
- Kadowaki, S.; Norman, A.W. Pancreatic vitamin D-dependent calcium binding protein: Biochemical properties and response to vitamin D. Arch. Biochem. Biophys. 1984, 233, 228–236. [Google Scholar] [CrossRef]
- Fadda, G.Z.; Akmal, M.; Lipson, L.G.; Massry, S.G. Direct effect of parathyroid hormone on insulin secretion from pancreatic islets. Am. J. Physiol. Metab. 1990, 258, E975–E984. [Google Scholar] [CrossRef]
- Dunlop, T.W.; Väisänen, S.; Frank, C.; Molnár, F.; Sinkkonen, L.; Carlberg, C. The human peroxisome proliferator-activated receptor delta gene is a primary target of 1alpha,25-dihydroxyvitamin D3 and its nuclear receptor. J. Mol. Biol. 2005, 349, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Giulietti, A.; Van Etten, E.; Overbergh, L.; Stoffels, K.; Bouillon, R.; Mathieu, C. Monocytes from type 2 diabetic patients have a pro-inflammatory profile. 1,25-Dihydroxyvitamin D(3) works as anti-inflammatory. Diabetes Res. Clin. Pr. 2007, 77, 47–57. [Google Scholar] [CrossRef]
- Nowotny, K.; Jung, T.; Höhn, A.; Weber, D.; Grune, T. Advanced glycation end products and oxidative stress in type 2 diabetes mellitus. Biomolecules 2015, 5, 194–222. [Google Scholar] [CrossRef] [PubMed]
- Durvasula, R.V.; Shankland, S.J. Activation of a local renin angiotensin system in podocytes by glucose. Am. J. Physiol. Renal Physiol. 2008, 294, F830–F839. [Google Scholar] [CrossRef] [PubMed]
- Valente, A.J.; Yoshida, T.; Murthy, S.N.; Sakamuri, S.S.V.P.; Katsuyama, M.; Clark, R.A.; Delafontaine, P.; Chandrasekar, B. Angiotensin II enhances AT1-Nox1 binding and stimulates arterial smooth muscle cell migration and proliferation through AT1, Nox1, and interleukin-18. Am. J. Physiol. Heart Circ. Physiol. 2012, 303, H282–H296. [Google Scholar] [CrossRef]
- Fatkhullina, A.R.; Peshkova, I.O.; Koltsova, E.K. The Role of Cytokines in the Development of Atherosclerosis. Biochemistry 2016, 81, 1358–1370. [Google Scholar] [CrossRef] [PubMed]
- Ohsawa, M.; Koyama, T.; Yamamoto, K.; Hirosawa, S.; Kamei, S.; Kamiyama, R. 1alpha,25-dihydroxyvitamin D(3) and its potent synthetic analogs downregulate tissue factor and upregulate thrombomodulin expression in monocytic cells, counteracting the effects of tumor necrosis factor and oxidized LDL. Circulation 2000, 102, 2867–2872. [Google Scholar] [CrossRef] [PubMed]
- Al-Rasheed, N.M.; Al-Rasheed, N.M.; Bassiouni, Y.A.; Hasan, I.H.; Al-Amin, M.A.; Al-Ajmi, H.N.; Mohamad, R.A. Vitamin D attenuates pro-inflammatory TNF-alpha cytokine expression by inhibiting NF-small ka, CyrillicB/p65 signaling in hypertrophied rat hearts. J. Physiol. Biochem. 2015, 71, 289–299. [Google Scholar] [CrossRef]
- Sharifi, A.; Vahedi, H.; Nedjat, S.; Rafiei, H.; Hosseinzadeh-Attar, M.J. Effect of single-dose injection of vitamin D on immune cytokines in ulcerative colitis patients: A randomized placebo-controlled trial. APMIS 2019, 127, 681–687. [Google Scholar] [CrossRef]
- Gombart, A.F.; Pierre, A.; Maggini, S. A Review of Micronutrients and the Immune System-Working in Harmony to Reduce the Risk of Infection. Nutrients 2020, 12, 236. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, J.; Ge, X.; Du, J.; Deb, D.K.; Li, Y.C. Vitamin D receptor inhibits nuclear factor kappaB activation by interacting with IkappaB kinase beta protein. J. Biol. Chem. 2013, 288, 19450–19458. [Google Scholar] [CrossRef]
- Deb, D.K.; Chen, Y.; Zhang, Z.; Zhang, Y.; Szeto, F.L.; Wong, K.E.; Kong, J.; Li, Y.C. 1,25-Dihydroxyvitamin D3 suppresses high glucose-induced angiotensinogen expression in kidney cells by blocking the NF-{kappa}B pathway. Am. J. Physiol. Renal Physiol. 2009, 296, F1212–F1218. [Google Scholar] [CrossRef] [PubMed]
- Turin, A.; Bax, J.J.; Doukas, D.; Joyce, C.; Lopez, J.J.; Mathew, V.; Pontone, G.; Shah, F.; Singh, S.; Wilber, D.J.; et al. Interactions Among Vitamin D, Atrial Fibrillation, and the Renin-Angiotensin-Aldosterone System. Am. J. Cardiol. 2018, 122, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Gao, P.; Zhao, T.; He, L.; Li, M.; Li, Y.; Shui, H.; Wu, X. Calcitriol regulates angiotensin-converting enzyme and angiotensin converting-enzyme 2 in diabetic kidney disease. Mol. Biol. Rep. 2016, 43, 397–406. [Google Scholar] [CrossRef]
- Riera, M.; Anguiano, L.; Clotet, S.; Ho, H.R.; Rebull, M.; Pascual, J.; Soler, M.J. Paricalcitol modulates ACE2 shedding and renal ADAM17 in NOD mice beyond proteinuria. Am. J. Physiol. Renal Physiol. 2016, 310, F534–F546. [Google Scholar] [CrossRef]
- Xu, J.; Yang, J.; Chen, J.; Luo, Q.; Zhang, Q.; Zhang, H. Vitamin D alleviates lipopolysaccharide-induced acute lung injury via regulation of the renin-angiotensin system. Mol. Med. Rep. 2017, 16, 7432–7438. [Google Scholar] [CrossRef]
- Pan, H.-C.; Chou, K.-M.; Lee, C.-C.; Yang, N.-I.; Sun, C.-Y. Circulating Klotho levels can predict long-term macrovascular outcomes in type 2 diabetic patients. Atherosclerosis 2018, 276, 83–90. [Google Scholar] [CrossRef]
- Lu, X.; Hu, M.C. Klotho/FGF23 Axis in Chronic Kidney Disease and Cardiovascular Disease. Kidney Dis. 2017, 3, 15–23. [Google Scholar] [CrossRef]
- Mencke, R.; Hillebrands, J.-L. NIGRAM consortium. The role of the anti-ageing protein Klotho in vascular physiology and pathophysiology. Ageing Res. Rev. 2017, 35, 124–146. [Google Scholar] [CrossRef] [PubMed]
- Krajisnik, T.; Björklund, P.; Marsell, R.; Ljunggren, O.; Åkerström, G.; Jonsson, K.B.; Westin, G.E.; Larsson, T. Fibroblast growth factor-23 regulates parathyroid hormone and 1a-hydroxylase expression in cultured bovine parathyroid cells. J. Endocrinol. 2007, 195, 125. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Liu, Y.; Goetz, R.; Fu, L.; Jayaraman, S.; Hu, M.C.; Moe, O.W.; Liang, G.; Li, X.; Mohammadi, M. α-Klotho is a non-enzymatic molecular scaffold for FGF23 hormone signalling. Nature 2018, 553, 461–466. [Google Scholar] [CrossRef]
- Six, I.; Okazaki, H.; Gross, P.; Cagnard, J.; Boudot, C.; Maizel, J.; Drueke, T.B.; Massy, Z.A. Direct, Acute Effects of Klotho and FGF23 on Vascular Smooth Muscle and Endothelium. PLoS ONE 2014, 9, e93423. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.C.; Shi, M.; Cho, H.J.; Adams-Huet, B.; Paek, J.; Hill, K.; Shelton, J.; Amaral, A.P.; Faul, C.; Taniguchi, M.; et al. Klotho and phosphate are modulators of pathologic uremic cardiac remodeling. J. Am. Soc. Nephrol. 2015, 26, 1290–1302. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.C.; O’Bryan, L.M.; Farrow, E.G.; Summers, L.J.; Clinkenbeard, E.L.; Roberts, J.L.; Cass, T.A.; Saha, J.; Broderick, C.; Ma, Y.L.; et al. Circulating αKlotho influences phosphate handling by controlling FGF23 production. J. Clin. Investig. 2012, 122, 4710–4715. [Google Scholar] [CrossRef] [PubMed]
- Brownstein, C.A.; Adler, F.; Nelson-Williams, C.; Iijima, J.; Li, P.; Imura, A.; Nabeshima, Y.; Reyes-Mugica, M.; Carpenter, T.O.; Lifton, R.P. A translocation causing increased α-Klotho level results in hypophosphatemic rickets and hyperparathyroidism. Proc. Natl. Acad. Sci. USA 2008, 105, 3455–3460. [Google Scholar] [CrossRef]
- Samefors, M.; Scragg, R.; Länne, T.; Vavruch, F.H.N.C.; Östgren, C.J. Association between serum 25(OH)D3 and cardiovascular morbidity and mortality in people with Type 2 diabetes: A community-based cohort study. Diabet. Med. 2017, 34, 372–379. [Google Scholar] [CrossRef]
- Lee, W.; Kim, S.-H.; Yoon, C.-H.; Suh, J.-W.; Cho, Y.-S.; Youn, T.-J.; Chae, I.-H. Impact of Long-term Glycosylated Hemoglobin in Patients with Acute Myocardial Infarction: A retrospective cohort study. Sci. Rep. 2020, 10, 6726. [Google Scholar] [CrossRef]
- Barbarawi, M.; Kheiri, B.; Zayed, Y.; Barbarawi, O.; Dhillon, H.; Swaid, B.; Yelangi, A.; Sundus, S.; Bachuwa, G.; Alkotob, M.L.; et al. Vitamin D Supplementation and Cardiovascular Disease Risks in More Than 83000 Individuals in 21 Randomized Clinical Trials: A Meta-analysis. JAMA Cardiol. 2019, 4, 765–776. [Google Scholar] [CrossRef]
- Durup, D.; Jørgensen, H.L.; Christensen, J.; Tjonneland, A.; Olsen, A.; Halkjaer, J.; Lind, B.; Heegaard, A.-M.; Schwarz, P. A Reverse J-Shaped Association Between Serum 25-Hydroxyvitamin D and Cardiovascular Disease Mortality: The CopD Study. J. Clin. Endocrinol. Metab. 2015, 100, 2339–2346. [Google Scholar] [CrossRef]
- ACE Inhibitor Myocardial Infarction Collaborative Group. Indications for ACE inhibitors in the early treatment of acute myocardial infarction: Systematic overview of individual data from 100,000 patients in randomized trials. Circulation 1998, 97, 2202–2212. [Google Scholar] [CrossRef]
- Pfeffer, M.A.; McMurray, J.J.; Velazquez, E.J.; Rouleau, J.-L.; Køber, L.; Maggioni, A.P.; Solomon, S.D.; Swedberg, K.; Van De Werf, F.; White, H.; et al. Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. N. Engl. J. Med. 2003, 349, 1893–1906. [Google Scholar] [CrossRef] [PubMed]
- Jorde, R.; Sneve, M.; Hutchinson, M.; Emaus, N.; Figenschau, Y.; Grimnes, G. Tracking of serum 25-hydroxyvitamin D levels during 14 years in a population-based study and during 12 months in an intervention study. Am. J. Epidemiol. 2010, 171, 903–908. [Google Scholar] [CrossRef] [PubMed]
| Variables | All Patients n = 1081 | HypoVitD | No HypoVitD | |||
|---|---|---|---|---|---|---|
| DM Group 1 n = 255 | No DM Group 2 n = 426 | DM Group 3 n = 106 | No DM Group 4 n = 294 | p Value | ||
| Age * | 66.7 (11.5) | 64.6 (10.53) | 67.7 (12.2) | 66.6 (8.8) | 62 (11.3) | 0.016 |
| Male gender (%) ˆ | 70.9 | 72.9 | 66.9 | 83 | 70.4 | 0.010 |
| BMI (kg/m2) †*• | 26.9 (4.4) | 28.2 (4.2) | 26.5 (4.1) | 27.3 (4.6) | 26.2 (4) | <0.001 |
| SBP (mmHg) | 136.9 (25.2) | 133.9 (24.8) | 138.4 (23.9) | 140.6 (29.8) | 131.9 (20.1) | 0.651 |
| DBP (mmHg) | 78.4 (13.9) | 78.5 (14.6) | 80.3 (14) | 79.6 (14.1) | 80.9 (11.5) | 0.748 |
| Heart rate (bpm) | 75.6 (16.8) | 76.4 (15.8) | 75.4 (13.8) | 83.4 (30.9) | 76.2 (15) | 0.308 |
| LBBB (%) | 4.4 | 5.1 | 4.9 | 5.7 | 2.7 | 0.404 |
| Cardiac arrest (%) | 2.7 | 2 | 2.2 | 1.9 | 4.1 | 0.369 |
| Diagnosis: (%) | ||||||
| STEMI | 59.9 | 57.3 | 59.6 | 55.7 | 63.9 | 0.314 |
| NSTEMI | 40.1 | 42.7 | 40.4 | 44.3 | 36.1 | |
| Killip > 1 (%) * | 21.4 | 27.8 | 21.6 | 20.8 | 15.6 | 0.007 |
| Killip Class (%) * | ||||||
| I | 78.6 | 72.2 | 78.4 | 79.2 | 84.4 | 0.071 |
| II | 15.4 | 18.4 | 16 | 14.2 | 12.6 | |
| III | 3.6 | 5.5 | 3.5 | 4.7 | 1.7 | |
| IV | 2.3 | 3.9 | 2.1 | 1.9 | 1.4 | |
| Hypertension (%) †* | 68.5 | 76.9 | 64.8 | 76.4 | 63.6 | 0.001 |
| Dislipidemia (%) †* | 56.9 | 64.3 | 54.3 | 65.1 | 51.4 | 0.004 |
| Smoking (%) | 38.9 | 38.4 | 34.9 | 36.4 | 38.9 | 0.354 |
| Family history for IHD (%) | 26.1 | 24.3 | 27.7 | 25.5 | 25.5 | 0.786 |
| Peripheral artery disease (%) †* | 9.1 | 18.8 | 5.9 | 11.3 | 4.4 | <0.001 |
| Previous MI/PTCA/CABG (%) *ˆ | 18.8 | 22 | 17.4 | 32.1 | 13.3 | <0.001 |
| Hb (g/dL) †* | 12.3 (11.2–13.6) | 12.2 (11.1–13.6) | 12.8 (11.4–14) | 12.6 (11.6–14) | 13 (12–14) | <0.001 |
| Total cholesterol (mg/dL) †*ˆ• | 183 (152.5–214.5) | 185 (147–230) | 193 (152.5–232) | 165 (148–197.5) | 193 (166.5–229.5) | <0.001 |
| HDL cholesterol (mg/dL) †*ˆ• | 43 (36–51) | 39 (31–46) | 44 (36–55) | 43 (32.5–55) | 46 (36.5–55.5) | <0.001 |
| LDL cholesterol (mg/dL) †* ˆ• | 113.6 (87.7–139.1) | 118.6 (81.6–144.4) | 119.2 (91.7–150) | 90.6 (76.1–122.9) | 124 (101.2–155.3) | <0.001 |
| Triglycerides (mg/dL) †* ˆ• | 110 (82–149.5) | 132 (100–202) | 111 (88–160.5) | 136 (95–196) | 110 (76.5–145.5) | <0.001 |
| HbA1C% †*ˆ• | 5.9 (5.5–6.5) | 7.1 (6.5–8.9) | 5.7 (5.6–6) | 6.7 (6.1–7.9) | 5.8 (5.6–6) | <0.001 |
| Creatinine (mg/dL) †* | 0.95 (0.79–1.2) | 1 (0.8–1.2) | 0.9 (0.8–1.1) | 1 (0.8–1.3) | 0.9 (0.8–1.1) | 0.026 |
| GFR mL/min/1.73 m2 per BSA † | 71.4 (54.3–92.1) | 68.4 (50.3–79.1) | 62 (51–74.6) | 61.1 (43.4–74.2) | 68.8 (57.5–83.6) | 0.081 |
| GRACE score 6 months * | 131.5 (34.4) | 122.8 (29.2) | 123.5 (29.5) | 128.8 (32) | 116.1 (34.3) | 0.020 |
| EDD_I (cm) | 2.2 (1) | 2.5 (0.5) | 2.6 (0.4) | 2.6 (0.3) | 2.6 (0.3) | 0.283 |
| ESD_I (cm) | 1.5 (0.8) | 1.7 (0.5) | 1.7 (0.4) | 1.8 (0.4) | 1.8 (0.4) | 0.862 |
| FS% | 33.7 (10.9) | 32.7 (12) | 34.8 (9.1) | 35 (13.7) | 32.2 (10.5) | 0.330 |
| LVEF% | 51.6 (11.3) | 52 (10) | 53 (9.9) | 49.7 (10.6) | 53.1 (8.01) | 0.027 |
| E/A | 0.9 (0.7–1.2) | 0.8 (0.7–1) | 0.9 (0.7–1.2) | 0.9 (0.7–1.1) | 0.9 (0.7–1.2) | 0.732 |
| E/E’ratio †*§• | 10.8 (9–14) | 11.4 (9–16) | 11 (9–13.8) | 11 (9.5–187) | 10 (8–11.1) | <0.001 |
| WMSI | 1.5 (0.4) | 1.5 (0.4) | 1.5 (0.4) | 1.5 (0.4) | 1.5 (0.3) | 0.145 |
| Mitral insufficiency: (%) | ||||||
| Absent | 37.8 | 36.3 | 38.5 | 36.7 | 38.4 | 0.801 |
| Mild | 54.8 | 55.9 | 54.3 | 54.1 | 54.8 | |
| Moderate | 6.4 | 6.1 | 6.9 | 8.2 | 5.3 | |
| Severe | 0.8 | 1.6 | 0.2 | 1 | 1.4 | |
| PCI (%) * | 66.2 | 59 | 69.1 | 64.2 | 68.9 | 0.034 |
| CABG (%) ~* | 13 | 20.7 | 9.7 | 11.3 | 11.6 | <0.001 |
| Medical therapy (%) | 20.8 | 20.3 | 21.1 | 24.5 | 19.5 | 0.734 |
| Time to PTCA (h) | 3 (2–4.6) | 3.1 (2.1–6.1) | 2.5 (2–4.4) | 2.4 (1.4–4.1) | 3.1 (2–5.1) | 0.440 |
| Multivessel disease 70% (%) †* | 38.4 | 47.4 | 36.5 | 46.2 | 30.7 | <0.001 |
| IIB/IIIA inhibitors (%) | 13.4 | 12.1 | 12.2 | 14.9 | 15.9 | 0.552 |
| NYHA class at discharge: (%) †ˆ | ||||||
| I | 83.1 | 71.8 | 87.1 | 77.1 | 89.3 | < 0.001 |
| II | 14.4 | 24.6 | 10.1 | 22.9 | 9 | |
| III | 2.3 | 3.6 | 2.6 | 0 | 1.7 | |
| IV | 0.1 | 0 | 0.2 | 0 | 0 | |
| Vitamin D (ng/mL) †~* ˆ§ | 16.7 (9.4–23.7) | 10.8 (7.2–15) | 13 (7.7–16.9) | 24.7 (22.3–29.2) | 26 (23.3–33.6) | <0.001 |
| CRP (mg/L) †~* | 22.7 (2.4–20.6) | 7.4 (2.8–22) | 6.3 (1.8–21.9) | 5.8 (1.4–14.8) | 3.9 (1.7–9.5) | <0.001 |
| ACEIs/ARBs (%) †* | 72.9 | 79.3 | 69.8 | 77.1 | 70.5 | 0.031 |
| Beta blockers (%) | 78.2 | 76.9 | 78.4 | 79 | 78.8 | 0.949 |
| Amiodarone (%) | 9.1 | 10.5 | 8.9 | 13.3 | 6.6 | 0.167 |
| Antialdosteronics (%) | 10.5 | 12.1 | 10.1 | 14.3 | 8.3 | 0.280 |
| Loop diuretics (%) †* | 23 | 33.2 | 21.3 | 25.7 | 15.6 | <0.001 |
| Aspirin (%) | 93.6 | 97.4 | 93.5 | 89.5 | 94.1 | 0.313 |
| P2Y12 inhibitors (%) | 79.3 | 76.2 | 80.3 | 72.4 | 83 | |
| Clopidogrel | 52.6 | 52.2 | 56.1 | 45.7 | 50.2 | 0.062 0.048 |
| Prasugrel | 15.8 | 13 | 15.6 | 19 | 17.3 | |
| Ticagrelor | 14.7 | 13.8 | 12.9 | 12.4 | 19 | |
| Statins (%) ˆ | 87.4 | 87.4 | 85.9 | 94.3 | 87.2 | 0.142 |
| Oral antidiabetics (%) †*ˆ | 18.9 | 53.8 | 0 | 56.2 | 1.4 | <0.001 |
| Insulin (%) †*ˆ | 8.8 | 24.3 | 0 | 29.5 | 0 | <0.001 |
| Warfarin (%) | 7.9 | 10.9 | 6.2 | 12.4 | 5.9 | 0.026 |
| Variables | HR | 95% CI | p |
|---|---|---|---|
| Age (for 1-year increase) | 1.04 | 1.03–1.05 | <0.0001 |
| Previous MI/PCI/CABG | 2.1 | 1.66–2.6 | <0.0001 |
| LVEF (for 10 point % increase) | 0.83 | 0.75–0.91 | <0.0001 |
| CRP (quartiles) | 1.18 | 1.07–1.3 | 0.001 |
| ACEI/ARBs (yes vs. no) | 0.79 | 0.63–1.005 | 0.055 |
| DM (yes vs. no) | 1.3 | 1.05–1.61 | 0.014 |
| Vitamin D (below 20 ng/mL vs. over 20 ng/mL) | 1.3 | 1.04–1.64 | 0.022 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aleksova, A.; Ferro, F.; Gagno, G.; Padoan, L.; Saro, R.; Santon, D.; Stenner, E.; Barbati, G.; Cappelletto, C.; Rossi, M.; et al. Diabetes Mellitus and Vitamin D Deficiency: Comparable Effect on Survival and a Deadly Association after a Myocardial Infarction. J. Clin. Med. 2020, 9, 2127. https://doi.org/10.3390/jcm9072127
Aleksova A, Ferro F, Gagno G, Padoan L, Saro R, Santon D, Stenner E, Barbati G, Cappelletto C, Rossi M, et al. Diabetes Mellitus and Vitamin D Deficiency: Comparable Effect on Survival and a Deadly Association after a Myocardial Infarction. Journal of Clinical Medicine. 2020; 9(7):2127. https://doi.org/10.3390/jcm9072127
Chicago/Turabian StyleAleksova, Aneta, Federico Ferro, Giulia Gagno, Laura Padoan, Riccardo Saro, Daniela Santon, Elisabetta Stenner, Giulia Barbati, Chiara Cappelletto, Maddalena Rossi, and et al. 2020. "Diabetes Mellitus and Vitamin D Deficiency: Comparable Effect on Survival and a Deadly Association after a Myocardial Infarction" Journal of Clinical Medicine 9, no. 7: 2127. https://doi.org/10.3390/jcm9072127
APA StyleAleksova, A., Ferro, F., Gagno, G., Padoan, L., Saro, R., Santon, D., Stenner, E., Barbati, G., Cappelletto, C., Rossi, M., Beltrami, A. P., & Sinagra, G. (2020). Diabetes Mellitus and Vitamin D Deficiency: Comparable Effect on Survival and a Deadly Association after a Myocardial Infarction. Journal of Clinical Medicine, 9(7), 2127. https://doi.org/10.3390/jcm9072127









