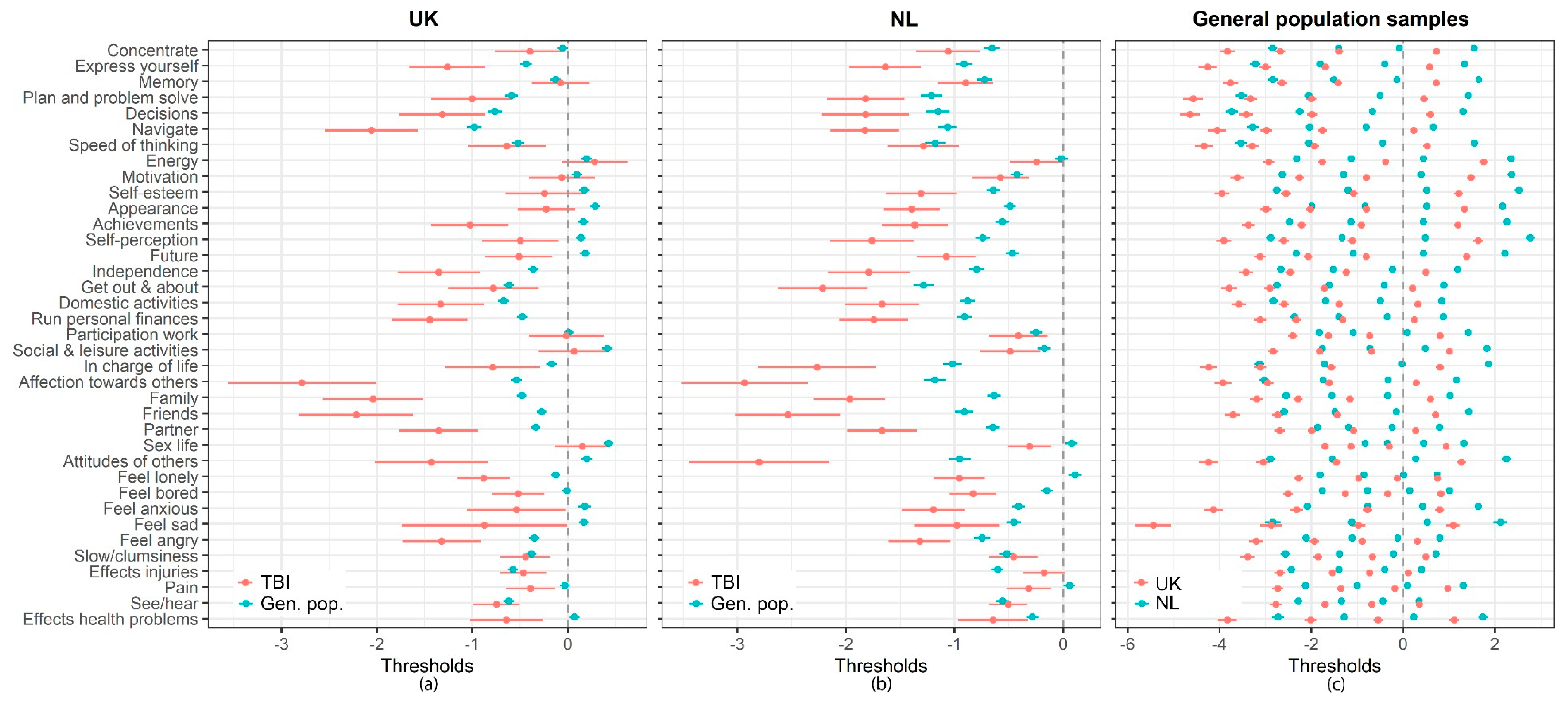
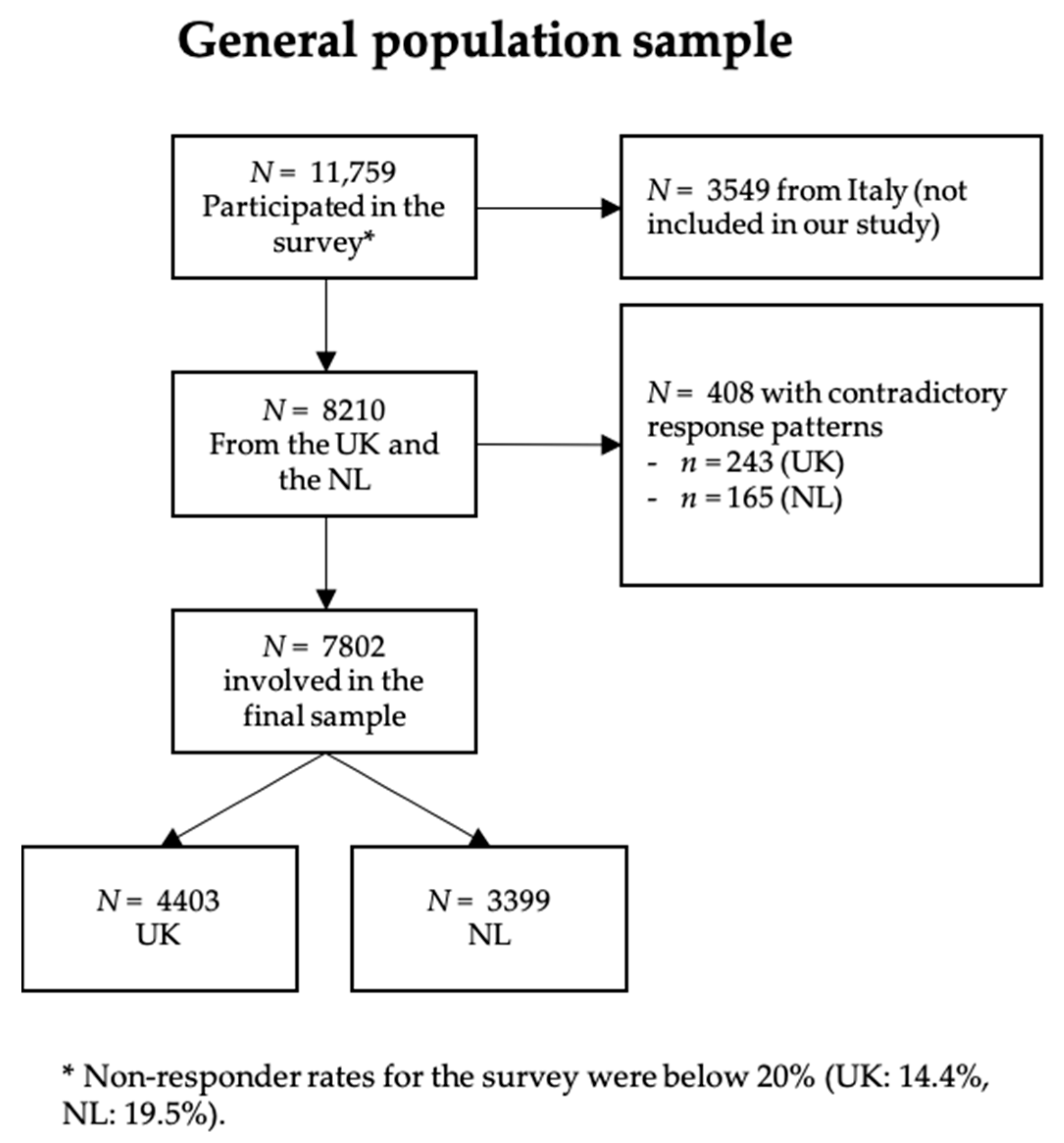
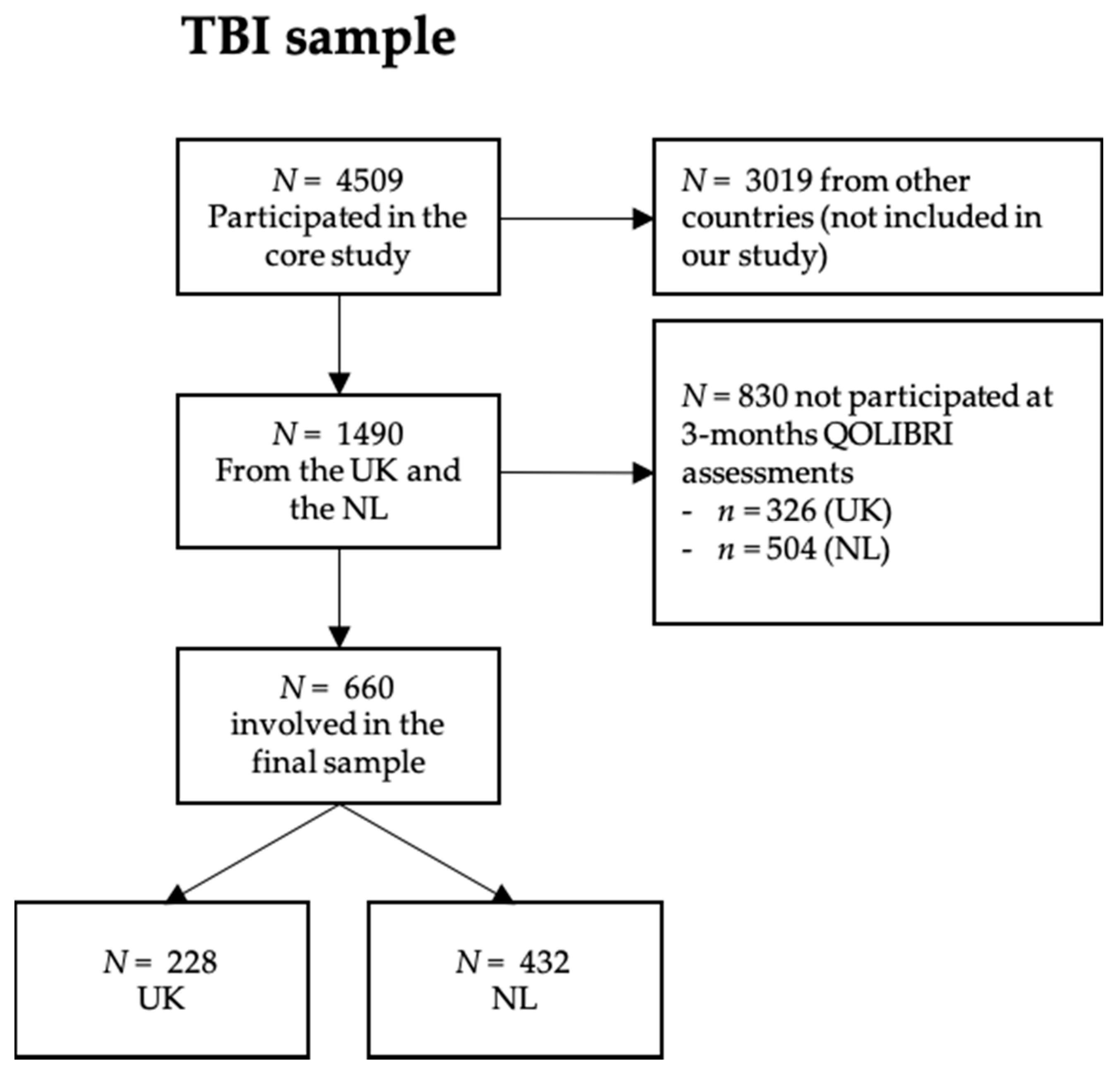
Appendix B. The CENTER-TBI Participants and Investigators
Cecilia Åkerlund 1, Krisztina Amrein 2, Nada Andelic 3, Lasse Andreassen 4, Audny Anke 5, Anna Antoni 6, Gérard Audibert 7, Philippe Azouvi 8, Maria Luisa Azzolini 9, Ronald Bartels 10, Pál Barzó 11, Romuald Beauvais 12, Ronny Beer 13, Bo-Michael Bellander 14, Antonio Belli 15, Habib Benali 16, Maurizio Berardino 17, Luigi Beretta 9, Morten Blaabjerg 18, Peter Bragge 19, Alexandra Brazinova 20, Vibeke Brinck 21, Joanne Brooker 22, Camilla Brorsson 23, Andras Buki 24, Monika Bullinger 25, Manuel Cabeleira 26, Alessio Caccioppola 27, Emiliana Calappi 27, Maria Rosa Calvi 9, Peter Cameron 28, Guillermo Carbayo Lozano 29, Marco Carbonara 27, Simona Cavallo 17, Giorgio Chevallard 30, Arturo Chieregato 30, Giuseppe Citerio 31,32, Iris Ceyisakar 33, Hans Clusmann 34, Mark Coburn 35, Jonathan Coles 36, Jamie D. Cooper 37, Marta Correia 38, Amra Čović 39, Nicola Curry 40, Endre Czeiter 24, Marek Czosnyka 26, Claire Dahyot-Fizelier 41, Paul Dark 42, Helen Dawes 43, Véronique De Keyser 44, Vincent Degos 16, Francesco Della Corte 45, Hugo den Boogert 10, Bart Depreitere 46, Đula Đilvesi 47, Abhishek Dixit 48, Emma Donoghue 22, Jens Dreier 49, Guy-Loup Dulière 50, Ari Ercole 48, Patrick Esser 43, Erzsébet Ezer 51, Martin Fabricius 52, Valery L. Feigin 53, Kelly Foks 54, Shirin Frisvold 55, Alex Furmanov 56, Pablo Gagliardo 57, Damien Galanaud 16, Dashiell Gantner 28, Guoyi Gao 58, Pradeep George 59, Alexandre Ghuysen 60, Lelde Giga 61, Ben Glocker 62, Jagoš Golubovic 47, Pedro A. Gomez 63, Johannes Gratz 64, Benjamin Gravesteijn 33, Francesca Grossi 45, Russell L. Gruen 65, Deepak Gupta 66, Juanita A. Haagsma 33, Iain Haitsma 67, Raimund Helbok 13, Eirik Helseth 68, Lindsay Horton 69, Jilske Huijben 33, Peter J. Hutchinson 70, Bram Jacobs 71, Stefan Jankowski 72, Mike Jarrett 21, Ji-yao Jiang 58, Faye Johnson 73, Kelly Jones 53, Mladen Karan 47, Angelos G. Kolias 70, Erwin Kompanje 74, Daniel Kondziella 52, Evgenios Koraropoulos 48, Lars-Owe Koskinen 75, Noémi Kovács 76, Ana Kowark 35, Alfonso Lagares 63, Linda Lanyon 59, Steven Laureys 77, Fiona Lecky 78,79, Didier Ledoux 77, Rolf Lefering 80, Valerie Legrand 81, Aurelie Lejeune 82, Leon Levi 83, Roger Lightfoot 84, Hester Lingsma 33, Andrew I.R. Maas 44, Ana M. Castaño-León 63, Marc Maegele 85, Marek Majdan 20, Alex Manara 86, Geoffrey Manley 87, Costanza Martino 88, Hugues Maréchal 50, Julia Mattern 89, Catherine McMahon 90, Béla Melegh 91, David Menon 48, Tomas Menovsky 44, Ana Mikolic 33, Benoit Misset 77, Visakh Muraleedharan 59, Lynnette Murray 28, Ancuta Negru 92, David Nelson 1, Virginia Newcombe 48, Daan Nieboer 33, József Nyirádi 2, Otesile Olubukola 78, Matej Oresic 93, Fabrizio Ortolano 27, Aarno Palotie 94,95,96, Paul M. Parizel 97, Jean-François Payen 98, Natascha Perera 12, Vincent Perlbarg 16, Paolo Persona 99, Wilco Peul 100, Anna Piippo-Karjalainen 101, Matti Pirinen 94, Horia Ples 92, Suzanne Polinder 33, Inigo Pomposo 29, Jussi P. Posti 102, Louis Puybasset 103, Andreea Radoi 104, Arminas Ragauskas 105, Rahul Raj 101, Malinka Rambadagalla 106, Jonathan Rhodes 107, Sylvia Richardson 108, Sophie Richter 48, Samuli Ripatti 94, Saulius Rocka 105, Cecilie Roe 109, Olav Roise 110,111, Jonathan Rosand 112, Jeffrey V. Rosenfeld 113, Christina Rosenlund 114, Guy Rosenthal 56, Rolf Rossaint 35, Sandra Rossi 99, Daniel Rueckert 62, Martin Rusnák 115, Juan Sahuquillo 104, Oliver Sakowitz 89,116, Renan Sanchez-Porras 116, Janos Sandor 117, Nadine Schäfer 80, Silke Schmidt 118, Herbert Schoechl 119, Guus Schoonman 120, Rico Frederik Schou 121, Elisabeth Schwendenwein 6, Charlie Sewalt 33, Toril Skandsen 122,123, Peter Smielewski 26, Abayomi Sorinola 124, Emmanuel Stamatakis 48, Simon Stanworth 40, Robert Stevens 125, William Stewart 126, Ewout W. Steyerberg 33,127, Nino Stocchetti 128, Nina Sundström 129, Anneliese Synnot 22,130, Riikka Takala 131, Viktória Tamás 124, Tomas Tamosuitis 132, Mark Steven Taylor 20, Braden Te Ao 53, Olli Tenovuo 102, Alice Theadom 53, Matt Thomas 86, Dick Tibboel 133, Marjolein Timmers 74, Christos Tolias 134, Tony Trapani 28, Cristina Maria Tudora 92, Peter Vajkoczy 135, Shirley Vallance 28, Egils Valeinis 61, Zoltán Vámos 51, Mathieu van der Jagt 136, Gregory Van der Steen 44, Joukje van der Naalt 71, Jeroen T.J.M. van Dijck 100, Thomas A. van Essen 100, Wim Van Hecke 137, Caroline van Heugten 138, Dominique Van Praag 139, Thijs Vande Vyvere 137, Roel P. J. van Wijk 100, Alessia Vargiolu 32, Emmanuel Vega 82, Kimberley Velt 33, Jan Verheyden 137, Paul M. Vespa 140, Anne Vik 122,141, Rimantas Vilcinis 132, Victor Volovici 67, Nicole von Steinbüchel 39, Daphne Voormolen 33, Petar Vulekovic 47, Kevin K.W. Wang 142, Eveline Wiegers 33, Guy Williams 48, Lindsay Wilson 69, Stefan Winzeck 48, Stefan Wolf 143, Zhihui Yang 142, Peter Ylén 144, Alexander Younsi 89, Frederick A. Zeiler 48,145, Veronika Zelinkova 20, Agate Ziverte 61, Tommaso Zoerle 27
1 Department of Physiology and Pharmacology, Section of Perioperative Medicine and Intensive Care, Karolinska Institutet, 17176, Stockholm, Sweden
2 János Szentágothai Research Centre, University of Pécs, 7622, Pécs, Hungary
3 Division of Surgery and Clinical Neuroscience, Department of Physical Medicine and Rehabilitation, Oslo University Hospital and University of Oslo, 0424, Oslo, Norway
4 Department of Neurosurgery, University Hospital Northern Norway, 9019, Tromso, Norway
5 Department of Physical Medicine and Rehabilitation, University Hospital Northern Norway, 9019, Tromso, Norway
6 Trauma Surgery, Medical University Vienna, 1090, Vienna, Austria
7 Department of Anesthesiology & Intensive Care, University Hospital Nancy, 54000, Nancy, France
8 Raymond Poincare hospital, Assistance Publique—Hopitaux de Paris, 75013, Paris, France
9 Department of Anesthesiology & Intensive Care, San Raffaele University Hospital, 20132, Milan, Italy
10 Department of Neurosurgery, Radboud University Medical Center, 6500 HB, Nijmegen, The Netherlands
11 Department of Neurosurgery, University of Szeged, 6720, Szeged, Hungary
12 International Projects Management, ARTTIC, 80333, Munchen, Germany
13 Department of Neurology, Neurological Intensive Care Unit, Medical University of Innsbruck, 6020, Innsbruck, Austria
14 Department of Neurosurgery & Anesthesia & intensive care medicine, Karolinska University Hospital, 17176, Stockholm, Sweden
15 NIHR Surgical Reconstruction and Microbiology Research Centre, B15 2TH, Birmingham, UK
16 Anesthesie-Réanimation, Assistance Publique—Hopitaux de Paris, 75013, Paris, France
17 Department of Anesthesia & ICU, AOU Città della Salute e della Scienza di Torino—Orthopedic and Trauma Center, 10126, Torino, Italy
18 Department of Neurology, Odense University Hospital, 5000, Odense, Denmark
19 BehaviourWorks Australia, Monash Sustainability Institute, Monash University, Victoria, Australia, 3004, Melbourne, Australia
20 Department of Public Health, Faculty of Health Sciences and Social Work, Trnava University, 91843, Trnava, Slovakia
21 Quesgen Systems Inc., Burlingame, CA 94010, California, CA, USA
22 Australian & New Zealand Intensive Care Research Centre, Department of Epidemiology and Preventive Medicine, School of Public Health and Preventive Medicine, Monash University, 3004, Melbourne, Australia
23 Department of Surgery and Perioperative Science, Umeå University, 90185, Umeå, Sweden
24 Department of Neurosurgery, Medical School, University of Pécs, Hungary and Neurotrauma Research Group, János Szentágothai Research Centre, University of Pécs, Hungary, 7622, Pécs, Hungary
25 Department of Medical Psychology, Universitätsklinikum Hamburg-Eppendorf, 20251, Hamburg, Germany
26 Brain Physics Lab, Division of Neurosurgery, Department of Clinical Neurosciences, University of Cambridge, Addenbrooke’s Hospital, CB2 0QQ, Cambridge, UK
27 Neuro ICU, Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico, 20122, Milan, Italy
28 ANZIC Research Centre, Department of Epidemiology and Preventive Medicine, Monash University, Melbourne, Victoria, Australia, 3004, Melbourne, Australia
29 Department of Neurosurgery, Hospital of Cruces, 48903, Bilbao, Spain
30 NeuroIntensive Care, Niguarda Hospital, 20122, Milan, Italy
31 School of Medicine and Surgery, Università Milano Bicocca, 20126, Milano, Italy
32 NeuroIntensive Care, ASST di Monza, 20900, Monza, Italy
33 Department of Public Health, Erasmus Medical Center-University Medical Center, Rotterdam, The Netherlands
34 Department of Neurosurgery, Medical Faculty RWTH Aachen University, 52074, Aachen, Germany
35 Department of Anaesthesiology, University Hospital of Aachen, 52074, Aachen, Germany
36 Department of Anesthesia & Neurointensive Care, Cambridge University Hospital NHS Foundation Trust, CB2 0QQ. Cambridge, UK
37 School of Public Health & PM, Monash University and The Alfred Hospital, Melbourne, Victoria, Australia, 3004, Melbourne, Australia
38 Radiology/MRI department, MRC Cognition and Brain Sciences Unit, CB 0QQ, Cambridge, UK
39 Institute of Medical Psychology and Medical Sociology, Universitätsmedizin Göttingen, 37073, Göttingen, Germany
40 Oxford University Hospitals NHS Trust, OX 0BP, Oxford, UK
41 Intensive Care Unit, CHU Poitiers, 86021, Potiers, France
42 Critical Care Directorate, Salford Royal Hospital NHS Foundation Trust, University of Manchester NIHR Biomedical Research Centre, M5 5AP, Salford, UK
43 Movement Science Group, Faculty of Health and Life Sciences, Oxford Brookes University, OX3 DU, Oxford, UK
44 Department of Neurosurgery, Antwerp University Hospital and University of Antwerp, 2650, Edegem, Belgium
45 Department of Anesthesia & Intensive Care, Maggiore Della Carità Hospital, 28100, Novara, Italy
46 Department of Neurosurgery, University Hospitals Leuven, 3000, Leuven, Belgium
47 Department of Neurosurgery, Clinical centre of Vojvodina, Faculty of Medicine, University of Novi Sad, 21000, Novi Sad, Serbia
48 Division of Anaesthesia, University of Cambridge, Addenbrooke’s Hospital, CB2 0QQ, Cambridge, UK
49 Center for Stroke Research Berlin, Charité—Universitätsmedizin Berlin, corporate member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, 13353, Berlin, Germany
50 Intensive Care Unit, CHR Citadelle, 4000, Liège, Belgium
51 Department of Anaesthesiology and Intensive Therapy, University of Pécs, 7622, Pécs, Hungary
52 Departments of Neurology, Clinical Neurophysiology and Neuroanesthesiology, Region Hovedstaden Rigshospitalet, 2100, Copenhagen, Denmark
53 National Institute for Stroke and Applied Neurosciences, Faculty of Health and Environmental Studies, Auckland University of Technology, 1010, Auckland, New Zealand
54 Department of Neurology, Erasmus MC, 3015 GD Rotterdam, the Netherlands
55 Department of Anesthesiology and Intensive care, University Hospital Northern Norway, 9019, Tromso, Norway
56 Department of Neurosurgery, Hadassah-hebrew University Medical center, 91120, Jerusalem, Israel
57 Fundación Instituto Valenciano de Neurorrehabilitación (FIVAN), 46005, Valencia, Spain
58 Department of Neurosurgery, Shanghai Renji Hospital, School of Medicine, Shanghai Jiaotong University, 200127, Shanghai, China
59 Karolinska Institutet, INCF International Neuroinformatics Coordinating Facility, 17176, Stockholm, Sweden
60 Emergency Department, CHU, 4000, Liège, Belgium
61 Neurosurgery clinic, Pauls Stradins Clinical University Hospital, 1002, Riga, Latvia
62 Department of Computing, Imperial College London, SW7 2BU, London, UK
63 Department of Neurosurgery, Hospital Universitario 12 de Octubre, 28041 Madrid, Spain
64 Department of Anesthesia, Critical Care and Pain Medicine, Medical University of Vienna, 1090, Vienna, Austria
65 College of Health and Medicine, Australian National University, 2601, Canberra, Australia
66 Department of Neurosurgery, Neurosciences Centre & JPN Apex trauma centre, All India Institute of Medical Sciences, 110029, New Delhi, India
67 Department of Neurosurgery, Erasmus MC, 3015 CE, Rotterdam, the Netherlands
68 Department of Neurosurgery, Oslo University Hospital, 0424, Oslo, Norway
69 Division of Psychology, University of Stirling, FK9 4LA, Stirling, UK
70 Division of Neurosurgery, Department of Clinical Neurosciences, Addenbrooke’s Hospital & University of Cambridge, CB2 0QQ, Cambridge, UK
71 Department of Neurology, University of Groningen, University Medical Center Groningen, 9700 RB, Groningen, Netherlands
72 Neurointensive Care, Sheffield Teaching Hospitals NHS Foundation Trust, S10 2JF, Sheffield, UK
73 Salford Royal Hospital NHS Foundation Trust Acute Research Delivery Team, M5 5AP, Salford, UK
74 Department of Intensive Care and Department of Ethics and Philosophy of Medicine, Erasmus Medical Center, 3015 CE, Rotterdam, The Netherlands
75 Department of Clinical Neuroscience, Neurosurgery, Umeå University, 90185, Umeå, Sweden
76 Hungarian Brain Research Program—Grant No. KTIA_13_NAP-A-II/8, University of Pécs, 7622, Pécs, Hungary
77 Cyclotron Research Center, University of Liège, 4000, Liège, Belgium
78 Centre for Urgent and Emergency Care Research (CURE), Health Services Research Section, School of Health and Related Research (ScHARR), University of Sheffield, S10 2TG, Sheffield, UK
79 Emergency Department, Salford Royal Hospital, M6 (HD, Salford, UK
80 Institute of Research in Operative Medicine (IFOM), Witten/Herdecke University, 58455, Cologne, Germany
81 VP Global Project Management CNS, ICON, 92000, Paris, France
82 Department of Anesthesiology-Intensive Care, Lille University Hospital, 59037, Lille, France
83 Department of Neurosurgery, Rambam Medical Center, 31096, Haifa, Israel
84 Department of Anesthesiology & Intensive Care, University Hospitals Southhampton NHS Trust, SO16 6YD, Southhampton, UK
85 Cologne-Merheim Medical Center (CMMC), Department of Traumatology, Orthopedic Surgery and Sportmedicine, Witten/Herdecke University, 58445, Cologne, Germany
86 Intensive Care Unit, Southmead Hospital, Bristol, BS10 5NB, Bristol, UK
87 Department of Neurological Surgery, University of California, CA 94110, San Francisco, CA, USA
88 Department of Anesthesia & Intensive Care, M. Bufalini Hospital, 47521, Cesena, Italy
89 Department of Neurosurgery, University Hospital Heidelberg, 69120, Heidelberg, Germany
90 Department of Neurosurgery, The Walton centre NHS Foundation Trust, L9 7LI, Liverpool, UK
91 Department of Medical Genetics, University of Pécs, 7622, Pécs, Hungary
92 Department of Neurosurgery, Emergency County Hospital Timisoara, 300736, Timisoara, Romania
93 School of Medical Sciences, Örebro University, 70281, Örebro, Sweden
94 Institute for Molecular Medicine Finland, University of Helsinki, 00290, Helsinki, Finland
95 Analytic and Translational Genetics Unit, Department of Medicine; Psychiatric & Neurodevelopmental Genetics Unit, Department of Psychiatry; Department of Neurology, Massachusetts General Hospital, MA 02114, Boston, MA, USA
96 Program in Medical and Population Genetics; The Stanley Center for Psychiatric Research, The Broad Institute of MIT and Harvard, MA 02142, Cambridge, MA, USA
97 Department of Radiology, University of Antwerp, 2650, Edegem, Belgium
98 Department of Anesthesiology & Intensive Care, University Hospital of Grenoble, 38043, Grenoble, France
99 Department of Anesthesia & Intensive Care, Azienda Ospedaliera Università di Padova, 35100, Padova, Italy
100 Department of Neurosurgery, Leiden University Medical Center, 2300 RC, Leiden, The Netherlands and Department of Neurosurgery, Medical Center Haaglanden, 2597 AX, The Hague, The Netherlands
101 Department of Neurosurgery, Helsinki University Central Hospital, 00290, Helsinki, Finland
102 Division of Clinical Neurosciences, Department of Neurosurgery and Turku Brain Injury Centre, Turku University Hospital and University of Turku, 20521, Turku, Finland
103 Department of Anesthesiology and Critical Care, Pitié -Salpêtrière Teaching Hospital, Assistance Publique, Hôpitaux de Paris and University Pierre et Marie Curie, 75013, Paris, France
104 Neurotraumatology and Neurosurgery Research Unit (UNINN), Vall d’Hebron Research Institute, 08035, Barcelona, Spain
105 Department of Neurosurgery, Kaunas University of technology and Vilnius University, 44249, Vilnius, Lithuania
106 Department of Neurosurgery, Rezekne Hospital, 4604, Rezenke, Latvia
107 Department of Anesthesia, Critical Care & Pain Medicine NHS Lothian & University of Edinburg, EH4 2XU, Edinburgh, UK
108 Director, MRC Biostatistics Unit, Cambridge Institute of Public Health, Cb2 0SR, Cambridge, UK
109 Department of Physical Medicine and Rehabilitation, Oslo University Hospital, University of Oslo, 0424, Oslo, Norway
110 Division of Orthopedics, Oslo University Hospital, 0424, Oslo, Norway
111 Institute of Clinical Medicine, Faculty of Medicine, University of Oslo, 0424, Oslo, Norway
112 Broad Institute, Cambridge MA Harvard Medical School, Boston MA, Massachusetts General Hospital, MA 02114, Boston, MA, USA
113 National Trauma Research Institute, The Alfred Hospital, Monash University, 3004, Melbourne, Victoria, Australia
114 Department of Neurosurgery, Odense University Hospital, 5000, Odense, Denmark
115 International Neurotrauma Research Organization, 1090, Vienna, Austria
116 Klinik für Neurochirurgie, Klinikum Ludwigsburg, 71640, Ludwigsburg, Germany
117 Division of Biostatistics and Epidemiology, Department of Preventive Medicine, University of Debrecen, 4032, Debrecen, Hungary
118 Department Health and Prevention, University Greifswald, 17489, Greifswald, Germany
119 Department of Anesthesiology and Intensive Care, AUVA Trauma Hospital, 5026, Salzburg, Austria
120 Department of Neurology, Elisabeth-TweeSteden Ziekenhuis, 5022 GC, Tilburg, the Netherlands
121 Department of Neuroanesthesia and Neurointensive Care, Odense University Hospital, 5000, Odense, Denmark
122 Department of Neuromedicine and Movement Science, Norwegian University of Science and Technology, NTNU, 7491, Trondheim, Norway
123 Department of Physical Medicine and Rehabilitation, St. Olavs Hospital, Trondheim University Hospital, 7491, Trondheim, Norway
124 Department of Neurosurgery, University of Pécs, 7622, Pécs, Hungary
125 Division of Neuroscience Critical Care, John Hopkins University School of Medicine, MD 21205, Baltimore, MD, USA
126 Department of Neuropathology, Queen Elizabeth University Hospital and University of Glasgow, G51 4TF, Glasgow, UK
127 Department of Department of Biomedical Data Sciences, Leiden University Medical Center, 2300 RC, Leiden, The Netherlands
128 Department of Pathophysiology and Transplantation, Milan University, and Neuroscience ICU, Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico, 20122, Milano, Italy
129 Department of Radiation Sciences, Biomedical Engineering, Umeå University, 90185, Umeå, Sweden
130 Cochrane Consumers and Communication Review Group, Centre for Health Communication and Participation, School of Psychology and Public Health, La Trobe University, 3004, Melbourne, Australia
131 Perioperative Services, Intensive Care Medicine and Pain Management, Turku University Hospital and University of Turku, 20521, Turku, Finland
132 Department of Neurosurgery, Kaunas University of Health Sciences, 50009, Kaunas, Lithuania
133 Intensive Care and Department of Pediatric Surgery, Erasmus Medical Center, Sophia Children’s Hospital, 3015 CE, Rotterdam, The Netherlands
134 Department of Neurosurgery, Kings college London, WC2R 2LS, London, UK
135 Neurologie, Neurochirurgie und Psychiatrie, Charité—Universitätsmedizin Berlin, 13353, Berlin, Germany
136 Department of Intensive Care Adults, Erasmus MC—University Medical Center Rotterdam, 30115 CE, Rotterdam, The Netherlands
137 icoMetrix NV, 3000, Leuven, Belgium
138 Movement Science Group, Faculty of Health and Life Sciences, Oxford Brookes University, OX3 0BP, Oxford, UK
139 Psychology Department, Antwerp University Hospital, 2650, Edegem, Belgium
140 Director of Neurocritical Care, University of California, CA 90095, Los Angeles, CA, USA
141 Department of Neurosurgery, St.Olavs Hospital, Trondheim University Hospital, 7491, Trondheim, Norway
142 Department of Emergency Medicine, University of Florida, Gainesville, FL 32611, FL, USA
143 Department of Neurosurgery, Charité—Universitätsmedizin Berlin, corporate member of Freie Universität Berlin, Humboldt-Universität zu Berlin, and Berlin Institute of Health, 13353, Berlin, Germany
144 VTT Technical Research Centre, 02044, Tampere, Finland
145 Section of Neurosurgery, Department of Surgery, Rady Faculty of Health Sciences, University of Manitoba, R3E 3P5, Winnipeg, MB, Canada