Impact of Acute Hemoglobin Falls in Heart Failure Patients: A Population Study
Abstract
1. Introduction
2. Subjects and Methods
2.1. Study Population and Baseline Data Collection
2.2. Acute Anemia and Kidney Injury Assessment
2.3. Cardiovascular Risk Factors Definition
2.4. Mortality and Hospitalization
2.5. Statistical Analysis
3. Results
3.1. General Characteristics of the Study Population
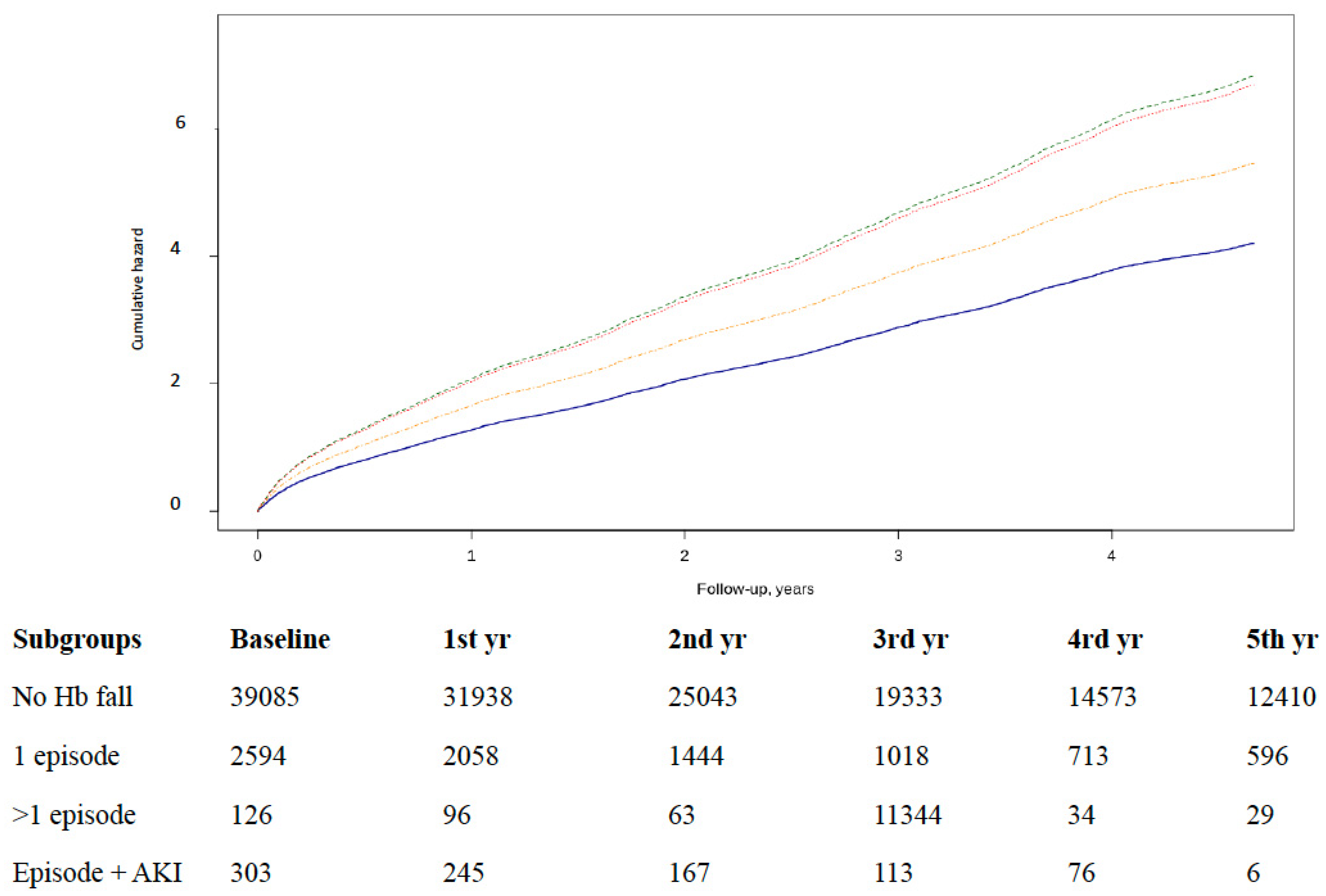
3.2. Episodes of Hb Falls and Acute Heart Failure Hospitalization
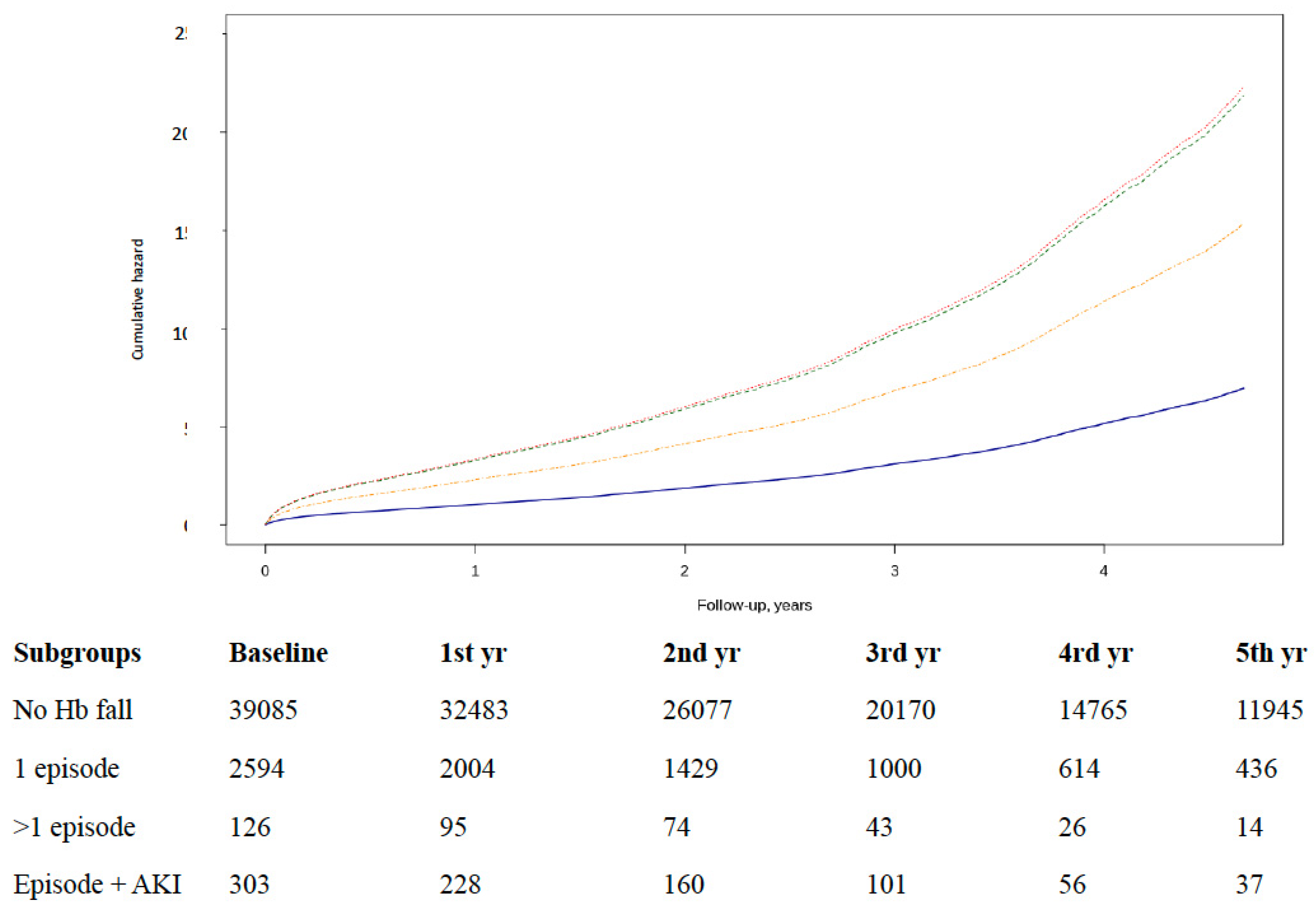
3.3. Episodes of Hb Falls and All-Cause Mortality
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roger, V.L. Epidemiology of heart failure. Circ. Res. 2013, 113, 646–659. [Google Scholar] [CrossRef]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; González-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. ESC Scientific Document Group. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200. [Google Scholar]
- Anand, I.; Gupta, P. Anemia and Iron Deficiency in Heart Failure. Circulation 2018, 138, 80–98. [Google Scholar] [CrossRef]
- Goodnough, L.T.; Schrier, S.L. Evaluation and management of anemia in the elderly. Am. J. Hematol. 2014, 89, 88–96. [Google Scholar] [CrossRef]
- Tang, Y.-D.; Katz, S.D. The prevalence of anemia in chronic heart failure and its impact on the clinical outcomes. Hear. Fail. Rev. 2008, 13, 387–392. [Google Scholar] [CrossRef]
- Van Der Meer, P.; Postmus, D.; Ponikowski, P.; Cleland, J.G.F.; O’Connor, C.M.; Cotter, G.; Metra, M.; Davison, B.; Givertz, M.M.; Mansoor, G.A.; et al. The Predictive Value of Short-Term Changes in Hemoglobin Concentration in Patients Presenting With Acute Decompensated Heart Failure. J. Am. Coll. Cardiol. 2013, 61, 1973–1981. [Google Scholar] [CrossRef]
- Magrì, D.; De Martino, F.; Moscucci, F.; Agostoni, P.; Sciomer, S. Anemia and Iron Deficiency in Heart Failure: Clinical and Prognostic Role. Hear. Fail. Clin. 2019, 15, 359–369. [Google Scholar] [CrossRef]
- Lopez, A.; Cacoub, F.; Macdougall, C.; Peyrin-Broulet, L. Iron deficiency anaemia. Lancet 2016, 387, 907–916. [Google Scholar] [CrossRef]
- Makris, K.; Spanou, L. Acute Kidney Injury: Definition, Pathophysiology and Clinical Phenotypes. Clin. Biochem. Rev. 2016, 37, 85–98. [Google Scholar]
- Chung, Y.J.; Luo, A.; Park, K.C.; Loonat, A.A.; Lakhal-Littleton, S.; Robbins, P.A.; Swietach, P. Iron-deficiency anemia reduces cardiac contraction by downregulating RyR2 channels and suppressing SERCA pump activity. JCI Insight 2019, 4. [Google Scholar] [CrossRef]
- Voigt, P.; Bussche, A.V.D. The EU General Data Protection Regulation (GDPR). In A Practical Guide, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.L.; Castro AF 3rd Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; Coresh, J. CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef] [PubMed]
- National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation classification and stratification. Am. J. Kidney Dis. 2002, 39 (Suppl. 1), S1–S266.
- Hata, N.; Yokoyama, S.; Shinada, T.; Kobayashi, N.; Shirakabe, A.; Tomita, K.; Kitamura, M.; Kurihara, O.; Takahashi, Y. Acute kidney injury and outcomes in acute decompensated heart failure: Evaluation of the RIFLE criteria in an acutely ill heart failure population. Eur. J. Hear. Fail. 2009, 12, 32–37. [Google Scholar] [CrossRef]
- Shirakabe, A.; Hata, N.; Kobayashi, N.; Shinada, T.; Tomita, K.; Tsurumi, M.; Matsushita, M.; Okazaki, H.; Yamamoto, Y.; Yokoyama, S.; et al. Long-term prognostic impact after acute kidney injury in patients with acute heart failure. Int. Hear. J. 2012, 53, 313–319. [Google Scholar] [CrossRef]
- Redon, J.; Tellez-Plaza, M.; Orozco-Beltran, D.; Gil-Guillén, V.F.; Pita-Fernández, S.; Navarro-Pérez, J.; Pallarés-Carratalá, V.; Valls, F.; Fernandez, A.; Perez-Navarro, A.M.; et al. Impact of hypertension on mortality and cardiovascular disease burden in patients with cardiovascular risk factors from a general practice setting. J. Hypertens. 2016, 34, 1–1083. [Google Scholar] [CrossRef]
- Williams, B.; Mancia, G.; Spiering, W.; Rosei, E.A.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. J. Hypertens. 2018, 36, 1953–2041. [Google Scholar] [CrossRef]
- Fortrie, G.; De Geus, H.R.H.; Betjes, M.G.H. The aftermath of acute kidney injury: A narrative review of long-term mortality and renal function. Crit. Care 2019, 23, 24. [Google Scholar] [CrossRef] [PubMed]
- Batchelor, E.K.; Kapitsinou, P.; Pergola, P.E.; Kovesdy, C.P.; Jalal, D. Iron Deficiency in Chronic Kidney Disease: Updates on Pathophysiology, Diagnosis, and Treatment. J. Am. Soc. Nephrol. 2020, 31, 456–468. [Google Scholar] [CrossRef]
- García-Compeán, D.; Del Cueto-Aguilera, Á.N.; Jiménez-Rodríguez, A.R.; González-González, J.A.; Maldonado-Garza, H.J. Diagnostic and therapeutic challenges of gastrointestinal angiodysplasias: A critical review and view points. World J. Gastroenterol. 2019, 25, 2549–2564. [Google Scholar] [CrossRef]
- Bielecka-Dabrowa, A.; Godoy, B.; Schefold, J.C.; Koziolek, M.; Banach, M.; Von Haehling, S. Decompensated Heart Failure and Renal Failure: What Is the Current Evidence? Curr. Hear. Fail. Rep. 2018, 15, 224–238. [Google Scholar] [CrossRef]
- Schefold, J.C.; Filippatos, G.; Hasenfuss, G.; Anker, S.D.; Von Haehling, S. Heart failure and kidney dysfunction: Epidemiology, mechanisms and management. Nat. Rev. Nephrol. 2016, 12, 610–623. [Google Scholar] [CrossRef]
- Berkovitch, A.; Maor, E.; Sabbag, A.; Chernomordik, F.; Elis, A.; Arbel, Y.; Goldenberg, I.; Grossman, E.; Klempfner, R. Precipitating Factors for Acute Heart Failure Hospitalization and Long-Term Survival. Medicine 2015, 94, e2330. [Google Scholar] [CrossRef] [PubMed]
- Fonarow, G.C.; Abraham, W.T.; Albert, N.M.; Stough, W.G.; Gheorghiade, M.; Greenberg, B.H.; O’Connor, C.M.; Pieper, K.; Sun, J.L.; Yancy, C.W.; et al. Factors Identified as Precipitating Hospital Admissions for Heart Failure and Clinical OutcomesFindings From OPTIMIZE-HF. Arch. Intern. Med. 2008, 168, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Martínez, F.; Martínez-Ibañez, L.; Pichler, G.; Ruiz-Hernández, A.; Redon, J. Multimorbidity and acute heart failure in internal medicine. Int. J. Cardiol. 2017, 232, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Komajda, M.; Anker, S.; Charlesworth, A.; Okonko, D.; Metra, M.; Di Lenarda, A.; Remme, W.; Moullet, C.; Swedberg, K.; Cleland, J.G.F.; et al. The impact of new onset anaemia on morbidity and mortality in chronic heart failure: Results from COMET. Eur. Hear. J. 2006, 27, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Parcha, V.; Patel, N.; Kalra, R.; Bhargava, A.; Prabhu, S.D.; Arora, G.; Arora, P. Clinical, Demographic, and Imaging Correlates of Anemia in Heart Failure with Preserved Ejection Fraction (from the RELAX Trial). Am. J. Cardiol. 2020, 125, 1870–1878. [Google Scholar] [CrossRef]
| All Subjects | No Hb Falls | One Episode Hb Fall | Two or More Hb Falls | AKI and Hb Falls | |
|---|---|---|---|---|---|
| Number | 45,437 | 42,064 | 2892 | 139 | 342 |
| Sex (M) | 20,504 (45.1) | 18,568 (44.1) | 1647 (57) * | 91 (65.5) * | 198 (57.9) * |
| Body mass index (kg/m2) | 31 ± 5.7 | 31.1 ± 5.7 | 30.4 ± 5.5 * | 29.4 ± 5.6 | 30.1 ± 5.4 |
| Age at diagnosis | 74.3 ± 11.0 | 74.3 ± 11.0 | 74.6 ± 10.8 | 73.4 ± 11.2 | 74 ± 11.1 |
| eGFR (mL/min/1.73 m2) | 64.1 ± 23.3 | 64.4 ± 22.9 | 62 ± 25.8 * | 59 ± 27.3 | 48.5 ± 27.2 *,&,$ |
| CKD stage 1 >90 | 7 | 6.8 | 8.7 | 11.5 | 6.4 |
| CKD stage 290–60 | 22.6 | 22.6 | 24.4 | 17.3 | 13.5 |
| CKD stage 360–30 | 18.2 | 18 | 20.9 | 23.7 | 24.3 |
| CKD stage 430–15 | 3.3 | 3.1 | 5.9 | 7.2 | 11.4 |
| Hb at baseline (g/dL) | 12.7 ± 1.9 | 12.8 ± 1.8 | 11.4 ± 2.2 * | 10.8 ± 2.2 * | 11.5 ± 1.9 * |
| Hb before event (g/dL) | 12.7 ± 2.0 | 12.8 ± 1.9 | 11.4 + 2.2 * | 10.2 ± 2.5 * | 10.8 ± 2.3 * |
| Acute HF hospitalization | 4673 (10.3) | 4260 (10.1) | 348 (12.0) * | 23 (16.5) | 42 (12.3) |
| Mortality | 10,490 (23.1) | 9037 (21.5) | 1191 (41.2) * | 83 (59.7) *,& | 179 (52.3) *,& |
| Comorbidities | |||||
| Anemia | 24,479 (53.9) | 21,816 (51.9) | 2274 (78.6) * | 128 (92.1) *,& | 261 (76.3) *,$ |
| Diabetes | 22,199 (48.9) | 20,501 (48.7) | 1456 (50.3) | 65 (46.8) | 177 (51.8) |
| Dyslipidemia | 27,653 (60.9) | 25,732 (61.2) | 1642 (56.8) * | 78 (56.1) | 201 (58.8) |
| Hypertension | 40,663 (89.5) | 37,614 (89.4) | 2608 (90.2) | 121 (87.1) | 320 (93.6) |
| Myocardial infarction | 10,087 (22.2) | 9178 (21.8) | 772 (26.7) * | 45 (32.4) * | 92 (26.9) |
| Atrial fibrillation | 20,759 (45.7) | 18,997 (45.2) | 1498 (51.8) * | 77 (55.4) | 187 (54.7) * |
| Treatments | |||||
| Diuretics | 36,676 (80.7) | 33,817 (80.4) | 2452 (84.8) * | 118 (84.9) | 289 (84.5) |
| Beta-blockers | 22,254 (49.0) | 20,526 (48.8) | 1489 (51.5) * | 66 (47.5) | 173 (50.6) |
| ACEi/ARB | 32,415 (71.3) | 30,073 (71.5) | 2005 (69.3) | 84 (60.4) * | 253 (74) $ |
| Calcium antagonists | 12,906 (28.4) | 11,937 (28.4) | 818 (28.3) | 54 (38.8) * | 97 (28.4) |
| NSAIDs | 14,697 (32.3) | 13,764 (32.7) | 801 (27.7) * | 48 (34.5) | 84 (24.6) * |
| Antiplatelets | 20,334 (44.8) | 18,735 (44.5) | 1375 (47.5) * | 76 (54.7) | 148 (43.3) |
| Anticoagulants | 33,478 (73.7) | 30,840 (73.3) | 2261 (78.2) * | 113 (81.3) | 264 (77.2) |
| Antialdosterone | 8579 (18.9) | 7869 (18.7) | 604 (20.9) * | 25 (18) | 81 (23.7) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopez, C.; Holgado, J.L.; Fernandez, A.; Sauri, I.; Uso, R.; Trillo, J.L.; Vela, S.; Bea, C.; Nuñez, J.; Ferrer, A.; et al. Impact of Acute Hemoglobin Falls in Heart Failure Patients: A Population Study. J. Clin. Med. 2020, 9, 1869. https://doi.org/10.3390/jcm9061869
Lopez C, Holgado JL, Fernandez A, Sauri I, Uso R, Trillo JL, Vela S, Bea C, Nuñez J, Ferrer A, et al. Impact of Acute Hemoglobin Falls in Heart Failure Patients: A Population Study. Journal of Clinical Medicine. 2020; 9(6):1869. https://doi.org/10.3390/jcm9061869
Chicago/Turabian StyleLopez, Cristina, Jose Luis Holgado, Antonio Fernandez, Inmaculada Sauri, Ruth Uso, Jose Luis Trillo, Sara Vela, Carlos Bea, Julio Nuñez, Ana Ferrer, and et al. 2020. "Impact of Acute Hemoglobin Falls in Heart Failure Patients: A Population Study" Journal of Clinical Medicine 9, no. 6: 1869. https://doi.org/10.3390/jcm9061869
APA StyleLopez, C., Holgado, J. L., Fernandez, A., Sauri, I., Uso, R., Trillo, J. L., Vela, S., Bea, C., Nuñez, J., Ferrer, A., Gamez, J., Ruiz, A., & Redon, J. (2020). Impact of Acute Hemoglobin Falls in Heart Failure Patients: A Population Study. Journal of Clinical Medicine, 9(6), 1869. https://doi.org/10.3390/jcm9061869





