Effects of a Red Orange and Lemon Extract in Obese Diabetic Zucker Rats: Role of Nicotinamide Adenine Dinucleotide Phosphate Oxidase
Abstract
1. Introduction
2. Experimental Section
2.1. Chemicals and Reagents
2.2. Ethics Statement
2.3. Experimental Protocol
2.4. RNA Extraction and Complementary DNA (cDNA) Synthesis
2.5. Selection of Gene of Interest and Primer Design
2.6. Reverse Transcription-Quantitative Polymerase Chain Reaction (RT-qPCR)
2.7. Western Blot Analysis
2.8. Evaluation of DNA Damage
2.9. Statistical Analysis
3. Results
3.1. Physiological Parameters: The Effect of RLE Treatment on Blood Pressure, and Food and Water Intake
3.2. Gene Expression Results
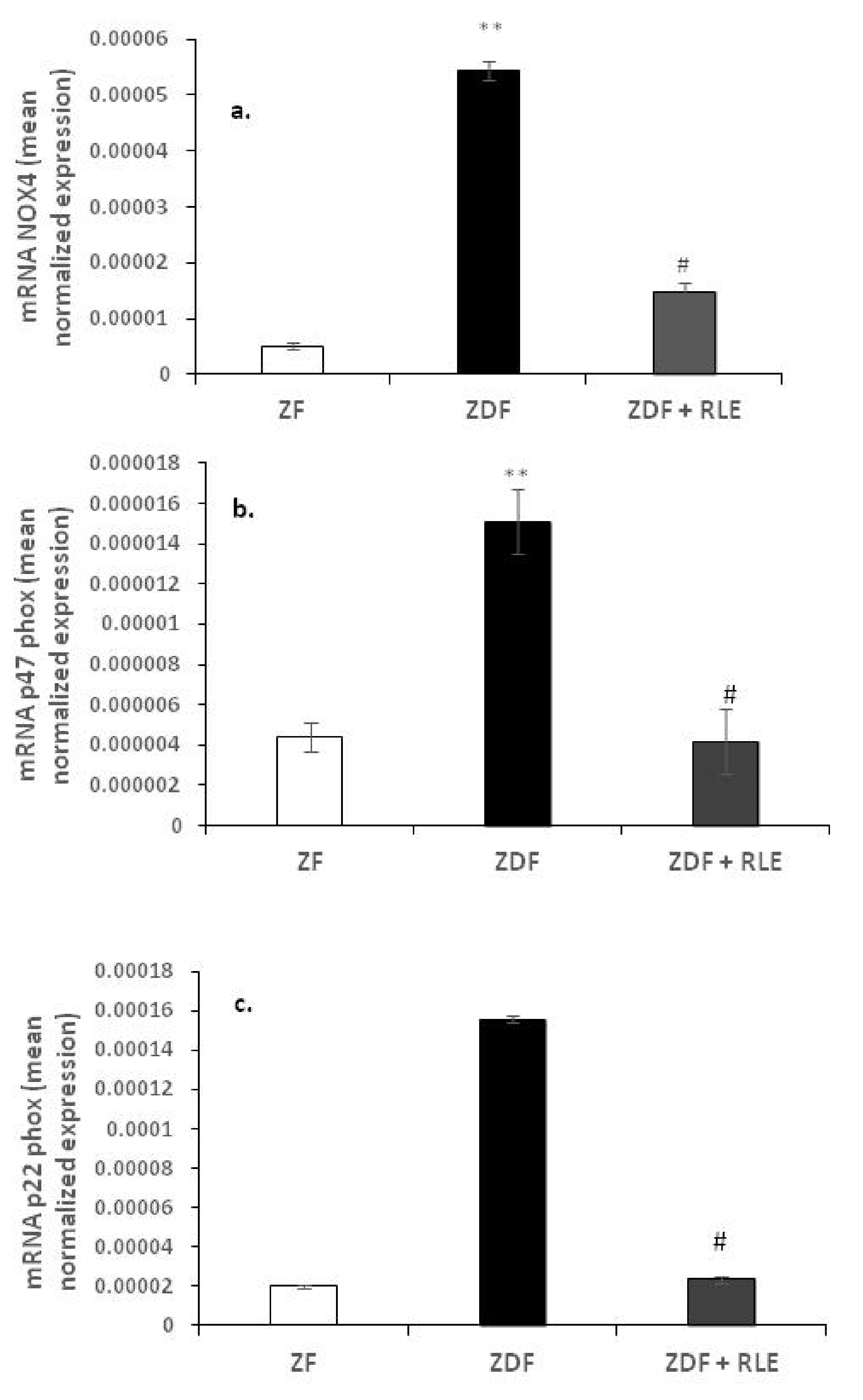
3.2.1. Nox 4, p22-phox, and p47-phox Gene Results
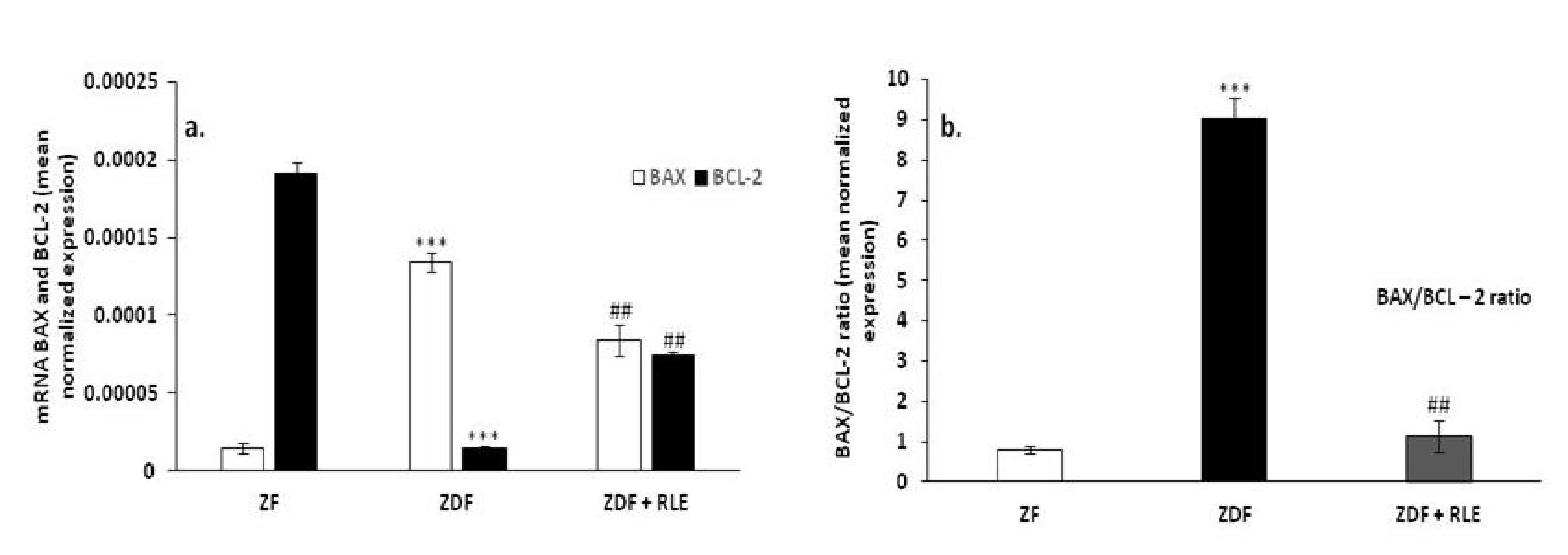
3.2.2. BAX, BCL-2, and BAX/BCL2 Ratio Gene Results
3.3. Western Blot Results
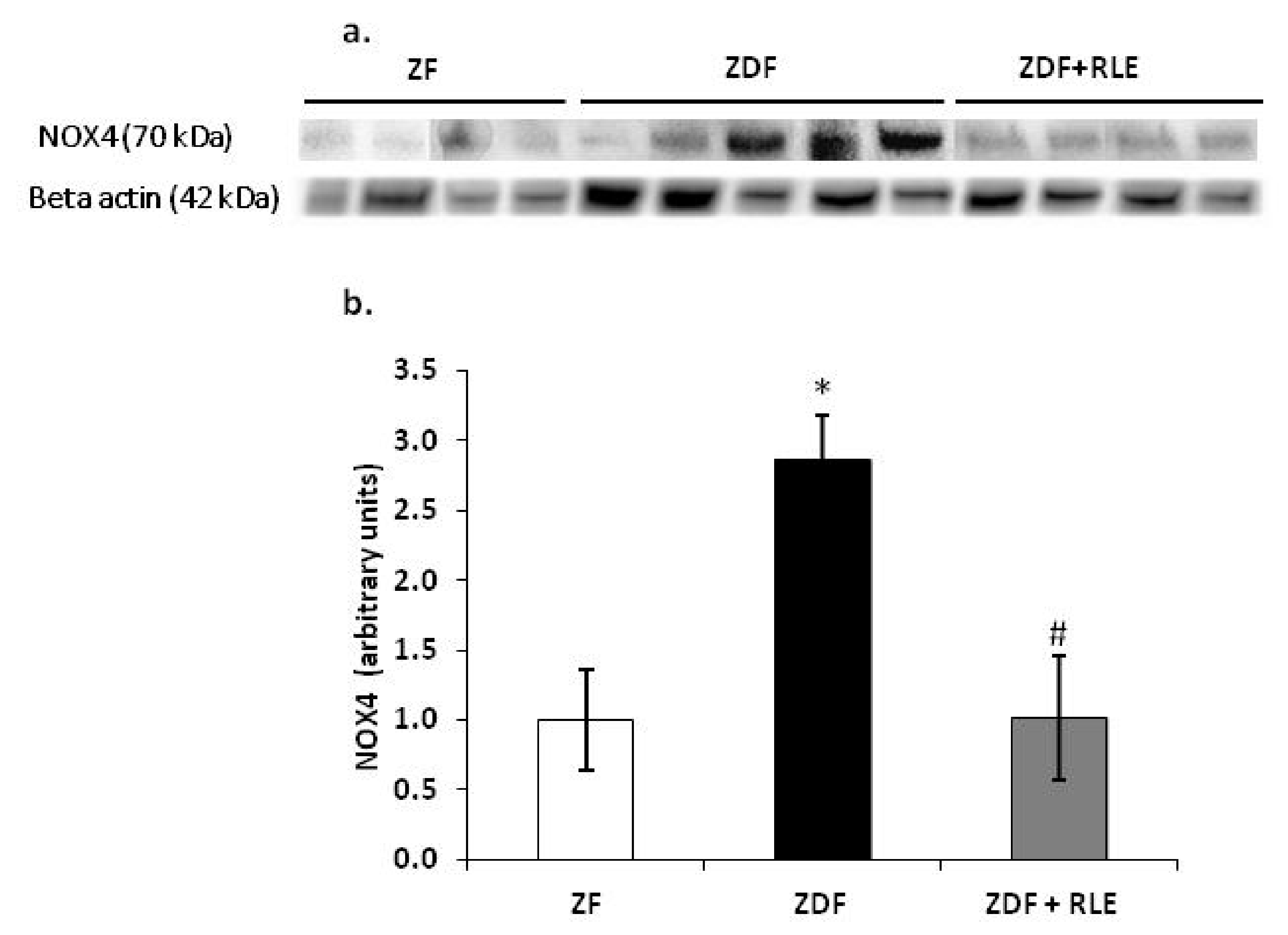
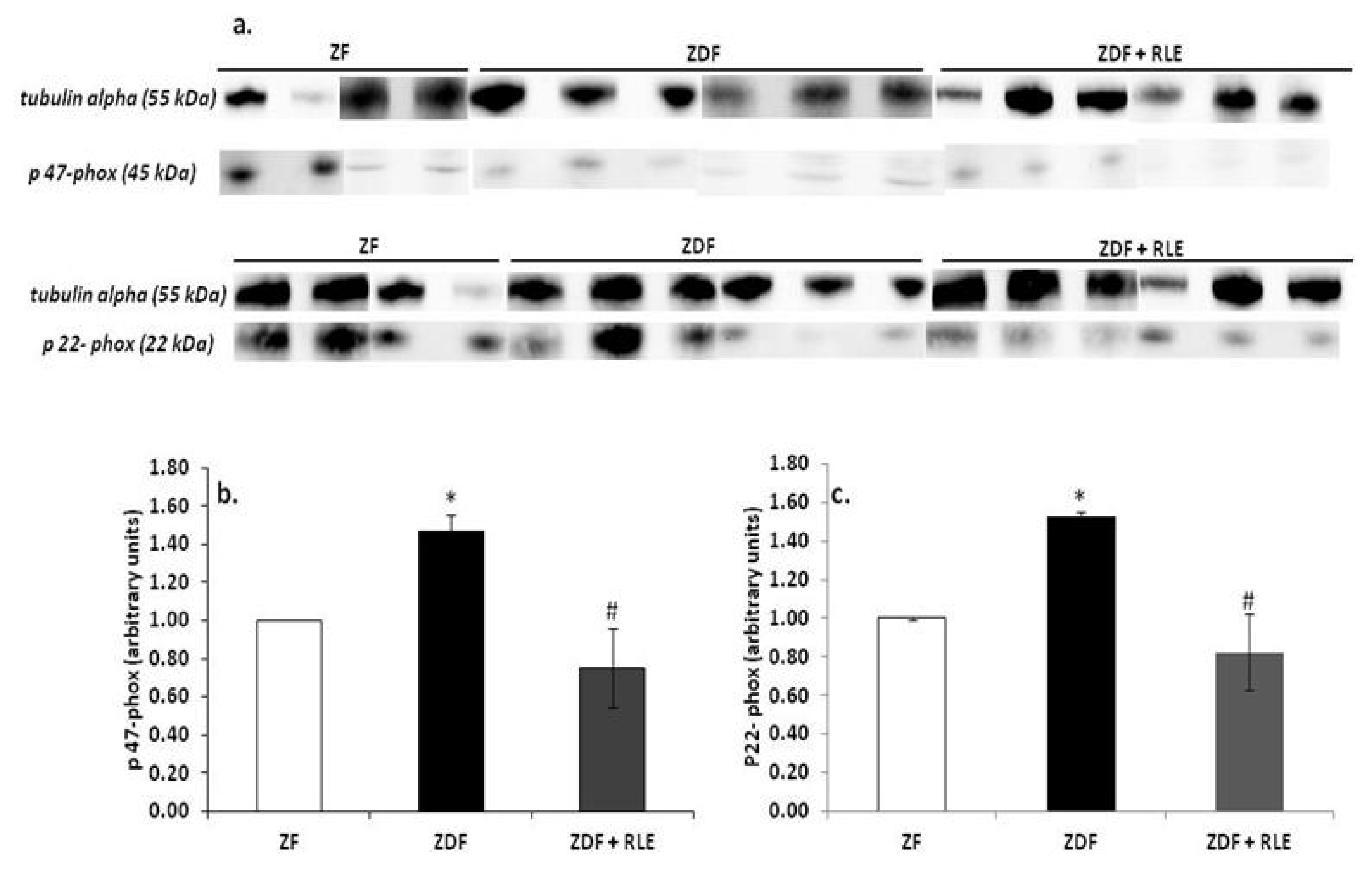
3.3.1. NOX4, p22-phox, and p47-phox Proteins Results
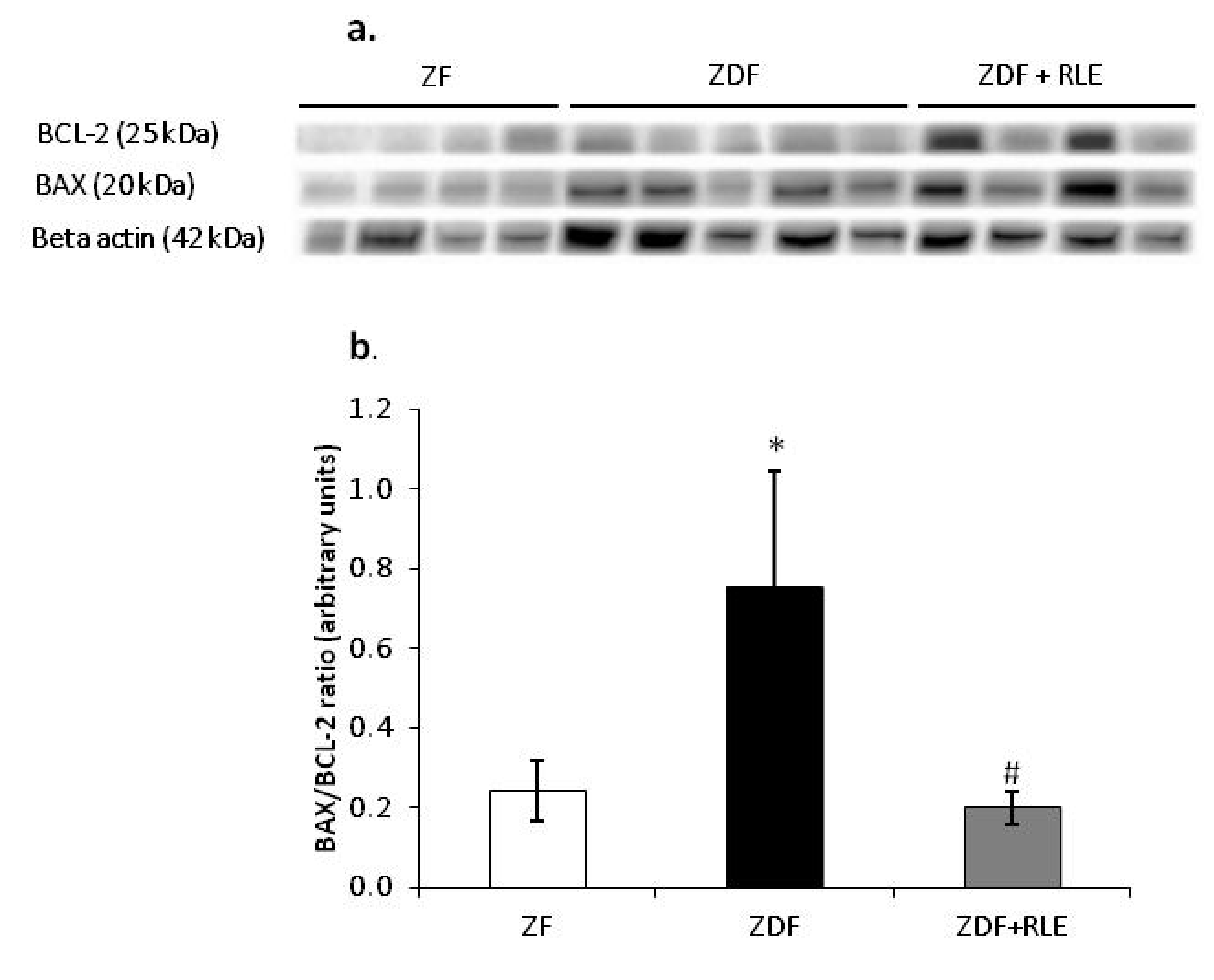
3.3.2. Ratio of the BAX/BCL-2 Expression Results
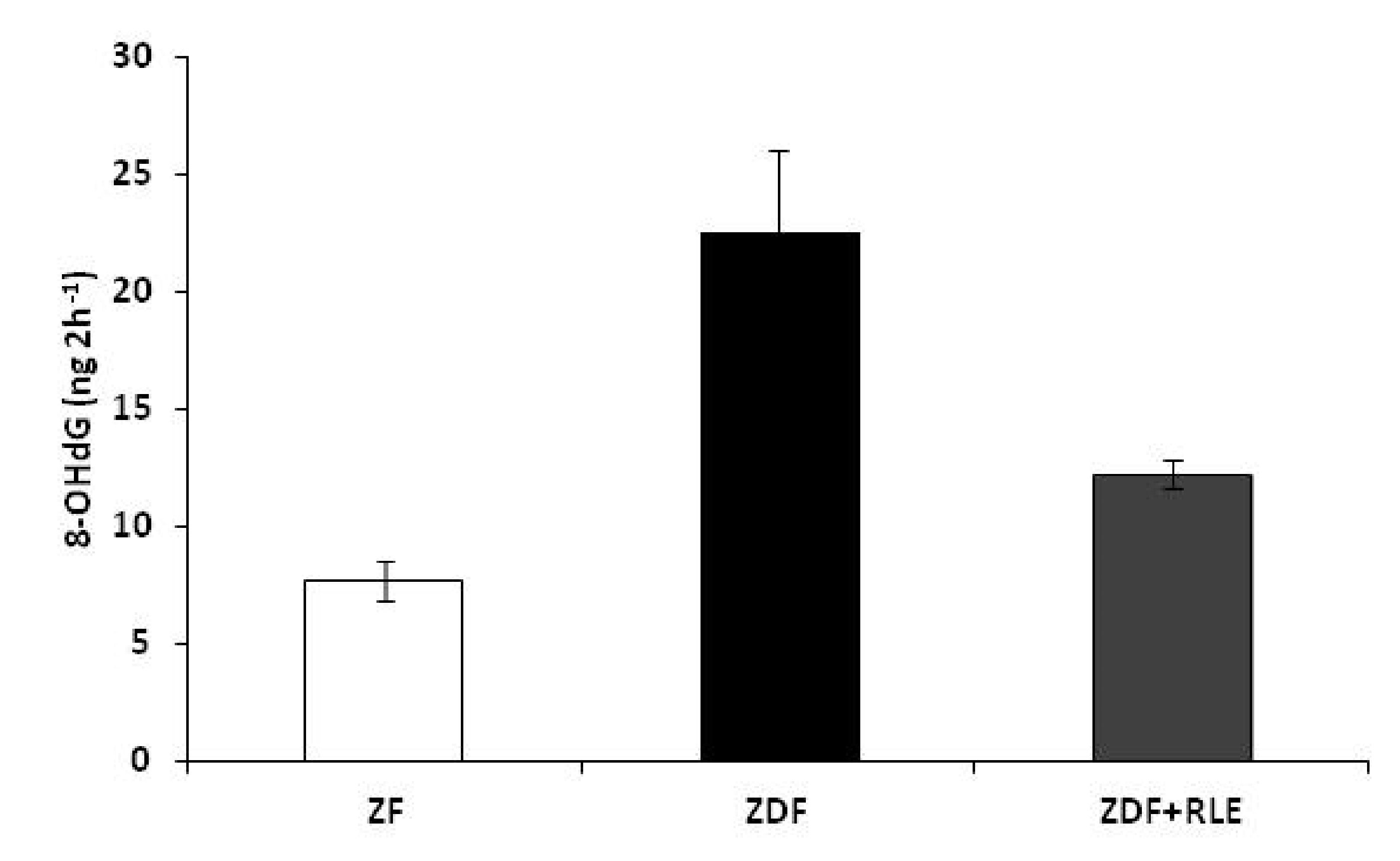
3.4. DNA Damage 8-Hydroxy-2-deoxy Guanosine (8-OHdG) Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gilbert, R.E.; Mifsud, S.A.; Hulthén, U.L.; Wilkinson-Berka, J.L.; Bertram, J.; Allen, T.; Cooper, M.E.; Kelly, D.J.; Wilkinson-Berka, J.L. Podocyte foot process broadening in experimental diabetic nephropathy: Amelioration with renin-angiotensin blockade. Diabetologia 2001, 44, 878–882. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Gao, Z.; Guo, Q.; Wang, T.; Lu, C.; Chen, Y.; Sheng, Q.; Chen, J.; Nie, Z.; Zhang, Y.; et al. Anti-Diabetic Effects of CTB-APSL Fusion Protein in Type 2 Diabetic Mice. Mar. Drugs 2014, 12, 1512–1529. [Google Scholar] [CrossRef] [PubMed]
- Cnop, M.; Welsh, N.; Jonas, J.-C.; Jörns, A.; Lenzen, S.; Eizirik, D.L. Mechanisms of pancreatic beta-cell death in type 1 and type 2 diabetes: Many differences, few similarities. Diabetes 2005, 54, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Reimann, M.; Bonifacio, E.; Solimena, M.; Schwarz, P.; Ludwig, B.; Hanefeld, M.; Bornstein, S. An update on preventive and regenerative therapies in diabetes mellitus. Pharmacol. Ther. 2009, 121, 317–331. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhao, M.; Wang, J.; Ding, Y.; Dai, X.; Li, Y. Oral Administration of Skin Gelatin Isolated from Chum Salmon (Oncorhynchus keta) Enhances Wound Healing in Diabetic Rats. Mar. Drugs 2011, 9, 696–711. [Google Scholar] [CrossRef]
- Singh, D.K.; Winocour, P.; Farrington, K. Oxidative stress in early diabetic nephropathy: Fueling the fire. Nat. Rev. Endocrinol. 2010, 7, 176–184. [Google Scholar] [CrossRef]
- Bhattacharjee, N.; Barma, S.; Konwar, N.; Dewanjee, S.; Manna, P. Mechanistic insight of diabetic nephropathy and its pharmacotherapeutic targets: An update. Eur. J. Pharmacol. 2016, 791, 8–24. [Google Scholar] [CrossRef]
- Miranda-Díaz, A.G.; Pazarin-Villaseñor, L.; Yanowsky-Escatell, F.G.; Andrade-Sierra, J. Oxidative Stress in Diabetic Nephropathy with Early Chronic Kidney Disease. J. Diabetes Res. 2016, 2016, 1–7. [Google Scholar] [CrossRef]
- Jha, J.C.; Banal, C.; Chow, B.S.; Cooper, M.E.; Jandeleit-Dahm, K.A. Diabetes and Kidney Disease: Role of Oxidative Stress. Antioxid. Redox Signal. 2016, 25, 657–684. [Google Scholar] [CrossRef]
- Kaneto, H.; Katakami, N.; Kawamori, D.; Miyatsuka, T.; Sakamoto, K.; Matsuoka, T.-A.; Matsuhisa, M.; Yamasaki, Y. Involvement of Oxidative Stress in the Pathogenesis of Diabetes. Antioxid. Redox Signal. 2007, 9, 355–366. [Google Scholar] [CrossRef]
- Palicz, A.; Foubert, T.R.; Jesaitis, A.; Maródi, L.; McPhail, L.C. Phosphatidic Acid and Diacylglycerol Directly Activate NADPH Oxidase by Interacting with Enzyme Components. J. Biol. Chem. 2000, 276, 3090–3097. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.X.; Tanaka, L.Y.; Wosniak, J.; Laurindo, F.R. Mechanisms and Implications of Reactive Oxygen Species Generation During the Unfolded Protein Response: Roles of Endoplasmic Reticulum Oxidoreductases, Mitochondrial Electron Transport, and NADPH Oxidase. Antioxid. Redox Signal. 2009, 11, 2409–2427. [Google Scholar] [CrossRef] [PubMed]
- Block, K.; Eid, A.; Griendling, K.; Lee, D.-Y.; Wittrant, Y.; Gorin, Y. Nox4 NAD(P)H oxidase mediates Src-dependent tyrosine phosphorylation of PDK-1 in response to angiotensin II: Role in mesangial cell hypertrophy and fibronectin expression. J. Biol. Chem. 2008, 283, 24061–24076. [Google Scholar] [CrossRef]
- Babior, B.M. NADPH oxidase: An update. Blood 1999, 93, 1464–1476. [Google Scholar] [CrossRef]
- Gill, P.S.; Wilcox, C.S. NADPH Oxidases in the Kidney. Antioxid. Redox Signal. 2006, 8, 1597–1607. [Google Scholar] [CrossRef]
- Gorin, Y.; Block, K. Nox as a target for diabetic complications. Clin. Sci. 2013, 125, 361–382. [Google Scholar] [CrossRef]
- Winiarska, K.; Dzik, J.M.; Labudda, M.; Focht, D.; Sierakowski, B.; Owczarek, A.; Komorowski, L.; Bielecki, W. Melatonin nephroprotective action in Zucker diabetic fatty rats involves its inhibitory effect on NADPH oxidase. J. Pineal Res. 2015, 60, 109–117. [Google Scholar] [CrossRef]
- Yanowsky-Escatell, F.G.; Andrade-Sierra, J.; Pazarín-Villaseñor, L.; Santana-Arciniega, C.; Torres-Vázquez, E.D.J.; Chávez-Iñiguez, J.S.; Zambrano-Velarde, M.; Ángel; Preciado-Figueroa, F.M. The Role of Dietary Antioxidants on Oxidative Stress in Diabetic Nephropathy. Iran. J. Kidney Dis. 2020, 14, 81–94. [Google Scholar]
- Wang, J.; Chen, Z.F.; He, C.X.; Wang, L.L.; Zheng, J.H.; Zhang, H.; Wang, Z.L.; Lu, Y.P. The effects of anthocyanin on chronic inflammatory pain induced by complete Freund’s adjuvant and its mechanism. Zhongguo Ying Yong Sheng Li Xue Za Zhi 2018, 34, 476–480. [Google Scholar] [CrossRef]
- Anjaneyulu, M.; Chopra, K. Quercetin attenuates thermal hyperalgesia and cold allodynia in STZ-induced diabetic rats. Indian J. Exp. Biol. 2004, 42, 766–769. [Google Scholar]
- Shih, P.-H.; Yeh, C.-T.; Yen, G.-C. Anthocyanins Induce the Activation of Phase II Enzymes through the Antioxidant Response Element Pathway against Oxidative Stress-Induced Apoptosis. J. Agric. Food Chem. 2007, 55, 9427–9435. [Google Scholar] [CrossRef] [PubMed]
- Damiano, S.; Iovane, V.; Squillacioti, C.; Mirabella, N.; Prisco, F.; Ariano, A.; Amenta, M.; Giordano, A.; Florio, S.; Ciarcia, R. Red orange and lemon extract prevents the renal toxicity induced by ochratoxin A in rats. J. Cell. Physiol. 2020, 235, 5386–5393. [Google Scholar] [CrossRef] [PubMed]
- Amorini, A.M.; Fazzina, G.; Lazzarino, G.; Tavazzi, B.; Di Pierro, D.; Santucci, R.; Sinibaldi, F.; Galvano, F.; Galvano, G. Activity and mechanism of the antioxidant properties of cyanidin-3-O-beta-glucopyranoside. Free Radic. Res. 2001, 35, 953–966. [Google Scholar] [CrossRef] [PubMed]
- Agil, A.; Rosado-Sánchez, I.; Ruiz, R.; Figueroa, A.; Zen, N.; Fernández-Vázquez, G. Melatonin improves glucose homeostasis in young Zucker diabetic fatty rats. J. Pineal Res. 2011, 52, 203–210. [Google Scholar] [CrossRef]
- Titta, L.; Trinei, M.; Stendardo, M.; Berniakovich, I.; Petroni, K.; Tonelli, C.; Riso, P.; Porrini, M.; Minucci, S.; Pelicci, P.G.; et al. Blood orange juice inhibits fat accumulation in mice. Int. J. Obes. 2009, 34, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3 - New capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef]
- Koressaar, T.; Remm, M. Enhancements and modifications of primer design program Primer3. Bioinformatics 2012, 23, 1289–1291. [Google Scholar]
- Lauritano, C.; Carotenuto, Y.; Vitiello, V.; Buttino, I.; Romano, G.; Hwang, J.-S.; Ianora, A. Effects of the oxylipin-producing diatom Skeletonema marinoi on gene expression levels of the calanoid copepod Calanus sinicus. Mar. Genom. 2015, 24, 89–94. [Google Scholar] [CrossRef]
- Lauritano, C.; De Luca, D.; Ferrarini, A.; Avanzato, C.; Minio, A.; Esposito, F.; Ianora, A. De novo transcriptome of the cosmopolitan dinoflagellate Amphidinium carterae to identify enzymes with biotechnological potential. Sci. Rep. 2017, 7, 11701. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, 45. [Google Scholar] [CrossRef]
- Muller, P.Y.; Janovjak, H.; Miserez, A.R.; Dobbie, Z. Processing of gene expression data generated by quantitative real-time RT-PCR. BioTechniques 2002, 32, 1372–1379. [Google Scholar]
- Varatharajan, R.; Sattar, M.Z.A.; Chung, I.; Abdulla, M.A.; Kassim, N.M.; Abdullah, N.A. Antioxidant and pro-oxidant effects of oil palm (Elaeis guineensis) leaves extract in experimental diabetic nephropathy: A duration-dependent outcome. BMC Complement. Altern. Med. 2013, 13, 242. [Google Scholar] [CrossRef] [PubMed]
- Forbes, J.M.; Coughlan, M.T.; Cooper, M.E. Oxidative Stress as a Major Culprit in Kidney Disease in Diabetes. Diabetes 2008, 57, 1446–1454. [Google Scholar] [CrossRef] [PubMed]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ. Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef]
- Skates, E.; Overall, J.; Dezego, K.; Wilson, M.; Esposito, D.; Lila, M.A.; Komarnytsky, S. Berries containing anthocyanins with enhanced methylation profiles are more effective at ameliorating high fat diet-induced metabolic damage. Food Chem. Toxicol. 2018, 111, 445–453. [Google Scholar] [CrossRef]
- Ravussin, Y.; Gutman, R.; LeDuc, C.A.; Leibel, R.L. Estimating energy expenditure in mice using an energy balance technique. Int. J. Obes. 2012, 37, 399–403. [Google Scholar] [CrossRef]
- Tsuda, T.; Horio, F.; Uchida, K.; Aoki, H.; Osawa, T. Dietary cyanidin 3-O-beta-D-glucoside-rich purple corn color prevents obesity and ameliorates hyperglycemia in mice. J. Nutr. 2003, 133, 2125–2130. [Google Scholar] [CrossRef]
- Tsuda, T. Regulation of Adipocyte Function by Anthocyanins; Possibility of Preventing the Metabolic Syndrome. J. Agric. Food Chem. 2008, 56, 642–646. [Google Scholar] [CrossRef]
- Vasović, L.; Trandafilović, M.; Vlajković, S.; Radenković, G. Congenital absence of the bilateral internal carotid artery: A review of the associated (ab)normalities from a newborn status to the eighth decade of life. Childs Nerv. Syst. 2017, 34, 35–49. [Google Scholar] [CrossRef]
- Lee, H.Y.; Eum, W.S.; Kim, D.W.; Lee, B.R.; Yoon, C.S.; Jang, S.H.; Choi, H.S.; Choi, S.H.; Baek, N.-I.; Kang, J.H.; et al. Isolation and identification of an antioxidant enzyme catalase stimulatory compound from Garnoderma lucidum. J. Biochem. Mol. Biol. 2003, 36, 450–455. [Google Scholar] [CrossRef]
- Wiernsperger, N.F. Oxidative stress: The special case of diabetes. BioFactors 2003, 19, 11–18. [Google Scholar] [CrossRef]
- Wu, L.L.; Chiou, C.-C.; Chang, P.Y.; Wu, J.T. Urinary 8-OHdG: A marker of oxidative stress to DNA and a risk factor for cancer, atherosclerosis and diabetics. Clin. Chim. Acta 2004, 339, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tarng, D.-C.; Huang, T.-P.; Wei, Y.-H.; Liu, T.-Y.; Chen, H.-W.; Chen, T.W.; Yang, W.-C. 8-Hydroxy-2′-Deoxyguanosine of leukocyte DNA as a marker of oxidative stress in chronic hemodialysis patients. Am. J. Kidney Dis. 2000, 36, 934–944. [Google Scholar] [CrossRef]
- Tarng, D.-C.; Chen, T.W.; Huang, T.-P.; Chen, C.-L.; Liu, T.-Y.; Wei, Y.-H. Increased oxidative damage to peripheral blood leukocyte DNA in chronic peritoneal dialysis patients. J. Am. Soc. Nephrol. 2002, 13, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Griendling, K.; Sorescu, D.; Ushio-Fukai, M. NAD(P)H Oxidase. Circ. Res. 2000, 86, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Etoh, T.; Inoguchi, T.; Kakimoto, M.; Sonoda, N.; Kobayashi, K.; Kuroda, J.; Sumimoto, H.; Nawata, H. Increased expression of NAD(P)H oxidase subunits, NOX4 and p22phox, in the kidney of streptozotocin-induced diabetic rats and its reversibity by interventive insulin treatment. Diabetologia 2003, 46, 1428–1437. [Google Scholar] [CrossRef]
- Kitada, M.; Koya, D.; Sugimoto, T.; Isono, M.; Araki, S.-I.; Kashiwagi, A.; Haneda, M. Translocation of Glomerular p47phox and p67phox by Protein Kinase C- Activation Is Required for Oxidative Stress in Diabetic Nephropathy. Diabetes 2003, 52, 2603–2614. [Google Scholar] [CrossRef]
- Sharma, N.M.; Rabeler, B.; Zheng, H.; Raichlin, E.; Patel, K.P. Exercise Training Attenuates Upregulation of p47(phox) and p67(phox) in Hearts of Diabetic Rats. Oxidative Med. Cell. Longev. 2016, 2016, 1–11. [Google Scholar] [CrossRef]
- Kassan, M.; Choi, S.-K.; Galán, M.; Lee, Y.-H.; Trebak, M.; Matrougui, K. Enhanced p22phox expression impairs vascular function through p38 and ERK1/2 MAP kinase-dependent mechanisms in type 2 diabetic mice. Am. J. Physiol. Circ. Physiol. 2014, 306, H972–H980. [Google Scholar] [CrossRef]
- Anjaneyulu, M.; Chopra, K. Nordihydroguairetic Acid, a Lignin, Prevents Oxidative Stress and the Development of Diabetic Nephropathy in Rats. Pharmacology 2004, 72, 42–50. [Google Scholar] [CrossRef]
- Álvarez-Cilleros, D.; López-Oliva, M.E.; Martin, M.A.; Ramos, S. Cocoa ameliorates renal injury in Zucker diabetic fatty rats by preventing oxidative stress, apoptosis and inactivation of autophagy. Food Funct. 2019, 10, 7926–7939. [Google Scholar] [CrossRef]
- Lin, C.-F.; Kuo, Y.-T.; Chen, T.-Y.; Chien, C.-T. Quercetin-Rich Guava (Psidium guajava) Juice in Combination with Trehalose Reduces Autophagy, Apoptosis and Pyroptosis Formation in the Kidney and Pancreas of Type II Diabetic Rats. Molecules 2016, 21, 334. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Wu, H.; Zhang, H.; Li, F.; Chen, S.; Hou, B.; Shi, Y.; Zhao, L.; Duan, H. Anthocyanins inhibit high glucose-induced renal tubular cell apoptosis caused by oxidative stress in db/db mice. Int. J. Mol. Med. 2018, 41, 1608–1618. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Fu, R.; Duan, Z.; Lu, J.; Gao, J.; Tian, L.; Lv, Z.; Chen, Z.; Han, J.; Jia, L.; et al. Sirt1 is essential for resveratrol enhancement of hypoxia-induced autophagy in the type 2 diabetic nephropathy rat. Pathol. Res. Pract. 2016, 212, 310–318. [Google Scholar] [CrossRef]
- Xu, X.H.; Ding, D.F.; Yong, H.-J.; Dong, C.-L.; You, N.; Ye, X.L.; Pan, M.-L.; Ma, J.-H.; You, Q.; Lu, Y.-B. Resveratrol transcriptionally regulates miRNA-18a-5p expression ameliorating diabetic nephropathy via increasing autophagy. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4952–4965. [Google Scholar]
| Gene Name | Primer F Primer R | Amplicon Size | E | R2 | Acc. Number |
|---|---|---|---|---|---|
| BAX | ACAACAACATGGAGCTGCAG-CTTGGATCCAGACAAACAGCC | 249 | 100 | 0.99 | U32098.1 |
| BCL2 | GCCTTCTTTGAGTTCGGTGG-CTGAGCAGCGTCTTCAGAGA | 221 | 100 | 0.99 | L14680.1 |
| NOX4 | TCGGGTGGCTTGTTGAAGTA-GTCTGTGGGAAATGAGCTTGG | 224 | 90 | 0.99 | NM_053524 |
| 18S | AGAAACGGCTACCACATCCA-CCCTCCAATGGATCCTCGTT | 158 | 93 | 0.99 | NR_046237.1 |
| p22 | ATCAAGCAGCCACCTACCAA-GGGAGCAACACCTTGGAAAC | 179 | 100 | 0.99 | AJ295951.1 |
| p47 | TACGCTGCTGTTGAAGAGGA-GATGTCCCCTTTCCTGACCA | 105 | 100 | 0.99 | AY029167.1 |
| Group Rat | BP (mmHg) | Water Intake (mL/day) | Food Intake (g/day) |
|---|---|---|---|
| ZF | 98.22 ± 9.6 | 14 ± 1.1 | 17 ± 0.8 |
| ZDF | 104.10 ± 12.6 | 58 ± 1.4 *** | 28 ± 1.0 * |
| ZDF + RLE | 103.71 ± 6.9 | 23 ± 1.7 *, ### | 21 ± 1.1 # |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Damiano, S.; Lauritano, C.; Longobardi, C.; Andretta, E.; Elagoz, A.M.; Rapisarda, P.; Di Iorio, M.; Florio, S.; Ciarcia, R. Effects of a Red Orange and Lemon Extract in Obese Diabetic Zucker Rats: Role of Nicotinamide Adenine Dinucleotide Phosphate Oxidase. J. Clin. Med. 2020, 9, 1600. https://doi.org/10.3390/jcm9051600
Damiano S, Lauritano C, Longobardi C, Andretta E, Elagoz AM, Rapisarda P, Di Iorio M, Florio S, Ciarcia R. Effects of a Red Orange and Lemon Extract in Obese Diabetic Zucker Rats: Role of Nicotinamide Adenine Dinucleotide Phosphate Oxidase. Journal of Clinical Medicine. 2020; 9(5):1600. https://doi.org/10.3390/jcm9051600
Chicago/Turabian StyleDamiano, Sara, Chiara Lauritano, Consiglia Longobardi, Emanuela Andretta, Ali Murat Elagoz, Paolo Rapisarda, Mattia Di Iorio, Salvatore Florio, and Roberto Ciarcia. 2020. "Effects of a Red Orange and Lemon Extract in Obese Diabetic Zucker Rats: Role of Nicotinamide Adenine Dinucleotide Phosphate Oxidase" Journal of Clinical Medicine 9, no. 5: 1600. https://doi.org/10.3390/jcm9051600
APA StyleDamiano, S., Lauritano, C., Longobardi, C., Andretta, E., Elagoz, A. M., Rapisarda, P., Di Iorio, M., Florio, S., & Ciarcia, R. (2020). Effects of a Red Orange and Lemon Extract in Obese Diabetic Zucker Rats: Role of Nicotinamide Adenine Dinucleotide Phosphate Oxidase. Journal of Clinical Medicine, 9(5), 1600. https://doi.org/10.3390/jcm9051600







